1 Center for Coronary Artery Disease, Beijing Anzhen Hospital, Capital Medical University, 100029 Beijing, China
2 Beijing Institute of Heart, Lung, and Blood Vessel Diseases, 100029 Beijing, China
Abstract
Coronary artery calcification (CAC) is a strong predictor of long-term adverse outcomes in patients with coronary artery disease (CAD). Meanwhile, insulin resistance (IR) is a key metabolic disorder that accelerates CAC progression through multiple pathways. Fibroblast growth factor 21 (FGF21) improves glucolipid metabolism and has been associated with vascular calcification. However, the relationship between serum FGF21 level and CAC severity in patients with varying degrees of IR remains unclear.
A total of 128 patients with CAD who underwent preprocedural coronary computed tomography angiography and percutaneous coronary intervention were enrolled. Patients were stratified by triglyceride–glucose (TyG) index into high (TyG >8.62, n = 62) and low (TyG ≤8.62, n = 66) groups. Associations between FGF21 levels and severe CAC were analyzed under varying degrees of IR.
In patients with a TyG index >8.62, serum FGF21 levels were significantly lower in those with severe CAC, and were negatively correlated with CAC scores. Multivariable analysis revealed that serum FGF21 levels were independently associated with severe CAC (odds ratio (OR) per 1-standard deviation (SD) increase: 0.261; 95% confidence interval (CI): 0.073, 0.933; p < 0.05). In contrast, serum FGF21 levels among patients with a TyG index ≤8.62 did not differ significantly between the severe and non-severe CAC groups, and no independent association between serum FGF21 level and severe CAC was observed after adjustment. Importantly, a significant interaction was observed between the TyG index and FGF21 level (p for interaction = 0.035). Moreover, the protective association between FGF21 and CAC was primarily observed in patients with a high TyG index.
Lower serum FGF21 levels in patients with CAD can identify individuals at increased risk of severe CAC, particularly among those with a higher degree of IR. Serum FGF21 levels may serve as a novel biomarker for CAC risk stratification in metabolically susceptible patients.
Keywords
- coronary artery disease
- coronary artery calcification
- fibroblast growth factor 21
- insulin resistance
- triglyceride-glucose index
Coronary artery disease (CAD) remains the major cause of mortality globally, with its pathogenesis intricately linked to atherosclerosis [1]. A critical hallmark of advanced atherosclerosis is the development of coronary artery calcification (CAC), which is recognized as a notable independent predictor associated with future cardiovascular events and all-cause mortality [2]. CAC is therefore a key target in the prevention and treatment of CAD, especially in CAD patients with multivessel disease [3, 4]. The progression of CAC is accelerated in individuals with metabolic disorders, particularly those with insulin resistance (IR) [5, 6]. IR fosters a pro-atherogenic milieu through multiple pathways, including chronic inflammation, endothelial dysfunction, dyslipidemia, and oxidative stress [7]. Collectively, these factors collectively promote the transition of vascular smooth muscle cells (VSMCs) toward an osteoblast-like phenotype, driving the deposition of hydroxyapatite crystals in the coronary arteries, which is a typical pathophysiological feature of CAC [8]. The triglyceride-glucose (TyG) index is a practical and reliable surrogate marker of IR that can independently predict both the presence and progression of CAC [9, 10]. Despite its established clinical significance, the precise molecular mechanisms linking IR to the accelerated calcification process have not been fully elucidated, highlighting the need for novel biomarkers and pathophysiological insights. Fibroblast growth factor 21 (FGF21), a member of the fibroblast growth factor family, is recognized as a pivotal metabolic regulator with multifaceted roles in glucose and lipid homeostasis [11]. FGF21 is primarily secreted by the liver and acts by enhancing insulin sensitivity, promoting glucose uptake, and improving lipid profiles [12]. These properties make FGF21 a critical defender against metabolic disorders such as IR, type 2 diabetes, and obesity [13, 14]. Our previous studies showed that FGF21 attenuated vascular calcification both in vivo and in vitro by inhibiting endoplasmic reticulum stress and reducing oxidative stress [15, 16, 17]. Hence, the exogenous administration of FGF21 could be a promising therapeutic intervention for vascular calcification [18].
The aim of this study was therefore to evaluate the association between serum FGF21 levels and the severity of CAC in a well-characterized cohort of patients with varying degrees of IR. By elucidating this relationship, we seek to identify a potential biomarker for risk stratification in metabolically susceptible patients with CAC.
This retrospective observational study consecutively included 128 CAD patients
aged
General patients’ data were extracted from the hospital’s electronic medical
record system, including demographic and clinical characteristics, laboratory
test results, angiographic results, and procedural details. Blood samples were
collected after overnight fasting. Routine biochemical parameters, including FBG
and lipid measurements, were processed the same day in the central laboratory
according to standardized laboratory techniques. The TyG index was calculated
using the formula: Ln [triglyceride (mg/dL)
CCTA scans were conducted using a 256-detector row CT system (Revolution CT, GE
Healthcare, Milwaukee, WI, USA). Non-contrast cardiac CT imaging was obtained
prior to CCTA, and all procedures adhered to the standards outlined in the
Society of Cardiovascular Computed Tomography guidelines [21]. The CAC score was
calculated automatically using the Agatston method [22], independent of any
clinical information. Patients with a score
After admission, fasting venous blood samples (5 mL) were collected from all patients in the morning using serum separator tubes. The samples were allowed to clot for 30 minutes at room temperature before centrifugation at 3000 r/min for 10 minutes. Subsequently, the serum was aliquoted into 1.5 mL EP tubes and stored at –80 °C. Serum FGF21 was measured by Enzyme-Linked Immunosorbent Assay (DF2100, R&D Systems, Minneapolis, MN, USA), according to the manufacturer’s instructions.
The Kolmogorov-Smirnov test was applied to assess the normality of continuous
variables. Data were expressed as the mean
Logistic regression models were constructed to evaluate the association between FGF21 and severe CAC in the overall population and subgroups with different TyG index levels. Odds ratios (ORs) were calculated per one SD increase in serum FGF21 levels. Covariates considered clinically relevant to severe CAC were selected in the multivariable analyses. The fully adjusted model was adjusted for sex, age, body mass index (BMI), history of hypertension, diabetes, dyslipidemia, smoking, and the use of antidiabetic agents. Multiplicative interaction terms were included in the adjusted models to assess whether TyG index levels modify the association between FGF21 and severe CAC. Receiver operating characteristic (ROC) curves were constructed to assess the diagnostic performance of serum FGF21 in predicting severe CAC. Youden’s index was calculated, together with the maximum value which corresponds to the optimal cutoff value of FGF21 levels. Sensitivity analyses were performed to assess the robustness of the primary findings. First, serum FGF21 levels were converted into a categorical variable based on the optimal cutoff identified by Youden’s index, and logistic regression analyses were re-performed to assess the robustness of the associations. Second, restricted cubic spline (RCS) curves (3 knots) were applied to explore the dose–response association between serum FGF21 levels and severe CAC in the overall population as well as in the subgroups stratified by TyG index.
Statistical analyses were performed with SPSS 26.0 (IBM SPSS, Armonk, NY, USA)
and R software (version 4.4.3, R Foundation for Statistical Computing, Vienna,
Austria). A two-sided p-value
A total of 128 CAD patients were enrolled and divided into high TyG index (n =
62, TyG index
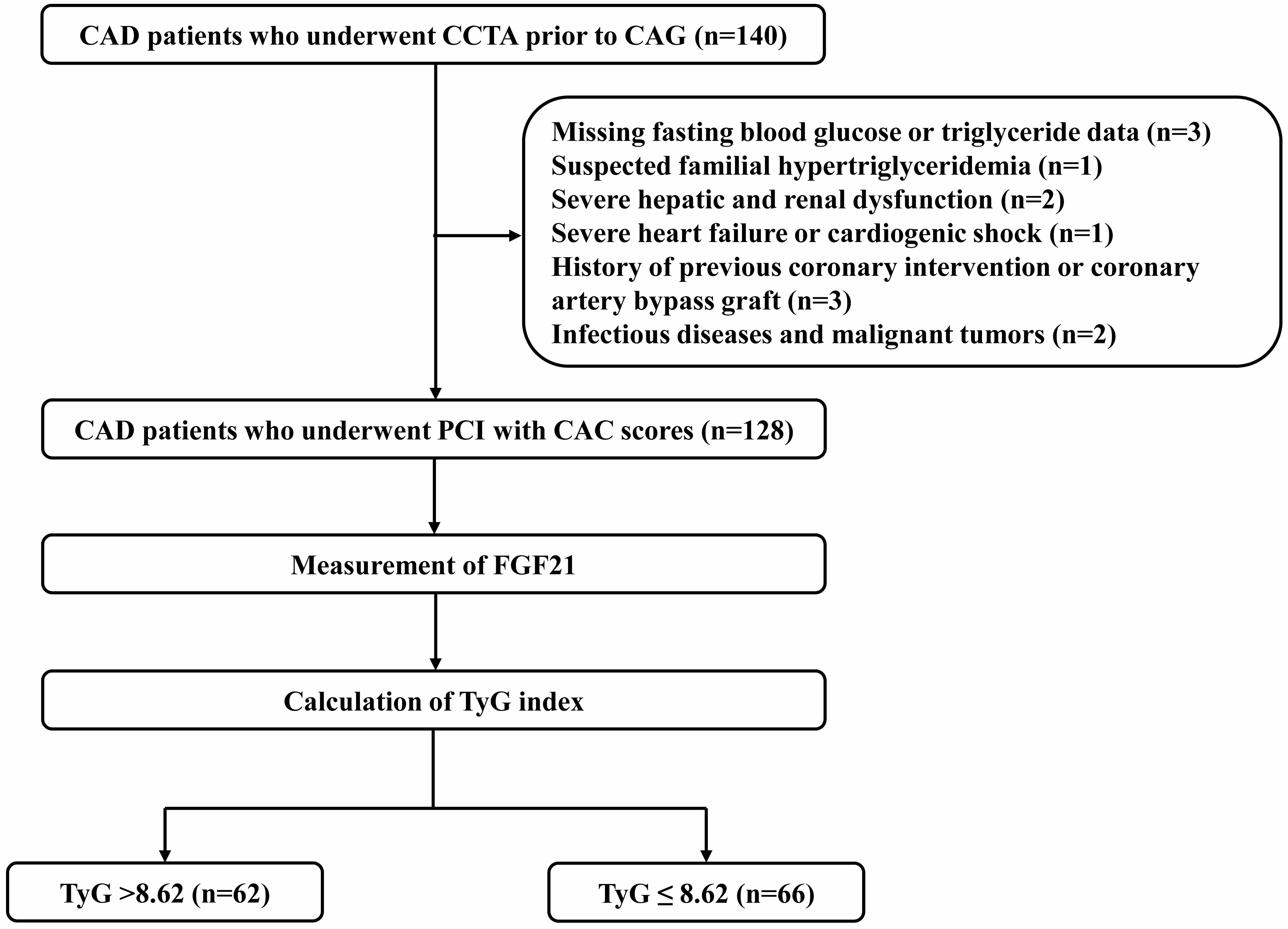 Fig. 1.
Fig. 1.
Flow diagram for patient enrollment. Abbreviations: CAC, coronary artery calcification; CAD, coronary artery disease; CAG, coronary angiography; CCTA, coronary computed tomography angiography; FGF21, fibroblast growth factor 21; PCI, percutaneous coronary intervention; TyG, triglyceride-glucose.
| Variables | Overall (n = 128) | TyG index |
TyG index |
p-value | ||
| Demographics | ||||||
| Age (years) | 58 |
57 |
59 |
0.186 | ||
| Male | 108 (84.4) | 52 (83.9) | 56 (84.8) | 0.879 | ||
| BMI (kg/m2) | 26.9 |
27.6 |
26.3 |
0.023 | ||
| Smoking | 67 (52.3) | 35 (56.5) | 32 (48.5) | 0.367 | ||
| Medical history | ||||||
| Hypertension | 83 (64.8) | 42 (67.7) | 41 (62.1) | 0.506 | ||
| Diabetes | 45 (35.2) | 30 (48.4) | 15 (22.7) | 0.002 | ||
| Dyslipidemia | 47 (36.7) | 44 (71.0) | 3 (4.5) | |||
| Prior MI | 33 (25.8) | 16 (25.8) | 17 (25.8) | 0.995 | ||
| Prior stroke | 12 (9.4) | 6 (9.7) | 6 (9.1) | 0.909 | ||
| Heart failure | 9 (7.0) | 5 (8.1) | 4 (6.1) | 0.658 | ||
| Laboratory tests | ||||||
| Creatinine (µmol/L) | 73.2 (64.1–83.0) | 74.7 (66.6–85.7) | 70.3 (62.0–82.6) | 0.234 | ||
| eGFR (mL/min/1.73 m2) | 96.8 (86.8–105.1) | 95.1 (86.1–103.8) | 96.8 (87.9–106.5) | 0.414 | ||
| FBG (mmol/L) | 5.1 (4.5–6.1) | 5.8 (5.0–6.6) | 5.2 (4.7–6.2) | |||
| TC (mmol/L) | 3.6 (3.1–4.1) | 3.8 (3.3–4.6) | 3.4 (3.1–3.8) | 0.006 | ||
| TG (mmol/L) | 1.4 (1.1–2.0) | 2.0 (1.6–2.3) | 1.1 (0.9–1.3) | |||
| LDL-C (mmol/L) | 1.9 (1.5–2.3) | 2.0 (1.5–2.7) | 1.8 (1.5–2.2) | 0.154 | ||
| HDL-C (mmol/L) | 0.95 (0.82–1.1) | 0.9 (0.8–1.0) | 1.0 (0.9–1.2) | 0.001 | ||
| TyG index | 8.62 (8.41–9.01) | 9.01 (8.86–9.34) | 8.41 (8.13–8.54) | |||
| Serum FGF21 (pg/mL) | 257.6 (156.5–370.9) | 270.6 (163.7–452.2) | 251.7 (139.4–337.3) | 0.291 | ||
| CAG and PCI results | ||||||
| CAC score | 310.0 (97.6–548.5) | 294.3 (96.4–600.0) | 340.9 (96.8–547.1) | 0.830 | ||
| Severe CAC | 48 (37.5) | 23 (37.1) | 25 (37.9) | 0.927 | ||
| Multivessel disease | 95 (74.2) | 45 (72.6) | 50 (75.8) | 0.569 | ||
| Target vessel territory | ||||||
| LAD | 50 (39.1) | 25 (40.3) | 25 (37.9) | 0.702 | ||
| LCX | 11 (8.6) | 4 (6.5) | 7 (10.6) | |||
| RCA | 67 (52.3) | 33 (53.2) | 34 (51.5) | |||
| Number of stents | ||||||
| 0 | 34 (26.6) | 16 (25.8) | 18 (27.3) | 0.710 | ||
| 1 | 28 (21.9) | 12 (19.4) | 16 (24.2) | |||
| 2 | 39 (30.5) | 22 (35.5) | 17 (25.8) | |||
| 27 (21.1) | 12 (19.3) | 15 (22.7) | ||||
| Medications during hospitalization | ||||||
| Aspirin | 128 (100.0) | 62 (100.0) | 66 (100.0) | - | ||
| P2Y12 inhibitors | 128 (100.0) | 62 (100.0) | 66 (100.0) | - | ||
| Statins | 128 (100.0) | 62 (100.0) | 66 (100.0) | - | ||
| Antidiabetic agents | 40 (31.3) | 25 (40.3) | 15 (22.7) | 0.032 | ||
| Oral hypoglycemic agents | 10 (7.8) | 6 (9.7) | 4 (6.1) | 0.446 | ||
| Insulin | 35 (27.3) | 21 (33.9) | 14 (21.2) | 0.108 | ||
Data are presented as the mean
Abbreviations: BMI, body mass index; CAC, coronary artery calcification; CAD, coronary artery disease; CAG, coronary angiography; eGFR, estimated glomerular filtration rate; FBG, fasting blood glucose; FGF21, fibroblast growth factor 21; HDL-C, high-density lipoprotein cholesterol; IR, insulin resistance; LAD, left anterior descending artery; LCX, left circumflex artery; LDL-C, low-density lipoprotein cholesterol; MI, myocardial infarction; PCI, percutaneous coronary intervention; RCA, right coronary artery; TC, total cholesterol; TG, triglyceride; TyG, triglyceride-glucose.
Table 1 shows the comparison of serum FGF21 levels and CAC scores in CAD
patients with varying degrees of IR. No significant differences in FGF21 levels
and CAC scores were observed between the high and low TyG index groups. In the
overall population, serum FGF21 levels were significantly lower in patients with
severe CAC compared to those with non-severe CAC [219.7 (111.5–319.6) vs. 273.7
(172.2–424.8) pg/mL, p = 0.019]. When stratified by TyG index, this
difference remained significant in patients with a TyG index
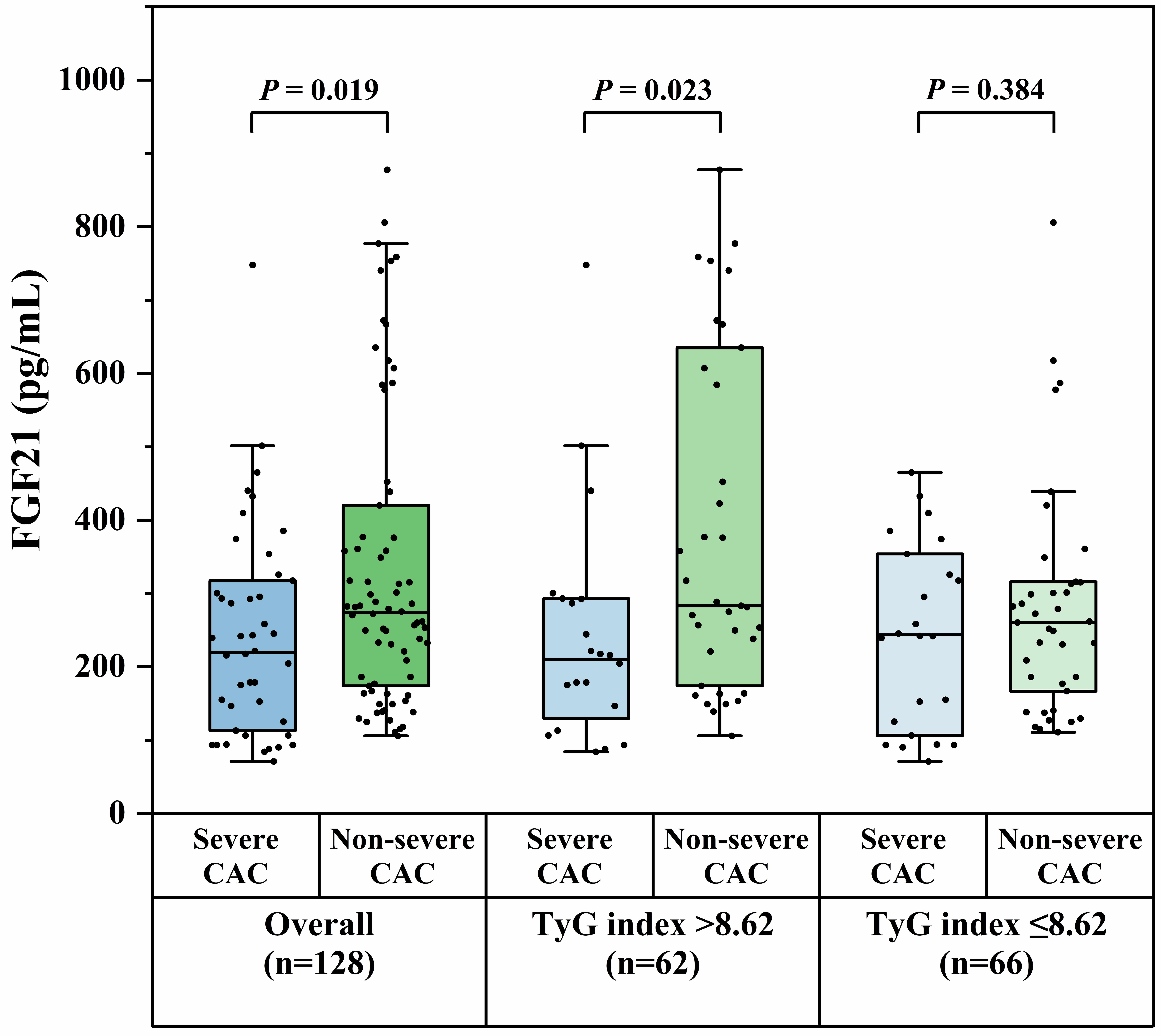 Fig. 2.
Fig. 2.
Comparison of serum FGF21 levels between severe and non-severe CAC groups in CAD patients with varying degrees of IR. Abbreviations: CAC, coronary artery calcification; CAD, coronary artery disease; FGF21, fibroblast growth factor 21; IR, insulin resistance; TyG, triglyceride-glucose.
| Variable | Overall (n = 128) | TyG index |
TyG index | |||
| r with CAC score | p-value | r with CAC score | p-value | r with CAC score | p-value | |
| Serum FGF21 (pg/mL) | –0.203 | 0.029 | –0.275 | 0.042 | –0.133 | 0.309 |
Abbreviations: CAC, coronary artery calcification; CAD, coronary artery disease; FGF21, fibroblast growth factor 21; IR, insulin resistance; TyG, triglyceride-glucose.
Table 3 shows the results of logistic regression analyses. In the overall
population, serum FGF21 levels were independently associated with severe CAC
after full adjustment. The OR per 1-SD increase in FGF21 level was 0.481 (95%
confidence interval [CI]: 0.244, 0.949; p
| FGF21 (per 1-SD) | Overall (n = 128) | TyG index |
TyG index | |||
| OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | |
| Model 1 | 0.482 (0.264, 0.880) | 0.018 | 0.335 (0.121, 0.927) | 0.035 | 0.674 (0.351, 1.296) | 0.237 |
| Model 2 | 0.479 (0.252, 0.910) | 0.025 | 0.301 (0.092, 0.986) | 0.047 | 0.685 (0.338, 1.386) | 0.293 |
| Model 3 | 0.481 (0.244, 0.949) | 0.035 | 0.261 (0.073, 0.933) | 0.039 | 0.766 (0.366, 1.601) | 0.478 |
Abbreviations: CAC, coronary artery calcification; CAD, coronary artery disease; CI, confidence interval; FGF21, fibroblast growth factor 21; IR, insulin resistance; OR, odds ratio; SD, standard deviation; TyG, triglyceride-glucose.
Model 1: unadjusted. Model 2: adjusted for sex, age, and BMI. Model 3: adjusted for Model 2 covariates plus hypertension, diabetes, dyslipidemia, smoking, and use of antidiabetic agents.
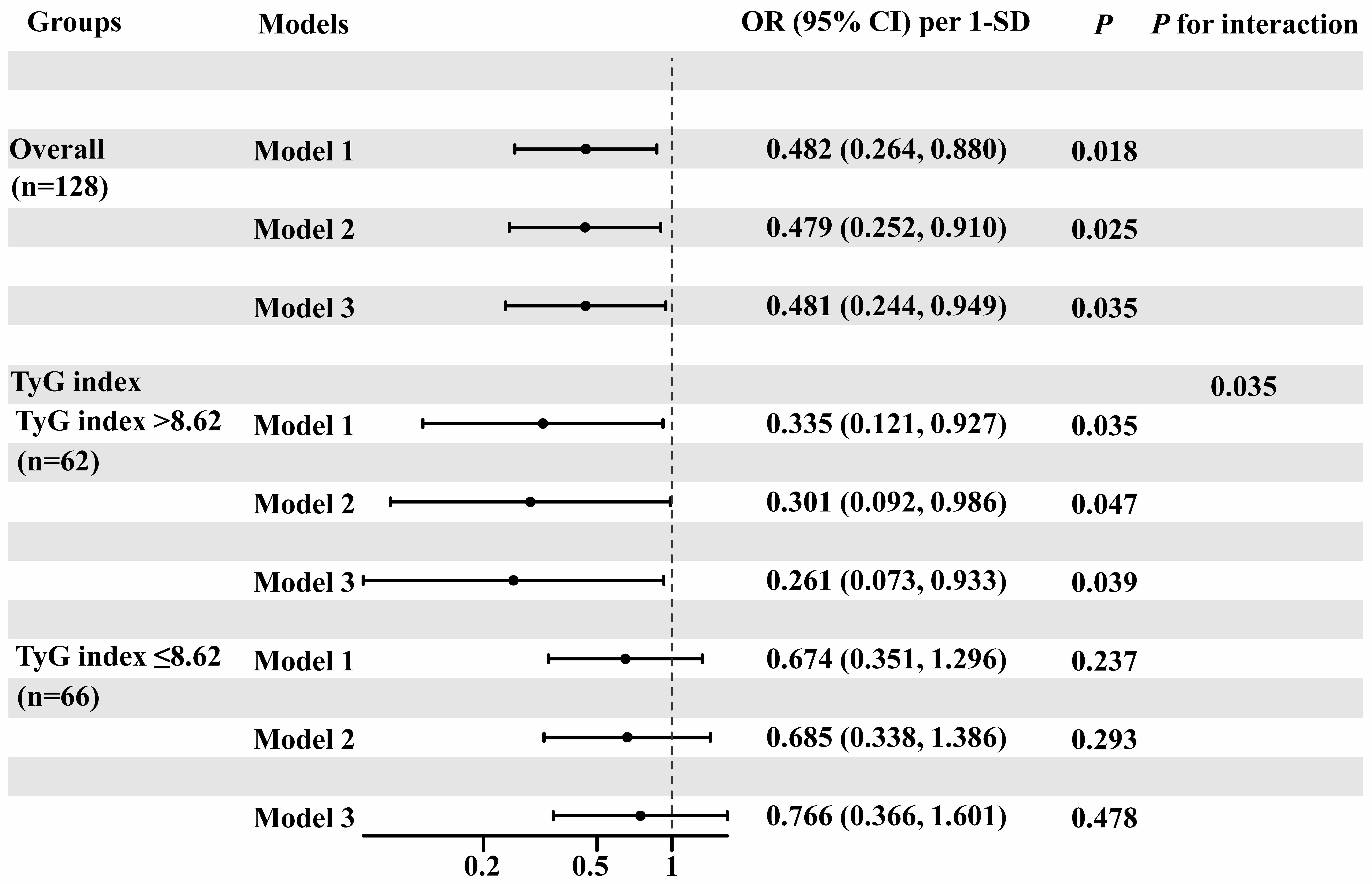 Fig. 3.
Fig. 3.
Forest plot of logistic regression analyses for the association between serum FGF21 and severe CAC in CAD patients with varying degrees of IR. Abbreviations: CAC, coronary artery calcification; CAD, coronary artery disease; CI, confidence interval; FGF21, fibroblast growth factor 21; IR, insulin resistance; OR, odds ratio; SD, standard deviation; TyG, triglyceride-glucose.
Supplementary Fig. 3 shows the diagnostic performance of FGF21 for
severe CAC, as determined by ROC curve analysis. In the overall population, the
area under the curve (AUC) for serum FGF21 was 0.632 (95% CI: 0.525, 0.739;
p = 0.019). The optimal cut-off value was determined as 114.2 pg/mL,
with a sensitivity of 97.3% and specificity of 26.2%. For patients with a TyG
index
Consistent with the main findings, when FGF21 was converted into a categorical
variable, elevated FGF21 levels (i.e., greater than the optimal cutoff) remained
significantly associated with a lower risk of severe CAC in both the overall
population (OR [95% CI]: 0.206 [0.075, 0.570], p = 0.002) and in the
high TyG index subgroup (OR [95% CI]: 0.113 [0.024, 0.540], p = 0.006)
after full adjustment. In contrast, no significant association was observed in
the low TyG index subgroup (Supplementary Table 3). Furthermore, RCS
curves revealed a generally linear, inverse dose-response relationship between
serum FGF21 levels and the risk of severe CAC in both the overall population and
the high TyG subgroup (p-overall = 0.057 and 0.074, respectively; both
p-nonlinear
The present study found that the association between serum FGF21 level and
severe CAC in a cohort of patients with CAD was significantly modified by the
degree of IR, as assessed by the TyG index. Specifically, among patients with a
higher degree of IR (TyG index
Notably, this relationship was significantly modified by the degree of IR, our
findings revealed a significant interaction between the TyG index and FGF21
levels in predicting severe CAC, with the protective association of FGF21
primarily observed in patients with a higher degree of IR (TyG index
Although IR is known to be closely related to vascular calcification and FGF21 secretion, our study did not find significant differences in CAC scores or serum FGF21 levels between the high and low TyG index groups. There may be several reasons for this. First, CAC is influenced by multiple factors, including age, gender, ethnicity, and cumulative exposure to other traditional cardiovascular risk factors, and is not entirely attributable to IR [37]. Therefore, the degree of IR defined by the TyG index may not directly translate into group-level differences in the calcification burden within this relatively small CAD cohort. Second, FGF21 is a stress-induced cytokine produced primarily by the liver, adipose tissue, and skeletal muscle. Consequently, circulating FGF21 levels are regulated by various factors beyond IR, including liver function, inflammation, and drug treatments [38, 39]. These factors may partially attenuate any significant differences between groups. Third, the relatively small sample size may have limited the ability to identify modest group-level differences.
In summary, our findings indicate the association of FGF21 with CAC is not static, but is modulated by the underlying metabolic environment. The interaction between the degree of IR and FGF21 is crucial for predicting CAC risk, and the inverse association of FGF21 with CAC is more pronounced in CAD patients with impaired metabolism. Therefore, in individuals with IR, FGF21 may not only serve as a valuable biomarker for CAC risk, but also as a complementary indicator for risk stratification in metabolically susceptible CAD patients. Given the growing evidence supporting FGF21 as a potential therapeutic target for various metabolic disorders [40, 41, 42], regular monitoring of FGF21 levels and the implementation of early preventive measures in high-risk populations may hold significant clinical importance.
The present study has several limitations that should be considered. First, as a single-center, observational study, our analysis cannot establish causality between FGF21 and CAC, and the influence of selection bias or unmeasured confounders cannot be fully ruled out. Furthermore, the relatively small sample size limits the statistical power of the analyses. This potentially affects the stability of our estimates, particularly in the subgroup analyses, and limits the external validity of our findings. Moreover, the TyG cutoff used in this study was derived from the median value of our cohort, rather than an externally validated threshold, and thus may not be directly applicable to other populations. Therefore, the current findings should be interpreted with caution, and larger studies are needed to validate the observed associations. Second, the TyG index was measured only at baseline and did not capture potential fluctuations over time, which may lead to misclassification bias. Finally, the study population was derived from a single-center East Asian cohort, and hence our findings may not be generalizable to other ethnic groups. Future prospective studies with larger sample sizes and multi-time point metabolic assessments are needed to further elucidate the complex relationship between FGF21, IR, and CAC.
This study found that the association between FGF21 and CAC was significantly
modified by the degree of IR. In CAD patients with a higher degree of IR (TyG
index
AUC, area under the curve; BMI, body mass index; CAC, coronary artery calcification; CAD, coronary artery disease; CAG, coronary angiography; CCTA, coronary computed tomography angiography; CI, confidence interval; eGFR, estimated glomerular filtration rate; FBG, fasting blood glucose; FGF21, fibroblast growth factor 21; HDL-C, high-density lipoprotein cholesterol; IQR, interquartile range; IR, insulin resistance; LAD, left anterior descending artery; LCX, left circumflex artery; LDL-C, low-density lipoprotein cholesterol; MI, myocardial infarction; OR, odds ratio; PCI, percutaneous coronary intervention; RCA, right coronary artery; ROC, receiver operating characteristic; SD, standard deviation; TC, total cholesterol; TG, triglyceride; TyG, triglyceride-glucose; VSMCs, vascular smooth muscle cells.
The data regarding this article will be shared by the corresponding author upon reasonable request.
CW: Conceptualization, Data curation, Formal analysis, Methodology, Writing—original draft. YL: Investigation, Writing—review & editing. HP: Conceptualization, Writing—review & editing. JL: Project administration, Supervision, Writing—review & editing. All authors contributed to the conception and editorial changes in the manuscript. All authors have read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Beijing Anzhen Hospital (Approval no. 2025174x). The participants provided their written informed consent to participate in this study.
Not applicable.
The study was supported by the National Natural Science Foundations of China (No. 82170344).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM46781.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



