1 Department of Neurology, Soonchunhyang University Seoul Hospital, Soonchunhyang University School of Medicine, 04401 Seoul, Republic of Korea
Abstract
Parkinson’s disease (PD) is increasingly being diagnosed in older adults. Despite this trend, the clinical features of geriatric patients with PD are not thoroughly defined. This study aimed to compare the clinical characteristics of geriatric patients (aged ≥75 years) with de novo PD against those of non-geriatric patients (aged <75 years) newly diagnosed with PD.
This retrospective analysis enrolled 110 patients aged 50 years or older with de novo PD from our hospital’s Parkinsonism registry between 2017 and 2023. Clinical evaluations included motor assessment via the Unified Parkinson’s Disease Rating Scale Part III and global cognitive function was measured using the Montreal Cognitive Assessment (MoCA). Nonmotor symptoms, including depression, anxiety, and fatigue, were assessed using other scales and autonomic dysfunction was assessed using the Scale for Outcomes in Parkinson’s Disease–Autonomic (SCOPA-AUT).
Geriatric patients with PD (n = 37) exhibited significantly lower cognitive performance (lower MoCA scores, p < 0.001) and more pronounced autonomic dysfunction (higher SCOPA-AUT scores, p = 0.0103) in comparison with non-geriatric PD patients (n = 73). In multivariate logistic regression analysis, lower MoCA scores (odds ratio [OR]: 0.7642, 95% confidence interval [CI]: 0.6712–0.8701, p < 0.001) and elevated SCOPA-AUT scores (OR: 1.0640, 95% CI: 1.0031–1.1286, p = 0.0391) emerged as significant independent predictors of geriatric PD.
These findings reveal a distinct clinical phenotype among geriatric patients with de novo PD, underscoring the value of early detection and proactive management of cognitive and autonomic impairments in this group. The results further emphasize the need for individualized assessment and therapeutic interventions tailored to the specific requirements of geriatric patients with PD.
Keywords
- aged
- cognition
- geriatrics
- Parkinson’s disease
Parkinson’s disease (PD) is showing growing prevalence among older adults, especially those of advanced age. There is an increasing influx of geriatric individuals with PD seeking care at movement disorders clinics [1]. This trend has been referred to as a global “Parkinson pandemic”, primarily attributed to population aging and changing demographics [2]. Furthermore, recent investigations have documented a substantial rise in the global burden of PD in recent decades [3, 4]. Collectively, these observations highlight an urgent need for targeted approaches to optimize the management of geriatric PD in aging societies.
In many countries, individuals aged 60 or 65 years and older are conventionally designated as “elderly” or “geriatric people”, based on chronological age. Nonetheless, the rapid expansion of the aging population during the 21st century has led several nations to become aging societies. In recent years, super-aged societies such as Japan have initiated a redefinition of the “geriatric” threshold, shifting it from 65 to 75 years [5]. This change recognizes both increased life expectancy and the enhanced health status of older adults, thereby encouraging a reconsideration of population classifications and tailored approaches to their care requirements. Furthermore, clinical research increasingly employs 75 years as the age cutoff for older patient groups, ensuring that study designs are better aligned with evolving demographic trends [6, 7, 8].
Despite these demographic changes, the clinical characteristics of newly diagnosed (de novo) PD in people aged 75 years and older have not been thoroughly explored. Our objective was to examine the clinical features of geriatric PD patients aged 75 and above, in comparison with non-geriatric PD patients. The findings of this study are intended to enhance our knowledge of PD manifestation in the geriatric cohort and provide important perspectives for the development of more personalized clinical management strategies.
The Institutional Review Board of our hospital approved this retrospective study
and granted a waiver of informed consent (approval number: 2025-01-003). Between
2017 and 2023, 179 patients were enrolled in our Parkinsonism registry. We
included only de novo PD patients who had a follow-up period exceeding
one year. At our movement disorders clinic, the diagnosis of PD was established
according to the UK Parkinson’s Disease Society Brain Bank criteria [9].
Additionally, brain magnetic resonance imaging (MRI) and
18F-fluoropropyl-carbomethoxy-3
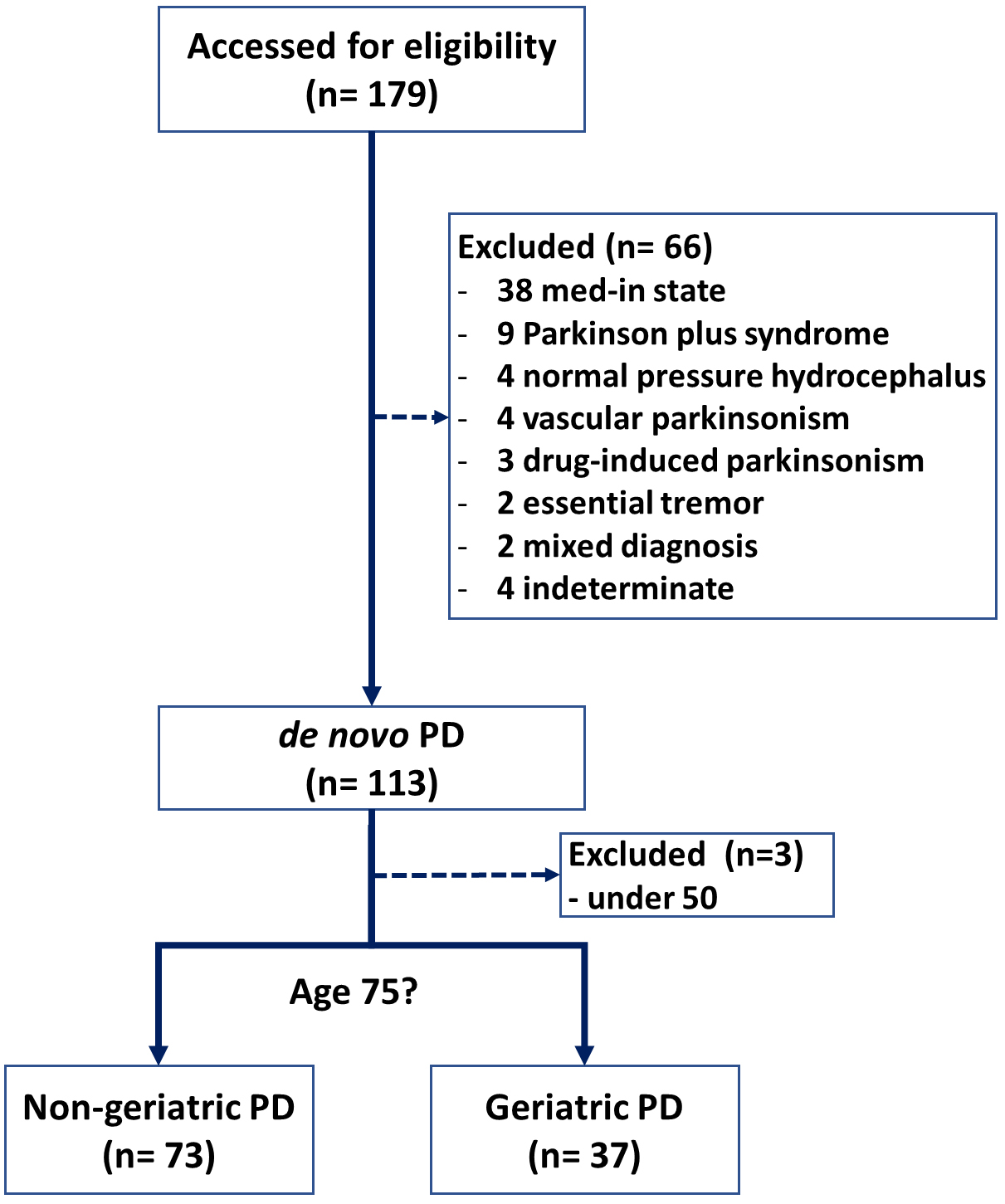 Fig. 1.
Fig. 1.
Flowchart of the study. PD, Parkinson’s disease.
All patients received thorough clinical assessments at the time of enrollment. Demographic and clinical information such as age, gender, body weight, height, and educational attainment was collected. Motor symptoms were evaluated with the Unified Parkinson’s Disease Rating Scale Part III (UPDRS-III), and disease severity was determined according to the Hoehn and Yahr (H&Y) staging [14]. Global cognitive performance was measured using the Korean version of the Montreal Cognitive Assessment (MoCA) [15], and depressive symptoms were assessed with the Beck Depression Inventory (BDI) [16]. Anxiety was evaluated with the Beck Anxiety Inventory (BAI) [17], and fatigue levels were measured using the Parkinson’s Disease Fatigue Scale (PFS) [18]. Furthermore, autonomic function was assessed using the Korean version of the Scales for Outcomes in Parkinson’s Disease – Autonomic (SCOPA-AUT) [19], with the sexual dysfunction domain omitted due to incomplete patient responses. The clinical data were examined to distinguish geriatric and non-geriatric Parkinson’s disease groups in terms of motor, cognitive, psychological, and autonomic domains.
Statistical evaluations were performed to contrast clinical characteristics
between the non-geriatric and geriatric PD groups. Continuous data are reported
as mean
Geriatric individuals (age
| Variable | Total (n = 110) | Non-geriatric PD (n = 73) | Geriatric PD (n = 37) | p value |
| Age, yr | 70.65 |
66.18 |
79.49 |
|
| Female gender | 57 (51.82%) | 36 (49.32%) | 21 (56.76%) | 0.5919 |
| Body weight, kg | 60.47 |
61.57 |
58.30 |
0.1206 |
| Height, m | 1.60 |
1.62 |
1.58 |
0.0265 |
| Body mass index | 23.37 |
23.41 |
23.28 |
0.8125 |
| Disease duration, yr | 1.29 |
1.35 |
1.17 |
0.3093 |
| Years of education | 10.14 |
11.29 |
7.88 |
|
| Diabetes mellitus, n (%) | 29 (26.36%) | 19 (26.03%) | 10 (27.03%) | |
| Hypertension, n (%) | 55 (50%) | 35 (47.95%) | 20 (54.05%) | 0.6865 |
| History of falls, n (%) | 44 (40%) | 27 (36.99%) | 17 (45.95%) | 0.4837 |
| UPDRS-III (motor) | 22.29 |
21.44 |
23.97 |
0.2668 |
| H&Y stage | 2 (2, 2) | 2 (2, 2) | 2 (2, 2) | 0.7705 |
| MoCA-K (cognitive assessment) | 23.85 |
25.32 |
20.95 |
|
| BDI (depressive symptoms) | 8.16 |
7.49 |
9.49 |
0.1529 |
| BAI (anxiety assessment) | 5.41 |
5.11 |
6.00 |
0.4734 |
| PFS (fatigue assessment) | 39.47 |
37.81 |
42.76 |
0.1246 |
| SCOPA-AUT (dysautonomia)# | 11.05 |
9.62 |
13.86 |
0.0103 |
This table was summarized appropriately based on the normality assessment using
the Kolmogorov-Smirnov test and the presence of a chi-squared test warning:
numerical data are expressed as mean
#Total score of SCOPA-AUT was calculated excluding the sexual domain, as a substantial number of patients did not respond to the question regarding sexual dysfunction.
UPDRS, Unified Parkinson’s Disease Rating Scale; H&Y, Hoehn and Yahr; MoCA-K, Korean version of the Montreal Cognitive Assessment; BDI, Beck Depression Inventory; BAI, Beck Anxiety Inventory; PFS, Parkinson’s disease Fatigue Scale; SCOPA-AUT, Scales for Outcomes in Parkinson’s disease – Autonomic.
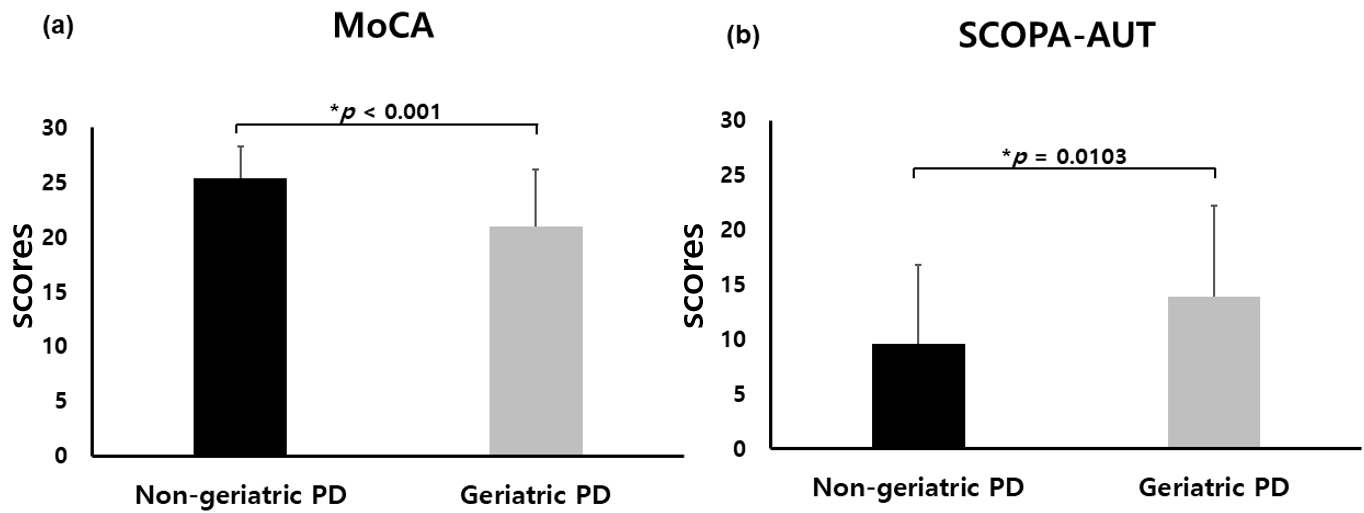 Fig. 2.
Fig. 2.
Clinical distinctions between geriatric and non-geriatric patients diagnosed with de novo Parkinson’s disease (PD). Geriatric patients with de novo PD exhibited lower MoCA scores (a) and elevated SCOPA-AUT scores (b) relative to their non-geriatric counterparts.
To further identify factors characteristic of geriatric PD, we conducted
logistic regression analyses as presented in Table 2. Univariable logistic
regression analysis demonstrated that shorter height (OR 0.0040, 95% CI
0.0001–0.4856, p = 0.0242), lower educational attainment (OR 0.8622,
95% CI 0.7882–0.9433, p = 0.0012), reduced MoCA scores (OR 0.7547,
95% CI 0.6621–0.8603, p
| Variable | Univariable | Multivariable | ||||
| Odd ratio | 95% CI | p-value | Odd ratio | 95% CI | p-value | |
| Gender-female | 1.3490 | 0.6086–2.9900 | 0.4611 | |||
| Body weight, kg | 0.9678 | 0.9294–1.0077 | 0.1127 | |||
| Height, m | 0.0040 | 0.0001–0.4856 | 0.0242 | |||
| Body mass index (kg/m2) | 0.9820 | 0.8468–1.1389 | 0.8105 | |||
| Disease duration, yr | 0.7972 | 0.5085–1.2500 | 0.3234 | |||
| Educational level, yr | 0.8622 | 0.7882–0.9433 | 0.0012 | |||
| Diabetes mellitus, n (%) | 1.0526 | 0.4304–2.5743 | 0.9105 | |||
| Hypertension, n (%) | 1.2773 | 0.5780–2.8229 | 0.5452 | |||
| History of previous falls, n (%) | 1.4481 | 0.6491–3.2306 | 0.3657 | |||
| UPDRS-III (motor) | 1.0212 | 0.9850–1.0587 | 0.2541 | |||
| H&Y stage | 1.0432 | 0.4050–2.6870 | 0.9302 | |||
| MoCA-K (cognitive function) | 0.7547 | 0.6621–0.8603 | 0.7642 | 0.6712–0.8701 | ||
| BDI (depressive symptoms) | 1.0410 | 0.9840–1.1013 | 0.1620 | |||
| BAI (anxiety) | 1.0244 | 0.9604–1.0927 | 0.4633 | |||
| PFS (fatigue) | 1.0180 | 0.9938–1.0427 | 0.1458 | |||
| SCOPA-AUT (dysautonomia)# | 1.0726 | 1.0176–1.1306 | 0.0091 | 1.0640 | 1.0031–1.1286 | 0.0391 |
Multivariable logistic regression with step-wise variable selection was implemented.
CI, confidence interval; UPDRS-III, the Unified Parkinson’s disease rating scale-part 3.
#Total score of SCOPA-AUT was calculated excluding the sexual domain, as a substantial number of patients did not respond to the question regarding sexual dysfunction.
In this study, we categorized the study population into two groups: individuals with PD younger than 75 years and geriatric individuals with PD aged 75 and above, with a particular emphasis on drug-naïve de novo patients. Until the early 2000s, PD was generally believed to develop primarily in individuals around the age of 60 [20]. As the incidence of PD has risen among the very elderly, research on the distinguishing features of geriatric Parkinson’s disease has gradually emerged. Recent investigations have demonstrated that individuals aged 75 and older with parkinsonism under medication exhibit unique clinical and functional profiles when compared to younger individuals, with age and disease duration playing a significant role in symptom trajectory and treatment efficacy [21, 22, 23]. Furthermore, as previously mentioned, we excluded young-onset PD patients under the age of 50, since it is well-established that these younger patients display clinical features that differ from those with onset around their 60s [12, 13]. Taken together, this represents the first study dedicated to examining the clinical features of geriatric PD. More specifically, the aim of this research was to assess differences in clinical traits between PD patients aged 75 or older and those around 60 years of age. By conducting this study, we sought to delineate critical clinical characteristics that warrant increased attention from clinicians when initially evaluating geriatric PD patients, not only in clinical practice but also within the context of research.
Geriatric PD patients were observed to be shorter in stature and to have attained a lower education level when compared with non-geriatric PD patients (Table 1). These disparities are likely attributable to the distinct socioeconomic background in Korea, heavily shaped by modernization during the 1900s and the Korean War in the 1950s. Nevertheless, there were no significant differences identified between the two groups with respect to metabolic conditions including diabetes and hypertension. Additionally, no substantial variations were detected regarding the timing of their first hospital visit for PD symptoms, the severity of motor symptoms at initial presentation, or the history of falls between the groups. These results indicate that, within a relatively stable society, geriatric PD patients are unlikely to show notable differences in demographic variables or motor symptoms when compared to non-geriatric PD patients, excepting differences related to age itself.
Interestingly, we observed that the pattern of non-motor symptoms varied between geriatric and non-geriatric patients with PD. No significant differences were found between the two groups regarding non-motor symptoms such as depression, anxiety, and fatigue. However, the geriatric group exhibited lower MoCA scores, indicating more pronounced cognitive impairment, and higher SCOPA-AUT scores, reflecting increased severity of autonomic dysfunction (Table 1 and Fig. 2). To assess whether these differences were independent of variables such as education level and age, we conducted logistic regression analysis and verified that both MoCA and SCOPA-AUT independently correlated with geriatric PD (Table 2). These results underscore the necessity for clinicians to closely monitor cognitive impairment and autonomic symptoms in the management of geriatric PD patients.
It is widely recognized that older individuals with PD experience a more rapid progression of cognitive decline and develop dementia earlier compared to younger patients [24]. Furthermore, a previous meta-analysis demonstrated that cognitive impairment in PD is associated with advanced age, lower educational levels, longer disease duration, higher levodopa dosages, greater severity of motor symptoms, as well as apathy and depression [25]. Consistent with existing literature, our findings show that cognitive impairment is a significant factor even at the very early stage of geriatric PD, regardless of other clinical characteristics (Table 2). The cognitive decline identified in geriatric PD patients highlights the importance of performing early and regular cognitive assessments in this demographic. To date, the exact pathophysiological mechanisms underlying cognitive deficits linked to aging in PD remain unclear. One plausible explanation is that more individuals in the older age group with PD have concomitant pathological alterations characteristic of Alzheimer’s disease [26]. Therefore, even in cases of de novo PD, cognitive decline could be more evident in this subgroup. Another potential contributing factor is that aging may disrupt the integrity of the blood-brain barrier, leading to impaired immune responses and subsequent neurodegeneration, which may facilitate cognitive deterioration in the geriatric population [27]. Furthermore, aging is associated with increased iron accumulation in the brain, particularly in deep gray matter structures such as the hippocampus and basal ganglia. This phenomenon may lead to cognitive decline, especially among geriatric patients with PD [28, 29].
Autonomic dysfunction in PD may manifest as early as the prodromal stage, with a
well-documented trend of progressive deterioration as the disease advances [30].
Recent evidence indicates that patients with PD who exhibit severe autonomic
symptoms tend to experience significantly poorer outcomes compared to those
presenting with milder symptoms [31]. The transmission of
Several limitations should be acknowledged for this study. First, the
retrospective design conducted within a single institution introduces the risk of
selection bias, potentially restricting the general applicability of our results.
Second, although global cognitive function was measured using the MoCA, we did
not incorporate in-depth assessments of specific cognitive domains. In addition,
cognitive impairment can be confounded by subclinical cerebrovascular disease,
sleep disorders, or polypharmacy. We could not address in this study. Third, the
evaluation of autonomic dysfunction relied upon the SCOPA-AUT, a subjective
instrument, and objective physiologic measurements were not included. Despite
these constraints, our study successfully identified unique clinical features in
geriatric patients newly diagnosed with PD. However, especially in geriatric
people, self-reported autonomic assessments may have limitations in accuracy and
reliability due to factors such as cognitive impairment or recall bias. Future
studies incorporating neuroimaging (e.g., hippocampal volume, white matter
hyperintensity) or biomarkers (e.g., plasma pTau,
In conclusion, geriatric patients with PD demonstrated lower cognitive performance and more pronounced autonomic dysfunction compared with non-geriatric patients with PD. In multivariable logistic regression analysis, decreased cognitive scores and higher autonomic dysfunction scores were identified as significant independent predictors of geriatric PD. Our data indicate that geriatric patients with de novo PD display more substantial cognitive deficits and greater autonomic dysfunction than non-geriatric de novo PD patients. These findings underscore the importance for clinicians to recognize these features when treating geriatric patients and to ensure these considerations are integrated into clinical research.
PD, Parkinson’s disease; BMI, Body mass index; MRI, magnetic resonance imaging; 18F-FP-CIT PET, 18F-fluoropropyl-carbomethoxy-3
The data underlying this article will be shared on reasonable request to the corresponding author.
Conceptualization, KYK; Methodology, KYK; Data curation, JY, ROK and KYK; Formal analysis, KYK; Funding acquisition, KYK; Investigation, JY, ROK and KYK; Writing - original draft, KYK; Writing - review & editing, JY, ROK and KYK; Supervision, KYK. All authors have read and agreed to the published version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All procedures were performed in accordance with ethical standards of the institution and the national research committee as well as with the 1964 Helsinki Declaration and its subsequent amendments. This study was retrospective and was approved with waiver of individual informed consent by the ethics committee of our Institutional Soonchunhyang University Seoul Hospital Review Board (IRB No. 2025-01-003).
We thank all staffs in our department of Neurology for their kind and enthusiastic support.
This research was supported by a grant of the Korea Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2023-KH134527) and was supported by the Soonchunhyang University Research Fund (grant number: 202413004).
The authors declare no conflict of interest.
During the preparation of the manuscript, we used ChatGPT-4o to improve readability and language. After using this tool, we reviewed and edited the content as needed and takes full responsibility for the final publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


