1 Department of Biology, College of Science, Jazan University, P.O. Box. 114, 45142 Jazan, Kingdom of Saudi Arabia
2 Faculty of Pharmacy, Cairo University, 12613 Cairo, Egypt
3 Biotechnology Department, Faculty of Science, Cairo University, 12613 Giza, Egypt
4 Department of Pharmacology and Toxicology, College of Pharmacy, Jazan University, 45142 Jazan, Kingdom of Saudi Arabia
Abstract
Allergic disorders rising in prevalence globally, affecting a substantial proportion of individuals in industrialized nations. The imbalance in the immune system, characterized by elevated allergen-specific T helper 2 (Th2) cells and immunoglobulin E (IgE) antibodies, is a key factor in allergy development. Allergen-specific immunotherapy (AIT) is the only treatment capable of alleviating allergic symptoms, preventing new sensitizations, and reducing asthma risk in allergic rhinitis patients. Traditional AIT, however, faces challenges such as frequent administration, adverse effects, and inconsistent patient outcomes. Nanoparticle-based approaches have emerged as a promising strategy to enhance AIT. This review explores the utilization of nanoparticles in AIT, highlighting their ability to interact with the immune system and improve therapeutic outcomes. Various types of nanoparticles, including polyesters, polysaccharide polymers, liposomes, protamine-based nanoparticles (NPs), and polyanhydrides, have been employed as adjuvants or carriers to enhance AIT’s efficacy and safety. Nanoparticles offer advantages such as allergen protection, improved immune response modulation, targeted cell delivery, and reduced side effects. This review provides an overview of the current landscape of nanoparticle-based allergen immunotherapy, discussing its potential to revolutionize allergy treatment compared to traditional immunotherapy.
Graphical Abstract
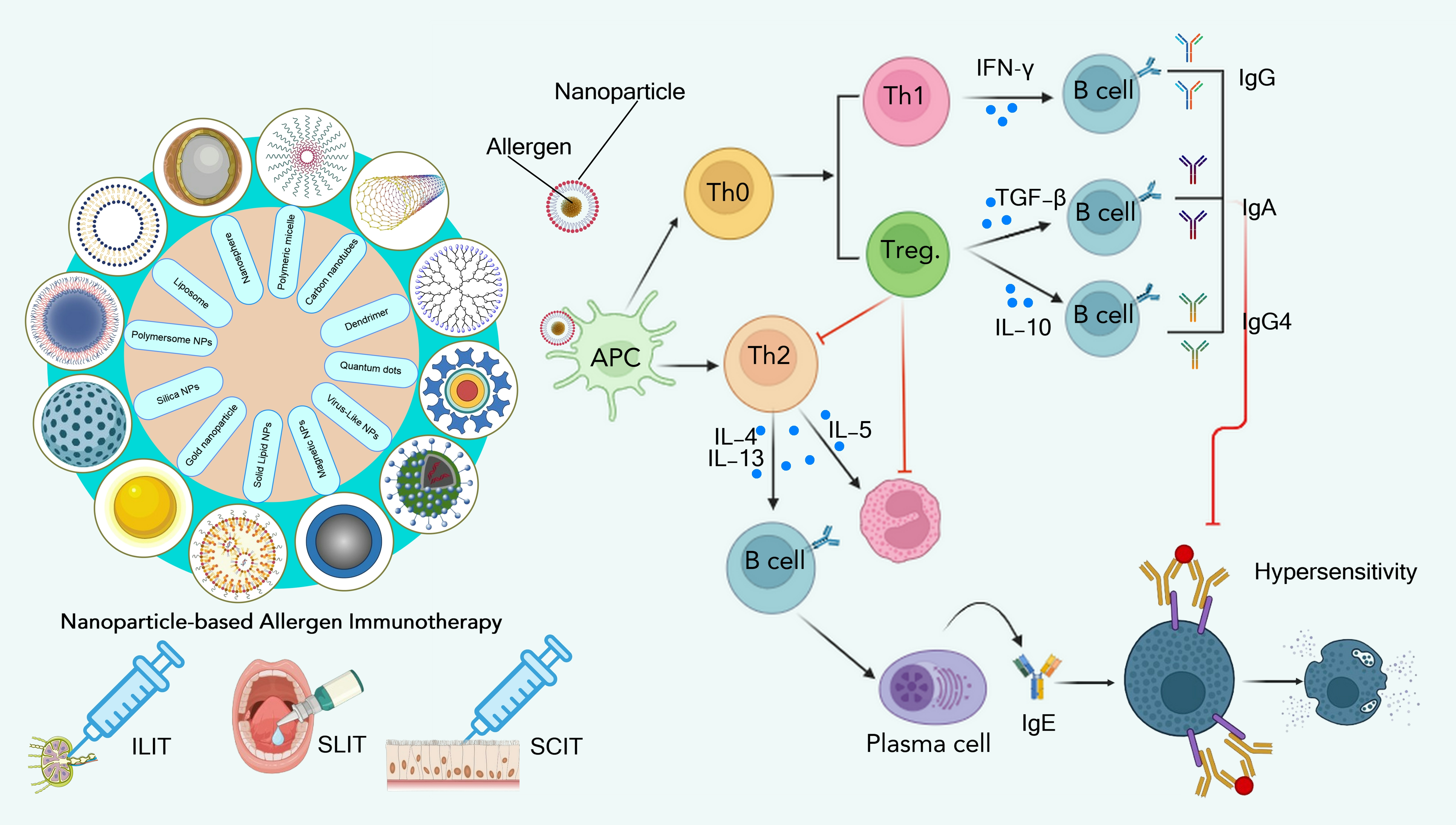
Keywords
- allergy
- immunotherapy
- allergic disease
- anaphylaxis
- nanoparticle
In the context of global health, the prevalence of allergic conditions is on the rise, posing a significant challenge. Industrialized nations witness a substantial impact, with over 25% of their population grappling with the consequences of allergies. This statistic emphasizes the critical need for advanced healthcare solutions to address this burgeoning issue [1, 2]. Allergic reactions are closely associated with an increased presence of allergen-specific CD4+ T helper 2 (Th2) cells in affected individuals. These Th2 cells release specific cytokines, including interleukin (IL)-4, IL-5, and IL-13, which play a crucial role in driving the allergic response. As a result, the body produces elevated levels of allergen-specific immunoglobulin E (IgE) antibodies, contributing to the manifestation of allergy symptoms [3, 4]. The imbalance in the immune response, characterized by the overproduction of certain cytokines and allergen-specific antibodies, is a critical factor in the development of allergic symptoms. Allergen-specific immunotherapy (AIT) stands out as a unique and effective treatment approach in this regard. AIT has proven to be the only therapeutic strategy capable of significantly improving allergic symptoms, preventing new allergen sensitizations, and reducing the risk of asthma in patients suffering from allergic rhinitis. This treatment modality addresses the underlying immune dysregulation, offering relief and long-term benefits to allergic individuals [5]. AIT works by inducing allergen-specific tolerance through various mechanisms, including changes in memory type allergen-specific T- and B-cell responses, suppression of allergen-specific IgE, and increased production of Immunoglobulin G1 (IgG1) and Immunoglobulin G4 (IgG4) antibodies [2, 6]. However, traditional AIT approaches have some limitations, such as the need for frequent administration, the risk of adverse reactions, and the variable efficacy across patients [7]. The field of immunotherapy has witnessed a promising advancement with the introduction of nanoparticles as a novel strategy in recent years. Nanoparticles have gained attention for their potential to overcome the limitations associated with traditional immunotherapy approaches. By utilizing nanoparticles (NPs), researchers aim to enhance both the safety and efficacy of immunotherapy treatments. This innovative approach involves designing nanoparticles to interact with the immune system, harnessing their immunomodulatory properties to achieve more targeted and effective therapeutic outcomes. The application of nanoparticles in immunotherapy holds great promise for improving patient care and managing various immune-related disorders [8]. Nanoparticles possess unique capabilities, allowing for their customization to engage with the immune system’s processes. This tailored design harnesses the immunomodulatory attributes of nanoparticles, presenting a novel therapeutic strategy with significant potential [9]. Various types of nanoparticles (NPs) have been extensively researched and developed to enhance the performance and safety of allergen-specific immunotherapy (AIT). These NPs, such as polyesters, polysaccharide polymers, liposomes, protamine-based nanoparticles, and polyanhydrides, serve as adjuvants or carriers, improving the overall efficacy of AIT. The diverse range of nanoparticle categories contributes to the advancement of immunotherapy by providing targeted and controlled delivery systems for allergens, thereby optimizing therapeutic outcomes [10]. NPs provide multiple benefits in the context of allergen-specific immunotherapy (AIT), particularly in the case of oral immunotherapy (OIT). One significant advantage is their ability to protect the encapsulated allergen by creating a barrier against proteolytic enzymes, ensuring the allergen’s integrity. This protection is crucial for OIT, as the allergen-containing vaccine must survive the challenging environment of the gastrointestinal tract during its journey to induce immune tolerance [4, 11]. The unique properties of nanoparticles make them highly suitable as prospective adjuvants in allergen-specific immunotherapy (AIT). Their small size facilitates efficient interaction with immune cells, while surface modification capabilities allow for targeted delivery and enhanced immunomodulatory effects. Additionally, biodegradability and biocompatibility ensure the safety and compatibility of nanoparticles with biological systems, making them an ideal candidate to improve AIT outcomes [4]. Nanoparticles can enhance the immune response to the allergen, promoting a desirable T helper 1 (Th1)/Treg response and suppressing the detrimental Th2 response [12, 13]. Nanoparticles can target specific cell populations, such as antigen-presenting cells, to improve the efficacy of allergen presentation and modulation of the immune response. Importantly, nanoparticle-based AIT formulations have shown the potential to reduce side effects, such as anaphylactic reactions, by decreasing IgE-mediated signaling and histamine release, thereby enhancing patient safety [10]. This comprehensive review will discuss the current state of nanoparticle-based allergen immunotherapy, including the various types of nanoparticle use.
Allergies are heightened immune responses to usually benign environmental substances known as allergens. There are various types of allergies, including respiratory allergies like hay fever (allergic rhinitis), caused by exposure to airborne allergens like pollen, dust mites, mold, and pet dander. Symptoms include sneezing, runny nose, nasal congestion, and itchy/watery eyes [14, 15, 16, 17]. Food allergies are immune reactions to specific proteins in foods like peanuts, tree nuts, eggs, milk, soy, wheat, fish, and shellfish [18, 19]. Symptoms can include hives, swelling, digestive issues, and, in severe cases, anaphylaxis [20, 21, 22]. Skin allergies, such as eczema (atopic dermatitis), are caused by contact with allergens that trigger inflammation and skin irritation. Symptoms include redness, itching, and rashes [15, 17, 23]. Drug allergies are adverse immune reactions to medications, which can range from mild rashes to life-threatening anaphylaxis [15, 24]. Allergic responses to insect bites or stings, for instance, from bees, wasps, or mosquitoes, may trigger swelling, rashes, and potentially life-threatening anaphylaxis in susceptible people [25, 26].
The most common allergens that trigger allergic reactions include pollen from trees, grasses, and weeds, which significantly contribute to seasonal respiratory allergies like hay fever [16, 17, 25]. Additionally, house dust mites and their waste products are prevalent indoor allergens that can provoke asthma and rhinitis symptoms [27]. Proteins found in the skin, saliva, and urine of pets like cats and dogs can cause allergic reactions in many people [17, 28]. Indoor and outdoor mold exposure can lead to allergic rhinitis, asthma, and other respiratory symptoms [17, 29]. The most common food allergens are peanuts, tree nuts, shellfish, fish, eggs, milk, soy, and wheat [21, 22]. Stings or bites from insects like bees, wasps, hornets, fire ants, and mosquitoes can trigger severe allergic reactions in susceptible individuals [30]. Natural rubber latex, found in many products like gloves and balloons, can cause contact dermatitis and other allergic responses [31]. The prevalence and specific types of allergens can vary by geographic region, climate, and other environmental factors [17, 32, 33].
The process begins when an allergen is encountered by antigen-presenting cells
(APCs), such as dendritic cells. These cells process the allergen and present its
peptides to naive T cells in the lymph nodes, leading to their differentiation
into T helper 2 (Th2) cells. Th2 cells secrete cytokines, notably interleukin-4
(IL-4), interleukin-5 (IL-5), and interleukin-13 (IL-13). IL-4 and IL-13 promote
class switching in B cells to produce immunoglobulin E (IgE), while IL-5 is
crucial for the activation and survival of eosinophils [34, 35]. The IgE
antibodies bind to high-affinity IgE receptors (Fc
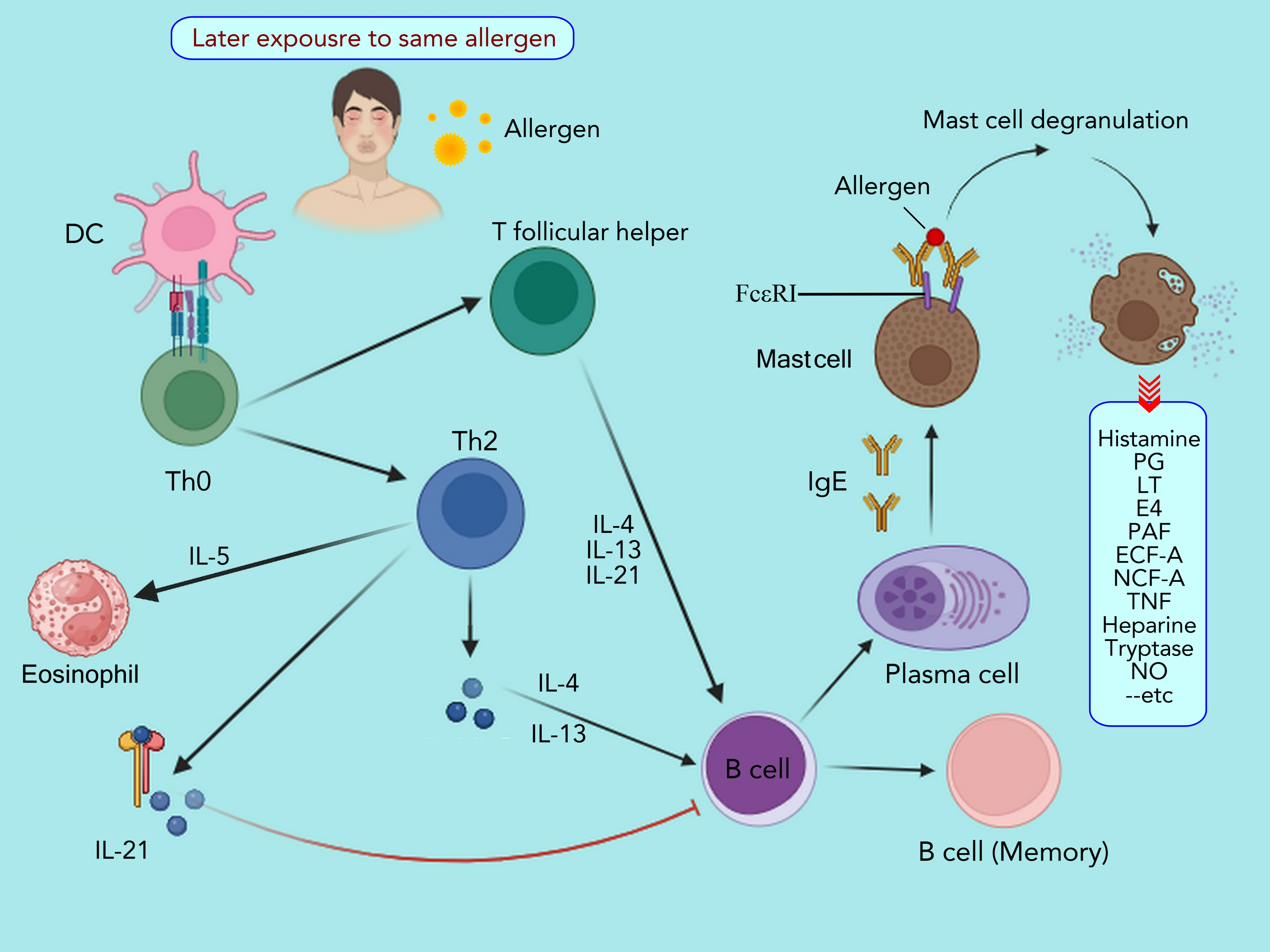 Fig. 1.
Fig. 1.
Mechanism of allergic disease. The process initiates
with dendritic cells (DCs) capturing allergens. These DCs then move to the
draining lymph nodes, where they present peptides derived from the allergens on
major histocompatibility complex (MHC) class II molecules to naive T cells. With
the aid of various co-stimulatory signals and cytokines, these T cells
differentiate into Th2 cells. Th2 cells primarily release type 2 cytokines that
contribute to allergic inflammation, such as IL-4, IL-5, and IL-13. Additionally,
T follicular helper (TFH) cells produce IL-21, IL-4, and IL-13, which
facilitate B-cell class switching to IgE, plasma cell maturation, and the
generation of allergen-specific IgE. The IgE antibodies released by plasma cells
attach to Fc
Allergen-specific immunotherapy (AIT) aims to modulate the underlying pathophysiological mechanisms of allergic reactions. The allergic response is initiated when an allergen is endocytosed by the dendritic cells in the airway epithelium [43]. These antigen-presenting cells then migrate to the nearby secondary lymphoid organs, presenting the allergen to naive T helper (Th0) cells [44]. The Th0 cells then differentiate into Th2 cells, which secrete a specific profile of interleukins, including IL-4, IL-5, IL-13, IL-25 and IL-33. The cytokines secreted by Th2 cells are fundamental to the allergic response and production of IgE, influencing various aspects of the immune reaction and contributing to the clinical manifestations of allergic diseases. Understanding these cytokines provides insights into potential therapeutic targets for managing allergic conditions [45, 46] (Fig. 2a, Ref. [9]).
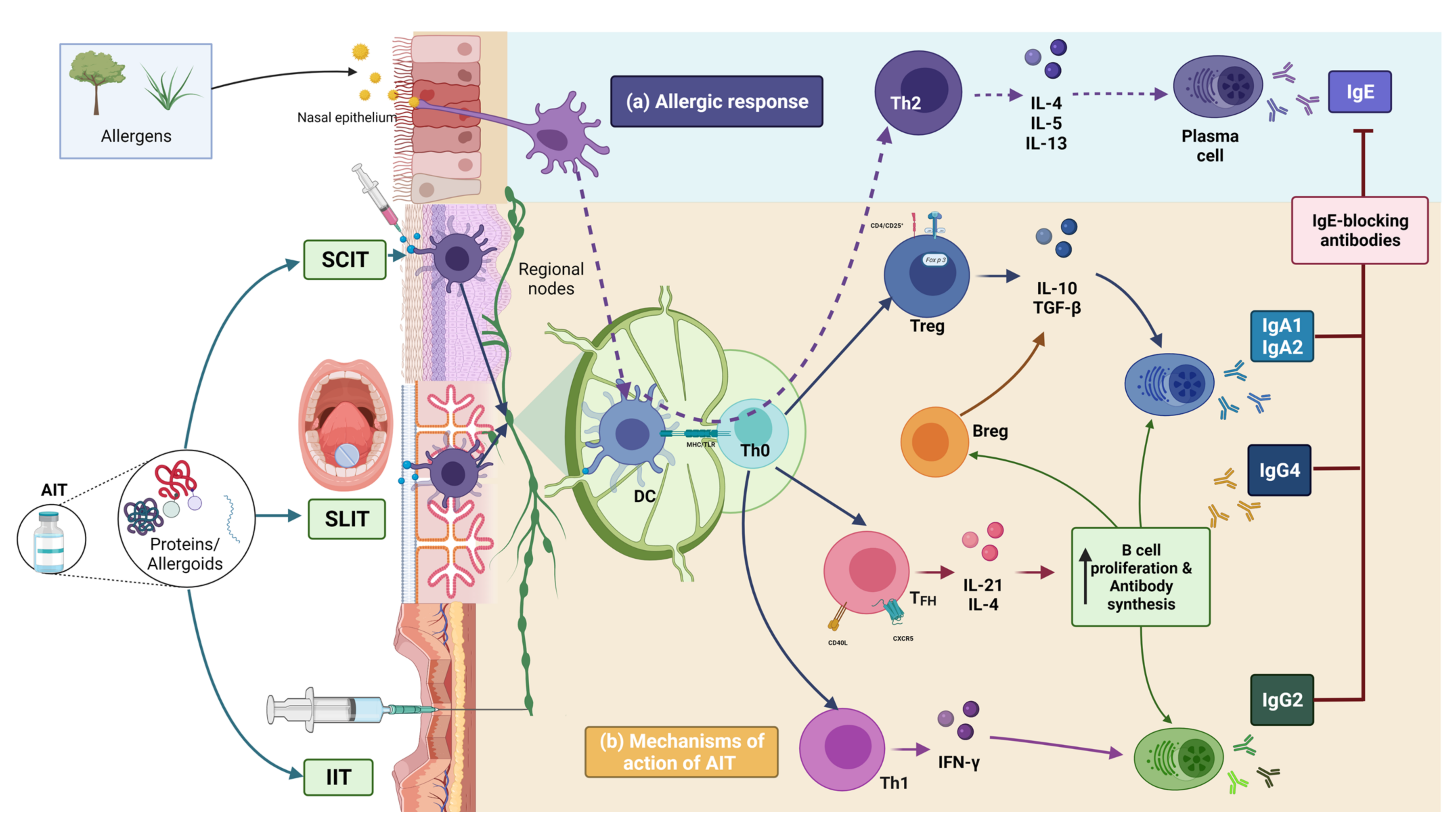 Fig. 2.
Fig. 2.
Mechanisms of Allergic Reactions and
Immunotherapy: From Allergen Recognition to Antibody Response. (a) The allergic
reaction initiates with the allergen being captured by airway epithelial
dendritic cells (DCs) through endocytosis. These DCs then migrate to nearby
secondary lymphoid organs, such as lymph nodes, where they present the antigen to
naive T helper cells (Th0). Subsequently, Th0 cells differentiate into Th2 cells,
which release specific interleukins that promote the production of
allergen-specific IgE antibodies, triggering an allergic response. In contrast,
(b) allergen-specific immunotherapy, including subcutaneous immunotherapy (SCIT),
sublingual immunotherapy (SLIT), and intralymphatic immunotherapy (IIT),
introduces the antigen in various forms (peptides, recombinant, or protein
complexes) to stimulate the differentiation of naive T cells into distinct
subsets. These subsets include Th1 cells, which produce interferon-gamma
(IFN-
Subcutaneous immunotherapy (SCIT) is an allergen immunotherapy (AIT) that involves injecting allergens under the skin to treat allergic diseases. This method is commonly used to manage conditions such as allergic rhinitis, asthma, and other respiratory allergies [47, 48]. SCIT exposes the body to small amounts of allergens, which helps desensitize the immune system over time [49, 50].
The mechanism of action of Subcutaneous Immunotherapy (SCIT) entails a complex
series of interactions within the immune system designed to promote tolerance to
allergens. Initially, the allergen, usually provided as an extract, is injected
under the skin and taken up by local immune cells [51]. Following this exposure,
antigen-presenting cells (APCs), particularly dendritic cells in the skin,
capture and process the allergen [52, 53]. This processing involves breaking down
the allergen into smaller peptides presented on the APC surface via Major
Histocompatibility Complex (MHC) class II molecules [54]. The interaction between
T cells and these allergen-derived peptides occurs through T cell receptors
(TCRs), leading to T cell activation and proliferation [55]. The polarization of
the immune response is significantly influenced by the cytokines produced by
APCs, such as interleukin-10 (IL-10) and transforming growth factor-beta
(TGF-
Sublingual immunotherapy (SLIT) is a form of allergen immunotherapy that involves administering allergen extracts under the tongue rather than through subcutaneous injections, as in traditional subcutaneous immunotherapy (SCIT) [65]. SLIT has emerged as an alternative to SCIT, offering potential advantages in terms of safety, convenience, and patient acceptance [66].
The mechanism of action of Sublingual Immunotherapy (SLIT) involves a series of
complex interactions within the immune system. Below is a detailed summary of the
critical processes involved: The allergen is introduced under the tongue and
absorbed through the oral mucosa [67]. This route allows direct interaction with
the immune system without needing injection [68]. Antigen-presenting cells (APCs)
capture the allergen, particularly Langerhans cells, in the oral mucosa [69].
These cells are abundant in IgE receptors, enhancing their ability to efficiently
uptake allergens [70, 71]. Once captured, APCs process the allergen, breaking it
into smaller peptides [72, 73]. These peptides are then presented on the surface
of the APCs using major histocompatibility complex (MHC) class II molecules,
which are crucial for T cell recognition [74, 75]. T cells interact with the
allergen peptides presented by APCs through their T cell receptors (TCRs) [76, 77]. This interaction leads to T cell activation and subsequent proliferation,
essential for mounting an immune response [78, 79]. Several factors influence the
polarization of the immune response: APCs secrete cytokines such as
interleukin-10 (IL-10) and transforming growth factor-beta (TGF-
Intralymphatic immunotherapy (ILIT) represents a novel approach to treating allergies, particularly allergic rhinitis [87]. This method aims to enhance the efficacy and safety of allergen immunotherapy by delivering allergens directly into the lymphatic system [3, 88]. Conventional allergen-specific immunotherapy (AIT) requires multiple injections over several years, which can be inconvenient and lead to poor patient compliance [89, 90]. ILIT offers a more efficient alternative, requiring fewer injections and potentially achieving faster desensitization [91, 92, 93]. This therapy particularly appeals to patients who struggle with the lengthy protocols of traditional immunotherapy [88].
ILIT injects allergens directly into a lymph node, a critical site for immune response modulation [94]. This method leverages the lymphatic system’s natural role in immune function, allowing for a more targeted and efficient immune response [95]. By delivering allergens directly to the lymph nodes, ILIT stimulates a robust immune response, promoting the production of specific antibodies (IgG) that can block the effects of IgE, the antibody responsible for allergic reactions [92, 96]. ILIT enhances the activation of regulatory T-cells, which play a crucial role in developing immune tolerance [97]. This shift in immune response can lead to long-term tolerance to allergens, reducing symptoms even after treatment cessation. Compared to traditional subcutaneous immunotherapy, ILIT has been associated with fewer systemic allergic reactions, as the allergen is introduced in a controlled manner directly into the lymphatic system rather than circulating through the bloodstream [98] (Fig. 2b).
Nanotechnology has been used in immunotherapy to improve effectiveness and reduce side effects, particularly in treating cancer and infectious diseases. In the last thirty years, a range of polymers have been employed to produce nanoparticles (NPs) that can transport proteins and nucleic acids for medicinal intentions [99, 100]. NPs are regarded as the primary element of nanomaterials (NMs), possessing distinct physical boundaries and at least one dimension within the nano-scale range [101]. These particles come in different forms with varying chemical compositions and physical properties, such as size, solubility, morphology, surface chemistry, and charge [102]. These properties can be adjusted to suit specific biomedical needs. The biocompatibility of NPs is crucial for their biological applications and is heavily influenced by their physicochemical properties. Moreover, attributes such as biodistribution, mechanisms of cellular uptake, and potential toxicity of nanoparticles are predominantly influenced by their surface properties and size [103].
Recent research has shown that nanoparticles (NPs) have the potential to positively impact the way allergenic extracts or single allergens interact with the immune system. The development of safer and more potent vaccines and immunosuppressive agents is a direct outcome of harnessing this innovative approach. NPs can act as adjuvants, stimulating the immune system to respond more effectively to vaccine antigens. The size of the particles can be adjusted to improve antigen delivery, and their chemical nature, solubility, and shape can protect antigens from degradation and allow for targeted delivery to specific tissues or cells. This strategy holds the potential to revolutionize treatment by significantly reducing the required antigen or drug dosage while maintaining optimal efficacy, particularly in oral or mucosal administration where antigens are vulnerable to degradation.
Furthermore, NPs can specifically target immune cells by incorporating ligands
or agonists, enhancing their ability to engage in adjuvant activities [104]. By
protecting antigens at the administration site and allowing for gradual release,
NPs can create a “depot” effect, maintaining stability and conformation of the
antigens. Additionally, the repetitive spatial organization of antigens within
NPs can further enhance their therapeutic potentially conjugating nanoparticles
(NPs) with specific ligands of Toll-like receptors (TLRs) or other receptors
present on antigen-presenting cells (APCs), it becomes possible to not only
target these cells precisely but also modulate their functional response. This
targeted approach can elicit a more tailored and effective immune reaction,
offering a promising strategy for various immunotherapeutic applications. The
ability to influence APCs behavior contributes to the development of customized
immune responses, which is essential for treating immune-related disorders [105].
Nanoparticles (NPs) not only promote immune tolerance but can also be utilized to
suppress or hinder undesired immune responses linked to autoimmune conditions and
allergies. This is achieved by directly exerting immunosuppressive effects on
immune cells such as antigen-presenting cells (APCs), T cells, and B cells or by
enhancing the delivery of immunosuppressive agents through increased solubility
and bioavailability. The elevation of cytokines like TGF-
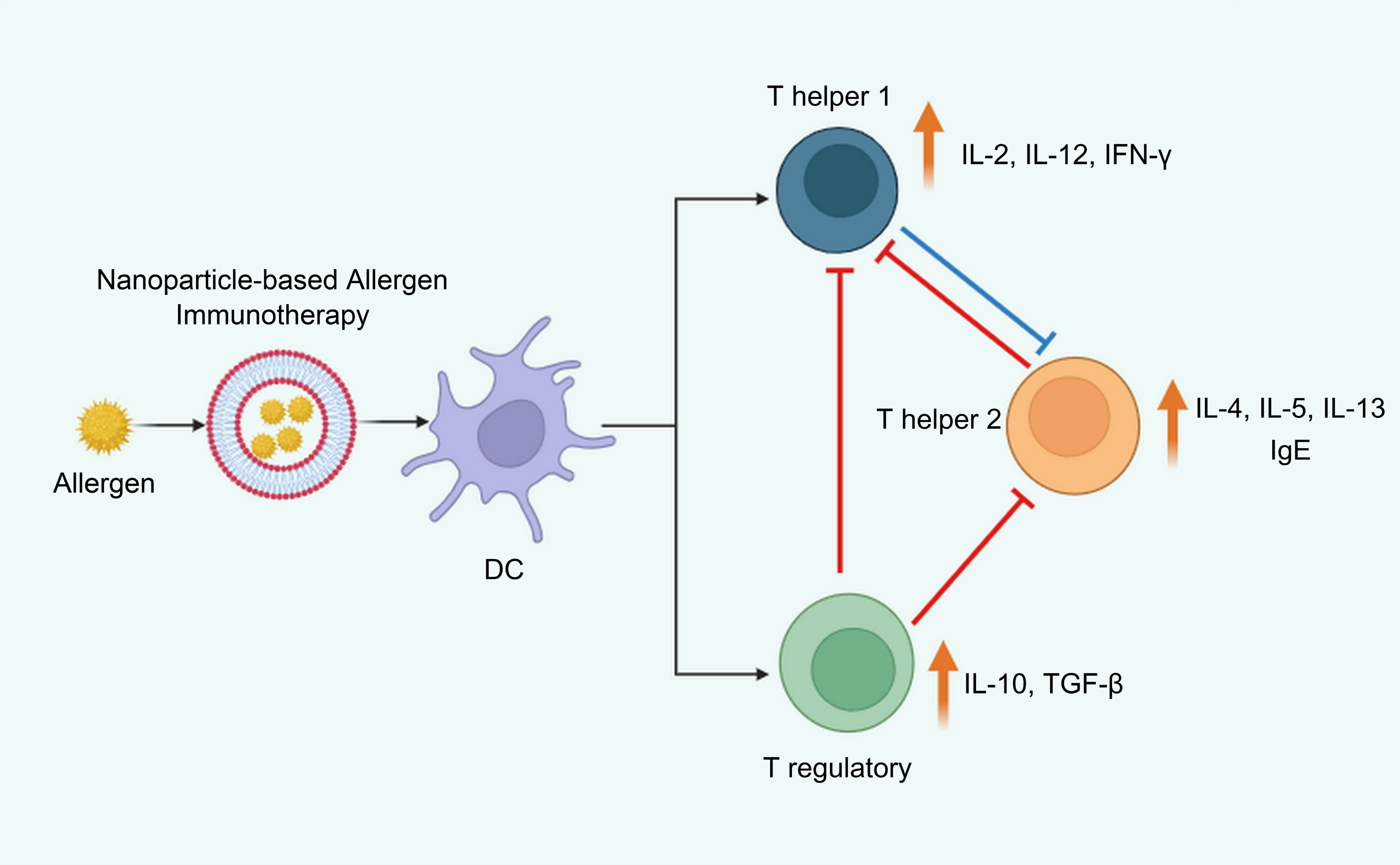 Fig. 3.
Fig. 3.
Immunomodulation allergic conditions through the utilization of nanoparticle-infused vaccines. Immune system alterations post-effective allergen-specific immunotherapy are represented by whole arrows; truncated arrows signify suppressive impacts. Shifting allergic TH2 responses towards the promotion of T regulatory cell generation and/or TH1 stimulation is illustrated. Created with BioRender.com.
An illustration of this concept is demonstrated by spherical fullerene
nanoparticles, which exhibit immunosuppressive properties in cases of
anaphylaxis. These nanoparticles exhibit the ability to mitigate IgE
receptor-mediated signaling, hindering the degranulation of human mast cells and
basophils in vitro. Furthermore, in vivo study demonstrate
their efficacy in preventing histamine release and reducing body temperature
during allergen exposure in a mouse anaphylaxis model, showcasing their potential
in managing allergic reactions [107]. Moreover, nanoparticles can be specifically
engineered to engage with the immune system, utilizing their immunomodulatory
properties for therapeutic purposes. This suggests that novel nanoparticle
formulations involving allergens could pave the way for alternative therapeutic
strategies or routes of administration [108]. Additionally, recent advancements
have introduced nanoparticle-based approaches in allergy diagnosis, showcasing
that encapsulating hydrophobic allergens within poly-
Nanoparticles have emerged as a promising approach for delivering allergens in immunotherapy, garnering significant attention due to their potential as adjuvants in allergy treatment. The incorporation of allergens into these delivery systems has been shown to significantly impact the effectiveness of allergy vaccines, highlighting the importance of this innovative approach in allergen immunotherapy [110].
Among the various types of nanoparticles in Table 1 (Ref. [110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120, 121, 122, 123]), biodegradable polymeric nanoparticles have been extensively studied for their potential in allergen delivery systems. These colloidal carriers, ranging in size from 10 to 1000 nm, can be categorized into nanocapsules and nanospheres based on their structure and drug delivery mechanisms [124]. The choice between these nanoparticle types depends on the specific preparation method, showcasing the versatility and customization options available in nanoparticle-based allergen-delivery systems. Categorizing NPs for allergen complex preparation based on chemical composition may involve polyesters with adjustable properties. Polysaccharide polymers like chitosan are favored for their ability to target mucosal tissues and boost mucosal immunity. Carbohydrate-based particles, liposomes, protamine-based nanoparticles, and polyanhydrides offer customization options for immunogenicity and delivery routes [125].
| Nanomaterials/nanoparticles | Definition | Advantage | Example of application | References |
| Polyesters | Polymeric nanoparticles are used as carriers for vaccines, allowing for precise delivery of antigens or adjuvants. | Biocompatibility, safety, biodegradability. | Poly (glutamic acid) (PGA) and poly (lactic-co-glycolic acid) (PLGA) as biodegradable synthetic polymer nanocarriers. | [111, 112, 113] |
| Polysaccharide polymers and carbohydrate-based particles | Polymers readily produced from natural sources include poly(d-glucosamine) and chitosan. This particular polysaccharide is notably abundant and is extracted from shrimp shells and other crustaceans. | Reduced production expenses, compatibility with biological systems, ability to decompose naturally, and lack of harmful effects. | Chitosan carries a cationic charge that can boost endocytosis and acts as an adjuvant by promoting the maturation of dendritic cells (DCs). | [114, 115] |
| Liposomes | Spherical vesicles are made of one or more layers of phospholipids that create water-filled spaces. This unique structure helps trap and transport both water-loving and water-repelling molecules. | Improve how well the protein dissolves and is absorbed, ensure it stays stable inside the body, and target the drug to the right place. | Oligomannose-coated liposomes, OMLs, made from mannotriose and dipalmitoyl phosphatidylethanolamine (Man3-DPPE), can trigger a robust Th1 immune response in mice, leading to high IFN- |
[116, 117, 118, 119] |
| Protamine-based NPs | Biodegradable peptides rich in arginine, around 4 kDa in size, have been used in human medicine for many years. | Protamine-based nanoparticles can effectively deliver their contents into the nucleus, enhancing the effectiveness of targeted gene therapy. | DNA or RNA oligonucleotides can form complexes with Protamine nanoparticles (NPs) via electrostatic interactions, leading to highly stable and biocompatible structures. | [120, 121] |
| Polyanhydrides | Amphiphilic polyanhydride nanocarriers have special abilities to control immune responses and are also considered safe. | The polymer composition can be adjusted to control release kinetics, antigen retention, and adjuvant effect, especially by focusing on the level of hydrophobicity. | Gantrez AN-119 is a unique copolymer synthesized from methyl vinyl ether and maleic anhydride, offering a versatile platform for allergen-based therapies. Its reactive nature towards amino groups facilitates the efficient loading and conjugation of various proteins, including allergens, making it a valuable tool in the development of allergen-specific immunotherapies. | [122] |
| Poly (gamma-glutamic acid) ( |
It is a substantial protein produced by specific strains of Bacillus bacteria. It is composed of |
The amphiphilic nature of the hydrophobically modified |
Ovalbumin (OVA) was effectively enclosed in the nanoparticles without causing harm to HL-60 cells. | [110, 123] |
Research has revealed the potential of poly (gamma-glutamic acid)
(
In the pursuit of effective allergen delivery systems, poly(vinylpyrrolidone) (PVP) nanoparticles have shown great promise. A notable study by Madan et al. [127], demonstrated the successful entrapment of antigens from Aspergillus fumigatus, a fungus associated with various allergic diseases. This research revealed that PVP nanoparticles provided sustained release, maintaining IgG antibody levels for an extended period of 12 weeks in male BALB/c mice, compared to just 7 days with free antigen immunization. Furthermore, the nanoparticles exhibited lower IgE levels compared to free allergens, suggesting their potential in reducing allergic reactions and providing a more controlled immunological response [106].
Studies are underway to investigate the use of non-biodegradable nanoparticles, crafted from diverse materials like latex, gold, silica, or polystyrene, as antigen carriers to enhance immune responses [egs. [128]]. These particles can present the antigen to the immune system for longer, enhancing immunogenicity [129]. Polystyrene nanoparticles provide technological advantages in immunotherapy due to their customizable surface functional groups. These groups enable effective binding with various antigens, ensuring a robust connection. When the antigen is chemically linked to the nanoparticle, it elicits stronger cellular and humoral immune responses compared to instances where the antigen is merely absorbed, highlighting the significance of this design in enhancing immunological reactions [130]. Latex particles have been demonstrated to exhibit superior presentation by MHC-Class I molecules compared to soluble antigens or those presented through MHC-Class II molecules. Additionally, gold nanoparticles have shown promise in enhancing the delivery of DNA vaccines into cells, further expanding the potential applications of nanoparticles in immunotherapy and gene delivery systems [131]. Recent studies have revealed that combining gold nanoparticles with alum can significantly boost the immune response to specific antigens, suggesting a synergistic effect. However, it is important to note that non-biodegradable particles generally demonstrate lower efficiency in antigen presentation compared to their biodegradable counterparts, which may impact their overall effectiveness in immunological applications [132, 133, 134].
Solid lipid nanoparticles (SLNs) are considered to be more advanced
nano-delivery systems than vesicular systems [135]. SLNs were created and hold
potential for extended stability, drug targeting, and controlled release [136, 137]. SLNs provide several significant advantages including improved solubility,
minimized side effects, enhanced drug bioavailability, ability to encapsulate
both hydrophilic and hydrophobic drugs, improved stability, specificity, and
potential for mass production [138, 139]. Herein, several studies demonstrate the
ability of lipid nanoparticles to be used in the industry as a method of
delivering allergens; Nassimi et al. [140] showed how phospholipid- and
triglyceride-based SLN in a 30:70 ratio can be used for potential pulmonary
purposes. In addition to evaluating cytokine activation, they also evaluated the
toxicity profile of these SLNs using in vitro, ex vivo, and
in vivo models. Upon nebulizing the SLN into mice, researchers observed
no activation of the pro-inflammatory cytokines keratinocyte-derived chemokine (KC) and TNF-
Current clinical trials explore nanoparticle formulations, including those combining allergens with immune-modulating agents to enhance safety and efficacy. One notable study highlighted the use of decorated nanoparticles that target mast cells, pivotal in allergic reactions. This dual-action approach delivers the allergen and inhibits the mast cells’ activation, potentially preventing severe allergic responses such as anaphylaxis [144]. The findings from these trials suggest that nanoparticle-based therapies could significantly reduce the risk of adverse effects commonly associated with traditional allergen immunotherapy. Nanotechnology is an emerging technology due to its ability to create innovative products with unique characteristics and functionalities. It holds significant promise in a wide range of applications and has a substantial influence on healthcare [145].
Furthermore, using nanoparticles in allergy immunotherapy encompasses food allergies and respiratory allergens. To improve patient compliance and treatment outcomes, nanoparticles can enable the precise delivery of allergens through different oral, sublingual, and intranasal routes [3]. The capacity to modify nanoparticle properties, including size, surface charge, and composition, enables customization according to individual patient profiles and particular allergies and sensitivities.
Despite the encouraging progress, obstacles persist in allergy immunotherapy based on nanoparticles. To fully comprehend and optimise the interactions between nanoparticles and immune cells, it is necessary to consider the intricate nature of the immune system. Current research is primarily concerned with clarifying these interactions to guarantee the safety and efficacy of these modalities [146]. Conducting comprehensive clinical trials to assess nanoparticle-based therapies’ long-term effects and durability is crucial as the area advances.
The unique physicochemical features and immunomodulatory capabilities of nanoparticles (NPs) have proven tremendous promise in boosting allergy immunotherapy (AIT) [4]. The efficient penetration of tissues by short nanoparticles enhances the accessibility of allergens to lymph nodes and blood arteries. Prior study has shown that amorphous silicon dioxide nanoparticles have more immunomodulatory properties than microparticles in allergic contact dermatitis models. Furthermore, mono-dispersal silica nanoparticles have demonstrated effectiveness in penetrating the epidermis and localizing lymph nodes, validating their effectiveness in anti-inflammatory therapy (AIT) [147]. The attraction of nanoparticles (NPs) is influenced by their size, shape, and surface charge. Small, negatively charged NPs demonstrate immunosuppressive effects in models of skin allergies. Significantly, biocompatible polymeric nanoparticles can mitigate the adverse effects of existing treatments, providing a reassuring prospect for future therapies [148]. Their surface features considerably influence the cellular absorption of nanoparticles (NPs), whereas macrophages demonstrate a greater preference for globular particles. Polylactic-co-glycolic acid particles functionalized with lectin are suggested as effective platforms for oral anionic immunotherapy (AIT), improving the absorption of allergens and increasing their resistance to breakdown in the gastrointestinal tract [149].
The depot effect is classified as a fundamental mechanism for stimulating immunological tolerance. Antigen persistence refers to the extended release of antigens, resulting in heightened exposure of immune cells and potentially causing immunomodulation [150]. The phenomenon above enables a decrease in the required therapeutic dosage. Within the realm of nanoparticles (NPs), allergens can be enclosed within carriers, and their release can be customized by altering the surface and polymer composition of the particles. Chitosan-coated polylactic-co-glycolic acid nanoparticles (NPs) exhibit a refined and regulated release of active components compared to uncoated NPs [151]. An investigation by Henriksen-Lacey et al. [152], also highlighted the immunologically enhancing depot impact of a liposome vaccination that includes antigens specific to TB. Therefore, the capacity of nanoparticles to form depots may provide several therapeutic benefits.
Nanoparticles (NPs) can protect antigens by encapsulation, sheltering them from
protease enzymes in the body, which is critical for oral immunotherapy (OIT),
particularly in overcoming the severe circumstances of the gastrointestinal
system [11]. OIT is a promising technique for treating food allergies, showing
success in 60–80% of instances tested. Research by Brotons-Canto et
al. [153], indicated that mannosylated NPs are acceptable for oral immunotherapy
against peanut allergies in mice, employing a new polymer system developed by
attaching mannosamine to a polyanhydride backbone. Similarly, a study has
demonstrated that polyanhydride NPs confer considerable benefits for peanut
allergy treatment by boosting Treg and Th1 responses while lowering Th2
activation. Srivastava and coworkers [154] also reported effectiveness using
CpG-coated polylactic-co-glycollic acid NPs in mouse models, leading to a
prolonged reduction in peanut-specific IgE/IgG1 and Th2 cytokines, accompanying
an increase in IgG2a and IFN-
Research into nanoparticle-based totally allergen immunotherapy (AIT) has gained momentum due to the growing incidence of allergic illnesses and the limitations of conventional therapies. This approach utilizes nanoparticles to beautify the efficacy and safety of allergen delivery, aiming to improve patient results [156].
Nanoparticle-based immunotherapy has emerged as a promising approach to cancer treatment. These nanoparticles can effectively deliver tumor antigens, costimulatory molecules, and immune adjuvants to enhance the immune response against cancer cells [157, 158]. The versatility of nanoparticles allows for improved efficacy of conventional cancer vaccines, sustained drug delivery, and modulation of the tumor microenvironment [158]. Various nanoparticle designs, including those made from polylactic-co-glycolic acid (PLGA), liposomes, and gold, have been explored for cancer immunotherapy applications [159]. These nanocarriers can be engineered to target specific immune cells, such as dendritic cells, and overcome limitations of traditional immunotherapies [160]. The ability of nanoparticles to co-deliver multiple immunotherapeutic agents and their potential for combination therapies make them a valuable tool in the fight against cancer [157, 159]. Studies in animal fashions have shown that nanoparticle-allergen complexes can improve balance, bioavailability, and centered delivery to precise immune cells, at the same time as reducing immunotoxicity in comparison to standard immunotherapy [1, 13].
While the ability of nanoparticle-based total cures is promising, protection remains a crucial subject. Research shows that nanoparticle formulations do not induce anaphylactic reactions, which is a significant benefit over traditional immunotherapy techniques that regularly bring a danger of severe detrimental consequences [4].
To alleviate the toxic effects of nanomaterials, researchers can alter the physicochemical features of nanoparticles (NPs), optimising their size, surface charge, and aggregation behaviour to diminish cytotoxicity while improving clearance from the body to lessen organ accumulation. Addressing oxidative stress and DNA damage generated by nanomaterials may involve employing antioxidants and producing particles with inherent antioxidant characteristics. To lessen allergic reactions and respiratory difficulties, effort can be directed on producing nanomaterials with minimal allergenic potential by changing surface features to prevent interaction with immune cells. Developing biocompatible and non-immunogenic nanomaterials for hypersensitive reactions while understanding their interactions with innate and adaptive immunity can boost safety. To handle pseudoallergy reactions, researchers should examine the complement system and inflammasome activation to design nanomaterials that do not trigger these pathways. Preventing skin harm from nanoparticles can be addressed by optimizing size, shape, and surface qualities to prevent skin penetration. Overcoming scientific barriers necessitates focussing research into nanoparticle-allergen interactions and completing long-term safety assessments. This underscores the urgency of improving understanding in the realm of nanotechnology. Establishing a robust regulatory framework is crucial for clear standards for evaluating and approving nanoparticle-based therapeutics. Practical impediments can be addressed by collaboration between researchers and pharmaceutical corporations to enhance development procedures and promote accessibility. Lastly, teaching healthcare providers about nanoparticle-based allergy immunotherapy through specialised training programs is vital for assuring informed practice. These methods strive to overcome the hurdles of nanomaterials and impediments to implementation, necessitating greater study, collaboration, and legislative developments to ensure patient safety and accessibility in nanomedicine.
Nanoparticle-based immunotherapy and traditional immunotherapy differ in their mode of action and administration. Nanoparticle-based immunotherapy uses nanoparticles to encapsulate allergens, allowing for targeted delivery and modulation of immune responses, which can be administered via oral, subcutaneous, or intranasal routes. This method promises enhanced efficacy and potentially faster results by improving allergen delivery and immune modulation. It also offers flexible delivery methods, which may improve patient compliance. On the other hand, traditional immunotherapy requires multiple treatment sessions and is administered through subcutaneous, sublingual, or intratympanic routes. While it is a well-established treatment for various allergies, it can be significantly expensive for patients. Nanoparticle-based immunotherapy, an emerging field with ongoing research, shows promising results with lower risks of anaphylactic reactions and adverse effects, potentially leading to lower long-term costs. However, it is not yet widely implemented, and the safety profile is still under investigation (Table 2, Ref. [6, 9, 10, 13, 68, 79, 108, 161, 162, 163, 164, 165, 166, 167, 168, 169, 170, 171, 172, 173, 174, 175, 176, 177, 178]).
| Aspect | Traditional immunotherapy | Nanoparticle-based immunotherapy |
| Mode of action | Gradual exposure to increasing doses of allergens to build tolerance [162, 163, 164]. | It uses nanoparticles to encapsulate allergens for targeted delivery and modulation of immune responses [10, 13, 79, 161]. |
| Administration | It can be delivered via various routes, including oral, subcutaneous, and intranasal [165, 166]. | It can be delivered via various routes, including oral, subcutaneous, and intranasal [165, 166]. |
| Efficacy | It is effective for many patients but may take years for full benefits [9]. | It promises enhanced efficacy by improving allergen delivery and immune modulation, potentially yielding faster results [10, 108]. |
| Safety profile | The risk of severe side effects, including systemic reactions, occurs in approximately 1–4% of patients, with anaphylaxis being a rare but severe concern, particularly within the first doses of treatment [167, 168]. | Studies suggest a lower risk of anaphylactic reactions and fewer adverse effects, though safety is still under investigation [10, 108]. |
| Dosage | Requires gradual dose escalation, which can be time-consuming [170, 171, 172, 173, 174]. | It allows tailored dosing through controlled release mechanisms, potentially reducing the overall dosage needed [169]. |
| Patient compliance | Requires regular visits for injections or sublingual tablets, which can affect adherence [175]. | Flexible delivery methods may improve patient compliance due to convenience [165, 166]. |
| Research status | It is established and widely used for many allergies, though with limitations [6]. | Emerging field with ongoing research demonstrating promising results but not yet widely implemented [169]. |
| Cost | Traditional immunotherapy is a well-established treatment for various allergies. It requires multiple treatment sessions, which can be significantly expensive for patients [68, 178]. | Due to reduced side effects and improved efficacy, there is potential for lower costs in the long term, though initial development costs may be high [176, 177]. |
The emergence of various applications utilizing diverse nanomaterials in biomedicine has necessitated an assessment of potential adverse consequences, specifically toxicity and hypersensitivity reactions [179].
Nanoparticles (NPs) induce cytotoxicity through many mechanisms, such as necrosis and apoptosis [180]. Substance toxicity is influenced by many physicochemical features such as morphology, size, surface charge, solubility, redox-active qualities, and aggregation capability [181, 182]. A study conducted in a laboratory setting have demonstrated a relationship between smaller size and increased toxicity. This is likely since tiny nanostructures are more effectively dispersed throughout the body, leading to their accumulation in certain organs and absorption by cells [183].
Nanostructures have the potential to induce the generation of reactive oxygen species (ROS), leading to oxidative stress within biological systems. This oxidative stress can cause damage to DNA molecules, affecting their structure and function. Understanding and mitigating these effects are crucial in nanomaterial research to ensure their safe utilization in various applications [184]. This has been demonstrated in keratinocytes, fibroblasts, and macrophages when exposed to carbon nanotubes [185]. Substance toxicity can be influenced by how they are administered, with intravenous injection often leading to enhanced toxicity due to increased dispersion throughout the body. However, while there are no absolute guidelines, toxicity may be altered by modifying nanostructure features, such as decreasing positive charges on the surface Cationic dendrimers have been associated with higher cytotoxicity [186], which may limit their biological applications. However, incorporating low cytotoxic groups, such as Zwitterionic moieties [187].
Immune cells can identify nanostructures as foreign substances, leading to two possible outcomes: they can either trigger an allergic or sensitizing response or enhance the immune response as boosters or adjuvants, potentially serving as immunomodulators [188]. Nanomaterials harm the respiratory system, worsening asthma symptoms and causing allergen reactions [188]. Nanoparticles, such as TiO2NPs, possess the unique ability to amplify allergen exposure by locally storing and gradually releasing allergens. This mechanism results in increased allergen levels, persistence, and prolonged release, thereby enhancing allergen presentation to the immune system. This property has significant implications for immunotherapy, offering a novel approach to desensitization and allergy management [189]. Interestingly, even without penetrating the epidermis, allergen-nanomaterial complexes have been found to exhibit this phenomenon. This observation highlights the complex interactions between nanomaterials and the immune system, providing valuable insights into the development of topical or transdermal allergy treatments [150].
Several nanostructures have been identified to exacerbate Th2-mediated allergic responses [190]. Pulmonary exposure to nanoparticles (NPs) can induce the release of inflammatory mediators in the lungs, even in the absence of allergens, suggesting an innate immune response. However, when an allergen is present, this effect may be amplified, leading to heightened inflammation. This observation is significant in understanding the complex interplay between NPs, allergens, and the respiratory system’s immune response [191]. Although these findings imply that tiny NPs may enhance allergic lung inflammation [192], the other study suggests that these reactions can be reduced [193]. The observed interaction between nanoparticles (NPs) and the immune system is indeed intricate and warrants further investigation. Understanding the complex relationship between NPs and immune responses is crucial for developing safe and effective nanomedicines, particularly in the context of allergy and immunotherapy. More research in this area can contribute to the advancement of precision medicine and personalized treatment strategies.
Nanomaterials have the potential to trigger hypersensitivity reactions by interacting with both innate and adaptive immunity. This involves antigen processing and presentation to T cells by antigen-presenting cells, particularly dendritic cells, ultimately resulting in the activation of effector cells. Such interactions highlight the complex role of nanomaterials in immune system engagement [188]. Nanomaterials possess the ability to activate multiple components of the immune system, including the complement system and pattern recognition receptors. They can also stimulate the release of alarmin molecules, which play a crucial role in triggering inflammasome activation. This multifaceted interaction between nanomaterials and immune pathways can significantly influence immune responses and inflammation [190]. Metal-based nanomaterials can provide an extra risk in terms of allergies due to the presence of metals known to induce allergic contact dermatitis, asthma, and allergy adjuvants [179]. TiO2NPs and ZnONPs are widely used in sunscreens and skin care products because they protect against UV radiation [194]. AgNPs are integrated for their antibacterial qualities, while SiNPs are used in cosmetics and to modify other materials’ characteristics [195]. Due to their wide range of applications and ability to deeply penetrate the skin, these substances can cause sensitization [179]. Smaller particles induce a more pronounced inflammatory reaction due to their ability to infiltrate tissues and increase surface area [196]. When the skin barrier fails, TiO2NP might worsen atopic dermatitis symptoms [197]. Polystyrene NPs can cause skin rashes without an allergen by stimulating CC-chemokines production.
Non-IgE-mediated hypersensitivity responses, known as pseudoallergy or
idiosyncratic reactions, have been linked to a broad spectrum of NPs [190]. A
proposed mechanism suggests that nanomaterials activate the complement system,
resulting in the release of anaphylatoxins. This activation subsequently
stimulates mast cells, basophils, and other inflammatory cells in the bloodstream
[198]. Additionally, certain studies revealed that nanoparticles (NPs) can
activate the NLRP3 inflammasome, an intracellular receptor, leading to the
production of pro-inflammatory cytokines IL-1
Nanoparticles (NPs) can contribute to the development of allergies and asthma by compromising the body’s protective barriers, such as the pulmonary and intestinal mucosa and the skin. This disruption affects the integrity of tight junctions, the mucus layer, efflux proteins, and cell viability, leading to increased permeability. Consequently, NPs can initiate an innate immune response and facilitate the entry of allergenic proteins, potentially triggering allergic reactions and exacerbating respiratory conditions [201, 202]. Before commercializing these nanomaterials, it is essential to conduct an assessment to identify potential adverse effects. In addition, it is essential to evaluate their safety and effectiveness. The generalization of these effects is impossible due to the strong dependence of immunological effects on the physicochemical structure and features of each specific nanomaterial. Furthermore, even when considering the same material, administration circumstances play a significant role. Therefore, it is necessary to determine the possible dangers in each specific situation through preclinical investigations [203].
Several barriers hinder the widespread adoption of nanoparticle-based allergen immunotherapy (AIT) in clinical settings. These challenges can be categorized into scientific, regulatory, and practical aspects.
Understanding the interactions between nanoparticles and allergens is crucial for developing effective therapies. Research is still needed to elucidate these interactions’ molecular mechanisms, including how nanoparticles influence immune responses, antigen processing, and T-cell activation [1, 10]. Although nanoparticle-based formulations show promise in enhancing the stability and bioavailability of allergens, there are unresolved questions regarding their long-term safety and potential adverse effects [10, 13, 204, 205, 206]. Concerns about immunotoxicity and the risk of inducing unwanted immune responses remain significant. Different types of nanoparticles (e.g., liposomes, dendrimers, metal-based) exhibit varying characteristics that can affect their performance in immunotherapy. This variability complicates the standardization of formulations and necessitates extensive testing to determine the most effective types for specific allergens [1, 4, 207]. In addition, nanoparticles must retain their stability in the biological milieu sufficiently to deliver their therapeutic target successfully [13].
The regulatory framework for nanoparticle-based therapies is still developing. Authorities must create clear protocols for testing, approving, and monitoring nanomedicines [208]. The development of nanoparticle-based AIT formulations necessitates a robust regulatory framework with clear guidelines for testing and approval. Regulatory bodies demand extensive data on safety, efficacy, and manufacturing processes, which can be both time-consuming and costly for pharmaceutical companies. This rigorous process is essential to ensure the quality and safety of novel treatments, ultimately benefiting patients and advancing medical care [1, 13]. Designing clinical trials for nanoparticle-based therapies poses unique challenges, including determining appropriate dosing regimens and assessing long-term outcomes [10]. The complexity of these formulations may also complicate the interpretation of trial results, making it challenging to establish apparent efficacy compared to conventional therapies [1, 10].
The development and production of nanoparticle-based therapies can be expensive, which may limit accessibility for patients [209]. High costs associated with research, development, and regulatory compliance can increase treatment prices, potentially making them unaffordable for many patients [13]. Immunotherapy often requires a long-term commitment from patients, and introducing nanoparticles may not significantly change the duration or frequency of treatment needed [1]. Additionally, patients may be hesitant to adopt new therapies due to unfamiliarity or concerns about safety [13]. Effective implementation of nanoparticle-based AIT requires healthcare providers to be well-informed about the technology and its benefits [13]. There may be a gap in knowledge and training among practitioners, which can hinder the integration of these therapies into clinical practice [4]. These limitations emphasize the necessity for ongoing study and advancement in nanoparticle technology for allergy therapy.
The prolonged stability of nanoparticles within the body raises substantial issues owing to their distinctive size and structural characteristics, which influence their ability to interact with biological systems. A significant concern is their interaction with the immune system. Immunological cells, including macrophages, can identify nanoparticles, triggering an immune reaction that might cause an inflammatory response or other negative consequences. To address this difficulty, one strategy is to engineer nanoparticles that elude immunological detection by encapsulating them in biocompatible substances like polyethylene glycol (PEG). PEGylation diminishes the immune system’s capacity to recognize nanoparticles, decreasing the likelihood of an immunological response [210]. Another technique is the fabrication of nanoparticles that emulate the body’s native chemicals, reducing the likelihood of their recognition as alien entities [211].
Nanoparticles pose the risk of causing allergic reactions. These reactions may result from the surface chemistry of nanoparticles, causing them to interact with proteins in the body to generate substances that may function as allergy triggers. A potential response to this issue is the surface enhancement of nanoparticles to improve their biological compatibility. Incorporating selected non-immunogenic agents or proteins might diminish the probability of an allergic reaction [212]. Moreover, comprehending the molecular pathways that govern these allergic reactions enables researchers to engineer nanoparticles with customized surface characteristics to prevent eliciting undesirable allergic reactions [213].
An alternative method to mitigate safety concerns is the creation of biologically degradable nanoparticles that decompose into non-toxic metabolites once they complete their therapeutic function. This mitigates the risk of chronic buildup and toxicity within the body. Nanoparticles composed of natural polymers such as chitosan or poly (lactic-co-glycolic acid) (PLGA) have demonstrated safe degradation in biological settings, reducing the possibility of persistent toxicity [214]. Moreover, enhanced in vivo study and modeling are crucial for accurately forecasting the long-term impacts of nanoparticles. Conventional in vitro assays frequently neglect the intricate relationships present inside the body. Consequently, researchers are concentrating on creating more precise animal models and computational simulations to forecast the performance of nanoparticles in living beings over prolonged durations [215]. These advancements provide a more thorough evaluation of possible dangers and contribute to ensuring the long-term safety of nanoparticles. Regulatory frameworks must advance to incorporate more rigorous criteria and sustained assessment of nanoparticles following their use in medicinal applications. Thorough regulatory control, encompassing post-market surveillance, may facilitate identifying and adequately managing any unanticipated long-term impacts [216].
Nanoparticle-based allergen immunotherapy holds significant potential but also comes with distinct challenges. In this context, we outline various strategies to effectively address these challenges, ultimately leading to more accessible and effective treatment options.
Several solutions exist for the difficulties mentioned above. Before large-scale manufacturing, it is imperative to utilize carriers with low toxicity to manufacture nanoparticles and streamline the preparation process [222]. Furthermore, nanoparticles with positive charges cannot penetrate airway mucus successfully [223]. Cationic carriers frequently need to be changed on their surfaces to mitigate potential toxicity, including neutral or anionic groups. Anti-inflammatory nanoparticles can regulate pharmaceutical release with specialized response capabilities. Nanoparticles can be engineered with pH-sensitive hydrophobic segments, allowing for targeted drug release in specific physiological conditions. For example, pH-responsive nanoparticles can be designed to release drugs selectively in inflamed organs, where the pH environment differs from healthy tissues. This innovative approach ensures that medications are delivered precisely where they are needed, enhancing treatment efficacy and minimizing off-target effects. Ultimately, to facilitate nanoparticle penetration through the mucus layer and enhance their retention, it is advantageous to create smaller nanoparticles, include targeted molecules, possess electrical neutrality, exhibit high encapsulation efficiency, and have low toxicity [224]. Substances like chitosan, known for their mucoadhesive properties, can be integrated into nanoparticles to enhance their capacity to linger within the organs for a prolonged duration. The surface of nanoparticles may be altered using polyethylene glycol. Research has demonstrated that nanoparticles modified with polyethylene glycol may readily traverse the mucus layer [225].
The latest allergy treatment strategies are marked by significant innovations, including precision medicine, endotype/phenotype analysis, biomarker-based diagnostics, and biologic therapies. These advancements are reshaping allergy management, offering personalized and targeted interventions for improved patient outcomes. Individualized therapeutic strategies and precision medicine for allergy patients have significant potential as the future of allergy treatment [226]. Nanotechnology has significant potential for enhancing targeted allergy therapy by precisely regulating medication release at specific sites [227]. In forthcoming research endeavors, researchers might have the potential to offer individuals personalized and meticulously customized therapies that precisely control the release of medications. This can be achieved by acquiring a thorough comprehension of allergy immunology [228]. Nanoparticle-based medications can be tailored to specific allergy subtypes, such as targeting eosinophil release in Th2-high allergies, offering personalized treatment. Similarly, pharmaceuticals designed to modulate neutrophil release could provide relief to patients with Th2-low allergies, highlighting the potential of customized therapies in addressing diverse allergic conditions [229]. The targeted approach to allergy treatment, focusing on specific immune pathways and cellular responses, holds great promise in enhancing therapeutic efficacy. By tailoring interventions to individual allergy subtypes, this strategy can lead to more successful outcomes and improved patient satisfaction. While numerous preclinical investigations have centered on nanotechnology as a potential alternative for treating allergies, there have been no clinical trials conducted as of yet [203]. Hence, nanotechnology is anticipated to be employed in treating allergy, from the laboratory to the clinical setting. Challenges still need to be addressed in this process. Developing nanoparticles designed explicitly for administration is crucial for allergies [4]. Additionally, it is critical to research the anti-allergy mechanism of these nanomaterials.
The potential of nanoparticle-based allergen immunotherapy warrants further exploration, and clinical trials play a pivotal role in advancing this field. Here, we propose a structured approach to guide future research endeavors, ensuring a rigorous and efficient process.
Phase I: Strategic Planning and Design
A comprehensive clinical trial strategy is imperative, outlining the study’s objectives, methodologies, and anticipated outcomes. This phase involves meticulous consideration of patient selection criteria, optimal dosing regimens, and the exploration of diverse nanoparticle combinations. By defining these parameters, researchers can pave the way for a well-structured and focused investigation.
Phase II: Navigating Regulatory Pathways
Obtaining regulatory approval is a critical step, requiring submission of the trial plan to competent authorities, such as the Food and Drug Administration (FDA). Compliance with established safety and efficacy guidelines for nanoparticles is essential. To streamline this process, leveraging previously FDA-approved materials can be a strategic advantage, facilitating a smoother regulatory journey.
Phase III: Enhancing Patient Engagement
Active collaboration with healthcare professionals and advocacy groups is key to facilitating patient recruitment. By incorporating feedback from research partners, trials can be tailored to address patient needs and preferences, thereby enhancing enrollment rates. A patient-centric approach ensures a more inclusive and successful clinical trial process.
Phase IV: Adaptive Trial Innovations
Implementing adaptive trial designs is a strategic move, allowing for dynamic adjustments based on interim results. This approach optimizes resource allocation and accelerates decision-making regarding trial continuation or modification. By embracing adaptability, researchers can enhance the efficiency and effectiveness of clinical studies, as evidenced by existing literature [230, 231].
By utilizing innovative strategies, researchers can develop precise and customized therapies for individual patients. This involves identifying specific biomarkers to understand immune responses, creating personalized nanoparticles with targeted allergens, and implementing individualized dosing regimens. The integration of real-time monitoring systems further enhances this approach, allowing healthcare providers to continuously track patient responses and adjust treatment plans accordingly. With these advancements, nanoparticle-based allergen immunotherapy becomes a powerful tool, offering improved patient outcomes and an enhanced quality of life. This patient-focused methodology paves the way for a new era of precision medicine, ensuring that each patient receives a treatment plan tailored to their unique needs [232].
Enhancing nanoparticle-based allergen immunotherapy through collaborative innovation requires a strategic approach that unites industry and academia. By initiating joint research projects, we can bridge the gap between theoretical knowledge and practical application, leveraging the expertise of both sectors. Creating knowledge-sharing platforms fosters a collaborative environment, accelerating innovation and driving the field forward. Securing funding through grants and investments is crucial for supporting these initiatives, ensuring the continuity and growth of nanoparticle research. Developing training programs that involve academic researchers in industry projects enhances practical skills and creates a pipeline of talented experts. Forming public-private partnerships aligns industry needs with academic capabilities, resulting in focused and impactful solutions to address specific health challenges. This collaborative framework empowers the field, leading to improved patient outcomes and a dynamic ecosystem for immunotherapy advancement [231].
Nanoparticles (NPs) offer several advantages over traditional Allergen Immunotherapy (AIT) methods. They can enhance allergen delivery to lymph nodes and blood vessels, modulate immune responses more effectively, and provide a depot effect for sustained antigen release. NPs also protect allergens from degradation, enabling new routes of administration like oral, sublingual, and intranasal, which could improve patient compliance. However, there are significant challenges to the widespread adoption of NP-based AIT, including scientific uncertainties about long-term safety and immunotoxicity, the need for robust regulations, and practical issues like high development costs and patient hesitancy due to unfamiliarity with the technology. While NP-based AIT shows great potential in improving allergy treatment, further research is required to fully understand the interactions between NPs and the immune system. Overcoming these barriers is essential to unlock the full potential of this innovative approach in clinical practice.
MAA, RME, NSS, MH, and AHI designed and wrote the manuscript. MAA, RME, NSS, WM, MH, and AHI were involved in original draft preparation; MAA, RME, NSS, WM, MH, and AHI participated in reviewing and editing; and MAA, RME, NSS, and AHI handled visualization. MAA and WM contributed to the design of the figures. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to thank all those who helped us during the preparation of this manuscript and all the reviewers for their valuable comments and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



