1 Department of Neurology and Neurophysiology, Liverpool Hospital, Liverpool, NSW 2170, Australia
2 Neurophysiology Department, Liverpool Hospital, Liverpool, NSW 2170, Australia
3 South Western Sydney Clinical School, University of New South Wales, Liverpool, NSW 2170, Australia
4 Ingham Institute for Applied Medical Research, Liverpool, NSW 2170, Australia
5 Neurology Department, Liverpool Hospital, Liverpool, NSW 2170, Australia
6 South Western Clinical School, University of NSW, Sydney, NSW 2170, Australia
7 Griffith University, Southport, QLD 4215, Australia
8 Western Sydney University, Sydney, NSW 2000, Australia
Abstract
Epilepsy is a neurological syndrome with a significant associated morbidity and mortality. An electroencephalogram (EEG) is an important tool to assist in the diagnosis of epilepsy. Sleep deprivation is a well-known risk factor for seizures and forms a basis for why sleep-deprived EEGs may assist in diagnosing epilepsy. There is mixed evidence regarding the utility of sleep deprivation in identifying signs of seizure activity, known as epileptiform activity. This study aimed to assess whether sleep-deprived EEGs increase the yield of epileptiform activity, compared with routine EEGs.
This was a retrospective observational study (January, 2018 to January, 2023) in patients of all ages who underwent routine and sleep-deprived EEGs at a major tertiary referral centre. Descriptive statistical analysis was undertaken in addition to the Fisher exact test and McNemar test to facilitate comparison of matched pairs.
There were 116 patients included in the study. The median age at time of first EEG was 32.5 years (interquartile range (IQR) 18–53). Fifty four percent of patients were male and 80% of patients were aged ≥18 years. The diagnostic yield of epileptiform activity was 21% when the results of both the routine and sleep-deprived EEG studies were included. There was no significant difference in the yield of epileptiform activity in routine versus sleep-deprived EEGs (12% versus 13% respectively, p = 1.0). Nine patients had epileptiform activity present on routine but not sleep-deprived EEG, and the reverse was true for 10 patients. There were several study limitations, including the varied time interval between EEG studies and limited referral documentation.
The study findings suggest that, in the correct clinical context, undertaking both a routine and sleep-deprived EEG, regardless of the order, may have benefit in identifying epileptiform activity. These findings also reinforce important data points that should be included with EEG referrals, to help optimise future research in this area.
Keywords
- sleep-deprivation
- electroencephalography
- epilepsy
- seizures
Epilepsy affects approximately 70 million people worldwide [1]. At least 75% of those with active epilepsy, in low-income countries, are inadequately treated, compared with less than 10% in high-income countries [2]. Even in well-resourced countries, diagnosing seizures and epilepsy can be difficult [3].
An electroencephalogram (EEG) is a standard tool used to help identify epileptiform activity, consistent with seizure activity [4]. One area of uncertainty is the usefulness of sleep-deprived EEGs, compared to routine (non-sleep-deprived) EEGs, in the diagnosis of seizures and epilepsy [5]. Sleep deprivation is a well-known risk factor for seizures in those with epilepsy [6]. This phenomenon forms the basis for why sleep-deprived EEGs are thought to assist in diagnosing seizures and epilepsy [7]. Sleep deprivation has also been shown to increase the yield of other manoeuvres that may trigger seizure episodes, such as photic stimulation and hyperventilation [8]. With respect to paediatric patients, where epileptiform discharges, in some seizure syndromes, are best seen during sleep, the American Clinical Neurophysiology Society recommends sleep recordings should be obtained whenever possible [9].
There exists some uncertainty as to the physiological basis by which epileptiform activity can be induced or activated by sleep-deprivation [10]. Cortical excitability appears to be enhanced with sleep-deprivation, particularly in some specific epilepsy syndromes, and is a potential explanation [10, 11, 12]. This mechanism may contribute to the phenomenon whereby the occurrence of epileptiform activity in sleep-deprived patients can occur independently of the state of alertness or sleep during the EEG study [13].
Whilst some studies have suggested no clinical benefit in detection of epileptiform activity with sleep-deprived EEGs [14, 15, 16, 17], others have suggested the opposite [8, 18, 19, 20]. One prospective study found that an EEG, with partial age-dependent sleep deprivation, identified epileptiform activity in one third of patients who initially had a normal standard EEG following an idiopathic or remote symptomatic seizure [20].
Clinicians and institutions often adopt a range of different protocols to undertake sleep-deprived EEGs. This adds further uncertainty to comparisons with routine EEGs [5]. The number of hours of sleep is one such example of heterogeneity in protocols [10]. Published studies have utilised different hours of sleep deprivation according to the participant’s age [17, 20]. One prospective, randomised, physician-blinded study concluded that partial sleep deprivation, as opposed to full-night sleep deprivation, is adequate to achieve any advantages of sleep deprivation for EEG [21].
Further confounding the yield of sleep deprivation is that routine EEGs tend to be of shorter duration than are sleep-deprived EEGs [10, 22]. A large prospective study of outpatient EEGs, at one institution, demonstrated that increasing the duration of a routine study, from 30 to 45 minutes or longer, increased the relative yield of new interictal epileptiform discharges by approximately 20% [23]. At the institution from the current study, routine EEGs are usually 20 minutes in duration versus 60 minutes for sleep-deprived EEGs.
Ordering sleep-deprived EEGs requires caution as they can be a significant burden and interruption to the patient’s lifestyle [5, 17, 24]. Sleep deprivation also carries the inherent risk of, at least temporarily, lowering the threshold for that patient to develop a seizure and can exacerbate behavioural difficulties in patients with developmental challenges [9]. A survey of children, undergoing sleep-deprived EEGs, found significant associated burden, including fatigue and the need for parents to stay awake with two reported cases of seizures the next day [24]. The 2022 National Institute for Health and Care Excellence guidelines recommend for consideration of sleep-deprived EEG following a normal routine EEG after discussion of risks and benefits [25].
This retrospective study aimed to assess whether sleep-deprived EEGs increased the yield of epileptiform activity, compared to routine EEGs. To improve the validity and reliability of the data, the study only included patients who underwent both a routine and sleep-deprived EEG. This research aimed to unravel the uncertain evidence base, regarding the utility of sleep-deprived EEGs. It was designed to assist in optimising protocols to improve yield whilst minimising patient and carer inconvenience associated with sleep deprivation.
This was a retrospective, single-centre observational study and did not affect routine care of participants. Baseline demographics included the age and gender of each participant. The current study utilised the Strengthening Reporting of Observational Studies in Epidemiology (STROBE) guidelines to optimise quality of reporting (Supplementary Material) [26].
There was no age restriction for patients. Data from EEG reports were retrospectively collected for patients who had undergone both a routine EEG and sleep-deprived EEG study during a five-year period (January 2018 to January 2023). The choice of test for each patient was pre-specified by the referring practitioner and may have originated from an inpatient, emergency department or outpatient setting. Referrers were not necessarily required to use the same referral form. Participants must have had both studies performed at Liverpool Hospital, a major tertiary referral and teaching hospital in south-western Sydney, in the state of New South Wales, Australia.
In situations where patients had more than two EEG studies, the routine and sleep-deprived EEG studies with the shortest inter-study interval were selected.
EEGs were recorded in accordance with the International 10–20 system. Duration of routine and sleep-deprived EEG recording time was 20 minutes and 60 minutes respectively. A Natus brain monitor EEG amplifier PK1274 (Natus Medical Incorporated, Middleton, MI, USA) was utilised and electrode placement followed the international 10–20 system. The protocol, at this institution, for sleep deprivation was for the patient to have undertaken 24 hours of sleep deprivation—any deviation from this protocol was noted. Patients were excluded if they had experienced at least four hours of sleep prior to the sleep-deprived EEG.
Routine triggering manoeuvres, including hyperventilation and photic stimulation, were utilised in both the routine and sleep-deprived studies. Major tertiary-referral hospitals in Sydney with epilepsy services were contacted to assess their protocols.
EEGs were reported by experienced clinical neurologists from the same institution who had level 2 or 3 Australian and New Zealand Association of Neurologists (ANZAN) certification for EEG training and education. Each of the reporting neurologists had at least 10 years of experience in clinical and EEG reporting practice. Inter-ictal epileptiform activity was identified by the reporting neurologist in accordance with previously specified criteria [27]. Topography and clinical phenotype of seizure semiology were not analysed. Neurophysiology technologist staff assisted the primary author with locating EEG data from the Liverpool Hospital Neurophysiology Department.
The primary author de-identified and collected data from each EEG report including: date of study; whether the study was normal or abnormal; presence and type of epileptiform activity; state of patient during epileptiform activity; details regarding non-epileptiform activity; any prescribed anti-seizure medications (ASMs); time elapsed between the study and seizure; speciality of the referrer; amount of sleep deprivation; and time elapsed between routine and sleep-deprived studies. In situations where there was suspicion of possible epileptiform activity, rather than confirmed epileptiform activity, this was also noted. If available, referral paperwork was analysed to provide these data.
The primary outcome was the yield of epileptiform activity for the same patient when comparing the routine to the sleep-deprived EEG. Sub-group analyses were performed based on age, gender, referrer status and order of study requests. If available, comparisons were made with reference to other collected data points.
The primary author analysed the data and applied statistical tests with the “R” statistical package (4.4.2, R Foundation for Statistical Computing, Vienna, Austria). Descriptive statistical analyses were performed to summarise the data. For continuous variables, median and interquartile range (IQR) were reported. McNemar’s test was used to compare between routine and sleep-deprived EEG studies with regards to the presence of epileptiform activity and undertake subgroup analysis. Fisher’s Exact Test was used to evaluate the effect of prespecified subgroups on diagnosis of epileptiform activity for each type of EEG. A p value of less than 0.05 was chosen as the significance level for all statistical tests.
There were 116 patients included in the study. Fifty four percent of patients were male and 80% of patients were aged at least 18 years of age. There were no patients under 13 years of age who underwent both a routine and sleep-deprived EEG during the study period. A total of 114 patients (98%) completed the prescribed 24 hours of sleep deprivation. In this study, 97 patients (84%) underwent the routine EEG prior to the sleep-deprived EEG. The group demographics are further outlined in Table 1.
| Routine EEG (n = 116) | Sleep-deprived EEG (n = 116) | ||
| Age at time of the study | |||
| median (IQR) | 32.5 (18–53) | 32.5 (18–54) | |
| 23 (20%) | 23 (20%) | ||
| 93 (80%) | 93 (80%) | ||
| Male gender, n | 63 (54%) | 63 (54%) | |
| Initial EEG study undertaken, n | 97 (84%) | 19 (16%) | |
| Completion of 24 hours sleep deprivation, n | N/A | 114 (98%) | |
| Documentation of antiseizure medication status, n | 4 (3%) | 13 (11%) | |
| nil antiseizure medication prescribed, n | 0 | 5 | |
| one antiseizure medication prescribed, n | 3 | 4 | |
| two antiseizure medications prescribed, n | 1 | 4 | |
| Documentation of time since last suspected seizure, n | 6 (5%) | 3 (3%) | |
| one day since last suspected seizure, n | 4 | 1 | |
| two days since last suspected seizure, n | 1 | 1 | |
| three days since last suspected seizure, n | 1 | 1 | |
| Documentation of source of referrer, n | 38 (33%) | 49 (42%) | |
| referred by a neurologist, n | 30 (26%) | 40 (34%) | |
| referred by a non-neurologist, n | 8 (7%) | 9 (8%) | |
| EEG report | |||
| within normal limits, n | 63 (54%) | 70 (60%) | |
| non-epileptiform abnormality, n | 42 (36%) | 33 (28%) | |
| epileptiform or suspicion for epileptiform activity, n | 21 (18%) | 21 (18%) | |
| epileptiform activity, n | 14 (12%) | 15 (13%) | |
EEG, electroencephalogram; IQR, interquartile range; N/A, not applicable.
Documentation as to whether a patient was prescribed an ASM was present more often in sleep-deprived EEG referrals, compared to routine EEG referrals (11% versus 3%). There were few EEG referrals that documented the time since the last suspected seizure (5% of the routine EEGs and 3% of the sleep-deprived EEGs). Documentation as to the identity of the referring physician was available for 33% of the routine EEGs and 42% of the sleep-deprived EEGs.
EEGs were reported as being within normal limits in 54% of routine and 60% of sleep-deprived EEGs. Non-epileptiform abnormalities were identified in 36% of routine and 28% of sleep-deprived EEGs.
Epileptiform activity was identified in 24 patients (21%) when the results of both routine and sleep-deprived EEG studies were included. There was no significant difference in the number of EEGs with epileptiform activity when comparing the routine and sleep-deprived group studies (12% versus 13% respectively, p = 1.0). Five patients (4.3% of the whole cohort) had epileptiform activity present on both routine and sleep-deprived EEGs.
Test order was found to significantly affect the overall diagnostic yield of undertaking both tests. Sleep-deprived EEGs were significantly more likely to demonstrate epileptiform activity if undertaken prior to the routine EEG (7/19 (37%) versus 8/97 (8%), p = 0.003). Routine EEGs were significantly more likely to demonstrate epileptiform activity if undertaken following a sleep-deprived EEG (6/19 (32%) versus 8/97 (8%), p = 0.011).
All eight patients with epileptiform activity on initial routine EEG subsequently had a sleep-deprived EEG undertaken. All seven patients, with epileptiform activity on an initial sleep-deprived EEG, subsequently had a routine EEG undertaken. Amongst patients who had epileptiform activity identified on their initial routine or sleep-deprived EEG, the proportion of subsequent EEGs with epileptiform activity did not differ significantly between the groups (2/8 (25%) versus 3/7 (43%) respectively, p = 0.61).
Epileptiform activity was identified on routine EEG but not sleep-deprived EEG in nine patients (7.8%) and the reverse was true for 10 patients (8.6%). Patients with an initial negative sleep-deprived EEG were more likely to have epileptiform activity on the subsequent EEG in comparison with patients who had an initial negative routine EEG (3/12 (25%) versus 6/89 (7%)), although this difference was not significant (p = 0.072). These data are further categorised by age group in Fig. 1.
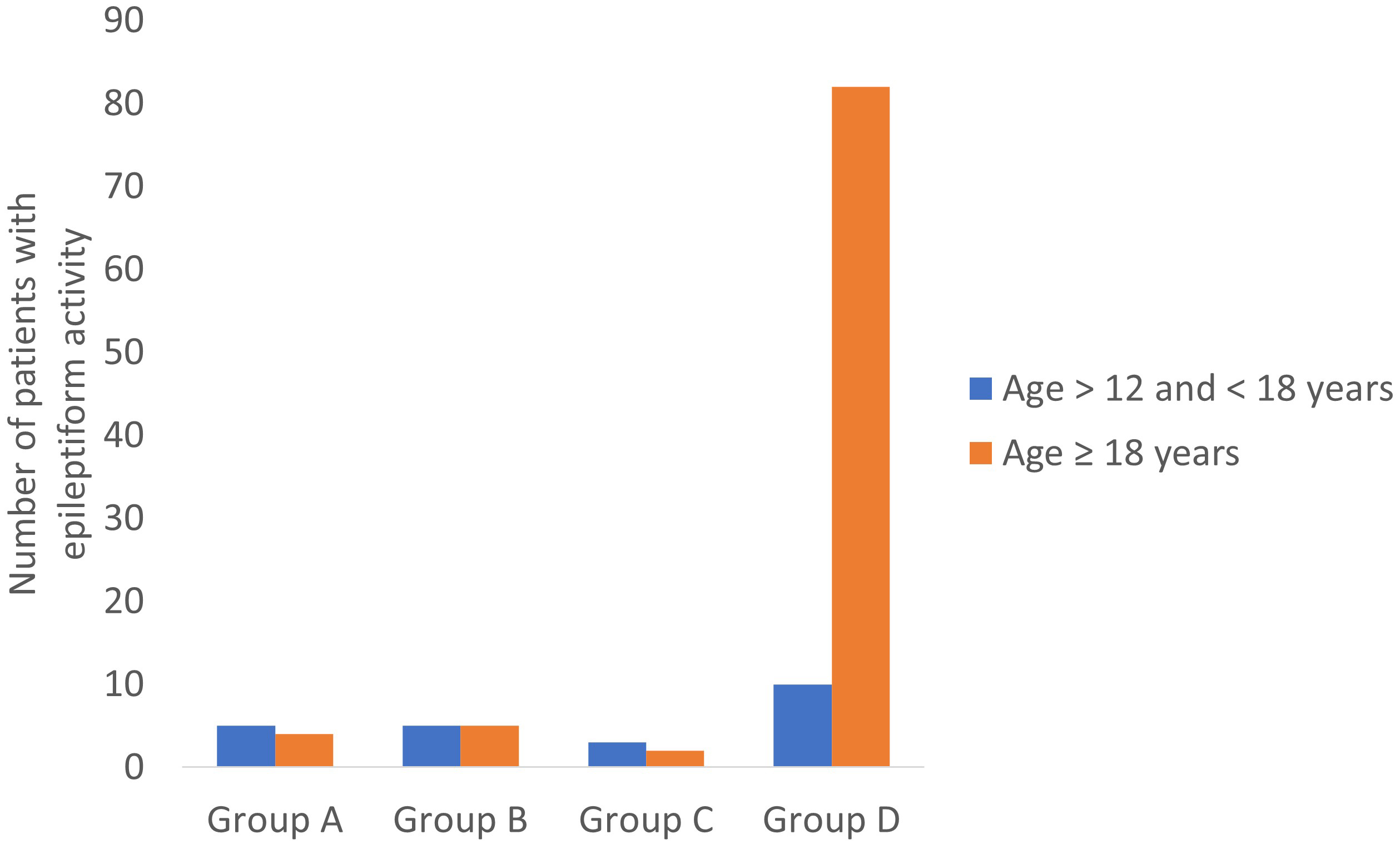 Fig. 1.
Fig. 1.
Presence of epileptiform activity on routine and sleep-deprived EEGs, grouped according to age. Group A—Epileptiform activity on routine but not sleep-deprived EEG; Group B—Epileptiform activity on sleep-deprived but not routine EEG; Group C—Epileptiform activity on both routine and sleep-deprived EEG; Group D—No epileptiform activity on routine or sleep-deprived EEG.
There was a significantly higher proportion of patients aged
| Routine EEG (n = 116) | Sleep-deprived EEG (n = 116) | |||||
| Epileptiform activity | No epileptiform activity | p value (Fisher exact) | Epileptiform activity | No epileptiform activity | p value (Fisher exact) | |
| Age |
8 (35%) | 15 (65%) | 0.001 | 8 (35%) | 15 (65%) | 0.002 |
| Age |
6 (6%) | 87 (94%) | 7 (8%) | 86 (92%) | ||
| Male gender, n | 6 (10%) | 57 (90%) | 0.40 | 9 (14%) | 54 (86%) | 0.78 |
| Female gender, n | 8 (15%) | 45 (85%) | 6 (11%) | 47 (89%) | ||
| Referrer identified, neurologist, n | 2 (7%) | 28 (93%) | 0.19 | 6 (15%) | 34 (85%) | 0.63 |
| Referrer identified, non-neurologist, n | 2 (25%) | 6 (75%) | 2 (22%) | 7 (78%) | ||
In a subgroup analysis, when comparing routine and sleep-deprived EEGs, there was no significant difference in the identification of epileptiform activity based on age group, gender and known identity of referrer (Table 3). More details as to the nature of epileptiform activity are stratified according to age group in Table 4.
| Presence of epileptiform activity, n (%) | p value (McNemar test) | ||
| Routine EEG | Sleep-deprived EEG | ||
| Age |
8 (35%) | 8 (35%) | 1.0 |
| Age |
6 (6%) | 7 (8%) | 1.0 |
| Male gender | 6 (10%) | 9 (14%) | 0.55 |
| Female gender | 8 (15%) | 6 (11%) | 0.72 |
| Referrer identified, neurologist | 2 (7%) | 6 (15%) | 1.0 |
| Referrer identified, non-neurologist | 2 (25%) | 2 (22%) | 1.0 |
| Routine EEG demonstrating epileptiform activity | Sleep-deprived EEG demonstrating epileptiform activity | ||||
| Age |
Age |
Age |
Age | ||
| Gender, n | |||||
| Male | 4 | 2 | 5 | 4 | |
| Female | 4 | 4 | 3 | 3 | |
| Order of EEG acquisition, n | |||||
| Routine EEG undertaken first | 4 | 4 | 3 | 5 | |
| Sleep-deprived EEG undertaken first | 4 | 2 | 5 | 2 | |
| Source of referrer, n | |||||
| Referred by a neurologist | 1 | 1 | 1 | 5 | |
| Referred by a non-neurologist | 2 | 0 | 2 | 0 | |
| Referrer not determined | 5 | 5 | 5 | 2 | |
| Type of epileptiform activity, n | |||||
| focal | 1 | 2 | 3 | 3 | |
| generalised | 7 | 4 | 5 | 4 | |
| State of the patient during epileptiform activity, n | |||||
| alert | 4 | 2 | 2 | 1 | |
| drowsy | 0 | 1 | 1 | 1 | |
| asleep | 1 | 0 | 1 | 1 | |
| alert and drowsy/asleep | 2 | 3 | 3 | 2 | |
| drowsy/asleep | 1 | 0 | 1 | 2 | |
| Type of epileptiform activity, n | |||||
| ictal | 1 | 0 | 0 | 0 | |
| inter-ictal | 7 | 6 | 8 | 7 | |
Amongst all patients, the median time elapsed between each pair of EEGs was 53 days (IQR 16–154 days). Regarding patients who underwent a routine EEG as the first study, the median time elapsed until a subsequent sleep-deprived EEG was 45 days (IQR 10–111 days). Regarding patients who underwent a sleep-deprived EEG as the initial study, the median time elapsed until a subsequent routine EEG was 192 days (IQR 49–414 days) (Fig. 2). Due to the relatively low sample size, any effect of variation in time between paired studies was difficult to further characterise.
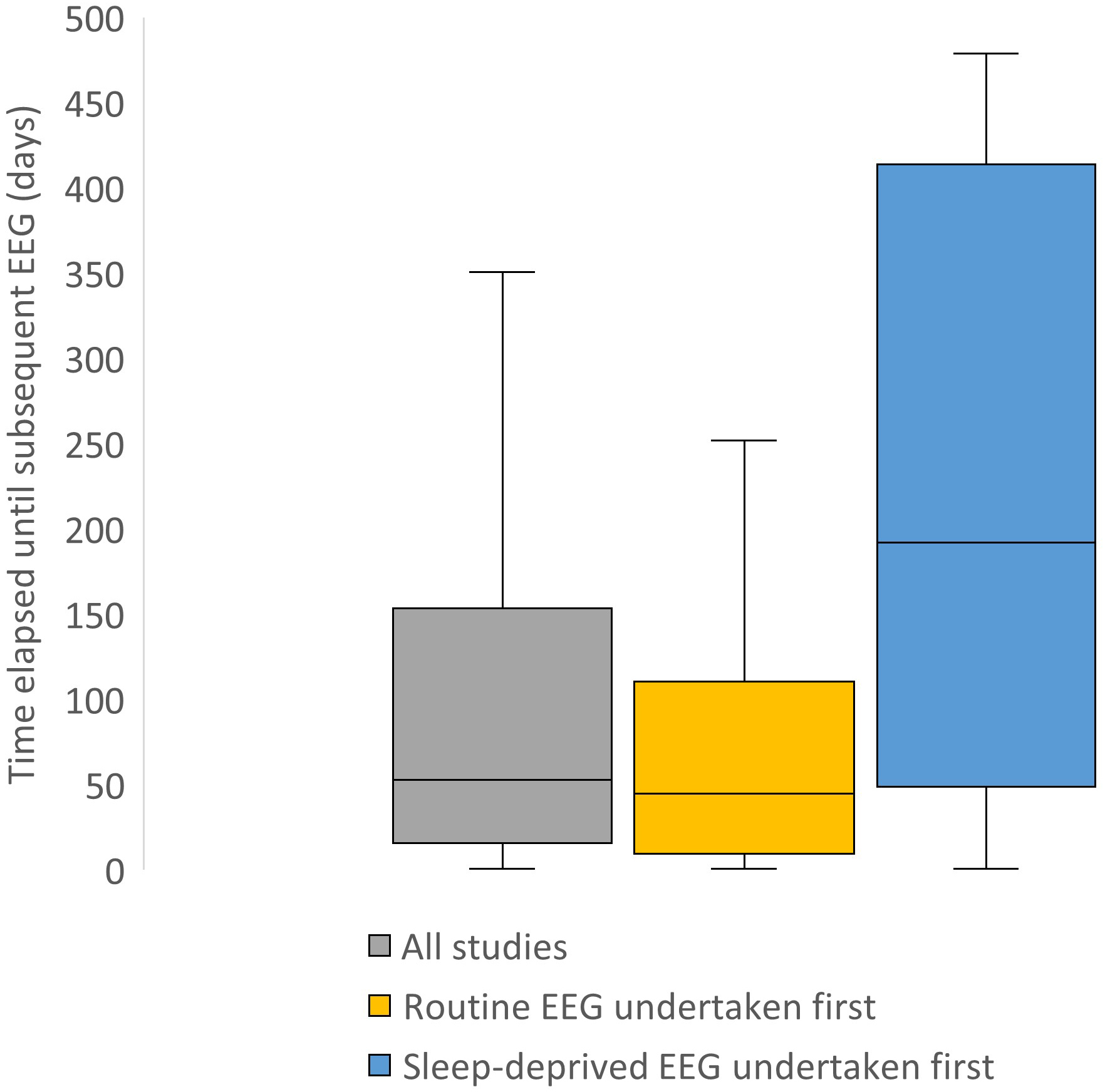 Fig. 2.
Fig. 2.
Box and whisker plot demonstrating time elapsed between each pair of EEG studies, measured in days. The grey box represents all patients in the study. The yellow box represents patients who had the routine EEG undertaken prior to the sleep-deprived EEG. The blue box represents patients who had the sleep-deprived EEG undertaken before the routine EEG. Outliers, defined as values greater than or equal to 1.5 times the interquartile range, have been removed to facilitate readability.
Of the tertiary-referral hospitals that responded with written protocols, heterogeneity existed amongst instructions for sleep-deprivation. Variations included: staying awake the entire night prior; losing at least half of normal sleep; allowing sleep between 1 and 5 AM; and age-specific criteria. The duration of sleep-deprived recordings varied from at least 40 minutes and up to 90 minutes in some cases.
The present study did not demonstrate a significant difference between routine and sleep-deprived EEGs with regards to the yield of epileptiform activity. This is not an unexpected result given the existing uncertainty in the literature regarding the utility of routine versus sleep deprived-EEGs.
In this study sleep-deprived EEGs were more likely to demonstrate epileptiform activity if undertaken first. An explanation for this may be related to the combined effect of a potentially higher test yield with sleep deprivation and shorter time interval to assessment following a clinical event [28]. Most initial EEGs in this study were routine studies (84%), which may have reduced the yield of sleep-deprived EEGs, but is in keeping with real world experience and protocols in the literature [10].
Complicating the interpretation of results from this study was the finding that routine EEGs were more likely to demonstrate epileptiform activity if undertaken as the second test. Although not answered by these data, in the setting of limited referral documentation, clinical experience suggests that this finding may be secondary to a selection bias–there may be a more undifferentiated population with lower pre-test probability of epileptiform activity referred for an initial routine EEG versus those initially referred for a sleep-deprived EEG. The ordering of a routine EEG after a sleep-deprived study may indicate a heightened suspicion of epileptiform activity, on the basis of clinical information, but this information was not available in the referral data.
An association was not found between the type of referrer and yield of epileptiform activity, although data in this study were limited as to the numbers of known referrers. There is some evidence in the literature as to increased yield with neurologist screening of referrals but comparison with this study is difficult [29]. Variation in referrers for EEG studies reflects real-world experience, which strengthens the external validity of results from this study.
A notable finding, partly related to the design of this study, was that all patients with an initial EEG demonstrating epileptiform activity went on to have a subsequent EEG study. Due to limited data available, the reasoning for this was unclear. It may be that the referring clinician requested for an additional EEG for diagnostic clarification following their review of raw EEG tracing. The subsequent EEG study may also have been undertaken for a different indication, such as for the optimisation of ASM dosing in some clinical contexts.
The significantly higher proportion of epileptiform activity identified on
routine and sleep-deprived EEGs in patients aged
A potential confounder in this study was the longer median time interval between each pair of EEG studies in patients who initially had a sleep-deprived EEG compared to those who initially had a routine EEG. This finding may suggest that referrers attribute greater value to the results of a sleep-deprived EEG. A “second” EEG undertaken also may have reflected a separate future clinical event, rather than assessment of the same initial clinical event.
The estimation of the yield of sleep-deprived EEGs was optimised in the present study in that 98% of patients achieved 24 hours of sleep deprivation. The inclusion criteria for less than four hours of sleep-deprivation was established to reduce any effect of poor adherence to sleep-deprivation instructions: the criteria for sleep-deprivation in the present study was equal to or more strict than multiple sleep-deprived protocols of some other nearby tertiary-referral centres. In this way, although two patients in the present study did not achieve a full 24 hours of sleep-deprivation, they had sufficient sleep-deprivation to allow for optimised external validity of our results.
The present study is potentially limited in that it did not explore the time spent awake versus asleep. Association of epileptiform activity with stages of sleep was not analysed. The added value of this information is questionable in that there is mixed evidence as to whether sleep-deprivation or sleep itself is the trigger for epileptiform activity in sleep-deprived patients [10].
There is a possibility that the longer duration of sleep-deprived EEG studies, in comparison to the routine studies, has contributed to an overestimation of their yield by virtue of more prolonged monitoring. The external validity of these results is supported by the typically longer duration of sleep-deprived recordings as noted in the literature [10].
Adding external validity to these results is the fact that Liverpool Hospital is a large tertiary centre, with a significant multicultural population. In a government publication, 49% of people, within the local health district, reported to speak a language other than English at home [31]. The most spoken languages, other than English, in the district were Arabic, Vietnamese, Mandarin, Cantonese and Greek [31]. This may have affected the level of adherence to the performance of the study but reflects real-world experience.
The significant methodological heterogeneity in the literature represents a challenge in comparing these results to other studies [10]. Although the current study only included patients who had undergone both routine and sleep-deprived EEG testing, some other studies in the literature have not necessarily required both EEG techniques to be undertaken on the same participant [16, 17, 21]. These studies also did not specify the seizure date in relation to the date of the EEG study [16, 17, 21]. A retrospective study, of adult patients who underwent emergency EEG, following a first unprovoked seizure, concluded that the diagnostic yield was highest when performed within 16 hours after the onset of the event [28]. One study, of 300 consecutive adults and children with unexplained seizures, found epileptiform abnormalities in 51% of patients who had an EEG within 24 hours versus 34% of those with a later EEG [32].
The retrospective nature of data collection in this study has potentially introduced confounders to the results. EEG abnormalities were identified from their respective reports so the threshold for reporting of epileptiform and non-epileptiform findings may have differed between individual reporting neurologists, thus introducing bias. The relatively small sample size and large variability in time intervals between pairs of EEG studies may also impact upon the results. Limited data were available as to the date of last known seizure and prescribed ASMs, factors that can influence the presence and nature of epileptiform activity on EEG [33].
Aside from sleep deprivation, there are other techniques outside the scope of this study that may increase the yield of identifying epileptiform activity. One such technique is known as “ambulatory-EEG monitoring” [34], in which prolonged EEG testing, over hours to days, can occur in an outpatient setting. This has its own limitations including accessibility and higher costs to patients. One prospective study [35] assessed patients with a historical suggestion of epilepsy but a normal or non-diagnostic initial routine EEG and found a greater benefit with ambulatory EEG, when compared with sleep-deprived EEGs. Contrasting this was a retrospective cohort study that found a similar diagnostic accuracy of a subsequent sleep-deprived or ambulatory EEG (sensitivity 45% versus 63% respectively) in patients who had a normal routine EEG after experiencing a first unprovoked seizure [36]. The 2022 National Institute for Health and Care Excellence guidelines recommend for consideration for a subsequent ambulatory EEG in some cases, after both a normal routine and sleep-deprived EEG, where the diagnosis of epilepsy remains obscure but probable [25].
In an unselected population of patients who underwent both routine and sleep-deprived EEGs, the diagnostic yield of epileptiform activity was 21%. There was no significant difference in the yield of epileptiform activity between routine and sleep-deprived EEGs. If an initial routine or sleep-deprived EEG did not identify epileptiform activity, a subsequent study of the opposite type (namely, sleep-deprived or routine EEG, respectively) had value in identifying epileptiform activity, despite a relatively low yield. Sleep-deprived EEGs were more likely to demonstrate epileptiform activity if undertaken first, although this may have been confounded by a selection bias making it difficult to proffer an argument that all initial EEGs should be sleep-deprived.
The findings, from this “real-world” study, support the notion that both types of EEGs may be helpful in the correct clinical context. The retrospective nature of this study in addition to other confounders, including variability in time elapsed between EEG studies, limit interpretation of these findings. To optimise the validity and reliability of future research into this topic, more uniform protocols should be established for sleep deprivation. Referrers should clearly document the date of last suspected seizure and currently prescribed ASMs. A prospective or randomised trial may also represent a way of addressing the multiple confounders between studies in the literature.
This research was initially presented as a conference poster, with the abstract being published on 16/8/24 at the following URL: https://neurologyopen.bmj.com/content/6/Suppl_1/A5.2.
EEG, electroencephalogram; ASM, anti-seizure medication.
In line with the established ethics approval for the study, data are not currently available to be shared. If there is a request for data, study authors can liaise with the local ethics committee for approval.
DG was the principal person who conducted the research study. DG was the primary person responsible for study conceptualisation, ethics application, study design, data collection, data analysis, manuscript preparation and manuscript editing. AJ assisted with data collection and manuscript editing. DC assisted with study design, data analysis and manuscript editing. RB assisted with study conceptualisation, study design, data analysis and manuscript editing. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and approved by the South Western Sydney Local Health District Human Research Ethics Committee (2023/ETH00699). As part of this approval, data were anonymised and the need to obtain consent, for the collection, analysis and publication, was waived.
The authors would like to acknowledge the efforts of the reporting neurologists and neurophysiology staff who contributed to the data used for this study. The authors extend appreciation to Wei Xuan, a senior biostatistician, for support with statistical analysis of the results.
This research received no external funding.
The authors declare no conflict of interest. Professor Roy Beran is serving as one of the Editorial Board members of this journal. We declare that Roy Beran had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Bettina Platt.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN36232.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


