1 Geriatrics Ward, Jiangsu Rongjun Hospital, 214062 Wuxi, Jiangsu, China
Abstract
Anxiety symptoms in elderly patients with cognitive impairment (CI) often reflect shared neurobiological processes rather than distinct psychiatric disorders. Current diagnostic approaches lack objective biomarkers for early identification. This study investigated the association between serum biomarkers and anxiety disorder status in elderly men with CI and to evaluate the exploratory discriminative ability of cognitive domains and biomarker profiles in differentiating CI patients with and without comorbid anxiety.
This cross-sectional retrospective study analyzed 86 elderly male CI patients (Group A: CI alone, n = 41; Group B: CI with anxiety, n = 45) at Jiangsu Rongjun Hospital (June–December, 2024). Anxiety disorder diagnosis was established through structured clinical interviews based on the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria conducted by two independent psychiatrists, with the Hamilton Anxiety Scale (HAMA) serving as an initial severity screening instrument. The Montreal Cognitive Assessment (MOCA) was used to evaluate cognitive function. Enzyme-linked immunosorbent assay (ELISA) was used to measure serum Tau protein (Tau), β-amyloid (Aβ), visinin-like protein 1 (VILIP-1), malondialdehyde (MDA), tumor necrosis factor-α (TNF-α), and interleukin-6 (IL-6); reverse transcription-polymerase chain reaction (RT-PCR) quantified microRNA-34c (MiR-34c). Patients with acute inflammation (C-reactive protein [CRP] >10 mg/L) were excluded. Bonferroni correction was used to address multiple comparisons across 25 simultaneous tests, and multivariate regression analysis was controlled for demographic and clinical confounders. Receiver operating characteristic (ROC) analysis was used to determine the discriminative ability.
Group B showed worse cognitive performance across the MOCA domains, with attention (area under the curve [AUC] = 0.738) and delayed recall (AUC = 0.742) demonstrating the strongest discriminative ability. Biomarker analysis revealed elevated Tau (AUC = 0.957), MDA (AUC = 0.941), and VILIP-1 (AUC = 0.914) in anxiety patients. Within-group analyses showed that anxiety severity correlated negatively with MiR-34c and positively with Tau, Aβ, MDA, IL-6, and VILIP-1. Under the Bonferroni-adjusted threshold (p < 0.002), only MDA in Group B (r = 0.478, p = 0.001) and MiR-34c in Group B (r = –0.523, p < 0.001) remained significant. Multivariate analysis identified these factors as independently associated with the outcome after controlling for demographics and comorbidities. However, given the substantial baseline imbalances between the groups, these associations should be interpreted with caution.
Combined cognitive assessment (attention, delayed recall) and serum biomarkers (Tau, MDA, VILIP-1, MiR-34c) demonstrate promising discriminative ability for identifying anxiety in elderly male patients with CI. These findings are exploratory and derived from a single-center cohort of retired male military veterans with pronounced baseline group imbalances, which substantially limits generalizability to the broader elderly CI population. The identified markers may reflect shared neuroinflammatory and oxidative stress pathways underlying both cognitive and emotional dysfunction, warranting further investigation as potential targets for integrated therapeutic approaches. Validation in prospective, multicenter, sex-inclusive cohorts with balanced comparison groups is essential before any clinical application can be considered.
Keywords
- aged
- cognitive dysfunction
- anxiety disorders
- Mental Status and Dementia Tests
- biomarkers
- cross-sectional studies
Anxiety disorders are prevalent among the elderly, though reported prevalence rates vary considerably depending on the population studied and the diagnostic criteria employed. In community-dwelling elderly populations, systematic reviews and meta-analyses have reported prevalence estimates ranging from 3.2% to 14.2%, while rates are substantially higher in clinical and institutionalized settings, where estimates may exceed 20%–28% in populations with comorbid chronic medical conditions [1, 2]. While prior research has predominantly examined younger populations, evidence suggests that anxiety disorders in elderly individuals cause greater psychological and physical distress, severely impacting their social engagement and mental well-being [3]. Therefore, early identification and intervention for anxiety disorders have become critical in clinical management.
The relationship between cognitive impairment (CI) and anxiety disorders in elderly populations is complex and bidirectional [4, 5]. Neurobiologically, both conditions share common pathophysiological mechanisms, including dysfunction in prefrontal-limbic circuits, dysregulation of neurotransmitter systems (particularly serotonin, gamma-aminobutyric acid (GABA), and dopamine), and alterations in the hypothalamic-pituitary-adrenal axis [6, 7, 8, 9]. In elderly patients with CI, structural brain changes in regions such as the amygdala, hippocampus, and prefrontal cortex can precipitate anxiety symptoms, while chronic anxiety may accelerate cognitive decline through sustained stress responses and inflammatory cascades [10, 11].
The temporal relationship between CI and anxiety is particularly relevant in clinical practice. While anxiety can manifest as a reaction to cognitive decline awareness, it can also represent an early neuropsychiatric symptom of underlying neurodegenerative processes [12]. Studies have shown that anxiety disorders in patients with mild cognitive impairment increase the risk of progression to dementia, suggesting shared pathological mechanisms [13, 14]. Furthermore, the cognitive domains most affected by anxiety disorders—attention, executive function, and memory—overlap significantly with those impaired in CI, creating a synergistic effect that compounds functional disability [14]. Importantly, in the context of neurodegenerative diseases, anxiety symptoms may constitute behavioral and psychological symptoms of dementia (BPSD) rather than primary anxiety disorders, and distinguishing between these entities remains a significant clinical challenge [13, 14].
Current diagnostic approaches, which rely on medical history and psychological scales, are subjective and often lead to misdiagnosis or delayed detection. Elderly patients are frequently diagnosed only after the manifestation of severe symptoms [5]. This challenge is compounded by the fact that anxiety symptoms in elderly patients with CI may be attributed to cognitive decline itself, leading to under-recognition and under-treatment [15, 16]. Although extensive research has explored the neurophysiological mechanisms and risk factors associated with anxiety disorders, there is limited research on early screening using serum biomarkers [6]. Thus, the development of objective assessment methods to identify high-risk individuals early is imperative.
Recent advancements in biomarker detection technologies have highlighted the
potential of serum markers in the diagnosis of neurological disorders [7, 10].
Tau protein (Tau) and
This study sought to analyze clinical data and serum samples from elderly male CI patients to identify biomarkers associated with anxiety disorders, offering novel perspectives for the characterization and future investigation of early detection and intervention in elderly men.
This study included elderly male CI patients undergoing short-term
rehabilitation between June and December 2024. A priori power analysis indicated
that a sample size of 80 participants (40 per group) would provide 80% power to
detect a medium effect size (Cohen’s d = 0.65) with
Participants met the following criteria: (1) Age
Exclusion criteria included: (1) Comorbid malignancies or terminal illnesses;
(2) History of anxiety, depression, or other psychiatric disorders prior to CI
onset; (3) History of traumatic brain injury, hemorrhagic stroke, or Parkinson’s
syndrome; (4) Acute inflammatory conditions (C-reactive protein [CRP]
Patients were consecutively enrolled upon admission for short-term rehabilitation. Cognitive impairment was identified using the MOCA assessment conducted during the admission evaluation. The enrolled population comprised patients whose cognitive symptoms were identified either during the current admission or through prior clinical records; however, the retrospective nature of the study limited the precise determination of disease duration for all participants. All participants underwent the anxiety assessment protocol described below at enrollment.
The Hamilton Anxiety Scale (HAMA) [10] was used as an initial screening
instrument for anxiety symptom severity. The HAMA is a clinician-administered
rating scale comprising 14 items, each scored on a 0–4 scale. A total score
A structured questionnaire was used to collect data on age, marital status, education, income, family structure, body mass index (BMI), cardiac function, smoking, alcohol use, trauma history, and medical history (hypertension, diabetes, coronary heart disease, and ischemic stroke). Fasting venous blood samples were collected after 12 h to measure blood cell counts, total bilirubin (TBIL), alanine aminotransferase (ALT), aspartate aminotransferase (AST), total cholesterol (TC), triglycerides (TG), low-density lipoprotein cholesterol (LDL-C), urea nitrogen (UREA), creatinine (CRE), uric acid (UA), fasting blood glucose (FBG), CRP, and homocysteine (HCY) levels.
Serum concentrations of Tau, A
Quantification of MiR-34c in serum was performed using reverse
transcription-polymerase chain reaction (RT-PCR) kits were obtained from Tianenze
Biotech Co., Ltd. (Cat. No. TNZ-M3421; Shanghai, China), adhering to the
manufacturer’s protocol: (1) drawing 3 mL of venous blood after fasting,
separating serum, and preserving at –80 °C; (2) isolating total RNA,
measuring concentration and purity via absorbance at 260 nm and 280 nm; (3)
Conducting RT-PCR following polyadenylation of miRNA, cDNA generation, and 40
cycles of amplification (95 °C for 15 s, 60 °C for 1 min).
Relative MiR-34c expression was determined using the
Analysis of data utilized SPSS 22.0 software (IBM Corp., Armonk, NY, USA). The
normality of continuous variables was assessed using the Shapiro-Wilk test.
Continuous measurements are presented as mean
Prior to presenting the outcome comparisons, the baseline demographic and
clinical characteristics of the study population were described. A total of 86
elderly male patients with CI were included, with 41 in Group A (CI alone) and 45
in Group B (CI with comorbid anxiety disorder). The exclusively male composition
of this cohort reflects the institutional setting: Jiangsu Rongjun Hospital
primarily serves retired military veterans, who are overwhelmingly male in the
current Chinese elderly population. During the study period, too few female CI
patients met the inclusion criteria to permit meaningful sex-stratified analysis;
the implications of this restriction are addressed in the Limitations section. As
presented in Table 1, substantial baseline differences were observed between
groups. Group B had a significantly higher proportion of patients aged
| Project | Group A (n = 41) | Group B (n = 45) | p | ||
| Age | 38.585 | ||||
| 10 (24.39) | 30 (66.67) | ||||
| 31 (75.61) | 15 (33.33) | ||||
| Marriage | 0.700 | 0.403 | |||
| Normal [case count (%)] | 40 (97.56) | 43 (95.56) | |||
| Widowed and divorced [case count (%)] | 1 (2.44) | 2 (4.44) | |||
| Education | 5.281 | 0.022* | |||
| Be educated |
28 (68.29) | 37 (82.22) | |||
| Be educated |
13 (31.71) | 8 (17.78) | |||
| Income | 0.023 | 0.879 | |||
| 13 (31.71) | 14 (31.11) | ||||
| 28 (68.29) | 31 (68.89) | ||||
| Family structure | 2.033 | 0.154 | |||
| Large family type [case count (%)] | 25 (60.98) | 23 (51.11) | |||
| Intermediate type [case count (%)] | 16 (39.02) | 22 (48.89) | |||
| BMI | 0.523 | 0.469 | |||
| 15 (36.59) | 19 (42.22) | ||||
| 26 (63.41) | 26 (57.78) | ||||
| Heart function | 4.775 | 0.029* | |||
| NYHA classification |
19 (46.34) | 14 (31.11) | |||
| NYHA classification |
22 (53.66) | 31 (68.89) | |||
| Smoking habit [case count (%)] | 20 (48.78) | 29 (64.44) | 4.596 | 0.032* | |
| Drinking habit [case count (%)] | 7 (17.07) | 10 (22.22) | 0.798 | 0.372 | |
| History of trauma [case count (%)] | 14 (34.15) | 24 (53.33) | 7.393 | 0.007* | |
| Combined hypertension [case count (%)] | 20 (48.78) | 31 (68.89) | 7.487 | 0.006* | |
| Combined diabetes [case count (%)] | 9 (21.95) | 23 (51.11) | 18.525 | ||
| Combined coronary heart disease [case count (%)] | 6 (14.63) | 12 (26.67) | 4.389 | 0.036* | |
| Combined ischemic stroke [case count (%)] | 1 (2.44) | 7 (15.56) | 13.473 | ||
CI, cognitive impairment; BMI, body mass index. NYHA, New York Heart
Association. *p
Group A demonstrated higher MOCA total scores and better performance in
visuospatial/executive function, attention, language, abstraction, and delayed
recall than Group B. Naming and orientation showed no significant differences.
HAMA scores confirmed elevated anxiety in Group B (Table 2; p
| Project | Group A (n = 41) | Group B (n = 45) | t/z | p |
| Total score of MOCA | 24.15 |
19.73 |
–7.661 | |
| Visuospatial and executive abilities | 3.49 |
2.76 |
–3.451 | |
| Naming | 2.71 |
2.73 |
–0.299 | 0.765 |
| Attention | 5.02 |
3.84 |
–7.922 | |
| Language | 2.51 |
1.93 |
–3.611 | |
| Abstraction | 1.56 |
1.00 |
–3.761 | |
| Delayed recall | 2.32 |
1.09 |
–4.631 | |
| Orientation | 5.83 |
5.67 |
–1.570 | 0.117 |
| HAMA | 5.24 |
18.67 |
–8.091 |
MOCA, Montreal Cognitive Assessment; HAMA, Hamilton Anxiety Scale. *p
Within-group correlation analysis revealed significant negative correlations
between anxiety levels and MOCA total scores (Group A: r = –0.423, p
| Project | Total score of MOCA | Visuospatial and executive | Attention | Language | Abstraction | Delayed recall | |
| Group A HAMA | |||||||
| r | –0.423 | –0.287 | –0.389 | –0.245 | –0.298 | –0.345 | |
| p | 0.006* | 0.068 | 0.012* | 0.121 | 0.059 | 0.028* | |
| Group B HAMA | |||||||
| r | –0.456 | –0.312 | –0.421 | –0.289 | –0.334 | –0.398 | |
| p | 0.002* | 0.037* | 0.005* | 0.055 | 0.024* | 0.007* | |
*Significant at p
ROC analysis evaluated the discriminative ability of cognitive measures in
differentiating between patients with CI with and without comorbid anxiety
disorder. ROC analysis indicated that the MOCA total scores,
visuospatial/executive function, attention, language, abstraction, and delayed
recall had area under the curve (AUC) values of 0.964, 0.693, 0.738, 0.661,
0.663, and 0.742, respectively, supporting their discriminative utility. MOCA
total scores showed excellent discrimination (AUC = 0.964, 95% CI: 0.921–1.000,
p
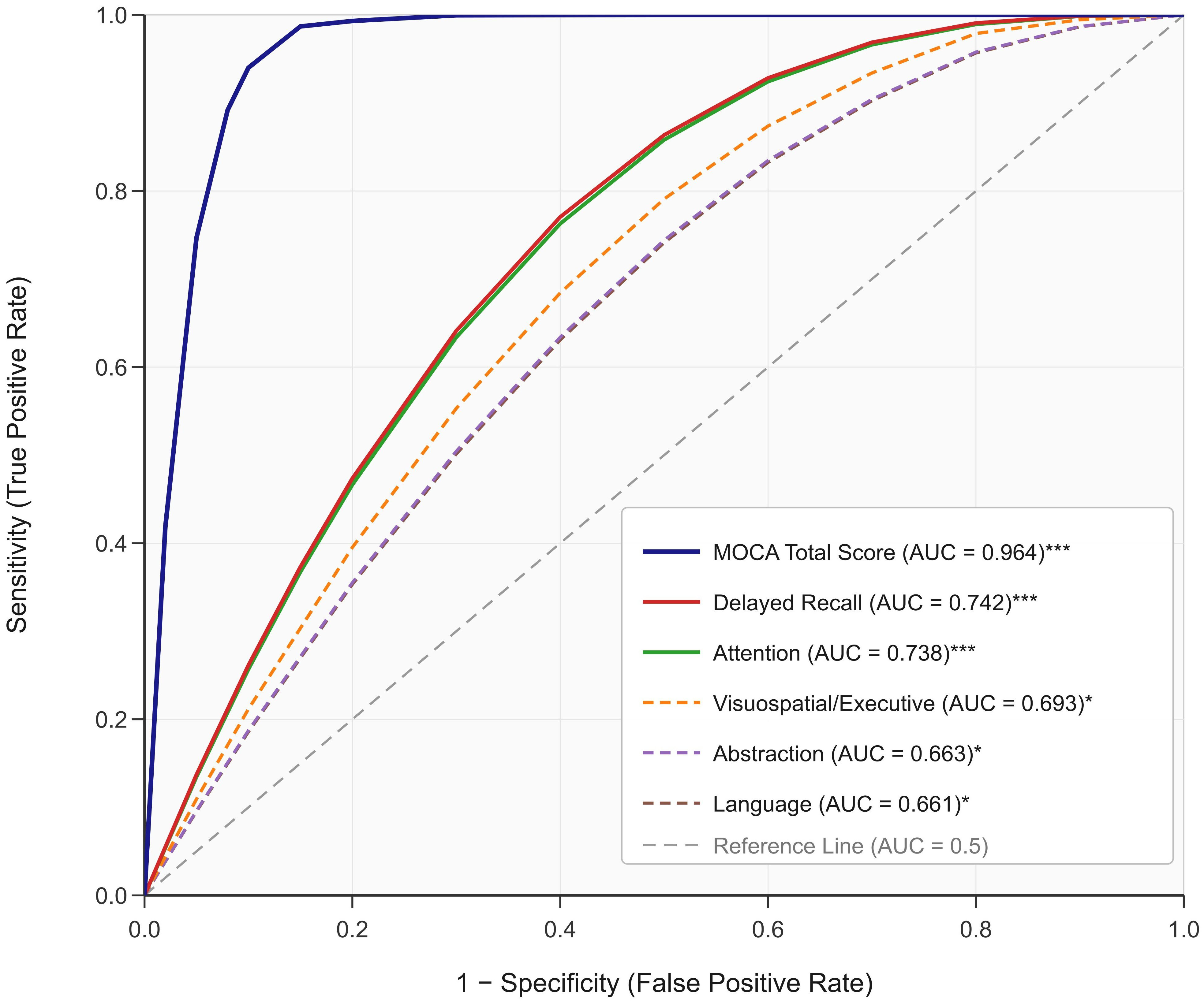 Fig. 1.
Fig. 1.
ROC curve of cognitive function assessment factors for
discriminating anxiety disorder status. Receiver operating characteristic curves
are shown for the MOCA total score and six cognitive subdomains. The diagonal
dashed reference line represents chance discrimination (AUC = 0.5). Solid lines
denote measures with AUC
Group A showed higher lymphocyte count (LYM) and MiR-34c levels, while Group B
had elevated UREA, HCY, Tau, A
| Project | Group A (n = 41) | Group B (n = 45) | t/z | p |
| Hemoglobin [Hb] (g/L) | 150.05 |
145.87 |
–1.138 | 0.255 |
| Lymphocyte count [LYM] (109/L) | 1.86 |
1.62 |
2.430 | 0.017* |
| Platelet count [PLT] (109/L) | 197.71 |
192.42 |
0.466 | 0.643 |
| TBIL (µmol/L) | 14.98 |
15.48 |
–0.553 | 0.580 |
| ALT (U/L) | 21.53 |
22.53 |
–0.368 | 0.713 |
| AST (U/L) | 20.41 |
23.20 |
–1.077 | 0.282 |
| TC (mmol/L) | 5.57 |
5.76 |
–0.601 | 0.548 |
| TG (mmol/L) | 1.64 |
1.26 |
–1.807 | 0.071 |
| LDL-C (mmol/L) | 2.85 |
2.86 |
–0.007 | 0.994 |
| UREA (mmol/L) | 6.20 |
7.01 |
–2.265 | 0.026* |
| CRE (µmol/L) | 79.59 |
83.26 |
–1.288 | 0.198 |
| UA (µmol/L) | 351.83 |
334.60 |
–0.968 | 0.333 |
| FBG (mmol/L) | 6.05 |
5.90 |
–0.636 | 0.525 |
| HCY (µmol/L) | 14.34 |
16.22 |
–2.504 | 0.012* |
| Tau (ng/L) | 167.66 |
200.86 |
–9.584 | |
| A |
152.78 |
169.97 |
–7.017 | |
| SOD (U/mL) | 55.78 |
53.59 |
–1.634 | 0.102 |
| MDA (µmol/L) | 2.69 |
3.67 |
–9.839 | |
| TNF- |
73.93 |
85.04 |
–4.657 | |
| IL-6 (ng/L) | 128.75 |
161.39 |
–5.789 | |
| VILIP-1 (pg/mL) | 491.13 |
642.75 |
–6.610 | |
| MiR-34c | 2.78 |
2.39 |
8.578 |
TBIL, total
bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TC,
total cholesterol; TG, triglycerides; LDL-C, low-density lipoprotein cholesterol;
UREA, urea nitrogen; CRE, creatinine; UA, uric acid; FBG, fasting blood glucose;
HCY, homocysteine; Tau, Tau protein; A
Within-group correlation analysis showed that anxiety levels correlated negatively with MiR-34c (Group A: r = –0.456, p = 0.003; Group B: r = –0.523, p
| Project | LYM | UREA | Tau | A |
MDA | HCY | TNF- |
IL-6 | VILIP-1 | MiR-34c | |
| Group A HAMA | |||||||||||
| r | –0.298 | 0.156 | 0.398 | 0.367 | 0.423 | 0.234 | 0.289 | 0.345 | 0.412 | –0.456 | |
| p | 0.058 | 0.329 | 0.010* | 0.018* | 0.006 | 0.140 | 0.067 | 0.027* | 0.007 | 0.003 | |
| Group B HAMA | |||||||||||
| r | –0.234 | 0.167 | 0.445 | 0.412 | 0.478 | 0.298 | 0.334 | 0.389 | 0.456 | –0.523 | |
| p | 0.125 | 0.272 | 0.002 | 0.005* | 0.001† | 0.047* | 0.024* | 0.008* | 0.002 | 0.000† | |
†Significant after Bonferroni correction (p
Multivariate logistic regression analysis, adjusting for age, education,
smoking, and comorbidities, identified the following factors independently
associated with anxiety disorders: Tau (odds ratio [OR] = 1.15, 95% CI:
1.08–1.23, p
ROC analysis was used to evaluate the discriminative performance of serum
biomarkers in differentiating patients with CI with and without comorbid anxiety
disorders. ROC analysis revealed that LYM, UREA, Tau, A
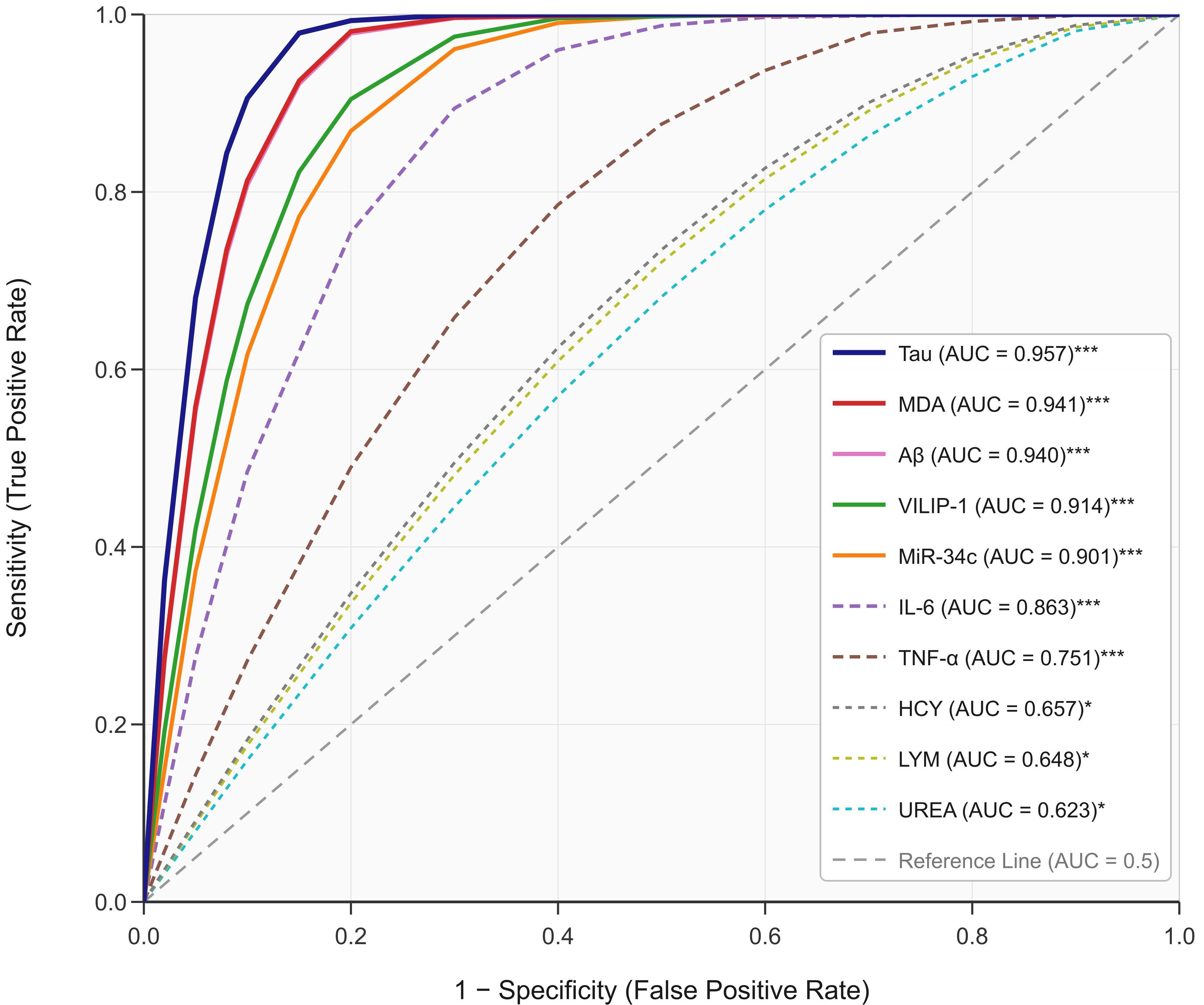 Fig. 2.
Fig. 2.
ROC curve of serum biomarkers for discriminating anxiety
disorder status. Receiver operating characteristic curves are shown for ten
serum biomarkers. The diagonal dashed reference line represents chance
discrimination (AUC = 0.5). Solid lines denote biomarkers with AUC
Understanding anxiety symptoms in elderly patients with cognitive impairment requires recognizing that both conditions often reflect shared neurobiological processes rather than representing distinct entities [13, 14]. In the context of neurodegenerative changes affecting elderly individuals, anxiety symptoms frequently emerge as neuropsychiatric manifestations of the same pathological mechanisms that drive cognitive decline.
The prefrontal-limbic circuits governing both cognitive processing and emotional regulation show overlapping dysfunctions in CI and anxiety disorders [15, 17]. A related diagnostic consideration is that the HAMA, while supplemented by DSM-5–based clinical interviews in this study, was originally developed for populations without significant cognitive impairments. In elderly patients with CI, somatic symptoms common to both conditions, such as sleep disturbance, fatigue, and concentration difficulties, may overlap, complicating the distinction between primary anxiety and anxiety arising as a behavioral and BPSD [13]. Although our exclusion of patients with pre-existing psychiatric histories was designed to reduce this ambiguity, the cross-sectional design cannot establish a temporal sequence between anxiety and cognitive decline. This diagnostic uncertainty should be considered when interpreting the associations reported below. When neurodegenerative processes affect these integrated networks, symptoms can manifest across both cognitive and emotional domains, explaining why anxiety symptoms are particularly prevalent in elderly CI patients and why our study found that Group B exhibited more severe deficits in multiple cognitive domains.
Our findings revealed that patients with CI and comorbid anxiety demonstrated
significantly worse performance across most MOCA domains, with attention and
delayed recall demonstrating the strongest discriminative ability (AUC
An important interpretative consideration is the possibility of bidirectional measurement bias. Anxiety can impair attentional resources, working memory capacity, and executive function, potentially resulting in lower MOCA scores that do not solely reflect underlying neurodegenerative pathology. This phenomenon has been well documented in the cognitive psychology literature, where state anxiety has been shown to consume working memory resources and disrupt prefrontal cortical efficiency. Consequently, the observed association between lower cognitive scores and higher anxiety severity may be partially attributable to the acute effects of anxiety on test performance rather than exclusively reflecting shared neurodegeneration. This bidirectional relationship represents a fundamental limitation of cross-sectional assessments and can only be disentangled through longitudinal designs with repeated cognitive evaluations under varying anxiety states.
The biomarker findings provide potential mechanistic insights into the shared
pathophysiology underlying CI-anxiety comorbidity, although these interpretations
remain speculative given the cross-sectional design. The inflammatory hypothesis
of neuropsychiatric disorders offers a framework for understanding these
observations [19, 20]. Elevated IL-6 and TNF-
The significantly elevated Tau and A
Oxidative stress represents another convergent pathway, as evidenced by markedly elevated MDA levels in the anxiety group. This finding reflects compromised cellular antioxidant defenses and increased lipid peroxidation [24, 25]. MDA retained a strong independent association with anxiety status in the multivariate model (OR = 3.45), suggesting that oxidative stress may represent a more robust biological correlate of anxiety in this population, although confirmatory longitudinal studies are needed.
VILIP-1 emerged as one of our strongest discriminators (AUC = 0.914), consistent with its role as a sensitive marker of neuronal calcium dysregulation and cellular stress [26, 27]. Its elevation suggests widespread neuronal vulnerability extending beyond traditional cognitive networks to encompass the emotional processing regions. Similarly, the significant reduction in MiR-34c levels in patients with anxiety may represent impaired stress resilience mechanisms and reduced neuroprotective capacity [28]. MiR-34c demonstrated the strongest independent association with anxiety in the multivariate model (OR = 0.12), suggesting that it may be a particularly informative molecular marker worthy of further investigation in prospective studies. Our findings are broadly consistent with emerging evidence implicating miRNA dysregulation in neuropsychiatric conditions, as reviewed by Albano et al. [28], although direct comparisons are limited by differences in study populations and biomarker panels.
Our multivariate analysis, incorporating the Bonferroni correction for multiple
testing, identified independent biomarker factors after controlling for
demographic and clinical confounders. The persistence of significant associations
for the oxidative stress indicator MDA and regulatory microRNA MiR-34c suggests
that these may represent informative biological correlates rather than secondary
effects, although validation in independent cohorts is essential. It must be
acknowledged, however, that the pronounced baseline imbalances between the study
groups—particularly the nearly threefold difference in the proportion of
patients aged
Regarding the generalizability of these findings, the restriction to male participants requires careful consideration. Sex differences in anxiety disorders and cognitive impairment are well documented, encompassing hormonal influences (e.g., estrogen-mediated neuroprotection), differential inflammatory and oxidative stress profiles, and variations in hypothalamic-pituitary-adrenal axis reactivity (HPA). These factors may substantially modify the biomarker associations observed in the present study, and our results should not be extrapolated to the female population. Beyond the sex restriction, the derivation of our study population from a single military veteran rehabilitation facility introduces a further dimension of selection that merits explicit consideration. Retired military veterans may carry a distinctive burden of cumulative psychological stress—including potential combat-related trauma exposure—that could independently modulate inflammatory, oxidative stress, and neurodegeneration biomarker profiles. Moreover, this population may differ from community-dwelling elderly individuals in terms of healthcare access patterns, medication use, lifestyle factors, and the nature and severity of chronic medical comorbidities. These selection-related characteristics represent sources of potential bias that are not captured by the covariates included in our regression model, and they further limit the extent to which the present findings can be extrapolated to the broader elderly CI population. Multicenter studies incorporating both sexes and adequate power for sex-stratified analyses are a critical priority for future research.
Future investigations would also benefit from employing more comprehensive neuropsychological batteries beyond the MOCA screening tool. While the MOCA is well-validated and appropriate for the clinical screening context of this study, detailed assessments of episodic memory (e.g., the Rey Auditory Verbal Learning Test), executive function (e.g., the Trail Making Test), language (e.g., the Boston Naming Test), and processing speed would provide more granular cognitive profiling and potentially improve the specificity of cognitive biomarker assessment approaches.
The clinical implications include the potential future development of biomarker-based screening approaches for anxiety identification in elderly CI populations, although the present findings are preliminary and hypothesis-generating. If validated in prospective studies with external cohorts, the identification of specific pathways (inflammation, oxidative stress, and neurodegeneration) could provide targets for therapeutic approaches, such as anti-inflammatory strategies and neuroprotective interventions.
This study had several limitations that warrant consideration. The cross-sectional retrospective design precludes causal or temporal inference, meaning that the observed associations cannot be interpreted as predictive or directional. Although DSM-5–based clinical interviews supplemented HAMA screening, the inability to definitively distinguish primary anxiety from BPSD-related symptomatology remains an inherent limitation in this cognitively impaired population. The male-only sample from a single military veteran rehabilitation facility limits generalizability to women and non-veteran populations, given the well-documented sex differences in anxiety epidemiology, hormonal neuroprotection, inflammatory profiles, and stress reactivity. As a highly selected subgroup of retired military veterans, this population may carry distinctive psychological stress exposure histories and comorbidity profiles that limit the generalizability of these findings to the broader elderly CI population. The modest sample size, despite adequate a priori power, raises concerns regarding overfitting in the multivariate model. Pronounced baseline imbalances in age, education, and comorbidity burden between groups represent a major concern; although multivariate adjustment was performed, regression adjustment alone may not fully resolve structural non-comparability of this magnitude, and residual confounding cannot be excluded from the results. The absence of external validation, moderate intercorrelation among biomarkers (as reflected in the VIF values and correlation matrix), and potential influence of subclinical inflammation on biomarker levels further limit the robustness and generalizability of these findings. Finally, the bidirectional relationship between anxiety and cognitive test performance complicates the interpretation of cognitive-anxiety associations, as anxiety may independently impair MOCA performance beyond any shared neurodegenerative substrate.
Anxiety symptoms in elderly men with cognitive impairment may reflect shared neurobiological processes rather than separate psychiatric disorders. MOCA attention and delayed recall domains, combined with serum biomarkers (Tau, MDA, VILIP-1, and MiR-34c), demonstrated the ability to discriminate anxiety status in this population. The inflammatory and oxidative stress pathways identified warrant further investigation as potential therapeutic targets for integrated cognitive-emotional interventions in elderly patients with cognitive impairment and comorbid anxiety. Clinicians should consider routine anxiety screening in elderly cognitive impairment patients using both cognitive assessment and biomarker profiling, pending validation in larger prospective studies. These findings are hypothesis-generating and require confirmation prior to clinical implementation. Given the single-center military veteran origin of this cohort and the pronounced baseline imbalances between groups, replication in diverse, multicenter, sex-inclusive populations with balanced comparison groups is essential before clinical translation can be considered.
This cross-sectional study identified associations between CI and anxiety disorders, demonstrating that MOCA scores (attention, delayed recall) and serum biomarkers (Tau, MDA, VILIP-1, and MiR-34c) exhibit discriminative ability for anxiety status in elderly men. These findings are consistent with a neurobiological model of CI-anxiety comorbidity involving inflammatory, oxidative stress, and neuronal damage pathways. However, these findings should be regarded as hypothesis-generating rather than confirmatory findings. Importantly, these results derive from a single-center cohort of retired male military veterans with pronounced baseline group imbalances, substantially limiting generalizability to the broader elderly CI population and necessitating that all reported biomarker associations be interpreted as exploratory. Limited by its cross-sectional design, use of severity screening rather than comprehensive diagnostic criteria for anxiety, inability to distinguish primary anxiety from BPSD-related symptomatology, small sample size, pronounced baseline imbalances between groups, male-only single-center focus, and absence of external validation, further prospective, longitudinal multi-center studies with comprehensive psychiatric assessment and sex-inclusive cohorts are needed to validate these findings before any clinical application can be considered.
The data presented in this study are available on reasonable request from the corresponding author.
XC, TL, JWZ, WZ, and JYD contributed to the study conception and design. Material preparation, data collection, and analysis were performed by XC, WZ, JWZ, TL, and JYD. The first draft of the manuscript was written by XC. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Ethics Committee of Jiangsu Rongjun Hospital (Ethics No. 20250925). We confirm that informed consent was obtained from all participants or their legal guardians. This study was conducted in accordance with the Declaration of Helsinki.
Not applicable.
This research was funded by the Research Project of Wuxi Municipal Health Commission (Major Project, Grant No. Z202322) and the Research Project of Wuxi City Science and Technology Association (Soft Project, Grant Nos. KX-25-A12 and KX-24-B24).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RN48908.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


