1 Department of Pharmaceutical Sciences, School of Health Science and Technology, UPES, 248007 Dehradun, Uttarakhand, India
2 School of Pharmacy, Dr Vishwanath Karad MIT World Peace University, 411038 Pune, Maharashtra, India
3 Lupin Research Park, 412115 Pune, Maharashtra, India
4 Department of Pharmacology & Toxicology, College of Pharmacy, Prince Sattam Bin Abdulaziz University, 11942 Al-Kharj, Saudi Arabia
5 Department of Pharmacognosy, College of Pharmacy, King Khalid University, 62529 Abha, Saudi Arabia
6 Department of Pharmacology, Delhi Pharmaceutical Sciences and Research University (DPSRU), 110017 New Delhi, India
Abstract
Psoriasis has been a rising concern for over a decade, imposing significant challenges to individuals and society. Traditional topical therapy is non-targeted and acts systemically, with associated side effects. This increases the global burden both socially and economically. This review covers the evolution of drug molecules and nanotechnology-based approaches for the topical treatment of psoriasis, a chronic inflammatory skin disorder with no known etiology. Nanotechnology-based approaches offer promising solutions by reducing side effects, providing targeted delivery, protecting drug molecules from degradation, enhancing skin retention, and providing controlled release. Researchers have investigated the incorporation of various conventional and non-conventional therapeutic agents into nanocarriers for psoriasis treatment. The current understanding of the disease and its treatment using various therapeutic agents combined with novel formulation strategies will reduce the duration of treatment and improve the quality of life in psoriatic disease conditions.
Keywords
- psoriasis
- topical drugs
- delivery systems
- inflammatory cytokines
- nanotechnology
The management of psoriasis, a chronic inflammatory skin disorder, requires innovative therapeutic approaches to overcome the limitations of traditional treatments. Natural agents are widely valued for their diverse biological effects and low side-effect profiles. A comprehensive approach integrating a healthy lifestyle, natural medicines, emerging drugs, and nanotechnology offers promising advancements. The integrative approach not only enhances treatment efficacy but also contributes to reducing the overall burden of psoriasis, improving patient outcomes and quality of life [1, 2]. Psoriasis is a chronic inflammatory skin disease characterized by hyperplasia and hyperkeratosis. In psoriasis, the skin undergoes various changes, manifesting as itchiness, redness, inflammation, scaliness, and emergence of skin lesions (Fig. 1) [3]. The prevalence of psoriasis varies, ranging from 0.27% to 11.4%, depending on genetic factors, sex, age, environment, ethnicity, and geography. It affects approximately 64.6 million people worldwide [4]. Psoriasis can affect the skin, nails, scalps, and joints. It is also associated with various comorbidities such as cardiovascular diseases, cancer, diabetes, anxiety, and depression. Common inflammatory pathways, genetic susceptibility, cellular mediators, and risk factors have been hypothesized to play contributory roles. Psoriasis affects quality of life at the physical, psychological, and social levels, resulting in poor mental health, reduced productivity, and even social exclusion [5].
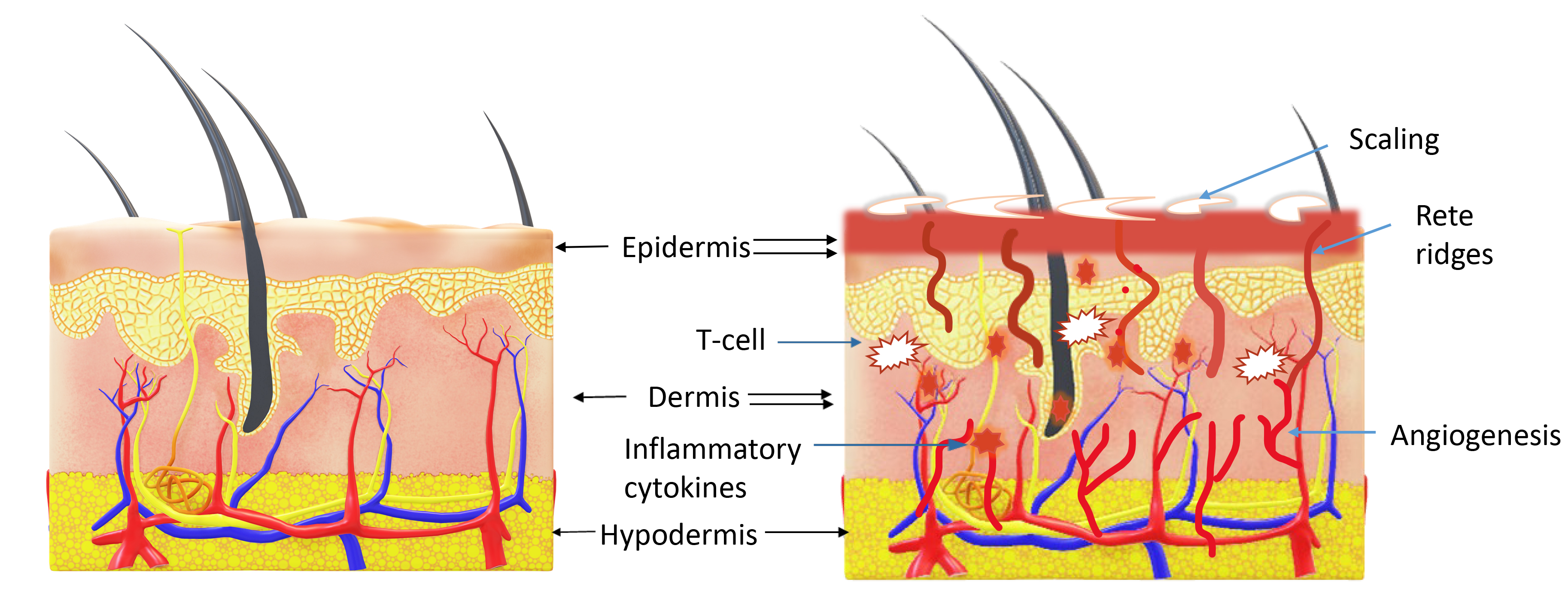 Fig. 1.
Fig. 1.
Comparison of the structure of normal skin and psoriatic skin. Normal skin is represented by single arrows, highlighting its organized structure. Psoriatic (diseased) skin is represented by double arrows, with unique features indicated by blue-colored arrows. In psoriatic skin, there is visible redness, scaling, and significantly increased epidermal thickness. In the dermis, psoriatic skin exhibits increased infiltration of inflammatory cytokines, accumulation of T cells, and increased angiogenesis, contributing to the disease’s characteristic symptoms. Drawn using Powerpoint.
Psoriasis is an autoimmune condition that is triggered by the activation of T lymphocytes. Activated T lymphocytes increase the infiltration of inflammatory cytokines in the dermis and marginally in the systemic circulation, thereby increasing the proliferation of keratinocytes and epidermal cell turnover [6]. One-third of patients with psoriasis have a genetic background. Various factors such as infection, stress, metabolic syndrome, obesity, and diabetes can also trigger it. These triggers can damage the skin, leading to a repair response in which skin keratinocytes undergo excessive proliferation without adequate differentiation. This abnormal process results in an impaired skin barrier and the formation of scaly plaques [7].
Management of psoriasis involves a multifaceted approach including adopting a healthy lifestyle and integrating therapeutic interventions. Key lifestyle modifications include effective stress management, regular physical activity, smoking cessation, avoiding alcohol consumption, maintaining an active skincare routine, dietary modification to reduce systemic inflammation, and utilizing therapeutic agents—essential components of a comprehensive management strategy [8, 9].
Many review articles have described various topical nanotechnology-based formulations and therapeutic agents available for the management of psoriasis [6, 10, 11]. However, study on emerging topical therapeutic agents are still required. Unlike previous reviews, it critically evaluates the integration of emerging and conventional therapies with nanocarriers. The present study attempts to provide insights into the existing state of research on topical therapeutics effective for psoriasis and proposes directions for future research. Using the query “TITLE-ABS-KEY (topical AND “drug delivery” AND psoriasis) AND (LIMIT-TO (DOCTYPE, “ar”)) AND (LIMIT-TO (LANGUAGE, “English”))”, Scopus and PubMed were retrieved at the mid of 2024. A total of 512 articles were retrieved by mid-2024, of which 207 duplicates were removed, leaving 305 articles for review. A total of 132 studies were scanned from 305 articles to confirm that only the relevant studies of interest were selected. After reviewing the title, abstract, and keywords, 156 articles were removed, and 37 were removed after reading the full text. A total of 112 articles were selected for this study.
Psoriasis is a multifactorial disease triggered by stress-induced keratinocyte
damage due to various factors. Its pathophysiology involves multiple components,
including immune cells, genetic predispositions, antimicrobial peptides, and
non-coding RNAs (Fig. 2). Psoriasis is an immune-driven condition characterized
by T lymphocyte activation. Dendritic cells, which function as antigen-presenting
cells (APCs), initiate an immune response by generating interleukin -12 (IL-12)
and interleukin -23 (IL-23), prompting the activation of T helper 1 (Th1) and T
helper 17 (Th17) cells. Th1 cells generate tumor necrosis factor-
 Fig. 2.
Fig. 2.
Key components involved in the pathophysiology of psoriasis. Th1, T helper 1 cells; Th17, T helper 17 cells; IL-17, Interleukin-17; PSORS, Psoriasis-susceptible. Drawn using Powerpoint.
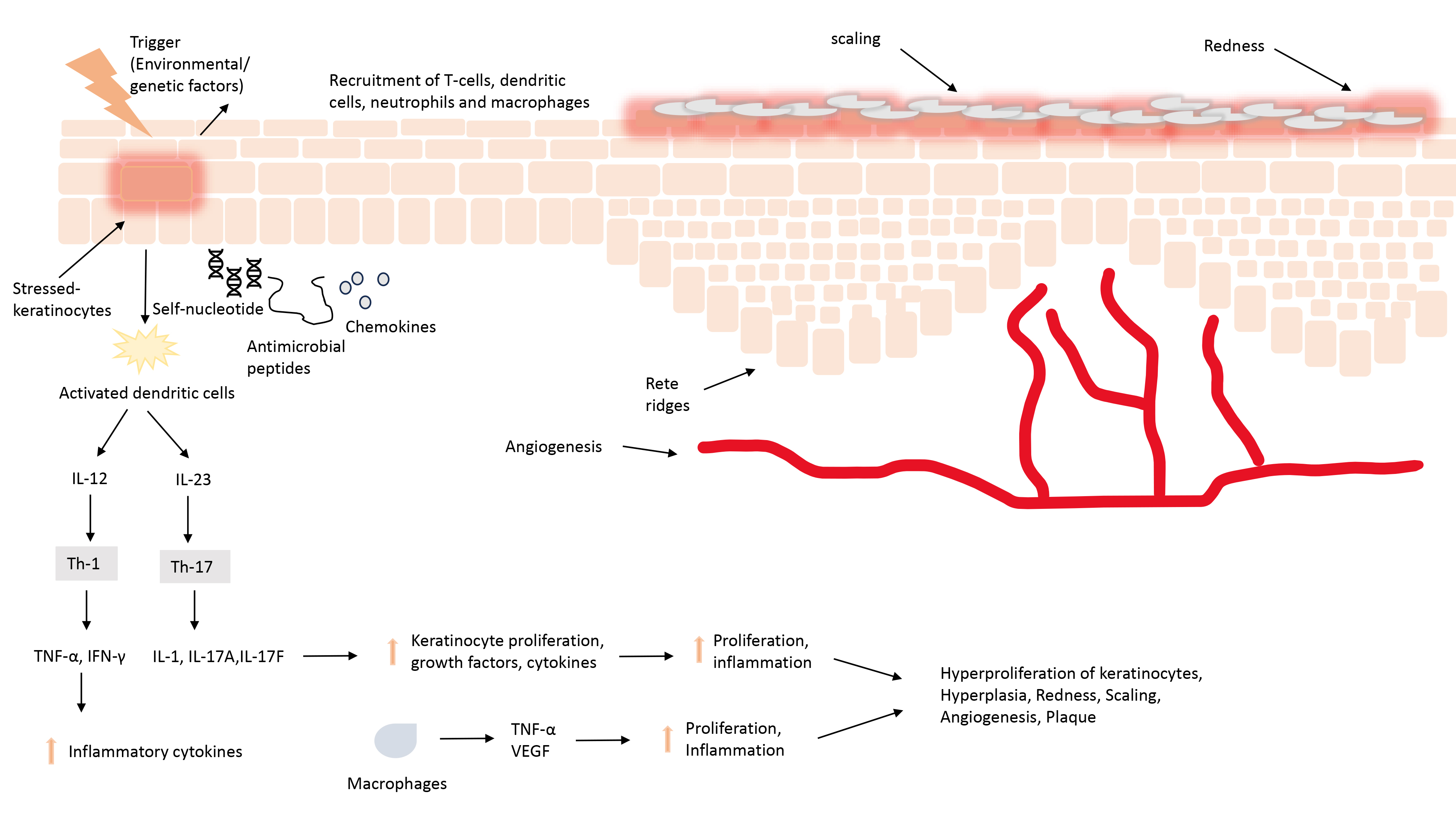 Fig. 3.
Fig. 3.
The pathophysiology of psoriatic skin involves stress-induced
activation of keratinocytes, which, when combined with self-nucleotides,
antimicrobial peptides, and various chemokines, leads to the activation of
dendritic cells. Abbreviations: IL, interleukin; Th, T helper cells,
TNF-
Additionally, Langerhans cells serve as mediators in activating distinct types
of T cells such as natural killer (NK) cells and NK-T cells. Th1 and Th2 utilize
the Janus kinase/signal transducers and activators of transcription (JAK-STAT)
pathway to perform their functions. In contrast, Th17 cells operate through the
activator 1 (ACT1) adapter protein and nuclear factor kappa B (NF-
Psoriasis is associated with significant changes in gene expression. Psoriasis occurs more frequently in monozygotic twins than in dizygotic twins. It is activated by environmental factors in individuals with genetic predisposition. Approximately 60 psoriasis-susceptible regions have been identified. According to linkage analysis, nine loci known as psoriasis-susceptible (PSOR1-9) are responsible for disease susceptibility. PSORS-1 was the most validated, whereas a weaker linkage was observed in PSORS-2 and PSORS-4. Linkage in the remaining regions was not replicated in independent research. PSORS-1 is the major histocompatibility complex (MHC) locus on chromosome 6p21. The 150 kb region of MHC-1 contains nine genes, of which three, CCHCR1, HLA-C, and CDSN, are significantly related to psoriasis. HLA-C codes for MHC class I receptors that participate in the immune response by acting as an antigen for CD8+ lymphocytes. In the PSORS-2 locus, the mutation in CARD14 is responsible for the phenotype of plaque and pustular psoriasis. PSORS-4 is located on the 1q21 chromosome; it stretches the epidermal differentiation cluster (EDC). Absence of the EDC gene is associated with psoriasis [15].
Antimicrobial peptides (AMPs) are 12–50 amino acid long, amphiphilic molecules.
Keratinocytes, T cells, dendritic cells, and neutrophils secrete antimicrobial
peptides such as S100,
Non-coding RNAs such as microRNAs (mRNAs) and long non-coding RNAs (lncRNAs) also play a role in the pathogenesis of psoriasis. More than 250 mRNA were atypically expressed in psoriatic lesions. Some mRNAs were upregulated, whereas others were downregulated. For example, mRNA miR-31 is upregulated; therefore, deletion of miR-31 alleviates inflammation and hyperplasia of keratinocytes. Conversely, miR-14a/b is highly expressed in psoriatic lesions and negatively regulates keratinocyte proliferation—so it has a protective effect. Genetic deficiency of miR-14a exacerbates psoriatic inflammation. lncRNAs control gene expression at both the transcriptional and post-transcriptional levels. In psoriasis, the non-protein-coding RNA induced by stress (PRINS), a type of lncRNA, is significantly overexpressed in the epidermis of affected patients. PRINS is activated by stress and its silencing decreases keratinocyte viability under stress conditions. However, the lncRNA maternally expressed gene 3 (MEG3) is downregulated in psoriatic skin. MEG3 suppresses keratinocyte proliferation and enhances apoptosis by targeting miR-21 and increasing Caspase 8 expression [18].
The available treatments for psoriasis aim to alleviate symptoms and improve the
patient’s quality of life, as psoriasis is a chronic disease with no cure and
requires long-term treatment. This creates economic challenges for the healthcare
and pharmaceutical industries [19]. Treatment of psoriasis consists of
combinations of therapeutic agents to address complications of psoriasis
depending on the severity of the disease, patient-specific factors, and the
presence of co-morbidities. The therapeutic agents alleviate symptoms, improve
skin health, and enhance the quality of life for patients. Broadly, therapeutic
approaches can be categorized into topical treatments, systemic therapies, and
advanced biologics. While topical treatments remain the cornerstone for managing
mild to moderate psoriasis, systemic therapies and biologics are pivotal for
severe cases. Topical agents are the first-line treatment for mild to moderate
cases. Topical agents include corticosteroids, vitamin D derivatives, retinoids,
anthralin, and salicylic acid (SA). Potent and super potent corticosteroids are
as effective as vitamin D derivatives and can be combined with keratolytic
agents, such as salicylic acid, to treat thick scaling. Vitamin D derivatives can
be used alone or with corticosteroids but should not be used with keratolytics or
phototherapy [3]. Systemic agents are used to treat moderate-to-severe psoriasis.
Systemic agents include the following: retinoids, such as acitretin;
immunosuppressants, such as methotrexate and tacrolimus; and immunomodulators,
such as cyclosporin. These agents can cause serious side effects, such as
nephrotoxicity, hepatotoxicity, and increased risk of infection. Therefore, these
agents are unsuitable for long-term treatment. Retinoids are teratogenic and
should not be used for women of childbearing age [20]. Phototherapy is used in
moderate-to-severe psoriasis; it includes narrow- and broad-band Ultraviolet B
(UV-B) and Ultraviolet A (UV-A) therapy with or without psoralen. Challenges
associated with phototherapy include the limited number of centers available,
frequent visits, cost, and increased risk of skin cancer [21]. Biologicals are a
relatively new treatment approved by the US FDA (United States Food and Drug
Administration) and are used to treat moderate to severe psoriasis but are costly
and lack long-term safety data. Biological agents included TNF-
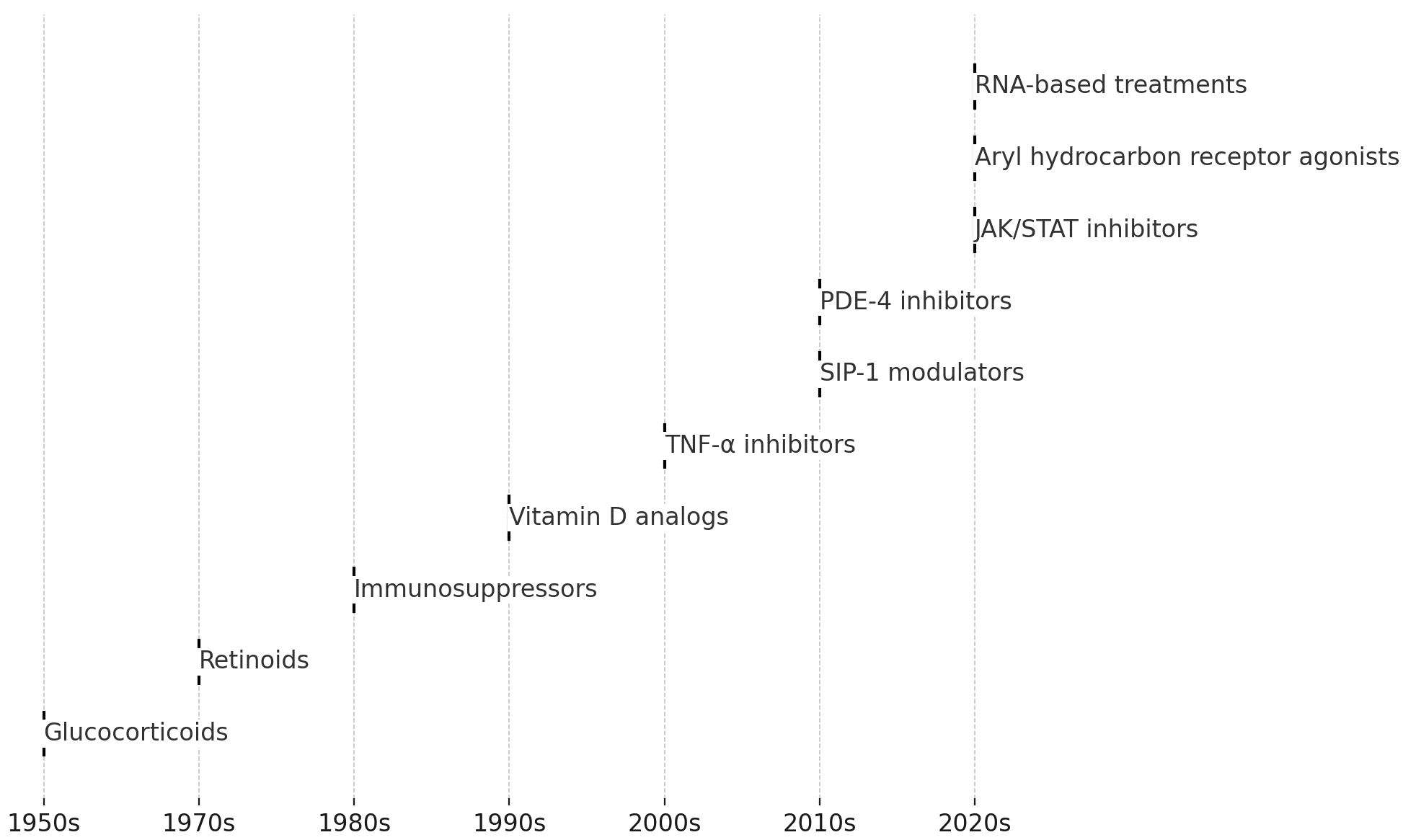
Topical treatment is effective in most patients with psoriasis, and only 20%
require systemic therapy [25]. Glucocorticoids, the primary drugs used for
treating psoriasis, were first introduced in the 1950s, followed by the
development of immunosuppressants and retinoids. Treatment has advanced
significantly with the approval of vitamin D analogs in the 1990s, followed by
the introduction of TNF-
Glucocorticoids are the first-line treatment, either alone or in combination. It has multiple functions, including antimitotic, anti-inflammatory, immunomodulatory, and vasoconstrictive effects. Therefore, it helps to reduce redness, swelling, scaling, and itching in psoriatic lesions. Glucocorticoids are chemically derived from androgens and pregnanes. Glucocorticoids consist of four lipophilic rings, which enable them to bind to glucocorticoid receptors [27]. Topical glucocorticoids were introduced in the 1950s and are still preferred for psoriasis management. Four classes of glucocorticoids are used to treat psoriasis: low potency (LPG), mid potency (MPG), high potency (HPG), and super high potency (SHPG). Low- and mid-potency glucocorticoids are used in folds, whereas high- and super-high-glucocorticoids are used on the exterior surfaces. Glucocorticoids act via two pathways, genomic and non-genomic. In the genomic pathway, cortisol binds to the glucocorticoid receptor, resulting in receptor dimerization and binding with the glucocorticoid responsive element (GRE), causing transcription of anti-inflammatory genes such as tyrosine aminotransferase (TAT), IL-10, antagonizing IL-1, and promoting dual-specificity protein phosphatase 1 (DUSP-1). Glucocorticoids also negatively regulate the expression of proinflammatory genes, such as cytokines, adhesion molecules, growth factors, nitric oxide, and autocoids. The non-genomic pathway does not involve de novo protein synthesis, so it rapidly affects binding to the glucocorticoid receptor, and a second messenger comes into action. It alters the activation and response of target cells such as T cells, platelets, and monocytes [27, 28].
The active form of VDA is calcitriol or 1,25(OH)2D3, whereas other synthetic analogs include calcipotriol, calcitriol, maxacalcitol, and tacalcitol. The first topical treatment for VDA was calcipotriol, which was introduced in Europe in 1987 and approved by the US FDA in 1993. It reverses epidermal dysregulation and hyperproliferation and induces apoptosis. In psoriasis, reduced vitamin D receptor expression is correlated with lower tight junction protein levels, which disrupts the skin barrier. Tight junctions are essential for keratinocyte adhesion and permeability and regulate cell differentiation through interactions with nuclear and cytoplasmic proteins. Vitamin D3 promotes keratinocyte terminal differentiation and inhibits proliferation, although the exact mechanism remains unclear. It also has immunomodulatory effects. It acts on cells involved in immunologic reactions, such as macrophages, lymphocytes, and Langerhans cells [29, 30, 31, 32].
Methotrexate is generally used as a systemic agent, but numerous topical formulations have been reported in the literature. It was introduced as a chemotherapeutic agent in the 1950s for treating cancer and autoimmune diseases, with its use in psoriasis beginning in the 1960s. Methotrexate (MTX) is a folic acid analog and antagonist. It binds to folic acid reductase with a much higher affinity than folic acid. It inhibits the synthesis of tetrahydrofolate and pyrimidine, which are required to synthesize DNA base pairs, leading to reduced DNA replication and inhibition of RNA and protein synthesis. It reduces epidermal cell proliferation and lymphocyte number [20].
Cyclosporine, a calcineurin inhibitor, was initially used as an immunosuppressant in organ transplants to prevent rejection, introduced for psoriasis treatment in the 1980s, and received FDA approval in 1997. The mechanism of action of cyclosporine is immunosuppression by binding with cyclophilin and inhibition of calcineurin, which is required for the transduction of calcium-dependent signals from T lymphocytes to cytokine promoters. It inhibits the proliferation of CD4+ helper T cells and keratinocytes. It regulates the immune system by explicitly inhibiting T-cell overproduction and impairing the function of antigen-presenting Langerhans cells. Whether taken orally or topically, cyclosporin has an affinity for skin cells. Cyclosporine reduced cell infiltration and epidermal thickness. It can be administered via a systemic or topical route; however, systemic treatment causes various side effects, which can be reduced with the use of topical formulations [20, 33, 34]. Other topical calcineurin inhibitors used for the treatment of psoriasis include tacrolimus and pimecrolimus. They exert an antipsoriatic effect by inhibiting the proliferation of T lymphocytes and dysregulation of mast cells. They do not affect fibroblasts and endothelial cells or cause skin atrophy [35].
Retinoids can be administered via both systemic and topical routes. Etretinate
is a synthetic analog of retinoic acid approved by the FDA for oral use in the
treatment of severe psoriasis in 1986. It is a metabolite of etretinate, has a
shorter half-life, and is preferred as a systemic agent for managing psoriasis
[36]. Tretinoin and isotretinoin are topical and systemic agents, respectively,
whereas adapalene and tazarotene are topical agents. Retinoids are teratogenic
when administered systemically. Retinoids have anti-hyperproliferative potential.
They modulate T lymphocyte responses, inhibit chemotactic responses, activate
polymorphonuclear leukocytes, and increase Langerhans cells. They also stimulate
natural killer lymphocytes and induce the migration of inhibitory factor-related
proteins (MRP-8) by inhibiting IFN-
Fingolimod was the first SIP1 modulator approved by the US FDA in 2010 for oral treatment of multiple sclerosis. Owing to its success in modulating the immune response, its potential in treating autoimmune diseases, including psoriasis, has been explored [38]. AKP-11 is a highly selective sphingosine-1-phosphate receptor modulator. SIP is a lipid that binds to G protein-coupled S1P receptors and modulates cell proliferation, cell differentiation, and proinflammatory cytokine production, thus helping maintain skin barrier function. The topical formulation of AKP-11 has been used in clinical trials for psoriasis treatment [39, 40]. Similarly, HWG-35D, a sphingosine kinase 2 inhibitor, is beneficial for psoriasis. Sphingosine kinase increases the phosphorylation of sphingosine phosphate and its inhibition blocks Th17 differentiation into CD4+ T lymphocytes. The topical application of HWG-35D (a novel sphingosine kinase 2 inhibitor) normalized the systemic and local immune responses induced by imiquimod (IMQ) treatment [40].
Tapinarof is the first aryl hydrocarbon inhibitor that received US FDA approval on May 23, 2022, to treat mild, moderate, and severe psoriasis. Tapinarof is a secondary metabolite obtained from the Gram-negative bacterium, Photorhabdus luminescens. The binding of tapinarof to the aryl hydrocarbon receptor leads to the translocation of the receptor from the cytoplasm to the nucleus after dimerization with the aryl hydrocarbon nuclear translocator, activating different genes. Aryl hydrocarbon signaling regulates Th17 and Th22 differentiation and IL-17 and IL22 expression. Tapinarof regulates keratinocyte function by inducing the expression of skin barrier genes. Therefore, it helps normalize the skin barrier disrupted by psoriasis. It reduces epidermal oxidative stress by directly scavenging reactive oxygen species and increasing the expression of antioxidant enzyme genes [23, 41].
Omiganan is a 12-amino-acid, antimicrobial cationic peptide active against many Gram-negative and Gram-positive microbes and fungi. It inhibits the activation of toll-like receptors (TLRs) on dendritic cells and other immune cells. This inhibition prevents the downstream inflammatory cascade that contributes to psoriasis. It also possesses anti-inflammatory properties. The liposomal gel formulation of omiganan substantially reduced psoriatic lesions by reducing the levels of proinflammatory cytokines [42]. Thiostrepton is a naturally occurring antimicrobial peptide that inhibits the activation of toll-like receptors 7-9 (TLR7-9) in dendritic cells. In psoriasis, these receptors are activated and contribute to the inflammatory response. Thiostrepton mitigates this activation through two primary mechanisms: the inhibition of proteasome function and endosome acidification. In mouse models, thiostrepton ameliorates psoriasis-like inflammation induced by imiquimod and LL37 [43].
PDE is expressed in keratinocytes and immune cells. It mediates the inflammatory
response by hydrolyzing cyclic adenosine monophosphate (cAMP). Inhibition of
PDE-4 increases intracellular cAMP concentrations, which activates various
downstream pathways. This process inhibits the inflammatory response by
suppressing the production of inflammatory cytokines and activating
anti-inflammatory mediators [44]. Apremilast was the first PDE-4 inhibitor to
receive US FDA approval in March 2014 for the treatment of plaque psoriasis and
psoriatic arthritis. Apremilast acts as an anti-inflammatory drug by inhibiting
various proinflammatory cytokines involved in the pathogenesis of psoriasis, such
as TNF-
Many herbal drugs have been explored for their antipsoriatic activity for a long
time. Curcumin has shown antipsoriatic activity, as reported in many clinical and
preclinical research. Curcumin has an inhibitory effect on IL-22-induced signal
transducer and activator of transcription 3 (STAT3) phosphorylation. It also
inhibits vesicular endothelial growth factor (VEGNF-
Nutraceuticals have both nutritional and medicinal properties [63]. Oleuropein, a nutraceutical product obtained from olive leaves, has anti-inflammatory and antioxidant properties, and its microemulsion formulation was found to be better than the marketed formulation Dermovate in patients with psoriasis, as reported by the authors [64]. (-)-epigallocatechin-3-gallate (EGCG), a catechin found in green tea, induces the expression of keratinocyte differentiation markers and has immunoregulatory and antiangiogenic properties [65].
TNF-
JAK inhibitors target specific proinflammatory cascades in psoriasis. Cytokines,
such as IL-6, IL-12, IL-22, and IL-23, play a role in the development of
psoriasis. They bind to type I and II receptors, which depend on JAKs for signal
transduction. Upon the binding of cytokines to their receptor, receptors undergo
conformational changes and activate and recruit two JAK proteins. Upon
activation, JAK alters the receptor, allows binding of STAT proteins, and results
in phosphorylation, dimerization, and translocation to the nucleus to change gene
expression. There were four JAK and seven STAT proteins. Inhibition of each
subtype of JAK can disrupt various downstream signaling pathways, making it an
effective treatment for reducing inflammation in psoriasis [70]. Deucravacitinib
is an oral drug approved by the US Food and FDA for the treatment of
moderate-to-severe psoriasis. It is a tyrosine kinase 2 (TYK2) inhibitor, a
member of the JAK family of inhibitors [71]. Topical JAK inhibitors include
tofacitinib, ruxolitinib, and baraticinib. Baraticinib inhibits JAK1 and JAK2
activities by disrupting the production of downstream signaling molecules and
proinflammatory mediators. Tofacitinib targets JAK1 and JAK3, whereas ruxolitinib
inhibits JAK1 and JAK2. JAK inhibitors prevent the phosphorylation and activation
of JAK, inhibiting dimerization and activation of STAT to move from the cell to
the nucleus to influence DNA transcription and gene expression. Tofacitinib
inhibits the expression of various interleukins, such as IL-16 and IL-23, thus
suppressing Th17 differentiation. It also inhibits IL-15 expression, which
increases keratinocyte apoptosis. Ruxolitinib suppresses the phosphorylation of
STAT3, resulting in reduced expression of IFN-
There is a change in the expression of various genes in psoriasis, and RNA-based treatments have been explored. RNA-based treatments are still in an investigational state. Although advances in delivery mechanisms and more targeted approaches have increased the interest in these treatments, they are still not widely approved or used in clinical practice for the treatment of psoriasis. Microinterfering RNAs (miRNAs) play vital roles in cell differentiation, proliferation, apoptosis, and immune responses. miRNA is highly expressed in psoriatic skin, and topical treatment with biomimetic reconstituted high-density lipoprotein nanogel of miRNA-210-antisense improved the psoriatic symptoms and reversed the immune disorder in the imiquimod-induced mouse model [24]. Similarly, elastic liposomal carriers of RNA interference (RNAi) downregulate human beta-defensin 2, a psoriasis marker, in a psoriatic tissue model [78]. Spherical nucleic acid nanoparticle conjugates (SNA-NCs) of short interfering RNA (siRNA) significantly reduce gene effects and cell proliferation [79].
Anthralin inhibits keratinocyte proliferation. It accumulates in the
mitochondria and impairs the energy supply. It also interferes with DNA
replication and slows cell division [80]. Tamoxifen, a selective estrogen
receptor modulator, inhibits keratinocyte viability and induces an immune shift
from Th1 to Th2. It releases anti-inflammatory cytokines such as IL-4, IL-10, and
IL-13. Squalene-integrated nanostructured lipid carriers increased the moisture
and lipid content of the skin and reduced the Psoriasis Area and Severity Index
(PASI) score and proinflammatory cytokine levels [81]. Leflunomide, a
disease-modifying anti-rheumatic drug, exerts an antipsoriatic effect by slowing
lymphocyte proliferation. Leflunomide rapidly metabolizes to teriflunomide and
inhibits dihydroorotate dehydrogenase, a rate-limiting enzyme in pyrimidine
synthesis in lymphocytes, thus modulating DNA synthesis [82]. Pentoxifylline is a
hemorheological agent. It inhibits TNF-
| Drug Class | Examples | Mechanism of Action | Effect | Reference |
| Glucocorticoids | Clobetasol propionate, halobetasol propionate, betamethasone dipropionate | Anti-inflammatory, immunosuppressive, and vasoconstriction by binding glucocorticoid receptors and regulating proinflammatory gene expression. | Reduces redness, swelling, and scaling of psoriatic lesions. | [27, 28] |
| Vitamin D3 Analogs | Calcitriol, calcipotriol, maxacalcitol | Promotes keratinocyte differentiation, inhibits proliferation, and modulates immune response. | Reverses dysregulated epidermal differentiation and inhibits keratinocyte proliferation. | [30] |
| Immunosuppressors | Methotrexate, cyclosporine | Methotrexate inhibits DNA synthesis, cyclosporine suppresses T-cell activity and immune response. | Reduces epidermal cell turnover and inflammation; decreases cell infiltration and epidermal thickness | [20, 33] |
| Retinoids | Acitretin, tazarotene, adapalene | Modulates T-cell response and keratinocyte proliferation, and has anti-inflammatory effects. | Reduces hyperkeratosis and normalizes keratinocyte function. | [37] |
| S1P Receptor Modulators | Fingolimod, AKP-11, HWG-35D | Modulates immune response by binding sphingosine-1-phosphate receptors and reducing cytokine production and cell proliferation. | Maintains skin barrier function and reduces inflammatory response. | [38, 40] |
| Aryl Hydrocarbon Receptor Agonist | Tapinarof | Regulates keratinocyte function, reduces oxidative stress, and modulates immune responses by activating AhR signaling pathways. | Normalizes skin barrier function, reduces oxidative stress, and inhibits proinflammatory cytokines | [23] |
| Antimicrobial Peptides | Omiganan, thiostrepton | Inhibits proinflammatory pathways and toll-like receptors on immune cells. | Reduces proinflammatory cytokines and ameliorates inflammation in psoriatic lesions. | [42, 43] |
| PDE-4 Inhibitors | Apremilast, roflumilast | Increases cAMP levels, which in turn suppresses inflammatory cytokine production. | Reduces inflammatory cytokines (TNF- |
[45, 46] |
| Herbal Drugs | Curcumin, berberine | Inhibits proinflammatory cytokines (IL-22, TNF- |
Decreases inflammation, blocks cell proliferation, and improves psoriatic lesions. | [49, 52] |
| TNF- |
Capsaicin, etanercept | Blocks TNF- |
Reduces inflammation and improves clinical symptoms of psoriasis. | [66, 67] |
| JAK/STAT Inhibitors | Tofacitinib, ruxolitinib, baraticinib | Targets JAK/STAT signaling, which is crucial in inflammatory pathways. | Reduces cytokine production, keratinocyte apoptosis, and inflammation in psoriatic lesions | [72] |
| Nutraceuticals | Oleuropein, EGCG (green tea extract) | Anti-inflammatory and antioxidant effects; supports skin health and immune function. | Supports other treatments. | [64, 65] |
| Endogenous Peptides | Reduces inflammatory cytokines, scavenges ROS, and normalizes immune responses in psoriasis. | [75, 77] | ||
| RNA-Based Treatments | miRNA-210, siRNA | Modulates gene expression related to inflammation, cell proliferation, and keratinocyte activity through RNA interference mechanisms. | Reduces inflammation and keratinocyte hyperproliferation in psoriatic models. | [24, 79] |
| Miscellaneous | Anthralin, tamoxifen, leflunomide | Inhibits keratinocyte proliferation and induces a shift from Th1 to Th2 immune response; also induces anti-inflammatory cytokines like IL-4, IL-10. | Reduces skin cell proliferation and inflammation, and improves psoriatic symptoms. | [80, 81, 82] |
Abbreviations:
Traditional topical formulations, such as creams, lotions, ointments, and gels, offer various benefits but have drawbacks. They may lead to itching, pain, inflammation, and local irritation and can be linked to erythema edema caused by drugs or excipients in the formulation [87]. Molecules with a small molecular weight can pass through the systemic circulation, which may be desired for the systemic delivery of therapeutic agents. However, when a local effect is required, the systemic absorption of the drug molecule may lead to undesirable side effects. For example, long-term use of super-potency corticosteroids may cause hypothalamus-pituitary-adrenal axis (HPA) suppression in young children [88]. Creams and ointments can be greasy, sticky, and have unpleasant odors. They may also be less effective and contain high drug concentrations, potentially affecting patient compliance. Creams, ointments, lotions, and gels exhibit imprecise and unpredictable drug delivery, and drugs do not remain at the site of action for the required period [89].
Nanotechnology plays an important role in overcoming the limitations of the conventional dosage forms. Nanocarriers increase the dermal retention of the drug, deliver the drug at the target site, and reduce the dose, thereby reducing side effects, and increasing patient compliance and acceptability [11]. Agarwal et al. [90] reported that the tolerability, efficacy, stability, and acceptability of liposomal gel formulations of dithranol increased in patients with plaque psoriasis compared to traditional cream formulations in an open-label trial. The co-delivery of clobetasol propionate and calcipotriol nanoemulsion loaded in the gel has high retention in the viable epidermis and dermis, negligible skin irritation despite high penetration, and controlled release, as reported by the authors [91]. These are just a few examples that demonstrate the added value of nanotechnology. Given their importance, the following sections provide a detailed overview of nano-based and other technical advances in drug delivery systems for the topical treatment of psoriasis.
Psoriasis is an immune-mediated skin condition that requires drugs targeting
cells in the epidermis and dermis, including dendritic cells, Langerhans cells,
keratinocytes, T lymphocytes, and mast cells. The stratum corneum of the skin is
a barrier that limits drug entry to molecules
Liposomes are bi-layered vesicles of cholesterol, phospholipids, and fatty acids
that encapsulate both hydrophilic and hydrophobic drugs. Liposome lipid
composition enables drug delivery via skin. Lipid bilayer vesicles containing
large concentrations of ethanol, known as ethosomes, increase permeation to
deeper layers of the skin by opening new channels [25, 96]. Ethosomes containing
anthralin demonstrated significantly superior results in enhancing the PASI,
scoring 81.84% compared with liposomes (68.66%) in clinical trials involving
patients with psoriasis [80]. Transferosomes, also known as deformable liposomes,
have an aqueous core that is surrounded by lipids and amphiphilic surfactants.
The elasticity of transferases is due to the presence of edge activators, along
with phospholipids, which destabilize the lipid bilayer [97, 98]. Transethosomes
have the combined properties of transferosomes and ethosomes, and are composed of
phospholipids, edge activators, ethanol, and water. They can pass through the
stratum corneum and enhance drug delivery to the dermis [25]. Rahangdale and
Pandey prepared a topical transethosome formulation of the anti-inflammatory and
PDE-4 inhibitor drug apremilast using sodium cholate, Lipiod S 100, and ethanol
and incorporated it into 1% carbopol gel. An ex vivo permeation study
on rat skin demonstrated that the drug penetrates the dermis layer where
psoriasis originates, indicating its accessibility for therapeutic action [99].
Niosomes are non-ionic surfactant-based vesicular drug delivery systems. Niosomes
are composed of an aqueous core surrounded by a surfactant macromolecule bilayer
and cholesterol. Niosomes are osmotically active, less immunogenic, more stable,
and less expensive [96]. Ceramide-based liposomes are known as cerosomes. These
are composed of sphingolipids. Sphingolipids are natural lipids that make up
45–50% of the intercellular lipids of the stratum corneum and thus increase
drug permeation and retention in the skin [100]. Lipospheres are lipid-based
nanocarriers with a solid hydrophobic core surrounded by a phospholipid layer
that acts as a stabilizer. Lipospheres are easy to prepare and have a low cost,
stability, and high aqueous dispersibility. Lipospheres are favored for topical
use in psoriasis because of their superior penetration across skin layers.
Tacrolimus and curcumin-based liposomes reduced TNF-
Nanoemulsions are colloidal isotropic drug delivery systems, similar to regular emulsions. They have oil and aqueous phases as dispersed or continuous phases, stabilized by surfactants, but at low concentrations, and the size of droplets ranges from 20 to 500 nm. Microemulsions are similar to nanoemulsions, but the droplet size range is 5–100 nm, and they are thermodynamically stable [6, 96]. A cyclosporin microemulsion containing isopropyl myristate, Tween 80, and isopropyl alcohol was formulated to achieve droplet sizes below 50 nm. The local drug depot allowed sustained release of viable skin for 24 h. An ex vivo permeation study using goat skin demonstrated that a cyclosporine microemulsion gel significantly enhanced the permeability of cyclosporine compared with a traditional cyclosporine suspension [105].
SLNs, which are solid at body and room temperature, encapsulate hydrophilic and
lipophilic drugs. They feature a lipid core stabilized by a surfactant, which
enhances drug penetration through the stratum corneum. SLNs also increase contact
with the skin, leading to enhanced skin hydration owing to their occlusive
nature. The topical SLN formulation of retinoid acitretin showed an encapsulation
efficiency of 89
Micelles are colloidal drug delivery systems composed of self-assembled amphiphilic molecules with a core-shell structure above the critical micelle concentration (CMC). Micelles enclose hydrophobic drugs in a nonpolar core, and the polar shell faces the aqueous medium. The small size of micelles mediates crossing the stratum corneum and increases drug deposition in a viable layer of the skin [6, 108].
Dendrimers are spheroidal, multivalent, hyperbranched, three-dimensional structures with active end groups. Dendrimers are biocompatible, increase the solubility of drugs, form prodrugs (drug-polymer conjugates), control the release of drugs, and increase the skin permeation of topically applied drugs [95].
Polymeric nanoparticles incorporate drugs into a polymeric matrix, releasing them through diffusion, erosion, or swelling. Microspheres (1–1000 µm) ensure controlled drug release, osmotically driven by polymer properties, drug amount, or drug-to-polymer ratio. Polymeric nanoparticles (10–100 nm) include nanospheres with dispersed drugs and nanocapsules with a core-shell structure for drug storage [98]. Coal tar-loaded PLGA nanoparticles exhibited a high drug loading of 92%, with a permeability study on the Strat-M membrane indicating 97% local drug accumulation within 24 h. The study also observed improved washability and reduced staining capacity of coal tar compared with crude coal tar [109].
Metallic nanoparticles are inert particles that are small in size and reactive against living cells [95]. Various metallic nanoparticles such as zinc oxide, silver, gold, and silica have been reported. They increase the permeation of drugs through the skin and exhibit anti-inflammatory properties [110, 111]. Methotrexate-loaded gold nanoparticles exhibited 70–80% loading efficiency, proved non-toxic to keratinocytes, demonstrated enhanced penetration in the epidermis and dermis of methotrexate from methotrexate-loaded gold nanoparticles compared to methotrexate alone, and displayed increased drug permeability through mouse skin [112].
Colloidal carriers offer many advantages such as high stability, high drug load, solubilization of poorly soluble drugs, and sustained release. Nanosponges (200–500 nm) can entrap hydrophobic and hydrophilic drugs and enhance cutaneous drug retention. Microsponges (size range 5–300 µm) provide prolonged contact time of the drug with the skin. It increases epidermal drug content while preventing absorption into the systemic circulation [113]. The microsponges of clobetasol propionate provide high encapsulation efficiency and a zero-order drug release pattern. In vivo drug efficacy research showed better antipsoriatic effects and reduced side effects compared to plain clobetasol propionate gel [114]. Cyclodextrin nanosponges of dithranol showed marked improvement in the antipsoriatic activity of the drug compared to the untreated group [115].
Microneedles, a non-invasive drug delivery system, penetrate the thick stratum corneum of the skin and enhance drug retention in the epidermis and dermis. Dissolvable microneedles improve drug delivery by dissolving them after their insertion into the skin [116]. Lu et al. [117] prepared a novel black phosphorus (BP)-loaded hydrogel inverse opal microneedle with photo and thermal responsive capability. The microneedle was composed of BP-loaded N-isopropyl acrylamide (NIPAM)/poly (ethylene glycol) diacrylate (PEGDA) inverse opal hydrogel scaffold and gelatin/agarose filled scaffold for drug loading. It was fabricated via nanoparticle assembly, and reversed-phase replication with micro-molding, which enables hydrophobic drug loading and photothermal responsive controlled drug delivery. This method enables good medication administration and efficiently improves psoriatic skin conditions by lowering the synthesis of inflammatory cytokines and epidermal hyperplasia in mice compared with calcipotriol ointment treatment. The dissolving microneedle array significantly increased the skin deposition of tofacitinib citrate (835 µg/cm2) compared with the control cream (143.98 µg/cm2) [73].
Various technical advances have also been explored to enhance drug delivery to the skin in psoriatic conditions. The increased epidermal thickness in psoriasis makes drug delivery via the topical route difficult, despite it being the preferred method for treating this condition. Skin pretreatment with a fractional laser using an Er: YAG laser device creates micropores and causes minimum coagulation, increasing drug penetration through the skin [67]. Iontophoresis uses a mild electric field to move a charged drug from the application site to the target site. The movement occurs because of electron repulsion or convection flow. Iontophoresis increases the transdermal penetration of drugs, depending on ionic strength [118]. Electroporation uses a high voltage of milliseconds to the skin to generate an electric pulse and create pores for transit time. Therefore, it increases drug transport due to electroosmosis and diffusion [119]. Recent clinical studies have shown that fractional CO2 laser technology for the delivery of methotrexate is a promising, efficacious, and well-tolerated substitute for the conventional method of intralesional injection in the treatment of nail psoriasis [120, 121]. There was a significant improvement in the nail bed and Nail Psoriasis Severity Index (NAPSI) scores for combined fractional CO2 laser and calcipotriol/betamethasone ointment preparation compared to fractional CO2 laser alone in a clinical trial in 30 patients. NAPSI scores were calculated at the beginning of the study and 3 months after the last laser session. The patients received six laser sessions at 4-month intervals [122]. Wang et al. [119] proposed that there is a six-fold increase in the penetration of cyclosporin solution in 40% ethanol compared to passive diffusion using multiple pulse electroporation. Iontophoresis-assisted delivery can also effectively enhance the skin permeation of cyclosporin, particularly when combined with lecithin vesicles [123]. Table 2 (Ref. [42, 45, 50, 52, 55, 56, 58, 64, 65, 67, 68, 73, 74, 76, 77, 79, 80, 83, 90, 99, 107, 110, 113, 114, 119, 120, 123, 124, 125, 126, 127, 128, 129, 130, 131, 132, 133, 134, 135, 136, 137, 138, 139, 140, 141, 142, 143, 144, 145, 146, 147, 148, 149, 150, 151, 152, 153, 154, 155, 156, 157, 158, 159, 160, 161, 162, 163, 164, 165, 166, 167, 168, 169, 170, 171, 172, 173, 174, 175, 176, 177, 178, 179, 180, 181, 182, 183, 184, 185, 186, 187, 188]) provides an overview of various nano-formulations of drugs used in psoriasis treatment. Detailed information on patented nanotechnology-based drug delivery systems can be found in existing literature.
| Drug | Class | Nanocarrier | Advantages | Reference |
| Clobetasol propionate | SHPG | Cyclodextrin nanosponge (CDNS) | Better payload, controlled release, reduced side effects, improved effectiveness | [113] |
| SLN | High skin permeation and deposition | [124] | ||
| Microsponge | Absence of burst release, sustained release | [114] | ||
| Polymeric nanoparticles | Delayed and high drug release, fewer side effects | [125] | ||
| Desoximetasone | SHPG | Niosomes | Reduced drug dose, dose frequency | [126] |
| Betamethasone | HPG | Hydrogel | Sustained release, cooling effect | [127] |
| Betamethasone 17-valerate | HPG | SLN | Controlled release, epidermis/dermis targeting | [128] |
| Mometasone furoate | HPG | Aspasomes | Prolonged release, skin smoothening | [129] |
| Triamcinolone acetonide | MPG | Transferosomes | Skin penetration and distribution, controlled release | [130] |
| Fluocinolone acetonide | MPG | Microemulsion and fractional laser | High efficiency, sustained release | [131] |
| NLC | Increased skin permeation and retention | [107] | ||
| Polypeptide drug conjugate | Drug permeation in epidermis, controlled release | [132] | ||
| SLN | Increased skin distribution, low systemic absorption | [133] | ||
| Hydrocortisone | LPG | Electrospun nanofibers | 100% drug release, no cytotoxicity | [134] |
| Microemulsion | Retention in the skin and low penetration through the skin | [135] | ||
| NLC | Protection against degradation, no fast-release | [136] | ||
| Calcipotriol | VDA | Liposomes | Increased skin penetration and deposition | [137] |
| Methotrexate | Immunosuppressant | ZnO/Ag hybrid mesoporous microspheres | Self-therapeutic ability, sustained release, enhanced drug delivery | [110] |
| Liquid crystals with lamellar phase | Extensive remodeling of skin microstructure- increased hair follicles, congestion of blood vessels | [138] | ||
| Fractional CO2 laser-assisted delivery for nail psoriasis | Reduced subungual hematoma and pain compared to intralesional injection | [120] | ||
| Transdermal patch | Sustained release, better skin permeation | [139] | ||
| ROS-responsive methotrexate prodrug nanoassemblies microneedle | Significant epidermal penetration, suppression of epidermal proliferation | [140] | ||
| Microporation and iontophoresis | High drug delivery to the skin | [141] | ||
| Nanogel | Enhanced transdermal flux | [142] | ||
| Microemulsion | Cutaneous drug distribution | [143] | ||
| Niosomes | High entrapment efficiency, stable | [144] | ||
| NLC | Increased drug deposition, deep skin penetration, prolonged release | [145] | ||
| Gold nanoparticles | Better anti-inflammatory efficacy, reduced proliferation and differentiation of keratinocytes | [146] | ||
| Liposomes | Improved drug retention and penetration in the skin | [147] | ||
| Deformable liposomes | 3–4-fold increase in skin permeation, high encapsulation efficiency | [148] | ||
| NLC | High skin penetration, fast drug release | [149] | ||
| Cyclosporine | Immunosuppressant | Ethosomes | Enhanced drug deposition | [150] |
| Polymeric micelles | Enhanced cutaneous delivery, deep skin penetration | [151] | ||
| Electroporation | High skin penetration, low systemic absorption | [119] | ||
| Iontophoresis | High skin permeation | [123] | ||
| Niosomes | 59-fold increase in skin deposition | [152] | ||
| Nanoemulsion | No cytotoxicity and skin irritation, increased hydration | [153] | ||
| Microemulsion | Quick cutaneous uptake. Increase skin accumulation | [154] | ||
| Microemulsion | High skin deposition, low distribution to other organs | [155] | ||
| Tacrolimus | Immunosuppressant | Polymeric nanoparticles | Increased skin deposition and retention | [156] |
| Self-assembled lipid-polymer hybrid nanoparticles | Increased drug loading, high cutaneous deposition | [157] | ||
| NLC | High skin retention and permeation | [158] | ||
| Hydrogel | 2-fold increase in skin deposition, high efficiency | [159] | ||
| Microemulsion | Enhanced permeation and deposition, increased cellular uptake | [160] | ||
| Liquid crystalline nanoparticles | Increased skin permeation and retention | [161] | ||
| Polymeric micelles | High deposition in hair follicles | [162] | ||
| Tazarotene | Retinoid derivative | SLN | Improved drug release | [163] |
| Microemulsion | Higher skin deposition and anti-psoriatic activity, no irritation | [164] | ||
| Magnetically responsive nanofiber patch | On-off response-based drug release, minimum skin irritation | [165] | ||
| Microemulsion | High skin deposition, high efficacy | [166] | ||
| Acitretin | Retinoid derivative | Solid dispersion | Increased solubility, increased skin permeation, and hydration | [167] |
| Niosomes | Enhanced skin permeation and deposition, less skin irritation | [168] | ||
| Retinol | Retinoid derivative | Silicon particles | Slow release, protection against degradation | [169] |
| Omiganan | Antimicrobial 0eptide | Liposomes | Better permeation, controlled release | [42] |
| Curcumin | Herbal drug | Nano-emulsion | 4.87-fold increase in skin permeation | [170] |
| Nano-hydrogel | Protection against degradation, increased skin penetration | [171] | ||
| NLC | 3.24-fold improvement in skin permeation, no cytotoxicity | [172] | ||
| Nanogel | Sustained release, good permeation and retention | [173] | ||
| Ethosomes | Targeted delivery, increased skin accumulation | [174] | ||
| Polymeric nanoparticles | Skin penetration, slow release | [175] | ||
| Polymeric nanoparticles | Enhances skin penetration and accumulation, sustained release | [176] | ||
| Nanofibers | Enhanced skin permeation and deposition | [177] | ||
| Erianin | Herbal drug | Mesoporous silica nanospheres | High drug retention, low drug penetration in skin | [56] |
| Capsaicin | Herbal drug | NLC | Increased drug accumulation in skin, no skin irritation | [178] |
| Babchi oil (Psoralea corylifolia) | Herbal drug | Nanogel | 2-folds high efficacy | [179] |
| Microemulsion | Significant skin permeation | [180] | ||
| Methoxsalen | Herbal drug | Microemulsion | 10-fold high deposition in the skin, no skin irritation | [181] |
| Microemulsion | Increased skin penetration and accumulation | [58] | ||
| Celastrol | Herbal drug | Niosomes | Accumulation in skin, enhanced cell uptake | [182] |
| Niosomes | Increased water solubility and permeation | [55] | ||
| Berberine oleate | Herbal drug | Liquid crystalline nanoparticles | 3-fold increase in drug deposition, enhanced permeation | [52] |
| Rosmarinic Acid | Herbal drug | Transethosomes | Increased solubility, sustained release | [50] |
| Oleuropein | Nutraceutical | Microemulsion | Better PASI scoring than the marketed formulation | [64] |
| (-)-epigallocatechin-3-gallate (EGCG) | Nutraceutical | Polymeric nanoparticles | Dose reduction, sustained release | [65] |
| Bilirubin | Endogenous peptide | Polymeric nanoparticles | Attenuation of oxidative stress in keratinocytes, reduced inflammatory cytokines | [77] |
| Diacerein | Endogenous peptide | Niosomes | Cutaneous penetration, high entrapment efficiency | [76] |
| Pentoxifylline | TNF- |
Niosomes | Deep skin penetration and deposition | [83] |
| NLC | High drug retention | [183] | ||
| Etanercept | TNF- |
Ablative fractional laser microporation | Well tolerated, mild local side effects | [67] |
| Er: YAG fractional laser ablation | Drug delivery to the epidermis and dermis | [68] | ||
| Thermosensitive hydrogel | Drug delivery to viable epidermis, stable | [184] | ||
| Tofacitinib citrate | JAK inhibitors | Microneedle arrays | Enhanced intradermal drug deposition | [73] |
| Alantolactone | STAT3 inhibitor | Polymeric nanoparticles | Abrogated keratocyte hyperproliferation and inflammation | [74] |
| Apremilast | PDE4 inhibitor | NLC | Enhanced skin retention by 3-folds, no skin irritation, no cytotoxicity | [185] |
| Nanocrystal | 2-folds increased aqueous solubility, enhanced skin penetration | [186] | ||
| Microemulsion | No cytotoxicity reduced inflammatory cytokines | [157] | ||
| Transethosomes | Better skin permeation, sustained release | [99] | ||
| NLC | Drug deposition in the skin, sustained release | [45] | ||
| SiRNA | IL-6 inhibitor | Polymeric nanoparticle and ablative laser (fractional CO2 laser and a fully ablative Er: YAG laser) | Low toxicity, up to 3.3-fold skin deposition, and 56% IL-6 knockdown in mice | [187] |
| EGF and EGFR knockdown | Gene−nanoparticle conjugate | Excellent penetration in skin, gene knockdown | [79] | |
| Anthralin | Anthracene derivative | Ethosomes | High permeation, low side effects | [80] |
| Liposomes/niosomes | Enhanced skin permeation of liposomes more than niosomes | [90] | ||
| Dendritic nanoparticles | Controlled release, increased skin accumulation | [188] |
Abbrevations: SHPG, super high potency glucocorticoids; CDNS, Cyclodextrin
nanosponge; SLN, solid lipid nanoparticles; HPG, high potency glucorticoids; MPG,
mid potency glucocorticoids; NLC, nanostructured lipid carriers; LPG, low potency
glucocrticoids; VDA, vitamin D analogues; ROS, reactive oxygen species;
TNF-
The combination of innovative therapeutic agents with nanotechnology is transforming psoriasis management. It bridges the gap between drug innovation and effective delivery. By using advancements in nanotechnology, the limitations of conventional formulations, such as poor bioavailability, instability, and systemic side effects can be systematically addressed, paving the way for more effective treatment approaches.
Psoriasis is driven by complex immune system dysregulation, leading to inflammation and the rapid turnover of skin cells. Despite advancements in psoriasis treatment, several needs are still unmet. Traditional topical therapies may lead to poor skin penetration and retention, leading to suboptimal therapeutic outcomes. High costs and accessibility barriers also confine the widespread utilization of these advanced treatments.
Nanotechnology-based therapies are promising, but not without limitations. The industrial scale-up of nanoformulations is challenging due to complex manufacturing processes, regulatory challenges, and the need for precise quality control to ensure reproducibility and stability. Furthermore, the long-term safety and efficacy of nanoformulations require further clinical validation.
Finally, while natural agents and emerging drugs offer potential, their
integration into scalable and clinically validated therapeutics still needs
extensive research and development. For instance, biologics such as monoclonal
antibodies that target specific components of the immune response (like
TNF-
Psoriasis is a chronic inflammatory skin condition that affects approximately 2–3% of the global population, posing significant challenges for both those affected and society as a whole. Although the specific mechanism involved in the development of psoriasis is still unclear, it mainly occurs due to abnormal differentiation and hyperproliferation of keratinocytes, leading to many structural and functional alterations in the skin. Conventional drug delivery systems provide insufficient skin penetration and retention and can also lead to systemic drug leaching. Therefore, nanoenabled drug delivery is required. With the increase in the understanding of the pathophysiology of psoriasis and various advances in nanotechnology-based drug delivery systems, it is possible to deliver the drug at the target site and enhance drug permeation and retention in the skin. The potential of many existing and new drug molecules is being explored, and various target sites are being identified. The exploration of both existing and novel drug molecules, along with the identification of new target sites, is underway and has shown potential. Combining possible therapeutic agents and nanotechnology could revolutionize the treatment of psoriasis, offering more efficient management and potentially shortening the treatment duration. Such advancements are likely to improve the quality of life of psoriasis patients. Future research should focus on developing disease-specific drugs and formulation techniques that can be scaled up to the industrial level while remaining safe, cost-effective, and providing more social benefits.
JU, NH, SP, TK, MN, KYT and MNA designed the study. TK, MN, and KYT wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for this work through a Large Research Project.
The work was supported by the Deanship of Research and Graduate Studies at King Khalid University for funding this work through a Large Research Project under grant number RGP2/580/45.
Sudeep Pukale is a consultant/proctor of Lupin Research Park, the judgments in data interpretation and writing were not influenced by this relationship. All other authors have no conflicts relevant to the contents of this paper to disclose.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.