1 Department of Cardiovascular Surgery, The First Affiliated Hospital of Guangxi Medical University, 530021 Nanning, Guangxi, China
Abstract
Heart failure (HF) is a major global cause of hospitalization and mortality, representing a complex clinical syndrome with significant unmet therapeutic needs. Sodium–glucose cotransporter 2 inhibitors (SGLT2is), originally developed for glycemic control, have recently demonstrated remarkable efficacy in the management of HF. This review comprehensively examines the mechanisms of action and therapeutic potential of SGLT2is in HF, with a focus on their multifaceted effects on hemodynamics, cardiac metabolism, inflammatory responses, oxidative stress, and neuroendocrine activation. In addition, clinical trial outcomes and safety profiles of SGLT2is in HF with reduced ejection fraction (HFrEF), HF with preserved ejection fraction (HFpEF), and HF with mid-range ejection fraction (HFmrEF) are thoroughly evaluated. Finally, this article discusses future research directions and clinical application prospects, aiming to provide novel insights and strategies for treating HF.
Keywords
- sodium–glucose cotransporter 2 inhibitors
- heart failure
- mechanisms of action
- clinical therapy
Heart failure (HF) represents the terminal stage of various cardiovascular
diseases, affecting over 64 million individuals worldwide, with its prevalence
continuing to rise due to aging populations and the increasing incidence of
metabolic disorders [1]. Despite advances in pharmacologic and device-based
therapies—such as renin–angiotensin system inhibitors,
Sodium–glucose cotransporter 2 inhibitors (SGLT2is), initially developed as antidiabetic agents that block renal glucose reabsorption, have emerged as a major therapeutic breakthrough in HF management [6]. Beyond their glucose-lowering effect, large-scale clinical trials and meta-analyses have demonstrated that SGLT2is significantly reduce cardiovascular death and HF hospitalizations and provide renal protection across diverse patient populations, including those without diabetes [6, 7, 8].
Despite the promising therapeutic potential of SGLT2is in HF, the complexity and heterogeneity of their mechanisms of action warrant further investigation [9]. This review aims to comprehensively explore the mechanisms and therapeutic potential of SGLT2is in HF, with an in-depth analysis of their multifaceted effects on hemodynamics, cardiac metabolism, inflammatory responses, oxidative stress, and neuroendocrine activation. Furthermore, we systematically evaluate clinical trial outcomes of SGLT2is in HF with reduced ejection fraction (HFrEF), HF with preserved ejection fraction (HFpEF), and HF with mid-range ejection fraction (HFmrEF), along with a detailed discussion of their safety profile in clinical practice [10]. By elucidating the multi-target effects of SGLT2is, this review seeks to provide more precise and effective therapeutic strategies for HF patients, ultimately improving their prognosis and alleviating the global healthcare burden.
SGLT2is represent a novel class of oral antihyperglycemic agents that have received significant attention in the medical field due to their unique mechanism of action and broad clinical applications [11]. SGLT2is primarily function by inhibiting the activity of SGLT2 in the proximal renal tubules, thereby reducing glucose reabsorption and promoting urinary glucose excretion, which effectively lowers blood glucose levels [6] (Fig. 1). Beyond glycemic control, SGLT2is exert beneficial hemodynamic effects by increasing urinary sodium excretion and inducing osmotic diuresis, which alleviates cardiac preload, reduces blood pressure, and preserves cardiac function [12]. In addition, SGLT2is inhibit sodium–glucose cotransporter 1 (SGLT1) in cardiomyocytes, mitigating intracellular sodium overload, calcium overload, and oxidative stress, thereby improving ventricular remodeling and protecting myocardial tissue [13]. SGLT2is also modulate adipose tissue metabolism and inflammation, attenuate myocardial fibrosis, and enhance cardiac metabolic remodeling [13].
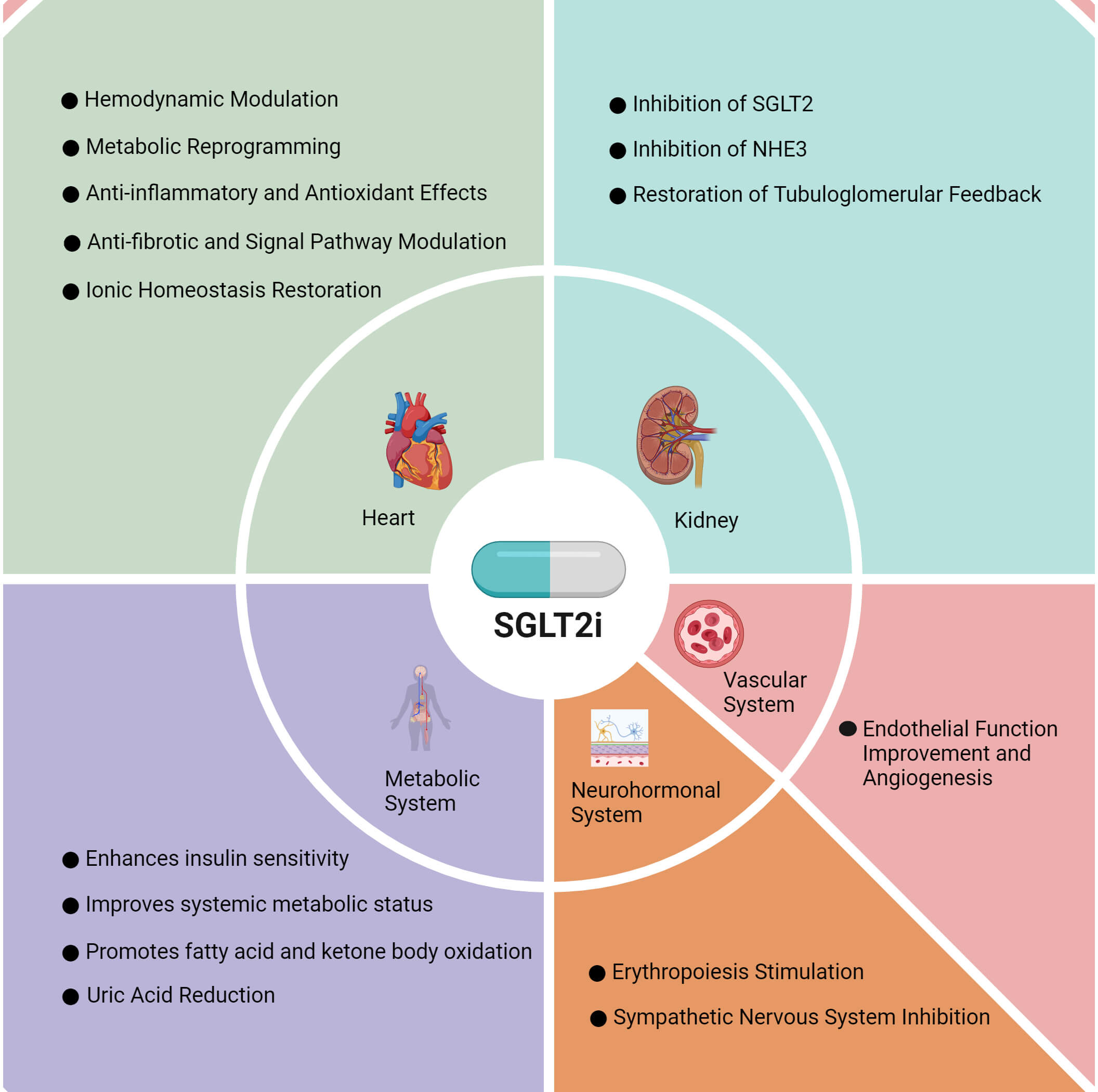 Fig. 1.
Fig. 1.
Therapeutic mechanisms of SGLT2is in HF. SGLT2is exert distinct therapeutic effects in the heart, kidney, metabolic system, neurohormonal system, and vascular system. NHE3, sodium–hydrogen exchanger 3; SGLT2is, sodium–glucose cotransporter 2 inhibitors; HF, heart failure. Fig. 1 was drawn using BioRender.
SGLT2is have a wide range of clinical applications, including use in adult patients with type 2 diabetes mellitus (T2DM) who have inadequate glycemic control with oral antihyperglycemic agents or basal insulin therapy, as well as in T2DM patients with comorbid cardiovascular disease, chronic kidney disease, or HF [14]. Commonly prescribed SGLT2is include dapagliflozin, empagliflozin, canagliflozin, and ertugliflozin. Numerous clinical trials have demonstrated that SGLT2is significantly reduce the composite endpoint of cardiovascular mortality and HF-related hospitalization in patients with both HFrEF and HFpEF [15, 16, 17]. SGLT2is also confer additional benefits, such as weight reduction, improved insulin sensitivity, decreased uric acid levels, and modest lowering of blood pressure [18]. In major heart failure trials, mean body weight reductions ranged between 1.5 and 2.5 kg within the first 6 months of therapy, and remained stable [19]. In Dapagliflozin And Prevention of Adverse-outcomes in Heart Failure (DAPA-HF), dapagliflozin led to an average weight loss of 1.8 kg compared with placebo at 8 months, while EMPEROR-Reduced and EMPEROR-Preserved trials reported similar findings [20]. Although SGLT2is are associated with an increased risk of urogenital infections and ketoacidosis, they are generally well tolerated in clinical practice, and most adverse effects are easily manageable [21]. Owing to their multifaceted mechanisms of action, SGLT2is have demonstrated remarkable efficacy and broad therapeutic potential in the management of T2DM and HF, offering new hope and expanded treatment options for patients.
SGLT2is improve hemodynamics and reduce blood pressure by inducing osmotic diuresis, which decreases intravascular fluid volume [22] (Fig. 2). This reduction in blood pressure not only lowers the resistance to cardiac output (afterload) but also reduces venous return (preload), thereby alleviating the initial workload on the heart and enabling more efficient cardiac pumping [23]. Studies have reported a modest antihypertensive effect of SGLT2is, with an estimated average blood pressure reduction of 2.46/1.46 mm Hg [24]. Notably, this antihypertensive effect persists even in patients with impaired renal function, suggesting mechanisms beyond simple diuresis, including improved endothelial function, reduced arterial stiffness, and modulation of sympathetic nervous activity [25].
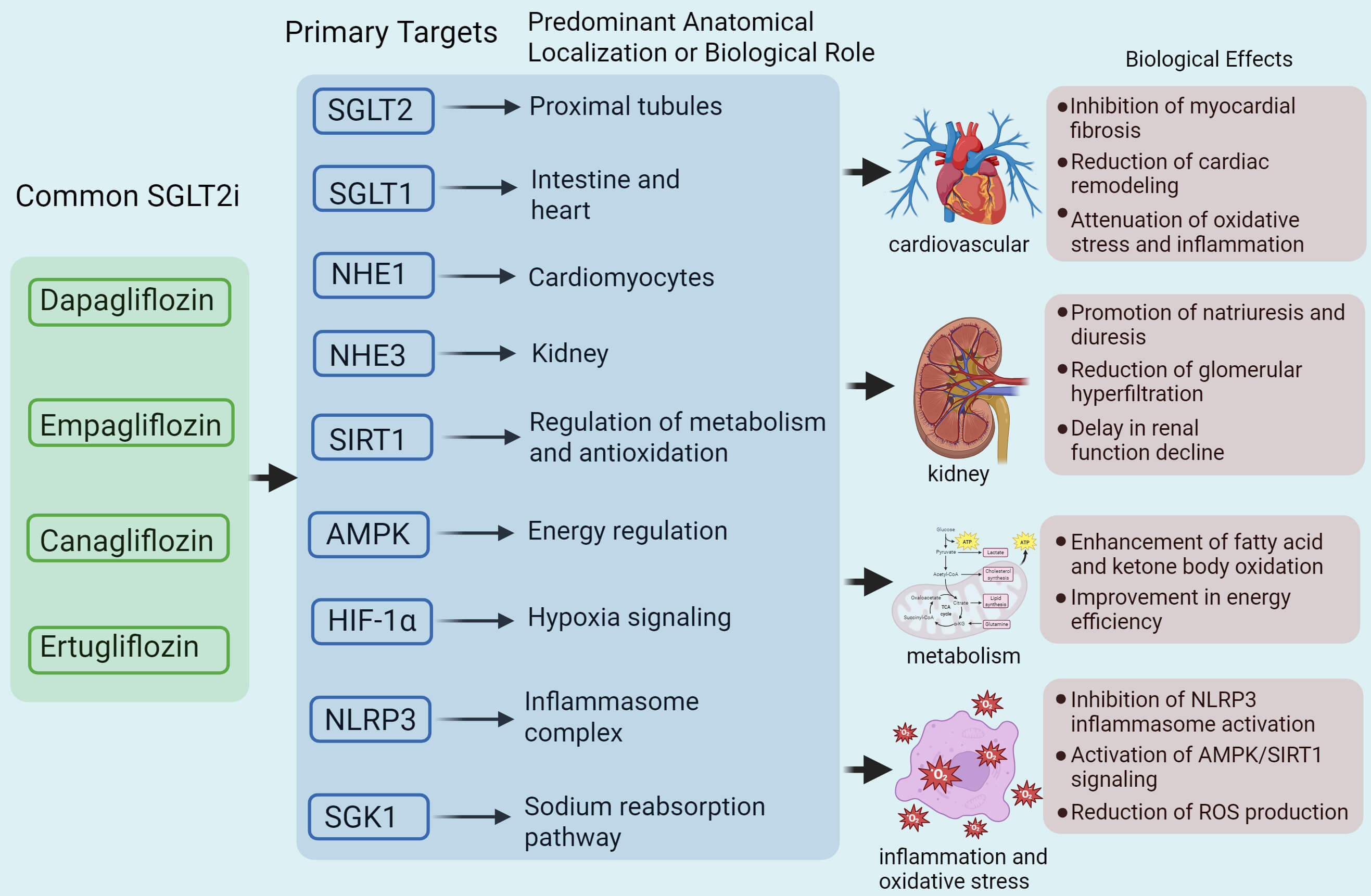 Fig. 2.
Fig. 2.
Mechanisms by which SGLT2is treat HF through different targets.
Dapagliflozin, empagliflozin, canagliflozin, and ertugliflozin exert various
functional effects by targeting multiple molecular pathways, including SGLT2,
sodium–glucose cotransporter 1 (SGLT1), sodium–hydrogen exchanger 1 (NHE1),
NHE3, sirtuin 1 (SIRT1), AMP-activated protein kinase (AMPK), hypoxia-inducible
factor 1-alpha (HIF-1
The reduction in preload is primarily achieved through diuretic and natriuretic effects, which decrease venous return [26]. Cardiac magnetic resonance imaging studies have demonstrated reduced end-diastolic volume following treatment with empagliflozin, consistent with decreased preload [27]. These changes collectively reduce cardiac workload, particularly in patients with HF. The DAPA-HF trial showed that dapagliflozin reduced the risk of the composite endpoint of cardiovascular death or worsening HF by 26% compared to placebo, suggesting that SGLT2is may directly enhance blood flow by optimizing cardiac loading conditions [28]. In addition, SGLT2is indirectly promote vasodilation by improving endothelial function, further contributing to improved hemodynamics [29]. While the primary mechanism involves renal-mediated volume regulation, emerging evidence also points to direct effects on cardiac cells, such as reductions in inflammation and oxidative stress [30].
Interstitial edema, characterized by fluid accumulation in tissue interstices, is common in HF patients [31]. SGLT2is mitigate interstitial edema by increasing urine output and sodium excretion, thereby reducing fluid retention [32]. Studies suggest that the diuretic effect of SGLT2is preferentially targets interstitial fluid rather than solely reducing plasma volume [33]. Mathematical modeling has shown that dapagliflozin has a lesser impact on plasma volume compared to the angiotensin-converting enzyme inhibitor (ACEI) enalapril, minimizing the risk of hypovolemia-induced reflex neurohumoral activation and electrolyte imbalances [34]. The RECEDE-CHF trial reported an increase in daily urine output of 545 mL with SGLT2is, of which 312 mL was electrolyte-free water clearance, contributing to the alleviation of fluid overload [35]. Although evidence on the long-term diuretic effects of SGLT2is remains inconsistent, these effects are considered beneficial in reducing pulmonary edema and improving congestive HF symptoms.
SGLT2is exert multifaceted regulatory effects on cardiac metabolism in HF, by coordinated modulation of glucose, energy, and lipid metabolism [36]. By inducing mild ketosis, SGLT2is influence glucose metabolism by promoting lipolysis and reducing glucose utilization, thereby enhancing ketone body production [37]. These ketone bodies serve as an efficient energy substrate for the heart, which is particularly beneficial in improving cardiac function in HF [38]. Studies suggest that SGLT2is reduce glucose uptake in cardiomyocytes by inhibiting glucose transporter 1 (GLUT1), which is beneficial in HF patients with impaired metabolic function [39].
In individuals with marked insulin resistance or poorly controlled type 2 diabetes, chronic suppression of glucose oxidation and elevated circulating free fatty acids create a highly inefficient metabolic environment [40]. Preclinical studies have shown that hearts from insulin-resistant models exhibit a greater shift toward ketone oxidation and mitochondrial coupling efficiency after SGLT2is exposure compared to non-diabetic controls [41]. Conversely, in non-diabetic or mildly insulin-resistant heart failure patients, baseline ketone oxidation is already relatively preserved, and the benefits of SGLT2is may derive more from systemic effects—such as improved renal—cardiac crosstalk, reduced inflammation, and enhanced autophagy—than solely from altering direct metabolic substrates [42]. Clinical metabolomic analyses from the DAPA-HF and EMPEROR studies have shown that although plasma ketone levels increase modestly in all patients, the correlation between increased ketones and clinical outcomes is strongest in those with higher baseline insulin resistance or hyperglycemia [43]. These findings support a model in which SGLT2is-mediated cardioprotection is multifactorial and context-dependent: the relative contribution of ketone metabolism predominates in metabolically impaired states, whereas hemodynamic and anti-inflammatory mechanisms may dominate in metabolically healthier individuals [42]. Recognizing this heterogeneity will be essential for tailoring therapeutic strategies and identifying patient subgroups that are most likely to benefit from SGLT2is-induced metabolic remodeling.
In terms of energy metabolism, SGLT2is enhances ATP production by promoting mitochondrial biogenesis and improving mitochondrial function [44]. They facilitate ketone body utilization, a metabolic pathway that is more efficient than fatty acid oxidation, thereby increasing the heart’s energy utilization efficiency [38]. Furthermore, SGLT2is appear to inhibit sodium–hydrogen exchanger 1 (NHE1), reducing intracellular sodium and calcium concentrations in cardiomyocytes [45]. This contributes to maintaining ion homeostasis and preventing calcium overload, which is critical for HF patients.
Regarding lipid metabolism, SGLT2is promote the oxidation of long-chain fatty acids, thereby reducing the accumulation of toxic lipid intermediates, a process particularly relevant in HF, where dysregulated lipid metabolism contributes to lipid accumulation and oxidative stress [46]. In addition, SGLT2is exert cardioprotective effects by modulating levels of lipid metabolism-related hormones, such as leptin and adiponectin [47].
Chronic inflammation is a hallmark of HF, and SGLT2is mitigate this inflammatory
state, thereby reducing myocardial injury [48]. Interleukin-1
Excessive activation of signal transducer and activator of transcription 3
(STAT3) is associated with myocardial hypertrophy and fibrosis, and its
inhibition mitigates pathological ventricular remodeling [52]. SGLT2is reduce
IL-6 levels, thereby inhibiting Janus kinase 2 (JAK2)/STAT3 phosphorylation and
blocking pro-inflammatory and pro-fibrotic signaling pathways [53]. SGLT2is
attenuate stimulation by interferon-
SGLT2is modulate immune responses by inhibiting the IL-6/STAT3 pathway, which reduces the generation of Th17 cells and promotes regulatory T cell (Treg) proliferation, thereby limiting excessive immune activation [57]. A decreased Th17/Treg ratio mitigates IL-17-mediated myocardial inflammation and fibrosis [58]. By alleviating hyperglycemia and oxidative stress, SGLT2is inhibit the release of neutrophil extracellular traps (NETs) by neutrophils, reducing microvascular embolism and inflammatory damage in the myocardium [59]. NETs are critical mediators of myocardial ischemia-reperfusion injury and HF progression, and their reduction helps protect the myocardial microcirculation [60].
Oxidative stress plays a pivotal role in the progression of HF, contributing to endothelial dysfunction, cardiac remodeling, and impaired contractility [25]. SGLT2is mitigate oxidative stress injury through multiple mechanisms. In mouse models of HF, empagliflozin significantly reduced cardiac ROS production while upregulating the expression of endogenous antioxidants [61]. SGLT2is may modulate intracellular sodium homeostasis by inhibiting NHE1 and the sodium channel Nav1.5, thereby attenuating oxidative stress [45].
Autophagy, a critical process for clearing damaged cellular components, particularly ROS-generating dysfunctional mitochondria, is enhanced by SGLT2is [62]. Studies indicate that SGLT2is promote autophagy by activating AMPK, SIRT1, and hypoxia-inducible factor pathways, facilitating the removal of these deleterious components [39]. Given the close interplay between oxidative stress and inflammation, the anti-inflammatory effects of SGLT2is may indirectly reduce oxidative stress [50]. By suppressing pro-inflammatory cytokines and modulating immune cell activity, SGLT2is mitigate oxidative damage due to inflammation [63].
SGLT2is reduce oxidative stress by altering myocardial energy substrate utilization, shifting from carbohydrates to more efficient substrates such as fatty acids and ketone bodies [37]. This metabolic shift enhances mitochondrial function, lowers ROS production, and further alleviates oxidative stress by improving insulin sensitivity and glucose control [9]. SGLT2is demonstrate significant reductions in oxidative stress in non-diabetic HF rat models, suggesting that their protective effects may be independent of glycemic control [64]. This indicates that SGLT2is exert broader cardioprotective effects through direct cardiac actions and metabolic regulation, beyond mechanisms associated with diabetes. Furthermore, SGLT2is enhance the antioxidant capacity of cardiomyocytes by activating antioxidant enzyme systems, providing additional protection against injury from oxidative stress [65].
SGLT2is exert their therapeutic effects in HF through modulation of multiple neuroendocrine systems. The DAPA-HF trial evaluated the efficacy of dapagliflozin in HF, while the EMPEROR-Reduced and EMPEROR-Preserved trials investigated empagliflozin in HF patients with varying ejection fractions [66, 67, 68]. These trials demonstrated that SGLT2is significantly reduce cardiovascular mortality and HF hospitalization rates, suggesting a potential role in improving cardiac function through neuroendocrine regulation [7].
SGLT2is promote urinary excretion, reducing plasma sodium levels, which in turn lowers renin and aldosterone levels, thereby suppressing the activity of the renin-angiotensin-aldosterone system (RAAS) [32]. Studies have shown that SGLT2is, such as empagliflozin, significantly decrease plasma aldosterone levels in HF patients, contributing to reduced fluid retention and vasoconstriction [69]. Excessive RAAS activation in HF exacerbates fluid retention and vascular constriction, and its inhibition helps alleviate these pathological processes [70].
In HF, increased activation of the sympathetic nervous system (SNS) leads to increases in heart rate, myocardial contractility, and vascular resistance. SGLT2is mitigate SNS activity by improving fluid balance and metabolic control [71]. Recent studies have reported decreased variability in heart rate and reduced resting heart rate following dapagliflozin treatment, indicating enhanced autonomic nervous system function, likely through decreased sympathetic activity, which reduces heart rate and cardiac workload [72].
Beyond their effects on traditional RAAS and SNS pathways, SGLT2is modulate additional hormones, such as brain natriuretic peptide (BNP) and glucagon-like peptide-1 (GLP-1), offering novel insights into their broad therapeutic benefits. SGLT2is may lower BNP levels, which is a reflection of reduced cardiac stress [73]. GLP-1, a hormone with cardioprotective properties, enhances myocardial contractility and reduces apoptosis [74]. Dapagliflozin has been shown to increase GLP-1 levels, potentially providing additional protection by improving cardiac contractile function [6].
Ventricular remodeling is a central pathological process in the progression of
HF, characterized by hypertrophy of cardiomyocytes, apoptosis, and interstitial
fibrosis, leading to ventricular dilation and contractile dysfunction [75].
SGLT2is reduce blood volume through osmotic diuresis (sodium and glucose
excretion), lowering preload, while also decreasing afterload, thereby reducing
cardiac workload [32]. This alleviation of cardiac stress decreases energy
demands, improves myocardial oxygen supply-demand balance, and suppresses
compensatory cardiomyocyte hypertrophy [72]. SGLT2is indirectly inhibit NHE1
activity in cardiomyocyte membranes, reducing intracellular sodium levels and
subsequently attenuating sodium–calcium exchanger (NCX)-mediated calcium
overload, which mitigates calcium-dependent contractile dysfunction and
mitochondrial oxidative stress [45]. Increased phosphorylation of AMPK suppresses
excessive mammalian target of rapamycin (mTOR) activation, restoring the balance
between autophagy and apoptosis, thereby reducing cardiomyocyte apoptosis and
necrosis [76]. SGLT2is induce mild ketosis (elevated
Myocardial fibrosis, a key contributor to increased ventricular stiffness and
diastolic dysfunction, is modulated by SGLT2is through multiple mechanisms.
SGLT2is downregulate transforming growth factor-beta (TGF-
The EMPEROR-Reduced trial demonstrated that SGLT2is significantly reduced left ventricular mass index (LVMI), which is indicative of the reversal of myocardial hypertrophy [8]. SGLT2is also limit ventricular dilation (reducing left ventricular end-systolic volume index (LVESVi)), restore sphericity index, and optimize contractile synchrony [27]. By reducing myocardial stiffness and the E/e′ ratio, SGLT2is improve left ventricular filling pressure, alleviating diastolic dysfunction [86]. The DAPA-HF and EMPEROR-Reduced trials confirmed that dapagliflozin and empagliflozin significantly lowered the risk of cardiovascular death and HFrEF, with subgroup analyses revealing improvements in left ventricular ejection fraction (LVEF) and reductions in N-terminal pro-B-type natriuretic peptide (NT-proBNP) [7, 87]. Cardiac magnetic resonance imaging studies have also shown that SGLT2is-treated groups exhibit reduced markers of myocardial fibrosis, such as ECV, and decreased myocardial fibrosis, and showed evidence of reverse ventricular remodeling [88].
SGLT2is have demonstrated significant efficacy and broad therapeutic potential
in the treatment of HFrEF, HFpEF, and HFmrEF [28]. Several clinical trials have
confirmed that SGLT2is significantly reduce the risk of cardiovascular death and
HF hospitalization across a broad range of ejection fractions [7] (Table 1, Ref.
[7, 8, 10, 23, 35, 87, 89, 90, 91]). The DAPA-HF trial showed that dapagliflozin reduced
the risk of cardiovascular death or HF hospitalization by 26% and all-cause
mortality by 17% in HFrEF patients [87]. Similarly, the EMPEROR-Reduced trial
demonstrated that empagliflozin reduced the risk of cardiovascular death or HF
hospitalization by 25% in HFrEF patients, with benefits that were independent of
the patient’s diabetes status [10]. The EMPEROR-Preserved trial was the first to
show that empagliflozin reduced the risk of cardiovascular death or HF
hospitalization by 21% in HFpEF patients, while the DELIVER trial reported an
18% reduction in the risk of the primary endpoint in HFpEF patients with an LVEF
| Trial name | HF classification | Intervention regimen | Key subgroup analysis findings | References |
| EMPEROR-Reduced | HFrEF | Empagliflozin vs. Placebo | Subgroup aged |
[7, 10] |
| EMPEROR-Preserved | HFpEF | Empagliflozin vs. Placebo | Consistent benefits across LVEF 50%–59% and LVEF |
[8, 89] |
| DEFINE-HF Trial | HFrEF | Dapagliflozin vs. Placebo | No significant difference in RRR between T2DM patients and non-T2DM patients; Benefit retained in eGFR 25–45 mL/min/1.73 m2 subgroup | [23] |
| RECEDE-CHF | HFrEF/HFpEF | Dapagliflozin vs. Placebo | No significant subgroup differences; No increased risk of acute kidney injury observed | [35] |
| DELIVER | HFpEF/HFmrEF | Dapagliflozin vs. Placebo | Patients with atrial fibrillation, no difference from those without atrial fibrillation | [89, 90] |
| DAPA-HF | HFrEF/HFpEF/HFmrEF | SGLT2is Group vs. Conventional Therapy Group (RAASis + |
Subgroup aged |
[87, 91] |
HFrEF, HF with reduced ejection fraction; HFpEF, HF with preserved ejection fraction; HFmrEF, HF with mid-range ejection fraction; LVEF, left ventricular ejection fraction; T2DM, type 2 diabetes mellitus; eGFR, estimated glomerular filtration rate; RAASis, Renin–Angiotensin–Aldosterone System inhibitors; MRAs, Mineralocorticoid Receptor Antagonists; RRR, Relative Risk Reduction.
SGLT2is also exhibit significant advantages in special high-risk populations, such as those with comorbid chronic kidney disease (CKD). Both dapagliflozin and empagliflozin significantly reduce the risk of cardiovascular death or HF hospitalization while slowing the progression of CKD [93]. Although SGLT2is are generally considered safe and well-tolerated, it is important to highlight the risk of euglycemic diabetic ketoacidosis (eDKA), a rare but potentially life-threatening complication. However, emerging evidence suggests that this condition can occasionally occur in non-diabetic heart failure patients receiving SGLT2is, particularly during catabolic or increased stress conditions such as prolonged fasting, infection, perioperative periods, or excessive alcohol consumption [94]. In patients with advanced CKD, the risk–benefit balance requires particular attention. Reduced renal clearance may alter the handling of glucose and ketone, and CKD-related malnutrition or infection could predispose patients to ketosis under stress [95]. Therefore, clinicians should educate patients and caregivers to recognize early symptoms of ketoacidosis (nausea, vomiting, abdominal pain, dyspnea) and advise temporary discontinuation of SGLT2is during acute illness, surgery, or reduced oral intake [96]. In patients with acute HF or worsening HF, SGLT2is, when used in combination therapy, increase urine output without compromising renal function [97]. SGLT2is also demonstrate synergistic effects when combined with conventional HF medications, further optimizing therapeutic outcomes [98].
The safety profile of SGLT2is in HF treatment has been extensively validated. Multiple large-scale randomized controlled trials (RCTs) and meta-analyses have confirmed that SGLT2is significantly reduce the risk of cardiovascular death and hospitalization across various HF phenotypes without introducing additional safety concerns [90, 99, 100]. Pooled analyses from the DAPA-HF and EMPEROR-Reduced trials demonstrated that dapagliflozin and empagliflozin significantly lowered mortality across the full range of ejection fractions, with adverse event rates comparable to placebo [7]. Real-world studies, such as the CVD-REAL study, further corroborate the safety of SGLT2is in HF management, showing high patient adherence and tolerability with low rates of adverse events [101].
Despite the significant advancements achieved with SGLT2is in the management of
HF, further in-depth studies are needed to elucidate their molecular mechanisms
and cellular signaling pathways to develop more effective therapeutic strategies
and optimize drug combinations. Future research should prioritize large-scale,
long-term RCTs to rigorously evaluate the efficacy and safety of SGLT2is across
diverse patient populations, with a particular emphasis on HFpEF and specific
subgroups. These subgroups include, but are not limited to, patients with LVEF
Optimizing the combination of SGLT2is with other HF therapies, including RAAS
inhibitors,
Personalized treatment strategies tailored to individual patient characteristics—such as comorbidities (e.g., T2DM, CKD, or coronary artery disease), renal function (e.g., eGFR levels), baseline LVEF, and frailty status—are essential to enhance treatment efficacy and safety. Natriuretic peptides (BNP, NT-proBNP) are well-established indicators of ventricular stress and may identify patients most likely to experience rapid hemodynamic improvement with SGLT2is [106]. Early reductions in NT-proBNP after initiation have been correlated with improved outcomes in both HFrEF and HFpEF populations [107]. Estimated glomerular filtration rate (eGFR) and albuminuria are key determinants of both efficacy and tolerability [108]. Clinical data suggest that SGLT2is retain cardiorenal protective effects as low as eGFR thresholds of 20–25 mL/min/1.73 m2, but initial eGFR decline and long-term slope stabilization vary by baseline renal status [109]. Parameters such as HbA1c, fasting ketone levels, and high-sensitivity C-reactive protein (hsCRP) may provide additional insights into metabolic adaptation and residual inflammatory risk during therapy [110].
A major paradigm shift introduced by large-scale randomized trials was the observation that the cardiovascular and renal benefits of SGLT2is extend beyond glucose lowering. Subgroup analyses from pivotal trials—including DAPA-HF, EMPEROR-Reduced, and EMPEROR-Preserved—have consistently shown that the relative risk reduction in heart failure hospitalization and cardiovascular death with SGLT2is therapy is comparable in patients with and without T2DM [76]. In trial DAPA-HF trial, for example, dapagliflozin reduced the composite outcome of worsening HF or cardiovascular death by 26% in patients without T2DM, closely matching the 25% reduction observed in those with T2DM [106]. Similar findings were reported in the EMPEROR-Reduced and DELIVER trials, underscoring that SGLT2is confer cardioprotective effects independent of glycemic status [103, 109]. However, subtle distinctions do exist between diabetic and non-diabetic HF populations. Patients with T2DM tend to have a higher baseline risk of renal dysfunction, volume overload, and microvascular disease, which may increase the absolute risk reduction observed with SGLT2is [107]. Conversely, non-diabetic HF patients may exhibit greater improvements in metabolic efficiency and myocardial energetics, albeit with slightly increased vigilance required for the development of volume depletion or euglycemic ketoacidosis [96]. Long-term safety monitoring of SGLT2is in non-diabetic HF patients is particularly important, especially for potential risks such as diabetic ketoacidosis, urogenital infections, and volume depletion [18]. Expanded real-world evidence studies and post-marketing surveillance are needed to assess these risks over extended periods and in varied clinical contexts. Furthermore, future research should clarify the optimal timing and sequencing of SGLT2is initiation relative to other HF therapies, particularly in patients with acute HF or recent decompensation [100]. Through these comprehensive research efforts, SGLT2is have the potential to further transform the landscape of HF management, offering improved prognosis and quality of life for a broader patient population.
SGLT2is exhibit remarkable efficacy and considerable potential in the management of HF. These agents exert their therapeutic effects through multiple mechanisms, including optimization of hemodynamics, regulation of cardiac metabolism, attenuation of inflammatory responses and oxidative stress, modulation of neuroendocrine pathways, and suppression of myocardial fibrosis. SGLT2is demonstrate robust performance in special populations and when used in combination with other HF therapies. Their safety has been consistently validated across numerous studies, with low rates of adverse events. Future research should prioritize elucidating the molecular mechanisms of SGLT2is, conducting additional large-scale clinical trials, exploring optimal combination therapies, developing personalized treatment strategies, and monitoring long-term safety in non-diabetic HF patients. With these advancements, SGLT2is are poised to play an increasingly pivotal role in the treatment of HF, thereby enhancing patient prognosis and quality of life.
ZH, GL, CF, ZW, SY, CL and BZ were responsible for the conception and design of the study, data analysis, and drafting of the manuscript. ZH, CZ and GL participated in data acquisition and interpretation. ZH, CL, and BZ contributed to the critical revision of the manuscript for important intellectual content. CL and BZ supervised the overall project, provided guidance throughout the study, and were responsible for the final approval of the version to be published. All authors have read and agreed to the published version of the manuscript. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research was funded by grants from the National Natural Science Foundation of China (Grant No.82360066) and the Natural Science Foundation of Guangxi (Grant No.2025GXNSFAA069056).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


