1 School of Medicine, Case Western Reserve University, Cleveland, OH 44106, USA
2 Department of Surgery, University Hospitals Cleveland Medical Center, Cleveland, OH 44106, USA
3 Division of Cardiovascular Surgery, University Hospital, 15706 Santiago de Compostela, Spain
4 Division of Cardiac Surgery, Harrington Heart and Vascular Institute, University Hospitals Cleveland Medical Center, Cleveland, OH 44106, USA
5 Faculty of Medicine, University of Toronto, Toronto, ON M5S 1A8, Canada
†These authors contributed equally.
Abstract
Postoperative atrial fibrillation, heart failure, and pericardial effusion remain frequent complications after cardiac surgery. Pericardial fluid is a localized inflammatory compartment in close contact with the myocardium and may provide information that complements systemic biomarkers. This narrative review summarizes inflammatory mediators identified in pericardial fluid after cardiac surgery and their associations with postoperative outcomes. We discuss potential mechanistic links between pericardial inflammation and pericardial fluid markers, as well as practical limitations related to sampling, timing, and standardization. Although pericardial fluid biomarkers may augment existing clinical risk models, prospective studies are needed to determine their incremental value and clarify their role in perioperative management.
Keywords
- pericardium
- pericardial fluid
- inflammatory markers
The pericardium is a two-layered, fibroserous sac that contributes to the mechanical protection and stability of the heart, as well as neurohumoral and immunological regulation. The space between the visceral and parietal layers of the serous pericardium contains the pericardial fluid (PCF). During pathological conditions, the pericardial space becomes a site of ‘dynamic inflammatory activity’ with several biochemical and molecular changes. Surgical trauma, myocardial disorders, and inflammatory changes can provoke an intense inflammatory response in the pericardial space, leading to cellular and molecular changes and the selective accumulation of inflammatory mediators in the pericardial compartment. Multiple studies have reported the role of local pericardial inflammation in the pathogenesis of adverse clinical outcomes in patients undergoing cardiac surgery. However, the utilization of these biomarkers in routine clinical practice remains very limited. This may be attributed, in part, to technical challenges in collecting and processing pericardial fluid samples, as well as the scarcity of solid evidence on the predictive value of these biomarkers in the clinical setting. This review aims to consolidate current evidence and provide an overview of pericardial inflammatory mediators and their potential role in predicting clinical outcomes in patients undergoing cardiac surgery.
This narrative review synthesizes current evidence on inflammatory mediators in pericardial fluid and their clinical relevance in cardiac surgery. Literature searches covered PubMed and the Cochrane Library for English-language, peer-reviewed articles published through February 2025, using combined search terms such as pericardial fluid, inflammation, cardiac surgery, cytokines, natriuretic peptides, galectin-3, troponin, and microRNAs. Both experimental, translational, and clinical studies were included in the review. Given the heterogeneity in study designs and outcomes, a systematic review or meta-analysis was not feasible; therefore, findings are summarized narratively, with particular emphasis on limitations and clinical relevance.
Pericardial fluid (PCF) is mainly an ultrafiltrate of plasma. It is derived from the visceral pericardial microvasculature [1]. This fluid originates in the cardiac interstitium and diffuses across the epicardial layer into the pericardial sac. The composition of PCF can represent the biochemical and hormonal microenvironment of the cardiac interstitium. The pericardial sac also encloses epicardial adipose tissue. This tissue is important for biochemical, hormonal, and metabolic regulation of cardiac homeostasis [2]. PCF has a low turnover rate. This is shown by extended residence times of radiolabeled growth factors and albumin in animal models. Therefore, PCF has emerged as an important marker for the diagnosis and prognosis of different cardiac disorders, such as myocardial ischemia, congestive heart failure, cardiac arrhythmias, and pathological pericardial effusions.
The pericardium can respond to injury in two ways: fluid exudation leading to PCF accumulation, or acute inflammation. Pericardial disorders may involve one main component, but both reactions often occur together [3]. Clinical presentation varies from subtle changes to life-threatening cardiac tamponade due to PCF accumulation and impaired cardiac output. These well-known features of the pericardium matter postoperatively because the pericardial space acts as a biologically active compartment, where inflammatory mediators can persist near the atrial and ventricular myocardium.
Cardiac surgery triggers a systemic inflammatory response that contributes substantially to postoperative organ dysfunction and coagulation disorders that can adversely impact clinical outcomes [4, 5, 6].
In the perioperative period of cardiac surgery, inflammatory markers display a
temporal pattern that reflects the sum of baseline patient risk profile, the
extent of surgical trauma, Cardiopulmonary bypass (CPB)-induced systemic
inflammation, and postoperative organ dysfunction. At baseline, patients
undergoing cardiac surgery often present with elevated markers such as C-reactive
protein (CRP) and interleukin-6 (IL-6) secondary to underlying cardiovascular
disease, complex comorbidities (e.g., congestive heart failure, coronary artery
disease), and chronic low-grade inflammation. Following surgery, an early
increase in acute-phase reactants and cytokines is consistently reported; serum
CRP typically peaks at ~48 hours postoperatively, while cytokines
such as IL-6 and IL-8 show a rapid surge within the first 24 h. Several recent
investigations have also shown that the pericardial fluid compartment undergoes a
more pronounced inflammatory response than plasma in the early postoperative
period, with marked elevations of IL-6, IL-8, tumor necrosis factor-
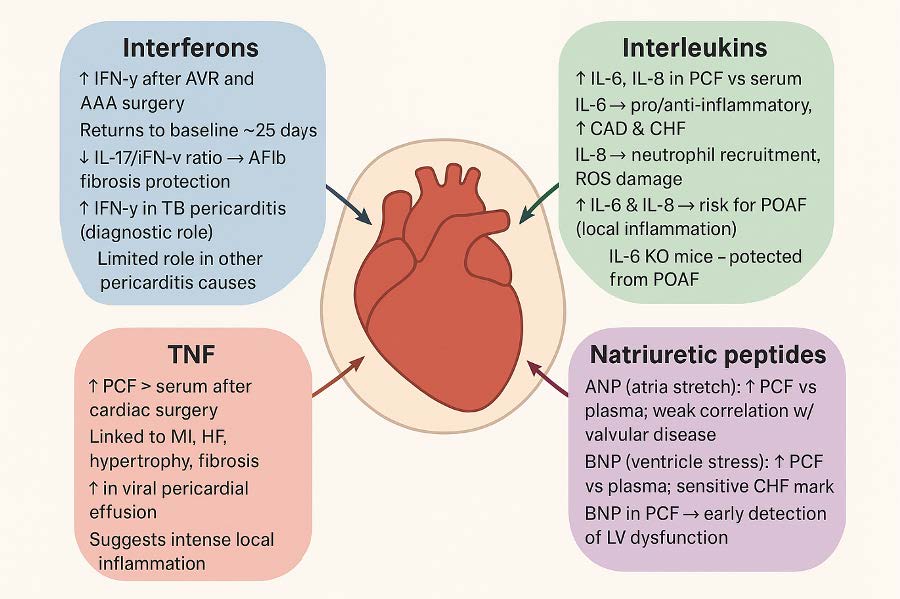 Fig. 1.
Fig. 1.
Biomarkers in the pericardial fluid. IFN, Interferon; AVR, Aortic Valve Replacement; AAA, Abdominal Aortic Aneurysm; PCF, Pericardial Fluid; CAD, Coronary Artery Disease; CHF, Congestive Heart Failure; ROS, Reactive Oxygen Species; POAF, Postoperative atrial fibrillation; KO, Knock-out; MI, Myocardial Infarction; HF, Heart Failure; ANP, Atrial Natriuretic Peptide; BNP, B-type Natriuretic Peptide; LV, Left Ventricle.
Pericardial inflammation can lead to several changes in the biochemical composition of pericardial fluid and serum. While serum markers such as CRP and erythrocyte sedimentation rate (ESR) are widely used in the management of pericardial inflammation, these represent systemic inflammation and lack specificity for pericardial processes. Furthermore, PCF contains a concentrated proportion of acute-phase reactants, cytokines, and pro-inflammatory mediators, which are upregulated during an inflammatory process. These may be useful in differentiating between infectious and non-infectious etiologies, in addition to their role as prognostic markers. Despite limited use in clinical practice, some of the most important ones are discussed below [7, 8]. These biomarkers are summarized in Table 1.
| Marker | Marker class | Key findings in pericardial fluid | Proposed clinical application/significance | Limitations | Timing and collection method |
| Interferon- |
Cytokine (Interferon) | Elevated PCF levels vs serum in cardiac surgery; high sensitivity and specificity for tuberculosis pericarditis; no PCF–serum gradient in autoimmune, neoplastic, or viral pericarditis | Potential diagnostic marker for tuberculous pericarditis; possible association with reduced atrial fibrosis and atrial fibrillation | Limited utility outside TB pericarditis; largely correlational data; inconsistent findings across etiologies | Intraoperative Collection |
| Interleukin-6 (IL-6) | Cytokine (Interleukin) | Markedly higher PCF vs serum levels; elevated in chronic heart failure; increased following cardiac surgery | Indicator of localized pericardial inflammation; potential predictor of postoperative atrial fibrillation (POAF) | Not disease-specific, reflective of inflammation but not etiology | Intraoperative Collection |
| Interleukin-8 (IL-8) | Cytokine (Interleukin) | Significantly higher levels of IL-8 in PCF vs serum | Marker of neutrophil-driven local inflammation; potential predictor of POAF | Limited prospective data; influenced by surgical trauma | Intraoperative Collection |
| Tumor Necrosis Factor (TNF) | Cytokine | Higher PCF than serum in cardiac surgery; elevated in viral pericardial effusions | Marker of intense local inflammatory response; potential indicator of viral pericarditis | Overlaps with other inflammatory conditions; unclear prognostic value | Intraoperative Collection |
| Atrial Natriuretic Peptide (ANP) | Natriuretic peptide | Higher PCF than plasma in cardiovascular surgery; weak correlation with valvular disease | Possible marker of cardiac wall stress or valvular pathology | Sparse and outdated literature; limited clinical validation | Intraoperative collection |
| B-type Natriuretic Peptide (BNP) | Natriuretic peptide | PCF concentrations up to 22-fold higher than plasma in LV dysfunction; correlates with CHF and AF | Potential early marker of ventricular dysfunction and heart failure | Serum BNP already well-validated marker of disease | Intraoperative collection |
| Galectin-3 (Gal-3) | Lectin | Experimental models show increased fibrosis when present in pericardial space | Potential biomarker and mediator of cardiac fibrosis and remodeling | Primarily preclinical evidence; not yet clinically validated | Intraoperative Collection |
| MicroRNAs (miRNAs) | Non-coding RNA | Distinct PCF miRNA profile; altered expression in STEMI; some miRNAs linked to HF and inflammation | Emerging diagnostic and therapeutic targets; possible paracrine modulators | Underdeveloped field; heterogeneous findings; unclear clinical thresholds | Intraoperative Collection |
| Troponin I (cTnI) | Myocardial injury marker | Elevated PCF levels post-cardiac surgery; higher levels associated with POAF | Possible marker of perioperative myocardial injury and POAF risk | Serum cardiac troponin is already well-validated marker of ischemia; | Postoperative Collection via Pericardial Drain |
| C-reactive Protein (CRP) | Acute-phase protein | Detectable in PCF despite size; reflects local inflammation | Marker of pericardial inflammation and disease activity | Poor specificity; limited etiologic discrimination | Intraoperative Collection |
The primary functions of Atrial natriuretic peptide (ANP) include natriuresis, diuresis, and vasodilation, thus controlling blood pressure [9]. The study by Soós et al. [10] found that, in patients undergoing cardiovascular surgery, pericardial ANP levels were significantly higher than venous plasma concentrations. Additionally, they found a weak correlation between PCF ANP concentrations and valvular heart disease [10]. However, research on ANP in pericardial fluid is scarce, with most studies published over 20 years ago. This raises concerns about the use of pericardial ANP in practice without further research.
B-type natriuretic peptide (BNP) promotes natriuresis, diuresis, and
vasodilation and inhibits the renin-angiotensin-aldosterone system during periods
of increased ventricular wall stress [11, 12]. In patients undergoing cardiac
surgery with impaired left ventricular function, pericardial BNP levels were
22-fold higher than plasma concentrations (p
Interleukins modulate immune response as well as cell proliferation and
differentiation. IL-6 concentrations were significantly increased in the PCF
(183.81
Interferons are cell-signaling glycoproteins that are typically released in
association with the innate immune system’s response to infection and neoplastic
proliferation. In patients receiving aortic valve replacement surgery, there was
a significantly increased interferon gamma (IFN-
Tumor necrosis factor (TNF) functions to promote leukocyte recruitment,
stimulation of acute phase reactants, and mediation of cell death. Increased
serum TNF has been associated with many forms of cardiac pathology, including
myocardial infarction, heart failure, hypertrophy, and fibrosis [23]. In patients
receiving cardiac surgery, the PCF levels of TNF were significantly higher than
those of the serum (Serum: 3.18
C-reactive protein (CRP) is a relatively large protein at 118 kDa, which cannot easily diffuse into the pericardial sac from the epicardium, which is typically permeable for molecules smaller than 40 kDa [24]. Despite this, CRP has been detected within the pericardial fluid of CABG patients who were receiving surgery for stable angina, unstable angina, and surgery following myocardial infarction [24]. This is likely due to increased vascular permeability caused by inflammatory changes local to the pericardium and surrounding tissues. CRP has also been proposed for monitoring acute pericarditis to assess disease severity and treatment response [25]. While CRP in the PCF can confirm the presence of local inflammation, it does not reliably distinguish between causes of disease or inflammation.
Galectin-3 (Gal-3) is a
MicroRNAs (miRNAs) in PCF act as paracrine signaling molecules that modulate gene expression in the nearby cardiac cells. PCF contains a distinct miRNA profile as compared to the heart. For example, miR-1, which is abundant in cardiac tissues, had very low levels in PCF and was detectable in only 28/51 samples [29]. There has been limited research on the significance of miRNAs already present in the PCF; however, some miRNAs have previously been identified as possible diagnostic biomarkers for heart failure [30]. Delivery of miRNAs to the pericardial sac has been proposed as a therapeutic approach for cardiovascular disease [31]. Several miRNAs have been shown to be upregulated in PCF in patients with ST-Elevation Myocardial Infarction (STEMI), and miR-22-3p levels are significantly higher in STEMI patients than in Non–ST-Elevation Myocardial Infarction (NSTEMI) patients or non-ischemic controls [32]. The study of miRNAs in PCF remains underdeveloped, and further research is needed to determine their clinical significance.
Within the PCF, there is evidence that there is increased troponin I (cTnI) in
the PCF following cardiac surgery, likely due to myocardial damage during surgery. Butts et al. [3] found that cTnI levels in the PF were
significantly increased 48 hours postoperatively in individuals who developed
POAF (0.22
Despite growing interest in pericardial fluid biomarkers, several important limitations must be acknowledged. First, most available evidence is derived from small, single-center, observational or translational studies, often with heterogeneous patient populations, surgical procedures, and sampling protocols. Differences in the timing of pericardial fluid collection (intraoperative vs. early or late postoperative), variability in drain-based sampling, and lack of standardized processing methods limit direct comparison across studies. Second, many associations between pericardial biomarkers and postoperative outcomes are correlational, and causality cannot be inferred in most clinical settings. While experimental data support mechanistic roles for certain mediators, such as IL-6, in fibrosis and electrical remodeling in humans, these biomarkers may primarily reflect underlying myocardial stress and inflammatory burden rather than act as independent drivers of pathology. Third, validated cutoff values, assay standardization, and reproducibility across laboratories are largely lacking, which currently restricts clinical implementation. Practical considerations, including the feasibility of routine pericardial fluid sampling, narrow postoperative time windows before drain removal, inter-institutional variability, and cost-effectiveness, further challenge real-world adoption. Finally, although pericardial fluid biomarkers may provide biologically enriched information compared with systemic markers, robust prospective studies demonstrating incremental predictive value beyond existing clinical risk scores and serum biomarkers are still needed.
Despite major advances, current clinical postoperative risk models rely heavily on preoperative variables (e.g., age, left atrial size, left ventricular function, surgery type, and CPB), intraoperative metrics (e.g., procedure type and urgency), and nonspecific systemic markers (e.g., leukocyte count, CRP). While useful, these models capture only a part of the risk spectrum and lack insight into the localized biological processes occurring within the pericardial environment [9]. Subsequently, clinicians often lack timely markers indicative of postoperative complications. Further efforts should be directed toward the PCF, which can provide insight into the dynamic responses of body organs to surgical trauma, thereby enabling early detection of complications [35].
Moreover, it has been well established that systemic biomarkers such as C-reactive protein and troponin are pivotal for clinical assessment, reflecting localized disease activity and myocardial involvement, but are limited in specificity. In the postoperative setting, when systemic inflammation is generalized, localized pericardial fluid biomarkers may provide insight into compartmental changes within the pericardium [36].
Furthermore, the pericardial space is uniquely placed as it holds the epicardium, autonomic fibers, and coronary vasculature. Pericardial cytokines and natriuretic peptides are consistently higher in PCF than in plasma, making PCF an early-phase reporter of myocardial stress, microinjury, and fibrosis [8]. Translational efforts to integrate the markers discussed in this review into perioperative care may allow clinicians to detect subclinical injury before clinical deterioration becomes clear [16]. Specifically, routine measurements of PCF biomarkers may enable proactive arrhythmia prevention, refined hemodynamic monitoring, heart failure management, improved identification of pericardial tamponade or effusion, and enhanced differentiation of superficial versus deep myocardial injury.
Measuring markers pre-operatively and in the immediate post-operative period (e.g., 24–48 h) in interventional randomized studies, via pericardial drain sampling, may help identify the subgroup of patients at higher risk of developing postoperative complications such as heart failure, cardiac tamponade, myocardial ischemia, or POAF, thus allowing for close monitoring and the implementation of early preventative and therapeutic strategies (e.g., the initiation of anti-coagulation or anti-inflammatory regimens, and the early start of anti-arrhythmic medications). Further, it has been demonstrated that adding biomarker-derived data to existing clinical risk assessment scores may improve discrimination and calibration by capturing the individualized biologic response to surgery rather than relying solely on clinical predictors [37]. Finally, understanding the biological and clinical roles of pericardial biomarkers, such as inflammatory cytokines, can enable the identification of novel therapeutic targets and the initiation of targeted therapies to prevent postoperative complications.
The pericardial fluid is a dynamic, mediator-rich environment that exhibits a compartmentalized, distinctive pattern of inflammation after cardiac surgery. The accumulation of pro-inflammatory and pro-fibrotic biomarkers within the pericardial cavity can play an important role in risk-stratification for patients undergoing cardiac surgery, as well as in the early detection and prevention of major postoperative complications such as atrial fibrillation and heart failure. The addition of biomarker-derived data to existing clinical risk scores can improve their performance, enable the implementation of patient-specific preventive strategies, and potentially lead to improved clinical outcomes.
ANP, Atrial Natriuretic Peptide; AF, Atrial Fibrillation; BNP, B-type Natriuretic Peptide; CABG, Coronary Artery Bypass Grafting; CAM, Cellular Adhesion Molecules; CPB, Cardiopulmonary Bypass; CRP, C-Reactive Protein; cTnI, Cardiac Troponin I; ECM, Extracellular Matrix; ESR, Erythrocyte Sedimentation Rate; Gal-3, Galectin-3; HF, Heart Failure; IFN, Interferon; IFN-
MB: Literature review, manuscript drafting and editing; HNC: Literature review, manuscript drafting and editing; ALFG: Manuscript editing, conception and design; YA-O: Manuscript editing, conception and design; AE: Manuscript editing, conception and design; ME-D: Primary Investigator, manuscript editing and final approval. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

