1 Department of Clinical Laboratory, Quanzhou First Hospital Affiliated to Fujian Medical University, 362000 Quanzhou, Fujian, China
2 Department of Neurology, Quanzhou First Hospital Affiliated to Fujian Medical University, 362000 Quanzhou, Fujian, China
Abstract
To evaluate the association of the purinergic receptor P2Y, G-protein coupled, 12 (P2Y12) gene polymorphisms with susceptibility to different etiological stroke subtypes.
A total of 459 first-ever acute ischemic stroke patients were classified into large-artery atherosclerosis (LAA, n = 163), small-vessel occlusion (SVO, n = 204), and cardioembolism (CE, n = 92) based on the Trial of Org 10172 in Acute Stroke Treatment (TOAST) criteria. Direct sequencing was used to screen these three stroke subtypes and non-stroke controls for P2Y12 polymorphisms: a T→C transition at 744 nucleotides (nt) downstream of intron 5's start site (i-T744C) and a C→T transition at 34 nt downstream of exon 2's start site (C34T). Based on the results of multivariate logistic analyses, a prediction model was established via a nomogram that incorporated genomic and clinical variables to quantify the risk of LAA stroke.
Significant differences in the P2Y12 i-T744C genotype and allele frequencies were observed between LAA patients and controls. After adjusting for confounding factors, the dominant model (p = 0.009) and additive model (p = 0.023) revealed that the i-T744C polymorphism was significantly associated with increased susceptibility to LAA. No significant associations were found for the SVO and CE stroke subtypes. Moreover, the C34T polymorphism was not an independent factor for any stroke subtype. We further constructed a nomogram prediction model for LAA stroke based on genomic and clinical variables, including age, hypertension, smoking, high-density lipoprotein cholesterol, and the i-T744C polymorphism. This nomogram exhibited satisfactory accuracy and predictive power for LAA stroke, as demonstrated by the area under the curve, calibration plot, and decision curve analysis.
The P2Y12 i-T744C polymorphism may serve as a predictor for LAA stroke. Furthermore, we constructed a genomic-clinical nomogram that may be valuable for predicting LAA stroke risk in the study population.
Keywords
- ischemic stroke
- receptors
- purinergic P2Y12
- polymorphism
- genetic
- large-artery atherosclerosis
- nomograms
The global burden of ischemic stroke (IS) keeps rising [1], with forecasts suggesting that IS-related deaths will increase to approximately 4.9 million worldwide by 2030 [2, 3]. IS is a complex disorder that can be classified into five etiological subtypes according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification: large-artery atherosclerosis (LAA), small-vessel occlusion (SVO), cardioembolism (CE), undetermined, and other determined etiology [4]. The clinical manifestations, targeted therapeutic strategies, and prognostic outcomes across the different etiological subtypes are distinct [4, 5], underscoring the significance of subtype-specific research in IS. The primary cause of LAA stroke is atherosclerotic plaque formation and subsequent rupture of large cerebral arteries [6]. LAA typically manifests with severe focal neurological deficits (e.g., hemiplegia, aphasia), accompanied by a high susceptibility to early neurological deterioration [4].
The P2Y purinoceptor 12 (P2Y12 receptor) gene (herein referred to as the
P2Y12 gene) localizes to chromosome 3, long arm bands q21 to q25 (3q21-q25) and encodes a
342-residue, G-protein-coupled receptor on the surface of platelets, microglia,
and vascular smooth muscle cells (VSMCs) [7]. The adenosine diphosphate
(ADP)-mediated P2Y12 receptor signaling pathway can promote platelet activation
and aggregation, pro-inflammatory cytokine release, formation of
platelet-leukocyte aggregates, and VSMC migration and proliferation [8]. These
processes contribute to the progression of atherosclerosis and thrombosis.
Consistent with the essential role of the P2Y12 receptor in atherosclerosis and
pathological thrombosis, one of its single-nucleotide polymorphisms (SNPs),
i-T744C (rs2046934, a T
Nomograms are a clinically valuable instrument for integrating diverse data types and can be used for multi-disease risk assessment with increased precision. The LAA subtype is a multi-factorial disease attributable to an interplay of multiple factors [16]. Integration of these variables into a predictive model may enhance the diagnostic accuracy of LAA. These considerations motivated us to develop a nomogram that integrates well-established genetic and clinical variables, enabling quantification of the weighted contribution of each input variable and making it intuitive for clinical practice.
Therefore, the aim of this study was to elucidate the relationship between two
common P2Y12 polymorphisms [i-T744C and C34T
(rs6785930, a C
Patients admitted to Quanzhou First Hospital in Fujian Province, China, for
their first-ever IS between February 2024 and March 2025 were recruited to this
research project. The diagnosis of IS was based on World Health Organization
(WHO) criteria and was radiologically confirmed by computed tomography scans (CT)
or magnetic resonance imaging (MRI) [2]. Eligible patients were categorized into
five etiological subtypes by experienced neurologists and according to TOAST
criteria [4, 17]. The diagnostic criteria for LAA, SVO and CE stroke were: (1)
LAA, cerebral hypodensities with a diameter of
During the same study period, non-stroke controls (age
Data on the genetic variable (P2Y12 i-T744C polymorphism) were
collected, as well as 12 clinical variables with
Venous whole blood was drawn from participants within 1 week of the IS event and placed into EDTA-K2 anticoagulant tubes. Genomic DNA was extracted from these blood samples using the TIANamp Genomic DNA Kit (DP319-02, TianGen Biotech Co., Beijing, China) and stored at –20 ℃ for subsequent genetic analysis. PCR was performed as follows: denaturation initiation at 94 °C for 5 min, with 30 amplification cycles (94 °C/30 sec, 56 °C/30 sec, and 72 °C/35 sec), a final 10-min extension at 72 °C, and hold at 4 °C [13]. A 50 µL PCR reaction system was utilized, consisting of 25 µL 2X SanTap PCR Mix (B532061, Sangon Biotech Co., Shanghai, China), 3 µL genomic DNA, 4 µL primer sets, and 18 µL sterile water. Genotyping of the amplicons with Sanger sequencing was performed by Sangon Biotech Co. using primers described previously [13].
Statistical analyses were conducted with IBM SPSS Statistics 27.0 (IBM Corp.,
Armonk, NY, USA). The nomogram was generated on the Beckman Coulter DxAI platform
based on R version 4.2.3 (https://www.xsmartanalysis.com/beckman/login/).
Continuous data are expressed as the mean
Non-stroke controls and LAA patients were randomly categorized at a 4:1 ratio.
The former (n = 282) served as the training cohort for construction of the model,
while the latter (n = 71) served as the validation cohort to assess the model’s
robustness. Detailed procedures for construction of the nomogram were as follows:
candidate variables associated with LAA stroke (p
A total of 459 IS patients with defined etiology participated in this study,
comprising 163 (35.5%) patients with LAA stroke, 204 (44.4%) with SVO stroke,
and 92 (20.1%) with CE stroke. Demographic and clinical characteristics of the
controls and different IS subtypes are presented in Table 1. The average age,
hypertension frequency, and HDL-C levels were significantly different between all
IS subtypes and controls (all p
| Variable | Controls (n = 190) | LAA stroke (n = 163) | p-value | SVO stroke (n = 204) | p-value | CE stroke (n = 92) | p-value |
| Age (years) | 59.69 |
63.12 |
0.002* | 62.63 |
0.004* | 68.73 |
|
| Men, n (%) | 91 (47.9) | 92 (56.4) | 0.109 | 95 (46.6) | 0.792 | 56 (60.9) | 0.041 |
| Hypertension, n (%) | 66 (34.7) | 119 (73.0) | 144 (70.6) | 67 (72.8) | |||
| Diabetes mellitus, n (%) | 44 (23.2) | 73 (44.8) | 78 (38.2) | 0.001* | 27 (29.3) | 0.262 | |
| Cigarette smoking, n (%) | 28 (14.7) | 49 (30.0) | 59 (28.9) | 22 (23.9) | 0.059 | ||
| Alcohol intake, n (%) | 19 (10.0) | 25 (15.3) | 0.126 | 31 (15.2) | 0.122 | 8 (8.7) | 0.730 |
| TG (mmol/L) | 1.48 |
1.75 |
0.013* | 1.63 |
0.040* | 1.39 |
0.431 |
| TC (mmol/L) | 5.07 |
5.20 |
0.336 | 5.11 |
0.725 | 4.88 |
0.141 |
| LDL-C (mmol/L) | 3.46 |
3.46 |
0.974 | 3.44 |
0.838 | 3.22 |
0.028* |
| HDL-C (mmol/L) | 1.42 |
1.26 |
1.28 |
1.22 |
|||
| Ischemic heart disease, n (%) | 11 (5.8) | 14 (8.6) | 0.307 | 6 (2.9) | 0.164 | 13 (14.1) | 0.019* |
| Atrial fibrillation, n (%) | 4 (2.1) | 7 (4.3) | 0.238 | 2 (1.0) | 0.617 | 67 (72.8) |
SVO, small-vessel occlusion; CE,
cardioembolism; TC, total cholesterol; TG, triglycerides; HDL-C, high-density
lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; *p
Table 2 presents the genotype distribution and allele frequencies of
P2Y12 polymorphisms in control participants and in patients with
different stroke subtypes. Both SNPs conformed to the Hardy-Weinberg equilibrium
in both the overall IS cohort and control group (all p
| SNP | Genotype/allele | Controls (n = 190) | LAA stroke (n = 163) | p | SVO stroke (n = 204) | p | CE stroke (n = 92) | p |
| i-T744C | Genotype | |||||||
| TT | 146 (76.8) | 100 (61.3) | 150 (73.5) | 63 (68.5) | ||||
| TC | 39 (20.5) | 56 (34.4) | 52 (25.5) | 25 (27.2) | ||||
| CC | 5 (2.6) | 7 (4.3) | 0.007* | 2 (1.0) | 0.253 | 4 (4.3) | 0.315 | |
| Allele | ||||||||
| T | 331 (87.1) | 256 (78.5) | 352 (86.3) | 151 (82.1) | ||||
| C | 49 (12.9) | 70 (21.5) | 0.002* | 56 (13.7) | 0.732 | 33 (17.9) | 0.111 | |
| C34T | Genotype | |||||||
| CC | 119 (62.6) | 107 (65.6) | 111 (54.4) | 59 (64.1) | ||||
| CT | 64 (33.7) | 48 (29.5) | 74 (36.3) | 28 (30.5) | ||||
| TT | 7 (3.7) | 8 (4.9) | 0.729 | 19 (9.3) | 0.049* | 5 (5.4) | 0.716 | |
| Allele | ||||||||
| C | 302 (79.5) | 262 (80.4) | 296 (72.5) | 146 (79.3) | ||||
| T | 78 (20.5) | 64 (19.6) | 0.768 | 112 (27.5) | 0.023* | 38 (20.7) | 0.972 |
SNP, single nucleotide polymorphism; C34T, a C
The genotype distribution of the i-T744C polymorphism differed significantly between the LAA subtype and control groups, with the C allele being more frequent in LAA patients (21.5% vs. 12.9%, p = 0.002). Multivariate logistic regression analysis showed the i-T744C polymorphism was associated with a significantly increased risk of LAA stroke in both the dominant (adjusted odds ratio (OR) = 2.024, 95% confidence interval (CI): 1.191–3.440, p = 0.009) and additive (adjusted OR = 1.703, 95% CI: 1.078–2.692, p = 0.023) models following adjustment for relevant confounders (Table 3). A significant association between the C34T polymorphism and SVO subtype risk was observed in the recessive model via univariate logistic regression analysis (p = 0.025). However, this positive association was lost after adjustment for other covariates (adjusted OR = 1.854, 95% CI: 0.708–4.852, p = 0.209). Additionally, both univariate and multivariable logistic regression analyses found no statistically significant relationships between the i-T744C and C34T polymorphisms and CE subtype risk.
| SNP | Genetic model | Crude OR (95% CI) | Crude p-value | Adjusted OR (95% CI) | Adjusted p-value | ||
| LAA strokea | |||||||
| i-T744C | Dominant | TT vs. TC+CC | 2.090 (1.318–3.316) | 0.002* | 2.024 (1.191–3.440) | 0.009* | |
| Recessive | TT+TC vs. CC | 1.660 (0.517–5.335) | 0.390 | 1.165 (0.313–4.334) | 0.820 | ||
| Additive | TT vs. TC vs. CC | 1.818 (1.219–2.712) | 0.003* | 1.703 (1.078–2.692) | 0.023* | ||
| C34T | Dominant | CC vs. CT+TT | 0.877 (0.567–1.358) | 0.557 | 0.919 (0.557–1.516) | 0.741 | |
| Recessive | CT+CC vs. TT | 1.349 (0.478–3.805) | 0.570 | 1.356 (0.410–4.487) | 0.618 | ||
| Additive | CC vs. CT vs. TT | 0.946 (0.655–1.367) | 0.769 | 0.978 (0.643–1.490) | 0.919 | ||
| SVO strokea | |||||||
| i-T744C | Dominant | TT vs. TC+CC | 1.195 (0.755–1.890) | 0.447 | 1.408 (0.835–1.438) | 0.199 | |
| Recessive | TT+TC vs. CC | 0.366 (0.070–1.911) | 0.391 | 0.196 (0.032–1.205) | 0.079 | ||
| Additive | TT vs. TC vs. CC | 1.075 (0.712–1.622) | 0.732 | 1.163 (0.729–1.441) | 0.526 | ||
| C34T | Dominant | CC vs. CT+TT | 1.404 (0.939–2.101) | 0.098 | 1.372 (0.952–1.976) | 0.090 | |
| Recessive | CT+CC vs. TT | 2.685 (1.102–6.540) | 0.025* | 1.854 (0.708–4.852) | 0.209 | ||
| Additive | TT vs. TC vs. CC | 1.185 (0.969–1.449) | 0.099 | 1.429 (0.910–2.244) | 0.121 | ||
| CE strokeb | |||||||
| i-T744C | Dominant | TT vs. TC+CC | 1.527 (0.878–2.658) | 0.133 | 1.721 (0.711–4.162) | 0.229 | |
| Recessive | TT+TC vs. CC | 1.682 (0.441–6.417) | 0.684 | 2.476 (0.415–14.778) | 0.320 | ||
| Additive | TT vs. TC vs. CC | 1.432 (0.900–2.278) | 0.129 | 1.625 (0.803–3.288) | 0.176 | ||
| C34T | Dominant | CC vs. CT+TT | 0.937 (0.559–1.573) | 0.807 | 0.693 (0.285–1.689) | 0.420 | |
| Recessive | CT+CC vs. TT | 1.502 (0.464–4.868) | 0.495 | 1.202 (0.158–9.159) | 0.859 | ||
| Additive | TT vs. TC vs. CC | 1.008 (0.652–1.557) | 0.972 | 0.786 (0.368–1.679) | 0.535 | ||
aAdjusted for age, hypertension, diabetes mellitus, smoking, TG and HDL-C;
bAdjusted for age, gender, hypertension, ischemic heart disease, LDL-C,
HDL-C, and atrial fibrillation; OR, odds ratio; *p
The characteristics of control and LAA patients in the training group are
compared in Table 4. As shown in Table 5, univariate and multivariable logistic
regression analyses identified five variables that appeared to be independent
predictors of LAA stroke: age (OR = 1.033, 95% CI: 1.004–1.064, p =
0.026), hypertension (OR = 4.857, 95% CI: 2.804–8.577, p
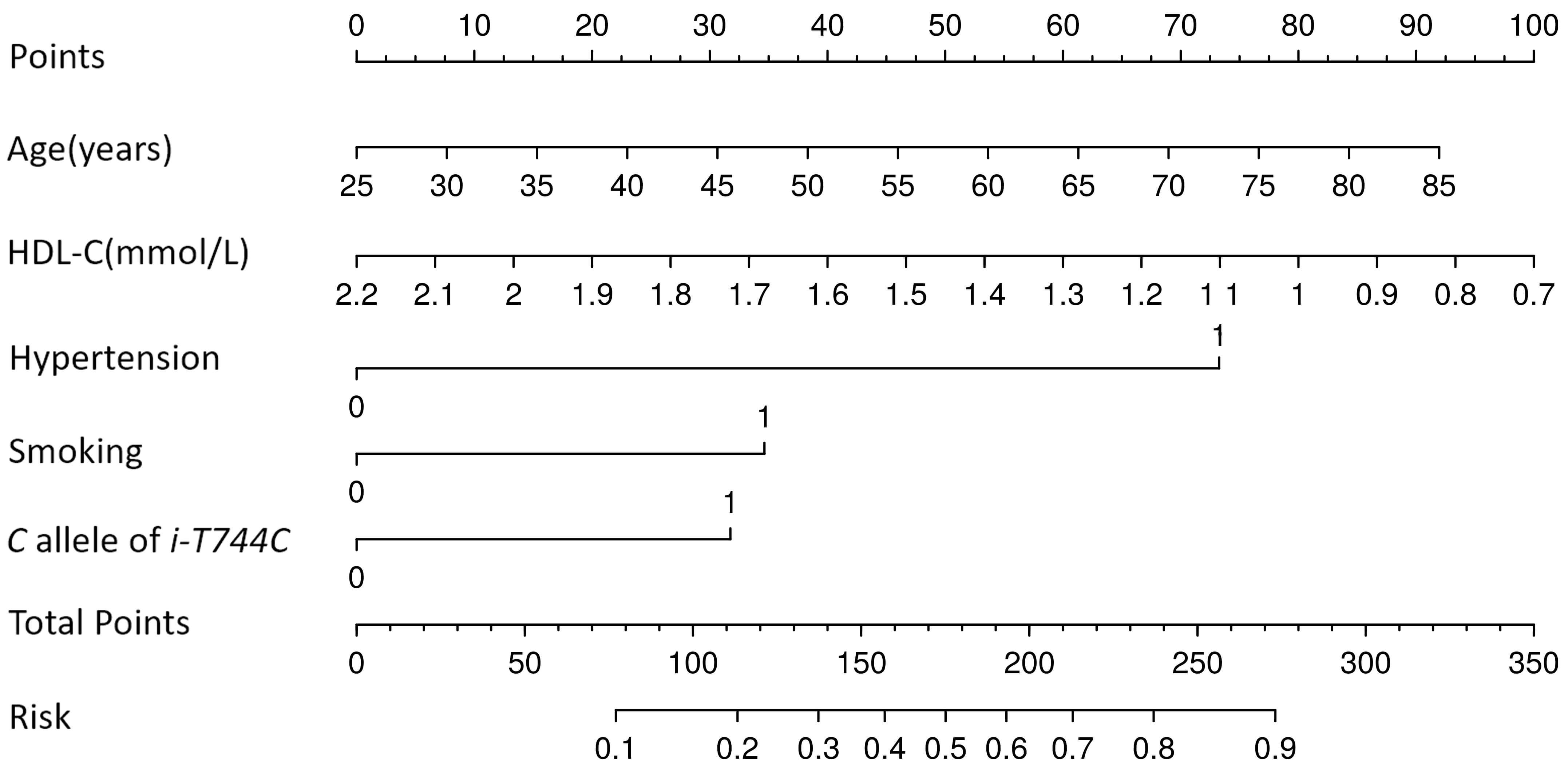 Fig. 1.
Fig. 1.
Risk prediction nomogram for large-artery atherosclerosis stroke. The predictors included age, hypertension, smoking, high-density lipoprotein cholesterol (HDL-C), and the i-T744C polymorphism. i-T744C, a T→C transition at 744 nucleotides (nt) downstream of intron 5’s start site of the P2Y12 gene.
| Variable | Controls (n = 152) | LAA stroke (n = 130) | p-value |
| Age (years) | 59.40 |
63.10 |
0.001* |
| Men, n (%) | 71 (46.7) | 72 (55.4) | 0.146 |
| Hypertension, n (%) | 48 (31.6) | 95 (73.1) | |
| Diabetes mellitus, n (%) | 35 (23.0) | 57 (43.8) | |
| Cigarette smoking, n (%) | 22 (14.5) | 39 (30.0) | 0.002* |
| Alcohol intake, n (%) | 15 (9.9) | 18 (13.9) | 0.300 |
| TG (mmol/L) | 1.50 |
1.74 |
0.043* |
| TC (mmol/L) | 5.09 |
5.14 |
0.739 |
| LDL-C (mmol/L) | 3.46 |
3.42 |
0.771 |
| HDL-C (mmol/L) | 1.42 |
1.25 |
|
| Ischemic heart disease, n (%) | 9 (5.9) | 11 (8.5) | 0.407 |
| Atrial fibrillation, n (%) | 4 (2.6) | 5 (3.8) | 0.563 |
| C allele of i-T744C | 35 (23.0) | 48 (36.9) | 0.011* |
*p
| Variable | Univariate regression analysis | Multivariate regression analysis | ||
| OR (95% CI) | p-value | OR (95% CI) | p-value | |
| Age | 1.041 (1.015–1.068) | 0.002* | 1.033 (1.004–1.064) | 0.026* |
| Hypertension | 5.881 (3.507–9.861) | 4.857 (2.804–8.577) | ||
| Diabetes mellitus | 2.610 (1.564–4.357) | 1.627 (0.899–2.951) | 0.107 | |
| Cigarette smoking | 2.532 (1.408–4.556) | 0.002* | 2.207 (1.102–4.493) | 0.027* |
| TG | 1.306 (1.010–1.689) | 0.042* | 1.054 (0.785–1.452) | 0.734 |
| HDL-C | 0.171 (0.076–0.384) | 0.264 (0.105–0.641) | 0.004* | |
| C allele of i-T744C | 1.957 (1.164–3.288) | 0.011* | 2.021 (1.100–3.765) | 0.024* |
*p
This predictive nomogram had AUCs of 0.8 (95% CI: 0.748–0.853) in the training cohort, and 0.723 (95% CI: 0.603–0.843) in the validation cohort, indicating consistent diagnostic efficacy for LAA stroke (Fig. 2a,b). The sensitivity and specificity were 0.8 and 0.697, respectively, in the training cohort, and 0.667 and 0.711, respectively, in the validation cohort. Furthermore, the calibration curve demonstrated good calibration of the predictive model (p = 0.392, Hosmer-Lemeshow test). The mean absolute error was 0.022 in the training group and 0.048 in the validation group (Fig. 2c,d). DCA revealed the model curves showed significant deviation from extreme values, thus demonstrating certain clinical utility in predicting LAA stroke (Fig. 2e,f). Collectively, these results indicate that the nomogram model incorporating the P2Y12 i-T744C polymorphism and clinical data holds substantial clinical significance for the prediction of LAA stroke risk.
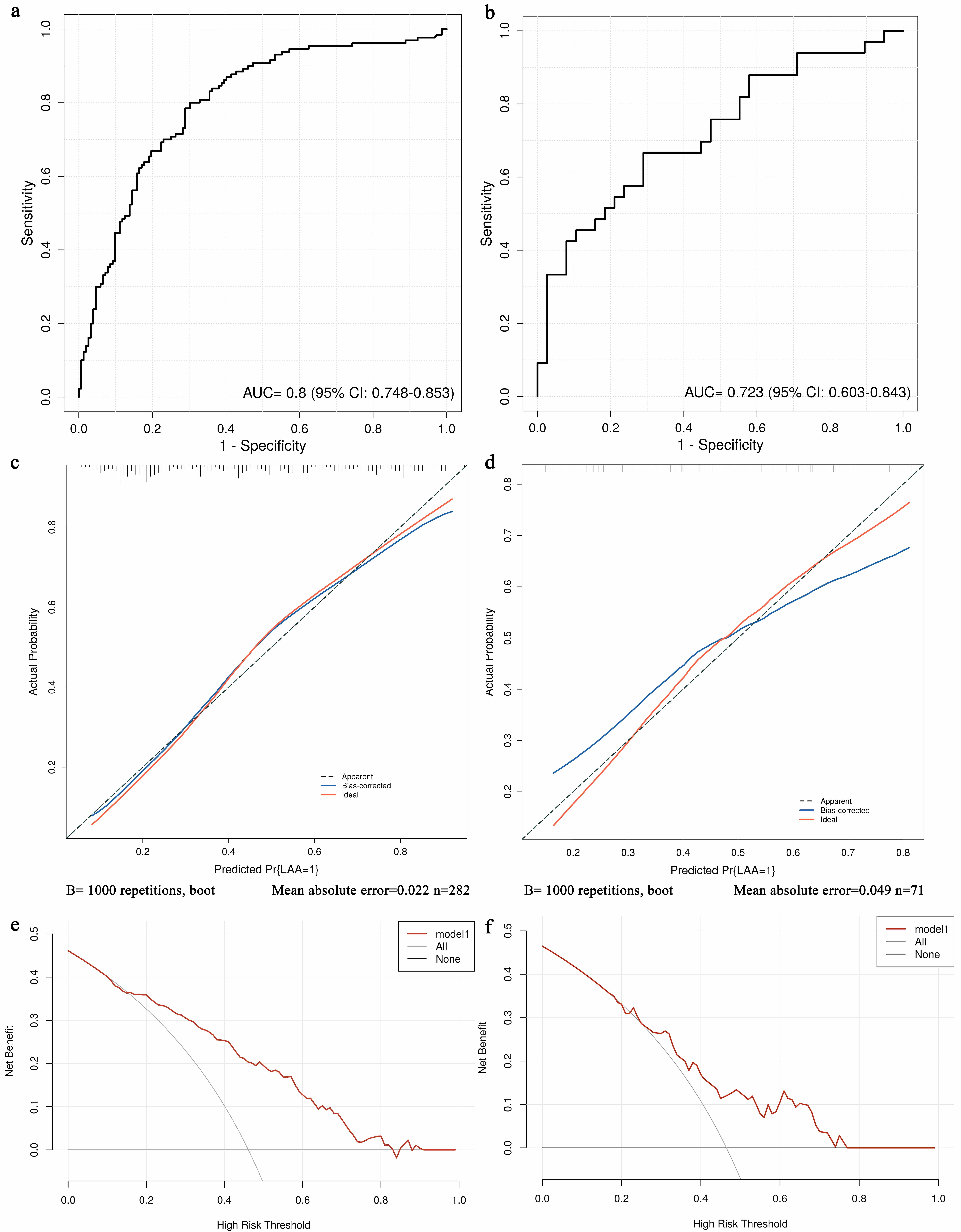 Fig. 2.
Fig. 2.
Validation of the nomogram. Receiver operating characteristic, calibration, and decision curve analysis of the training cohort (a,c,e) and validation cohort (b,d,f). ROC, receiver operating characteristic curve; AUC, area under the ROC curve; CI, confidence interval; LAA, large-artery atherosclerosis.
This study provides the first evidence of a significant association between the P2Y12 i-T744C polymorphism and an elevated risk of LAA stroke. Furthermore, we developed a nomogram model that incorporates traditional risk factors along with the i-T744C polymorphism for predicting LAA stroke. Patients with high scores in this predictive nomogram have a high probability of developing LAA stroke, and may therefore derive clinical benefit from early prediction and preventive care.
Studies have established that the P2Y12 receptor, encoded by the P2Y12
gene, plays an essential role in atherosclerosis and atherothrombosis. Activation
of P2Y12 on platelets triggers the phosphoinositide 3-kinase (PI3K)/protein
kinase B (PKB/AKT) pathway via coupled G protein
Other investigations have shown that P2Y12 genetic variants were
associated with susceptibility to atherosclerosis-related disorders and also
influenced platelet reactivity. Fontana et al. [10] reported that four
variants in the P2Y12 gene—i-T744C, i-C139T (a
C
Consistent with previous findings [15], the P2Y12 C34T polymorphism did not correlate with any IS subtypes in the current study. The P2Y12 C34T polymorphism has also been investigated for its association with clopidogrel resistance (CR). The results showed the T allele of C34T polymorphism was associated with increased susceptibility to CR and concomitant adverse cardiac or cerebrovascular outcomes in Chinese cohorts [26]. However, conflicting results have been reported in other ethnic groups [27], and further studies should focus on correlations between the C34T polymorphism and adverse clinical outcomes in clopidogrel-treated stroke patients.
The usefulness of nomograms for the diagnosis and prognostic evaluation of IS
and its subtypes has previously been reported through the integration of relevant
risk factors [28, 29, 30]. For example, Chen et al. [28] devised a nomogram
with good discrimination for the prediction of early IS. Their nomogram comprised
6 clinical parameters: gender, diabetes, family history, coronary heart disease,
smoking, and age. Similar to a report in the literature [16], we identified age,
hypertension, smoking, and HDL-C as four clinical correlates of LAA stroke.
Additionally, we also found that the 744C polymorphism may be a potential
predictor for the development of LAA stroke. IDI and NRI-common metrics quantify
the incremental predictive value of one model over another in clinical research
[31]. These suggested the i-T744C polymorphism might offer important
additional clinical information for LAA stroke risk prediction. Based on the
above findings, we constructed a novel nomogram for early LAA stroke prediction
that includes the i-T744C polymorphism and clinical variables. This may
serve as a reliable tool for personalized LAA stroke monitoring, as evidenced by
its satisfactory calibration, discriminative capacity (AUC of 0.8 in the training
cohort and 0.723 in the validation cohort), and favorable net clinical benefit.
Our nomogram provides an immediate predicted risk of LAA stroke in clinical
practice based on patient-specific variables, helping clinicians to rapidly
identify high-risk individuals (predicted probability
Our study had several limitations. First, the sample sizes of the stroke subtype and control groups were relatively small. Further studies with larger and ethnically diverse cohorts are needed to confirm our results. Second, complex gene-environment interactions drive the development of different stroke subtypes, whereas our predictive nomogram focuses on limited variables and is exclusively applicable to LAA stroke. Therefore, future studies should incorporate more genetic and environmental variations, as well as systematic analysis of gene-environment interaction effects within stroke subtypes. Finally, this analysis was limited to genotype-phenotype associations and requires further in-depth exploration of the potential mechanisms underlying the observed associations.
Our findings revealed that the P2Y12 i-T744C polymorphism was associated with LAA stroke, with the C allele being a significant predisposing factor. We constructed a nomogram model that combines the i-T744C polymorphism and clinical variables. This model displayed a favorable capacity to discriminate individual LAA risk, and could thus help to identify high-risk individuals and enable personalized prevention. Our findings require further validation in large-scale, multi-ethnic studies.
The datasets used and analyzed in the current article can be available from the corresponding author upon reasonable request.
CW—carried out the experiment and wrote the manuscript, YC, XW—collected the samples, JC—analyzed and interpreted the data, ZZ—designed the study and revised the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was conducted in accordance with the Declaration of Helsinki. All procedures were approved by the Ethics Committee of Quanzhou First Hospital (Approval Number: [2024] K112). Written informed consent was signed by all participants before enrollment in the study.
We are grateful to the patients for their participation in our study.
This work was funded by the Startup Fund for scientific research, Fujian Medical University (2023QH1312).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


