1 The First Clinical College, Ningxia Medical University, 750004 Yinchuan, Ningxia, China
2 Department of Intervention, Tongxin County People’s Hospital, 751300 Wuzhong, Ningxia, China
3 Department of Cardiology, Peking University First Hospital, 100034 Beijing, China
4 Heart Centre & Department of Cardiovascular Diseases, General Hospital of Ningxia Medical University, 750004 Yinchuan, Ningxia, China
5 The First School of Clinical Medicine, Southern Medical University, 51000 Guangzhou, Guangdong, China
6 Institute of Medical Sciences, General Hospital of Ningxia Medical University, 750004 Yinchuan, Ningxia, China
7 National Health Commission Key Laboratory of Metabolic Cardiovascular Diseases Research, Ningxia Medical University, 750004 Yinchuan, Ningxia, China
8 Ningxia Key Laboratory of Vascular Injury and Repair Research, Ningxia Medical University, 750004 Yinchuan, Ningxia, China
†These authors contributed equally.
Abstract
Residual cardiovascular risk remains substantial despite aggressive low-density lipoprotein cholesterol (LDL-C) lowering in coronary heart disease (CHD). Consequently, this elevated risk has spurred the search for non-lipid targets, such as homocysteine (HCY). However, the combined effect of HCY with LDL-C and the overall potential for combined risk stratification remain unclear.
This retrospective cohort study included patients with CHD confirmed by coronary angiography or computed tomography angiography at the General Hospital of Ningxia Medical University between January 2019 and December 2021. Participants were stratified by baseline LDL-C levels (<1.8 vs. ≥1.8 mmol/L) and HCY (<15 vs. ≥15 μmol/L). Major adverse cardiovascular events (MACEs) were employed as the primary endpoint, defined as a composite of all-cause death, stroke, non-fatal myocardial infarction, or unplanned revascularization.
A total of 744 MACEs were recorded during the 25-month follow-up. Elevated levels of LDL-C (adjusted hazard ratio (aHR) = 1.38, 95% confidence interval (CI): 1.09–1.73) and HCY (aHR = 1.47, 95% CI: 1.19–1.81) were independently associated with a higher risk of MACEs. The risk was synergistic when both factors were elevated, as patients in the high LDL-C and high HCY group had a significantly increased risk (aHR = 1.97, 95% CI: 1.34–2.90) compared to the reference group with low levels.
LDL-C and HCY are independent predictors of MACEs in patients with CHD, and the combined use of these indices improves risk stratification. Thus, integrating these indices into clinical practice could improve personalized management strategies and outcomes in this high-risk population.
Keywords
- coronary heart disease
- low-density lipoprotein cholesterol
- homocysteine
- major adverse cardiovascular events
- combined effect
Cardiovascular disease (CVD) persists as a predominant cause of global mortality [1]. Within this category, coronary heart disease (CHD) represents a significant component, often resulting in serious outcomes including death, myocardial infarction, and the need for revascularization procedures. Low-density lipoprotein cholesterol (LDL-C) is an acknowledged contributor to atherosclerosis and serves as a fundamental biomarker for risk evaluation and management in atherosclerotic cardiovascular disease [2]. Robust evidence confirms a causative link between LDL-C and CHD [3], leading international guidelines to uniformly advocate for aggressive LDL-C reduction in secondary prevention strategies [3, 4]. However, achieving target LDL-C levels does not eradicate all cardiovascular risk, indicating the necessity to discover other modifiable factors.
Conventional risk factors such as hypertension, diabetes, dyslipidemia, and smoking do not completely elucidate this residual risk [5]. Homocysteine (HCY), a sulfur-containing amino acid produced during methionine metabolism, has been recognized as an independent cardiovascular risk factor [6, 7]. Increased HCY concentrations are correlated with impaired endothelial function, enhanced oxidative stress, and a propensity for thrombosis [8, 9, 10]—key processes that facilitate the development and instability of atherosclerotic plaque. Longitudinal research has demonstrated that individuals with high HCY levels face a greater likelihood of CHD and mortality from all causes [11]. However, the causal role of HCY remains debated, and large-scale trials of HCY-lowering with B vitamins have yielded largely neutral results [12], leading to the current treatment that does not recommend such therapy for CHD management.
The pro-atherogenic effects of HCY may intersect with and amplify those of LDL-C. Biologically, HCY-induced endothelial dysfunction promotes LDL retention and modification, while concomitant oxidative stress accelerates foam cell formation and plaque progression [13, 14], suggesting a plausible synergistic risk. Furthermore, the role of lipids exhibits paradoxes; whereas LDL-C unequivocally drives atherosclerosis, its link to arrhythmias like atrial fibrillation is complex, as highlighted in a pertinent meta-analysis [15]. Despite this mechanistic interplay, the combined effect of LDL-C and HCY on clinical outcomes in CHD remains inadequately evaluated, with most prior studies examining these factors in isolation.
Owing to the constraints of relying on a single biomarker, the integration of multiple biomarkers into clinical practice could refine cardiovascular risk prediction. This study therefore sought to examine the individual and combined influences of LDL-C and HCY on the incidence of major adverse cardiovascular events (MACEs) in patient with CHD.
We performed a retrospective cohort study involving patients diagnosed with CHD
who received coronary angiography or coronary computed tomography angiography at
the General Hospital of Ningxia Medical University from January 2019 to December
2021. CHD was characterized by the presence of
Inclusion criteria were: (1) patients aged 18 years or older; (2) a confirmed diagnosis of CHD (encompassing stable or unstable angina or myocardial infarction); (3) availability of baseline LDL-C and HCY measurements. Exclusion criteria included: (1) absent or incomplete clinical or laboratory data; (2) severe concomitant illnesses (such as advanced cancer or end-stage renal disease); (3) loss to follow-up within the 25-month study period.
From an initial screening of 6917 patients, 1153 were excluded due to insufficient data, and an additional 627 were lost to follow-up, yielding a final analytical cohort of 5137 individuals (Fig. 1). The study protocol was approved by the General Hospital of Ningxia Medical University ethics committee, with a waiver for patient informed consent.
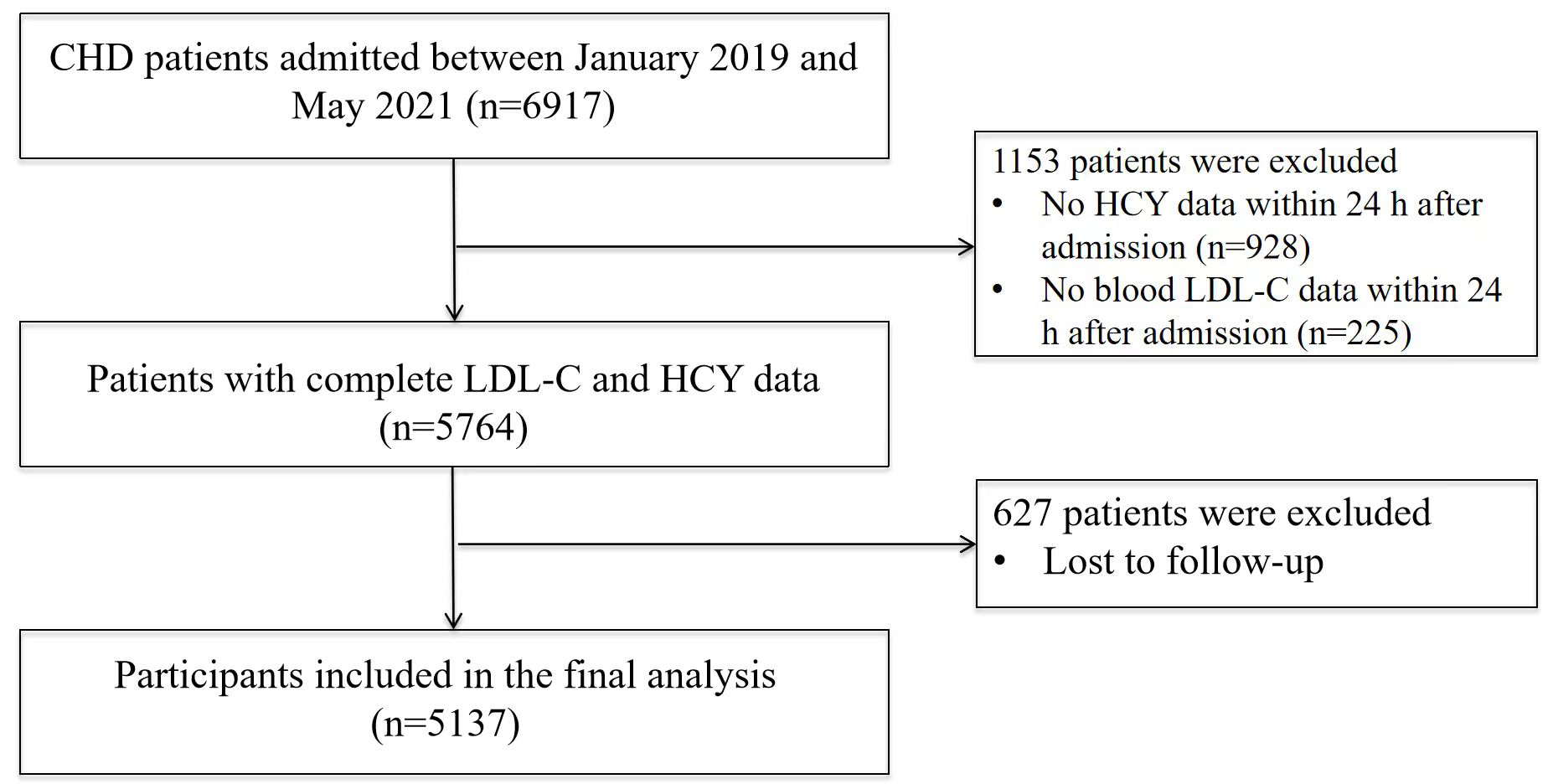 Fig. 1.
Fig. 1.
Flowchart illustrating the selection process of study participants. CHD, coronary heart disease.
Participants were stratified by baseline LDL-C and HCY levels using clinically relevant cut-off points. The LDL-C cut-off of 1.8 mmol/L was selected in accordance with the recommended target for secondary prevention in current international guidelines [3, 4]. The HCY cut-off of 15 µmol/L is widely used to define hyperhomocysteinemia in clinical practice and epidemiological studies in our region, and aligns with the reference standard of our institutional laboratory.
Demographic information, lifestyle factors, clinical history (including age, sex, smoking status, alcohol consumption, hypertension, diabetes, and body mass index [BMI]), and laboratory parameters (LDL-C, HCY, total cholesterol [TC], triglycerides [TG], high-density lipoprotein [HDL-C], fasting glucose, creatinine) were obtained from electronic medical records. Blood specimens were drawn from a peripheral vein into Ethylenediaminetetraacetic acid containing tubes within 24 hours of hospital admission and analyzed using a Sysmex automated hematology analyzer (Sysmex Corporation, Kobe, Japan) and a Beckman Coulter series automated biochemistry analyzer (Beckman Coulter, Inc., Brea, CA, USA). Information on interventional procedures, including percutaneous coronary intervention (PCI), was also documented.
The follow-up period extended to 25 months after patient discharge. The primary endpoint was the occurrence of MACEs, a composite of all-cause mortality, stroke, acute myocardial infarction (AMI), or unplanned revascularization. Follow-up data were acquired through review of electronic health records or structured telephone interviews. Standardized follow-up procedures were implemented by trained staff, and any ambiguous events were reviewed by two senior clinicians.
All-cause mortality was defined as death from any cause. Stroke was identified as a disabling neurological deficit attributable to cerebral ischemia or hemorrhage. Non-fatal myocardial infarction was diagnosed based on ischemic symptoms, elevated cardiac biomarker levels, characteristic electrocardiographic changes, or imaging evidence of new myocardial necrosis, in the absence of fatal outcome. Unplanned revascularization referred to any urgent coronary procedure performed in response to an acute coronary syndrome or other acute ischemic event.
Continuous variables are summarized as mean
The continuous associations of LDL-C and HCY with MACE risk were visualized using generalized additive models with smoothing splines. Cumulative event incidence was plotted with Kaplan–Meier curves, and group differences were assessed with the log-rank test. Univariate and multivariable Cox proportional hazards models were employed to compute hazard ratios (HRs) and corresponding 95% confidence intervals (CIs). Multivariable models were adjusted for age, sex, hypertension, diabetes, smoking status, alcohol use, TC, TG, BMI, and creatinine levels.
All statistical analyses were conducted using R software (version 4.1.0; R Foundation for Statistical Computing, Vienna, Austria) and EmpowerStats (version 4.0; X&Y Solutions, Inc., Boston, MA, USA), a statistical platform commonly used in biomedical research. A two-sided p-value below 0.05 was deemed statistically significant.
Table 1 displays the baseline characteristics for the 5137 participants,
categorized according to their LDL-C and HCY levels. Significant differences were
observed across groups regarding demographic, lifestyle, and clinical variables
(all p
| Variable | Total (N = 5137) | Low LDL-C & low HCY (n = 448) | High LDL-C & low HCY (n = 1095) | Low LDL-C & high HCY (n = 957) | High LDL-C & high HCY (n = 2637) | p value | |
| Demographics | |||||||
| Age, years (SD) | 62.66 |
61.65 |
60.42 |
65.04 |
62.89 |
||
| Male, n (%) | 3525 (68.6) | 252 (56.2) | 595 (54.3) | 738 (77.1) | 1940 (73.6) | ||
| BMI, kg/m2 (SD) | 25.10 |
24.69 |
25.19 |
24.92 |
25.21 |
0.011 | |
| Lifestyle | |||||||
| Smoking, n (%) | 2423 (47.2) | 174 (38.8) | 422 (38.5) | 475 (49.6) | 1352 (51.3) | ||
| Drinking, n (%) | 1198 (23.3) | 102 (22.8) | 236 (21.6) | 231 (24.1) | 629 (23.9) | 0.429 | |
| Clinical factors | |||||||
| Hypertension, n (%) | 3504 (68.2) | 324 (72.3) | 749 (68.4) | 683 (71.4) | 1748 (66.3) | 0.006 | |
| Diabetes mellitus, n (%) | 1612 (31.4) | 183 (40.8) | 416 (38.0) | 285 (29.8) | 728 (27.6) | ||
| Biochemical markers | |||||||
| Glucose, mmol/L (SD) | 7.09 |
7.24 |
7.33 |
6.83 |
7.06 |
0.003 | |
| HCY, µmol/L [IQR] | 17.98 [14.24–24.28] | 12.70 [11.36–13.74] | 12.80 [11.33–13.90] | 20.82 [17.38–26.99] | 21.12 [17.64–29.12] | ||
| LDL-C, mmol/L (SD) | 2.37 |
1.43 |
2.69 |
1.45 |
2.72 |
||
| HDL-C, mmol/L (SD) | 0.96 |
0.97 |
1.02 |
0.91 |
0.96 |
||
| TC, mmol/L (SD) | 3.96 |
2.97 |
4.36 |
2.95 |
4.33 |
||
| TG, mmol/L (SD) | 1.75 |
1.61 |
1.85 |
1.54 |
1.82 |
||
| Creatinine, µmol/L (SD) | 74.22 |
64.69 |
64.39 |
81.19 |
77.38 |
||
| Procedure | |||||||
| PCI, n (%) | 3134 (61.0) | 259 (57.8) | 665 (60.7) | 563 (58.8) | 1647 (62.5) | 0.102 | |
Notes: Data are presented as mean (standard deviation, SD), median [interquartile range], or number (percentage). LDL-C, low-density lipoprotein cholesterol; HCY, homocysteine; BMI, body mass index; TC, total cholesterol; TG, triglycerides; PCI, percutaneous coronary intervention; HDL-C, high-density lipoprotein.
Multivariable-adjusted spline curves indicated a positive continuous
relationship between LDL-C concentration and the risk of MACEs (Fig. 2A). In the
unadjusted model, participants with elevated LDL-C (
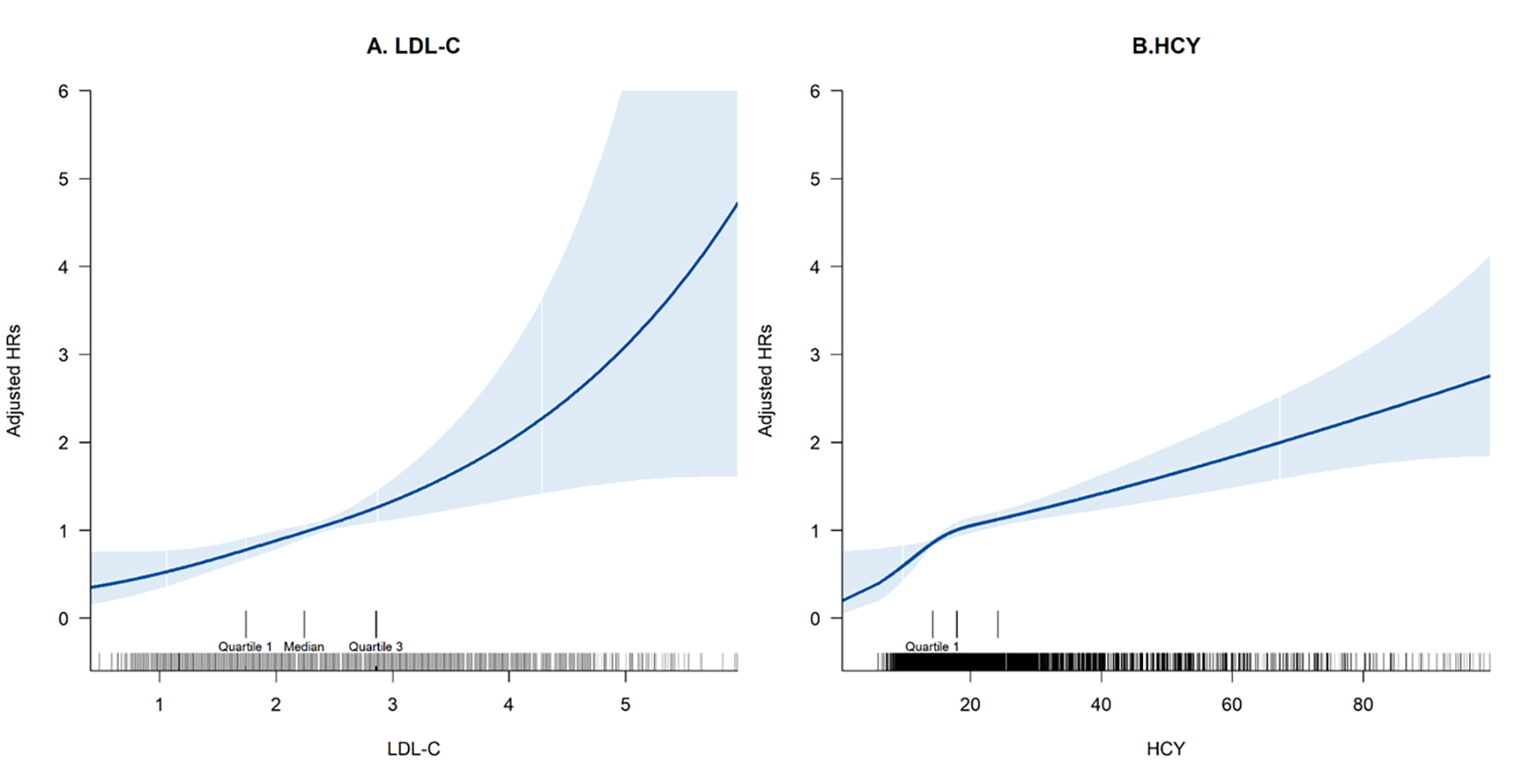 Fig. 2.
Fig. 2.
Multivariable adjusted spline curves for the associations of (A) LDL-C and (B) HCY with the risk of MACEs. HR, hazard ratio; MACEs, major adverse cardiovascular events.
| Group | n | Events, n (%) | Crude HR (95% CI) | p value | Adjusted HR†† (95% CI) | p value | |
| LDL-C categories | |||||||
| LDL-C |
1405 | 177 (12.6) | Ref | — | Ref | — | |
| LDL-C |
3732 | 567 (15.2) | 1.32 (1.12–1.57) | 0.001 | 1.38 (1.09–1.73) | 0.006 | |
| HCY categories | |||||||
| HCY |
1543 | 139 (9.0) | Ref | — | Ref | — | |
| HCY |
3594 | 605 (16.8) | 1.56 (1.29–1.88) | 1.47 (1.19–1.81) | |||
| Combined LDL-C and HCY | |||||||
| Low LDL-C & low HCY | 448 | 37 (8.3) | Ref | — | Ref | — | |
| High LDL-C & low HCY | 1095 | 102 (9.3) | 1.23 (0.84–1.79) | 0.285 | 1.35 (0.88–2.06) | 0.169 | |
| Low LDL-C & high HCY | 957 | 140 (14.6) | 1.46 (1.01–2.09) | 0.043 | 1.44 (0.97–2.14) | 0.067 | |
| High LDL-C & high HCY | 2637 | 465 (17.6) | 1.93 (1.38–2.70) | 1.97 (1.34–2.90) | 0.001 | ||
| p for trend | — | — | — | — | |||
Notes: ††Adjusted for age, sex, hypertension, diabetes mellitus, smoking, drinking, TC, TG, BMI and creatinine. CI, confidence interval; Ref, reference group.
Fig. 2B illustrates the association between HCY levels and MACEs. Participants
with high HCY (
Kaplan-Meier curves displayed a graded increase in cumulative MACE incidence
across the groups (Fig. 3). The lowest risk was observed in the group with low
LDL-C and low HCY, followed successively by high LDL-C & low HCY, low LDL-C &
high HCY, and finally the high LDL-C & high HCY group (log-rank p
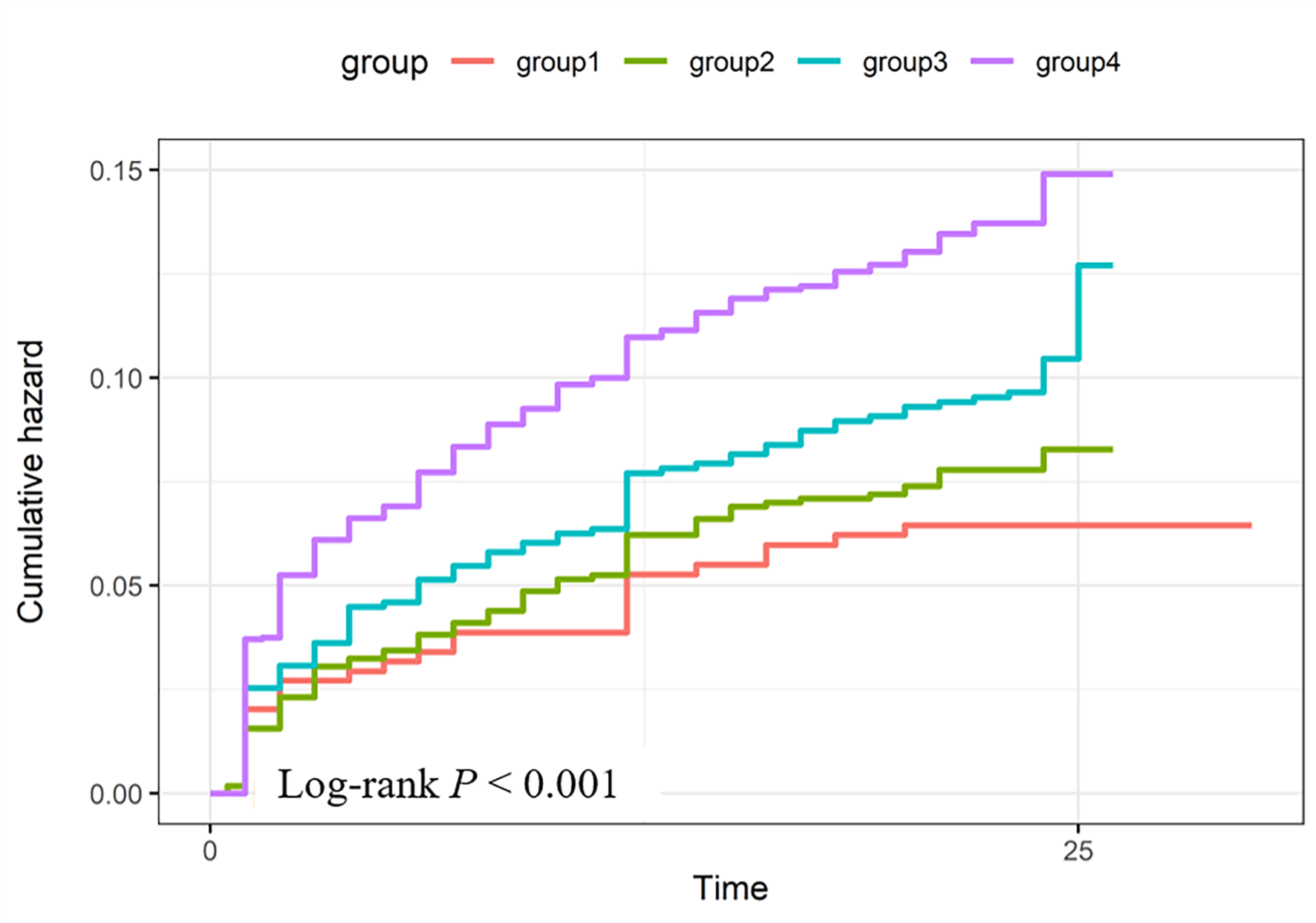 Fig. 3.
Fig. 3.
Kaplan–Meier survival curves for cumulative incidence of MACEs
stratified by LDL-C and HCY categories. Groups include: Group1 (low LDL-C & low
HCY), Group2 (high LDL-C & low HCY), Group3 (low LDL-C & high HCY), and Group4
(high LDL-C & high HCY). Log-rank test indicated significant differences among
groups (p
This investigation confirms significant associations between LDL-C and HCY with
the risk of MACEs in patients diagnosed with CHD. These observations are
consistent with prior studies: LDL-C is a validated risk factor for
atherosclerosis [2], and elevated HCY promotes endothelial dysfunction, oxidative
stress, and thrombotic processes, thereby accelerating cardiovascular disease
progression [8, 9, 10]. Importantly, our results further reveal a combined effect of
LDL-C and HCY on MCAE risk. Patients with concurrent high LDL-C (
LDL-C is universally acknowledged as a primary pathogenic agent in coronary atherosclerosis, a view substantiated by extensive evidence. For instance, investigations in renal transplant recipients have indicated that a high LDL-C/HDL-C ratio markedly increases cardiovascular morbidity and mortality, emphasizing its utility as a risk indicator [16]. Likewise, in individuals with high-risk hypercholesterolemia, LDL-C levels exhibit a linear correlation with cardiovascular disease incidence, affirming that reducing LDL-C decreases risk [17]. Furthermore, in patients presenting with acute coronary syndrome (ACS), reduced HDL-C levels are independently associated with increased cardiovascular event rates, underscoring the critical influence of lipid metabolism on clinical outcomes [18]. Despite effective LDL-C-lowering therapies, residual risk remains, implicating other factors such as triglycerides [19] and inflammation [20]. Our results reinforce LDL-C as a crucial predictor of cardiovascular risk in CHD patients and further identify elevated HCY as a factor contributing to residual risk.
Elevated plasma HCY is associated with pro-atherogenic and pro-thrombotic mechanisms [21, 22]. A meta-analysis found that each 25% increase in plasma HCY concentration corresponds to a 10% greater risk of cardiovascular disease and a 20% increased risk of stroke [20]. Similarly, an increment of 5 µmol/L in HCY was associated with a 52% elevated risk of new-onset heart disease and a 32% higher mortality risk [23]. Clinically, high levels of HCY levels predict short-term adverse events in acute myocardial infarction patients [24] and are an independent predictor of MACEs in patients with ACS [25]. Moreover, the co-existence of hypertension and hyperhomocysteinemia has been strongly linked to carotid plaque development [26], amplifying overall cardiovascular risk. Although randomized controlled trials on HCY-lowering treatments have not uniformly shown clinical benefits [27], our findings suggest that elevated HCY retains clinical relevance.
The most compelling finding of this study was the combined effect of LDL-C and HCY on MACE risk. Patients with high concentrations of both indices the highest risk of MACEs. LDL-C primarily facilitates lipid accumulation within the arterial wall, advancing atherosclerosis and stenosis [28], whereas HCY aggravates vascular damage through endothelial dysfunction, oxidative stress, and inflammatory processes [26]. This combined effect stems from interconnected pathways where HCY-induced endothelial dysfunction and oxidative stress promote the retention and modification of LDL-C—a pivotal atherogenic step. Conversely, the inflammatory milieu from oxidized LDL exacerbates HCY-mediated metabolic disturbances, creating a vicious cycle that amplifies atherosclerosis.
The inconsistent outcomes of large-scale HCY-lowering trials with B vitamins [12], despite effectively reducing plasma HCY levels [29], may be explained by several factors: intervention timing may be too late in established CHD, HCY may be a marker of underlying pathology rather than a modifiable target, and benefits may be restricted to unselected genetic or nutritional subgroups. Our findings—that risk is greatest with concurrent hyperlipidemia—suggest future trials must account for this synergy. It is critical to emphasize that our results do not contradict the current clinical guidelines; they do not support the routine use of B-vitamin supplementation for CHD patients.
Current clinical guidelines prioritize LDL-C reduction as the foundation of secondary prevention [3, 4], yet residual risk remains substantial. Our study suggests that HCY assessment may help identify high-risk individuals. For patients with both high LDL-C and high HCY, aggressive lipid-lowering (e.g., with Proprotein Convertase Subtilisin/Kexin type 9 inhibitors) combined with HCY-lowering interventions might improve outcomes [30]. A dual-biomarker approach could enable more precise and personalized cardiovascular risk management.
Our findings advocate for a dual-biomarker strategy to identify high-risk CHD patients who may benefit from intensified management. However, optimizing secondary prevention extends beyond risk identification to ensuring long-term adherence to prescribed therapies. The challenge now lies in converting risk identification into sustained adherence. Digital strategies provide a solution: Digital Health Interventions, such as remote monitoring and messaging, boost medication adherence, as evidenced in post-ACS care [31]. Simultaneously, clinicians must steer patients toward trustworthy online health information [32]. Embedding these tools into care pathways is vital for ensuring long-term risk reduction.
This study has several limitations. First, the retrospective, single-center design may affect the generalizability of the findings and introduce selection bias. Second, only baseline measurements of LDL-C and HCY were available; fluctuations over the follow-up period were not captured. Third, despite multivariate adjustment, residual confounding cannot be entirely ruled out due to unmeasured variables such as dietary habits, genetic factors, inflammatory markers, or medication compliance. Fourth, loss to follow-up may have introduced bias. Finally, the study cohort comprised only Chinese patients, potentially limiting the extrapolation of findings to other ethnic populations.
Both LDL-C and HCY were independent predictors of MACEs in CHD patients. When elevated together, they exhibited a combined effect, nearly doubling the risk of MACEs. These results support the clinical utility of combined biomarker assessment for improved cardiovascular risk stratification. Future prospective, multicenter studies are needed to validate these findings and explore integrated treatment strategies targeting both LDL-C and HCY.
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
Conceptualization: SBJ, GZC; Methodology: GZC, RY, YRM, PW, ZYW; Data acquisition and investigation: BW, TSM, JWY, BZZ, XYL; Formal analysis: BZZ, XYL; Writing—original draft: BZZ, XYL; Writing—review and editing: BZZ, XYL; Supervision: SBJ; Resources: SBJ, ZYW. All authors commented on previous versions of the manuscript. All authors have read and agreed to the published version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Ethics Committee of the General Hospital of Ningxia Medical University approved the study protocol and informed consent procedure (KYLL-2024-0088). This study was performed in line with the principles of the Declaration of Helsinki. All data were analyzed in an entirely de-identified manner. The requirement for written informed consent was waived by the approving ethics committee due to the retrospective and nature of this study.
We would like to gratefully acknowledge all of the investigators and patients participating in this work.
This work was supported by Open competition mechanism to select the best candidates for key research projects of Ningxia Medical University (grant number: XJKF230205) and the Excellent Talent Support Program of Ningxia Province.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



