1 The First Clinical Medical College, Lanzhou University, 730000 Lanzhou, Gansu, China
2 Department of Cardiology, The First Hospital of Lanzhou University, 730000 Lanzhou, Gansu, China
3 Gansu Provincial Clinical Research Center for Cardiovascular Diseases, 730000 Lanzhou, Gansu, China
Abstract
Residual inflammation and persistent lipid abnormalities substantially increase the risk of adverse clinical outcomes in patients with acute myocardial infarction (AMI) undergoing primary percutaneous coronary intervention (PPCI). Although proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors have been shown to improve cardiovascular outcomes, the efficacy, safety, and prognosis of these inhibitors when administered as a single dose after PPCI in real-world practice remain unclear.
A retrospective study of patients with acute ST-segment elevation myocardial infarction (STEMI) admitted between May 2023 and May 2024. Patients were assigned to an alirocumab group or a conventional treatment group based on whether a single dose of alirocumab was administered within 6 hours of PPCI. Baseline differences between groups were balanced using 1:1 propensity score matching (PSM). The occurrence of major adverse cardiovascular events (MACEs) at 12 months post-procedure was applied as the primary endpoint. Secondary endpoints included lipid profiles, inflammatory markers, cardiac function, quality-of-life changes, and safety outcomes.
A non-significant downward trend in the incidence of MACEs at 12 months post-PPCI was observed in the alirocumab group compared with the conventional treatment group (log-rank p = 0.242). A single dose of alirocumab significantly reduced low-density lipoprotein cholesterol (LDL-C) at 1 month (p = 0.011) and attenuated inflammation markers at 24 hours postoperatively. At 12 months, the alirocumab group showed improved cardiac function with significantly reduced left ventricular end-systolic volume (LVESV, p = 0.009) and modest but statistically significant improvement in quality of life (p = 0.012), primarily driven by enhanced physical activity (p < 0.001), alongside reduced insecurity (p < 0.001). No increased incidence of adverse events was observed (p > 0.05).
This study demonstrated that a single dose of alirocumab in STEMI patients undergoing PPCI was associated with significant improvement in LDL-C levels, attenuation of early postoperative inflammation, and a favorable trend toward improved cardiac function and quality of life, while maintaining an acceptable safety profile.
Keywords
- alirocumab
- percutaneous coronary intervention
- myocardial infarction
Acute myocardial infarction (AMI) is a leading cause of death and disability worldwide. The global epidemiologic burden of AMI continues to rise. According to the World Health Organization, the annual incidence of AMI is approximately 50–200 cases per 100,000 people, and approximately 30% of patients face the risk of recurrent cardiovascular events or death within one year of onset [1]. Although primary percutaneous coronary intervention (PPCI) has significantly improved revascularization efficiency among patients with AMI, adverse outcomes including left ventricular remodeling, reinfarction, and heart failure still occur in approximately 15%–20% of patients due to residual inflammation, oxidative stress, and lipid metabolism disorders [2]. Consequently, optimization of perioperative pharmacologic strategies to further improve long-term outcomes has become an important focus of research in cardiovascular medicine.
In recent years, the role of proprotein convertase subtilisin/kexin type 9 (PCSK9) in atherosclerotic cardiovascular disease has garnered substantial attention [3]. Basic research has demonstrated that PCSK9 not only modulates lipid metabolism through promoting low-density lipoprotein receptor (LDLR) degradation but also directly participates in pathological processes, including macrophage activation, plaque destabilization, and cardiomyocyte apoptosis following AMI [4]. PCSK9 inhibitors, represented by evolocumab and alirocumab, have been proven in multiple randomized controlled trials to significantly reduce the incidence of major adverse cardiovascular events (MACE) by effectively lowering low-density lipoprotein cholesterol (LDL-C) levels by approximately 50%–62% and potentially exerting anti-inflammatory effects [5, 6]. The American College of Cardiology/American Heart Association (ACC/AHA) guidelines recommend that patients who have recently experienced acute coronary syndrome (ACS) should first be treated with ezetimibe (Class I recommendation) in addition to intensive statin therapy. If LDL-C levels are still not within the target range, PCSK9 inhibitors may be considered as an additional treatment (Class IIa recommendation) [2].
However, unique clinical challenges exist during the acute phase of ACS,
including significant fluctuations in patients’ baseline LDL-C levels caused by
stress-induced metabolic changes upon admission, and a delayed maximal
lipid-lowering effect of statins, which requires 4–6 weeks of continuous
treatment to become apparent. This makes it challenging to accurately evaluate
the feasibility of a stepwise lipid-lowering regimen in the early post-PPCI
period, and delaying initiation of PCSK9 inhibitors may result in missing the
critical window of opportunity to intervene in inflammation and disordered lipid
metabolism. Therefore, whether PCSK9 inhibitor therapy should be initiated during
the early stages of ACS has become a subject of considerable interest [7].
Several studies published by the European Society of Cardiology (ESC) in 2023
have demonstrated that initiating a combined lipid-lowering strategy involving
PCSK9 inhibitors immediately after ACS onset, during hospitalization, or at
discharge can rapidly and markedly reduce LDL-C levels, significantly enhancing
LDL-C goal achievement rates up to 98% (LDL-C
This study was designed as a single-center, retrospective, observational cohort study in which clinical data from patients diagnosed with STEMI, admitted to the Cardiology Center of Lanzhou University First Hospital from May 2023 to May 2024, were collected. Throughout the data collection process, the guidelines for strengthening the reporting of observational studies in epidemiology (STROBE) were strictly followed [11]. Patients were assigned to either an alirocumab group (n = 96) or a conventional treatment group (n = 96) based on the administration of a single 75 mg subcutaneous dose of alirocumab within 6 hours following PPCI. This dose was chosen in accordance with the standard clinical starting regimen and pharmacokinetic data showing that a single 75 mg dose produces clinically meaningful LDL-C reductions, while providing a conservative and feasible option for an exploratory single-dose strategy in the acute STEMI setting. Standard guideline-directed medical therapy according to the 2023 ESC guidelines was administered to both groups, including aspirin (loading dose 300 mg, maintenance dose 100 mg/day), ticagrelor (loading dose 180 mg, maintenance dose 90 mg bid), or clopidogrel (loading dose 300 mg, maintenance dose 75 mg/day), and anticoagulant therapy as needed. All patients were initiated on moderate-intensity statin therapy during the index hospitalization (atorvastatin 20 mg/day or rosuvastatin 10 mg/day), and longitudinal prescription records from the electronic medical and pharmacy systems confirmed that all patients in both groups maintained continuous moderate-intensity statin therapy from discharge through 12 months, without ezetimibe co-therapy and without any additional PCSK9 inhibitor use during follow-up. This retrospective cohort study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. Ethical approval was obtained from the Ethics Committee of the First Hospital of Lanzhou University, and the requirement for informed consent was waived.
Patients aged between 18 and 75 years;
Meets the ESC guidelines for STEMI diagnosis (typical chest pain lasting
First episode with concomitant PPCI, and post-procedural culprit vessel thrombolysis in myocardial infarction (TIMI) flow grade 3;
Baseline LDL-C
Previous use of PCSK9 inhibitors or known allergies to monoclonal antibodies;
Concurrent structural heart disease;
Severe liver dysfunction: alanine aminotransferase (ALT) or aspartate
aminotransferase (AST)
Patients with active infections, autoimmune diseases, malignant tumors, or psychiatric disorders;
Pregnant or breastfeeding women;
Incomplete clinical data;
Patients who were discharged without authorization.
The primary endpoint was a composite of MACE at 12 months post-PPCI, including cardiac death (defined as death due to sudden cardiac death, heart failure, or reinfarction), non-fatal reinfarction, unplanned repeat revascularization, malignant arrhythmia, and readmission for heart failure.
Lipid metabolism markers: LDL-C and total cholesterol (TC) levels at 1, 6, and
12 months post-PCI; Inflammation markers: Neutrophil/lymphocyte ratio (NLR) and
C-reactive protein (CRP) at 24 hours post-PPCI; Cardiac function: Cardiac injury
markers troponin I (cTNI) and N-terminal pro-B-type natriuretic peptide
(NT-proBNP) levels at 6 and 24 hours postoperatively; left ventricular ejection
fraction (LVEF), left ventricular end-systolic volume (LVESV), and left
ventricular end-diastolic volume (LVEDV) at 6 months and 1 year postoperatively;
Health-related quality of life was assessed using the Myocardial Infarction
Dimensional Assessment Scale (MIDAS) at hospital discharge (baseline) and at 12
months. At discharge, once patients met clinical stability and discharge
criteria, the MIDAS questionnaire was self-completed by patients in a quiet area
of the cardiology ward under the supervision of trained cardiology nurses or
research staff who were not involved in clinical decision-making. Standardized
written and verbal instructions were provided, emphasizing that the items
referred to the preceding week, that responses were scored on a 0–4 Likert scale
(0 = ‘never’ to 4 = ‘always’), and that there were no right or wrong answers.
Staff were available to clarify item wording without suggesting responses,
ensuring a uniform administration procedure. Baseline assessments were therefore
obtained in clinically stable patients at the time of discharge, reflecting early
recovery health status rather than the immediate acute phase. The Scale is a
35-item, myocardial infarction (MI)-specific health status questionnaire with seven domains: physical
activity (12 items), insecurity (sense of safety, 9 items), emotional reaction (4
items), dependency (3 items), diet (3 items), concerns about medication (2
items), and side effects (2 items). Each item is rated from 0 (“never”
impaired) to 4 (“always” impaired), yielding a total score range of 0–140
points, where higher scores denote worse post-MI health status. This scale has
been validated in MI patients, with good internal consistency (Cronbach’s
Drug-related adverse events were defined as injection-site reactions (erythema, swelling, persistent pain), upper respiratory tract symptoms (sore throat, runny nose, sneezing), skin itching, hypersensitivity reactions, and muscle pain.
Outpatient follow-up was scheduled at 1, 3, 6, and 12 months after PPCI and included collection of medical history, laboratory testing, and echocardiography. Patients who did not attend outpatient follow-up visits were contacted by telephone to ascertain endpoint events and other prespecified outcomes. As summarized in Supplementary Fig. 1, the treatment pathway and dose exposure were identical in both groups except for a single 75 mg alirocumab injection within 6 hours after PPCI in the alirocumab group, on top of continuous moderate-intensity statin therapy without additional lipid-lowering drugs, including ezetimibe or further PCSK9 inhibitor use during the 12-month follow-up.
PSM was performed using a multivariable logistic regression model to estimate
propensity scores. A comprehensive set of covariates, all measured before the
administration of alirocumab, was included in the model, encompassing demographic
characteristics (age, male), clinical characteristics (Killip grade
According to the inclusion and exclusion criteria, 396 patients were initially enrolled, among whom 9 (2.3%) were excluded for nonadherence to discharge medication instructions, 37 (9.3%) were excluded for failure to regularly attend outpatient follow-up visits, and 9 (2.3%) were lost to follow-up (including 5 who lost contact and 4 who declined to participate). After exclusion of these patients, the baseline data of the remaining patients underwent PSM, resulting in a final cohort of 192 matched patients (Fig. 1). Before propensity score matching, there were significant differences between the alirocumab group and the conventional treatment group in terms of age, hypertension, TC, LDL-C, CRP, cTNI, NT-proBNP, LVEDV, and ticagrelor use. After matching, no significant differences were present between the groups for any covariate. The quality of baseline data matching was good (Table 1, Supplementary Fig. 2).
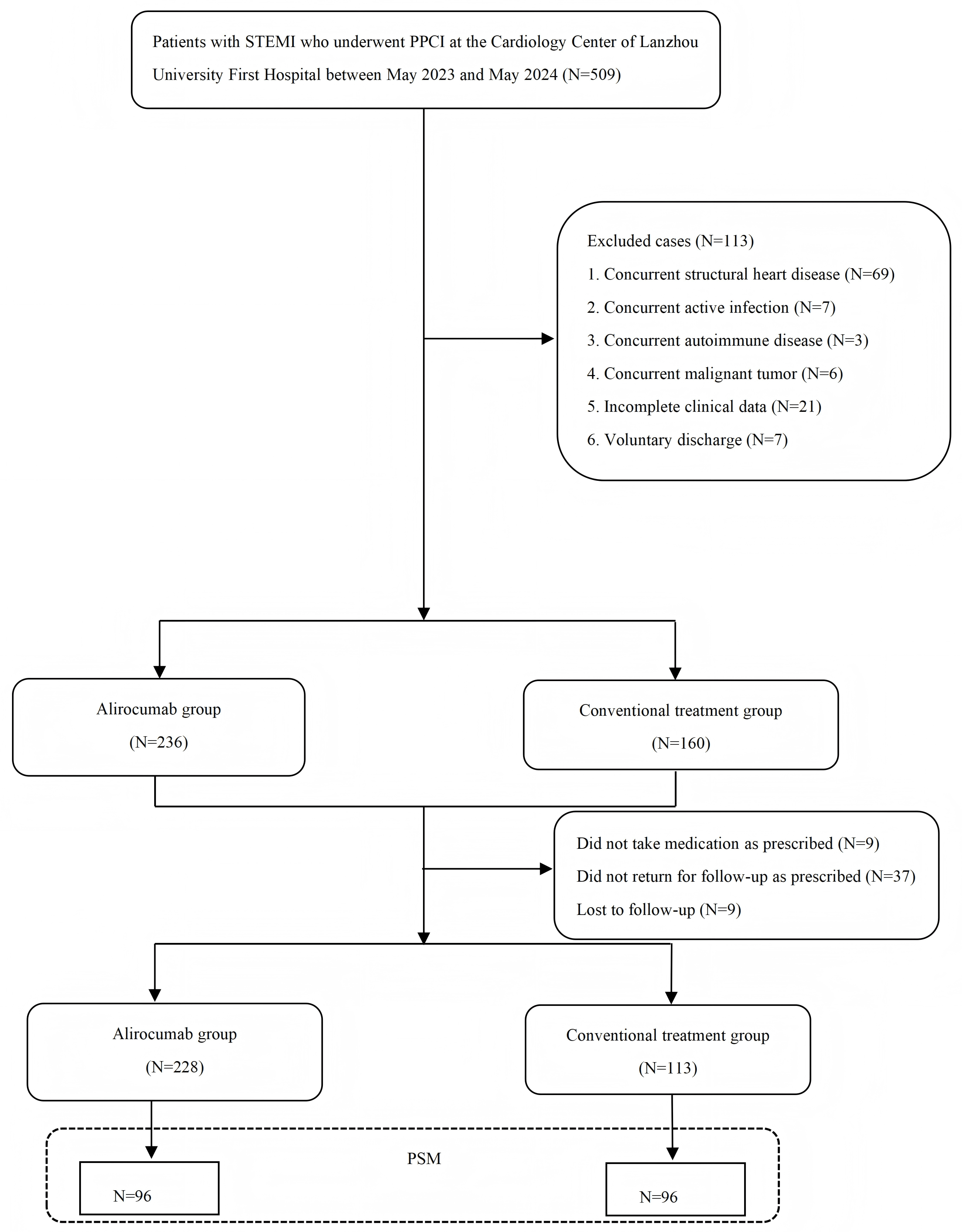 Fig. 1.
Fig. 1.
Flowchart of case screening. STEMI, ST-segment elevation myocardial infarction; PPCI, primary percutaneous coronary intervention; PSM, propensity score matching.
| Variable | Before PSM | After PSM | ||||||||
| Alirocumab (n = 228) | Conventional (n = 113) | SMD | p value | Alirocumab (n = 96) | Conventional (n = 96) | SMD | p value | |||
| Demographic characteristics | ||||||||||
| Age, year | 58.3 |
61.7 |
–0.31 | 0.007 | 59.1 |
60.3 |
–0.09 | 0.419 | ||
| Male, n (%) | 164.0 (71.9%) | 82.0 (72.6%) | 0.01 | 0.903 | 70.0 (72.9%) | 69.0 (71.9%) | 0.02 | 0.872 | ||
| Clinical characteristic, n (%) | ||||||||||
| Killip grade |
35.0 (15.4%) | 21.0 (18.6%) | 0.09 | 0.449 | 16.0 (16.7%) | 13.0 (13.5%) | 0.09 | 0.547 | ||
| Hypertension | 160.0 (70.2%) | 57.0 (50.4%) | 0.41 | 58.0 (60.4%) | 50.0 (52.1%) | 0.08 | 0.246 | |||
| Diabetes | 82.0 (36.0%) | 43.0 (38.1%) | 0.04 | 0.707 | 21.0 (21.9%) | 12.0 (12.5%) | 0.08 | 0.086 | ||
| Smoking | 115.0 (50.4%) | 62.0 (54.9%) | 0.09 | 0.442 | 58.0 (60.4%) | 56.0 (58.3%) | 0.04 | 0.769 | ||
| Laboratory indexes | ||||||||||
| TC, mmol/L | 4.1 |
4.7 |
–0.64 | 4.0 |
4.2 |
–0.09 | 0.193 | |||
| TG, mmol/L | 1.8 |
1.9 |
–0.14 | 0.172 | 1.7 |
1.8 |
–0.06 | 0.264 | ||
| LDL-C, mmol/L | 2.7 |
2.9 |
–0.28 | 0.007 | 2.7 |
2.8 |
–0.07 | 0.328 | ||
| NLR | 5.1 (3.8–9.5) | 5.5 (3.2–11.1) | –0.41 | 0.402 | 4.8 (3.9–7.6) | 5.4 (3.2–8.9) | –0.08 | 0.444 | ||
| CRP, mg/L | 12.5 (2.9–39.5) | 8.1 (1.9–20.2) | 0.61 | 0.003 | 10.0 (2.9–18.5) | 12.6 (2.4–19.8) | –0.08 | 0.531 | ||
| cTNI, µg/L | 3.2 (0.2–15.0) | 1.50 (0.3–6.8) | 0.32 | 0.003 | 2.5 (0.3–6.1) | 1.9 (0.4–5.8) | 0.07 | 0.661 | ||
| NT-proBNP, pg/mL | 1250.3 (580.2–5930.4) | 580.8 (198.1–1600.4) | 0.65 | 1290.4 (700.4–1940.4) | 1080.3 (630.1–1590.2) | 0.09 | 0.083 | |||
| Glucose, mmol/L | 7.8 |
8.0 |
–0.10 | 0.423 | 7.7 |
7.9 |
–0.09 | 0.478 | ||
| HbA1c, % | 6.8 |
6.9 |
–0.09 | 0.482 | 6.7 |
6.8 |
–0.07 | 0.548 | ||
| ALT, U/L | 32.4 (18.1–50.4) | 35.2 (20.2–55.1) | –0.19 | 0.241 | 33.1 (17.4–49.1) | 34.2 (19.3–53.4) | –0.07 | 0.517 | ||
| AST, U/L | 45.5 (25.6–67.2) | 48.3 (28.1–72.2) | –0.21 | 0.320 | 46.4 (24.1–66.3) | 47.0 (26.3–70.4) | –0.06 | 0.617 | ||
| Cr, µmol/L | 78.0 |
82.0 |
–0.16 | 0.116 | 79.0 |
80.1 |
–0.03 | 0.742 | ||
| Imaging characteristics | ||||||||||
| LVEF, % | 48.3 |
46.9 |
0.17 | 0.147 | 47.6 |
47.8 |
–0.02 | 0.828 | ||
| LVESV, mL | 65.2 |
68.1 |
–0.16 | 0.164 | 60.0 |
56.2 |
0.08 | 0.096 | ||
| LVEDV, mL | 125.0 |
130.4 |
–0.24 | 0.010 | 117.1 |
121.1 |
–0.09 | 0.269 | ||
| Lesion characteristics | ||||||||||
| Area of infarction, % | 0.440 | 0.860 | ||||||||
| Anterior | 115.0 (50.4%) | 45.0 (39.8%) | 0.22 | 48.0 (50.0%) | 46.0 (47.9%) | 0.06 | ||||
| Posterior | 25.0 (11.0%) | 15.0 (13.3%) | 0.07 | 10.0 (10.4%) | 7.0 (7.3%) | 0.07 | ||||
| Lateral | 28.0 (12.3%) | 15.0 (13.3%) | 0.03 | 12.0 (12.5%) | 16.0 (16.7%) | –0.09 | ||||
| Inferior | 42.0 (18.4%) | 28.0 (24.8%) | 0.16 | 18.0 (18.8%) | 20.0 (20.8%) | –0.08 | ||||
| Multifocal | 18.0 (7.9%) | 10.0 (8.8%) | 0.04 | 8.0 (8.3%) | 7.0 (7.3%) | 0.04 | ||||
| PCI-related indicators | ||||||||||
| Preoperative TIMI flow Grade |
85.0 (37.3%) | 38.0 (33.6%) | 0.08 | 0.510 | 34.0 (35.4%) | 28.0 (29.2%) | 0.09 | 0.356 | ||
| Post-PCI reflux abnormality | ||||||||||
| Slow blood flow, % | 18.0 (7.9%) | 10.0 (8.8%) | 0.04 | 0.763 | 8.0 (8.3%) | 5.0 (7.3%) | 0.08 | 0.390 | ||
| No-reflow, % | 7.0 (3.1%) | 5.0 (4.4%) | 0.07 | 0.523 | 3.0 (3.1%) | 3.0 (3.1%) | 0.00 | |||
| S2PCI time, h | 6.1 (2.2–15.1) | 6.0 (2.3–12.2) | 0.16 | 0.337 | 6.2 (2.1–15.4) | 6.2 (2.4–10.1) | 0.09 | 0.156 | ||
| Postoperative medication, % | ||||||||||
| Ticagrelor | 180.0 (78.9%) | 75.0 (66.4%) | 0.28 | 0.012 | 78.0 (81.3%) | 70.0 (72.9%) | 0.08 | 0.171 | ||
| Atorvastatin | 160.0 (70.2%) | 70.0 (61.9%) | 0.18 | 0.128 | 78.0 (81.3%) | 80.0 (83.3%) | –0.06 | 0.706 | ||
| Dapagliflozin | 45.0 (19.7%) | 18.0 (15.9%) | 0.10 | 0.395 | 19.0 (19.8%) | 18.0 (18.8%) | 0.03 | 0.855 | ||
| RAAS inhibitors | 90.0 (39.5%) | 50.0 (44.2%) | 0.10 | 0.400 | 38.0 (39.6%) | 40.0 (41.7%) | –0.04 | 0.769 | ||
| ARNI | 28.0 (12.3%) | 12.0 (10.6%) | 0.05 | 0.654 | 11.0 (11.5%) | 6.0 (6.3%) | 0.07 | 0.205 | ||
| 210.0 (92.1%) | 102.0 (90.3%) | 0.07 | 0.567 | 89.0 (92.7%) | 88.0 (91.7%) | 0.04 | 0.789 | |||
SMD, standardized mean differences; TG, triglyceride; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; NLR, neutrophil/lymphocyte ratio; CRP, c-reactive protein; cTNI, cardiac injury markers troponin I; NT-proBNP, n-terminal pro-B-type natriuretic peptide; HbA1c, glycated hemoglobin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; Cr, Creatinine; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; LVEDV, left ventricular end-diastolic volume; PCI, percutaneous coronary intervention; TIMI, thrombolysis in myocardial infarction; S2PCI, onset to PCI; ARNI, angiotensin receptor-neprilysin inhibitor; RAAS inhibitors, angiotensin receptor blockers or Angiotensin converting enzyme inhibitor.
During the follow-up period, a total of 21 patients experienced MACE (overall
incidence, 10.9%). Kaplan-Meier analysis showed a gradual increase in the
cumulative incidence of MACE in both groups, with numerically lower event rates
in the alirocumab group compared with the conventional treatment group from
approximately day 5 post-PCI; however, this difference did not reach statistical
significance (log-rank p = 0.242) (Fig. 2). In Cox regression analysis,
the incidence of MACE was 8.3% in the alirocumab group and 13.5% in the
conventional treatment group, again without a statistically significant
between-group difference (p = 0.250). The individual components of
MACE—cardiac death (2.1% vs. 1.0%), non-fatal myocardial infarction (2.1%
vs. 3.1%), unplanned repeat revascularization (2.1% vs. 4.2%), malignant
arrhythmia (1.0% vs. 3.1%), and readmission for heart failure (1.0% vs.
2.1%)—also did not differ significantly between the two groups (all p
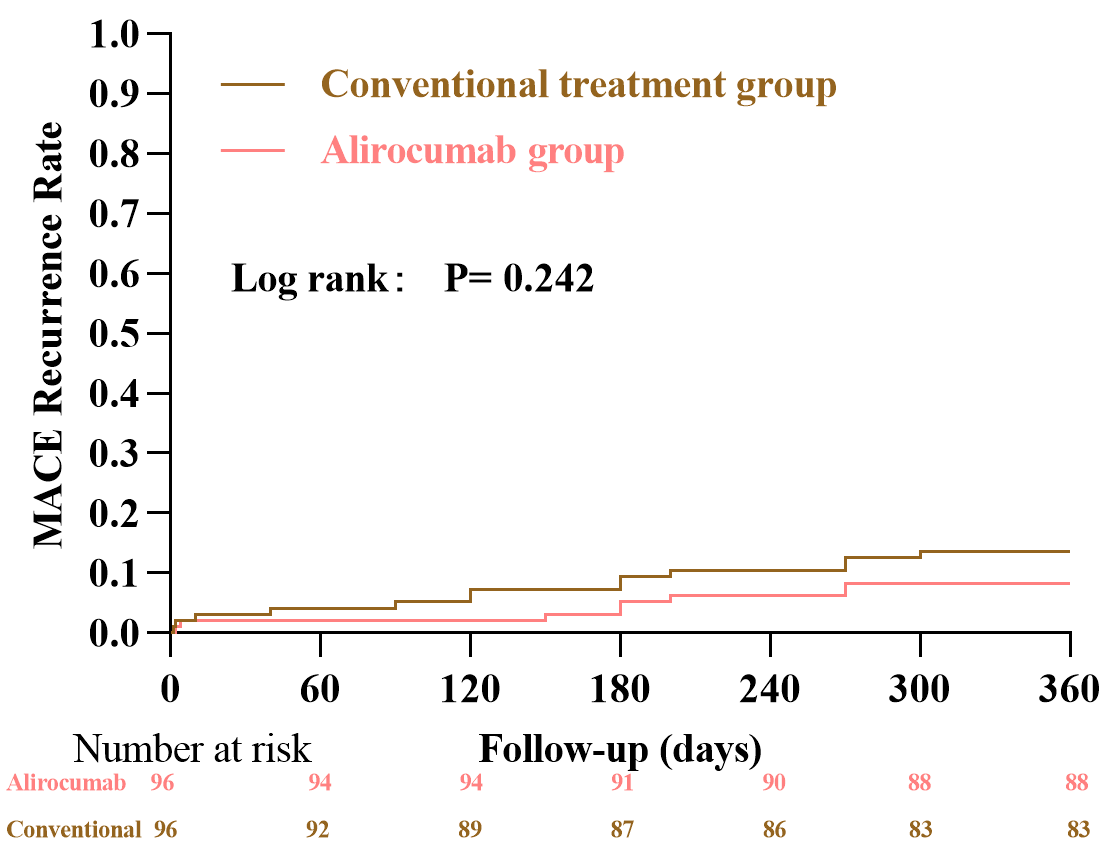 Fig. 2.
Fig. 2.
Cumulative incidence of MACE. MACE, major adverse cardiovascular events.
| Endpoint | Alirocumab group | Conventional treatment group | Adjusted hazard ratio (95% CI) | p value |
| MACE | 8.0 (8.3) | 13.0 (13.5) | 0.606 (0.258–1.423) | 0.250 |
| Cardiac death | 2.0 (2.1) | 1.0 (1.0) | 1.973 (0.206–18.947) | 0.556 |
| Non-fatal reinfarction | 2.0 (2.1) | 3.0 (3.1) | 0.651 (0.104–4.088) | 0.647 |
| Unplanned revascularization | 2.0 (2.1) | 4.0 (4.2) | 0.513 (0.099–2.663) | 0.427 |
| Malignant arrhythmia | 1.0 (1.0) | 3.0 (3.1) | 0.319 (0.032–3.148) | 0.328 |
| Readmission for heart failure | 1.0 (1.0) | 2.0 (2.1) | 0.486 (0.043–5.467) | 0.559 |
CI, confidence interval.
In terms of lipid-lowering effects, there were no significant differences in TC
levels between the two groups at 1, 6, and 12 months post-PCI (p
| Variable | Alirocumab group | Conventional treatment group | p value | ||
| TC, mmol/L | |||||
| 1 month | 3.2 |
3.5 |
0.066 | ||
| 6 months | 3.1 |
3.3 |
0.251 | ||
| 12 months | 3.1 |
3.2 |
0.432 | ||
| LDL-C, mmol/L | |||||
| 1 month | 1.9 |
2.2 |
0.011 | ||
| 6 months | 2.2 |
2.4 |
0.043 | ||
| 12 months | 2.3 |
2.4 |
0.196 | ||
| CRP, mg/L | |||||
| 24 hours | 3.5 (3.0–7.9) | 6.3 (1.9–11.0) | 0.040 | ||
| NLR | |||||
| 24 hours | 3.2 |
3.9 |
0.025 | ||
| cTNI, µg/L | |||||
| 6 hours | 4.8 (2.2–16.1) | 5.50 (3.6–13.0) | 0.541 | ||
| 24 hours | 1.5 (0.2–5.2) | 2.20 (1.2–6.5) | 0.129 | ||
| NT-pro BNP, pg/mL | |||||
| 6 hours | 580.2 (329.8–1089.8) | 650.2 (219.5–1279.8) | 0.463 | ||
| 24 hours | 349.8 (230.1–680.3) | 420.3 (329.6–799.8) | 0.358 | ||
| LVEF, % | |||||
| 6 months | 48.6 |
49.9 |
0.255 | ||
| 12 months | 47.8 |
48.6 |
0.512 | ||
| LVESV, mL | |||||
| 6 months | 46.5 |
50.2 |
0.067 | ||
| 12 months | 45.2 |
49.6 |
0.009 | ||
| LVEDV, mL | |||||
| 6 months | 109.2 |
115.5 |
0.157 | ||
| 12 months | 98.1 |
109.3 |
0.212 | ||
| MIDAS score | |||||
| Before discharge (Total score) | 75.6 |
78.3 |
0.213 | ||
| Physical Activity | 29.1 |
27.9 |
0.356 | ||
| Diet | 2.8 |
3.0 |
0.512 | ||
| Emotional Reaction | 8.2 |
8.9 |
0.426 | ||
| Dependency | 5.9 |
5.8 |
0.351 | ||
| Security | 19.9 |
21.5 |
0.167 | ||
| Medication Concerns | 5.2 |
5.6 |
0.138 | ||
| Medication Side-effects | 4.5 |
5.6 |
0.239 | ||
| 12 months (Total score) | 36.6 |
38.9 |
0.012 | ||
| Physical Activity | 12.8 |
14.8 |
|||
| Diet | 2.6 |
1.9 |
0.107 | ||
| Emotional Reaction | 4.2 |
3.4 |
0.246 | ||
| Dependency | 2.9 |
2.1 |
0.430 | ||
| Security | 9.5 |
12.0 |
|||
| Medication Concerns | 2.2 |
2.3 |
0.532 | ||
| Medication Side-effects | 2.4 |
2.4 |
0.769 | ||
MIDAS, Myocardial Infarction Dimensional Assessment Scale.
During the follow-up period, 7 adverse events were recorded: 5 (5.2%) in the alirocumab group and 2 (2.1%) in the conventional treatment group: persistent pain at the injection site (2.1% in the alirocumab group vs. 0% in the control group, p = 0.156), sore throat (2.1% in the alirocumab group vs. 0% in the control group, p = 0.156), skin itching (1.0% in the alirocumab group vs. 0% in the control group, p = 0.317), muscle pain (alirocumab group 0% vs. control group 2.1%, p = 0.156). Overall, administration of a single dose of alirocumab was not associated with an increased incidence of adverse reactions (alirocumab group: 5.2% vs. control group: 2.1%, p = 0.249) (Table 4).
| Variable | Alirocumab group | Conventional treatment group | p value | |
| Injection site reactions | ||||
| Erythema | 0 | 0 | ||
| Swelling | 0 | 0 | ||
| Persistent pain | 2 (2.1) | 0 | 0.156 | |
| Upper respiratory tract symptoms | ||||
| Sore throat | 2 (2.1) | 0 | 0.156 | |
| Runny nose | 0 | 0 | ||
| Sneezing | 0 | 0 | ||
| Skin itching | 1 (1.0) | 0 | 0.317 | |
| Hypersensitivity reactions | 0 | 0 | ||
| Muscle pain | 0 | 2 (2.1) | 0.156 | |
| Total | 5 (5.2) | 2 (2.1) | 0.249 | |
In recent years, the importance of early intensive lipid-lowering therapy in ACS has been increasingly emphasized by international guidelines. According to cholesterol management guidelines in Europe, the United States, and China, high-intensity statin therapy should be initiated at the time of hospital admission for patients with ACS, regardless of baseline LDL-C levels. For patients at extremely high risk whose LDL levels remain above target, other lipid-lowering drugs such as ezetimibe should be added as soon as possible, and PCSK9 inhibitors should be introduced when necessary to rapidly reduce LDL-C to guideline-recommended target levels [2, 13]. However, in real-world clinical practice, rates of lipid-lowering therapy intensification and LDL-C target achievement among patients with ACS remain generally low, providing a rationale for earlier initiation of PCSK9 inhibitor therapy [14]. Several clinical studies, including EVACS [15], EVOPACS [8] and EPIC-STEMI [9], have demonstrated that initiating PCSK9 inhibitor therapy at admission significantly reduces LDL-C levels within days following ACS onset, enabling patients to achieve lipid targets rapidly. A meta-analysis of nine studies involving 2869 ACS patients confirmed that early initiation of PCSK9 inhibitor therapy in the hospital setting can rapidly and significantly lower LDL-C levels, improve lipid control rates, and significantly reduce the risk of MACE in the short term, with good safety [16]. These studies suggest that initiating PCSK9 inhibitor-enhanced lipid-lowering therapy in the early stages of ACS onset is a promising strategy. However, in actual clinical application, factors such as the injection route, storage requirements, high cost, and compliance challenges associated with long-term therapy have resulted in low patient adherence and high discontinuation rates. In routine clinical practice, the use of PCSK9 inhibitors is frequently limited to single in-hospital injections [10]. Previous studies have primarily evaluated long-term, multiple-dose regimens, while the short-term and long-term impacts of single-dose administration have rarely been assessed. The present study addresses this gap by evaluating, for the first time, the efficacy and safety of a single dose of alirocumab in patients with STEMI undergoing primary PCI in a real-world clinical setting.
In this real-world, propensity score–matched cohort, the cumulative incidence of MACE over 12 months was numerically lower in the alirocumab group than in the conventional treatment group; however, this difference was not statistically significant in either Kaplan-Meier or Cox regression analyses, and the individual components of the composite endpoint (cardiac death, non-fatal myocardial infarction, unplanned revascularization, malignant arrhythmia, and rehospitalization for heart failure) were also similar between groups. Given the modest sample size (n = 192) and low absolute event count (n = 21), the study was likely underpowered to detect anything other than large treatment effects, as the statistical power of time-to-event analyses is strongly driven by the number of events. Consequently, the observed numerical differences should be regarded as exploratory and hypothesis-generating rather than as evidence of a definite clinical benefit of single-dose alirocumab. Our neutral primary endpoint contrasts with the robust risk reductions reported in large randomized trials of long-term PCSK9 inhibition, such as FOURIER (evolocumab) and ODYSSEY OUTCOMES (alirocumab), in which sustained LDL-C lowering over several years led to significant reductions in MACE. Thus, while our findings are directionally consistent with the pharmacologic effects of PCSK9 inhibition, they do not demonstrate a clinical efficacy signal for a single peri-procedural dose and instead underscore the need for adequately powered, multicenter studies with longer follow-up to clarify whether short-term or peri-PCI PCSK9 inhibition can meaningfully influence prognosis after STEMI.
Although no significant difference in the primary endpoint of MACE was observed,
superiority of the alirocumab group was evident across several secondary
endpoints: (1) In terms of lipid levels, the LDL-C levels in the alirocumab group
were significantly lower than those in the conventional treatment group at 1
month post-PPCI. Nevertheless, the mean LDL-C values observed in the alirocumab
group (1.9
In addition, the safety analysis showed that the incidence of adverse reactions was low in both groups and that the between-group difference was not statistically significant. Administration of a single dose of alirocumab was not associated with a significant increase in the risk of adverse events. Most adverse events were mild, including injection-site pain and throat discomfort, and all resolved spontaneously within 3 days. This finding is consistent with the safety profile observed after a single dose of alirocumab in healthy subjects.
Several limitations should also be acknowledged in this study: (1) The
single-center, retrospective, observational design may have introduced selection
and information bias. Moreover, as with all observational ‘real-world’ analyses,
although PSM was employed to balance known baseline confounders, unmeasured or
residual biases may still have influenced outcome comparability, thus limiting
the ability to draw conclusions with the same level of certainty as randomized
clinical trials. Differences in patient demographics, healthcare systems,
procedural techniques, and background medical therapy across regions and centers
may limit the external validity of our results. (2) The modest overall sample
size and the limited number of primary endpoint events not only reduced the
statistical power of the study but also constrained the performance of the
propensity score matching, thereby increasing the uncertainty around the
estimated treatment effects. Additionally, the follow-up duration was limited to
1 year, which may have been insufficient to capture long-term clinical benefits.
For atherosclerotic cardiovascular disease, the long-term benefits of many
interventions often become apparent only after longer-term follow-up. (3) As a
retrospective analysis, data collection was limited to predefined follow-up
protocols, restricting the availability of additional clinical indicators and
thereby weakening the strength of the conclusions. (4) Potential discrepancies
between patient recall and actual clinical events may have introduced recall
bias. Adherence to statin therapy was assessed indirectly based on longitudinal
prescription and refill records rather than objective measures such as pill
counts or electronic monitoring. Although these data suggest that all patients
remained on moderate-intensity statins during follow-up, this approach cannot
fully capture actual medication intake, and residual misclassification of
adherence cannot be excluded. (5) CRP and NLR are dynamic inflammatory markers
that can be influenced not only by the acute ischemic event itself but also by
multiple peri-procedural factors, including contrast load, procedural complexity,
periprocedural myocardial injury, perioperative infections, hemodynamic
instability, and concomitant medications such as heparins, statins, and
In summary, this study suggests that, in patients with STEMI undergoing PPCI, postoperative administration of a single dose of alirocumab may provide more effective LDL-C lowering, attenuation of early inflammatory responses, improvement in ventricular remodeling, and enhancement of quality of life than conventional therapy, while maintaining a favorable safety profile. Overall, these real-world findings support the potential clinical value of administering a single dose of alirocumab after PPCI in patients with STEMI.
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
PW: writing—original draft, performed the research. HW: writing—review and editing, analyzed the data. DY: writing—review and editing, designed the research study. ZZ: writing—original draft, Supervision, Conceptualization. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This retrospective cohort study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. Ethical approval was obtained from the Ethics Committee of the First Hospital of Lanzhou University (Approval No. LDYYLL2025-967). Given the retrospective design of the study and the use of anonymized data, the requirement for informed consent was waived.
Not applicable.
This work was supported by the Clinical Cooperative Pilot: Project of Traditional Chinese and Western Medicine for Major Diseases (no. Administration of State Administration of Traditional Chinese Medicine [2018], no. 3); National Key R&D Program of China (no. 2018YFC1311505); Gansu Provincial Clinical Research Center for Cardiovascular Diseases (no. 18JR2FA005).
The authors declare no conflict of interest.
During the preparation of this work the authors used ChatGpt-5.0 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM47437.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


