1 Peripheral Vascular Ward (Cardiac Surgery Ward 1), Fuwai Hospital Chinese Academy of Medical Sciences, 518001 Shenzhen, Guangdong, China
2 Department of Vascular Surgery, State Key Laboratory of Cardiovascular Disease, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, 100037 Beijing, China
†These authors contributed equally.
Abstract
Thoracic endovascular aortic repair (TEVAR) in Zone 2 frequently necessitates coverage of the isolated left vertebral artery (ILVA), a congenital vascular anomaly, to ensure adequate proximal sealing. However, the clinical requirement of ILVA revascularization remains uncertain. Thus, this study aimed to compare the outcomes between ILVA coverage and fenestration during Zone 2 TEVAR.
We retrospectively analyzed the clinical records of patients with ILVA who underwent Zone 2 TEVAR between September 2010 and August 2023. Patients were divided into two groups: Coverage Group (n = 23) and Fenestration Group (n = 33). Baseline characteristics, surgical outcomes, and changes in left and right vertebral artery diameters pre- and postoperatively were compared. Continuous variables were compared using Student's t-test or Mann-Whitney U test, depending on the distribution. Categorical variables were analyzed using the chi-square test or Fisher's exact test.
The overall cohort had a mean age of 54.48 ± 10.31 years, with 89.29% of participants male and a mean body mass index (BMI) of 25.88 ± 3.5 kg/m2. The Fenestration Group was significantly older than the Coverage Group (56.82 ± 8.78 vs. 51.13 ± 11.56; p = 0.04). Technical success of the TEVAR was achieved in both groups in 98.21% of cases, with no perioperative mortality. Simultaneous left subclavian artery stenting was performed more frequently in the Fenestration Group (57.58% vs. 21.74%; p = 0.008). At discharge, patients in the Coverage Group demonstrated a significantly greater reduction in left vertebral artery diameter compared with the Fenestration Group (13.64% [5.52%, 22.4%] vs. 0 [–3.29%, 5.13%]; p < 0.001). The incidence of vertebral artery diameter reduction was significantly higher in the Coverage Group compared with the Fenestration Group (39.13% vs. 6.06%; p < 0.01). Follow-up computed tomography angiography demonstrated a greater reduction in left vertebral artery diameter in the Coverage Group (52.94% vs. 14.29%; p = 0.020), while occlusion rates were comparable between groups (29.41% vs. 4.76%; p = 0.070).
Fenestration is associated with a lower incidence of postoperative ILVA diameter reduction compared with direct coverage during Zone 2 TEVAR. These findings highlight the potential benefit of ILVA revascularization and underscore the need for further validation in larger studies.
Keywords
- isolated left vertebral artery
- physician-modified fenestration
- aortic arch
- thoracic endovascular aortic repair
Open surgical repair remains the gold standard for the treatment of aortic arch pathologies, particularly in patients with low-to-moderate surgical risk, where it is strongly recommended. However, for patients with high surgical risk, hybrid procedures or thoracic endovascular aortic repair (TEVAR) are considered reasonable alternatives and are classified as Class IIb recommendations [1]. With ongoing advancements in endovascular techniques and device design, TEVAR has been increasingly adopted in the treatment of aortic arch pathologies [2]. In TEVAR involving the aortic arch, effective proximal sealing frequently requires intentional coverage of one or more supra-aortic branches. In Zone 2 TEVAR, achieving an adequate proximal landing zone often necessitates coverage of the left subclavian artery (LSA). Additionally, anatomical variants, such as an isolated left vertebral artery (ILVA), may be encountered in patients undergoing TEVAR for aortic arch pathologies [3].
Historically, the ILVA was often covered during TEVAR without careful consideration of its contribution to cerebral circulation. However, recent studies have increasingly emphasized the importance of ILVA preservation, given its critical role in maintaining posterior circulation through the circle of Willis. Although several surgical strategies have been proposed to preserve the ILVA, the impact of physician-modified fenestration (PMF) on ILVA preservation and clinical outcomes remains insufficiently characterized [4]. Therefore, the objective of this study was to assess the clinical significance of ILVA preservation during Zone 2 TEVAR and its association with postoperative changes in vertebral artery diameter.
This retrospective, single-center study included patients with aortic arch pathologies who underwent TEVAR at our institution between September 2010 and August 2023. Inclusion criteria were: (1) diagnosis of aortic arch pathology, including but not limited to acute aortic syndrome (AAS), aortic aneurysm, or related conditions; (2) planned Zone 2 TEVAR with coverage of the LSA, potentially involving coverage of the ILVA; and (3) preoperative computed tomography angiography (CTA) confirming the presence of an ILVA. Exclusion criteria were: (1) poor-quality CTA imaging that could not be reliably evaluated, and (2) patients deemed unsuitable for TEVAR due to other medical reasons. AAS refers to a spectrum of life-threatening conditions, including aortic dissection, intramural hematoma, and penetrating aortic ulcers, which are recognized indications for TEVAR under current clinical guidelines [2, 5]. This study was approved by the Ethics Committee of Fuwai Hospital. Owing to the retrospective design, the requirement for informed consent was waived.
Two experienced radiologists independently measured vascular diameters using
Endosize software (Therenva, Rennes, France). To minimize measurement errors,
diameters were assessed 2 cm from the origin of both vertebral arteries.
Postoperative occlusion of the isolated left vertebral artery was defined as the
absence of intraluminal blood flow following TEVAR. Postoperative stenosis of the
isolated left vertebral artery was defined as a
All procedures were performed under general anesthesia. Standard disinfection
and draping were applied to the bilateral femoral and left brachial artery
regions. A 5F gold marker pigtail catheter was introduced through the femoral
artery into the ascending aorta for digital subtraction angiography (DSA) (Fig. 1A). Measurements of the aortic lesion and its morphological characteristics were
obtained from CTA and DSA images. The aortic stent graft was selected with a
diameter oversized by 15–20% relative to the measured aortic diameter. Systemic
heparinization was administered in all patients. An Ankura thoracic
aortic-covered stent (Lifetech Scientific Co., Ltd., Shenzhen, Guangdong, China) was used in
every procedure. This device features a longitudinally distributed metallic
support structure that provides additional reinforcement along the greater
curvature of the aorta and incorporates a radiopaque marker to delineate the
transition between the bare metal and covered segments. When aligned
perpendicularly to the X-ray beam, the marker appears as a “
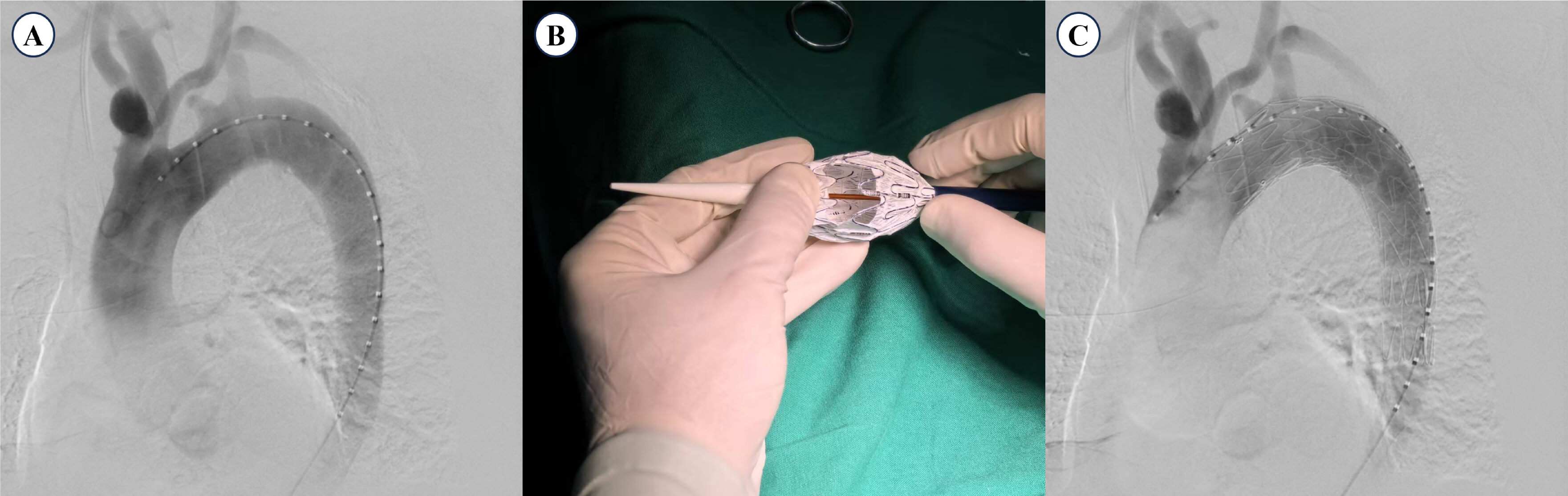 Fig. 1.
Fig. 1.
Procedural steps for the PMF technique in TEVAR. (A)
Preoperative DSA showing aortic pathology and vertebral artery anatomy. (B)
Fenestration of the ILVA and LSA openings performed with electrocautery, guided
by the “
Upon confirmation of the “
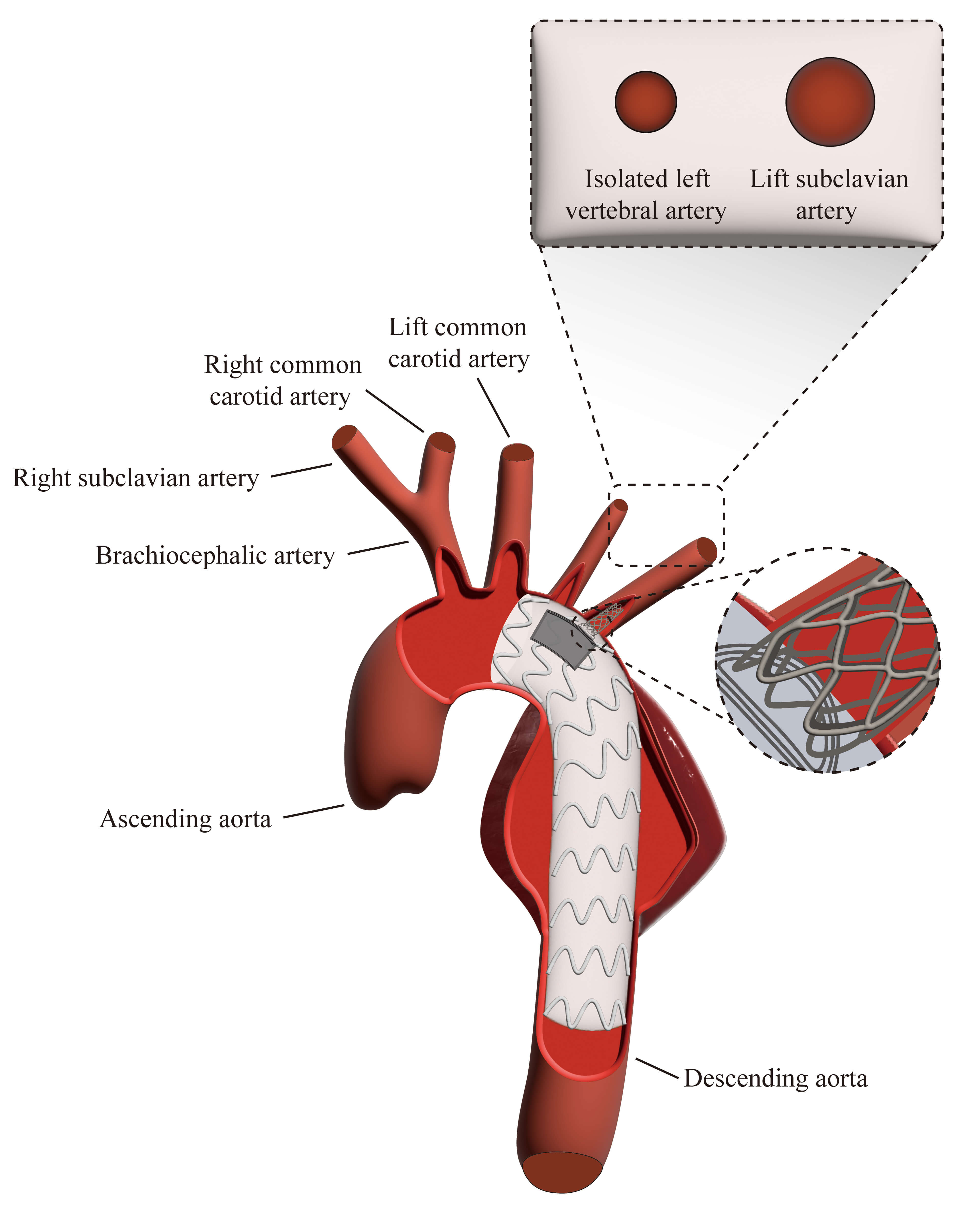 Fig. 2.
Fig. 2.
Illustration of TEVAR procedure with PMF and bridging stent. The diagram demonstrates the key steps of TEVAR with PMF for ILVA preservation and bridging stent placement for the LSA.
All patients with a history of hyperlipidemia or diabetes mellitus received
standard lipid-lowering and antidiabetic treatment during the perioperative
period, applied consistently across both groups. In the Fenestration Group,
antithrombotic therapy was tailored according to the diameter of the bridging
stent: patients with stents
Continuous variables were expressed as mean
Patients were followed up through outpatient visits and telephone interviews. The follow-up duration ranged from 1 month to 13 years. All patients were recommended to undergo CTA prior to discharge and annually thereafter, or earlier if clinically indicated, to monitor for complications such as endoleaks, stent migration, or changes in vertebral artery patency.
A total of 56 patients with aortic arch disease and ILVA who underwent TEVAR
were included in this study (Fig. 3). Patients were divided into either the
Coverage Group (N = 23) or the Fenestration Group (N = 33), depending on whether
fenestration was performed (Table 1). The mean age of the overall cohort was
54.48
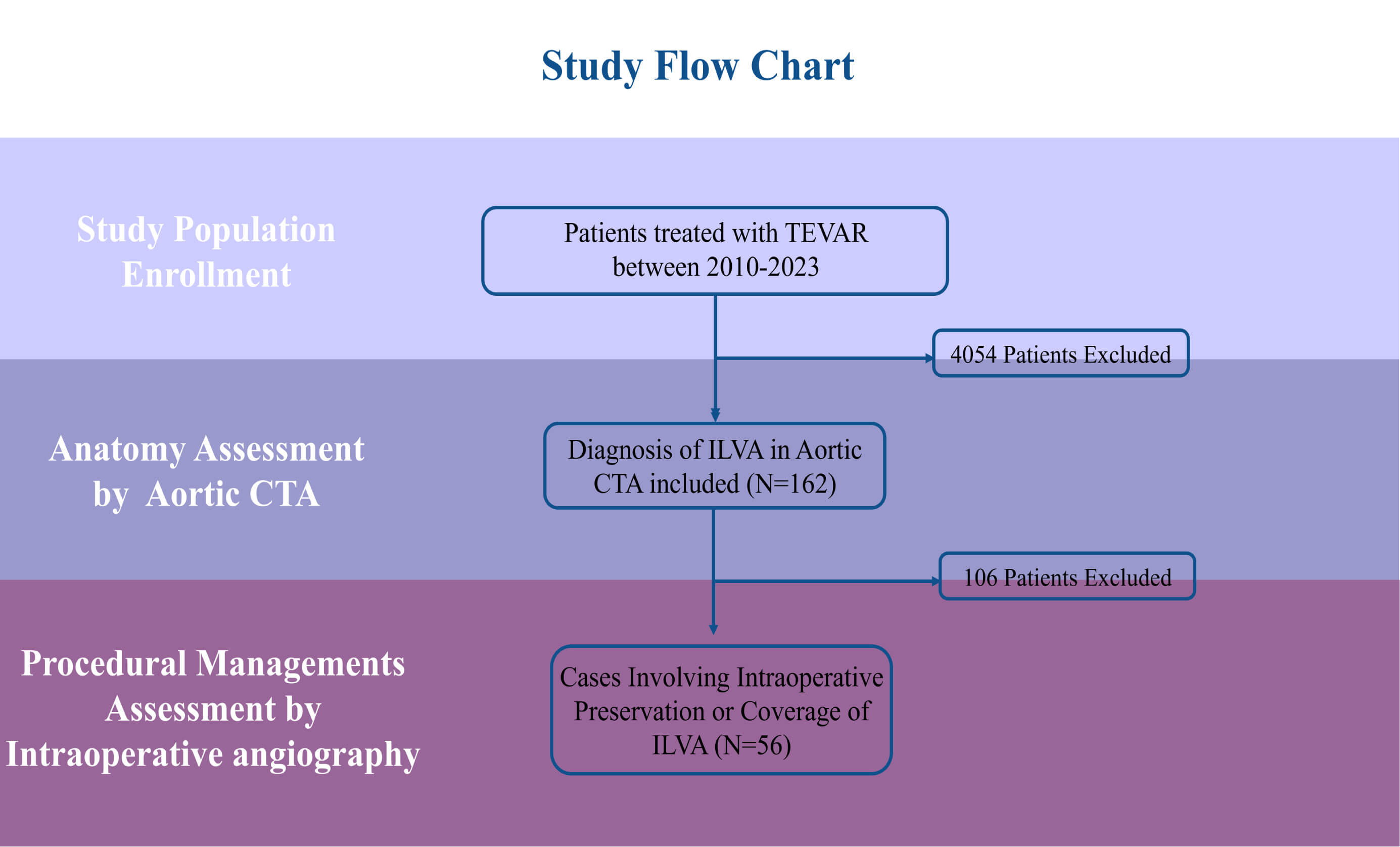 Fig. 3.
Fig. 3.
Patient selection and study flowchart. CTA, computed tomography angiography.
| Parameters | Total (N = 56) | Coverage Group (N = 23) | Fenestration Group (N = 33) | p-value | |
| Age (years) | 54.48 |
51.13 |
56.82 |
0.04 | |
| Male, n (%) | 50 (89.29) | 23 (100) | 27 (81.82) | 0.09 | |
| Body Mass Index (kg/m2) | 25.88 |
25.47 |
26.17 |
0.47 | |
| Clinical Presentation, n (%) | 0.20 | ||||
| Asymptomatic | 15 (26.79) | 5 (21.74) | 10 (30.30) | ||
| Chest Pain | 36 (64.29) | 14 (60.87) | 22 (66.67) | ||
| Abdominal Pain | 5 (8.93) | 4 (17.39) | 1 (3.03) | ||
| Medical History, n (%) | |||||
| Hyperlipidemia | 18 (32.14) | 6 (26.09) | 12 (36.36) | 0.42 | |
| Hypertension | 44 (78.57) | 19 (82.61) | 25 (75.76) | 0.78 | |
| Diabetes | 5 (8.93) | 0 | 5 (15.15) | 0.14 | |
| Coronary Artery Disease | 8 (14.29) | 2 (8.70) | 6 (18.18) | 0.54 | |
| History of Stroke | 2 (3.57) | 2 (8.70) | 0 | 0.16 | |
| Chronic Obstructive Pulmonary Disease | 1 (1.79) | 0 | 1 (3.03) | 1.00 | |
| Smoking | 26 (46.43) | 8 (34.78) | 18 (54.55) | 0.14 | |
| History of Open Surgery | 1 (1.79) | 1 (4.35) | 0 | 0.41 | |
| Left Ventricular Ejection Fraction (%) | 61.61 |
61.61 |
61.61 |
1.00 | |
| Clinical Diagnosis, n (%) | 0.12 | ||||
| Acute Aortic Syndrome | 31 (55.36) | 13 (56.52) | 18 (54.55) | ||
| Aortic Ulcer | 12 (21.43) | 2 (8.70) | 10 (30.30) | ||
| True Thoracic Aortic Aneurysm | 5 (8.93) | 4 (17.39) | 1 (3.03) | ||
| Pseudo Thoracic Aortic Aneurysm | 3 (5.36) | 1 (4.35) | 2 (6.06) | ||
| Chronic Type B Aortic Dissection | 5 (8.93) | 3 (13.04) | 2 (6.06) | ||
| Preoperative Aortic CTA | |||||
| Left Vertebral Artery Diameter (mm) | 3.06 |
3.07 |
3.06 |
0.97 | |
| Right Vertebral Artery Diameter (mm) | 3.74 |
3.85 |
3.66 |
0.25 | |
The procedural outcomes are detailed in Table 2. The overall technical success rate of TEVAR was 98.21%, and no perioperative mortality occurred. A significantly higher proportion of patients in the Fenestration Group underwent simultaneous left subclavian artery stenting compared with the Coverage Group (57.58% vs. 21.74%, p = 0.008). No significant differences were observed between the two groups with respect to endoleak rates, perioperative complications, or postoperative length of stay.
| Parameters | Total (N = 56) | Coverage Group (N = 23) | Fenestration Group (N = 33) | p-value |
| Technical success rate, n (%) | 55 (98.21) | 23 (100) | 32 (96.97) | 1.000 |
| Simultaneous LSA stenting, n (%) | 24 (42.86) | 5 (21.74) | 19 (57.58) | 0.008 |
| Perioperative mortality, n (%) | 0 | 0 | 0 | 1.000 |
| Endoleak, n (%) | 4 (7.14) | 2 (8.70) | 2 (6.06) | 1.000 |
| Postoperative length of stay, days | 5.29 |
5.39 |
5.21 |
0.730 |
| Pre-discharge CTA measurements, n (%) | 54 (96.43) | 23 (100.00) | 31 (93.94) | 0.510 |
| LVA diameter, mm | 2.79 |
2.47 |
3.02 |
0.020 |
| LVA reduction in diameter, mm | 0.10 (0, 0.5) | 0.3 (0.15, 0.70) | 0 (–0.10, 0.15) | |
| Change in LVA diameter (% decrease) | 3.03% (0, 13.61) | 13.64% (5.52, 22.4) | 0 (–3.29, 5.13) | |
| RVA diameter, mm | 3.75 |
3.98 |
3.58 |
0.009 |
| RVA reduction in diameter, mm | 0 (–0.37, 0.20) | –0.10 (–0.40, 0.05) | 0.10 (–0.25, 0.35) | 0.030 |
| Change in RVA diameter (% decrease) | 0 (–10.26, 5.71) | –2.56% (–10.98, 0.98) | 2.38% (–6.98, 9.41) | 0.050 |
| LVA occlusion rate, n (%) | 2 (3.57) | 2 (8.70) | 0 | 0.160 |
| Incidence of vertebral artery diameter reduction, n (%) | 11 (19.64) | 9 (39.13) | 2 (6.06) |
LSA, left subclavian artery; LVA, left vertebral artery; RVA, right vertebral artery.
A total of 54 patients (96.43%) underwent CTA at discharge. Compared with
preoperative and postoperative measurements, the Coverage Group exhibited a
significantly greater reduction in LVA diameter at discharge than that of the
Fenestration Group 13.64% [5.52, 22.4%] vs. 0 [–3.29%, 5.13%], p
Among the 38 patients who underwent follow-up CTA, the median follow-up CTA interval was 41.38 (21.42, 73.33) months. The follow-up CTA interval was significantly longer in the Coverage Group than in the Fenestration Group (79.33 [34.25, 106.73] vs. 36.43 [19.67, 55.67] months, p = 0.010). The incidence of complications during follow-up was comparable between groups (17.39% vs. 9.09%, p = 0.61). In the Fenestration Group, one patient experienced an ischemic stroke and two patients developed endoleaks on follow-up CTA. In the Coverage Group, adverse events included one sudden unexplained death, one type II endoleak from the LSA that was treated with coil embolization at 9 months, one reintervention with repeat TEVAR for a new distal aortic ulcer, and one case of LSA stent occlusion. Additionally, a type III endoleak occurred in one Coverage Group patient, which resolved spontaneously at the 3-month CTA follow-up. All endoleaks were either self-limiting or successfully treated, and no long-term adverse events were reported. No patient in the Coverage Group required carotid-subclavian bypass surgery for posterior circulation hypoperfusion or upper limb ischemia.
A total of 38 patients underwent follow-up CTA, including 17 in the Coverage
Group and 21 in the Fenestration Group, with a mean interval of 23.29
| Parameters | Total (N = 38) | Coverage (N = 17) | Fenestration (N = 21) | p-value |
| Follow-up CTA interval, months | 41.38 (21.42, 73.33) | 79.33 (34.25, 106.73) | 36.43 (19.67, 55.67) | 0.010 |
| LVA diameter at follow-up, mm | 2.42 |
1.78 |
2.93 |
0.006 |
| LVA reduction, mm | 0.25 (–0.10, 1) | 0.80 (0.30, 2.3) | 0 (–0.10, 0.3) | 0.007 |
| LVA decrease, % | 8.22 (–2.58, 28.57) | 28.57 (10, 100) | 0 (–3.03, 8.33) | 0.005 |
| RVA diameter at follow-up, mm | 3.88 |
4.20 |
3.62 |
0.003 |
| RVA reduction, mm | –0.20 (–0.57, 0.17) | –0.30 (–0.90, –0.2) | 0 (–0.20, 0.4) | 0.005 |
| RVA decrease, % | –4.89 (–16.31, 3.75) | –10.34 (–24.39, –5.13) | 0 (–5.41, 11.43) | 0.003 |
| Incidence of LVA diameter occlusion, n (%) | 6 (15.79) | 5 (29.41) | 1 (4.76) | 0.070 |
| Incidence of LVA diameter reduction, n (%) | 12 (31.58) | 9 (52.94) | 3 (14.29) | 0.020 |
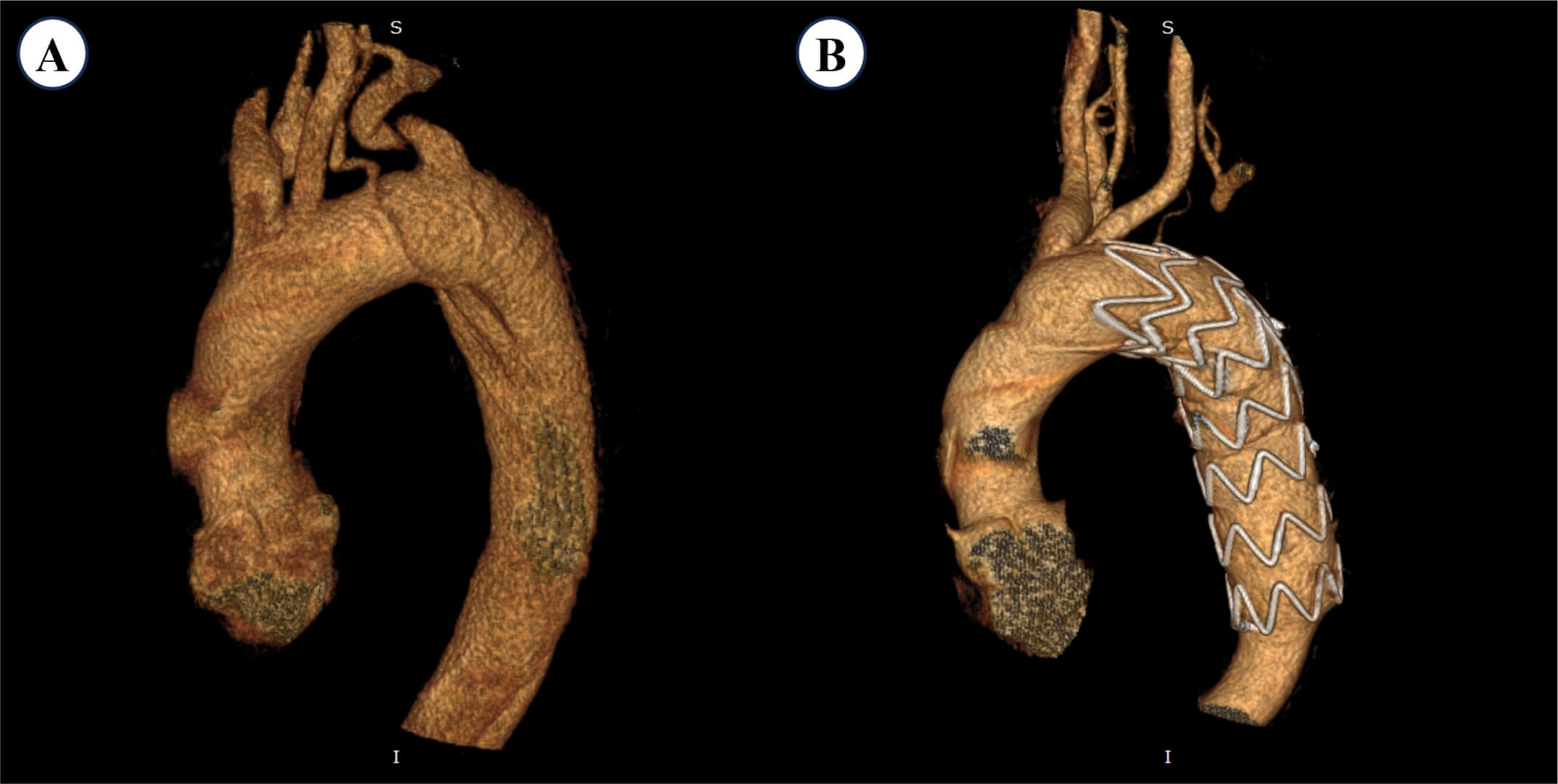 Fig. 4.
Fig. 4.
Preoperative and postoperative 3D-CTA images of a patient in the Coverage Group. (A) Preoperative image showing a prominent ILVA arising directly from the aortic arch, with normal caliber and course. (B) Four-year postoperative image showing a marked reduction in ILVA diameter, indicating chronic hypoperfusion, and absence of contrast opacification in the LSA. S, superior; I, inferior.
Following the carotid arteries, the vertebral arteries play a critical role in cerebral perfusion and constitute a crucial component of the circle of Willis. Posterior circulation infarctions account for approximately 25–30% of all ischemic strokes [5]. Prior studies have shown that reduced blood flow in the left vertebral and subclavian arteries increases the risk of symptomatic vertebrobasilar insufficiency, spinal cord injury (SCI), and stroke [7, 8, 9]. In the management of complex thoracic aortic pathologies, particularly in patients with an ILVA, achieving an adequate proximal landing zone often requires coverage of both the LSA and LVA. The isolated left vertebral artery typically arises directly from the aortic arch between the left common carotid artery and LSA. This anatomical variant represents the second most common variation of the supra-aortic trunks, with a reported prevalence of approximately 4.81% [10]. Current guidelines recommend that in patients with an ILVA arising directly from the thoracic aorta, vertebral artery revascularization should be considered when TEVAR involves its origin [1]. Compared with LSA revascularization, revascularization of an isolated left vertebral artery presents greater technical challenges during TEVAR.
In recent years, recognition of the importance of preserving the ILVA during TEVAR has grown. Several studies have investigated various strategies to achieve this objective in the setting of complex thoracic aortic disease. Yang et al. [11] demonstrated that hybrid procedures combining TEVAR, ILVA transposition, and left common carotid-subclavian artery bypass are both safe and feasible for managing thoracic aortic pathologies involving the ILVA. Luo et al. [12] reported the use of a physician-modified Castor branched stent with fenestration to reconstruct the ILVA during aortic arch surgery. Their findings indicated that the PMF technique with the Castor stent, performed under local anesthesia, was both safe and effective [12]. Shen et al. [13] highlighted the feasibility of in situ fenestration for ILVA reconstruction in complex aortic arch disease. This method was also shown to be both safe and effective, although it required direct ILVA exposure and sheath placement via puncture [13]. In a multicenter retrospective study, Zhang et al. [4] compared three techniques: a novel chimney approach using right brachial-left brachial crossover, external fenestration, and arterial transposition. At our center, we utilized the PMF technique to preserve the ILVA in cases of complex thoracic aortic disease. In the present study, fenestration did not significantly affect overall surgical outcomes, and no substantial differences were observed in endoleak incidence between groups.
Most previous studies have focused on the technical methods and immediate outcomes of ILVA preservation, whereas limited attention has been directed toward the hemodynamic consequences of ILVA preservation after TEVAR. In this study, we innovatively evaluated the hemodynamic impact of preserving the ILVA using PMF during TEVAR. Our findings showed that, for patients undergoing Zone 2 TEVAR involving the ILVA, the Fenestration Group had a significantly lower incidence of postoperative ILVA stenosis compared with the Coverage Group. Previous research has demonstrated that preoperative revascularization of the LSA in patients requiring LSA coverage during TEVAR reduces the risk of stroke and SCI [14, 15]. This protective effect was likely due to the maintenance of posterior circulatory perfusion through the left vertebral artery. Therefore, preserving the ILVA during TEVAR is particularly important for patients with dominant left vertebral arteries or incomplete circles of Willis [7, 16]. Our study also found that in the Coverage Group, the non-stenosis rate was 52.17%. We hypothesize that although the left vertebral artery origin was covered, the relatively short coverage length and incomplete apposition of the stent graft to the aortic wall may have minimized the hemodynamic impact on the left vertebral artery in these patients.
Owing to varying levels of understanding of the disease, not all interventional vascular specialists at our center routinely use fenestration techniques to preserve ILVA flow. However, as on-table fenestration techniques have advanced in recent years and in the absence of dedicated devices for managing aortic arch pathologies with vascular anomalies, we recommend PMF as an effective treatment option. Based on our experience and the anatomical characteristics of the aortic arch branches, the origin of the ILVA is typically located along the centerline of the aortic arch, within the contour of the LSA opening on the greater curvature of the arch. Therefore, if adequate blood flow to the LSA is maintained, coverage of the ILVA origin is unlikely.
This study has several limitations. First, being a retrospective, single-center study, inherent biases related to patient selection, data collection, and analysis may affect the generalizability of our findings. For instance, patients in the Fenestration Group were significantly older, which may reflect a clinical preference for preserving posterior circulation in older patients who have more comorbidities, thus introducing potential selection bias. Second, the relatively small sample size may have limited the statistical power to detect subtle differences between the coverage and fenestration groups. Third, although our study focused on the hemodynamic impact of ILVA preservation, neurological events were not predefined as primary endpoints, and no postoperative neuroimaging, such as brain MRI, was routinely performed. Finally, long-term functional outcomes, including cerebrovascular events and patient-reported quality of life, were not evaluated. These additional endpoints may provide further insights into the clinical significance of ILVA revascularization.
In patients with ILVA undergoing Zone 2 TEVAR, fenestration was associated with a lower incidence of postoperative ILVA diameter reduction compared to direct coverage. These findings suggest a potential benefit of ILVA revascularization; however, due to the study’s limited sample size, further research is needed to confirm these results.
All data generated or analyzed during this study are included in this published article.
ZP and KF designed the study, selected methodology and software, analyzed the data and drafted the manuscript. JL, YZ, JZ, BF, YL, CS, and ML contributed to patient enrollment, data collection, imaging evaluation, and clinical follow-up. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This retrospective cohort study was approved by the ethics committee of Fuwai Hospital Chinese Academy of Medical Sciences (2025-2982) and was performed in accordance with the Declaration of Helsinki. Individual informed consent was waived due to the retrospective nature of the study and the use of anonymized data.
We thank all the patients and doctors from Fuwai hospital involved in this study.
This work was supported by shenzhen Clinical Research Center for Cardiovascular Diseases Fund (No.20220819165348002), National High Level Hospital Clinical Research Funding (2022-GSP-GG-16), Shenzhen Municipal Special Program for Major Public Platforms and Core Technology Breakthroughs in the Biopharmaceutical Industry (No. XMHT20220104045), Shenzhen Development and Reform Commission Document No. 64 (2023), and Young Talent Program of the Academician Fund (No. YS-2022-013).
The authors declare no conflict of interest.
During the preparation of this manuscript, the authors used ChatGPT to assist with language polishing and grammar checking. After using this tool, the authors carefully reviewed and edited the content as needed and take full responsibility for the integrity and accuracy of the work. The AI tool was not used for study design, data analysis, or generation of scientific conclusions.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




