1 Department of Pharmacology and Dental Therapeutics, College of Dentistry, Chosun University, 61452 Gwangju, Republic of Korea
Abstract
Although catalase is a crucial antioxidant enzyme widely used in industrial and environmental systems to decompose hydrogen peroxide (H2O2), its low stability and reusability limit its practical applications. This study aimed to enhance catalase performance by immobilising an enzyme on titanium dioxide nanoparticles (TNPs) in the anatase phase to form catalase-immobilised TNPs (CITNPs) and to evaluate their catalytic activity, stability, and reusability under various conditions. TNPs were modified using four chemical intermediates to introduce functional groups for enzyme immobilisation: (3-aminopropyl)triethoxysilane for amine groups (method A), (3-mercaptopropyl)trimethoxysilane for thiol groups (method B), dihydrocaffeic acid for carboxylic groups (method C), and tannic acid for phenolic groups (method D). Catalase was immobilised onto each modified TiO2 intermediate to obtain CITNPs. The catalase activity, thermal and pH stabilities, reusability, and functional capability in beverage solutions were evaluated using ultraviolet-visible analyses and enzyme activity assays. All four immobilisation routes successfully produced CITNPs with a high catalase loading efficiency (approximately 75.5%–87.3%). CITNPs demonstrated optimal catalytic activity at 40 °C and pH 6.0–7.0. Thermal stability was markedly improved compared to free catalase, with CITNPs retaining 28.9% residual enzyme activity after exposure to 70 °C for 60 min, whereas free catalase lost approximately all activity under the same conditions. Reusability testing over 10 reaction cycles revealed sustained catalytic performance, with 41.8%–76.3% activity retained after the final cycle. In real sample applications, CITNPs effectively eliminated H2O2 artificially generated in hot tea and coffee solutions, confirming functional activity in food-associated environments. Immobilisation of catalase onto chemically modified TNPs significantly enhanced their stability and reusability without compromising enzymatic efficiency. CITNPs demonstrated strong catalytic performance even under high-temperature stress and repeated operational cycles and successfully removed H2O2 in beverage systems. These findings suggest that CITNPs are promising candidates for catalase-based applications in industrial processing, food-related systems, biotechnology, and environmental detoxification.
Keywords
- catalase
- enzyme
- immobilisation
- TiO2 nanoparticle intermediates
Catalase (EC 1.11.1.6) is an essential enzyme that decomposes H2O2 into water and oxygen and plays a vital role in protecting tissues from oxidative damage caused by reactive oxygen species (ROS) [1, 2]. Due to its high catalytic efficiency and substrate specificity, catalase exhibits significant potential for applications in environmental biotechnology, food processing, pharmaceuticals, and the biomedical industry [3, 4, 5, 6, 7]. However, the practical use of free catalase is limited by its insufficient operational stability under non-ideal processing conditions such as prolonged use, repeated catalytic cycling, storage stress, elevated temperatures, and local pH fluctuations.
Immobilisation of catalase on solid supports is a promising strategy for enhancing enzyme stability and extending its functional lifespan [8]. Nanomaterials have recently emerged as attractive enzyme carriers because of their high surface-to-volume ratios, tunable surface chemistries, and unique physicochemical properties [9, 10]. Various nanomaterials, including silica [11], magnetic materials [12], gelatin [13], chitosan [14], and metal oxides such as Fe2O3 [15], CuO [16], and ZnO [17], have been effectively used as matrices for enzyme immobilisation. These nanostructured materials provide numerous binding sites and create protective microenvironments that help preserve enzyme conformation and function. Among them, titanium dioxide (TiO2) is widely utilized as a support material because of its chemical stability, biocompatibility, affordability, and ease of surface modification [18, 19, 20, 21]. TiO2-based supports have been successfully used for enzyme immobilisation, often resulting in improved thermal and operational stability, as compared to free enzymes [18, 19, 21].
The immobilisation of catalase onto titanium dioxide nanoparticles (TNPs) was explored in this study using four different surface modifications to facilitate either covalent binding or the physical adsorption of catalase. Surface-modified TNPs were prepared using (i) (3-aminopropyl)triethoxysilane (APTES) to introduce amine groups (method A), (ii) (3-mercaptopropyl)trimethoxysilane (MPTMS) to introduce thiol groups (method B), (iii) dihydrocaffeic acid (DCA) to introduce carboxylic groups (method C), and (iv) tannic acid (TA) to introduce phenolic groups (method D). Following catalase immobilisation, the catalytic activity and enzyme stability of the resulting catalase-immobilised titanium dioxide nanoparticles (CITNPs) were characterised under various thermal and pH conditions. Additionally, the ability of CITNPs to degrade hydrogen peroxide (H2O2) was evaluated in real samples such as tea extract and coffee solution. The results demonstrate that catalase immobilised via all four surface-modified TNP intermediates exhibits high loading capacity and retains enhanced thermal stability and reusability under varying environmental conditions while maintaining significant catalytic activity.
The TNPs used in this study were synthesised following our previously reported procedure [20] and were characterised as the anatase phase with an average particle size of approximately 20–30 nm. APTES, MPTMS, DCA, TA, glutaraldehyde (GLA), 1-ethyl-3-(3-dimethylamino)propyl carbodiimide hydrochloride (EDC), N-hydroxysuccinimide (NHS), H2O2, bovine serum albumin (BSA), and catalase were purchased from Sigma-Aldrich (St. Louis, MO, USA). Deionised water (DW) was purified using a Millipore Milli-Q purification system (Merck Millipore, Molsheim, France).
Amino-TNP or thiol-TNP (TNP-APTES or TNP-MPTMS, respectively) was prepared via silanisation of the TNP surface [22, 23]. Briefly, 50 mg TNPs was added to 20 mL DW containing 0.5 mL APTES or MPTMS (molar ratio of TNP to APTES or MPTMS = 6.25:1), and the reaction mixture was stirred overnight. Carboxylic-TNP (TNP-DCA) was prepared by slowly adding 50 mg TNPs to 10 mL sodium phosphate buffer (0.2 M) containing 10 mg DCA, and the reaction solution was stirred for 1 h at 25 °C. The phenolic-TNP (TNP-TA) intermediates were synthesised by a coordination reaction between the Ti ions on the TNP surface and the phenolic groups of TA [24]. Briefly, 50 mg TNPs were dispersed in 10 mL TA (10 mM) at a molar ratio of 5:1. The mixture was stirred for 1 h at 25 °C. The modified TNP intermediates were separated by centrifugation after washing three times with distilled water and dried at 40 °C under vacuum.
Ultraviolet-visible (UV-Vis) spectra were recorded using a Hitachi U-1900 spectrophotometer (Hitachi High-Tech, Tokyo, Japan). Samples (0.1 mg/mL) were prepared in a 1 cm quartz cuvette and scanned over a wavelength range of 300–600 nm. Fourier-transform infrared (FT-IR) spectra of the pristine and modified TNP intermediates were obtained over the wavenumber range of 400–4000 cm-1 using a Nicolet 6700 FT-IR spectrometer (Thermo Fisher Scientific, Waltham, MA, USA).
Catalase was immobilised on the surface of the TNPs using GLA as a crosslinker and covalently attached to the silane-grafted TNP intermediates (Fig. 1). Briefly, 10 mg TNP-APTES was dispersed in sodium phosphate buffer (50 mM, pH 7). Subsequently, 0.5 mL catalase (5 mg/mL) and 50 µL GLA (100 mM) were slowly added over 5 min at 25 °C under stirring. The immobilised catalase was collected by centrifugation (10,000 rpm for 5 min) and washed three times with sodium phosphate buffer. To immobilise catalase on TNP-DCA (10 mg), 3 mg NHS and 1.8 mg EDC were added to 5 mL 2-(N-morpholino)ethanesulfonic acid (MES) buffer solution (100 mM, pH 6.5) and stirred for 1 h at room temperature. Subsequently, 0.5 mL catalase (5 mg/mL) in MES buffer (pH 6.5) was added, and the solution was stirred for a further 4 h at 4 °C. The immobilised catalase was collected by centrifugation (10,000 rpm for 5 min) and washed three times with MES buffer. Catalase was immobilised on TNP-TA or TNP-MPTMS by the direct interaction between catalase and the functional groups (phenolic or thiol) on the TNP intermediates. Briefly, 0.5 mL catalase solution (5 mg/mL) in phosphate buffer was slowly added to 10 mL sodium phosphate buffer containing TNP-TA (10 mg) or TNP-MPTMS particles (10 mg). The mixed solution was stirred at 4 °C for 6 h. The immobilised catalase was collected by centrifugation (10,000 rpm for 5 min) and washed three times with sodium phosphate buffer. Finally, the prepared CITNPs were lyophilised and stored at –20 °C.
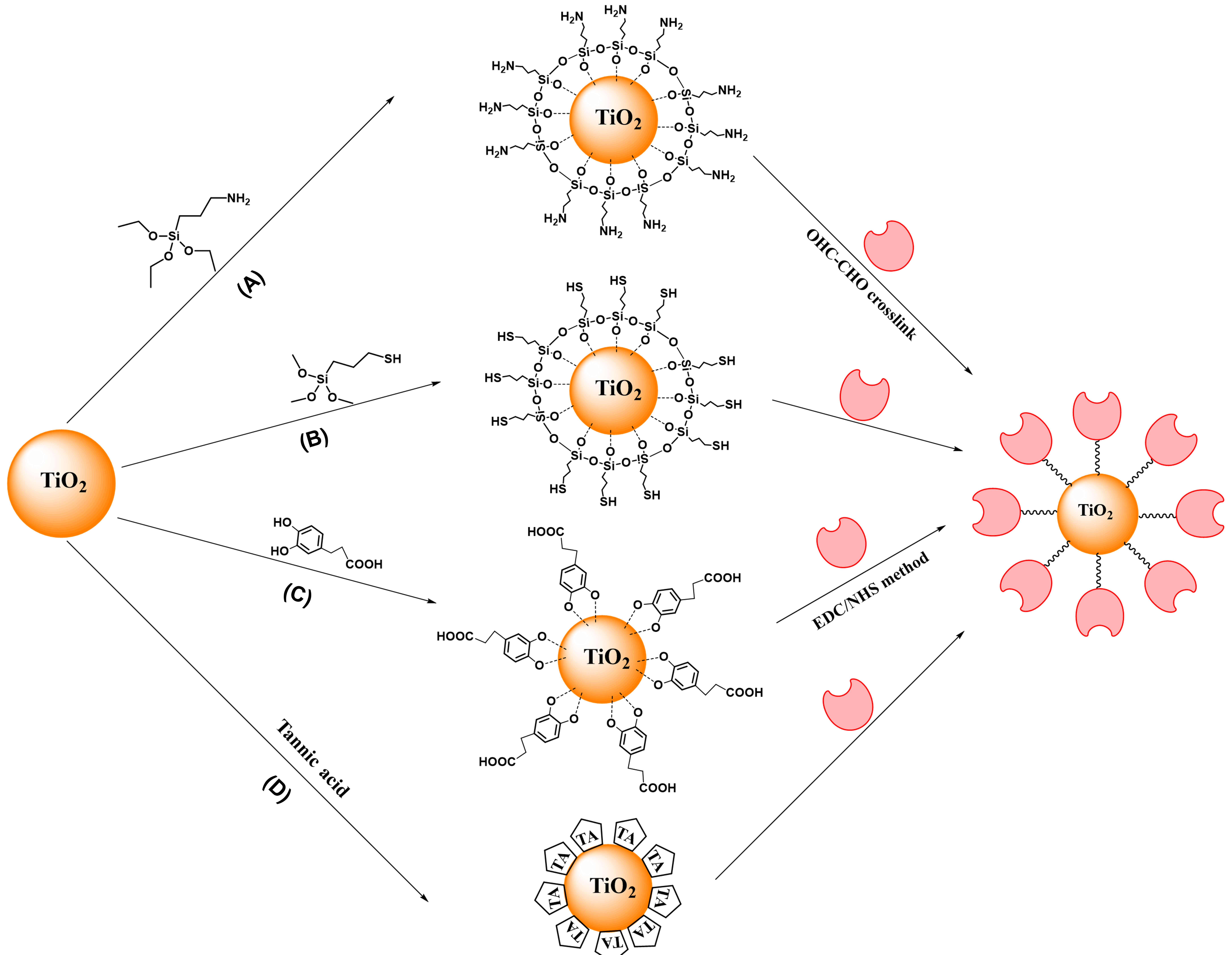 Fig. 1.
Fig. 1.
Schematic diagram illustrating the preparation and surface functionalisation of TNPs with four distinct functional groups. (A) amine (using APTES); (B) thiol (using MPTMS); (C) carboxylic (using DCA); and (D) phenolic (using TA). Catalase was subsequently immobilised onto these functionalised TNPs via covalent bonding or surface adsorption, resulting in CITNPs. TNPs, titanium dioxide nanoparticles; APTES, (3-aminopropyl)triethoxysilane; MPTMS, (3-mercaptopropyl)trimethoxysilane; DCA, dihydrocaffeic acid; TA, tannic acid; CITNPs, catalase-immobilised TNPs. The figure was created with ChemDraw Ultra 12.0 (Waltham, MA, USA).
To determine the catalase activity, 50 µL catalase (5 mg/mL) or 5 mg CITNPs was added to 500 µL H2O2 (5 mM) in sodium phosphate buffer (50 mM, pH 7.0) and incubated for 5 min at 25 °C. The H2O2 concentration was measured via UV absorbance at 240 nm [25] using a UV-Vis spectrophotometer (Hitachi U-1900, Tokyo, Japan). The catalase concentration was determined via the Lowry method using a Multiskan SkyHigh microplate reader (Thermo Fisher Scientific, Waltham, MA, USA), with BSA as the standard [26]. The amount of catalase bound to CITNPs was calculated by subtracting the amount of residual catalase in the supernatant after immobilisation from the total amount used initially. The immobilisation efficiency of the CITNPs was calculated using Eqns. 1,2:
The effects of temperature and pH on the activity of immobilised catalase were examined at 10–80 °C and pH 3.0–9.0 (pH 3.0–6.0 in 50 mM sodium citrate; pH 6.0–8.0 in 50 mM sodium phosphate; pH 8.0–9.0 in 50 mM Tris-HCl). Each sample was incubated for 5 min under specific conditions and the activity was determined using the enzyme assay described in Section 2.5. The relative percentage activity of catalase was calculated based on the highest observed activity, which was set to 100%.
CITNPs were incubated in sodium phosphate buffer (50 mM, pH 7.0) at 70 °C for 60 min, and the catalase activity was determined using the enzyme assay described in Section 2.5. The reusability of catalase in the CITNPs (10 mg/mL) was evaluated at ambient temperature for 10 min, as described in the catalase activity assay. After each test cycle, the CITNPs were recovered by centrifugation (12,000 rpm for 2 min) and washed three times with phosphate buffer. H2O2 solution (5 mM), used as a catalase substrate, was freshly prepared for each assay cycle. The catalase activity at time zero or during the first cycle was assumed to be 100% of the initial catalase activity.
Commercially available dried green tea leaves and coffee powder purchased from a local market (Gwangju, Republic of Korea) were used as model phenolic-containing beverages to prepare beverage extracts [27]. For the tea extract, 2 g dried tea leaves were steeped in 100 mL of DW at 80 °C for 5 min and then filtered through Whatman No. 1 filter paper (GE Healthcare, Chicago, IL, USA). For the coffee extract, 2 g ground coffee powder was brewed with 100 mL boiling DW and filtered. All extracts were cooled to room temperature, diluted to 1:10 (v/v) with 50 mM phosphate buffer (pH 7.0), and further filtered using a Sep-Pak C18 cartridge (Waters Corporation, Milford, MA, USA) followed by a 0.45 µm microfilter (Millipore, Billerica, MA, USA) before analysis. For the H2O2 removal assay, CITNPs (or free catalase) were added to the beverage extracts at an equivalent catalase loading of 100 U/mL (enzyme activity determined as described in Section 2.5). The mixtures were gently mixed at 25 °C, and samples were collected at specific time intervals (0, 1, 3, 5, and 10 min) for residual H2O2 analyses.
H2O2 was indirectly detected via the formation of a ternary complex
(Ti-TAR-H2O2), as previously described [27]. Briefly,
50 µL of the reaction sample was mixed with 50 µL of
10 mM titanium(III) chloride and 50 µL of 10 mM
4-(2-thiazolylazo)resorcinol (TAR) and incubated at room temperature for 5 min.
The resulting mixture containing the ternary complex was injected into a CE
system (P/ACE 5500, Beckman Coulter, Kraemer, CA, USA) equipped with a photodiode
array detector set at 530 nm (
The reusability of the CITNPs was evaluated by performing the H2O2 removal reaction over 10 consecutive cycles. After each cycle (5 min reaction), the CITNPs were separated by centrifugation (10,000 rpm, 5 min), washed twice with phosphate buffer (pH 7.0), and reused under identical conditions. The relative catalase activity for each cycle was calculated by comparing the H2O2 removal rate of that cycle with that of the first cycle.
As previously reported [20], X-ray diffraction analyses and transmission electron microscopy observations confirmed that the TNPs employed in this study exhibited a well-defined anatase crystal structure and possessed a relatively uniform particle size distribution with diameters of approximately 20–30 nm. To prepare the TNP intermediates, the TNP surface was modified with four types of functional groups (amine, thiol, carboxylic acid, and phenolic groups), abbreviated as APTES, MPTMS, DCA, and TA, respectively. As shown in Fig. 2, the functional groups on the TNP intermediates were confirmed using UV-Vis and FT-IR spectroscopy as well as the sedimentation behaviour. A rapid increase in UV absorbance was observed for TNP-TA and TNP-DCA at 300–400 nm, indicating the presence of phenolic groups on the modified surfaces, whereas both TNP-APTES and TNP-MPTMS exhibited weak UV absorbance (Fig. 2A). As shown in Fig. 2B, the FT-IR spectra of all modified TNP intermediates exhibited a characteristic Ti-O-Ti stretching band at 483–683 cm-1. TNP-APTES and TNP-MPTMS exhibited Ti-O-Si asymmetric stretching bands with a small peak at 985 cm-1 and weak peaks at 2853 and 2925 cm-1 corresponding to the C-H asymmetric and symmetric stretching vibrations of the coupling agents [28]. TNP-TA exhibited the characteristic bands of TA at 1000–1700 cm-1 owing to the chemical interactions between TA and the TiO2 surface. Moreover, the sedimentation behaviours of TNP-APTES and TNP-MPTMS differed from those of TNP-TA and TNP-DCA. That is, the TNP-APTES or TNP-MPTMS suspensions in water aggregated and precipitated after 1 h, whereas those of TNP-DCA and TNP-TA did not (Fig. 2C).
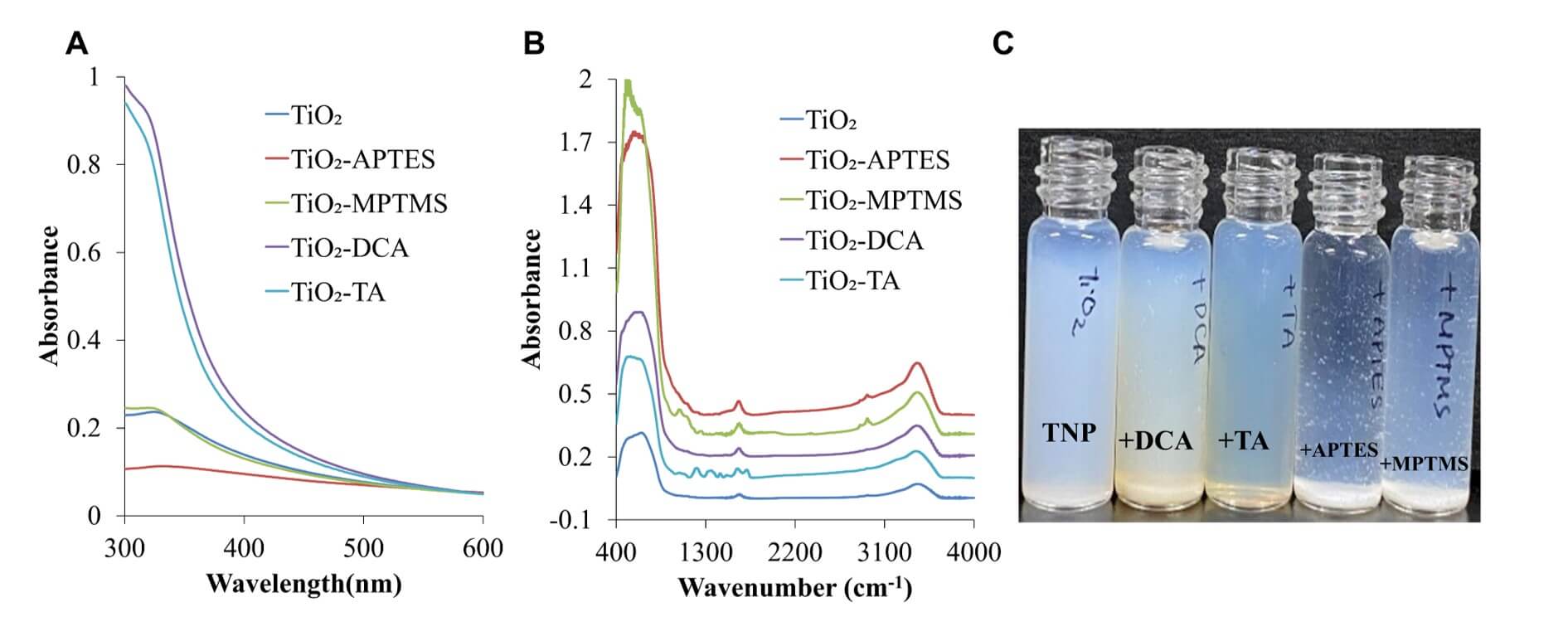 Fig. 2.
Fig. 2.
Characterisation of the TNP intermediates functionalised with various surface groups. (A) UV-Vis spectra: TNP functionalised with APTES (amine), MPTMS (thiol), DCA (carboxylic acid), and TA (phenolic) exhibited distinct absorption profiles. TNP-TA and TNP-DCA exhibited strong absorbance at 300–400 nm, attributable to phenolic group interactions. (B) FT-IR spectra: all surface-modified TNPs exhibited the characteristic Ti-O-Ti stretching band (483–683 cm-1). TNP-APTES and TNP-MPTMS also exhibited Ti-O-Si asymmetric stretching (~985 cm-1) and C–H stretching vibrations (~2853 and 2925 cm-1), confirming successful organosilane coupling. TNP-TA exhibited distinct bands at 1000–1700 cm-1, corresponding to phenolic TA structures. (C) Photographic images of the TNP suspension stability: TNP-APTES and TNP-MPTMS aggregated and precipitated within 1 h, whereas TNP-DCA and TNP-TA remained well-dispersed, indicating improved colloidal stability. UV-Vis, ultraviolet-visible; FT-IR, Fourier-transform infrared.
Catalase was immobilised on the TNP intermediates, each possessing a specific
functional group (amine, thiol, carboxylic, or phenolic) on its surface. Table 1
compares the catalase loading capacity and relative activity of the catalases
after the immobilisation reaction with the TNP intermediates (TNP-APTES,
TNP-MPTMS, TNP-DCA, and TNP-TA). The catalase loading on TNP-APTES, TNP-MPTMS,
TNP-DCA, and TNP-TA followed the order B
| Functional group | Catalase loading (%) | Relative activity (%) | |
| CITNP-A | Amine | 85.5 | 77.16 |
| CITNP-B | Thiol | 87.3 | 73.93 |
| CITNP-C | Carboxylic | 78.5 | 70.57 |
| CITNP-D | Phenolic | 75.5 | 60.21 |
The effect of temperature and pH on the activity of the catalase immobilised on the TNPs was investigated by determining the relative catalase activity over a broad range of temperatures (10–80 °C) and pH values (2.0–9.0). As shown in Fig. 3, both free and immobilised catalase exhibit maximum enzyme activity at approximately 40 °C. However, the CITNPs are thermally more stable than free catalase, retaining higher catalase activity over a broader temperature range, particularly at higher temperatures (50–80 °C) (Fig. 3A). Additionally, CITNP catalase, as compared with free catalase, exhibits high pH tolerance, maintaining substantial activity across a wide pH range (pH 2.0–9.0) (Fig. 3B). CITNPs demonstrated optimal catalytic activity at 40 °C and pH 6.0–7.0. Although enhanced stability after immobilisation is typically expected, the extent of activity retention varies across different surface functional groups, indicating that the immobilisation chemistry influences enzyme performance independently of simple loading effects.
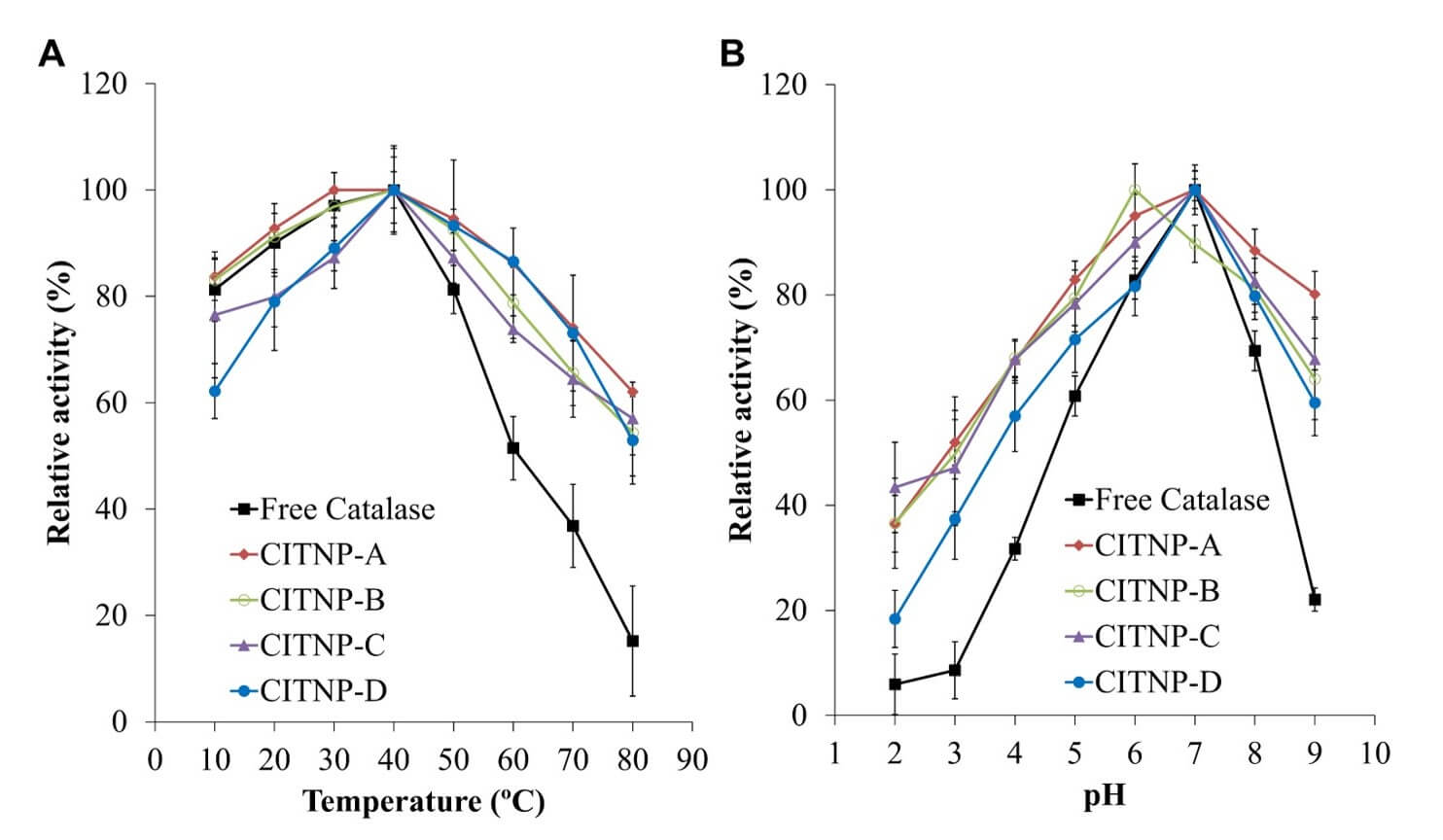 Fig. 3.
Fig. 3.
Effect of temperature and pH on the catalase activity of the
CITNPs. (A) Temperature profile: the catalase activity of the CITNPs derived
from each TNP intermediate was measured at 10–80 °C. The activities are
expressed relative to the maximum observed at 40 °C. Compared to free
catalase, the CITNPs exhibited broader thermal tolerance, retaining higher
relative activity at elevated temperatures. (B) pH profile: the catalase activity
was evaluated at pH 2.0–9.0 using appropriate buffer systems. The activities are
expressed relative to the maximum observed at pH 6. The CITNPs retained higher
activity than free catalase under both acidic and alkaline conditions, indicating
enhanced pH stability after immobilisation. Data is presented as mean
The thermal stability of catalase was examined by incubating CITNPs in 50 mM phosphate buffer at 70 °C for 60 min. Fig. 4A shows that the catalase activity of CITNPs prepared using various TNP intermediates decreases with increasing incubation time. Notably, the CITNPs exhibit a slower catalase activity reduction rate than free catalase, retaining 9%–28.9% of the initial catalase activity, depending on the preparation method, after 60 min of incubation at 70 °C. Additionally, CITNP catalase prepared with TNP-APTES demonstrates high reusability, as compared with free catalase, retaining 41.8%–76.3% of the initial catalase activity even after 10 reuse cycles (Fig. 4B), depending on the TNP intermediate. Notably, differences in operational stability were observed despite comparable immobilisation efficiencies, suggesting that enzyme–support interactions play a critical role in long-term performance.
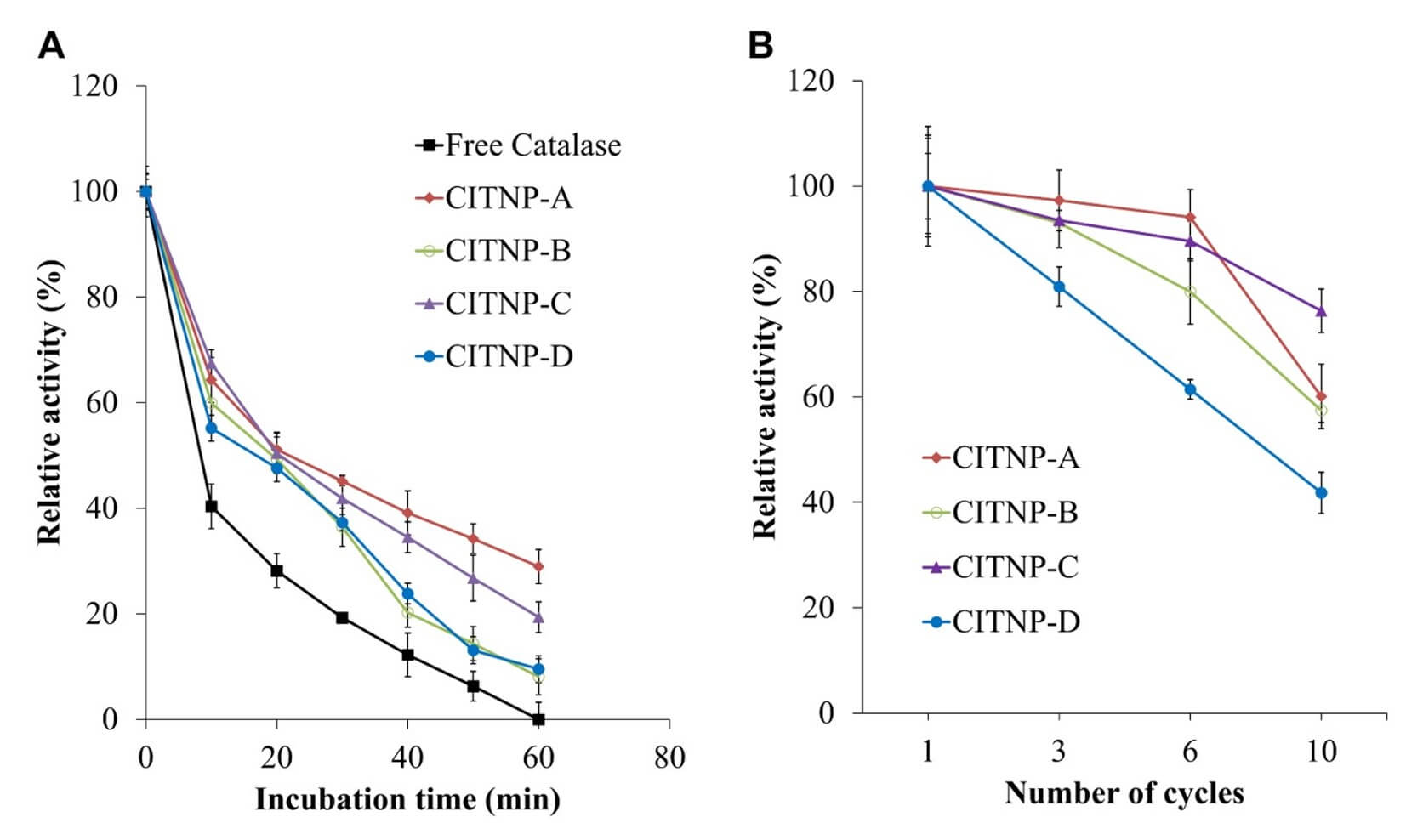 Fig. 4.
Fig. 4.
Thermal stability and reusability of CITNPs. (A) Thermal
stability: CITNPs were incubated at 70 °C in 50 mM phosphate buffer
(pH 7.0) for up to 60 min. The residual activity was measured and expressed
relative to the untreated controls. The CITNPs retained significantly higher
residual activity than free catalase, ranging from 9%–28.9% after 60 min. (B)
Reusability: the catalytic activity of the free catalase and CITNPs was assessed
over 10 consecutive reaction cycles. The activity was measured after each cycle
and expressed as a percentage of the initial activity (cycle 1). The CITNPs
retained 41.8%–76.3% activity after 10 cycles, while the activity of free
catalase declined by
Since the CITNPs prepared using TNP-APTES (method A) exhibited superior catalase
activity than the other TNP intermediates, it was selected for further evaluation
in removing H2O2 from tea or coffee extracts through the use of a CE
system (indirectly detected the Ti-TAR-H2O2 complex). As shown in Fig. 5A,B, the H2O2 peak rapidly decreases with an increasing CITNP
treatment time with the extract of tea or coffee, degrading
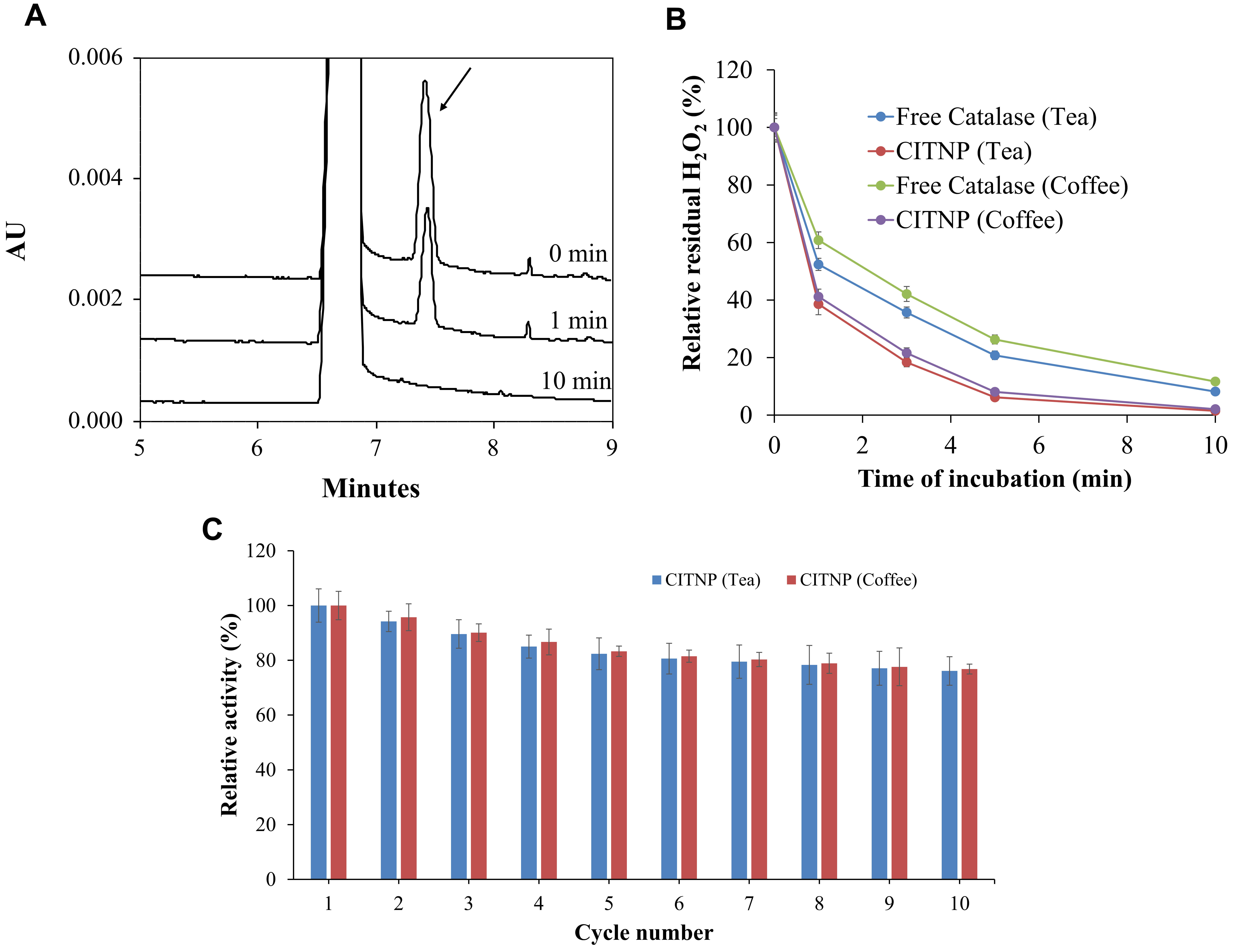 Fig. 5.
Fig. 5.
Catalytic activity and reusability of CITNPs in the removal of
H2O2 from tea and coffee extracts. (A) CE analysis of the
H2O2 concentration in tea extract before and after treatment with
CITNPs. The peak corresponding to the Ti-TAR-H2O2 complex (indicated by
an arrow at a migration time of 7.4 min) decreased progressively following
treatment with CITNPs (10 µg/mL) at 0, 1 and 10 min. (B) Time-course
of H2O2 removal during 10 min of incubation at 40 °C. Residual
H2O2 concentrations are expressed as a percentage of the initial
concentration in both tea and coffee extracts. (C) Reusability of CITNPs for
H2O2 removal. The enzyme activity was measured after each cycle and
expressed as a percentage of the initial activity (cycle 1). The CITNPs retained
CITNPs were prepared by modifying the surfaces of anatase-phase TNPs with distinct functional groups, namely, amino (method A), thiol (method B), carboxylic (method C), and phenolic (method D) groups. Each modified TNP intermediate exhibited unique properties that are likely influenced by the surface charge characteristics of the respective functional groups. For instance, TNP intermediates functionalised with amino or thiol groups (methods A and B) tended to undergo sedimentation in the suspension, whereas those functionalised with phenolic or carboxylic groups (methods C and D) remained well dispersed. Additionally, the TNP intermediates obtained from methods C and D exhibited strong UV absorbance at 300–400 nm, which was attributed to the presence of chromophores (Fig. 2A).
Covalent immobilisation of catalase onto TNP intermediates bearing amine
(-NH2), thiol (-SH), or carboxyl (-COOH) groups resulted in catalytic
activity recoveries of 70.6%–77.2%. Notably, TNP functionalised with APTES
(method A) exhibited the highest activity recovery (77.2%), suggesting that
amino groups facilitate efficient covalent bonding with catalase through Schiff
base formation or amide linkages, thereby enhancing enzyme retention and
stability [29, 30, 31]. Notably, the CITNPs prepared via method D (TA-modified TNPs),
in which catalase was non-covalently immobilised, also exhibited moderate
catalytic activity and stability comparable to those of its covalently bonded
counterparts (APTES, MPTMS, and DCA). This may be attributed to strong hydrogen
bonding and
CITNPs exhibited high thermal stability, retaining up to 28.9% of their initial activity after 60 min of incubation at 70 °C, whereas free catalase rapidly denatured under the same conditions. This enhanced thermal resistance can be attributed to the protective confinement effect of the TNP matrix, which restricts thermal unfolding and conformational changes in catalase, thereby preserving its structural integrity and catalytic function. This finding aligns with the results of a previous study by Wu et al. [35], who reported approximately 25% residual activity for catalase immobilised on titania submicrospheres under similar thermal stress. Comparable thermal resilience has been reported for other inorganic carriers such as mesoporous silica [36] and ZnO-based nanocomposites [37]. The CITNPs also exhibited broad pH tolerance and maintained substantial activity across a wide pH range (2.0–9.0). This represents a significant improvement over free catalase, which functions optimally near pH 6.5 but rapidly loses its activity in strongly acidic or alkaline environments. The increased pH stability likely resulted from the TNP matrix providing a protective microenvironment that buffered the enzyme against pH-induced denaturation [35, 38]. Similar pH resilience has been reported for catalase immobilised on chitosan-based and polymeric supports, which maintains activity at pH 5.0–9.0 [39]. The observed differences between the amine-, thiol-, carboxyl-, and phenolic-functionalised supports indicate that the effect of immobilisation is not solely determined by enzyme loading. In particular, the superior performance of amine-functionalised TiO2 suggests a more favourable balance between the binding strength and conformational freedom. Remarkably, CITNPs prepared with TNP-APTES exhibited high reusability, retaining 76.3% of their initial activity after 10 reuse cycles, surpassing other matrices, such as titanate nanotubes, which retained only 50% activity after nine cycles [38]. This suggests that the modified TNP matrix prevents catalase leaching and preserves its conformation. The catalytic activity and reusability of the CITNPs for H2O2 degradation were subsequently validated using CE. The CITNPs effectively decomposed the H2O2 in hot tea and coffee extracts and retained their catalytic activity after 10 cycles, demonstrating potential for industrial applications.
Tea and coffee extracts were selected as representative complex aqueous matrices rather than simple buffer systems. These beverages contain endogenously generated H2O2 resulting from polyphenol auto-oxidation during heating and storage, as previously reported by our group using CE. Therefore, their use enables the validation of catalytic performance and analytical robustness under realistic matrix conditions. Quantitative kinetic parameters, such as Km and Vmax, were not evaluated in this study because accurate kinetic analyses of immobilised enzymes require accounting for substrate diffusion and mass-transfer limitations. Future work will involve detailed kinetic modelling to further elucidate the catalytic behaviour of the immobilised system.
Catalase was successfully immobilised on TiO2 nanoparticles using different surface functionalisation strategies, and the resulting systems exhibited distinct activity retention and operational stability despite similar enzyme loading efficiencies. Among these, amine-functionalised TiO2 exhibited the highest stability and reusability, retaining a large fraction of its initial activity under repeated use and thermal stress. The novelty of this study lies in the direct comparative evaluation of multiple functional intermediates on a single TiO2 platform, demonstrating that the immobilisation chemistry critically governs enzyme performance. These findings highlight the potential of functionalised TiO2-catalase systems for practical H2O2 removal in food, environmental, and biotechnological applications.
TNP, titanium dioxide nanoparticles; TiO2, titanium dioxide; CITNP, catalase-immobilised titanium dioxide nanoparticles; H2O2, hydrogen peroxide; APTES, (3-aminopropyl)triethoxysilane; DCA, dihydrocaffeic acid; MPTMS, (3-mercaptopropyl)trimethoxysilane; TA, tannic acid; GLA, glutaraldehyde; EDC, 1-ethyl-3-(3-dimethylamino)propyl carbodiimide hydrochloride; NHS, N-hydroxysuccinimide; BSA, bovine serum albumin; DW, deionised water; UV-Vis, ultraviolet-visible; FT-IR, Fourier-transform infrared spectroscopy; MES, 2-(N-morpholino)ethanesulfonic acid; CE, capillary electrophoresis; Ti-TAR-H2O2, ternary complex of titanium(III) chloride, 4-(2-thiazolylazo)resorcinol, and hydrogen peroxide; ROS, reactive oxygen species.
The datasets generated in this study are available from the corresponding author upon request.
PDV conducted the experiments and analysed the data. HY supervised the research and contributed to the interpretation of the results. Both authors wrote and reviewed the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research was supported by the Basic Science Research Program through the NRF of Korea, funded by the Korean Government (No. RS-2023-00222390).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





