1 Neurology Department, The People's Hospital of Chizhou, 247100 Chizhou, Anhui, China
2 Clinical Nutrition Department, Guangdong Provincial Hospital of Chinese Medicine, 510030 Guangzhou, Guangdong, China
3 School of Public Health, Southern Medical University, 528305 Guangzhou, Guangdong, China
4 The First Clinical Medical School, Guangdong Pharmaceutical University, 510006 Guangzhou, Guangdong, China
5 Research and Development Center, Center of Human Microecology Engineering and Technology of Guangdong Province, 510700 Guangzhou, Guangdong, China
†These authors contributed equally.
Abstract
Insomnia, the most prevalent sleep disorder, is clinically defined as difficulty initiating or maintaining sleep. Although many medications are effective for insomnia treatment, they carry risks of drug dependence and abuse. The microbiota-gut-brain axis (MGBA) facilitates bidirectional signaling between the gastrointestinal tract and the central nervous system via gut microbes. Probiotics that provide mental and behavioral benefits through MGBA (psychobiotics) offer broad therapeutic potential.
A non-toxic, drug-resistant strain of Lactobacillus reuteri E9 was isolated and characterized. Its effects were evaluated in a pentylenetetrazol (PTZ)-induced zebrafish model of sleep disorder. Neurotransmitter levels (glycine, serine, taurine, γ-aminobutyric acid (GABA)) and gene expression of GABA/melatonin receptors were analyzed.
E9 significantly upregulated inhibitory neurotransmitters, including GABA, taurine, glycine, and serine (p < 0.05). In PTZ-induced zebrafish, E9 exerted sedative effects by reducing seizures and hyperactivity. Concurrently, E9 upregulated the expression of GABA receptor genes and melatonin receptor (Mtnr1aa) genes in zebrafish neural tissue.
Lactobacillus reuteri E9 demonstrates potential as a psychobiotic for sleep disorder management by modulating key inhibitory neurotransmitters and sleep-related receptor expression via the MGBA pathway, offering a non-pharmacological alternative to conventional treatments.
Keywords
- Lactobacillus reuteri
- gamma-aminobutyric acid
- sleep disorders
- zebrafish
Insomnia, particularly sleep-onset and sleep-maintenance subtypes, ranks among the most prevalent sleep disorders. Sleep deficiency can lead to symptoms including amnesia, irritability, depression, attention deficits, and fatigue. Beyond impairing cognitive function, sleep disorders are associated with metabolic syndromes such as obesity, inflammation, diabetes, and cardiovascular disease [1]. Although multiple pharmacological treatments exist, including benzodiazepine receptor agonists, antihistamines, melatonin receptor agonists, anxiolytics, antidepressants, and antipsychotics, these agents carry significant disadvantages. Notably, they pose risks of dependence, abuse, and adverse effects such as vertigo, headache, somnolence, amnesia, cognitive impairment, and even increased mortality [2]. Consequently, a safe therapeutic alternative is needed to reduce reliance on sedative-hypnotics, thereby improving sleep quality and efficiency in insomnia patients without substantial side effects.
Gut microbiota (GM) influences host physiology through diverse pathways, including nutritional modulation, immune regulation, and neurotransmitter and hormone production. Studies indicate that sleep disorders may contribute to metabolic dysregulation, particularly through aberrant neurotransmitter release and subtle GM alterations [3]. The microbiome-gut-brain axis (MGBA) represents a bidirectional communication network between the gastrointestinal tract and central nervous system, mediated by GM through mechanisms such as immune activation and vagal nerve signaling. Consequently, balanced GM composition may improve sleep via MGBA modulation [4].
Probiotics, defined as live microorganisms conferring health benefits when
adequately dosed, have been shown to enhance sleep quality under stress and
ameliorate sleep-related memory dysfunction and cognitive impairment [5, 6, 7, 8].
Supplementation with
This study investigated the therapeutic mechanism of Lactobacillus reuteri E9 (E9) for insomnia using a zebrafish model. E9 was selected based on its documented role in neurotransmitter metabolism. Our findings demonstrate that E9 upregulates neurotransmitters including glycine, serine, taurine, and GABA. Furthermore, E9 exerted sedative effects in pentylenetetrazol (PTZ)-induced insomnia by reducing seizure activity and upregulating GABA receptor and melatonin receptor (Mtnr1aa) gene expression in zebrafish.
Optimal Growth Assessment: To establish the optimal incubation parameters for Lactobacillus reuteri E9 (deposited at the China General Microbiological Culture Collection Center under accession number CGMCC No. 21768), the strain was cultured aerobically at 37 °C for 48 hours. Colony morphology on agar plates was then evaluated using the streak plate technique. This analysis documented key characteristics of individual colonies, including shape, dimensions, surface texture, margin appearance, opacity, pigmentation, and other morphological traits.
Origin and Molecular Identification: The E9 isolate originated from traditional fermented vegetables (pickles) sourced in Anhui Province, China. Following repeated purification (involving more than three sequential single-colony isolation streaks), the purified culture was subjected to 16S rDNA sequencing for species-level identification. Amplification employed universal primers 27F (5′-AGAGTTTGATCCTGGCTCAG-3′) and 1492R (5′-GGTTACCTTGTTACGACTT-3′). Sanger sequencing services were provided by Sangon Biotech (Shanghai, China). The resulting 16S rDNA amplicon sequence was subsequently aligned against the GenBank nucleotide database utilizing the BLAST algorithm (Basic Local Alignment Search Tool).
The API 50CHL system (BioMérieux, Lyons, France) employs a standardized panel comprising 49 microtubes containing fermentable carbohydrates, designed for identifying Lactobacillus and related bacteria [10]. Each well was inoculated with a standardized suspension of the test organism. During incubation, acid production from metabolized carbohydrates lowers the pH, triggering a chromogenic shift in the pH indicator. This acidification profile constitutes the strain’s biochemical fingerprint, used for identification or typing.
Result Interpretation: Test strips were evaluated after 24 and 48 hours of incubation. A color change to yellow in any well indicates substrate acidification, recorded as a positive reaction (+), as the phenol red indicator turns yellow at acidic pH. Similarly, a transition from red to black in the esculin control well (tube 25) signifies a positive control reaction. Wells showing no color change were recorded as negative (–).
Biochemical profiles were subsequently analyzed using the Apiweb™ software (Google LLC, Mountain View, CA, USA). This platform queries the reference database, generating identification results that include the species designation. Identification confidence is reflected by two parameters: the % ID value (higher percentages indicate closer matches) and the T-index (values approaching 1.0 represent superior identification quality.
The E9 strain was isolated and cultured under standard laboratory conditions. Cultures were maintained on MRS agar plates and incubated at 37 °C for 24 hours. For liquid cultures, E9 was grown in nutrient broth (Haibo Biotechnology Co., Ltd., Qingdao, China) with shaking at 200 rpm at 37 °C. A panel of antimicrobial agents was selected for testing, including: Penicillin (PEN), Ampicillin (AM), Meropenem (MP), Vancomycin (VA), Erythromycin (EM), Clindamycin (CM), Linezolid (LZ). These antibiotics were all purchased from Sangon Biotech (Shanghai) Co., Ltd. (Shanghai, China). Stock solutions of these antibiotics were prepared according to the manufacturer’s instructions and stored at –20 °C until use.
Antibiotic susceptibility profiles were assessed using the standardized Kirby-Bauer disk diffusion method (KB method). Strains were categorized as either susceptible (S) or resistant (R) based on inhibition zone measurements. Interpretation adhered to National Committee for Clinical Laboratory Standards (NCCLS) guidelines for non-fastidious bacteria, whereby bacterial sensitivity correlates directly with the diameter of the growth inhibition zone surrounding each antibiotic disk. Specifically, larger zones indicate greater susceptibility. Interpretive criteria varied for different antibiotic-impregnated disks (BioMérieux, France), as zone diameter thresholds defining susceptibility differ between antimicrobial agents.
A single colony of E9 was picked from the nutrient agar plate and inoculated
into 5 mL of nutrient broth. The culture was incubated overnight at 37
°C with shaking. The overnight culture was then diluted to an optical
density (OD) of 0.1 at 600 nm, corresponding to approximately 1
Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) Conditions:
Chromatographic separation employed an ACE Excel2C-18PFP analytical column (100
Mass spectrometric detection utilized Heated Electrospray Ionization (HESI) under optimized conditions: spray voltage (3.5 kV), capillary temperature (300 °C), sheath gas flow (50 arb), auxiliary gas flow (10 arb), S-lens RF level (40), and S-lens voltage (25 V). Full scans were acquired at 70,000 resolution, with MS/MS spectra collected at 17,500 resolution.
Data Processing & Metabolite Identification: Raw data files (.d) were converted to .abf format using AbfConverter. Subsequent data processing—including peak detection, alignment, and annotation—was performed in MS-DIAL (v4.90). Compound identification leveraged MS1/MS2 spectral matching against public repositories (MASSBANK, METLIN).
Peak Filtering & Quantification: Low-quality features (detection frequency
Peak stability was assessed via QC sample coefficient of variation (CV = RSD).
Features with CV
Adult zebrafish were maintained in a controlled environment with a 14/10 hour light/dark cycle and fed twice daily. Anxiety was induced by exposing zebrafish to a 7.5 mM solution of pentylenetetrazol (PTZ) (Aladdin, Shanghai, China) for 20 minutes. Zebrafish were treated with E9 by adding the bacterial culture supernatant to the tank water at a final concentration of 1% (v/v) for 1 hour before PTZ exposure. After PTZ exposure, zebrafish behavior was recorded for 5 mins using a video tracking system. Key parameters included time spent in the top half of the tank, distance traveled, and freezing behavior. Zebrafish were stained with a neural activity marker (e.g., c-Fos) and imaged using a fluorescent microscope (Minghui NIB950-FL, Guangzhou, China) to assess neural activity. The definition and quantification of areas based on Green fluorescent protein-Calmodulin-M13 Peptide (GCaMP) signal correlation with neuronal activity, or areas exhibiting enhanced calcium signaling, can be found in previously reported literature [12].
Experimental equipment utilized in zebrafish studies including Intelligent Breeding Management Suite (ZIBS), Microfluidics AutoLoader System (ZMFAL-100), Behavioral Imaging Analysis System (BH-100), Physiological Imaging Analysis System (PY-100) and Neuroimaging Analysis System (NS-100). All equipment was obtained from Guangdong Longseek Test Co., Ltd. (Guangzhou, China).
Normally developed 5 dpf AB zebrafish were randomly selected and placed in a
6-well cell plate. The experiment included the following groups (24 zebrafish per
group): control group, model group (PTZ), positive group (GABA), GABA+NCS-382
group, E9 group, and E9+NCS-382 group. The control and model groups were treated
with E3 water, while the positive group received 500 µM GABA solution. The
GABA+NCS-382 group was administered a mixture of 500 µM GABA and 10
µM NCS-382. The E9 group was treated with 1
Zebrafish Maintenance: Adult zebrafish were housed under standard laboratory
conditions with a 14/10 hour light/dark cycle. Zebrafish were treated with GABA
(positive group, 500 µM) or E9 with different concentration (1
All data analyses were conducted in SPSS 25.0 (IBM Corp., Chicago, IL, USA).
Results are presented as means
Lactobacillus reuteri E9, isolated from traditional Anhui pickles, was cultured on MRS agar under anaerobic conditions at 37 °C. Colonies appeared white, spherical, smooth-surfaced, and entire-margined (Supplementary Fig. 1). 16S rRNA gene sequencing revealed 99.72% homology with Limosilactobacillus reuteri JCM 1112T (AP007281), with phylogenetic analysis presented in Supplementary Fig. 2.
Biochemical characterization of E9 revealed the production of D-galactose,
sucrose, maltose, D-ribose, D-glucose, arginine, D-maltose, and L-arabinose
following fermentation (Supplementary Table 1). Concurrently, enzyme
activity assays detected alanine-phenylalanine-proline arylamidase, urease,
leucine arylamidase,
Antimicrobial susceptibility testing of E9 revealed sensitivity to penicillin (PEN), ampicillin (AM), meropenem (MP), erythromycin (EM), clindamycin (CM), and linezolid (LZ), but resistance to vancomycin (VA) (Table 1; Supplementary Fig. 3). These findings support the potential of E9 for further development and utilization.
| Antibacterial drug | Minimum Inhibitory Concentration (µg/mL) | Drug sensitivity |
| Penicillin (PEN) | 1.5 | Sensitiveness |
| Ampicillin (AM) | 0.38 | Sensitiveness |
| Meropenem (MP) | 0.047 | Sensitiveness |
| Vancomycin (VA) | Resistance | |
| Erythromycin (EM) | 0.25 | Sensitiveness |
| Clindamycin (CM) | 0.047 | Sensitiveness |
| Linezolid (LZ) | 1.0 | Sensitiveness |
Zebrafish were administered E9 for 7, 14, or 21 days commencing at 3 days
post-fertilization (dpf), and neurotransmitter levels were quantified [15, 16].
The entire fish was homogenized and processed to obtain samples for untargeted
metabolomic analysis [17]. Compared to the control group, concentrations of the
inhibitory neurotransmitters GABA, taurine, glycine, and serine were
significantly elevated (p
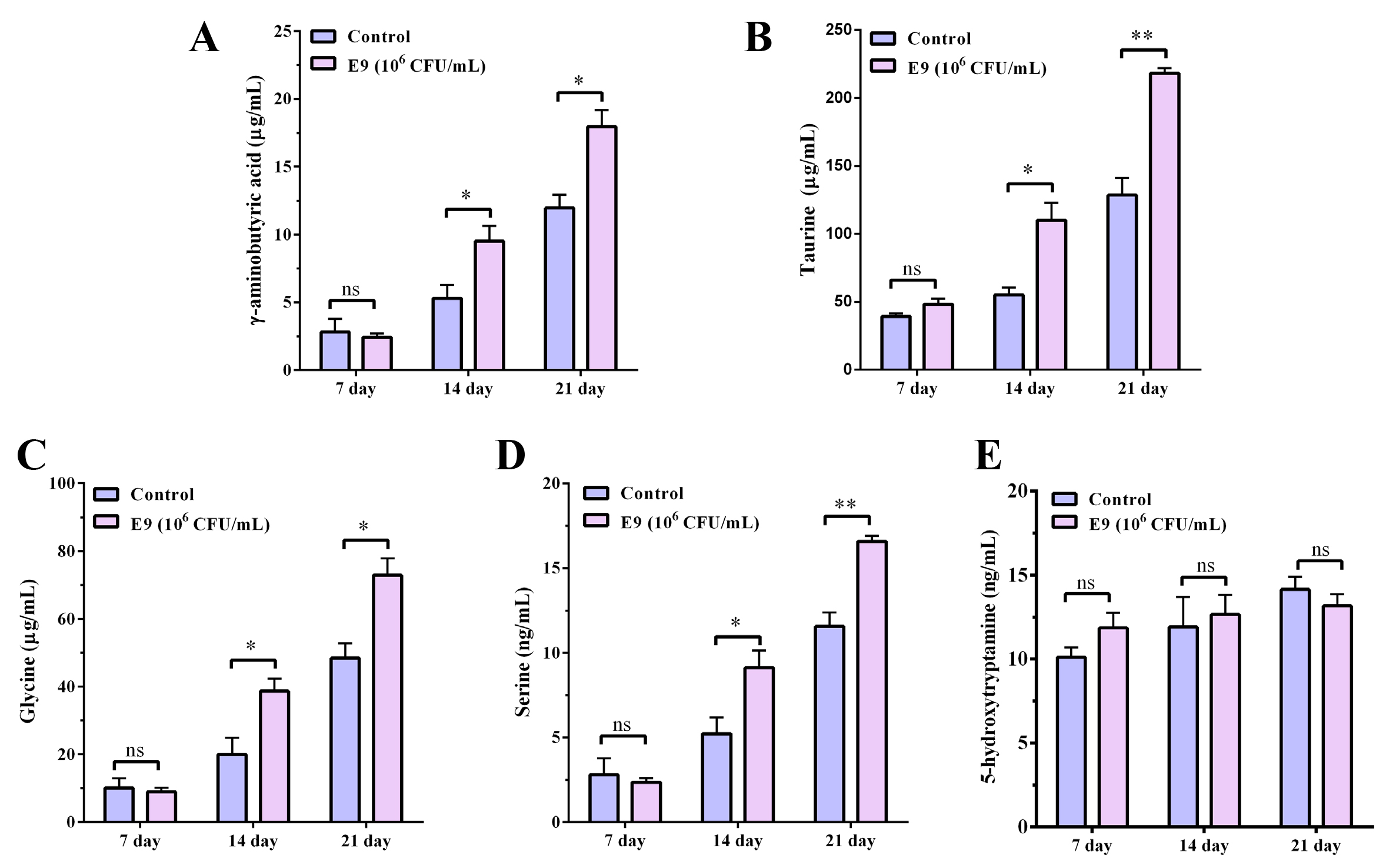 Fig. 1.
Fig. 1.
Effects of Lactobacillus reuteri E9 on
inhibitory neurotransmitter levels in zebrafish. (A)
In this experiment, zebrafish were immobilized in a small microfluidic chip, allowing them to survive while maintaining a certain degree of mobility [20]. A microscope was focused on a single zebrafish to continuously capture images, which were then processed and superimposed by a computer to generate the heatmap. The color gradient from blue to red indicates an increasing number of superimposed images, reflecting higher zebrafish activity. By quantifying the tail swing speed and amplitude of the zebrafish, Average tail velocity and average range of motion angle were obtained, respectively. Figures illustrate the tail swing speed and amplitude in the control group, model group, positive control group (GABA), and three experimental groups treated with different concentrations of E9.
As shown in Fig. 2A, the green and red areas represent the tail movement
trajectory coverage for zebrafish. Compared to the normal group, zebrafish in the
PTZ-induced model group exhibited a significantly larger tail movement area,
indicating increased locomotor activity. Concurrently, the model group showed
significantly elevated tail movement angular velocity and amplitude (p
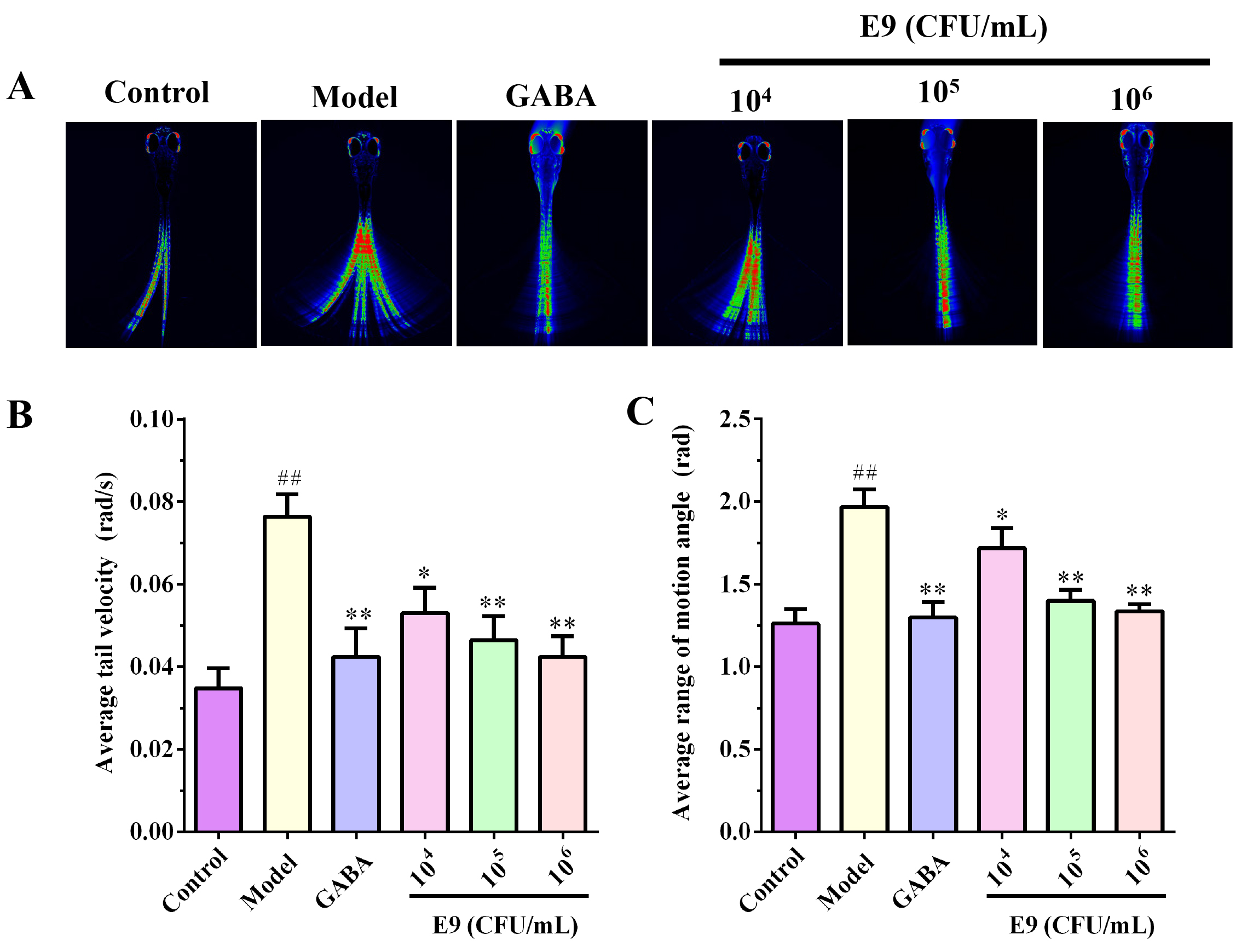 Fig. 2.
Fig. 2.
Behavioral analysis of PTZ-induced anxiety in zebrafish treated
with Lactobacillus reuteri E9. (A) Representative tail movement
trajectories. (B) Average tail swing angular velocity and (C) average tail swing
angle of zebrafish in control, PTZ-induced model, GABA-treated, and E9-treated
groups. Data are mean
Similar to GABA, zebrafish treated with E9 exhibited a reduced tail movement
area compared to the model group (Fig. 2A), indicating attenuated locomotor
activity in the PTZ-induced insomnia model. Furthermore, E9 administration
significantly decreased tail movement angular velocity and amplitude relative to
the model group (p
Tg(Elavl3:H2B-GCaMP6f) zebrafish were utilized to assess the effects of E9 on neuronal activity [21, 22]. Lin et al. [12] present an in vivo drug screening platform integrating high-throughput brain-wide activity mapping with machine learning-based predictive analytics. This approach leverages calcium-sensitive fluorophore fluorescence as a well-established neuronal activity indicator. To enable large-scale investigation of central nervous system (CNS) physiology, the team engineered an autonomous system for parallel imaging of awake, non-anesthetized zebrafish larvae. The system utilizes a hydrodynamically operated microfluidic chip capable of trapping, positioning, and orienting multiple larvae simultaneously for brain-wide neuronal recordings [12].
Fig. 3 depicts brain regions exhibiting baseline (blue) and elevated (yellow) fluorescence, representing neuronal discharge. Compared to the normal group, PTZ-exposed zebrafish showed significantly increased neuronal activation area, confirming PTZ-induced CNS excitation. The GABA treatment group exhibited markedly reduced activation area versus the model group, demonstrating GABA’s inhibition of PTZ-induced neuronal discharge and its sedative-hypnotic effect. Similarly, E9 administration significantly suppressed PTZ-induced neuronal activation area relative to the model group. This indicates that E9 attenuates pathological neuronal discharge, consistent with sedative-hypnotic activity.
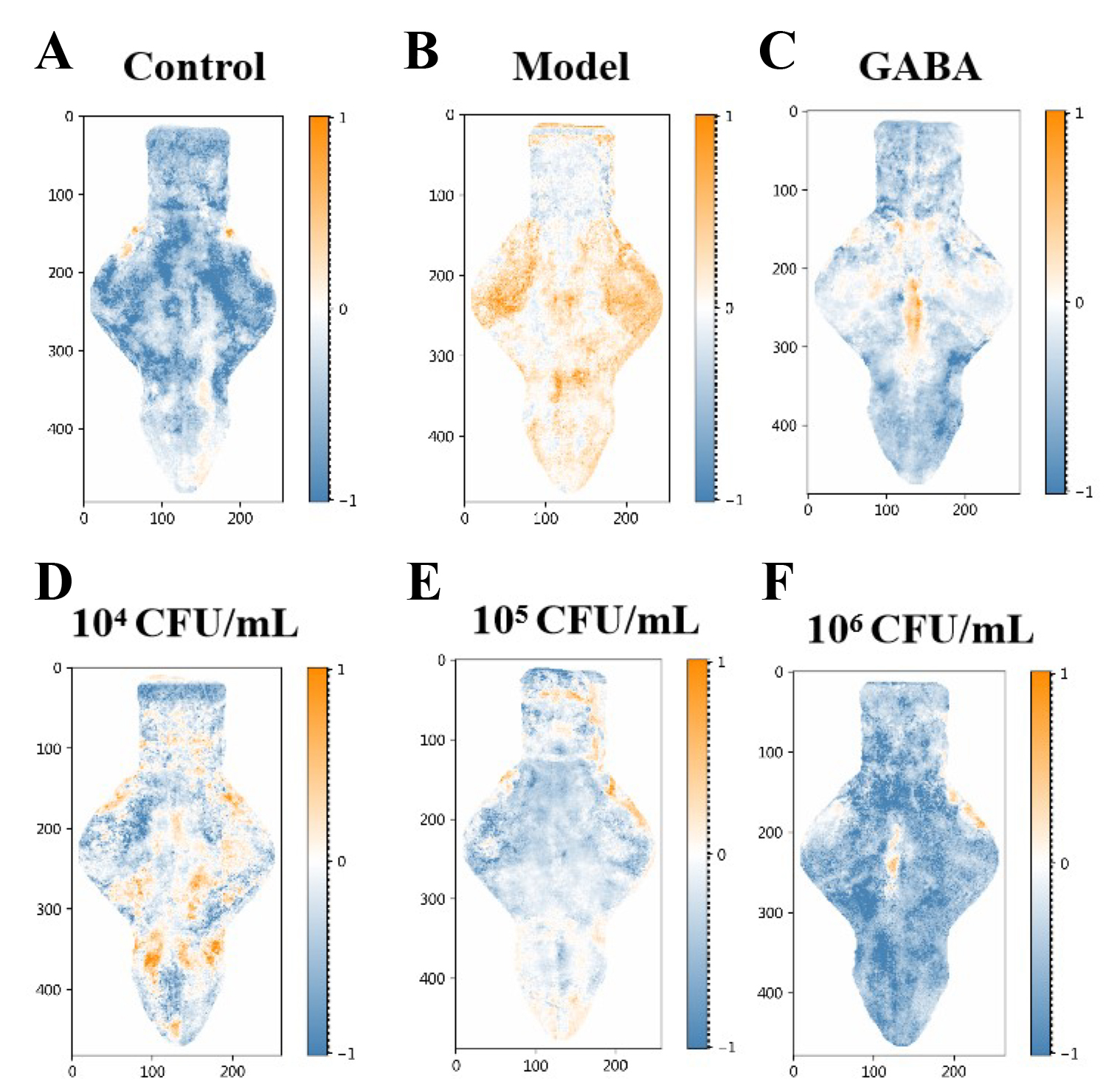 Fig. 3.
Fig. 3.
Lactobacillus reuteri E9 suppresses PTZ-induced
neuronal hyperactivity in zebrafish brains. Zebrafish in (A) normal (control
group), (B) PTZ-induced model (model group), (C) GABA-treated (positive group),
and E9-ttreated with different concentrations (expriment groups): (D) 1
Analysis of sleep-related pathway gene expression provides RNA-level assessment
of sedative-hypnotic efficacy [23, 24, 25]. As shown in Fig. 4: PTZ-exposed zebrafish
exhibited significantly downregulated expression of the GABA receptor gene and
melatonin receptor gene (Mtnr1aa) versus the normal group (p
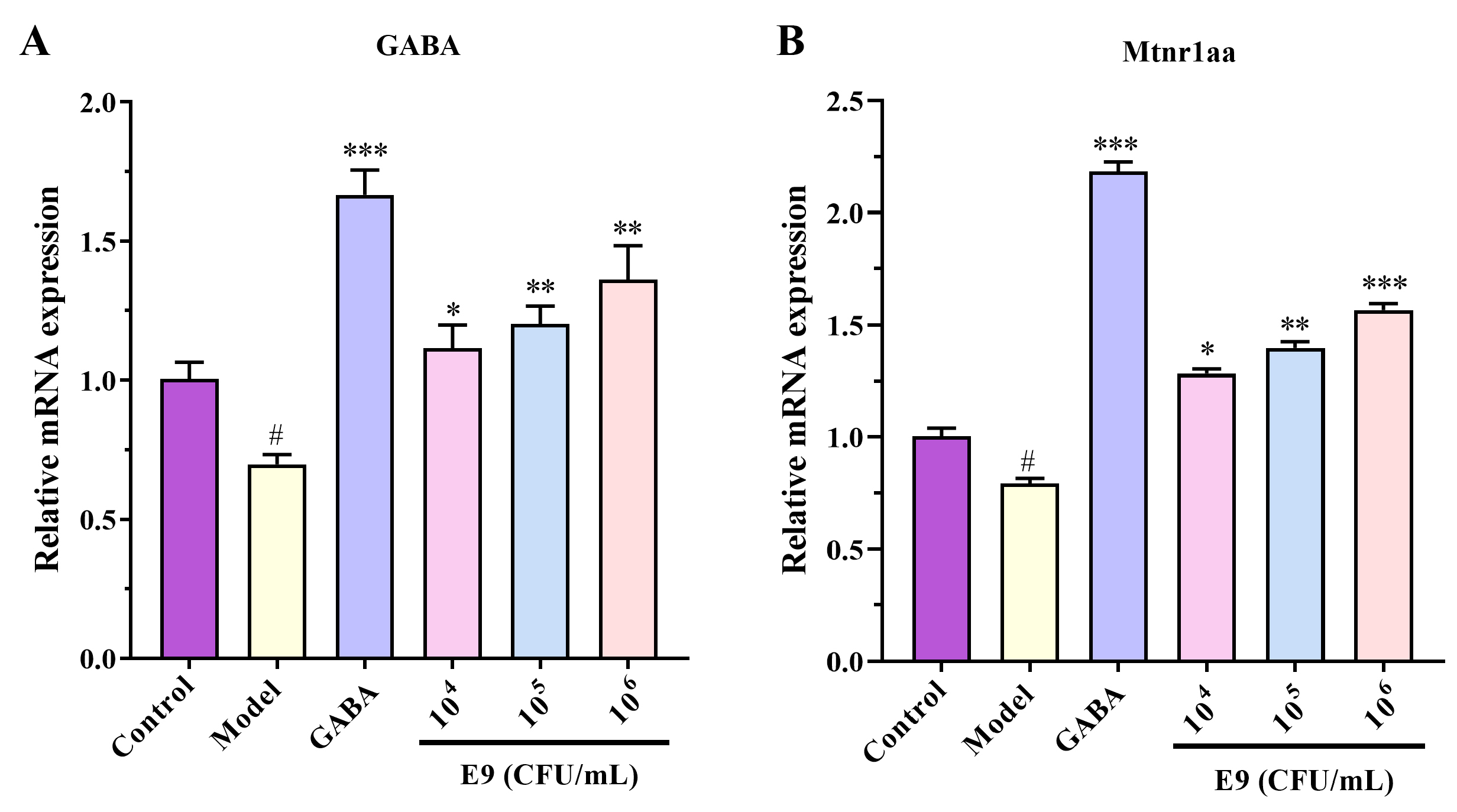 Fig. 4.
Fig. 4.
Lactobacillus reuteri E9 upregulates GABA and melatonin
receptor gene expression in zebrafish. Relative mRNA levels of (A) GABA receptor
and (B) melatonin receptor 1aa (Mtnr1aa) in zebrafish brains. Data are normalized
to
As shown in Fig. 5. Compared with the model group, both GABA and E9
significantly reduced the total swimming distance and average swimming speed of
zebrafish (p
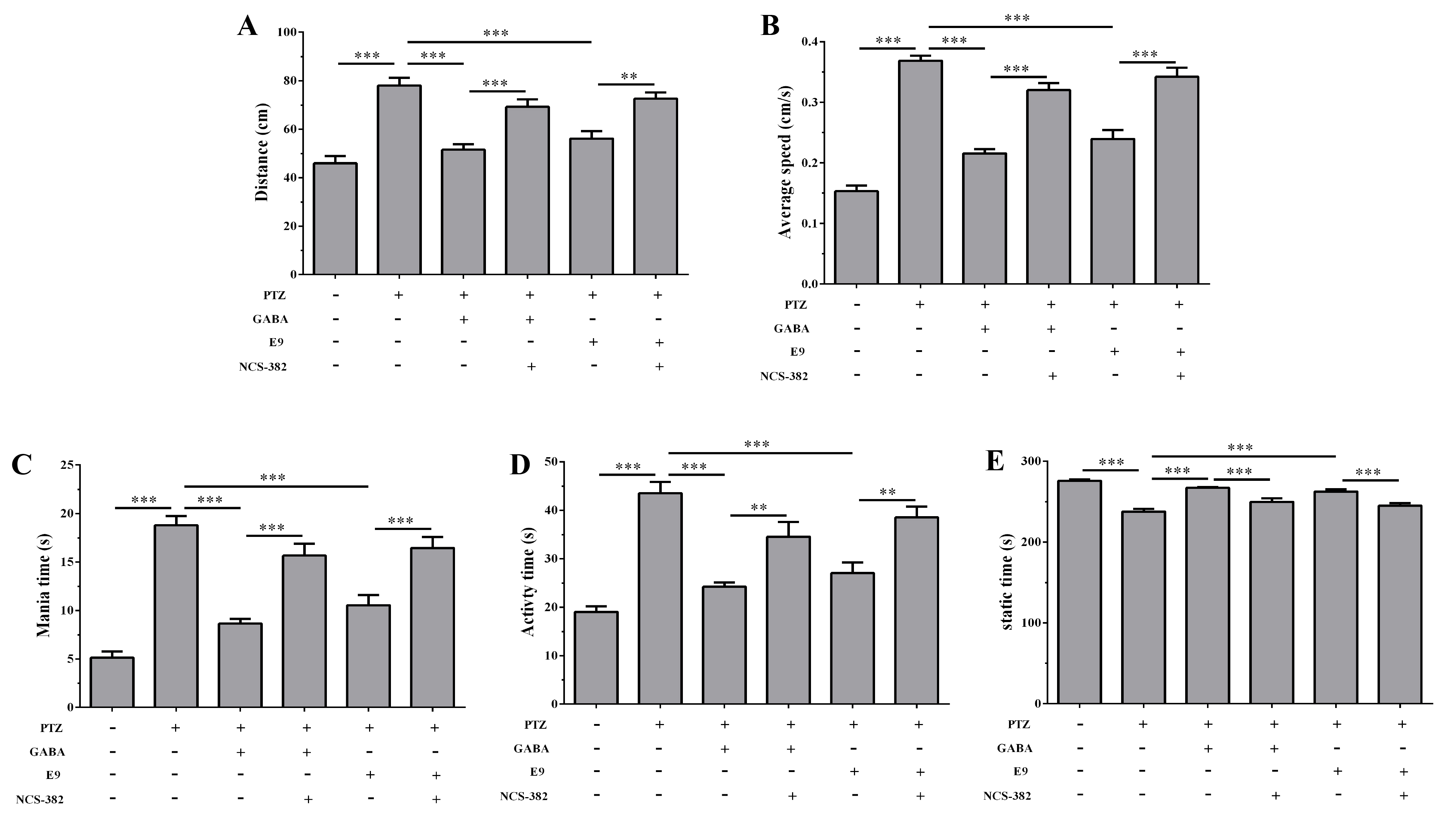 Fig. 5.
Fig. 5.
Behavioral analysis of the inhibitory effect of GABA receptor
antagonist NCS-382 on the anxiolytic activity of E9 in zebrafish with PTZ-induced
anxiety. (A) Total swimming distance. (B) Average swimming speed. (C) Mania
time. (D) Activity time. (E) Static time(s). Data are presented as mean
The E9 strain demonstrated superior antimicrobial susceptibility profiles, indicating robust potential against diverse pathogens. This high efficacy likely stems from strain-specific bioactive metabolites that require further isolation and characterization [26, 27]. Probiotic has significant pathogen-inhibiting capacity positions it as a promising candidate for augmenting antimicrobial strategies amid rising antibiotic resistance. In PTZ-induced zebrafish anxiety models, E9 significantly ameliorated anxiety-like behaviors, as evidenced by improved behavioral trajectories and reduced neurological hyperactivity. These findings strongly suggest E9 confers neurologically mediated anxiolytic effects, supporting its therapeutic potential for anxiety disorders.
Zebrafish were selected as the model organism based on their high genetic homology to humans and well-documented utility in neurobehavioral research [28, 29]. The observed anxiolytic effects demonstrate translational relevance for developing novel human anxiety therapeutics, though validation in mammalian models and subsequent clinical trials remains essential [30]. qPCR analysis elucidated molecular mechanisms underlying probiotic efficacy against severe insomnia, revealing modulation of sleep- and anxiety-regulatory gene networks. These findings indicate E9 likely mediates its effects through pathways governing neurotransmitter dynamics, receptor sensitization, and neuroinflammatory processes.
The specific genes and pathways identified represent promising targets for elucidating probiotic therapeutic mechanisms. And defining precise molecular interactions will be essential for developing targeted therapies leveraging probiotic benefits [31, 32]. Further characterization of E9 metabolite-pathway crosstalk may yield novel therapeutic compounds. While current findings are promising, key areas require investigation: Isolation and characterization of probiotic bioactive components responsible for antimicrobial/anxiolytic effects to enable standardized formulation development. Besides, long-term safety and efficacy assessment for chronic administration, including potential side effects and drug interactions. What’s more, Validation in mammalian models to advance toward clinical translation and exploration of therapeutic potential for other neuropsychiatric conditions (e.g., depression, PTSD, neurodegenerative diseases) given probiotic neuromodulatory properties [33, 34].
In summary, E9 holds considerable promise as a dual-function agent for antimicrobial interventions and anxiety behavior modulation. The integrated mechanistic and behavioral evidence presented establishes a robust foundation for translational development. Through continued elucidation of its mode of action and expanded therapeutic exploration, E9 emerges as a compelling candidate for addressing both infectious disease challenges and neuropsychiatric disorders.
In summary, Lactobacillus reuteri E9 demonstrates dual therapeutic potential as both an antimicrobial agent and a neuromodulator for sleep disorders. Our findings establish that E9 produces inhibitory neurotransmitters (GABA, glycine, serine, taurine) and upregulates GABA/Mtnr1aa receptor expression in zebrafish, reversing PTZ-induced insomnia. It significantly suppresses neuronal hyperactivity and ameliorates anxiety-like behaviors in PTZ model. The strain exhibits broad-spectrum antibiotic susceptibility with intrinsic bioactive metabolites. These results validate E9’s mechanistic role in GABA pathway modulation via the microbiota-gut-brain axis, positioning it as a promising non-pharmaceutical intervention for sleep disorders. Future work should isolate active metabolites, validate efficacy in mammalian models, and explore clinical translation.
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Conceptualization, YJ, WZ; methodology, YJ, LG, and YL; validation, WZ; investigation, YJ, LG, HH, HC, TC, YL; resources, WZ; data curation, HH, HC and TC; writing—preparation of the original project, YJ, YL; writing—reviewing and editing, LG, YL and WZ; visualization, YL; supervision, YL, WZ. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All zebrafish procedures followed the 3R principles (Replacement, Reduction, Refinement) and complied with GB/T 35892-2018 ethical guidelines, under approval from the Laboratory Animal Welfare and Ethics Committee. All zebrafish experiments were approved and met the ethical standards of the Institutional Review Board of the Laboratory Animal Ethics Committee, Center of Human Microecology Engineering and Technology of Guangdong Province (approval number: IACUC MC 0925-01-2024).
We thank Zhao Zhang and Kangdi Zheng (Center of Human Microecology Engineering and Technology of Guangdong Province, Guangdong Longsee Biomedical Corporation, Guangzhou, China) for statistical consultation and technical support provided by the Longseek high-throughput zebrafish screening platform for drug and probiotic evaluation. Also we acknowledge Dr. Chongxi Yang from La Trobe University (Australia) for language polishing.
This study was funded by 2023 Chizhou City major science and technology special project.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL39587.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





