1 Department of Rehabilitation Medicine, Affiliated Hospital of North Sichuan Medical College, 637000 Nanchong, Sichuan, China
2 Department of Radiology, Affiliated Hospital of North Sichuan Medical College, 637000 Nanchong, Sichuan, China
3 Department of Rehabilitation Medicine, Qinghai Provincial People's Hospital, 810000 Xining, Qinghai, China
†These authors contributed equally.
Abstract
Post-stroke dysphagia (PSD) is a common and serious complication, yet conventional rehabilitation therapies have limited efficacy. Repetitive transcranial magnetic stimulation (rTMS) is a promising treatment, but its optimal intervention strategy remains undetermined.
Seventy-five PSD patients were randomly assigned in a 1:1:1 ratio to the sham rTMS group, affected rTMS group and bilateral rTMS group. All groups received the corresponding rTMS intervention and conventional rehabilitation therapy. Swallowing function was assessed at baseline (T0) and after treatment (T1) using the standardized swallowing assessment (SSA), penetration-aspiration scale (PAS), yale pharyngeal residue severity rating scale (YPR-SRS), and suprahyoid motor evoked potentials (MEP). Adverse reactions and dropouts were recorded.
After treatment, the SSA scores of all three groups were significantly improved. Bilateral rTMS showed significantly greater improvement in SSA and a higher treatment response rate (77.27%) compared to both the sham group and the affected rTMS group (p < 0.001). Mixed-effects model and intention-to-treat analyses both supported the optimal efficacy of bilateral rTMS (interaction effect p < 0.01). Regarding swallowing safety (PAS), the bilateral rTMS group's score was significantly lower than that of the sham group (p = 0.017). In terms of pharyngeal residue clearance (YPR-SRS), the bilateral rTMS group showed significantly greater improvement in the piriform sinuses compared to the other two groups, and superior improvement in the vallecula compared to the sham group (p < 0.05). After treatment, MEP amplitudes increased in all groups. Notably, only the bilateral rTMS group not only significantly increased MEP amplitudes on both sides (p < 0.01) but also significantly shortened the latency on the contralesional side (p = 0.046). The bilateral rTMS group achieved a “large effect size” in improving SSA scores, increasing MEP amplitudes, and shortening latency on the contralesional side, with the SSA effect size (D = 2.339) far exceeding that of the other groups. All treatment regimens were well-tolerated, with only 5 cases of transient scalp discomfort reported and no serious adverse events.
Conventional rehabilitation combined with 10 Hz rTMS targeting the swallowing cortex can effectively improve swallowing function in PSD patients. Bilateral rTMS is a superior strategy. Its therapeutic advantage may stem from the synergistic modulation of bilateral cortical excitability and neural conduction efficiency, providing a better multi-target neuromodulation option for clinical practice.
No: ChiCTR2300068730. https://www.chictr.org.cn/showproj.html?proj=182568.
Graphical Abstract
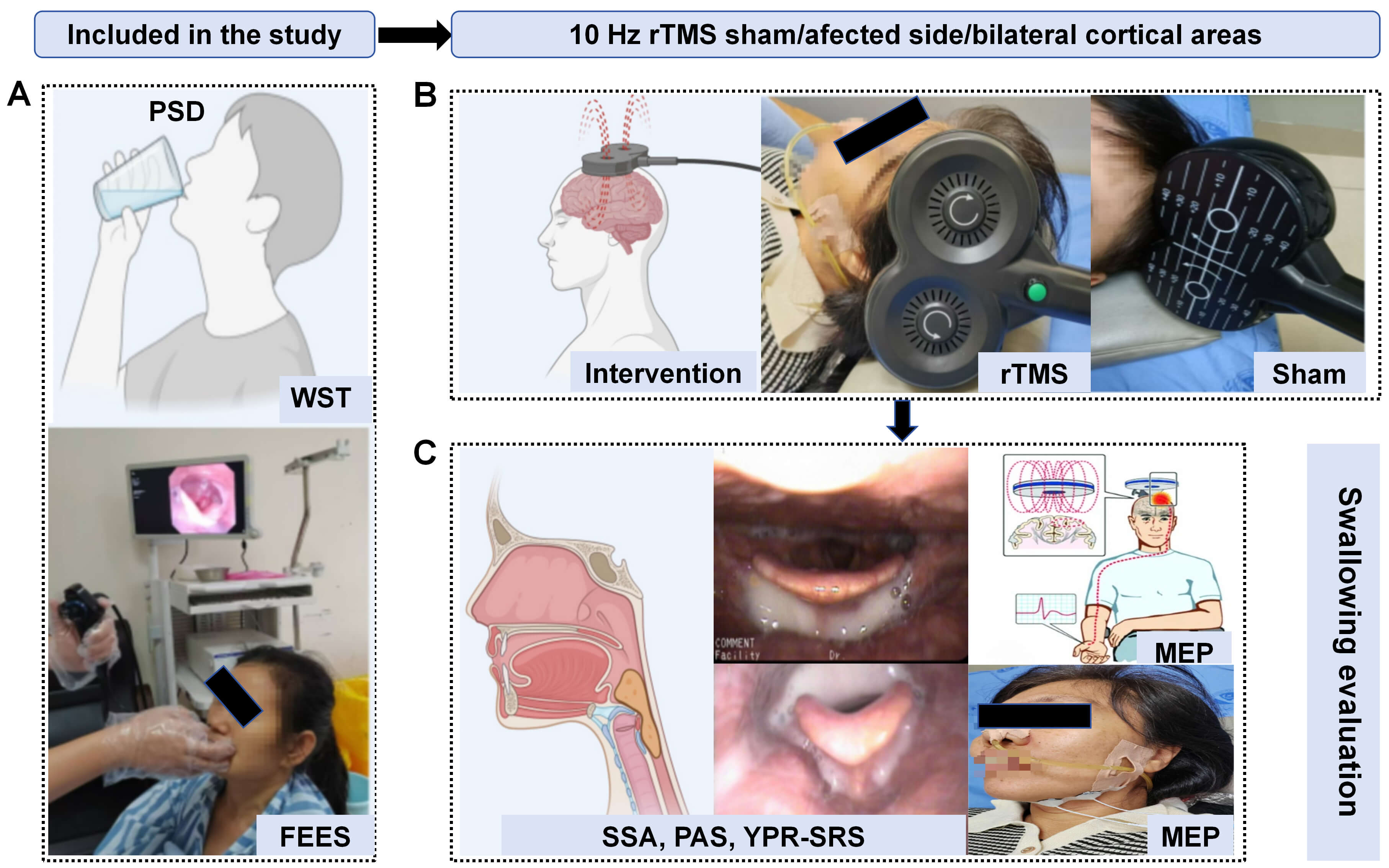
Keywords
- transcranial magnetic stimulation
- stroke
- post-stroke dysphagia
- randomized controlled trial
Dysphagia is one of the common complications following stroke, with an incidence rate ranging from 21% to 64% [1]. Post-stroke dysphagia (PSD) can lead to dehydration, malnutrition, aspiration and lung infections, in severe cases, can be life-threatening, imposing a heavy burden on both patients and their families [2]. Current conventional treatment options for PSD include swallowing exercises, physical factor therapy based on electrical swallowing stimulation, traditional Chinese medicine therapy, posture training and dietary modifications. Although these methods have some efficacy, they often encounter limitations. Finding ways to rapidly improve swallowing function, reduce various complications and enhance the quality of life for patients remains a key focus and challenge [3, 4].
Repetitive transcranial magnetic stimulation (rTMS) is one of the commonly used and well-supported neuromodulation techniques in stroke rehabilitation [5, 6]. In recent years, the use of rTMS in patients with PSD has increased significantly, demonstrating promising clinical potential [5, 7]. The therapeutic mechanisms of rTMS for PSD are currently understood through several theoretical models: The interhemispheric competition theory posits that post-stroke interhemispheric inhibition via the corpus callosum is disrupted, leading to reduced excitability in the affected hemisphere and increased inhibition from the unaffected hemisphere, which is considered a key contributor to swallowing dysfunction [8]. According to this model, applying high-frequency rTMS to the affected hemisphere can help restore excitability balance between the two hemispheres, thereby improving swallowing function [9, 10]. The compensation theory of the unaffected hemisphere suggests that when the affected hemisphere is severely damaged and corticobulbar pathways are compromised, activating compensatory pathways in the unaffected hemisphere becomes crucial for functional recovery [11]. Furthermore, the dual-balance model emphasizes that simultaneous modulation of excitability in both hemispheres may optimize neural network function more effectively than unilateral stimulation alone [12]. Beyond targeting the cerebral cortex, modulating the cerebellum and its functional connections with cortical regions involved in swallowing has emerged as a new research direction, aiming to improve swallowing function by influencing cortical–cerebellar circuits [13, 14].
Although the aforementioned theories provide a basis for different stimulation strategies, there is still no consensus on which strategy is optimal. While preliminary evidence from previous studies and the present study suggests that high-frequency rTMS targeting bilateral cerebral cortices may be a superior approach, this hypothesis still requires validation through rigorously designed, high-quality clinical trials [13, 15, 16, 17, 18].
Therefore, this study adopted a high-frequency (10 Hz) rTMS protocol with
multi-target combined modulation. This protocol is based on prior evidence
indicating that high-frequency rTMS (
Patients with PSD were recruited from the Affiliated Hospital of North Sichuan Medical College between March 2023 and October 2025. All participants met the predefined inclusion and exclusion criteria. Before the commencement of the study, written informed consent was obtained from each patient or their legal guardian/immediate family member, ensuring full comprehension of the study’s purpose, procedures, potential risks, and benefits. The trial protocol was approved by the Medical Ethics Committee of the Affiliated Hospital of North Sichuan Medical College (approval numbers: 2023ER031-1) and registered in the Chinese Clinical Trial Registry (registration number: ChiCTR2300068730).
This study was designed as a single-center, single-blind, randomized controlled trial with an integrated multimodal evaluation system to comprehensively examine intervention effects and underlying mechanisms. Assessment modalities included the standardized swallowing assessment (SSA), fiberoptic endoscopic evaluation of swallowing (FEES), and suprahyoid muscle motor evoked potentials (MEP). The study aimed to systematically elucidate the intervention’s pathways and neuromodulatory mechanisms from both behavioral and neurophysiological perspectives, providing a more precise scientific basis for the rehabilitation of patients with PSD.
Block randomization was employed, with an independent statistician not involved in patient recruitment or assessment generating a random sequence using computer software. The sequence allocated patients in a 1:1:1 ratio to the sham group, affected rTMS group and bilateral rTMS group. Allocation concealment was ensured using sequentially numbered, opaque, sealed envelopes containing the group assignment. Eligible patients who provided informed consent were assigned to a group by a study coordinator who opened the corresponding envelope in sequential order, thereby determining the group allocation and treatment protocol. Blinding was applied to both patients and outcome assessors; only the treating operator was aware of the group assignment. All outcome measures were assessed at baseline (before treatment) and immediately post-intervention by the same therapist, who was blinded to group allocation and not involved in the intervention. This assessor received standardized training prior to the trial to ensure consistency in evaluation. Throughout the trial, adverse events (including epilepsy, headache, dizziness, syncope, dyspnea, scalp/neck skin redness, tinnitus, etc.) and participant dropout were recorded in detail to comprehensively evaluate treatment safety and tolerability.
This trial employed a random grouping design, consisting of a total of three
groups. The significance level (
(1) Unilateral stroke confirmed by computed tomography or magnetic resonance
imaging [22]; (2) First-ever stroke occurring within 2 weeks to 6 months after
onset; (3) Age
(1) Dysphagia attributable to neurological conditions other than stroke, such as Parkinson’s disease, Alzheimer’s disease, traumatic brain injury, cerebellar or brainstem strokes, and head/neck tumors; (2) Pregnancy or lactation; (3) Acutely ill or medically unstable patients (e.g., unstable hemodynamics, active progressive illness); (4) Inability to comply with FEES, MEP, or swallowing function assessments due to cognitive, behavioral, or communication impairments; (5) Contraindications to transcranial magnetic stimulation (e.g., intracranial metal implants, epilepsy) or severe adverse reactions during previous rTMS sessions; (6) Disease progression or recurrent stroke during the trial; (7) Withdrawal of informed consent or unwillingness to continue participation.
During the treatment phase, to simulate the real-world scenario of multimodal integrated rehabilitation, all patients received standardized conventional swallowing rehabilitation therapy alongside their group-specific interventions. This conventional therapy consisted of a 2-week regimen administered once daily. The conventional rehabilitation included the following components: swallowing function training, oral sensory stimulation, acupuncture therapy, and postural compensatory training. Swallowing function training, oral sensory stimulation, and postural compensatory training were conducted by qualified rehabilitation therapists [3, 4, 5, 23]. Acupuncture treatment was performed by a certified acupuncturist. The acupoint selection protocol followed the principles of “local point selection, stage-specific treatment, and integration of pattern differentiation with disease diagnosis”. Based on these principles, differentiated acupoint prescriptions were designed to address the distinct stages of PSD (oral phase, pharyngeal phase, and oral-pharyngeal mixed phase) [23, 24]. Additionally, stimulation was delivered using a figure-of-8 coil connected to a MagNeuro 60 transcranial magnetic stimulator (Nanjing Vishee Medical Technology Co., Ltd., Nanjing, China). The sham rTMS group received bilateral sham stimulation over the swallowing cortex; affected rTMS group received real stimulation on the affected hemisphere combined with sham stimulation on the contralesional side; and bilateral rTMS group received real stimulation to both hemispheres. The rTMS parameters were set as follows: frequency of 10 Hz, intensity at 100% of the resting motor threshold (RMT), train duration of 2 s, inter-train interval of 10 s, and a total of 1200 pulses per session. Treatment was administered once daily, 7 days a week, for 2 weeks. Sham stimulation was performed using the flipped-coil method, where the figure-of-8 coil was rotated 180°, aligning the induced magnetic field tangentially to the scalp. This approach produced acoustic artifacts similar to real stimulation without delivering an effective magnetic field intracranially. Prior validation tests confirmed that participants could not reliably distinguish between real and sham stimulation. Immediately after the first treatment session, a blinding assessment was conducted by asking patients, “Do you believe you received real or sham stimulation in this session?” to evaluate their awareness of group assignment. Based on previous research indicating that the excitatory effects of rTMS can last approximately 30 minutes and remain effective over intermittent periods of 20–30 minutes, the bilateral rTMS group received stimulation to the unaffected hemisphere first, followed by the affected hemisphere, with the aim of achieving an additive effect. During treatment, patients were positioned comfortably in a supine or seated posture. A figure-of-eight coil was placed over the primary motor cortex representation area (swallowing cortical hotspot) of the suprahyoid muscles, as determined by MEP mapping. Therapists administered the corresponding stimulation according to group assignment. Treatment was immediately discontinued if patients experienced intolerable discomfort.
This study systematically collected baseline demographic and clinical data from all enrolled patients prior to the trial commencement. Information included age, gender, stroke location, and stroke type, along with relevant outcome measures, to evaluate the comparability of baseline characteristics across groups. The primary and secondary outcome measures comprised the SSA, yale pharyngeal residue severity rating scale (YPR-SRS), and MEP. All assessments were conducted by a rehabilitation therapist blinded to the intervention at two time points: baseline (T0) and after two weeks of intervention (T1). Throughout the trial period, any adverse events (including seizures, headache, dizziness, syncope, dyspnea, local scalp/neck redness, and tinnitus-as well as participant dropouts) were meticulously documented to comprehensively evaluate treatment safety and tolerability. Prior to the trial, all assessors received standardized training on the relevant outcome measures to ensure consistency and accuracy in data collection.
In this study, SSA was used as the primary outcome indicator. SSA is a simple, safe, and easily promotable tool for assessing swallowing function, demonstrating good sensitivity, specificity, reliability, and validity in the evaluation of swallowing disorders related to stroke and other conditions. The scale consists of three parts: clinical examination, a 5 mL water swallow test, and a 60 mL water swallow test, with a total score ranging from 18 to 46 points. A higher score indicates a more severe swallowing dysfunction. Multiple studies have shown that SSA scores have high consistency with gold standard results such as swallow imaging, effectively identifying the risk of swallowing disorders and associated complications, and possessing good clinical predictive value [25, 26].
The other measures, including the Penetration-Aspiration Scale (PAS) score and Yale Pharyngeal Residue Severity Rating Scale (YPR-SRS) score derived from FEES, as well as MEP parameters, were pre-defined as exploratory secondary outcome measures. The analysis of these measures was intended to generate preliminary evidence and hypotheses for future research.
2.6.2.1 PAS and YPR-SRS Based on FEES
FEES is one of the “gold standards” for swallowing function assessment. It allows for direct observation of the pharyngeal structures, food residue, and dynamic changes in swallowing by having patients swallow boluses of varying viscosities. This method has high sensitivity for detecting pharyngeal residue and can effectively assess the speed of swallowing initiation, pharyngeal clearance efficiency, and the degree of aspiration [9, 25, 27]. (1) The PAS scale is used to quantify the severity of airway penetration and aspiration, with a scoring range of 1 to 8, where a score of 8 represents silent aspiration. This scale has good sensitivity, specificity, and reliability and has been widely used in swallowing disorder research [26]. (2) The YPR-SRS scale is a five-point visual assessment tool focused on evaluating the location and amount of residue in the valleculae and piriform sinuses. A higher score indicates a greater amount of residue and lower pharyngeal clearance efficiency [27].
2.6.2.2 Localization of Motor Cortex Hotspots and Measurement of MEP for the Suprahyoid Muscles
MEP is a non-invasive neurophysiological testing technique mainly used to assess the function of the neural conduction pathways from the motor cortex to the muscles, including their overall synchrony and structural integrity [9, 26]. The analysis of MEP primarily relies on two indicators: amplitude and latency. The amplitude reflects the number and synchrony of the activated motor neurons, while the latency indicates the conduction velocity of the neural impulses. These indicators provide critical neurophysiological evidence for assessing the excitability and integrity of the corticospinal tract [14].
MEPs were measured in all patients before repetitive rTMS intervention to individualize the stimulation site and intensity. The specific procedure was as follows: after the patient assumed a comfortable position and relaxed fully, the operator placed the rTMS positioning cap and recording electrodes according to the International 10–20 electroencephalogram electrode placement system. The examiner then delivered a single-pulse stimulus to the primary motor cortex at 30% of the maximum output intensity. The stimulus intensity was gradually increased and the coil position was finely adjusted until the surface electrodes over the abductor pollicis brevis (APB) muscle recorded MEPs with the largest amplitude and good reproducibility; this location was identified as the cortical “hot spot” for the APB. Subsequently, at this “hot spot”, the stimulus intensity was progressively decreased [28]. The minimum intensity required to elicit MEPs with an amplitude exceeding 50 µV in at least 5 out of 10 consecutive stimuli was defined as the RMT [29].
After completing the above measurements, the recording electrode was placed on the suprahyoid muscle (Location: 2 cm lateral to the midpoint of the line connecting the middle of the hyoid bone and the midpoint of the mandible), the reference electrode was placed 2 cm lateral to the recording electrode, and the ground electrode was placed on the proximal forearm. The coil was positioned approximately 3 cm anterior to the vertex (Cz point) and about 7.5 cm laterally [30]. Single-pulse stimuli were delivered at 100% RMT intensity, and the coil was finely adjusted to locate the cortical hot spot for the suprahyoid muscle that could evoke consistent and stable MEP waveforms. Once the hot spot was identified, 10 consecutive stimuli were delivered. The five MEP waveforms with the best reproducibility were selected to calculate and record the RMT, latency, and amplitude for the suprahyoid muscle group. If no MEP could be elicited from the affected side, the corresponding data from the mirror region of the hot spot on the healthy side were used as a substitute [28, 30]. All operations were performed by the same professional neurodiagnostic physician, strictly adhering to MEP detection standards to ensure accurate localization of the swallowing-related cortex and the effectiveness of the therapeutic stimulation intensity (100% RMT).
Statistical analyses were performed using SPSS 27.0 (IBM Corp., Armonk, NY, USA).
Normality of continuous variables was assessed using the Shapiro-Wilk test and
histograms. Normally distributed data are presented as mean
Between March 2023 and October 2025, a total of 75 patients with PSD were
enrolled in this study. During the trial, five patients were lost to follow-up:
two in the affected rTMS group withdrew one week after intervention due to
conflicts with other treatment schedules, and three in the bilateral rTMS group
dropped out before completing one week of intervention due to hospital discharge
or personal reasons. Ultimately, 70 patients completed all scale assessments,
FEES, and MEP data collection, and were included in the statistical analysis. The
detailed trial flow is illustrated in Fig. 1. Analysis of baseline
characteristics showed no statistically significant differences among the three
groups in terms of age, sex, disease duration, stroke location, or stroke type
(p
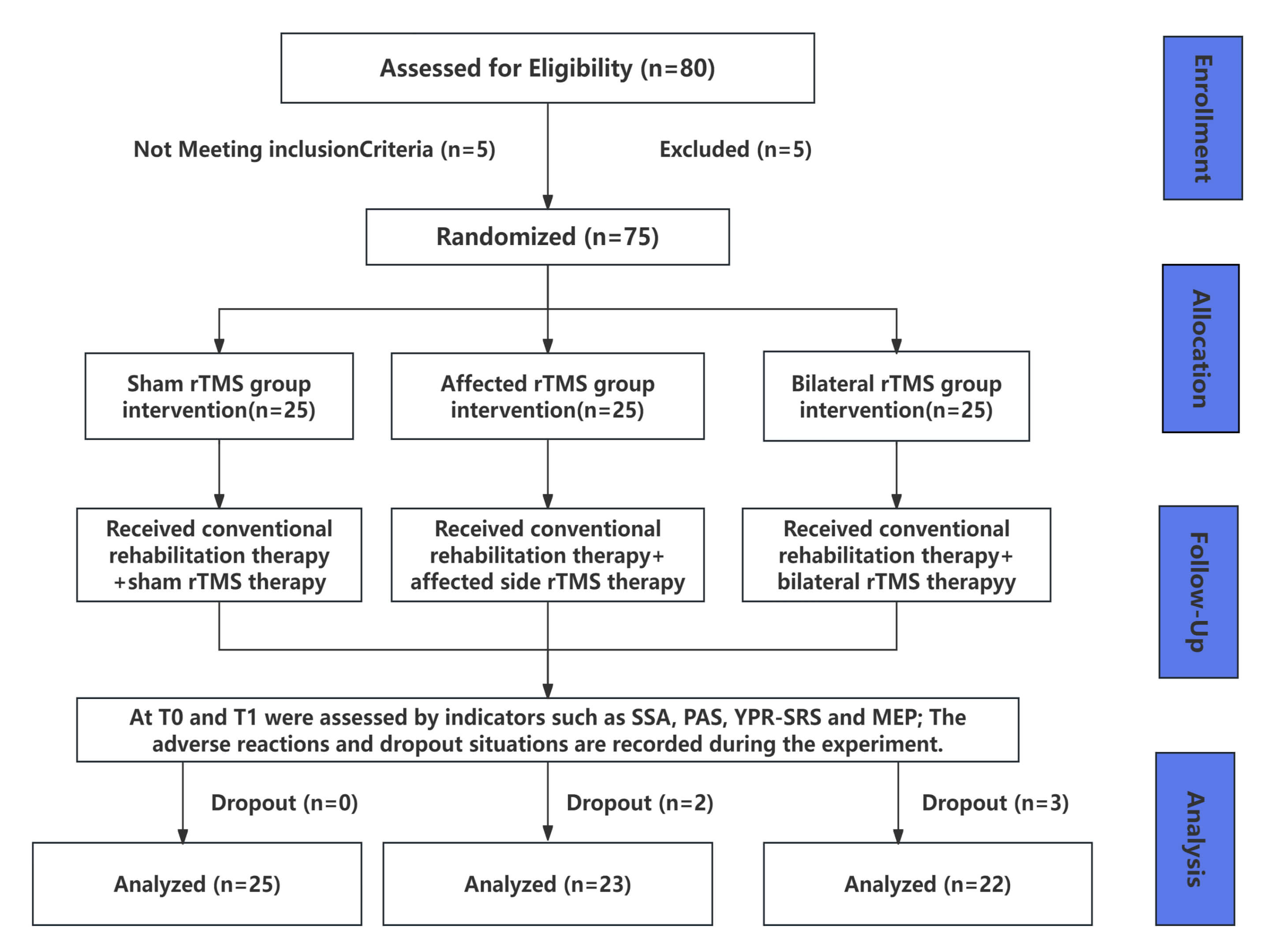 Fig. 1.
Fig. 1.
Flowchart of the experiment. rTMS, Repetitive transcranial magnetic stimulation; SSA, Standardized swallowing assessment; PAS, Penetration aspiration scale; YPR-SRS, yale pharyngeal residue severity rating scale; MEP, Motor evoked potential. T0, Baseline before the intervention; T1, 2 weeks after rTMS intervention.
| Variables | Sham rTMS | Affected rTMS | Bilateral rTMS | Statistics | p-value | ||
| (n = 25) | (n = 23) | (n = 22) | |||||
| Age (years, |
61.800 |
65.570 |
57.590 |
2.070 | 0.134 | ||
| Gender | |||||||
| Male | 16 | 15 | 15 | 0.095 | 0.954 | ||
| Female | 9 | 8 | 7 | ||||
| Time since stroke onset (days, mean |
83.84 |
82.70 |
86.64 |
0.166 | 0.920 | ||
| Stroke types | |||||||
| Ischemic | 14 | 17 | 17 | 2.911 | 0.233 | ||
| Hemorrhagic | 11 | 6 | 5 | ||||
| Stroke sites | |||||||
| Left | 10 | 13 | 13 | 2.063 | 0.356 | ||
| Right | 15 | 10 | 9 | ||||
| T0 | |||||||
| SSA ( |
32.960 |
33.570 |
32.000 |
0.551 | 0.579 | ||
| PAS | 4 (3, 5) | 4 (3, 6) | 4 (3, 5) | 0.437 | 0.804 | ||
| Md (P25, P75) | |||||||
| Oral residual scores | |||||||
| Vallecular residue | 4 (4, 4) | 4 (4, 4) | 4 (3, 4) | 1.050 | 0.592 | ||
| Md (P25, P75) | |||||||
| Pyriform sinus residue | 4 (3, 4) | 4 (3, 4) | 4 (4, 4) | 1.555 | 0.457 | ||
| Md (P25, P75) | |||||||
| MEP | |||||||
| Amplitude | |||||||
| Ipsilesional ( |
160.2 |
173.5 |
157.5 |
2.308 | 0.315 | ||
| Contralesional ( |
131.9 |
140.3 |
127.7 |
1.594 | 0.451 | ||
| Latencyperiod | |||||||
| Ipsilesional ( |
4.941 |
4.813 |
4.943 |
0.474 | 0.789 | ||
| Contralesional ( |
5.000 |
4.948 |
5.409 |
5.258 | 0.072 | ||
The SSA scores of the three groups at T0 and T1 are presented in Table 2 and
Fig. 2A. Overall, the SSA scores improved significantly in all groups after
treatment, with T1 scores being significantly lower than those at T0 (p
| Variables | Sham rTMS | Affected rTMS | Bilateral rTMS | p-value | |||
| (n = 25) | (n = 23) | (n = 22) | |||||
| SSA | |||||||
| T0 ( |
32.960 |
33.570 |
32.000 |
0.579 | |||
| 95% CI | (30.92, 35.00) | (31.49, 35.64) | (29.62, 34.38) | ||||
| T1 ( |
29.880 |
28.350 |
21.090 |
||||
| 95% CI | (27.63, 32.13) | (25.79, 30.90) | (19.39, 22.79) | ||||
| p-value | |||||||
| Change value (ΔSSA) | 3.080 |
5.217 |
10.910 |
||||
| Effective rate (ΔSSA |
32.00% | 47.83% | 77.27% | 0.008 | |||
| PAS | |||||||
| T0 Md (P25, P75) | 4 (3, 5) | 4 (3, 6) | 4 (3, 5) | 0.804 | |||
| 95% CI | (3, 5) | (3, 5) | (3, 5) | ||||
| T1 Md (P25, P75) | 2 (2, 6) | 2 (1, 3) | 1 (1, 2.25) | 0.017 | |||
| 95% CI | (2, 6) | (1, 3) | (1, 2) | ||||
| p-value | 0.126 | ||||||
| PAS |
|||||||
| T0 | 3 (12.0%) | 3 (13.0%) | 4 (18.2%) | 0.815 | |||
| T1 | 13 (52.0%) | 16 (69.6%) | 17 (77.3%) | 0.170 | |||
| PAS |
|||||||
| T0 | 20 (80.0%) | 17 (73.9%) | 19 (86.4%) | 0.580 | |||
| T1 | 21 (84.0%) | 20 (87.0%) | 21 (95.5%) | 0.448 | |||
| Vallecular residue | |||||||
| T0 Md (P25, P75) | 4 (4, 4) | 4 (4, 4) | 4 (3, 4) | 0.592 | |||
| 95% CI | (4, 4) | (4, 4) | (3, 4) | ||||
| T1 Md (P25, P75) | 3 (2, 4) | 2 (2, 3) | 2 (2, 2) | ||||
| 95% CI | (3, 4) | (2, 3) | (2, 2) | ||||
| p-value | 0.037 | ||||||
| Pyriform sinus residue | |||||||
| T0 Md (P25, P75) | 4 (3, 4) | 4 (3, 4) | 4 (4, 4) | 0.457 | |||
| 95% CI | (3, 4) | (3, 4) | (4, 4) | ||||
| T1 Md (P25, P75) | 2 (2, 3) | 2 (2, 3) | 2 (2, 2) | 0.004 | |||
| 95% CI | (2, 3) | (2, 3) | (2, 2) | ||||
| p-value | |||||||
| MEP Amplitude | |||||||
| Ipsilesional | |||||||
| T0 ( |
160.20 |
173.50 |
157.50 |
0.315 | |||
| 95% CI | (141.7, 178.7) | (156.9, 190.0) | (138.0, 176.9) | ||||
| T1 ( |
269.20 |
275.50 |
314.40 |
0.023 | |||
| 95% CI | (232.7, 305.8) | (253.9, 297.1) | (287.2, 341.6) | ||||
| p-value | |||||||
| Effect size (D) | 2.435 | 2.295 | 2.944 | ||||
| Contralesional | |||||||
| T0 ( |
131.90 |
140.30 |
127.70 |
0.451 | |||
| 95% CI | (116.3, 147.6) | (125.4, 155.1) | (110.8, 144.5) | ||||
| T1 ( |
217.30 |
208.50 |
267.30 |
0.007 | |||
| 95% CI | (196.4, 238.3) | (182.4, 234.5) | (238.5, 296.1) | ||||
| p-value | |||||||
| Effect size (D) | 1.908 | 1.391 | 2.622 | ||||
| Latency period | |||||||
| Ipsilesional | |||||||
| T0 ( |
4.941 |
4.813 |
4.943 |
0.789 | |||
| 95% CI | (4.513, 5.369) | (4.728, 4.898) | (4.725, 5.161) | ||||
| T1 ( |
4.776 |
4.713 |
4.845 |
0.196 | |||
| 95% CI | (4.673, 4.879) | (4.621, 4.805) | (4.735, 4.956) | ||||
| p-value | 0.593 | 0.079 | 0.873 | ||||
| Effect size (D) | 0.219 | 0.490 | 0.251 | ||||
| Contralesional | |||||||
| T0 ( |
5.000 |
4.948 |
5.409 |
0.789 | |||
| 95% CI | (4.866, 5.134) | (4.750, 5.146) | (5.073, 5.745) | ||||
| T1 ( |
4.912 |
4.835 |
4.957 |
0.061 | |||
| 95% CI | (4.820, 5.004) | (4.746, 4.924) | (4.858, 5.056) | ||||
| p-value | 0.270 | 0.486 | 0.046 | ||||
| Effect size (D) | 0.317 | 0.318 | 0.810 | ||||
Effect sizes of 0.2, 0.5, and 0.8 correspond to small, medium, and large, respectively.
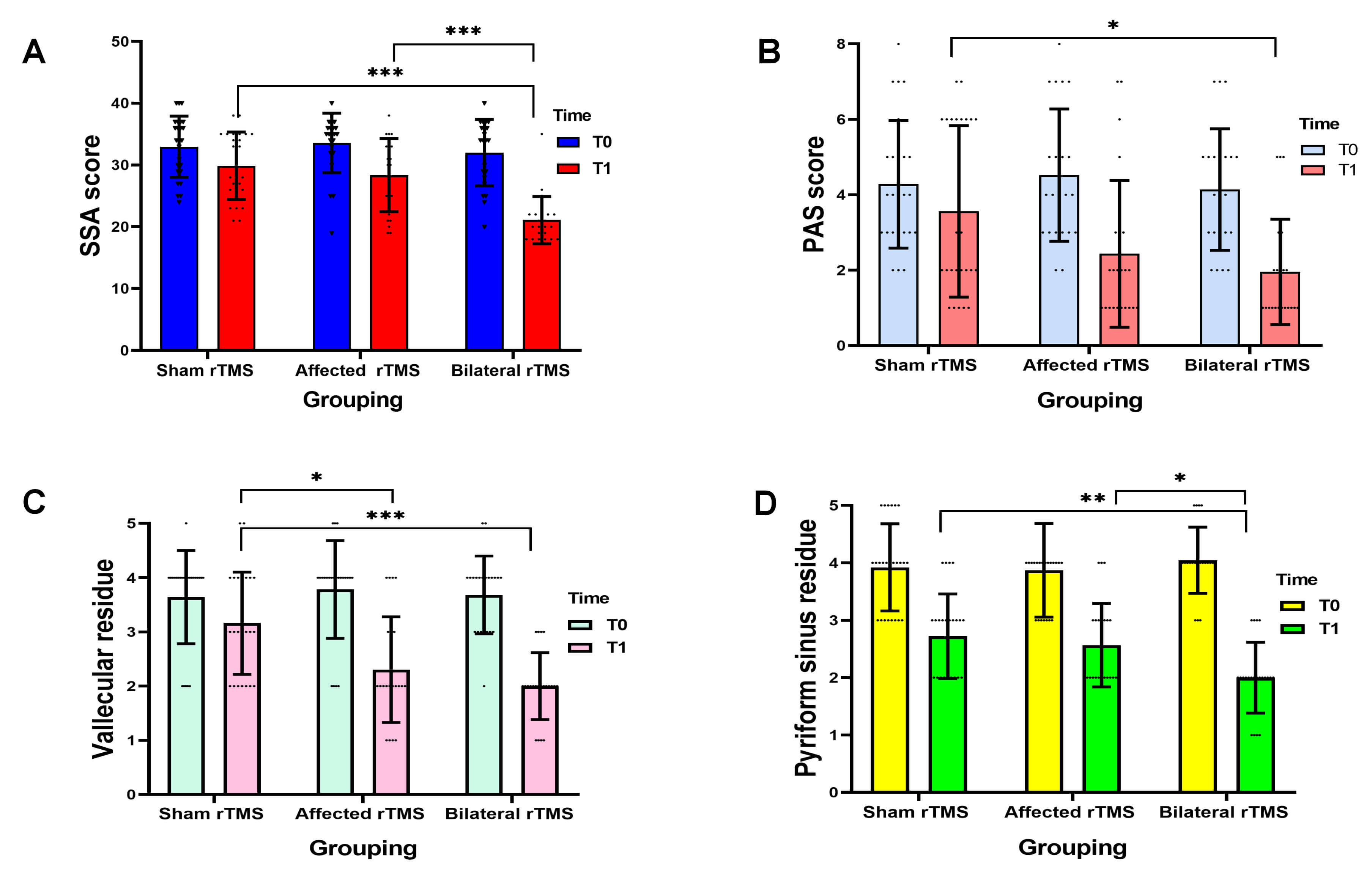 Fig. 2.
Fig. 2.
Comparison of SSA scores (A), PAS scores (B), vallecular residue
scores (C), and pyriform sinus residue scores (D) before and after treatment in
the three groups. *p
To systematically quantify the independent and interactive effects of group and
time on SSA scores, a linear mixed-effects model was constructed based on data
from patients who completed the full intervention. The model results were as
follows: a significant main effect of group (F = 54.920, p
| Model effect | F (DFn, DFd) | p-value |
| Between-group main effect | 54.920 (2, 134) | |
| Time main effect | 12.640 (1, 134) | |
| Group |
7.230 (2, 134) | 0.001 |
To further evaluate the magnitude of improvement in swallowing function, the
change in SSA score (
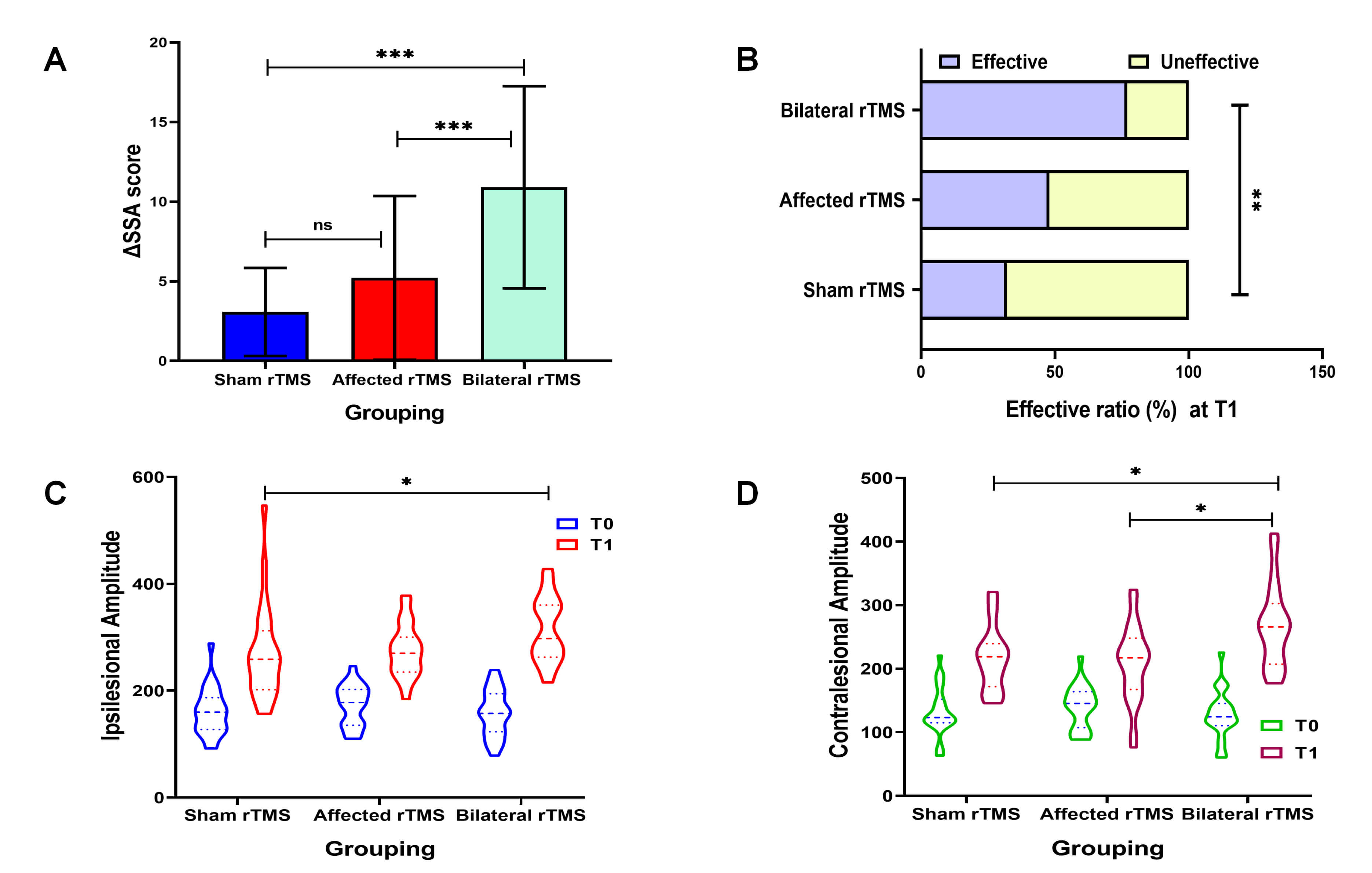 Fig. 3.
Fig. 3.
Comparison of the
Additionally, an intention-to-treat analysis was conducted, applying the
baseline observation carried forward (BOCF) method to the five patients who
dropped out. The results showed that SSA scores still differed significantly
among the three groups after treatment (p
The PAS scores of the three groups of patients at T0 and T1 are shown in Table 2
and Fig. 2B. Statistical analysis revealed that the PAS scores in the affected
rTMS group and the bilateral rTMS group at T1 were significantly lower than those
at T0 (p
Furthermore, the proportion of patients achieving safe swallowing (PAS
Table 2 and Fig. 2C,D summarize the results of the vallecular residue and
pyriform sinus residue scoring for the three groups of patients at T0 and T1.
Overall, the residue scores for both the vallecula and the pyriform sinus showed
significant improvement over time, with scores at T1 being significantly lower
than those at T0 (p
The MEP parameters for the three groups at T0 and T1 are summarized in Table 2
and Fig. 3C,D. Overall, the MEP amplitude significantly increased from T0 to T1
in all groups (p
At T1, this study calculated the Cohen’s D values for the changes in functional outcomes to assess the effect sizes of different indicators, with the effect sizes for each group summarized as follows (Table 2):
Sham rTMS Group: SSA score improvement demonstrated a medium effect size (D = 0.592); MEP amplitude showed large effects on both the unaffected side and affected side (D = 2.435 and 1.908, respectively); Changes in MEP latency were classified as a small effect (unaffected side D = 0.219; affected side D = 0.317).
Affected rTMS Group: SSA score improvement showed a large effect size (D = 0.969); MEP amplitude on both the unaffected side and affected side also exhibited large effects (D = 2.295 and 1.391, respectively); Changes in MEP latency remained a small effect (unaffected side D = 0.490; affected side D = 0.318).
Bilateral rTMS Group: SSA score improvement resulted in a large effect size (D = 2.339); MEP amplitude on both the unaffected side and affected side demonstrated large effects (D = 2.944 and 2.622, respectively); Notably, the MEP latency on the affected side achieved a large effect size (D = 0.810), while the change in latency on the unaffected side remained a small effect (D = 0.251).
The blinding assessment conducted after the first treatment session revealed
that the proportion of patients who correctly identified their actual group
assignment was 68% in the sham group, 60% in the affected rTMS group, and 76%
in the bilateral rTMS group. A chi-square test showed no statistically
significant difference in the accuracy rates among the three groups
(
All patients successfully completed the treatment, with no cases of dropout due to intolerance, indicating overall good tolerability. During the study, a total of 5 cases of transient scalp discomfort were reported (1 case in the sham rTMS group, 2 cases in the affected rTMS group, and 2 cases in the bilateral rTMS group). Symptoms were alleviated after pausing stimulation and taking a short break. No other serious adverse events, such as headache, epilepsy, tinnitus, or psychological discomfort, were observed.
This randomized controlled trial investigated the efficacy, safety, and underlying neural mechanisms of 10 Hz rTMS applied to the affected-side or bilateral swallowing cortical regions, combined with conventional rehabilitation, for functional recovery in patients with PSD. Utilizing a multidimensional assessment framework encompassing behavioral and neurophysiological outcomes, the study results indicate that: (1) while rTMS combined with conventional rehabilitation (regardless of stimulation target) effectively improved swallowing function in PSD patients, the bilateral rTMS group demonstrated superior comprehensive efficacy, showing significantly greater improvements in overall swallowing efficiency, reduction of aspiration risk, and decrease in pharyngeal residue compared to both the sham and affected rTMS groups; (2) neurophysiological data revealed that all interventions enhanced cortical excitability in swallowing-related areas, and the bilateral rTMS paradigm not only augmented excitability but also potentially optimized the conduction efficiency of neural pathways; (3) effect size analysis further supported these findings, with bilateral rTMS showing a “large effect” for improvement in the primary functional outcome; (4) regarding safety, no serious adverse events occurred with any intervention protocol, and all were well tolerated, confirming the safety of 10 Hz rTMS in this clinical application.
Swallowing function involves multi-stage coordinated activities from the oral cavity to the esophagus, relying on a complex neural network regulated by bilateral cerebral cortices, subcortical structures, the brainstem, and the cerebellum [31, 33]. Within this network, the bilateral cerebral hemispheres jointly control swallowing via corticofugal projections, typically with one hemisphere serving as the dominant side and the other playing a synergistic and compensatory role [11, 34]. When stroke injures key nodes of this network-such as the primary motor cortex, anterior insula, anterior cingulate cortex, or frontal operculum-or disrupts the white matter pathways connecting them, the integrity and coordination of the network are compromised. The core pathological mechanisms involve desynchronization of cortico-subcortical neural circuits and impaired neuromuscular control of muscles such as the suprahyoid group, ultimately leading to clinical symptoms including pharyngeal residue, aspiration, and delayed swallowing initiation [3, 35, 36]. This understanding of network dysfunction provides a theoretical basis for the use of neuromodulation techniques, such as rTMS, to target and repair neural circuits.
The bilateral rTMS intervention strategy adopted in this study was primarily
based on the theoretical frameworks of the interhemispheric competition model and
the dual-mode balance recovery model [9, 11, 17, 18]. Following stroke, the
excitability of the affected hemisphere decreases, while the unaffected
hemisphere may become hyperactive due to loss of interhemispheric inhibition,
thereby impeding the recovery of swallowing function [12]. In contrast to
conventional unilateral intervention approaches, bilateral high-frequency rTMS
(e.g., 10 Hz) simultaneously modulates excitability in both hemispheres: it
promotes functional reorganization in the affected hemisphere while suppressing
excessive activity in the unaffected hemisphere, ultimately restoring the overall
function of the swallowing network. As swallowing is a physiologically bilateral
cortical process, its neural network relies on compensatory mechanisms from the
unaffected hemisphere after unilateral damage [37, 38, 39]. Thus, bilateral
synchronous stimulation may activate potential compensatory pathways, yielding
synergistic effects. Although there is no unified standard for optimal rTMS
parameters in PSD treatment, high-frequency stimulation (
The findings of this study clearly demonstrate that bilateral high-frequency
rTMS offers significant advantages in improving swallowing function in patients
with PSD. Specifically: Comprehensive improvement in swallowing function: The
bilateral rTMS group showed significantly greater improvements in the SSA score
(reflecting overall swallowing ability), PAS score (reflecting swallowing
safety), and pharyngeal residue score (reflecting swallowing efficiency) compared
to both the sham stimulation group and the unilateral rTMS group. High clinical
response rate: Using a
Changes in MEPs provide an objective neurophysiological metric for elucidating the mechanisms of rTMS. The results of this study showed that the MEP amplitude significantly increased from baseline in all intervention groups after treatment, suggesting that conventional rehabilitation combined with rTMS may effectively enhance cortical excitability in swallowing-related areas. Notably, the bilateral rTMS group demonstrated a unique advantage: the improvement in MEP amplitude on the affected side was significantly greater than that in the affected-side rTMS group, indicating that bilateral stimulation may more effectively facilitate the recruitment and synchronized firing capacity of neural pathways in the affected hemisphere, potentially by modulating interhemispheric interactions [13, 14]. Particularly important is that only the bilateral rTMS group showed a significant shortening of MEP latency on the unaffected side. Given that latency reflects the neural conduction velocity from the cortex to the target muscles, improvement in this measure suggests that bilateral rTMS not only increased cortical excitability but may also have enhanced the conduction speed of swallowing-related neural pathways, possibly by optimizing myelination or synaptic transmission efficiency [45, 46, 47]. The underlying mechanisms may involve rTMS-induced modulation of neurotransmitter systems (e.g., GABA and glutamate) and upregulation of brain-derived neurotrophic factor expression, as observed in other studies [47, 48]. Therefore, bilateral rTMS may promote neural remodeling and functional recovery related to swallowing through a dual mechanism of “enhancing excitability” and “optimizing conductivity”.
All patients completed the treatment without any dropouts due to intolerance, indicating good overall tolerability of the intervention protocols across all groups. A total of five cases of transient scalp discomfort were reported among the three groups, with no significant difference in incidence rates between groups. Symptoms resolved after briefly pausing stimulation and allowing a short rest. No serious adverse events such as headache, seizures, tinnitus, or psychological discomfort were observed. These findings are consistent with the majority of reported studies, further supporting the favorable safety profile of rTMS in treating post-stroke dysphagia [49]. In this study, bilateral rTMS did not increase the risk of adverse reactions, demonstrating that both dual-target combined intervention and single intervention modalities possess reliable safety and clinical applicability [44].
This study has several limitations. First, the single-center design and the lack of subgroup analyses based on stroke type, location, or severity (SSA or FEES) may limit the generalizability of the findings. Future research should employ multicenter, large-sample randomized controlled trials incorporating machine learning-based patient stratification to further validate the efficacy of bilateral rTMS in PSD. Second, the 2-week intervention period without a follow-up assessment precludes evaluation of long-term effects. Subsequent studies should consider extending the intervention duration and incorporating follow-ups at 4, 8, and 12 weeks post-intervention (e.g., via telephone-administered SSA combined with on-site FEES or MEP assessments) to determine the persistence of therapeutic effects and neuroplastic changes. Furthermore, while MEP was used to localize stimulation hotspots, the absence of MRI neuronavigation may have affected targeting precision. Although MEP can reflect cortical excitability, its utility in elucidating underlying mechanisms remains limited. Future investigations should integrate multimodal techniques such as fNIRS, fMRI, and biomarkers to provide deeper insights into the neural mechanisms of swallowing function recovery.
It should be noted that studies have shown that acupuncture exerts independent effects on the central nervous system. In the present study, acupuncture was included as a fixed component of conventional rehabilitation therapy to simulate real-world clinical practice [24]. Consequently, the current study design does not allow for a precise delineation of the individual contributions of acupuncture and rTMS to the observed therapeutic outcomes. To more purely evaluate the incremental benefit of rTMS on top of conventional rehabilitation, future studies could consider a more rigorous three-arm trial design.
This study demonstrates that conventional rehabilitation combined with 10 Hz rTMS targeting the swallowing cortex effectively improves PSD, with the bilateral rTMS strategy yielding superior therapeutic outcomes. Specifically, bilateral rTMS significantly enhanced swallowing efficiency, swallowing safety, and pharyngeal residue clearance, with a large effect size observed for the primary functional outcome. The underlying mechanism may involve simultaneous enhancement of cortical excitability and optimization of neural conduction velocity, which collectively promote functional improvement and neuroplastic remodeling of swallowing-related neural pathways. All treatment regimens were well tolerated, with no severe adverse events reported, confirming their safety profile. In summary, bilateral 10 Hz rTMS, as a multi-target neuromodulation strategy that synchronously modulates the swallowing cortices of both hemispheres, demonstrates clear clinical benefits and holds promise for broader application. Future research should extend the intervention period, conduct multicenter large-sample randomized controlled trials, and integrate multimodal neuroimaging techniques to further validate its long-term efficacy and elucidate its neural mechanisms in greater depth.
PSD, Post-stroke dysphagia; WST, Water swallowing test; FEES, Fiberoptic endoscopic evaluation of swallowing; rTMS, Repetitive transcranial magnetic stimulation; SSA, Standardized swallowing assessment; PAS, Penetration aspiration scale; MEP, Motor evoked potential.
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
BH performed the experiments and wrote the paper. KP, RZ and XC analyzed and interpreted the data. YL and YW contributed data analysis. YX conceived and designed the experiments. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was performed in accordance with the Declaration of Helsinki, and was reviewed and approved by the Medical Ethics Committee of the Affiliated Hospital of North Sichuan Medical College, with approval numbers 2023ER031-1. Additionally, it has completed registration with the Chinese Clinical Trial Registry, with the registration number ChiCTR2300068730. All participants/patients (or their proxies/legal guardians) provided informed consent to participate in the study.
We gratefully acknowledge all participants for their involvement in this study.
This study was supported by the Sichuan Rehabilitation Medical Association Scientific Research Project [SCKFKY20250204] and Sichuan Provincial Medical Science and Technology Innovation Research Association Research Project [YCH-KY-YCZD2024-160].
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RN49912.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



