1 Department of Cardiology, Sorbonne University, 75013 Paris, France
2 Department of Systems Medicine, “Tor Vergata” University, 00133 Rome, Italy
3 Department of Cardiology, Royal Stoke University Hospital, University Hospitals of North Midlands NHS Trust, ST4 6QG Stoke-on-Trent, UK
4 Keele Cardiovascular Research Group, Centre for Prognosis Research, Keele University, ST5 5BG Keele, UK
A recent meta-analysis suggests greater clinical benefit of Proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9i) among patients with baseline Low-Density-Lipoprotein cholesterol (LDL-C) levels of 100 mg/dL or higher, with cardiovascular mortality reduction observed in the alirocumab-treated post-acute coronary syndrome (ACS) population included in the ODYSSEY OUTCOMES [1]. Importantly, no head-to-head randomized trials have directly compared alirocumab and evolocumab across different baseline LDL-C strata, and available network meta-analyses do not demonstrate meaningful differences in relative risk reduction for major cardiovascular events (MACE) between the two agents. Therefore, any apparent heterogeneity in clinical benefit according to baseline LDL-C should not be interpreted as evidence of intrinsic differences in drug efficacy.
This Editorial does not aim to compare the intrinsic efficacy of alirocumab and evolocumab, but rather to discuss how baseline LDL-C, residual patient risk, trial design, and clinical context modulate the observed magnitude of clinical benefit and may contribute to the perception of differential effects between these two PCSK9 inhibitors (Fig. 1).
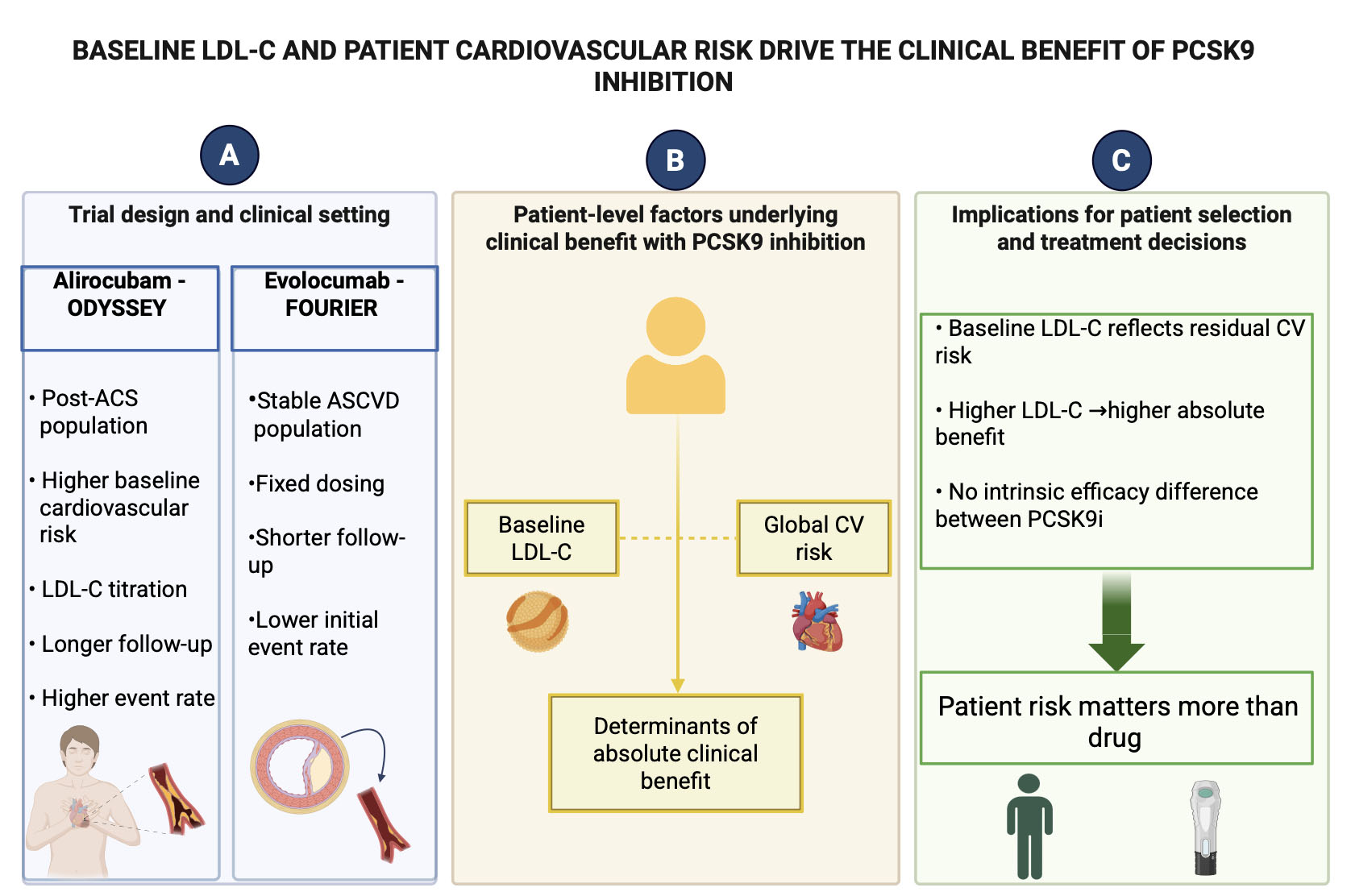 Fig. 1.
Fig. 1.
Baseline LDL-C and patient cardiovascular risk drive the clinical benefit of PCSK9 inhibition. (A) Summarizes the main differences in trial design and clinical setting between alirocumab in ODYSSEY OUTCOMES and evolocumab in FOURIER. ODYSSEY OUTCOMES enrolled patients after acute coronary syndrome, characterized by higher baseline cardiovascular risk, LDL-C titration and longer follow-up, whereas FOURIER included patients with stable atherosclerotic cardiovascular disease treated with fixed-dose evolocumab and lower initial event rates. Figure legends: ACS (acute coronary syndrome), LDL-C (Low-Density-Lipoprotein cholesterol), ASCVD (stable atherosclerotic cardiovascular disease), CV (cardiovascular), PCSK9i (Proprotein convertase subtilisin/kexin type 9 inhibitors). (B) Schematically illustrates the patient-related factors underlying the magnitude of absolute clinical benefit with PCSK9 inhibition. Baseline LDL-C levels and global cardiovascular risk are shown as coexisting and complementary determinants that define residual cardiovascular risk and, consequently, the potential absolute benefit of treatment. The schematic emphasizes that clinical benefit is primarily driven by patient characteristics rather than by differences between PCSK9 inhibitor molecules. (C) Outlines the clinical implications, indicating that baseline LDL-C reflects residual cardiovascular risk and potential absolute benefit, without intrinsic efficacy differences between PCSK9 inhibitors; therefore, patient risk profile rather than molecule choice should guide clinical decision-making.
Both alirocumab and evolocumab provide large and consistent relative risk reductions in MACE across a wide range of baseline LDL-C levels. However, the apparent modulation of absolute benefit by baseline LDL-C is more clearly illustrated in the ODYSSEY OUTCOMES, conducted in a post-ACS setting, than with evolocumab in stable atherosclerotic cardiovascular disease (ASCVD), since the baseline risk was higher and the follow-up longer in ODYSSEY OUTCOMES. These factors directly influence MACE rate, as demonstrated by the randomised controlled trial (RCT) meta-analysis and by comparison between the FOURIER and OLE FOURIER trials [2]. Indirect and real-world comparisons do not support a major intrinsic difference between the two monoclonal PCSK9i; observed gradients by baseline LDL-C mostly reflect trial design, background risk, and targeting strategies rather than molecule-specific biology [3].
In ODYSSEY OUTCOMES, patients enrolled after recent ACS had a median baseline LDL-C of approximately 92–93 mg/dL, with enrichment of higher-risk individuals, and were treated with a titration-based alirocumab strategy aimed at achieving very low LDL-C levels. In contrast, FOURIER enrolled patients with ASCVD and a similar average baseline LDL-C, who were treated with fixed-dose evolocumab without titration and achieved LDL-C levels of approximately 30 mg/dL early during follow-up [4]. Thus, despite comparable baseline LDL-C levels, the two programs differed substantially in the clinical setting, baseline cardiovascular risk, treatment algorithm, and event rates factors that are expected to critically influence absolute risk reduction and overall clinical benefit.
In ODYSSEY OUTCOMES, alirocumab was associated with a significant reduction in MACE, with a pronounced gradient of absolute benefit across baseline LDL-C strata. Patients with baseline LDL-C of 100 mg/dL or higher derived the greatest benefit, including a significant reduction in cardiovascular mortality. Importantly, this finding should not be interpreted as evidence of the superiority of alirocumab over evolocumab, but rather as a consequence of higher baseline cardiovascular risk, longer follow-up, and a treat-to-target strategy in a post-ACS population. In contrast, patients with lower baseline LDL-C experienced smaller absolute risk reductions despite similar proportional LDL-C lowering, consistent with observations from other randomized controlled trials [5]. Meta-regression analyses across PCSK9i and statin trials further support a relationship between higher baseline LDL-C and greater mortality benefit, in line with the cholesterol-years concept and cumulative exposure to cardiovascular risk.
In FOURIER, evolocumab was associated with marked LDL-C reduction and a consistent relative risk reduction in MACE across a broad range of patient subgroups [4]. Analyses according to LDL-C levels achieved early in follow-up demonstrated a monotonic association between lower achieved LDL-C and lower event rates [6]. In contrast to ODYSSEY OUTCOMES, FOURIER did not show a strong qualitative interaction between baseline LDL-C and relative treatment efficacy, with broadly similar relative risk reductions across baseline LDL-C categories and differences mainly reflected in absolute risk reduction. This pattern is consistent with the inclusion of a lower-risk, stable atherosclerotic cardiovascular disease population and a fixed-dose treatment strategy. Accordingly, the absence of a mortality benefit signal in FOURIER should be interpreted in the context of lower baseline cardiovascular risk, a stable disease setting, and a shorter duration of follow-up, rather than as a lack of efficacy of evolocumab. Overall, FOURIER and its open-label extension support the principle that “lower and longer is better”, highlighting the importance of cumulative exposure to LDL-C lowering over time [2].
Network meta-analyses suggest that evolocumab may achieve slightly greater percentage LDL-C reductions than standard alirocumab doses at comparable time points [3]. However, indirect comparisons of cardiovascular outcomes do not demonstrate meaningful differences in relative risk reduction for major adverse cardiovascular events between the two agents after accounting for baseline risk and follow-up duration. Real-world studies in secondary prevention report broadly similar LDL-C reductions and on-treatment LDL-C levels with both agents, despite differences in baseline LDL-C and background lipid-lowering therapy, including more frequent ezetimibe use among patients treated with alirocumab [7]. Safety profiles are largely comparable, with no major differences across clinically relevant adverse events. Taken together, these findings support the interpretation that apparent differences in clinical benefits according to baseline LDL-C are driven predominantly by patient risk profiles, clinical setting (post-ACS vs chronic ASCVD), and treatment strategies, rather than by intrinsic differences between the two monoclonal antibodies [8].
In stable ASCVD with LDL-C
PS and MM substantial contributions to the conception or design of the work. DMG made the central figure, MM has reviewed the first draft of manuscript. All authors have reviewed and approved the final manuscript. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

