1 Nephrology Department, Hangzhou Hospital of Traditional Chinese Medicine, 310007 Hangzhou, Zhejiang, China
2 Hangzhou Clinical College, Zhejiang Chinese Medical University, 310053 Hangzhou, Zhejiang, China
3 Department of Orthopedics, The Second Affiliated Hospital of Zhejiang Chinese Medical University, 310003 Hangzhou, Zhejiang, China
4 Department of Orthopedics, The First Affiliated Hospital, Zhejiang University, 310000 Hangzhou, Zhejiang, China
5 The First School of Clinical Medicine, Zhejiang Chinese Medical University, 310053 Hangzhou, Zhejiang, China
†These authors contributed equally.
Abstract
Diabetic nephropathy (DN) is a prevalent complication of diabetes, with current treatment options offering limited effectiveness, particularly in advanced stages. Human pluripotent stem cells (hPSCs), particularly induced PSCs (iPSCs), show promising potential in the treatment of DN due to their pluripotency, capacity for differentiation into kidney-specific cells, and suitability for personalized therapies. iPSC-based personalized approaches can effectively mitigate immune rejection, a common challenge with allogeneic transplants, thus enhancing therapeutic outcomes. Clustered regularly interspaced short palindromic repeats (CRISPR) gene editing further enhances the potential of hPSCs by enabling the precise correction of disease-associated genetic defects, increasing both the safety and efficacy of therapeutic cells. In addition to direct treatment, hPSCs have proven valuable in disease modeling and drug screening, particularly for identifying and validating disease-specific targets. Kidney organoids derived from hPSCs replicate key features of DN pathology, making them useful platforms for validating therapeutic targets and assessing drug efficacy. Comparatively, both hPSCs and mesenchymal SCs (MSCs) have shown promise in improving renal function in preclinical models, with hPSCs offering broader differentiation capacity. Integration with tissue engineering technologies, such as three-dimensional bioprinting and bioengineered scaffolds, expands the regenerative potential of hPSCs by supporting the formation of functional renal structures and enhancing in vivo integration and regenerative capacity. Despite current challenges, such as tumorigenicity, genomic instability, and limited direct research, advances in gene editing, differentiation protocols, and tissue engineering promise to address these barriers. Continued optimization of these approaches will likely lead to successful clinical applications of hPSCs, potentially revolutionizing treatment options for DN.
Keywords
- diabetic nephropathy
- human pluripotent stem cells
- induced pluripotent stem cells
- CRISPR gene editing
- kidney organoids
Diabetic nephropathy (DN) is a prevalent microvascular complication in diabetes and a primary cause of end-stage renal disease (ESRD). As diabetes rates rise, the incidence of DN has escalated globally alongside the rising rates of diabetes, severely impacting quality of life and straining healthcare systems [1]. DN progression is marked by proteinuria, glomerulosclerosis, and tubulointerstitial fibrosis, often necessitating dialysis or kidney transplantation [2]. Current treatments, including renin-angiotensin-aldosterone system (RAAS) blockers and glycemic control, offer limited efficacy, especially in advanced stages, highlighting the urgent need for new therapeutic strategies [3].
Emerging therapies have shown promise. Sodium-glucose cotransporter 2 (SGLT2) inhibitors, for example, have demonstrated the potential to slow DN progression, particularly in combination with RAAS blockers [4]. Additionally, kidney-targeted agents based on natural antioxidants, such as astragaloside IV, exhibit significant nephroprotective effects by reducing oxidative stress and fibrosis in vitro and in vivo [5]. Despite these advances, the complex pathophysiology, especially fibrosis and chronic inflammation, presents ongoing challenges. Further research is needed to understand DN mechanisms and develop effective therapies [6]. While new drug developments offer hope, DN remains challenging due to its multifaceted pathology involving chronic inflammation, oxidative stress, and fibrosis [7]. Current drugs provide some renal protection in the early stages but often fail as DN progresses, underscoring the importance of exploring alternative therapeutic options.
Human pluripotent stem cells (hPSCs), encompassing embryonic SCs (ESCs) and
induced PSCs (iPSCs), hold great potential in regenerative medicine. hPSCs
possess robust self-renewal capabilities and can differentiate into nearly all
cell types under specific conditions. While ESCs, derived from embryos, exhibit
unlimited differentiation potential [8], their use involves ethical concerns [9].
By contrast, iPSCs are generated by reprogramming adult somatic cells to a
pluripotent state, bypassing ethical issues and demonstrating similar
multi-lineage differentiation potential to ESCs [10]. Due to their ability to
differentiate into various cell types, such as renal and pancreatic
With advances in regenerative medicine, hPSCs show promising potential for
repairing damaged renal tissues. Differentiated renal cells derived from hPSCs
not only simulate kidney development in vitro but also hold potential
for cell-based replacement therapy for damaged kidney cells [2]. The study
indicate that hPSC-derived renal progenitor cells can form mature kidney
structures, such as glomeruli and tubules, both in vitro and in
vivo, and have demonstrated significant renal function improvement in animal
models [17]. Patient-specific iPSCs offer the advantage of reducing immune
rejection risks associated with allogeneic transplants, enabling personalized
therapy. This approach could be transformative for DN, allowing tailored
treatments through reprogramming patient-derived cells into renal or pancreatic
ESCs are derived from the inner cell mass of early embryos and exhibit high pluripotency, enabling differentiation into various cell types from all three germ layers, namely, neurons, muscle cells, and endothelial cells [9]. Their strong self-renewal and differentiation capabilities make them crucial in regenerative medicine and developmental biology. However, obtaining ESCs raises ethical concerns due to embryo destruction [20]. These concerns are tied to the moral status of embryos, leading many countries to impose strict regulations on ESCs research [21]. As a result, researchers have turned to iPSCs to avoid ethical issues. iPSCs are generated by reprogramming somatic cells (e.g., skin or blood cells) into a pluripotent state using specific transcription factors such as octamer-binding transcription factor 4, SRY-Box Transcription Factor 2, Krüppel-like factor 4 [22]. This process eliminates the need for embryo destruction, addressing ethical dilemmas associated with ESCs [23]. iPSCs allow for the personalized reprogramming of patient-derived cells, facilitating tailored therapies [12]. Their development not only resolves ethical concerns but also enhances precision medicine.
The iPSC reprogramming technique represents a significant advancement, enabling somatic cells to revert to an embryonic-like state through specific transcription factors that activate pluripotency genes [24]. Recent advancements, such as non-integrating viral vectors and mRNA reprogramming, improve the safety of iPSCs by reducing genetic mutation risks [25]. iPSCs provide several advantages; for example, they bypass ethical controversies linked to ESCs and can be derived from the patient’s own cells, minimizing the risk of immune rejection post-transplantation. This personalized source enhances the feasibility of tailored treatments in regenerative medicine [26]. Furthermore, iPSCs can be utilized for disease modeling, drug screening, and gene editing, offering extensive application potential in disease research [27].
hPSCs possess the ability to differentiate into all three germ layers—ectoderm, mesoderm, and endoderm—each of which further develops into diverse tissues and organs, including the nervous system, muscles, blood, and liver tissue [28]. Under carefully controlled culture conditions and with specific signaling molecules, hPSCs can be directed in vitro to become desired cell types, a capability that makes hPSCs invaluable for developmental biology research, disease modeling, and regenerative medicine [29].
To direct the differentiation of hPSCs in vitro, researchers commonly replicate developmental cues using specific media and signaling factors. For instance, the addition of growth factors and pathway modulators such as bone morphogenetic protein 4 (BMP4), Wingless-related integration site (Wnt), and fibroblast growth factor (FGF) guides hPSCs to differentiate into cardiomyocytes, neurons, or hepatocytes (Fig. 1) [30]. Enhanced differentiation efficiency can be achieved with three-dimensional (3D) culture systems or co-culture methods, resulting in organoids or cellular clusters that closely resemble the functions of native tissues [31]. For example, the development of 3D microfluidic co-culture systems, for example, has enabled the generation of functional liver cells from hPSCs [32].
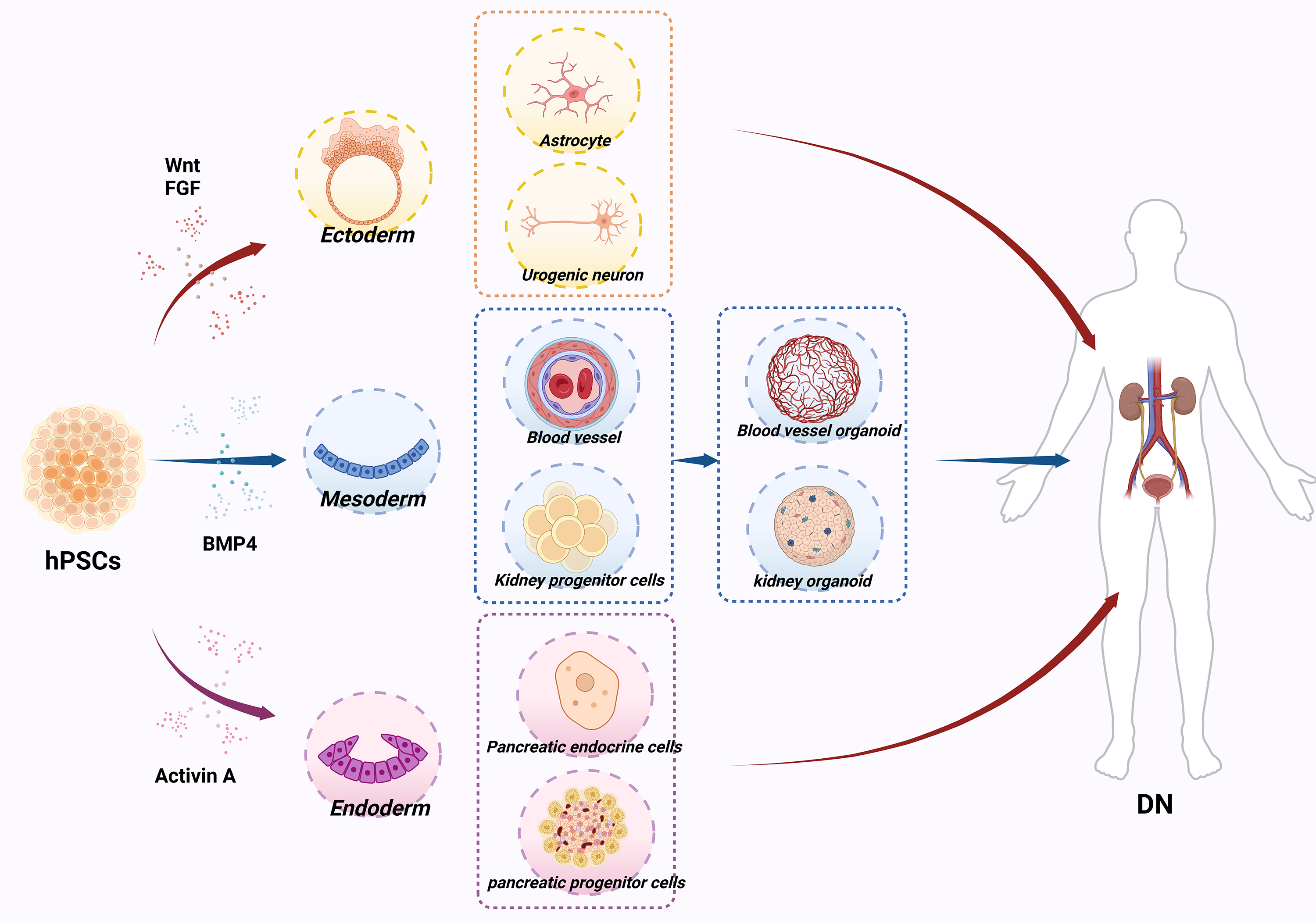 Fig. 1.
Fig. 1.
Differentiation potential of human pluripotent stem cells (hPSCs) and their application in diabetic nephropathy (DN) models. Adding growth factors and pathway modulators, such as bone morphogenetic protein 4 (BMP4), Wingless-related integration site (Wnt), and fibroblast growth factor (FGF) can guide hPSCs to differentiate into cardiomyocytes, neurons, or hepatocytes. (Created with BioRender.com).
In DN research, hPSCs have shown promise in multiple applications. They are
widely used to model the dysfunction of pancreatic
Significant advances have also been made in using hPSCs to generate kidney progenitor cells and kidney organoids, offering new approaches to DN treatment. Bantounas et al. [17] developed a method for deriving kidney progenitor cells from hPSCs and successfully transplanted these cells into immunodeficient mice, where they matured into functional kidney units. Bajaj et al. [36] expanded on this by creating a 3D kidney organoid platform from hPSCs, enabling the assessment of nephrotoxicity in drug development and opening new paths for kidney disease research. Vascular organoids derived from hPSCs have also been used to model diabetes-related vascular damage, providing valuable insight into potential therapeutic targets for diabetic complications [37].
In diabetes cell therapy and transplantation studies, hPSCs show remarkable potential. For example, Ghila et al. [38] successfully used pancreatic progenitor cells derived from hiPSCs to perform transplantation treatments in diabetic mouse models, demonstrating an approach that could lead to future therapeutic applications. Such strategies, particularly when enhanced by cell encapsulation techniques, offer improved cell survival post-transplant, representing a significant step forward in cell-based diabetes treatments.
Furthermore, hPSCs are crucial for studying complications of diabetes in vascular and neural systems. Morizane [37] used vascular organoids derived from hPSCs to model diabetes-induced vascular damage, identifying new therapeutic targets for vascular complications. In another study, Gorashi et al. [39] created a model of endothelial dysfunction in diabetes using patient-specific iPSCs, highlighting the role of hPSCs in vascular disease research. hPSC-derived urogenic neurons and astrocytes have also been applied to central nervous system models of diabetes-related complications, providing a novel platform for studying the neurological effects of diabetes [40].
As regenerative medicine and precision medicine continue to evolve, the applications of hPSCs have expanded considerably. Memon and Abdelalim [19] highlighted the crucial role of hPSCs in precision medicine in diabetes, noting that hPSC-derived cells can replicate disease states in vitro, offering an effective model for drug development and personalized treatment. Moreover, combining hPSCs with gene-editing technology allows researchers to introduce known pathogenic mutations into disease models, offering deeper insights into the specific contributions of these mutations and advancing research into diabetes mechanisms [41].
hPSCs have the remarkable ability to differentiate into a wide variety of cell types, a feature that plays a significant role in the study and treatment of DN [9]. By adding specific growth factors, such as Wnt and BMP4, hPSCs can be directed to differentiate into kidney progenitor cells, which can further mature into key kidney cells such as tubular epithelial cells and podocytes—cells essential for kidney function [17]. The successful generation of these progenitor cells in vitro lays a crucial foundation for exploring cell replacement therapies for DN [11].
Combining hPSCs with biomaterial scaffolds effectively mimics the natural kidney developmental process and significantly enhances the production efficiency of kidney progenitor cells [17]. This approach allows for better control of cell culture conditions, ultimately generating different kidney cell types more efficiently [9]. Furthermore, using modular differentiation systems enables the creation of both tubular and collecting duct cells, providing a robust basis for the reconstruction of complex kidney organ structures in vitro [11].
To generate specific kidney cell types, factors such as activin A and FGF9 can be added to the culture medium to successfully induce hPSCs into functional podocytes and tubular epithelial cells [17]. These differentiated cells exhibit similar functional characteristics to native kidney cells and can be used in DN models to facilitate tissue repair [13]. Additionally, combining two-dimensional (2D) and 3D culture systems have significantly increased the efficiency and maturity of differentiated hPSCs, resulting in kidney cells that more closely resemble their natural counterparts [9].
Kidney cells derived from hPSCs show immense potential in cell replacement therapies, particularly podocytes and tubular epithelial cells, which can be transplanted to replace damaged kidney tissues and restore kidney function partially [10]. In animal studies, transplanted podocytes demonstrated a significant protective effects on the kidneys, reducing proteinuria and inflammation, which are key symptoms of DN [42]. Furthermore, when these differentiated cells were transplanted into immunodeficient mice, they formed mature kidney structures, including glomeruli and tubules, effectively mitigating kidney damage [42].
As differentiation techniques continue to advance, hPSC-derived kidney cells hold promise for future clinical applications in DN, potentially offering a way to replace damaged kidney tissue and restore kidney function [6]. Researchers have also developed a new method using synthetic mRNA encoding transcription factors, which allows for the rapid and efficient differentiation of hPSCs into critical kidney cells such as tubular epithelial cells and podocytes, providing new avenues for kidney regenerative therapies [11].
The potential therapeutic application of hPSCs in DN extends beyond cell
replacement therapy and includes significant paracrine effects, particularly
through the secretion of exosomes that help protect kidney function. Exosomes
derived from hPSCs are rich in microRNAs (miRNAs), such as miR-16-5p, which can
inhibit vascular endothelial growth factor A (VEGFA), effectively protecting
podocytes from damage induced by high glucose conditions [2]. Moreover, hPSCs
secrete specific factors that inhibit transforming growth factor beta 1
(TGF-
In DN models, exosomes and paracrine factors secreted by hPSCs have shown
notable protective effects. These include mitigating kidney damage by regulating
inflammatory responses and significantly reducing fibrosis and inflammation
through modulation of the TGF-
Beyond their paracrine effects, hPSCs also exhibit strong immunomodulatory capabilities. By secreting exosomes and anti-inflammatory factors, hPSCs can reduce the infiltration of pro-inflammatory macrophages and regulate T-cell activity. The study has shown that hPSC-derived exosomes can decrease the levels of pro-inflammatory cytokines while enhancing the population of anti-inflammatory macrophages, significantly alleviating the chronic inflammation commonly seen in DN [45]. In addition, the paracrine actions of hPSCs can alter the immune microenvironment in the kidney, reducing the infiltration of inflammatory cells into kidney tissues and offering further protection to renal structures [18].
The antifibrotic effects of hPSCs in DN are primarily achieved through the
regulation of multiple signaling pathways, reducing excessive deposition of the
extracellular matrix (ECM) and thereby mitigating the progression of kidney
fibrosis. The study has shown that exosomes secreted by hPSCs contain miRNAs,
such as miR-16-5p, which can suppress the expression of VEGFA, thus helping to
reduce fibrosis in the kidney [2]. Additionally, hPSCs have been shown to
regulate the TGF-
hPSCs can also interfere with multiple fibrosis-related signaling pathways,
including TGF-
Another important mechanism of action for hPSCs is their ability to reduce extracellular matrix deposition. By decreasing the accumulation of key matrix components such as collagen and fibronectin, hPSCs are able to inhibit epithelial-mesenchymal transition in tubular epithelial cells as well as fibroblast transdifferentiation. These effects are largely mediated by exosomal miRNAs, such as miR-24-3p, and circular RNAs, such as circ_0080425, which collectively contribute to reducing the progression of kidney fibrosis [49, 50].
Kidney fibrosis is a critical step in the development of DN and a major contributor to ESRD. The antifibrotic properties of hPSCs offer promising opportunities to delay or even reverse the pathological progression of DN, positioning them as a potential therapeutic tool [51]. These attributes make hPSCs a highly promising candidate for DN treatment, offering a novel approach to addressing kidney fibrosis and providing hope for patients with advanced kidney disease (Fig. 2).
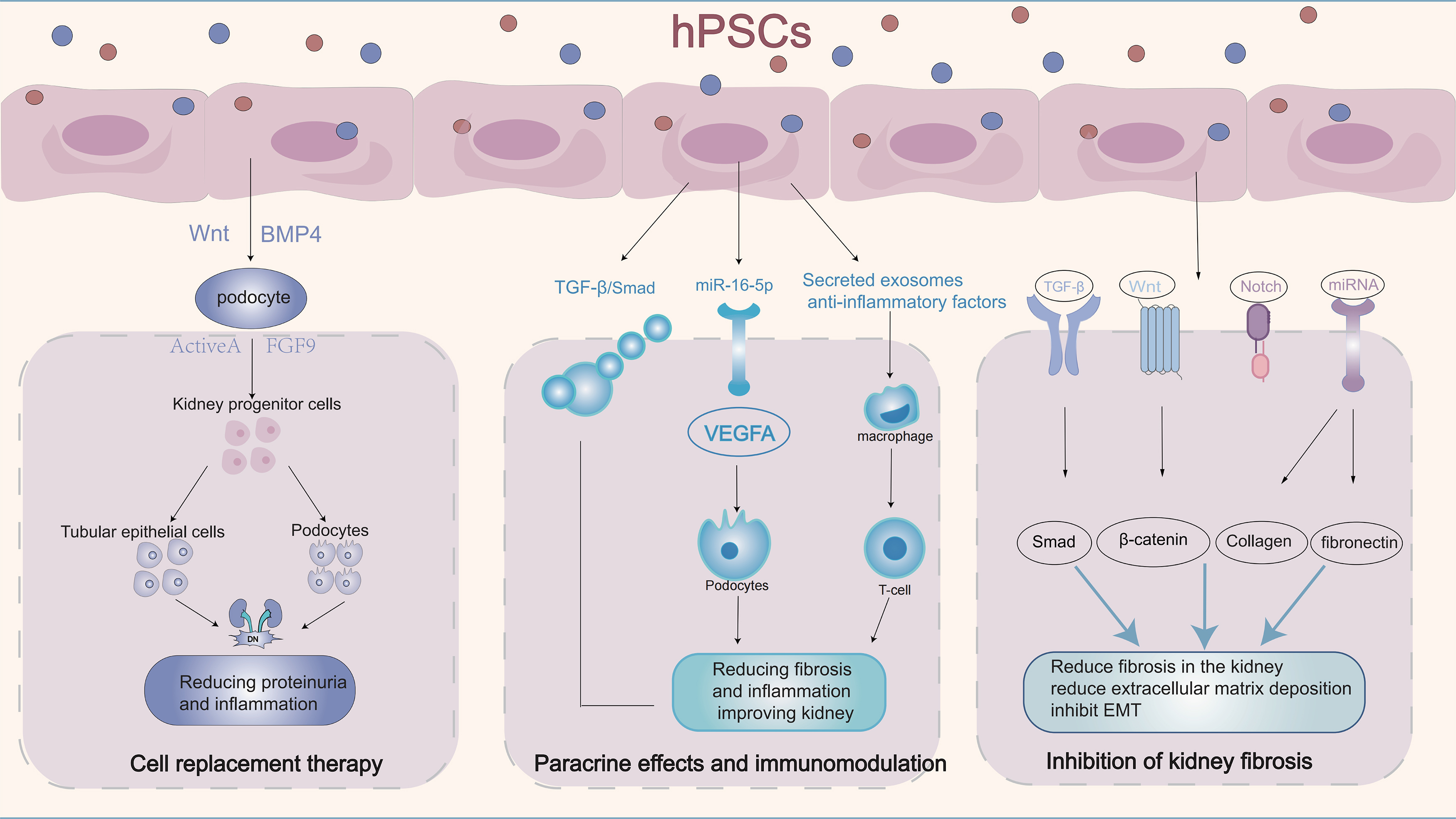 Fig. 2.
Fig. 2.
Application of hPSCs in DN regeneration. hPSCs can
differentiate into other cells by adding growth factors and pathway modulators,
secreting exosomes for paracrine and immunomodulatory effects, and interfering
with multiple fibrosis-related signaling pathways, achieving therapeutic effects
for DN. TGF-
hPSCs have demonstrated significant potential in DN research, particularly in
modeling the disease mechanisms and exploring novel treatment strategies. In one
study, Leite et al. [33] successfully used iPSCs to differentiate into
pancreatic endocrine cells, effectively modeling the immune response of patients
with type 1 diabetes. This model highlighted the specific attack of autoimmune
cells on
In terms of cell replacement therapy, Ghila et al. [38]. utilized
pancreatic progenitor cells derived from iPSCs for transplantation in a diabetic
mouse model, demonstrating improved insulin secretion and increased survival
rates of the transplanted cells, offering promising avenues for future cell-based
therapies. Similarly, Balboa et al. [34] reviewed the role of hPSCs in
modeling
In the development of disease models and personalized therapies, Gheibi et al. [35] generated insulin and glucose-responsive cells for disease modeling and diabetes treatment, demonstrating the ability to recreate disease states in vitro and providing an effective platform for drug development and individualized treatment. Liu et al. [40] utilized urine-derived iPSCs from diabetic patients to generate neurons, astrocytes, and microvascular endothelial cells, offering new tools for investigating diabetes-related central nervous system disorders and expanding possibilities for personalized treatment. Concurrently, Gorashi et al. [39] developed a model for diabetic endothelial dysfunction using patient-specific iPSCs, which is valuable for studying vascular complications of diabetes and screening potential therapeutic drugs.
Furthermore, Bajaj et al. [36] developed an hPSC-based kidney model to evaluate drug nephrotoxicity, providing a novel research avenue for assessing the safety of DN-related drugs. Qi et al. [41] applied gene-editing techniques to introduce specific point mutations into hPSCs, creating disease model cells that are instrumental in understanding DN pathology and developing personalized treatment strategies. Memon and Abdelalim [19] discussed the potential of using hPSCs to achieve precision medicine in diabetes research, emphasizing their role in creating personalized disease models and developing therapeutic approaches, thus paving the way for individualized diabetes treatment.
Currently, hPSCs in DN research are mainly in the experimental and preclinical stages, and have not yet been widely applied in clinical practice [2, 53]. By contrast, mesenchymal SCs (MSCs) have already made their way into clinical use. For instance, a randomized, placebo-controlled, multicenter study evaluated the efficacy of allogeneic bone marrow (BM)-derived mesenchymal precursor cells in patients with DN, demonstrating good safety and tolerability along with positive effects on renal function in some patients [54]. These findings suggest that MSCs have shown initial clinical utility in DN treatment, laying the groundwork for further large-scale clinical trials.
MSCs, with their multipotent differentiation capability and immunomodulatory
properties, have been extensively studied for DN treatment. They can be derived
from a variety of tissues, including bone marrow, adipose tissue, umbilical cord,
placenta, dental pulp, and amniotic fluid, with each source offering unique
advantages in terms of efficacy, mechanism, and therapeutic potential (Table 1,
Ref.
[42, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81]).
BM-derived MSCs (BM-MSCs) were the first to be discovered and widely applied, and
are known for their strong differentiation potential and immunomodulatory
effects. Zhou et al. [55] found that intraventricular administration of
BM-MSCs in DN rat models resulted in the cells colonizing the kidney,
significantly reducing the proteinuria and renal mass index. Lang and Dai [56]
demonstrated that BM-MSCs could inhibit renal fibrosis through suppression of the
TGF-
| Research (author and year) | Mechanism/Principle | Research results | |
| Bone marrow-derived MSCs, BM-MSCs | |||
| Zhou et al. (2009) [55] | BM-MSCs colonization in kidneys without sustained proliferation | Reduction in urinary protein and renal mass index | |
| Lang and Dai (2016) [56] | Inhibition of TGF- |
Reduction in 24-hour urinary protein, serum creatinine, and renal mass index | |
| Luznik et al. (2008) [57] | Promotion of pancreatic |
Lowered blood glucose, preventing DN onset | |
| Mousa et al. (2016) [70] | Regulation of metabolic markers, reducing inflammation | Improvements in renal, hepatic, and glucose levels | |
| He et al. (2020) [71] | Promote insulin-secreting cell regeneration, activating GSK-3 |
Improved renal function, reduced urinary protein | |
| Lin et al. (2020) [72] | Activation of GSK-3 |
Reduction in urinary protein and renal damage | |
| Akagun et al. (2017) [73] | Improvement of metabolic abnormalities in diabetes patients | BM-MSCs improve blood glucose and metabolic irregularities, aiding diabetes management | |
| Adipose tissue-derived MSCs, AD-MSCs | |||
| Takemura et al. (2020) [58] | Direct transplantation of AD-MSC cell sheets into kidney | Suppression of renal injury progression | |
| Alicka et al. (2019) [59] | Comparative study on AD-MSC function in diabetic vs. non-diabetic patients | Reduced proliferation, secretion, and mitochondrial function in diabetic AD-MSCs may affect therapeutic potential | |
| Timper et al. (2006) [74] | AD-MSCs differentiated into insulin-secreting cells | Potential for AD-MSCs as cell replacement therapy in diabetes | |
| Wang et al. (2018) [75] | AD-MSCs improve insulin sensitivity, reduce inflammatory responses | Lowered inflammatory markers, improved insulin resistance in diabetic models | |
| Ni et al. (2015) [76] | AD-MSCs reduce inflammation, improve metabolism | Improved glucose metabolism through inflammation reduction | |
| Habib et al. (2021) [77] | Synergistic effect of AD-MSCs combined with other therapies | Enhanced efficacy in diabetes treatment with combined therapies, indicating synergy | |
| Umbilical cord-derived MSCs, UC-MSCs | |||
| Park et al. (2012) [60] | Paracrine action reducing TGF- |
Renal protection in DN, reducing renal injury | |
| Montanucci et al. (2016) [61] | Restoration of Treg/Th17 balance, improving immune dysfunction | Microencapsulated UC-MSCs improve immune dysfunction in type 1 diabetes | |
| Chen et al. (2020) [62] | Inhibition of ASK1 and P38 MAPK pathways, reducing apoptosis | Enhanced renal function, reduced renal cell apoptosis | |
| Li et al. (2020) [42] | Inhibition of TGF- |
Reduction in fibrosis, improvement in DN pathology | |
| Guan et al. (2015) [78] | UC-MSCs differentiate into insulin-secreting cells, enhancing pancreatic function | Increased insulin secretion, lowered glucose in diabetes models | |
| Zang et al. (2023) [79] | UC-MSCs improve glucose metabolism, reduce insulin resistance | Reduced or discontinued insulin use in some type 2 diabetes patients post-transplantation | |
| Placenta-derived MSCs, PL-MSCs | |||
| Jiang et al. (2011) [63] | Intravenous PL-MSC infusion to assess safety and efficacy | Reduced insulin use, increased C-peptide levels, improved renal function | |
| Han et al. (2023) [64] | Activation of SIRT1-PGC-1 |
Alleviated podocyte damage in DN, showing potential in DN treatment | |
| Liang et al. (2017) [80] | Promotion of angiogenesis through pro-angiogenic and immune factors | Improved ischemia in diabetic rats; essential in treating diabetes complications | |
| Dental pulp-derived MSCs, DP-MSCs | |||
| Rao et al. (2019) [65] | Inhibition of AGEs-induced epithelial-mesenchymal transition, reducing renal fibrosis | Improvement in renal damage in DN models, protective effects | |
| Kanafi and Bhonde (2024) [66] | Differentiation into insulin-secreting cells, promoting pancreatic regeneration | Therapeutic potential in diabetes treatment | |
| Yamada et al. (2019) [81] | Pluripotency and self-renewal for systemic disease treatments | Broad application prospects in regenerative medicine; potential in DN | |
| Amniotic fluid-derived MSCs, AF-MSCs | |||
| Feng et al. (2018) [67] | SIRT3 regulates mitochondrial autophagy, enhancing AFSCs tolerance in high glucose | SIRT3-modified AFSCs improved glucose metabolism, reduced renal fibrosis and injury, showing therapeutic potential in DN | |
| Kim et al. (2012) [68] | Promotes angiogenesis and re-epithelialization, accelerating wound healing | Accelerated wound healing in diabetic NOD/SCID mice; strategy for chronic diabetic wound treatment | |
| Harrell et al. (2019) [69] | Highlighted AF-MSCs’ therapeutic potential in degenerative and inflammatory diseases | Emphasizing their differentiation capacity and immunomodulatory effects, especially in DN | |
MSCs, mesenchymal SCs; GSK-3
Adipose-derived MSCs (AD-MSCs) have gained attention due to their relatively easy acquisition and minimally invasive nature. Takemura et al. [58] found that directly transplanting AD-MSC sheets into the kidneys of DN rats effectively slowed renal damage progression. In a Phase 1 clinical trial, Purwati et al. [82] evaluated the effects of autologous AD-MSC transplantation in patients with type 2 diabetes and observed significant reductions in blood glucose levels. However, Alicka et al. [59] noted that diabetic patients had reduced proliferation capacity in their AD-MSCs, potentially impacting their therapeutic efficacy. Umbilical cord-derived MSCs (UC-MSCs) have strong proliferative capabilities and low immunogenicity. Park et al. [60] found that UC-MSCs exert nephroprotective effects in DN through paracrine signaling. Montanucci et al. [61] demonstrated that UC-MSCs could improve immune dysfunction in patients with type 1 diabetes by restoring the regulatory T cell/T helper 17 (Th17) cell balance. Chen et al. [62] found that UC-MSCs significantly improved renal function in DN models by inhibiting the apoptosis signal-regulating kinase 1 and p38 mitogen-activated protein kinase pathways.
Placenta-derived MSCs (PL-MSCs), known for their non-invasive acquisition and immunomodulatory and tissue repair capabilities, have also been studied for diabetes treatment. In a clinical trial, Jiang et al. [63] infused PL-MSCs into patients with type 2 diabetes, observing reduced insulin requirements and improved renal function. Furthermore, Han et al. [64] showed that PL-MSCs reduced podocyte injury in DN by activating the sirtuin 1 (SIRT1)/peroxisome proliferator-activated receptor gamma coactivator-1 alpha/mitochondrial transcription factor A signaling pathway. Dental pulp-derived MSCs (DP-MSCs) have also garnered attention due to their powerful regenerative potential. Rao et al. [65] demonstrated that SCs from human exfoliated deciduous teeth (SHED) significantly improved kidney damage in DN models. Kanafi and Bhonde [66] highlighted the potential of DP-MSCs to differentiate into insulin-secreting cells, suggesting promising therapeutic potential for pancreatic regeneration.
Amniotic fluid-derived MSCs (AF-MSCs) possess multi-lineage differentiation potential and low immunogenicity, offering promising clinical applications. Feng et al. [67] investigated the role of SIRT3 in AFSCs, showing that it regulated mitochondrial autophagy and enhanced AFSCs’ therapeutic efficacy in DN. Kim et al. [68] found that AF-MSCs promoted angiogenesis and re-epithelialization in diabetic mice, accelerating wound healing and further demonstrating their potential in treating diabetic complications.
iPSCs exhibit more significant advantages than MSCs in the treatment of DN, primarily due to their pluripotency, enhanced differentiation potential, immunomodulatory capacity, and suitability for personalized therapy.
iPSCs possess high pluripotency, enabling them to differentiate into various specialized cell types, which is particularly crucial for cell replacement therapy in DN. Osafune [53] emphasized that the pluripotency of iPSCs allows them to generate renal lineage cells, providing new possibilities for DN treatment. By contrast, MSCs have relatively limited differentiation potential and cannot generate specific renal cells. Thanaskody et al. [83] pointed out that iPSCs can be generated from adult somatic cells through genetic reprogramming without involving embryonic destruction, thus avoiding ethical controversies. Their ability for self-renewal and differentiation into multiple specialized cell types makes iPSCs a significant cell source in regenerative medicine, offering broader regenerative potential in DN treatment compared to MSCs.
iPSC-derived MSCs (iPSC-MSCs) exhibit significant advantages in immunomodulation and therapeutic efficacy. Cheng et al. [84] demonstrated that iPSC-MSCs combined with low-dose rapamycin can significantly prolong the survival time of transplanted islets, enhancing immunosuppressive effects. iPSC-MSCs display remarkable immunomodulatory functions by inhibiting the release of Th1 pro-inflammatory cytokines, thereby enhancing the ability to counteract diabetes-related immune responses. Similarly, Ozay et al. [85] noted that iPSC-MSCs have tri-lineage differentiation capacity similar to BM-derived MSCs and significantly extend graft survival time in transplantation models. This suggests that iPSC-MSCs may be an effective strategy for treating diabetic complications, including DN. Wang et al. [75] compared the therapeutic effects of iPSC-MSCs and adult MSCs, and in acute kidney injury. The results showed that iPSC-MSCs are comparable to adult MSCs and, in some cases, exhibit superior tissue repair capabilities and promote angiogenesis. Lee et al. [86] demonstrated that iPSC-MSCs can prevent the progression of DN by improving mitochondrial function and inhibiting inflammation, indicating their potential in regulating metabolic processes and providing therapeutic effects.
Another significant advantage of iPSCs is their ability to generate patient-specific cells, thereby avoiding immune rejection reactions. Päth et al. [87] emphasized that iPSCs can produce patient-specific renal cells, offering a more personalized treatment approach, which is especially important for chronic diseases like DN. Moreover, iPSCs hold substantial value in disease modeling and drug development. Liu and Tang [88] pointed out that iPSCs can not only be used to construct disease models but also directly treat kidney injuries by generating renal cells. Wu et al. [89] highlighted the potential of iPSCs in generating specific types of renal cells, providing possibilities for future personalized DN therapies. Additionally, Himeno et al. [90] found that iPSC-derived MSC-like cells can significantly improve DN, demonstrating their potential in treating diabetic complications. The ability of iPSCs to generate specific cell types for repairing damage underscores their multifunctionality compared to MSCs.
The differentiation process of iPSCs is complex, involving multiple stages of cell culture and induction. Compared to MSCs, the use of iPSCs demands a higher level of operational complexity, presenting technical bottlenecks and longer timelines for clinical translation. To improve the differentiation efficiency and cell maturity, optimizing culture systems is essential. Technologies such as 3D bioreactors and microfluidic chips allow for better simulation of the in vivo microenvironment, providing more physiologically relevant conditions that enhance the functionality of the derived cells for DN treatment [91]. Dynamic monitoring with single-cell RNA sequencing (scRNA-seq) allows real-time tracking of differentiation, while precise modulation of signaling pathways like Wnt, BMP, and Notch improves differentiation accuracy and iPSC therapeutic potential [92]. MSCs, on the other hand, offer a simpler therapeutic approach with rapid efficacy, particularly advantageous in the acute or early stages of DN, where their effects can be swiftly realized through intravenous infusion [43]. However, for iPSCs, optimizing cell delivery methods remains a challenge. Techniques such as renal artery injection, tail vein injection, and local tissue implantation have been explored to improve cell engraftment and ensure their long-term survival in the kidney [93]. Furthermore, the success of iPSC-based therapies depends on immune and inflammation modulation. Gene editing to reduce the expression of major histocompatibility complexes (MHC) or the use of immunosuppressive agents can minimize immune rejection and enhance integration with host tissues [94]. Finally, improving the renal microenvironment using biomaterials (e.g., hydrogels and scaffolds) along with local delivery of pro-angiogenic or anti-inflammatory factors can support the long-term functionality of transplanted cells [95].
The risk of tumorigenesis is a significant concern in the clinical application
of iPSCs. Han et al. [96] reported a case in which a diabetic patient
developed an immature teratoma following an injection of iPSC-derived
hPSCs also present a substantial risk of immune rejection, particularly when using allogeneic cells, which may necessitate prolonged immunosuppressive therapy. By contrast, MSCs exhibit inherently low immunogenicity, resulting in better compatibility for allogeneic transplantation and a reduced risk of immune rejection [100]. Pellegrini and Sordi [100] pointed out that despite the significant potential of iPSCs in regenerative medicine, their application is still hindered by challenges such as immunogenicity and tumorigenic risk. They suggested that the implementation of more rigorous differentiation and purification protocols is essential to enhance the safety of iPSC-based treatments.
The long-term efficacy of hPSCs in treating DN remains uncertain, necessitating further research and validation. By contrast, the application of MSCs in chronic kidney disease has been well documented, with numerous clinical studies demonstrating their effectiveness, particularly in enhancing renal function and slowing disease progression [101]. Liu and Tang [88], in their comprehensive review, highlighted both the potential and limitations of iPSCs in treating chronic conditions such as DN. They emphasized that, despite the promise shown by iPSCs in terms of differentiation potential and immunomodulatory capacity, issues related to tumorigenesis and immune rejection remain major obstacles that must be addressed before broader clinical application.
iPSCs show considerable promise for treating DN, but their clinical application is hindered by several significant challenges, including risks of tumorigenesis, immune rejection, low differentiation efficiency, difficulties in large-scale production, and issues with genetic and epigenetic stability. Additionally, the complex and time-consuming differentiation process of iPSCs presents obstacles, particularly in scenarios where rapid therapeutic intervention is required, making MSCs a more favorable option in such cases. To advance the use of iPSCs in DN treatment, it is crucial to enhance differentiation protocols, employ precise gene-editing tools [102], and implement stringent quality control measures, thereby ensuring a safe and effective approach to personalized therapy.
hPSCs show substantial promise in the treatment of DN, particularly in personalized therapy, genetic correction, and tissue engineering. With advancements in gene editing and bioengineering technologies, hPSCs are expected to offer safer and more effective treatments for DN in the near future (Fig. 3).
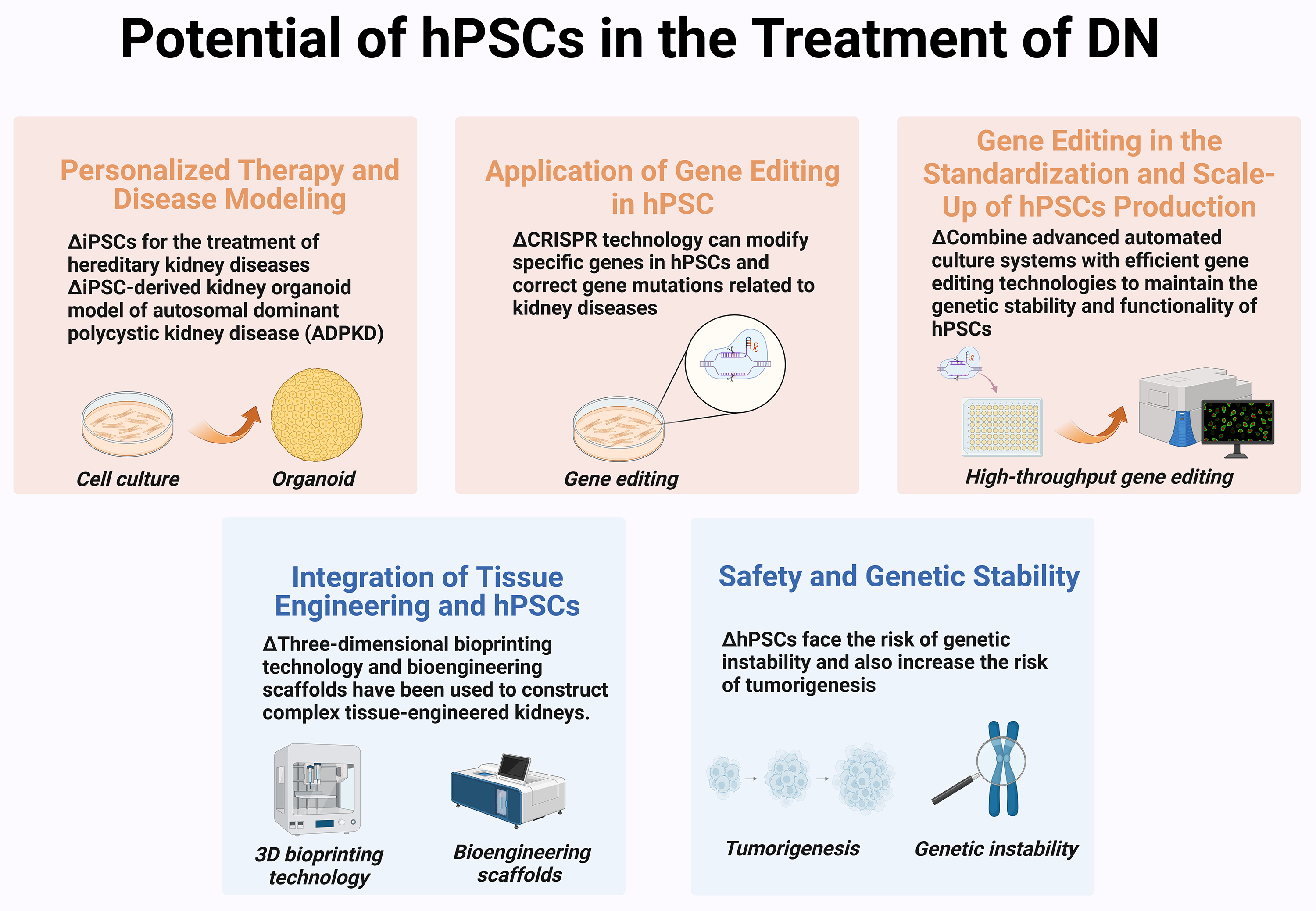 Fig. 3.
Fig. 3.
Potential of hPSCs in the treatment of DN. Therapeutic potential of hPSCs is mainly reflected in their ability to provide personalized, targeted treatments, enhance efficacy through gene editing, enable large-scale standardized production, integrate with tissue engineering, and offer reliable safety and genetic stability. (Created with BioRender.com).
hPSCs possess high pluripotency, allowing differentiation into various renal cell types, which provides an opportunity for personalized therapy. Patient-specific iPSCs can be used to prevent immune rejection associated with allogeneic transplantation, thereby enhancing therapeutic efficacy [103]. For instance, Lim et al. [104] corrected gene mutations in iPSCs derived from patients with Gitelman syndrome using clustered regularly interspaced short palindromic repeats (CRISPR) technology, leading to the development of functional renal organoids with restored normal function, highlighting the potential of iPSCs in treating genetic kidney diseases. Furthermore, hPSCs have demonstrated significant value in disease modeling. Shamshirgaran et al. [105] used CRISPR/Cas9 to generate iPSC-derived kidney organoid models, accurately recapitulating the polycystic kidney disease (ADPKD) phenotype, thus providing an effective platform for target validation and treatment strategy exploration.
hPSCs also play a crucial role in disease modeling. iPSC-derived kidney organoids can partially mimic glomerular diseases, providing valuable tools for studying disease mechanisms and drug screening [106]. Rota et al. [107] highlighted the significant potential of iPSCs in generating kidney-specific cells like podocytes and tubular epithelial cells, though challenges such as post-transplant cell survival and functional integration remain. Moreover, iPSC-derived kidney organoids can effectively mimic pathological processes in diabetic nephropathy (DN), such as foot cell damage and tubulointerstitial fibrosis, providing an important model for DN research [105]. High-throughput drug screening platforms utilizing these organoids can more accurately replicate the human kidney’s response to drug treatments compared to traditional monolayer cell cultures or animal models, making them ideal for drug screening [108]. By integrating kidney organoids into microfluidic chips, multiple drug combinations can be screened in parallel in small volumes across multiple channels. This approach, combined with assays for cell viability, inflammatory markers, and histological staining, enables a comprehensive evaluation of drug effects and toxicity [109].
The application of CRISPR gene editing in hPSCs has shown remarkable potential,
particularly in repairing disease-specific gene mutations. Through CRISPR
technology, researchers can modify specific genes in hPSCs to correct gene
mutations associated with renal diseases, thereby improving therapeutic outcomes
[110]. For example, Brandão et al. [111] introduced specific
mutations in human iPSCs using CRISPR/Cas9, which provides an efficient tool for
modeling genetic kidney diseases. Further advances in gene editing have enabled
multiplex editing and large-scale gene correction, which could be crucial for
treating complex genetic conditions. Ma [112] utilized CRISPR-Cpf1
for simultaneous multi-gene editing, improving the modeling efficiency of complex
kidney diseases. This capability enhances the accuracy of the models, making it
feasible to treat conditions with complex genetic backgrounds. Maguire et
al. [113] developed a CRISPR/Cas9 gene editing method combining two
oligonucleotide repair templates to achieve high precision during specific gene
modifications, thereby enhancing the therapeutic potential of hPSCs. CRISPR has
also been used to enhance the immune evasion capability of hPSC-derived renal
organoids, further improving their therapeutic effectiveness. Gaykema et
al. [114] knocked out
The production of hPSCs is complex and faces challenges regarding standardization and scale-up, particularly in maintaining genetic stability and ensuring batch-to-batch consistency. To meet clinical demands, it is critical to combine advanced automated culture systems with efficient gene editing technologies to maintain the genetic stability and functionality of hPSCs [115]. Jurlina et al. [116] developed a tetracycline-inducible high-fidelity CRISPR platform by integrating high-fidelity Cas9 into the adeno-associated virus integration site 1 (AAVS1) safe harbor locus, which significantly improved editing efficiency and reduced quality variations between batches during large-scale production. This high-precision gene editing technique ensures that hPSCs maintain genetic stability during mass production, thus reducing tumorigenic risk. Wang et al. [117] demonstrated the feasibility of using CRISPR/Cas ribonucleoproteins for high-throughput gene editing, providing a rapid and efficient approach to repairing genetic defects in hPSCs, which is beneficial for producing stable cells on a large scale for therapeutic purposes. These developments will help address challenges in production, particularly regarding the consistency and safety of cells during large-scale production.
The integration of tissue engineering technologies with hPSCs further expands their application potential. For instance, 3D bioprinting has been utilized to construct complex tissue-engineered kidneys, enhancing the integration and regenerative capabilities of hPSCs in vivo [115]. In another study, Kim et al. [118] used decellularized ECM hydrogels to improve the vascularization and maturation of hPSC-derived renal organoids, which successfully recapitulated the renal pathology of Fabry disease, promoting clinical feasibility. Additionally, the use of bioengineered scaffolds can provide an ideal microenvironment for the growth of hPSCs, thereby improving cell integration and overall therapeutic efficacy post-transplantation [119, 120]. Hammad et al. [121] successfully used CRISPR/Cas to introduce a fluorescent biosensor gene into iPSCs, enabling real-time monitoring of cellular behaviors during the generation of kidney organoids. This technology allows more precise tracking of cell differentiation and maturation processes, thus paving the way for further optimization of hPSC-based therapies.
During large-scale in vitro expansion, hPSCs face the risk of genetic instability, which affects therapeutic efficacy and safety [115]. Genetic instability not only compromises therapeutic reliability but also increases tumorigenic risk. Therefore, the development of efficient and precise gene editing methods is essential for maintaining genetic stability in hPSCs. For example, CRISPR/Cpf1-mediated multigene editing can effectively reduce the accumulation of genetic mutations, thereby decreasing tumor risk and enhancing cellular safety [112].
To mitigate genetic instability, researchers have also explored non-integrative CRISPR/Cas9 knockout methods to avoid random integration of exogenous gene segments. Mamun and Bukhari [122] proposed a simplified CRISPR/Cas9 gene editing method that effectively avoids foreign DNA integration, improves editing efficiency, and enhances the applicability of hPSCs for clinical use. High-fidelity CRISPR variants and base editing offer advanced solutions for precise genetic manipulation. Modifying the Cas9/Cpf1 protein structures significantly reduces off-target effects while improving gene editing efficiency [116]. Base editing, which allows base substitution without causing double-strand breaks, reduces the risk of large-scale genomic rearrangements, making it a safer approach for gene correction. Additionally, prime editing further extends the editing potential by handling complex base substitutions and small insertions [123]. The concept of a “body-on-a-chip” offers a novel approach to studying the effects of drugs on multiple systems in patients with DN. By interconnecting multiple microphysiological systems, researchers can more comprehensively simulate drug metabolism and toxicity within the human body. This integrated approach provides a holistic perspective, aiding in the accurate assessment of drug safety and efficacy in DN research [124]. To ensure the safety and consistency of hPSCs, large-scale culture and quality control are essential. Techniques such as digital PCR and next-generation sequencing (NGS) can be employed to monitor off-target edits and genomic structural changes, ensuring the consistency and safety of cell products [125]. In vivo experiments and long-term monitoring are also critical to verifying the stability and functionality of transplanted cells. Animal models can be used to track whether transplanted cells exhibit chromosomal abnormalities or tumorigenic tendencies. Regular assessments of kidney function markers are also necessary to confirm the clinical potential of the cell therapies [108, 126].
DN remains a major complication of diabetes with limited therapeutic options, especially in advanced stages. hPSCs, particularly iPSCs, offer promising avenues for both treatment and research. Their ability to differentiate into kidney-specific cells and enable personalized therapies makes them valuable tools in regenerative medicine. Advances in CRISPR-based gene editing further enhance their therapeutic potential by allowing precise correction of genetic abnormalities. Additionally, hPSC-derived kidney organoids serve as effective models for disease mechanism studies and drug screening. Compared to mesenchymal stem cells, hPSCs provide broader differentiation capabilities, particularly when integrated with tissue engineering strategies such as 3D bioprinting and bioengineered scaffolds. Despite ongoing challenges like tumorigenicity and genomic instability, continued advancements in stem cell biology, gene editing, and biomaterial technology are paving the way for the clinical application of hPSC-based therapies in DN, potentially transforming the future of diabetic kidney disease management.
JY and CZ designed the study; FY and WX drafted the manuscript, HL, XZ and ZS performed literature selection and drew the figures; YD and WZ revised the manuscript. WX, FY, YD, and WZ contributed to the literature selection and acquisition. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We gratefully acknowledge the assistance and instruction from biorender.com and Adobe Illustrator (AI) for creating the figure.
The study was funded by Zhejiang Provincial Science and Technology Program Project (2025C02191). The funding bodies had no role in the design of the study; in the collection, analysis, interpretation of data and in drafting the manuscript.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



