1 Medical College of Yangzhou University, 225001 Yangzhou, Jiangsu, China
2 Department of Cardiology, The Affiliated Taizhou People’s Hospital of Nanjing Medical University, 225300 Taizhou, Jiangsu, China
3 Department of General Surgery, The Hanjiang District Maternal and Child Health Care Hospital, 225100 Yangzhou, Jiangsu, China
4 Hubei University of Medicine, 442000 Shiyan, Hubei, China
†These authors contributed equally.
Abstract
Sodium–glucose cotransporter 2 (SGLT2) inhibitors, employed as antidiabetic agents, have been shown to effectively improve the prognosis of patients with chronic and stable heart failure, chronic kidney disease, and diabetes in the context of cardiovascular–renal–endocrine integrated management. However, the safety and clinical benefits of the early application of SGLT2 inhibitors in hospitalized patients with acute heart failure remain controversial. This study aimed to evaluate the safety and prognostic impact of early SGLT2 inhibitor therapy in patients with acute heart failure.
A systematic literature search of the PubMed, Web of Science, and Cochrane Library databases was conducted to identify studies on the use of SGLT2 inhibitors in acute heart failure. Two researchers independently screened studies, extracted data, and assessed the risk of bias in the included studies. The meta-analysis was performed using STATA 16.0 software (StataCorp, College Station, TX, USA).
A total of 23 studies involving 47,291 patients with acute heart failure were included in this analysis (10 randomized controlled trials and 13 observational studies). Early use of SGLT2 inhibitors in hospitalized patients with acute heart failure was associated with a reduction in the incidence of composite events in the short term (relative risk (RR) = 0.64, 95% confidence interval (CI) (0.56, 0.74)), all-cause mortality (RR = 0.72, 95% CI (0.60, 0.86)), and heart failure rehospitalization rates (RR= 0.77, 95% CI (0.63, 0.87)); however, the early use of SGLT2i did not improve the incidence of cardiogenic death (RR = 0.74, 95% CI (0.51, 1.08)). Additionally, the early administration of SGLT2 inhibitors significantly reduced the incidence of cardiogenic mortality (RR = 0.77, 95% CI (0.60, 1.0); p = 0.045), as well as decreasing heart failure rehospitalization rates (RR = 0.77, 95% CI (0.70, 0.86)) and all-cause mortality (RR = 0.49, 95% CI (0.41, 0.60)), without increasing the incidence of adverse drug reactions such as acute kidney injury, urinary tract infections, diabetic ketoacidosis, hypoglycemia, or hypotension.
Early in-hospital use of SGLT2 inhibitors can safely and effectively reduce the incidence of all-cause mortality, cardiogenic rehospitalization, and composite events in acute heart failure patients in both the short term and over one year.
Keywords
- sodium-glucose cotransporter-2 inhibitors
- acute heart failure
- heart failure
- prognosis
Acute heart failure (AHF) is a clinical syndrome characterized by a sudden onset
or exacerbation of left ventricular dysfunction, leading to decreased myocardial
contractility and increased cardiac load, resulting in a rapid decline in acute
cardiac output [1]. Globally, the prevalence of heart failure (HF) continues to
rise and has become one of the most common causes of hospitalization in
individuals aged 65 and older. Despite modern treatment options, the in-hospital
mortality rate for AHF patients remains at 8–10%, with a 30-day readmission
rate reaching 25%, thus posing a significant public health burden [2].
Therefore, recent research has focused on multi-target drugs that can effectively
improve prognosis. Sodium-glucose co-transporter 2 inhibitors (SGLT2i) are a
novel class of oral hypoglycemic agents that primarily target the SGLT2 receptors
in the renal proximal tubules, inhibiting glucose reabsorption [3]. Recent
clinical randomized trials have demonstrated that SGLT2i have beneficial effects
in cardiovascular and renal diseases, and this effect extends to non-diabetic
populations. The 2023 European Society of Cardiology (ESC) guidelines included
SGLT2i as one of the four cornerstone therapies for heart failure with reduced
ejection fraction (HFrEF (Heart Failure with Reduced Ejection Fraction), left
ventricular ejection fraction (LVEF)
This study was designed and conducted under the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines for the systematic evaluation and Meta-analysis.
Using English keywords and their synonyms (acute heart failure) AND (Sodium glucose transporter 2 inhibitor OR SGLT-2 inhibitor) for a combined search in PubMed, Web of Science, and Cochrane Library, limited to English-language literature, with the search period from October 2013 to June 2025. Secondary screening of the literature will be performed through reading the articles.
(1) Randomized controlled trials or retrospective studies. (2) Study population consisting of patients diagnosed with acute heart failure who require pharmacological treatment. The control group should consist of patients receiving conventional drug therapy/SLGT-2 inhibitor therapy after discharge/placebo therapy, while the study group should involve patients who start using SGLT-2 inhibitors upon hospitalization. (3) Studies must provide clear information on patient demographics, follow-up duration, and related endpoint events.
(1) Sample size (patients receiving SGLT-2 inhibitors) less than 10. (2) Duplicate publications. (3) Article types: conference abstracts, reviews, commentaries, case reports, or studies involving cells or animals. (4) Studies that do not provide detailed information about the study population, endpoint event definitions, and major adverse cardiovascular events (MACEs) occurrence, and where contacting the authors for further information is not possible.
Two researchers will independently extract data from studies that meet the inclusion and exclusion criteria, then cross-check the extracted information. The data to be extracted includes:
(1) Basic study information: first author, publication year, inclusion criteria, etc. (2) Basic information on enrolled patients: number of patients, age, gender, relevant medical history, follow-up duration, etc. (3) Outcome measures: short-term/medium- to long-term major adverse cardiovascular events (MACEs) and drug-related adverse events timeline. (4) Bias risk assessment-related elements.
Stata 16.0 software (StataCorp, College Station, TX, USA) will be used to
conduct a meta-analysis of the length of hospital stay, short-term/medium- to
long-term major cardiovascular adverse events, and drug-related adverse event
timelines for studies included. Relative risk (RR) will be used to assess the
differences in the occurrence of clinical events between different treatment
regimens. I2 statistics will be used to assess the heterogeneity of the
studies. I2 values of 75%, 50%, and 25% represent high, medium, and low
heterogeneity, respectively. If there is no statistical heterogeneity between the
studies, a fixed-effect model will be used for meta-analysis. If heterogeneity
exists, subgroup analysis, sensitivity analysis, and trim-and-fill method will be
used to further explore the sources of heterogeneity. Funnel plots and Egger’s
test will be used to assess publication bias. A p-value of
Literature inclusion status: A total of 1145 articles were retrieved, including 429 from PubMed, 584 from Web of Science, and 132 from Cochrane Library. After excluding 378 duplicate articles, 258 articles were preliminarily screened by reading the titles and abstracts. Following a detailed review of the full texts of the initially screened articles, 23 articles were ultimately included. Among these, 13 observational studies [6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18] and 10 randomized controlled trials [19, 20, 21, 22, 23, 24, 25, 26, 27, 28] were selected. The specific search process is shown in Fig. 1. The evaluation process was carried out by two researchers separately and cross-checked. The bias in the included articles was evaluated using the quality assessment criteria from the Cochrane Handbook and The Newcastle-Ottawa Scale, as seen in the Supplementary Table 1 and Supplementary Fig. 1.
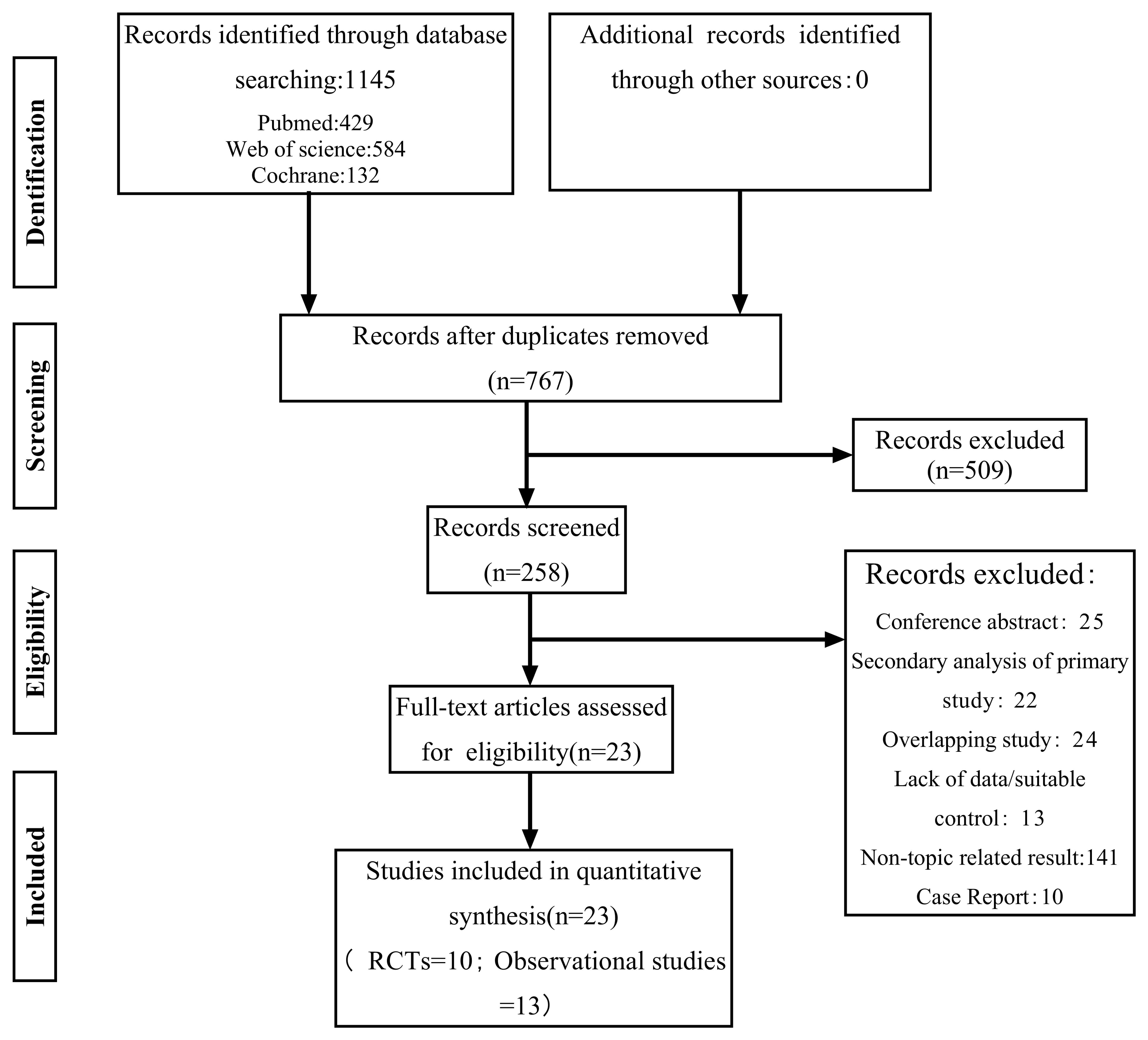 Fig. 1.
Fig. 1.
Study screening procedures and outcomes. RCTs, randomized controlled trials.
This study included 23 studies involving a total of 47,261 acute heart failure (AHF) patients, among whom 7294 received early SGLT2 inhibitor (SGLT2i) treatment, and 39,967 received standard heart failure treatment, delayed SGLT2i, or placebo. The baseline characteristics of all included patients are shown in Table 1 (Ref. [6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28]).
| Author | Year | Sample size | Age | Male | Diabetes mellitus | NYHA functional class | Duration of SGLT2i use and dosage | |||||
| (N (%)) | N (%) | N (%) | III-IV | |||||||||
| SGLT2 | NO-SGLT2 | SGLT2 | NO-SGLT2 | SGLT2 | NO-SGLT2 | SGLT2 | NO-SGLT2 | SGLT2 | NO-SGLT2 | SGLT2 | ||
| Llorens P et al. [6] | 2025 | 366 | 2693 | 81 (72−88) | 86 (79−90) | 193 (52.7) | 1085 (40.3) | 178 (49.6) | 931 (34.9) | 50 (16.5) | 428 (18.6) | before discharge |
| Park S et al. [7] | 2023 | 818 | 28,472 | 69.1 (12.2) | 74.2 (10.7) | 406 (49.6) | 12,445 (43.7) | all | all | 225 (27.5) | 9437 (33.1) | before discharge |
| Pérez-Belmonte LM et al. [8] | 2022 | 99 | 109 | 84.5 |
86.4 |
54 (54.5) | 50 (45.9) | all | all | 40 | 41 | before discharge |
| Matsukawa R et al. [9] | 2023 | 92 | 76 | 71.4 |
72.0 |
66 (71.7) | 53 (69.7) | all | all | 75 | 67 | within 48 hours of admission |
| Nakagaito M et al. [10] | 2021 | 56 | 30 | 69.6 |
75.8 |
38 (68) | 17 (57) | all | all | / | / | before discharge |
| Echeverría LE et al. [11] | 2025 | 1275 | 395 | 68.0 (59.0–76.0) | 69.0 (58.5–77.0) | 384 (30.1) | 84 (24.3) | 222 (17.4) | 73 (18.5) | 855 (67.1) | 237 (60.0) | within 48 hours of admission |
| Calcagno T et al. [12] | 2025 | 2043 | 2043 | 68.1 |
68.5 |
928 (45.4) | 929 (45.5) | 1649 (80.7) | 1661 (81.3) | before discharge | ||
| Kambara T et al. [13] | 2019 | 12 | 19 | 73 |
75 |
9 (75) | 14 (73) | all | all | 10 | 17 | within 24 hours of admission |
| Burgos LM et al. [14] | 2024 | 237 | 141 | / | / | / | / | / | / | / | / | before discharge |
| Amioka M et al. [15] | 2025 | 163 | 198 | 82.5 |
83.3 |
90 (55.2) | 103 (52) | 68 (41.7) | 71 (35.9) | 79 (48.5) | 98 (49.5) | within 48 hours of admission |
| Aklilu AM et al. [16] | 2023 | 356 | 2949 | 69.5 (60.4–78.5) | 78.7 (68.1–87.3) | 219 (61.5) | 1428 (48.4) | 241 (67.7) | 1470 (49.8) | / | / | within 10 days of admission |
| Guzmán-Carreras A et al. [17] | 2024 | 210 | 540 | 82 |
84 |
102 (48.6) | 215 (39.8) | 132 (62.9) | 213 (39.4%) | 91 | 212 | before discharge |
| Wu D et al. [18] | 2024 | 206 | 193 | 75.00 (60.75, 79.25) | 75.00 (67.00, 82.00) | 102 (49.51) | 96 (49.74) | 102 (49.51) | 89 (46.11) | 160 | 180 | within 6 days of admission |
| El-Gazar RA et al. [19] | 2025 | 71 | 71 | 55 (40–63) | 56 (48–61) | 54 (76.1) | 48 (67.6) | 30 (42.3) | 35 (49.3) | 70 | 67 | within 24 hours of admission; 100 mg/day of canagliflozin |
| Emara AN et al. [20] | 2023 | 45 | 42 | 63.9 (10) | 61.1 (11.8) | 35 (77.8) | 27 (64.3) | 16 (35.6) | 22 (52.4) | / | / | |
| Cox ZL et al. [21] | 2024 | 119 | 119 | 65 (56–73) | 64 (55–74) | 78 (66) | 67 (56) | 84 (71) | 85 (71) | / | / | within 24 hours of admission; Dapagliflozin 10 mg/day |
| Schulze PC et al. [22] | 2022 | 30 | 29 | 72.9 |
76.5 |
19 (63.3) | 17 (58.6) | 13/30 (43.3) | 10/29 (34.5) | 27 | 24 | within 24 hours of admission; Empagliflozin 10 mg/day |
| Damman K et al. [23] | 2020 | 40 | 39 | 79 (73–83) | 73 (61–83) | 24 (60) | 29 (74.4) | 38 | 28 | 92 | 97 | within 24 hours of admission; Empagliflozin 10 mg/day |
| Voors AA et al. [24] | 2022 | 265 | 265 | 71 (62–78) | 70 (59–78) | 179 (67.5) | 172 (64.9) | 124 (46.8) | 116 (43.8) | 160 | 168 | within 24 hours of admission; Empagliflozin 10 mg/day |
| Charaya K et al. [25] | 2022 | 50 | 52 | 72.6 |
74.2 |
29 (58) | 27 (52) | 15 (30) | 16 (30) | 16 (34) | 23 (44) | within 24 hours of admission; Empagliflozin 10 mg/day |
| Ibrahim A et al. [26] | 2020 | 50 | 50 | 62.02 |
60.64 |
28 (56) | 26 (52) | all | all | / | / | within 24 hours of admission; Dapagliflozin 10 mg/day |
| Bhatt DL et al. [27] | 2021 | 608 | 614 | 69 (63–76) | 70 (64–76) | 410 (67.4) | 400 (65.1) | / | / | / | / | within 3 days before or after discharge; sotagliflozin 200 mg/day |
| López-Vilella R et al. [28] | 2022 | 83 | 453 | 72.4 |
73.4 |
53 | 256 | 54 | 140 | 13 | 72 | before discharge |
SGLT2, Sodium–glucose cotransporter 2; NYHA, New York Heart Association.
The short-term composite event incidence was included in 7 studies [6, 7, 8, 9, 14, 19, 23]. These studies exhibited high heterogeneity (I2 = 78.6%). Through sensitivity analysis, we found that the study by Pérez-Belmonte LM [8] had a potential risk of bias (Supplementary Fig. 2). After excluding this study, we found that early use of SGLT2 inhibitors could reduce the incidence of short-term composite events in patients [RR = 0.64, 95% CI (0.56, 0.74)] (Fig. 2A).
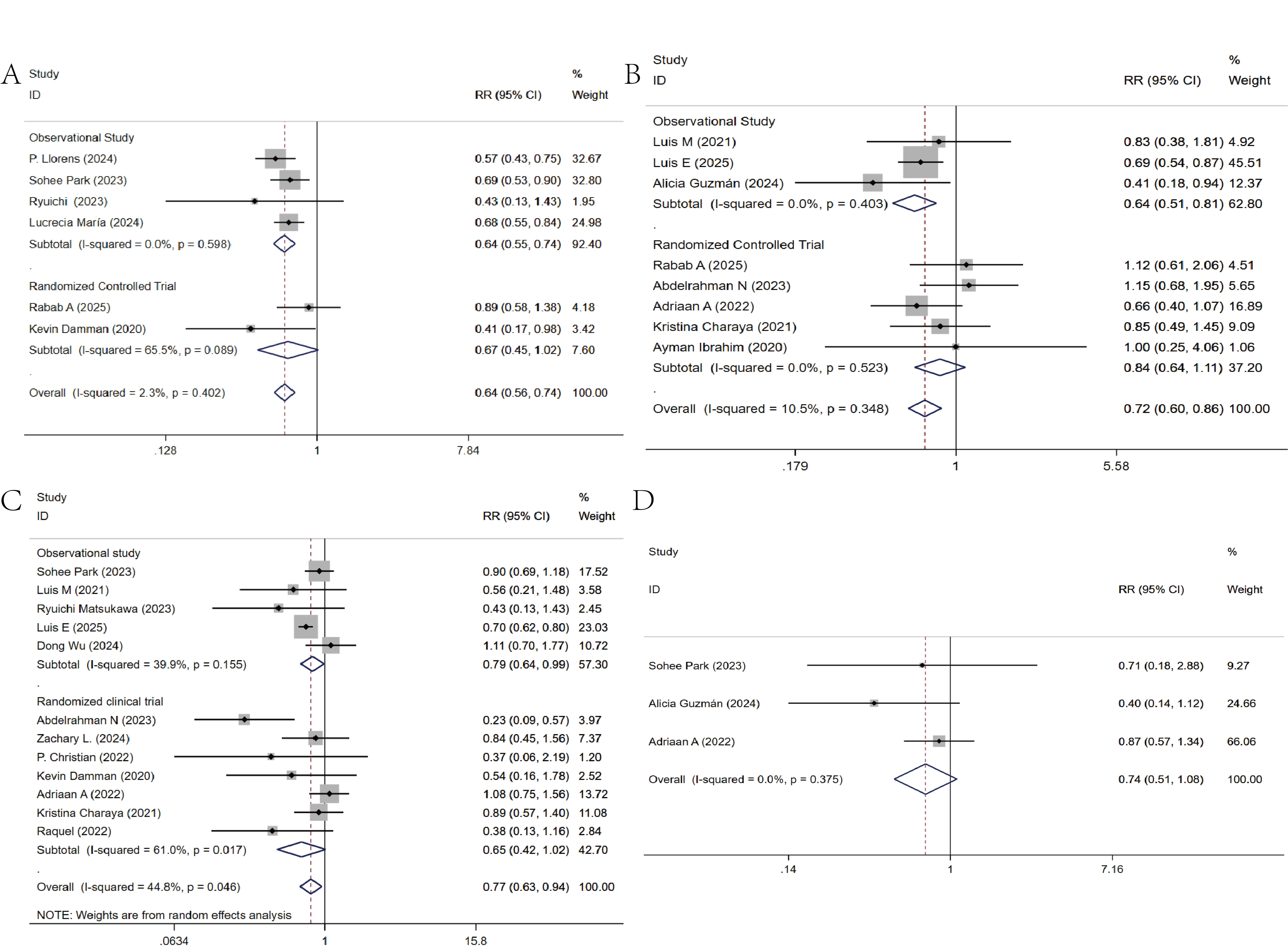 Fig. 2.
Fig. 2.
Meta-analysis of short-term prognostic impacts. (A) Composite event rates. (B) All-cause mortality. (C) Heart failure rehospitalization rates. (D) Cardiovascular mortality. CI, confidence interval; RR, relative risk.
The all-cause mortality rate in the short term was included in 13 studies [8, 11, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26]. These studies exhibited high heterogeneity (I2 = 83.4%). Through sensitivity analysis, we found that the studies by DONG Wu [18] and Aklilu AM [16] had significant risk of bias (Supplementary Fig. 3). After excluding these two studies, we found that early use of SGLT2 inhibitors could reduce the all-cause mortality rate in the short term [RR = 0.72, 95% CI (0.60, 0.86)] (Fig. 2B).
The incidence of heart failure-related rehospitalization in the short term was included in 12 studies [7, 8, 9, 11, 18, 20, 21, 22, 23, 24, 25, 28]. These studies exhibited moderate heterogeneity (I2 = 44.8%). Using a random-effects analysis, we found that early use of SGLT2 inhibitors could reduce the incidence of heart failure-related rehospitalization in the short term [RR = 0.77, 95% CI (0.63, 0.94)] (Fig. 2C).
The short-term cardiogenic mortality rate was included in three studies [7, 17, 24], and there was no heterogeneity (I2 = 0%). Fixed-effect analysis showed that early use of SGLT2 inhibitors did not affect short-term cardiogenic mortality [RR = 0.74, 95% CI (0.51, 1.08), p = 0.121] (Fig. 2D).
The specific analysis results are shown in Table 2.
| Heterogeneity test | META analysis | ||||
| I2 | p | RR 95% CI | Z | p | |
| Composite event rates | 2.3% | 0.402 | 0.64 (0.56, 0.74) | 6.2 | |
| All-cause mortality | 10.5% | 0.348 | 0.72 (0.60, 0.86) | 3.66 | |
| Heart failure rehospitalization rates | 44.8% | 0.046 | 0.77 (0.63, 0.94) | 2.54 | 0.011 |
| Cardiovascular mortality | 0% | 0.375 | 0.74 (0.51, 1.08) | 1.55 | 0.121 |
For the incidence of composite events within 1 year, four studies were included [7, 12, 15, 27], with significant heterogeneity among the studies (I2 = 78.9%, p = 0.003). Sensitivity analysis showed no significant reduction in heterogeneity after excluding any single study (Supplementary Fig. 4). A random-effects model analysis revealed that early use of SGLT2 inhibitors significantly reduced the incidence of composite events within 1 year in patients [RR = 0.74, 95% CI (0.63, 0.87)], (Fig. 3A).
 Fig. 3.
Fig. 3.
Meta-analysis of one-year prognostic impact. (A) Composite event rates. (B) Cardiovascular mortality. (C) Heart failure rehospitalization rates. (D) All-cause mortality.
For the incidence of cardiogenic mortality within 1 year, four studies were included [7, 10, 15, 27], with no heterogeneity (I2 = 0%). A fixed-effects model analysis showed that early use of SGLT2 inhibitors significantly reduced the cardiogenic mortality rate within 1 year [RR = 0.84, 95% CI (0.70, 0.99)], (Fig. 3B).
For the heart failure rehospitalization rate within 1 year, five studies were included [7, 10, 12, 15, 27], with moderate heterogeneity among the studies (I2 = 31.3%). A random-effects model analysis revealed that early use of SGLT2 inhibitors significantly reduced the heart failure rehospitalization rate within 1 year [RR = 0.77, 95% CI (0.70, 0.86)], (Fig. 3C).
For all-cause mortality within 1 year, three studies were included [6, 12, 17], with significant heterogeneity among the studies (I2 = 36.1%). A random-effects model analysis showed that early use of SGLT2 inhibitors significantly reduced all-cause mortality within 1 year [RR = 0.49, 95% CI (0.41, 0.60)], (Fig. 3D).
The specific analysis results are shown in Table 3.
| Heterogeneity test | META analysis | ||||
| I2 | p | RR 95% CI | Z | p | |
| Cardiovascular mortality | 0% | 0.595 | 0.84 (0.70, 1.0) | 1.95 | 0.05 |
| Heart failure rehospitalization rates | 31.3% | 0.393 | 0.77 (0.70, 0.86) | 4.99 | |
| All-cause mortality | 36.1% | 0.209 | 0.49 (0.41, 0.60) | 7.14 | |
Regarding the safety of SGLT2i in AHF patients, we compared the most common adverse drug events and found that SGLT2i did not increase the incidence of acute kidney injury [19, 20, 21, 22, 23, 24, 25, 27], (Fig. 4A), urinary tract infections [19, 22, 23, 24, 27], (Fig. 4B), ketoacidosis [20, 21, 22, 23, 28], (Fig. 4C), hypotension [19, 20, 21, 22, 25, 27], (Fig. 4D), or hypoglycemia [19, 20, 21, 24, 27], (Fig. 4E) (Table 4).
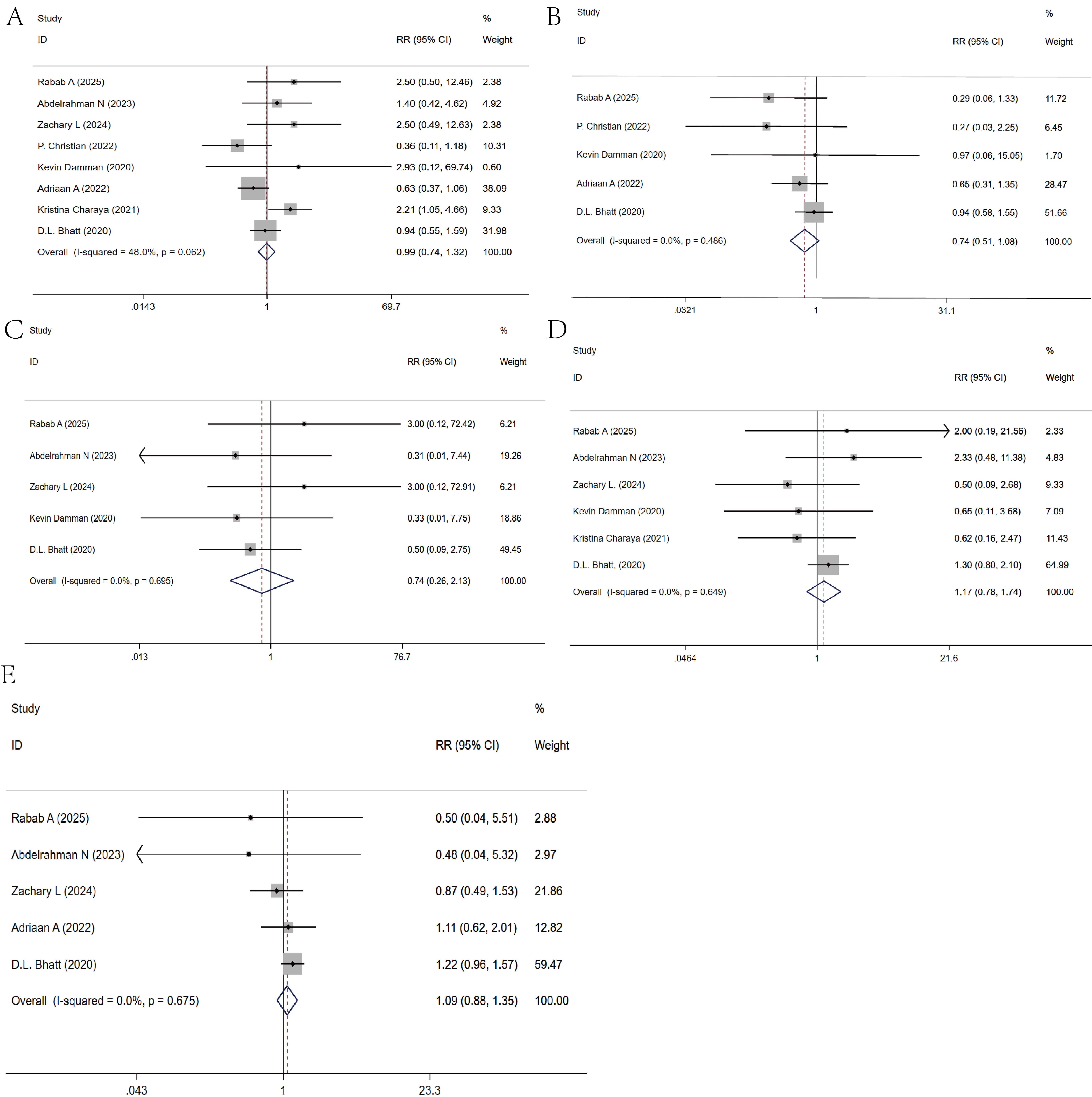 Fig. 4.
Fig. 4.
Meta-analysis of drug safety. (A) Acute kidney injury. (B) Urinary tract infections. (C) Ketoacidosis. (D) Hypotension. (E) Hypoglycemia.
| Heterogeneity test | META analysis | ||||
| I2 | p | RR 95% CI | Z | p | |
| Acute kidney injury | 48% | 0.062 | 0.99 (0.74, 1.32) | 0.1 | 0.924 |
| Urinary tract infections | 0% | 0.468 | 0.74 (0.51, 1.08) | 1.56 | 0.119 |
| Ketoacidosis | 0% | 0.695 | 0.74 (0.26, 2.13) | 0.55 | 0.581 |
| Hypotension | 0% | 0.649 | 1.17 (0.78, 1.74) | 0.76 | 0.446 |
| Hypoglycemia | 0% | 0.723 | 1.08 (0.88, 1.35) | 0.77 | 0.441 |
Bias assessment of the above results indicated a low likelihood of bias, confirming the robustness of the meta-analysis findings. See Supplementary Figs. 5,6.
This may be the largest meta-analysis and systematic review to date in terms of the number of studies included and sample size. It comprehensively explores and demonstrates that the early use of SGLT2i in-hospital can safely and effectively reduce the incidence of composite events, such as all-cause mortality and cardiogenic rehospitalization, both in the short term and within 1 year, in patients with acute heart failure (AHF). This suggests potential benefits in improving patient prognosis.
Initially, SGLT2i were introduced as a treatment for diabetes. However, with the emergence of numerous randomized controlled trials and evidence from evidence-based medicine, the clinical indications for SGLT2i have expanded to include non-diabetic populations and other diseases. The DELIVER study highlighted that SGLT2i significantly reduced the risk of major composite outcomes in patients with chronic heart failure (HF), including 18% of those with HF with improved ejection fraction (HFimpEF) [HR 0.74, 95% CI (0.41–0.96)], and reduced the risk of the first heart failure worsening event by 22% [HR 0.78, 95% CI (0.61–1.14)] [29]. Following this, some researchers hypothesized that SGLT2i might also provide clinical benefits in AHF. The EMPULSE study was the first to confirm this hypothesis, showing that, compared to placebo, administration of dapagliflozin 10 mg within 3 days of hospitalization for newly diagnosed AHF significantly improved 90-day clinical outcomes in AHF patients (stratified win ratio: 1.36; 95% CI: 1.09–1.68; p = 0.0054), including improvements in the Kansas City Cardiomyopathy Questionnaire (KCCQ) total symptom score, time to all-cause mortality, or first heart failure event [24]. However, the DAPA-RESPONSE-AHF study pointed out that within 24 hours of hospitalization, randomization to dapagliflozin reduced rehospitalization within 30 days after discharge, but had no impact on the incidence of heart failure worsening or mortality during hospitalization [20]. Additionally, potential adverse effects of SGLT2 inhibitors may also affect the safety of AHF patients. Due to the complex physiological status of patients in the acute phase, common side effects such as hypoglycemia, dehydration, and further renal function impairment may pose a greater risk to these patients [30]. A study by Voors AA et al. [31] noted that SGLT2 inhibitors may cause early renal impairment in AHF patients, though this damage was no longer significant after 90 days, with similar rates of acute kidney events between groups. However, this has led to concerns regarding the early use of SGLT2 inhibitors in AHF patients. Therefore, SGLT2 inhibitors still require more evidence from randomized controlled trials and further evidence-based studies.
A meta-analysis conducted by Carvalho PEP et al. [32] summarized the treatment effects of
SGLT2i within 30 days of acute AHF onset. It found that compared to traditional
treatment groups, SGLT2i reduced all-cause mortality (OR: 0.75; 95% CI
0.56–0.99; p = 0.049), heart failure (HF) readmission (OR: 0.54; 95%
CI 0.44–0.66; p
Although existing evidence confirms that early use of SGLT2i can safely and effectively yield clinical benefits for AHF patients in the short and long term, some issues remain unresolved. First, the mechanisms underlying these benefits are still unclear. Kasperova BJ et al. [33] have pointed out that SGLT-2 inhibitors significantly downregulate pro-inflammatory gene expression in epicardial adipose tissue (EAT) and reduce macrophage and T lymphocyte infiltration in EAT, thereby decreasing oxidative stress and ferroptosis, which protects the myocardium. However, the protective mechanism of SGLT2i in AHF remains uncertain. Second, the optimal timing for early use of SGLT2i is still undefined. Research by Echeverría LE et al. [11] has suggested that administering SGLT2i within 48 hours of AHF hospitalization was associated with a reduction in in-hospital mortality (RR 0.37; 95% CI, 0.17–0.77), shorter hospital stay, and lower 30-day composite mortality/HF readmission rates (RR 0.72; 95% CI, 0.53–0.98). Our study mainly compared the clinical benefits of early inpatient use versus no use or post-discharge use, but the exact timing (immediately, 24 hours, or 48 hours after admission) for optimal benefit in AHF patients still requires further clinical validation. Lastly, more subgroup analyses are needed to investigate the use of SGLT2i in different populations, such as those with or without diabetes, hypertension, renal insufficiency, and gender differences. Currently, the latest guidelines for AHF recommend that SGLT2i treatment should be initiated in AHF patients either before discharge or early after discharge. Additionally, the DAPA ACT HF-TIMI 68 Trial, the largest randomized controlled trial to date, will soon release its follow-up results, and we look forward to its findings, which may provide further evidence for the early use of SGLT2i in AHF patients [34].
(1) Since only one study reported the improvement of quality of life with SGLT2 inhibitors, this study did not perform a comparative analysis on the impact of early use of SGLT2 inhibitors on quality of life.
(2) Due to the differences in the definition of “early” treatment across studies, this research did not define or categorize specific time points for early treatment.
(3) Due to differences in study populations, objectives, and the lack of specific individualized data, this study only performed subgroup analysis based on study type. Future randomized controlled trials targeting specific populations, heart failure classifications, and treatment duration are anticipated.
(4) This study only included relevant English-language literature, which may have excluded studies published in other languages.
In summary, early use of SGLT2i effectively reduces the incidence of all-cause mortality, cardiogenic readmission, and other composite events in AHF patients over the short term and up to one year, without increasing the occurrence of adverse drug events such as hypoglycemia, hypotension, renal impairment, or ketoacidosis. However, further high-quality and large-scale clinical evidence is needed to confirm its hemodynamic effects and clinical benefits in non-diabetic populations.
All data in this study are available from the original cited studies.
YFD is responsible for the organization, verification, and writing of the relevant literature data for this article. YM and HL each completed literature retrieval, data organization, statistical analysis, and revisions of the article. LZ is responsible for the conception of the study, drafting the manuscript, and making substantial revisions to its content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This study was financially supported by Postgraduate Research & Practice Innovation Program of Jiangsu Province (NO: SJCX24-2350).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM45590.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




