1 Headache Unit, Neurology Department, Vall d'Hebron University Hospital, 08035 Barcelona, Spain
2 Headache Research Group, Vall d'Hebron Research Institute, Autònoma University of Barcelona, 08035 Barcelona, Spain
Abstract
Chronic migraine is a disabling and prevalent neurological disease that significantly impacts patients' quality of life. OnabotulinumtoxinA (OnabotA) is a safe and effective chronic migraine preventive treatment. The objective was to evaluate adherence to the recommended time interval between consecutive OnabotA injections according to the Phase III Research Evaluating Migraine Prophylaxis Therapy (PREEMPT) protocol, and to analyze changes in the interval adherence before and after the introduction of an Advanced Practice Nurse (APN) in a specialized Headache Unit of a tertiary hospital.
This was a retrospective study performed in a Spanish specialized Headache Clinic. Patients diagnosed with chronic migraine and treated with OnabotA following the PREEMPT protocol were included, before and after the introduction of APNs. Treatment was considered interval-compliant if the injection interval ranged from 75 to 105 days. The data collected included three 18-month periods. Statistical analyses examined differences across periods using R-Studio.
A total of 2991 participants were included, of whom 83.8% (2507/2991) were women, with a median age [interquartile range (IQR)] of 48.0 [40.0–57.0] years old. A statistically significant association between the introduction of the APN and improved adherence to the PREEMPT protocol was observed over time (Z = –19.60, p < 0.001). Adherence rates increased from 52.1% in P1 to 76.1% in both P2 and P3. The median time between visits decreased from 105 days in P1 to 96 days in P2, with a slight increase to 98 days in P3.
The involvement of advanced practice nurses in OnabotA administration decreased the time interval between infiltrations and improved adherence to the PREEMPT protocol. These findings highlight the APN's role in optimizing patient care.
Keywords
- migraine
- botulinum toxin type A
- treatment
- headache
- advanced practice nursing
Chronic migraine (CM) is a debilitating, prevalent, neurological disease that significantly impacts patients’ quality of life [1]. Migraine affects over 1 billion people worldwide and in Spain, it affects 5 million people, of which 500,000 suffer CM. For this reason, specific headache consultations are usually very overburdened [2, 3]. In this population, preventive treatment is mandatory and is key to reducing the burden in people with CM [4]. OnabotulinumtoxinA (OnabotA) is an effective and safe preventive treatment for CM reducing headache frequency, medication use, and improving quality of life as demonstrated by the PREEMPT studies which used a 155 U to 195 U fixed-dose protocol with pericranial subcutaneous injections every 12 weeks [5]. In Spain, OnabotA was approved and financed in 2012 for the preventive treatment of CM and is widely administered by neurologists specializing in headache management [6].
An Advanced Practice Nurse (APN) is a highly trained nurse with advanced clinical skills, capable of managing illnesses, providing specialized health education to society, overseeing chronic disease treatment, and continuously updated with recent technological advancements, methodologies, and healthcare practices [7]. If individuals respond to OnabotA treatment, the administration and evaluation of OnabotA is done every 12 weeks. This creates increasing demand among responders who require treatment, especially straining procedural capacity within Headache Clinics. The growing number of patients requiring a 3-month follow-up also increases the need for patient education and monitoring. An APN was recruited and incorporated into the team. This change was evaluated by comparing it to previous clinical practice, including adherence to the Phase III Research Evaluating Migraine Prophylaxis Therapy (PREEMPT) protocol schedule.
In 2019, an APN joined the Headache Unit as part of an organizational initiative to improve access to care for patients with chronic migraine. The nurse received direct training from neurologists specialized in headache medicine and was closely supervised during the initial phase until formal delegation of the OnabotA administration technique was approved. Additionally, the APN completed the Headache Specialization Course of the Catalan Society of Neurology, ensuring the necessary theoretical and practical competence for performing the procedure.
Hence, the objective was to evaluate adherence to the recommended time interval between consecutive OnabotA injections according to the PREEMPT protocol, and to analyze changes in the interval adherence before and after the introduction of an APN in a specialized Headache Unit of a tertiary hospital.
This is a retrospective study performed in a Spanish specialized Headache Clinic.
We included adult patients attended at the Headache Outpatient Clinic diagnosed
with CM and treated with OnabotA following the PREEMPT protocol. For the outlined
objective, the variables extracted from the electronic medical record system were
the patient record number, age, sex, visit dates of each OnabotA administration,
and the healthcare professional (neurologist or APN) who performed the procedure.
Treatment was considered interval-compliant with the PREEMPT protocol if the
interval between injections ranged from 75 to 105 days. Although the PREEMPT
protocol recommends administration every 12 weeks, in clinical practice these
intervals is operationalized as approximately three months (90 days). To account
for real-world scheduling variability, a
Statistical analysis was conducted using R-Studio v4.1.2 (The R Foundation for Statistical Computing, Vienna, Austria; https://www.r-project.org). Nominal variables (sex, healthcare professional, and time-intervals) were presented as frequencies (percentages), while median and interquartile range (IQR) were reported for quantitative variables, including age and waiting time (in days) between OnabotA visits. Normality of quantitative variables was confirmed through Q-Q plots and the Shapiro-Wilk test. Differences between healthcare professionals (neurologist vs. APN) were evaluated using an unpaired t-test, and the linear-by-linear association test was used to examine trends across study periods for categorical variables. Effect sizes for ordered associations were calculated using Kendall’s tau-b with 95% confidence intervals. A 5% significance level was used for all tests.
A total of 2991 participants were included, of whom 83.8% (2507/2991) were women, with a median age [IQR] of 48.0 [40.0–57.0] years old. During P1, 2718 visits were recorded, with all infiltrations performed exclusively by neurologists. In P2, 4267 administrations were documented, with 71.0% (3031/4267) of infiltrations conducted by neurologists and 29.0% (1236/4267) by APNs. During P3, 3826 administrations were recorded, with infiltrations nearly evenly split between neurologists (50.2%; 1921/3826) and APNs (49.8%; 1905/3826) (Fig. 1).
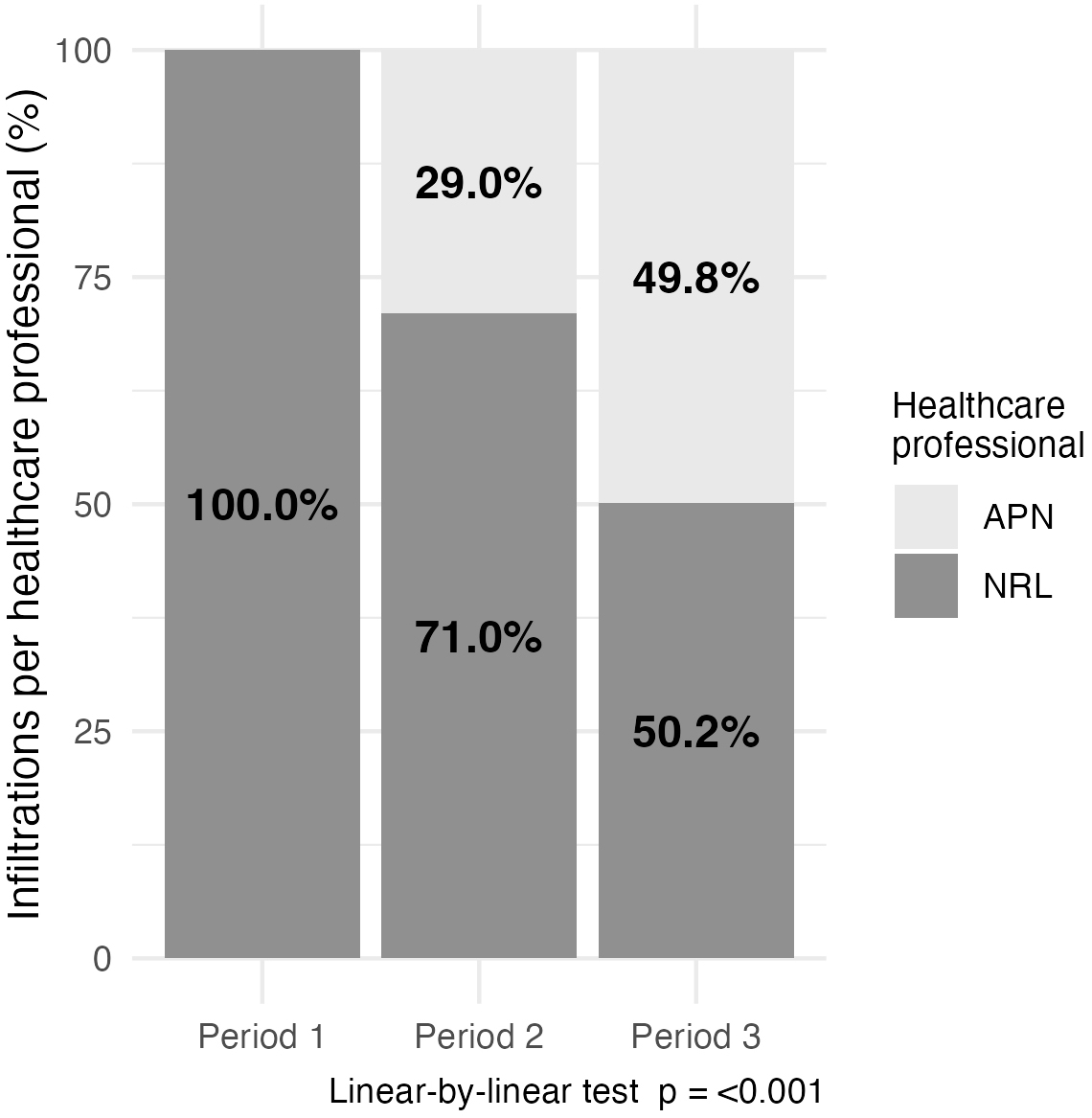 Fig. 1.
Fig. 1.
Infiltrations per healthcare professional (%). APN, advanced practice nurse; NRL, neurologist.
Adherence to the PREEMPT protocol showed a statistically significant association
with the study period (Z = –19.60, p
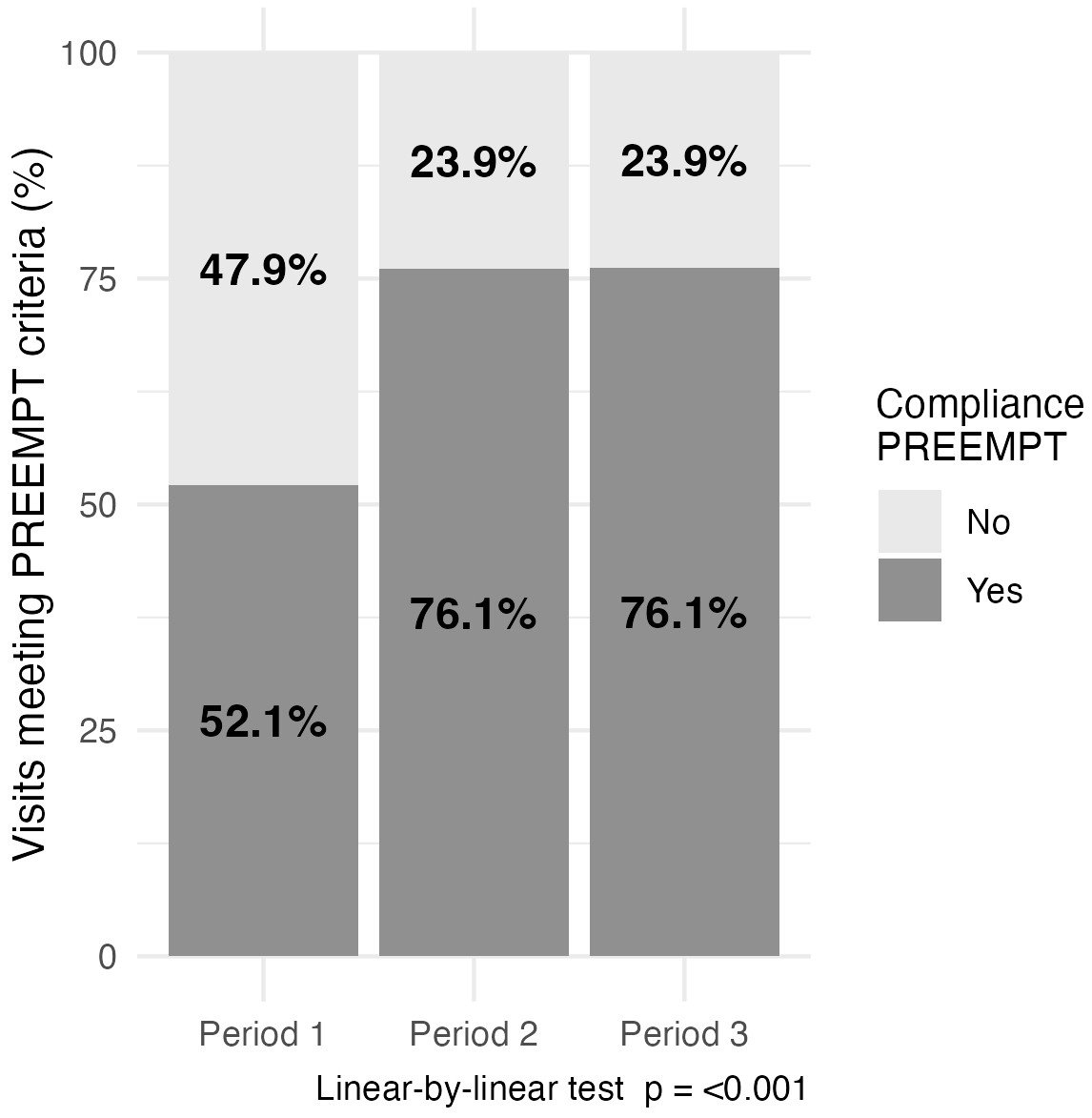 Fig. 2.
Fig. 2.
Visits meeting Phase III Research Evaluating Migraine Prophylaxis Therapy (PREEMPT) criteria (%).
The administration of OnabotA by headache nurses is a key component of APN professional practice [8]. In this study, the involvement of APNs in the administration of OnabotA was associated with shorter injection intervals and improved adherence to the PREEMPT protocol, which is critical for the effective management of CM [9]. Adherence to the PREEMPT protocol directly benefits individuals with CM by improving the quality of care [9], while for the medical team, the presence of the APNs reduces outpatient care pressure by reducing the number of visits exclusively managed by neurologists. It promotes interdisciplinary collaboration. We believe that improved adherence to the recommended OnabotA administration interval directly enhances quality of care because consistent 12-week dosing minimizes symptom fluctuation and ensures more stable migraine control, while the increased scheduling efficiency provided by the APN reduces waiting times and reinforces a patient-centred care pathway [10]. This improvement may be partially attributed to an increase in human resources allocated to treatment administration. However, other aspects, such as enhanced communication between patients and APNs, greater access to the Headache Clinic, and management of agendas, may have also contributed to the improved adherence observed. Similar European experiences have described the integration of nurse-led OnabotA administration as a safe and efficient model of care within multidisciplinary headache services, improving access and treatment continuity for patients with chronic migraine [8].
The slight increase of +2 days observed in P3 suggests that the system may be reaching its capacity with one nurse, potentially indicating a saturation of available resources. Although the total number of OnabotA administrations decreased in the last period, adherence rates remained stable. Therefore, this variation does not seem to be related to the introduction or availability of other preventive treatments, but rather to internal organizational adjustments in appointment scheduling and patient flow. This trend underscores the need for further reinforcement of APN with another nurse to maintain optimal adherence rates and ensure continued accessibility to treatment. Expanding the involvement of APNs could help sustain the improvements observed in P2 while preventing potential delays in treatment administration as patient demand continues to grow.
It is important to note that, in clinical practice, some well-controlled patients may tolerate slightly extended treatment intervals without loss of efficacy [11]. Although this study focused on adherence to the PREEMPT-recommended 12-week schedule rather than clinical outcomes, this aspect could be relevant when interpreting adherence data. Future studies could explore whether individual variability in response allows flexibility in injection timing without compromising treatment effectiveness.
A limitation of this study is that adherence was used as a surrogate outcome, without including direct clinical efficacy measures such as headache frequency, acute medication use, or patient-reported outcomes. Therefore, although we could not objectively confirm clinical improvement in our cohort, prior evidence suggests that maintaining regular 12-week dosing is associated with positive migraine control [12]. These data were not available within the retrospective design; however, future prospective studies within our group are already addressing these outcomes, as well as evaluating the tolerability of OnabotA administration, to further assess the clinical impact of improved adherence. A further limitation is that the study was conducted in a single tertiary headache unit, which may restrict the generalizability of the findings to other healthcare settings or models of nurse-led care. Although the COVID-19 period was not included, post-pandemic organisational changes may still act as residual confounders. Nonetheless, a key strength of this work is the large real-world sample spanning multiple time periods, which enhances the reliability of the observed associations.
The approval and integration of the APN in Headache Clinics and care has played a pivotal role in developing this activity. This organizational change has allowed for greater scheduling flexibility, optimized appointment management, and improved continuity of care for patients requiring OnabotA administration. This is the first study in Spain to quantify the organizational impact of introducing an APN into chronic migraine management, providing real-world evidence on improved adherence to recommended injection intervals. These findings offer a valuable insight for healthcare planning and optimization of multidisciplinary headache models.
Future research could explore patients’ perspectives on adherence and perceived differences between nurse- and physician-led care, as an integrated approach to care seems to be positively impacting treatment adherence.
This study demonstrated that the incorporation of APNs was associated with a higher number of infiltrations, shorter administration intervals, and improved adherence to the PREEMPT protocol. These changes contributed directly to enhanced care for individuals with chronic migraine.
The data supporting the findings of this study are not publicly available due to institutional data protection and confidentiality policies. Data availability complies with the journal’s editorial policies and formats as outlined by IMR Press. The datasets are available from the corresponding author upon reasonable request.
EGC and MTF conceptualized and designed the study. EGC was responsible for data collection and coordination of the study procedures. VJG performed the statistical analysis. EGC drafted the original manuscript. AAR, EC, LGD, and PPR contributed to the interpretation of the data, provided critical intellectual input, and participated in the revision of the manuscript for important scientific content. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This retrospective study was conducted in accordance with local regulations and complied with the ethical principles of the Declaration of Helsinki. Written informed consent was obtained from all participants. The study was approved by the Vall d’Hebron Ethics Committee (approval number: PR(AG)53/2017).
The authors would like to thank all patients and staff from the Headache Clinic for their collaboration.
This research received no external funding.
EGC has received honoraria for participation in medical education activities from AbbVie, Eli Lilly, Organon, Novartis, Teva, Lundbeck and Dr Reddy’s. Additionally, the author has received consulting fees from AbbVie, and Lundbeck. EGC has also received research grants from Sociedad Española de Enfermería Neurológica. PPR has received honoraria for participation in medical education activities from AbbVie, Eli Lilly, Organon, Novartis, Teva, Lundbeck and Dr Reddy’s. EC has received honoraria from Novartis, Chiesi, Lundbeck, MedScape, Lilly, Organon, TEVA, Dr Reddy’s; his salary has been partially funded by Río Hortega grant Acción Estratégica en Salud 2017–2020 from Instituto de Salud Carlos III (CM20/00217) and Juan Rodés fellowship, Subprograma Estatal de Incorporación de la Acción Estratégica en Salud 2023 (JR23/00065). He is a junior editor for Cephalalgia. MTF has received honoraria for participation in medical education activities from AbbVie, Chiesi, Eli Lilly, Organon, Medscape, Novartis, and Teva. Additionally, the author has received consulting fees from AbbVie, Eli Lilly, and Organon. Marta Torres-Ferrús has also received research grants from Instituto Investigación Carlos III, Mutual Médica, Sociedad Española de Neurología, and Societat Catalana de Neurologia. AAR has received honoraria for participation in medical education activities from AbbVie, Eli Lilly, Novartis, and Teva. Other authors do not have any conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


