1 Department of Cardiology, West China Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
2 West China Hospital School of Nursing, Sichuan University, 610041 Chengdu, Sichuan, China
Abstract
The clinical value of B-type natriuretic peptide (BNP) or N-terminal pro-B-type natriuretic peptide (NT-proBNP)-guided therapy for improving outcomes in patients with heart failure (HF) remains controversial. Thus, this meta-analysis synthesizes the available evidence from randomized controlled trials (RCTs) to determine whether a biomarker-guided strategy reduces all-cause mortality and HF-related hospitalizations compared with clinically guided management.
This systematic review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We conducted a systematic search of PubMed, Embase, the Cochrane Library, and Web of Science databases from inception to May 2025 for RCTs comparing biomarker-guided versus clinically guided management in patients with HF. Pooled risk ratios (RRs) were calculated using a random-effects model. We performed extensive supplementary analyses, including a subgroup analysis, sensitivity analysis, and trial sequential analysis (TSA).
We included 17 articles (reporting on 17 distinct RCTs) comprising 5069 patients. The primary meta-analysis showed that biomarker-guided therapy was associated with a significant reduction in all-cause mortality (RR 0.84, 95% confidence interval (CI) 0.73–0.96; I2 = 12.2%) and HF-related hospitalizations (RR 0.79, 95% CI 0.65–0.96; I2 = 53.7%). However, the robustness of these findings was undermined by subsequent analyses. Meanwhile, a sensitivity analysis restricted to studies with a low risk of bias rendered the mortality benefit non-significant (RR 0.90, 95% CI 0.79–1.03). Egger's test indicated potential publication bias (p = 0.0285), and TSA suggested the cumulative evidence was insufficient to draw a definitive conclusion.
Although there is a trend toward benefit, the existing evidence for biomarker-guided HF therapy is deemed “very low” quality based on the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) assessment. The results were compromised by methodological deficiencies in primary studies and potential publication bias. Therefore, the evidence is inadequate to support the routine use of this strategy in clinical practice. Further large-scale, high-quality RCTs are warranted.
CRD420250652134, https://www.crd.york.ac.uk/PROSPERO/view/CRD420250652134.
Graphical Abstract

Keywords
- heart failure
- brain natriuretic peptide
- biomarkers
- systematic review
- meta-analysis
Heart failure (HF) represents a growing global health challenge, affecting an estimated 64 million individuals and imposing a substantial public health and economic burden [1, 2]. Pathophysiologically, HF is defined by congestion or fluid overload, which are the primary drivers of symptom aggravation, organ dysfunction, and recurrent hospitalizations [3, 4]. Despite notable advancements in guideline-directed medical therapy (GDMT), including angiotensin receptor-neprilysin inhibitors (ARNIs) and sodium-glucose cotransporter 2 (SGLT2) inhibitors, hospitalizations for HF remain prevalent, highlighting an ongoing necessity for improved management strategies [5, 6, 7]. Up to fifty percent of patients experience readmission within six months, often as a result of inadequately managed congestion [8].
Traditional fluid management relies on clinical assessment, such as monitoring symptoms and physical signs. These signs are sometimes subjective and not very sensitive, and they usually show up late in the process of hemodynamic deterioration [9, 10]. This can delay required treatment modifications, while overly aggressive diuretic therapy based on these indications may induce adverse outcomes like renal damage and electrolyte abnormalities [11].
B-type natriuretic peptide (BNP) and its N-terminal pro-B-type natriuretic peptide (NT-proBNP) are released from the ventricles in response to increased wall stress, serving as objective and dynamic markers of hemodynamic congestion [12]. Theoretically, titrating HF therapies based on natriuretic peptide levels could enable a more proactive and precise management approach, potentially improving clinical outcomes [13]. However, after more than two decades of investigation, the clinical utility of this strategy remains highly contested. While some trials, like the recent STRONG-HF study, demonstrated that an intensive, NT-proBNP-informed strategy improved outcomes post-discharge for acute HF [14], other large, well-designed trials, most notably GUIDE-IT, found no benefit compared to standard care in high-risk heart failure with reduced ejection fraction (HFrEF) patients [15]. This conflict is further complicated by trials such as TIME-CHF and BATTLESCARRED, which suggested potential age-dependent effects [16, 17].
This evidentiary dissonance has resulted in cautious recommendations from major clinical practice guidelines. Both the 2022 American Heart Association/American College of Cardiology/Heart Failure Society of America (AHA/ACC/HFSA) and 2023 European Society of Cardiology (ESC) guidelines strongly endorse natriuretic peptides for diagnosis and prognostication but decline to issue a Class I recommendation for their use in therapeutic guidance, citing insufficient and conflicting evidence [6, 7]. This creates a critical evidence-practice gap: while biomarker-guided therapy is theoretically attractive for precise management, its inconsistent performance in large RCTs has prevented its clinical adoption. Previous meta-analyses have also yielded inconsistent conclusions, often limited by the inclusion of older, smaller studies [18, 19]. Therefore, this study aims to conduct an updated systematic review and meta-analysis of all eligible randomized controlled trials (RCTs) to clarify whether a biomarker-guided strategy reduces all-cause mortality and HF-related hospitalizations compared to clinically guided management, and to rigorously assess the quality and robustness of the current evidence base.
This systematic review and meta-analysis were conducted and reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement [20]. The study protocol was prospectively registered with the PROSPERO international register of systematic reviews (CRD420250652134).
We conducted a systematic electronic literature search of PubMed, Embase, the Cochrane Central Register of Controlled Trials (CENTRAL), and Web of Science from their inception to May 2025. The search strategy combined Medical Subject Headings (MeSH) and free-text terms related to “Heart Failure”, “Natriuretic Peptides”, and “Guided Therapy”. The literature screening process was conducted independently by two reviewers. Initially, titles and abstracts were screened, followed by a full-text review of potentially eligible articles to determine final inclusion. Discrepancies were resolved through consensus or by consulting a third reviewer. The full search strategy for all databases is provided in Supplementary Material 1.
Studies were included if they met the following criteria: (1) Study design:
Parallel-group RCTs. (2) Participants: Adult patients (age
Two researchers separately extracted data utilizing a standardized form. The extracted data comprised study parameters (author, year, sample size), patient demographics (age, sex, HF type, left ventricular ejection fraction (LVEF)), intervention specifics (biomarker target), follow-up length, and outcome metrics (event counts for each group). The Cochrane Risk of Bias tool 2.0 (RoB 2) (The Cochrane Collaboration, London, UK) was used to rate the overall risk of each RCT as “low risk”, “some concerns”, or “high risk”.
We performed statistical analyses using R software (version 4.2.1, The R
Foundation for Statistical Computing, Vienna, Austria). We calculated pooled risk
ratios (RRs) and 95% confidence intervals (CIs) for dichotomous outcomes using a
Mantel-Haenszel random-effects model. We quantified heterogeneity via the I2
statistic, with I2
The literature search identified 5895 records. Following multi-stage screening, 17 articles reporting on 17 unique RCTs were included in the final analysis. Cross-verification confirmed no patient overlap. The entire literature screening process is depicted in Fig. 1.
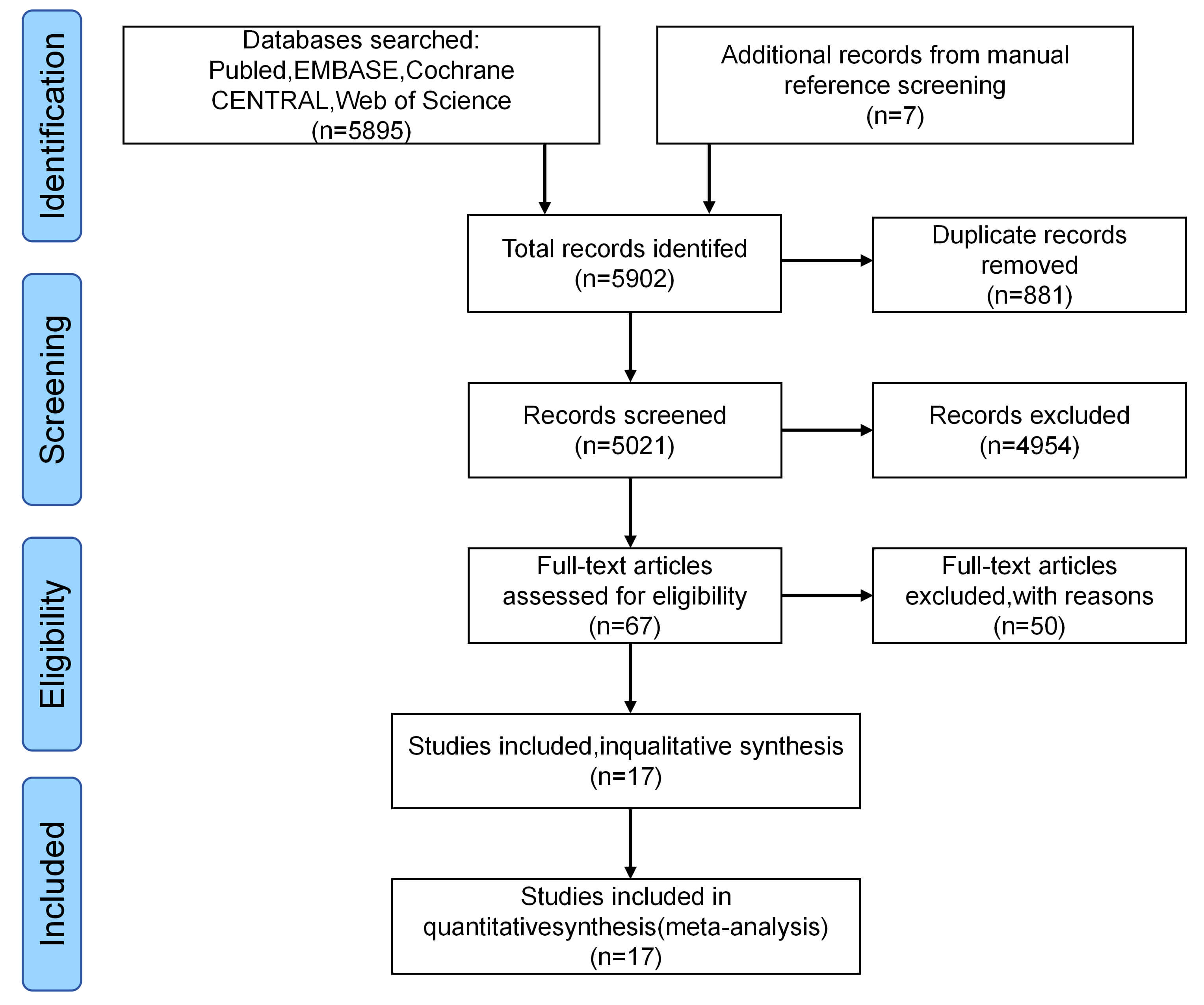 Fig. 1.
Fig. 1.
PRISMA 2020 flow diagram for study selection. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
This meta-analysis included 5069 patients (2528 in the biomarker-guided group;
2541 in the clinically guided group). Most trials (n = 14) enrolled patients with
chronic HF, while three focused on acute decompensated HF. The majority of trials targeted heart failure with reduced ejection
fraction (HFrEF; LVEF
| Study (authors, year) [Ref] | Trial name/registry ID | N (intervention/control) | Population type | Mean age (years) | LVEF (%) | Follow-up (months) | Biomarker target |
| Troughton et al. (2000) [13] | - | 33/36 | Chronic HFrEF | 69 | 27 | 6 | NT-proBNP target: decrease |
| Jourdain et al. (2007) [23] | STARS-BNP | 110/110 | Chronic HFrEF | 74 | 30 | 15 | BNP target: |
| Pfisterer et al. (2009) [16] | TIME-CHF | 251/248 | Chronic HFrEF ( |
79 | 30 (Median) | 18 | NT-proBNP target: |
| Lainchbury et al. (2009) [17] | - | 66/68 | Chronic HFrEF | 72 | 28 | 10 | NT-proBNP target: decrease |
| Eurlings et al. (2010) [24] | PRIMA | 151/159 | Chronic HF | 74 | 35 | 12 | NT-proBNP target: individual |
| Januzzi et al. (2011) [26] | PROTECT | 74/77 | Chronic HFrEF | 58 | 25 | 12 | NT-proBNP target: |
| Karlström et al. (2011) [27] | - | 66/61 | Chronic HFrEF ( |
81 | 30 | 12 | BNP target: decrease |
| Felker et al. (2017) [15] | GUIDE-IT | 446/448 | High-risk HFrEF | 62 | 26 | 15 (Median) | NT-proBNP target: |
| Stienen et al. (2018) [30] | PRIMA II | 204/202 | Acute Decompensated HF | 76 | 34 (Median) | 6 | NT-proBNP target: |
| Adamo et al. (2023) [14] | STRONG-HF | 542/536 | Acute HF | 64 | 28 (Median) | 6 | High-intensity care with NT-proBNP monitoring |
| Berger et al. (2010) [21] | - | 40/44 | Chronic HF | 72 | 34 | 9 | NT-proBNP-guided |
| Bajraktari et al. (2018) [31] | - | 60/60 | Outpatient HF | 63 | 34 | 12 | Echo |
| Mekontso Dessap et al. (2012) [28] | - | 151/153 | ICU mechanical ventilation with cardiac dysfunction | 68 | NR | 2 | NP-driven fluid management |
| Anguita et al. (2010) [22] | - | 64/65 | Chronic HF | 70 | 36 | 12 | BNP-guided |
| Kim and Kim (2012) [29] | - | 35/35 | Chronic HFrEF | 57 | 26 | 6 | BNP-guided beta-blocker titration |
| Persson et al. (2010) [25] | SIGNAL-HF | 185/189 | Chronic HF (Primary Care) | 75 | NR | 12 | NT-proBNP-guided |
| Saraya et al. (2015) [32] | - | 50/50 | Chronic HFrEF | 56 | 29 | 6 | BNP-guided |
Abbreviations: LVEF, left ventricular ejection fraction; HF, heart failure; HFrEF, heart failure with reduced ejection fraction; ULN, upper limit of normal; mgt., management; BB, beta-blocker; BNP, B-type natriuretic peptide; NT-proBNP, N-terminal pro-B-type natriuretic peptide; ICU, intensive care unit.
Using the Cochrane RoB 2 tool, we assessed the 17 included studies. Only 7 were rated as having an overall “low risk” of bias. The remaining 10 were rated as having “some concerns”, primarily due to the open-label design of the interventions, which poses a risk of performance bias, and the lack of pre-registered protocols in older studies, which increases the risk of selective reporting bias. The detailed risk of bias assessment is summarized in Fig. 2.
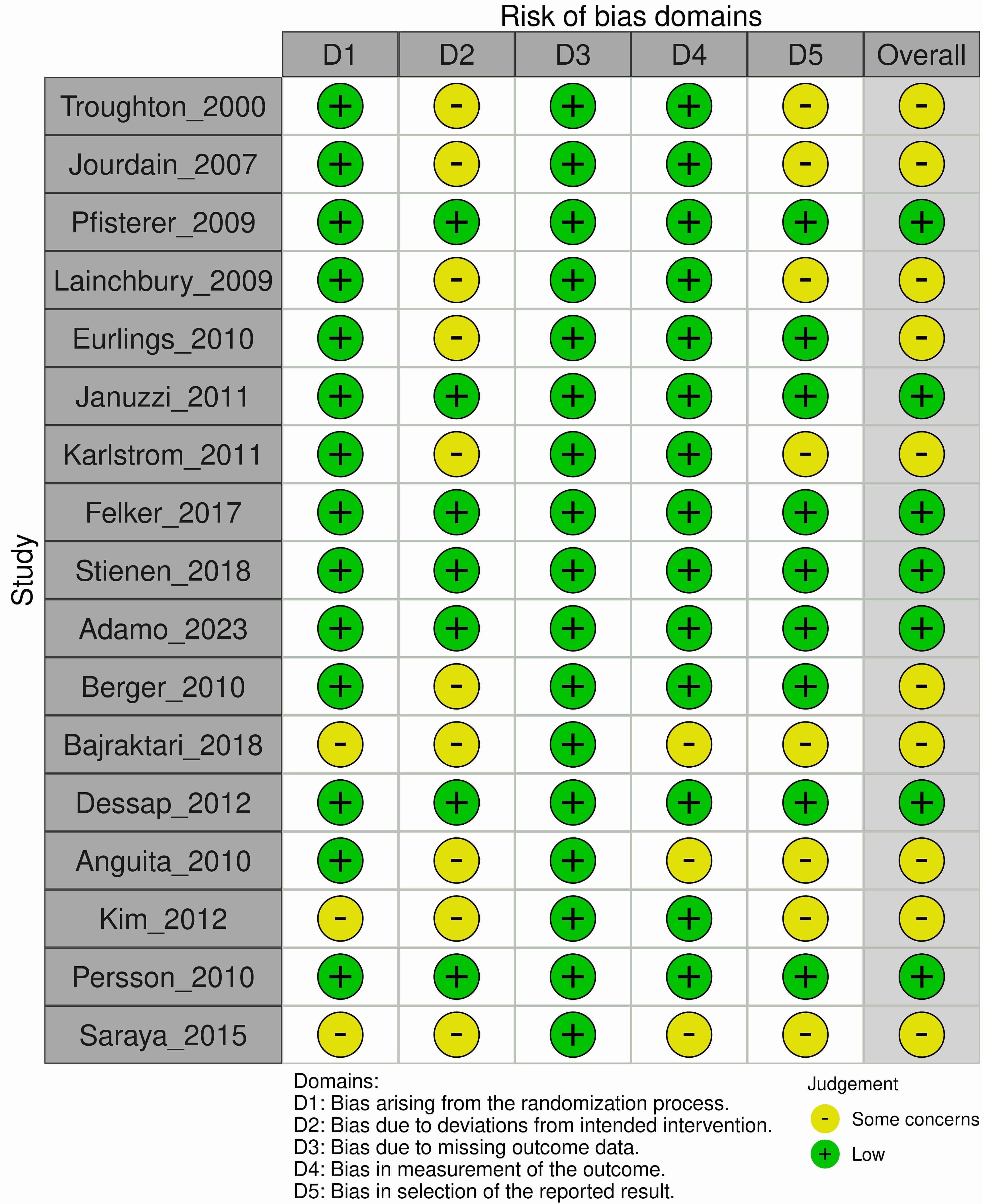 Fig. 2.
Fig. 2.
Risk of bias summary: review authors’ judgements about each risk of bias item for each included study.
Seventeen studies (5069 patients) reported data on all-cause mortality. The random-effects meta-analysis showed that biomarker-guided therapy was associated with a statistically significant 16% relative risk reduction in all-cause mortality compared to clinical guidance (RR 0.84, 95% CI 0.73–0.96, p = 0.015), with low heterogeneity (I2 = 12.2%) (Fig. 3).
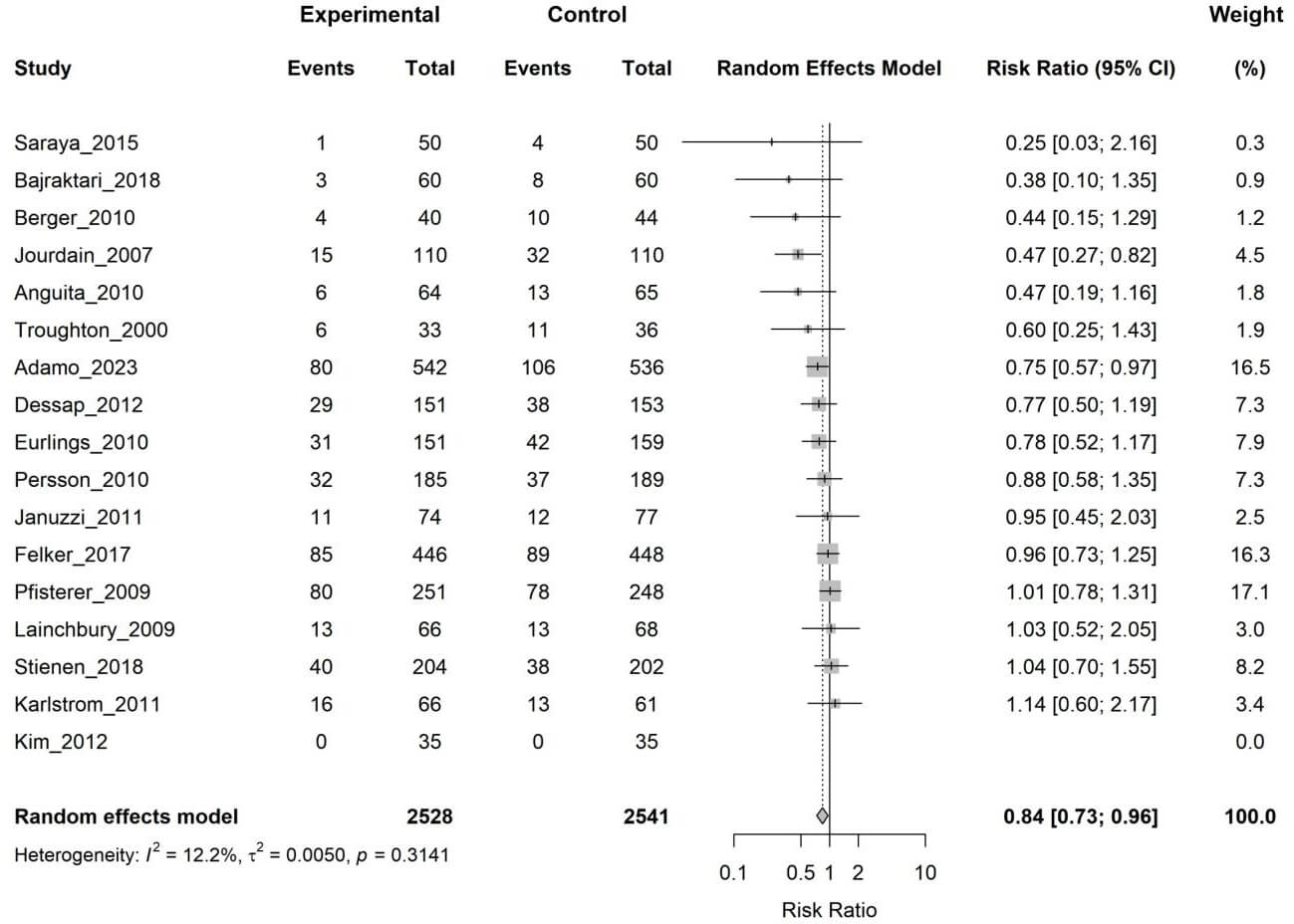 Fig. 3.
Fig. 3.
Forest plot of the effect of biomarker-guided therapy versus clinically guided therapy on all-cause mortality. CI, confidence interval.
Eight studies (3932 patients) provided data on HF-related hospitalizations. The pooled analysis demonstrated that the biomarker-guided group had a 21% lower risk of HF hospitalization (RR 0.79, 95% CI 0.65–0.96, p = 0.024), though with moderate heterogeneity (I2 = 53.7%) (Fig. 4).
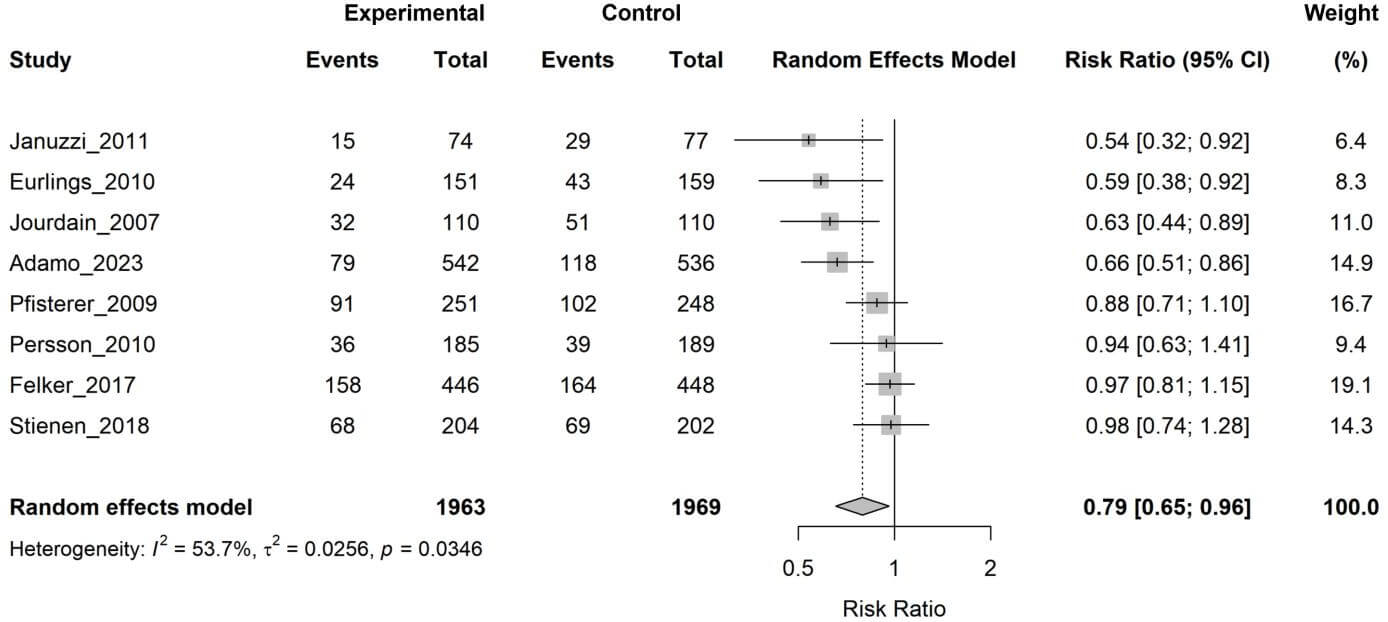 Fig. 4.
Fig. 4.
Forest plot of the effect of biomarker-guided therapy versus clinically guided therapy on heart failure-related hospitalization.
A pre-specified subgroup analysis stratified by clinical setting (chronic vs. acute HF) did not explain the heterogeneity observed for HF-related hospitalization (p for subgroup interaction = 0.92).
Critically, a sensitivity analysis restricted to the seven low-risk-of-bias studies showed that the pooled effect for all-cause mortality was no longer statistically significant (RR 0.90, 95% CI 0.79–1.03, p = 0.097), underscoring the fragility of the primary finding. Additionally, a leave-one-out sensitivity analysis using the Hartung-Knapp method was performed to challenge the robustness of our findings (Supplementary Figs. 1,2). This analysis confirmed our primary results were fragile. For all-cause mortality, omitting the influential Adamo 2023 trial (16.5% weight) caused the result to lose statistical significance (New RR 0.87, 95% CI 0.75–1.004, p = 0.056). Similarly, the HF-hospitalization finding also lost significance when several individual studies were omitted (e.g., omitting Jourdain 2007 yielded RR 0.82 [0.66–1.01]). This strongly supports that the ‘naïve’ pooled estimates are not robust and are highly influenced by single studies.
The funnel plot for all-cause mortality was asymmetric (Fig. 5), and Egger’s test confirmed a significant risk of publication bias (p = 0.0285).
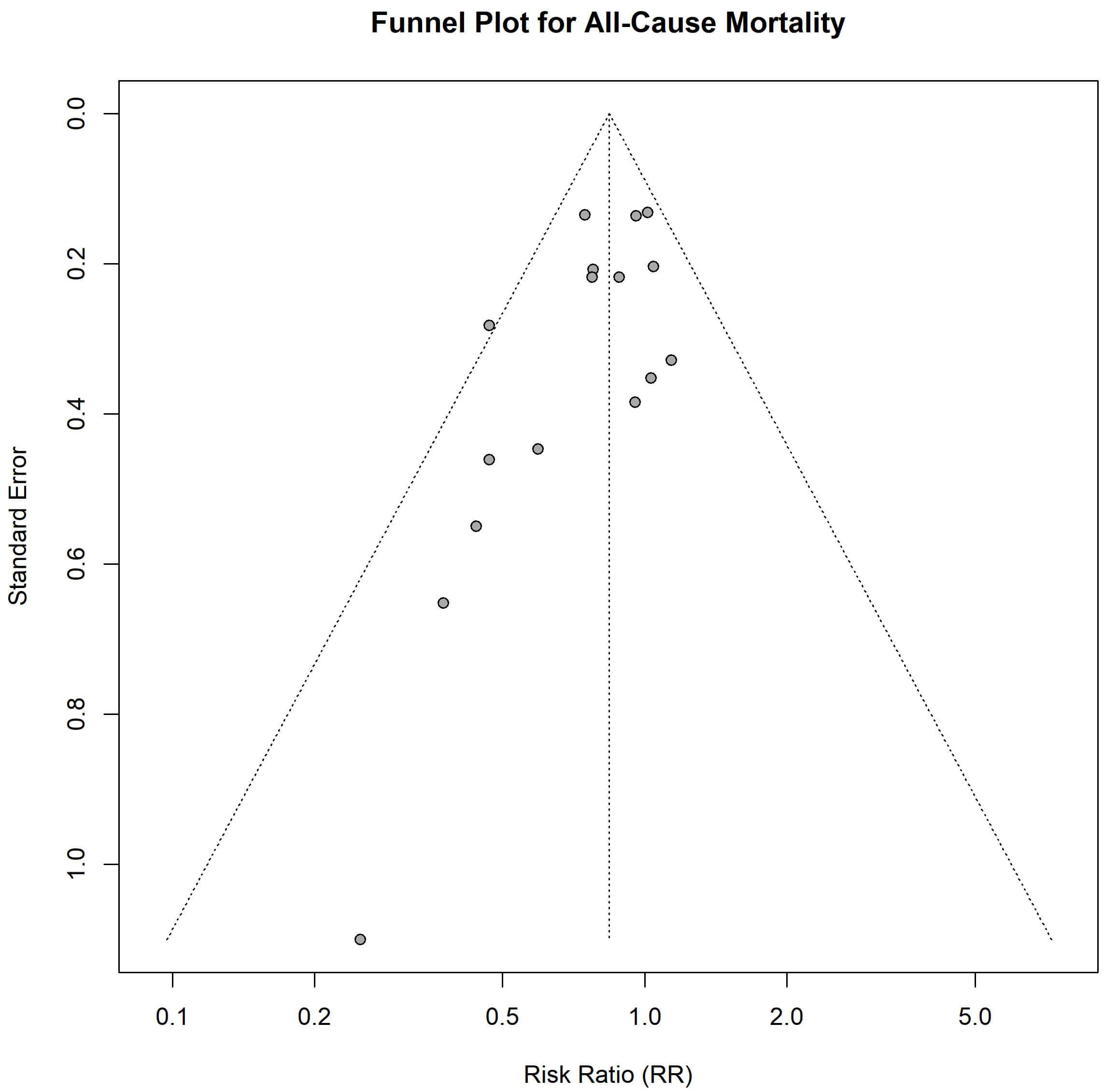 Fig. 5.
Fig. 5.
Funnel plot for the assessment of publication bias for the outcome of all-cause mortality.
Furthermore, TSA showed that while the cumulative Z-curve crossed the conventional significance boundary, it failed to cross the TSA-defined monitoring boundary for efficacy. The total sample size (5069) was substantially smaller than the required information size (14,888), indicating that the cumulative evidence is insufficient to draw a definitive conclusion (Fig. 6).
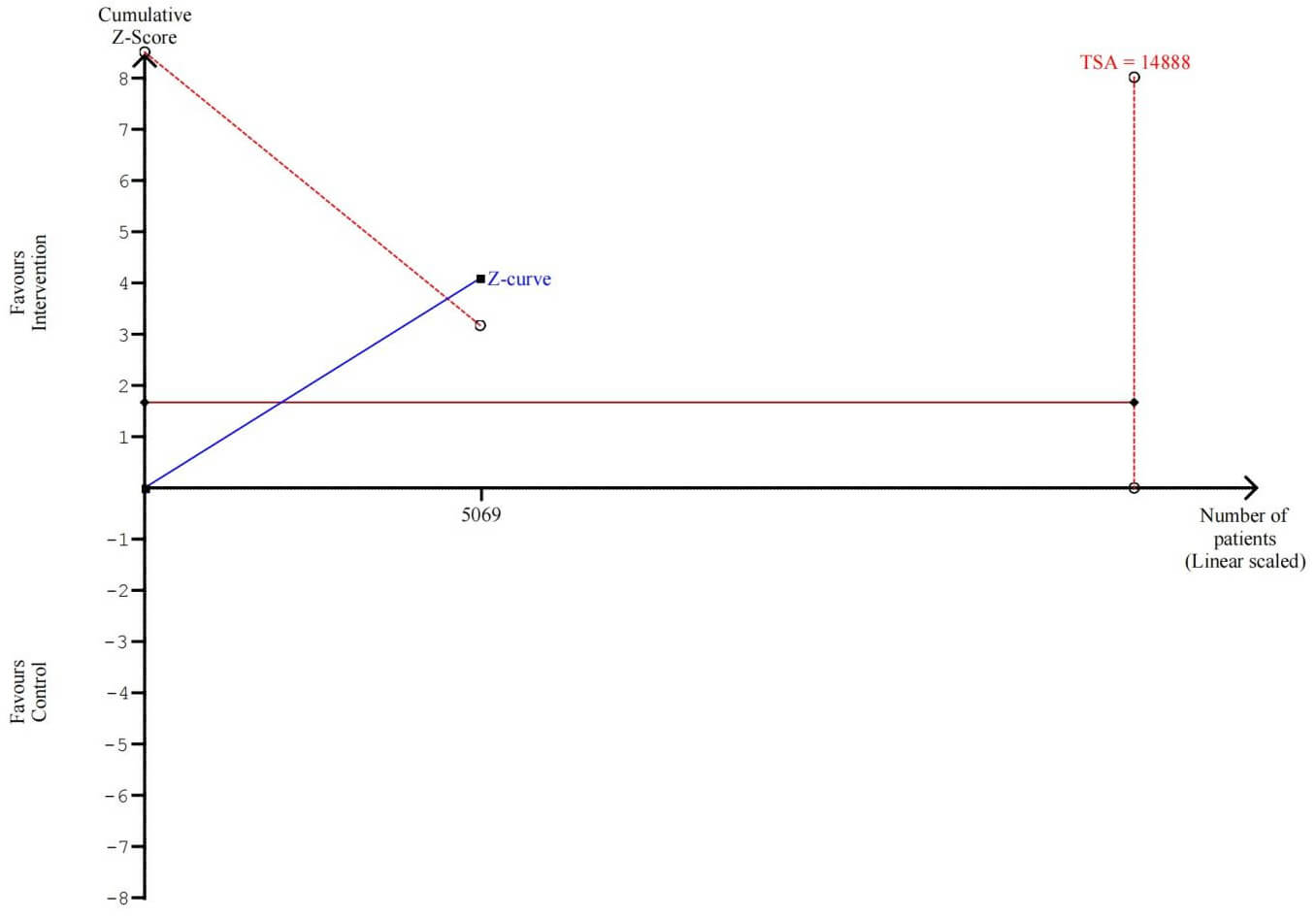 Fig. 6.
Fig. 6.
Trial Sequential Analysis for all-cause mortality. TSA, trial sequential analysis.
The nominal 16% reduction in all-cause mortality (RR 0.84, 95% CI 0.73–0.96) is consistent in direction with previous meta-analyses but must be interpreted with extreme caution [18, 19]. We contend that this ‘naïve’ pooled result is prone to overestimation and does not accurately reflect the intervention’s true clinical benefit. The core issue, illuminated by our sensitivity analysis, is that this mortality benefit disappears entirely when analysis is confined to the most methodologically sound trials (RR 0.90, 95% CI 0.79–1.03). We believe this non-significant finding from the high-quality studies represents the most credible estimate of effect.
This discrepancy strongly suggests that the observed benefit may be an artifact driven by older, smaller, open-label studies which are at high risk of performance bias [33]. In such trials, the “intensified care” effect—whereby patients and clinicians in the intervention arm, aware of the novel strategy, engage more intensively—may contribute more to improved outcomes than the biomarker guidance itself [34, 35]. This concept is supported by the GUIDE-IT trial, which failed to show a benefit, arguably because its control group also received highly structured, intensive clinical follow-up, thereby equalizing the intensity of care between groups [15, 36].
Our conclusion that the primary finding is a false positive (Type I error) is further strengthened by two key analyses. The detection of significant publication bias further weakens the evidence. The tendency for smaller studies with null or negative findings to remain unpublished can create a skewed and overly optimistic representation of an intervention’s efficacy in the published literature [37]. Furthermore, the TSA results provide the most compelling argument against the certainty of the findings, indicating that the cumulative evidence is underpowered and that the statistically significant result from the primary analysis is likely a false positive (Type I error) [38].
The moderate heterogeneity (I2 = 53.7%) for the hospitalization outcome likely stems from substantial clinical and methodological diversity across trials [39]. Key sources of heterogeneity include varying natriuretic peptide targets, heterogeneous patient populations (e.g., HFrEF vs. HFpEF, chronic vs. acute), and variable control arm care intensity [15, 40, 41]. The issue of “varying targets” is more problematic than it first appears. Natriuretic peptides are not intrinsically stable metrics. First, they fluctuate significantly within patients and between patients, driven by significant modulation by age, renal function, body mass index (BMI), and comorbidities. Second, different commercial assays produce different readings for the same sample, each with distinct analytical performance and reference ranges. This “noise” from both biological and analytical sources directly fuels what can be termed ‘threshold bias’. A review of Table 1 reveals this lack of consensus: targets ranged from absolute values to relative changes in others. This means the “intervention” was not a uniform strategy across trials. The therapeutic intensity required to meet these disparate goals varied dramatically. We argue this fundamental inconsistency, originating from the biomarker itself and amplified by trial design, is a major, unresolved driver of the heterogeneity we found. As HF is increasingly recognized as a collection of heterogeneous phenotypes, a “one-size-fits-all” biomarker-guided approach may be inherently flawed [42, 43]. Future strategies may need to be tailored to specific patient profiles, potentially integrating multiple biomarkers to capture different pathophysiological domains like inflammation, fibrosis, and renal dysfunction [44, 45].
Based on our comprehensive analysis and the resulting “very low” GRADE rating, as detailed in Table 2, the current evidence is insufficient to endorse the routine use of biomarker-guided therapy in clinical practice. The potential benefits do not yet outweigh the uncertainties and the additional resources required [46]. Our findings support the cautious stance of current international guidelines [6, 7].
| Outcome | Control group risk | Intervention group risk (95% CI) | Relative effect (95% CI) | Absolute effect (per 1000 people) | Quality of evidence (GRADE) |
| All-cause mortality | 214 per 1000 | 180 per 1000 (156 to 205) | RR 0.84 (0.73–0.96) | 34 fewer (9 fewer to 58 fewer) | |
| HF hospitalization | 312 per 1000 | 246 per 1000 (203 to 300) | RR 0.79 (0.65–0.96) | 66 fewer (12 fewer to 109 fewer) |
Abbreviations: GRADE, Grading of Recommendations, Assessment, Development and
Evaluation; RR, risk ratio; CI, confidence interval. GRADE Quality Rating:
The path forward requires a new generation of clinical trials that learn from the shortcomings of the past [47]. Future research should focus on: (1) Methodological rigor: To eliminate bias, conduct large-scale RCTs with blinded outcome adjudication [48]. (2) Patient selection: Focus on well-defined, high-risk subgroups (rather than broad HF populations) most likely to benefit, such as those with persistent congestion despite initial therapy [19, 49]. (3) Standardized protocols: Developing and validating clear, actionable, and standardized treatment algorithms linked to specific biomarker changes to ensure interventions are consistent and reproducible [50]. (4) Integration with modern therapies: Evaluating biomarker guidance in the context of contemporary GDMT, including SGLT2 inhibitors, which themselves profoundly impact natriuretic peptide levels and volume status [51, 52].
This review’s strengths include a comprehensive search method and the use of advanced statistical techniques, such as TSA and the GRADE framework, to critically appraise the evidence and estimate the certainty of the overall conclusions. However, the quality of the original research included in the analysis limits the conclusions. The identified risks of bias, severe publication bias, and statistical imprecision are major limitations.
In summary, the prospective benefit of biomarker-guided therapy in HF is suggested by a pooled analysis of existing RCTs; however, this conclusion is based on very low-quality evidence and lacks robustness. Prevalent methodological flaws, statistical imprecision, and a high risk of publication bias erode confidence in the effect estimate. This combination of factors leads to our conclusion that the current evidence is insufficient to support the routine implementation of this strategy. There is a clear and urgent need for large-scale, methodologically rigorous RCTs to definitively define the role, if any, of biomarker-guided therapy in contemporary HF management.
All data generated or analyzed during this study are included in this published article.
HZ, TL, FL, KL, XW, and YX made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or critically revising it for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by the Key R&D Project of Sichuan Science and Technology Planning Project (Grant No. 2022YFS0356).
The authors declare no conflict of interest. The funder had no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM46184.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






