1 Department of Cardiology - Valve Clinic, Vrije Universiteit Brussel (VUB), Universitair Ziekenhuis Brussel (UZ Brussel), B-1090 Brussels, Belgium
2 Department of Cardiology, Université Libre de Bruxelles (ULB), CHU Saint-Pierre, B-1000 Brussels, Belgium
Abstract
Significant tricuspid regurgitation (TR) is increasingly recognized as a major determinant of morbidity and mortality, yet the clinical impact of significant TR has long been underestimated. Assessment of right ventricular (RV) systolic function is central to understanding and managing TR and represents the principal determinant of symptoms, therapeutic response, and long-term outcomes. The unique sensitivity of the RV to alterations in preload and afterload leads to maladaptive remodeling, making accurate functional assessment essential for risk stratification and for optimizing the timing and type of intervention, especially given the expanding range of available surgical and transcatheter treatment options. Echocardiography remains the primary imaging modality, providing qualitative and quantitative evaluations of RV function through parameters such as tricuspid annular plane systolic excursion (TAPSE), RV fractional area change (RVFAC), and tissue Doppler systolic velocity (S′). Advances in speckle-tracking echocardiography for RV free-wall longitudinal strain and in three-dimensional imaging have improved accuracy; however, all echocardiographic measures remain limited by the complex geometry of the RV. When feasible and available, cardiac magnetic resonance (CMR) imaging serves as the reference standard for precise assessment of RV volumetric and functional parameters. Impaired RV systolic function, both before and after intervention, irrespective of the imaging parameter used for the assessment, consistently predicts adverse outcomes in patients with severe TR, including heart failure progression, reduced exercise tolerance, and decreased survival. Therefore, early recognition and quantification of RV dysfunction are crucial to enable timely therapy, as interventions before the development of advanced RV impairment provide symptomatic and survival benefits. This review summarizes the pathophysiology, quantitative thresholds, and prognostic significance of RV function assessment, emphasizing the pivotal role this evaluation plays in the contemporary management of significant TR.
Keywords
- right ventricular function
- tricuspid valve
- tricuspid valve insufficiency
- ventricular remodeling
- risk assessment
- review
For decades, clinical and research attention has largely focused on left ventricular (LV) structure and function, while the right ventricle (RV) was primarily considered as a conduit for pulmonary blood flow, rather than a contributor to systemic physiology. This concept slowed advances in our knowledge of the pathophysiology of right-sided heart disease [1]. Consequently, the tricuspid valve (TV) has long been regarded as the “forgotten heart valve” [2]. However, large population-based registries have demonstrated a 1.5–2% prevalence of significant (at least moderate) tricuspid regurgitation (TR) among the general population, with increasing prevalence of clinically-relevant TR with advancing age, reaching nearly 4% in individuals over 75 years of age [3, 4, 5]. There is increasing evidence from several patient cohorts that the presence of significant secondary TR (STR) has prognostic implications, and that, if left untreated, significant TR is associated with adverse clinical outcomes. These outcomes include quality of life, exercise capacity, mortality, and heart failure-related hospitalization, which are largely independent of LV and RV systolic function and of the presence of pulmonary hypertension (PHT) [5, 6, 7, 8]. Surgical and transcatheter TV interventions are increasingly available for the treatment of significant TR but are still underused. One of the major clinical challenges is to determine the optimal timing for intervention, because patients may remain asymptomatic for a long time if receiving adequate diuretic treatment, and referrals often occur when patients have advanced right heart failure and irreversible end-organ dysfunction [9, 10, 11]. Current guidelines for the management of valvular heart disease issued by the European Society of Cardiology (ESC) [12] and by the American Heart Association/American College of Cardiology (AHA/ACC) [13] recommend intervention based on a combination of clinical and echocardiographic factors, including TR severity, symptom burden, anatomical parameters such as tricuspid annular dilation, and, importantly, the presence and extent of PHT and RV dysfunction. Nonetheless, evaluating the right heart in the setting of TR remains challenging in current clinical practice, because of the complex three-dimensional anatomy resulting in difficult image acquisition, specific right-sided hemodynamic patterns, the load dependency of common RV indices, and the complex interplay between the RV, the pulmonary vasculature, and the LV [1]. This review provides an overview of current evidence on the assessment, clinical features, and prognostic impact of right heart function in patients with significant TR (Fig. 1).
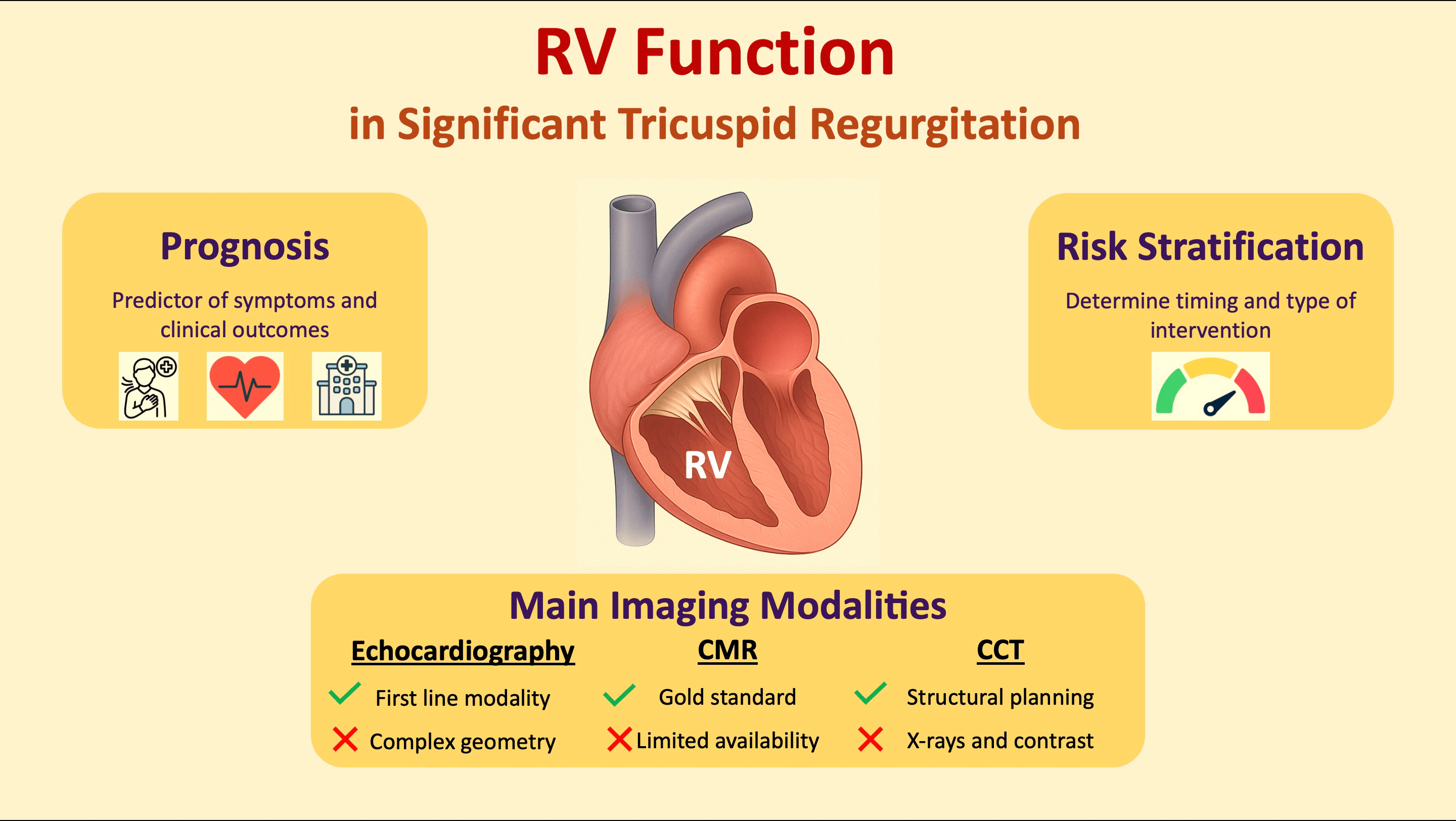 Fig. 1.
Fig. 1.
Right ventricular function in patients with significant tricuspid regurgitation. Abbreviations: CCT, cardiac computed tomography; CMR, cardiac magnetic resonance; RV, right ventricular.
The RV is located anteriorly within the thoracic cavity, directly behind the sternum. It is bordered by the TV annulus (TA) proximally and by the pulmonary valve distally. The RV is classically divided into three structural components: (1) the inlet region, which comprises the TV apparatus, including the leaflets, chordae tendineae, and papillary muscles; (2) the trabeculated apical myocardium; and (3) the infundibulum, also known as the conus arteriosus, i.e., the smooth-walled outflow tract that directs blood toward the pulmonary valve [1, 14]. For functional and imaging purposes, the RV can also be divided into anterior, lateral, and inferior walls, as well as into basal, midventricular, and apical segments [1].
The RV has a unique, complex, three-dimensional geometry. Whereas the LV is ellipsoid, the RV has a triangular shape in the sagittal plane and a crescent-shaped profile in cross-sectional views [1]. Under normal loading conditions and in the absence of conduction abnormalities, the interventricular septum is concave toward the LV during both systole and diastole. In adults, the RV end-diastolic volume typically exceeds that of the LV [1]. However, due to thinner myocardial walls, the RV’s mass is approximately one-sixth that of the LV [15].
The RV myocardium is composed of two main muscle layers: a superficial and a deep one. Superficial fibers run parallel to the atrioventricular (AV) groove and are oriented circumferentially [14, 16]. On the sternocostal surface, the superficial fibers adopt an oblique trajectory towards the apex and continue into the superficial myocardial layer of the LV, reflecting the myocardial fiber continuity between the ventricles. In contrast, the deep RV muscle fibers are arranged longitudinally, extending from the base to the apex; they are primarily responsible for the longitudinal shortening that characterizes normal RV contraction [1, 14, 16].
Normal RV function is the result of the precise interaction of several factors, including systemic venous return (preload), intrinsic myocardial systolic and diastolic performance, pulmonary vascular resistance (afterload), and pericardial compliance. Under physiological conditions, RV systolic function is primarily driven by the shortening of longitudinal myocardial fibers, accounting for about 80% of RV stroke volume [17]. However, in certain clinical scenarios, such as early after cardiac surgery, a transient but significant reduction in longitudinal function is often observed [18]. In these cases, the recruitment of circumferential myocardial fibers compensates for the impaired longitudinal mechanics, enabling overall cardiac output to be preserved [18].
The chronic volume overload of the RV observed in significant TR results from the addition of regurgitant flow to systemic venous return. This increases RV preload and consequently also RV afterload. In the early stages of chronic TR, the RV undergoes homeometric adaptation in accordance with Anrep’s effect, enhancing myocardial contractility while maintaining chamber dimensions and a relatively low end-systolic volume. This compensatory mechanism effectively preserves RV stroke volume and ejection fraction over a prolonged period despite ongoing volume overload [19, 20, 21]. However, as the severity of TR worsens, RV systolic function begins to decline, particularly in the longitudinal plane. The radial and anteroposterior components of contraction initially remain preserved. With further disease progression, a heterometric adaptation, as described by Starling’s law, becomes predominant. This phase is characterized by increases in RV end-diastolic and end-systolic volumes in an attempt to preserve forward stroke volume, albeit at the expense of adverse RV remodeling and RV hypertrophy. Although RV wall thickness may remain within normal limits, total RV mass increases. Eventually, elevated RV filling pressures and diastolic interventricular septal displacement (flattening) can impair LV filling and function, a phenomenon known as ventricular interdependence [19, 20, 21].
In patients with chronic significant TR, the substantial volume that regurgitates into the right atrium (RA) results in elevated RA pressures, which are subsequently transmitted to the systemic venous circulation, resulting in systemic venous congestion. This congestion manifests clinically as hepatomegaly, ascites, peripheral edema, and edema of the gastrointestinal tract [22]. Persistent venous congestion can further impair RV function through several mechanisms, including pericardial constraint, interventricular septal shift due to elevated RV diastolic pressure, and reduced coronary perfusion pressure; the latter particularly affects the subendocardial layers of the RV myocardium, making these layers more vulnerable to ischemia. This process initiates a vicious cycle in which progressive RV dysfunction promotes venous congestion, and prolonged congestion further deteriorates RV function. If left untreated, this cycle ultimately results in advanced, refractory right heart failure [22].
In 90% of cases, TR is the result of dilatation of the RA, RV, or TA, leading to STR. The most common form of STR is ventricular STR (VSTR), in which dilation of the RV causes tricuspid leaflet tethering during systole [23]. Atrial STR (ASTR), which occurs as a result of RA dilatation or dysfunction, often in the setting of atrial fibrillation, has long been neglected but has recently emerged as an important etiology of STR, accounting for 10%–15% of cases [24, 25]. There may be some overlap between VSTR and ASTR. Indeed, chronic significant VSTR may result in dilatation of the RA and TA due to volume overload (‘VSTR begets ASTR’), and chronic significant ASTR may eventually result in RV dilatation or dysfunction (‘ASTR begets VSTR’; Fig. 2) [23]. Therefore, in advanced disease, the chronic volume overload imposed by ASTR on the RV may have a deleterious effect on RV function, and some patients may present with complex forms of STR that have characteristics of both ASTR and VSTR [26].
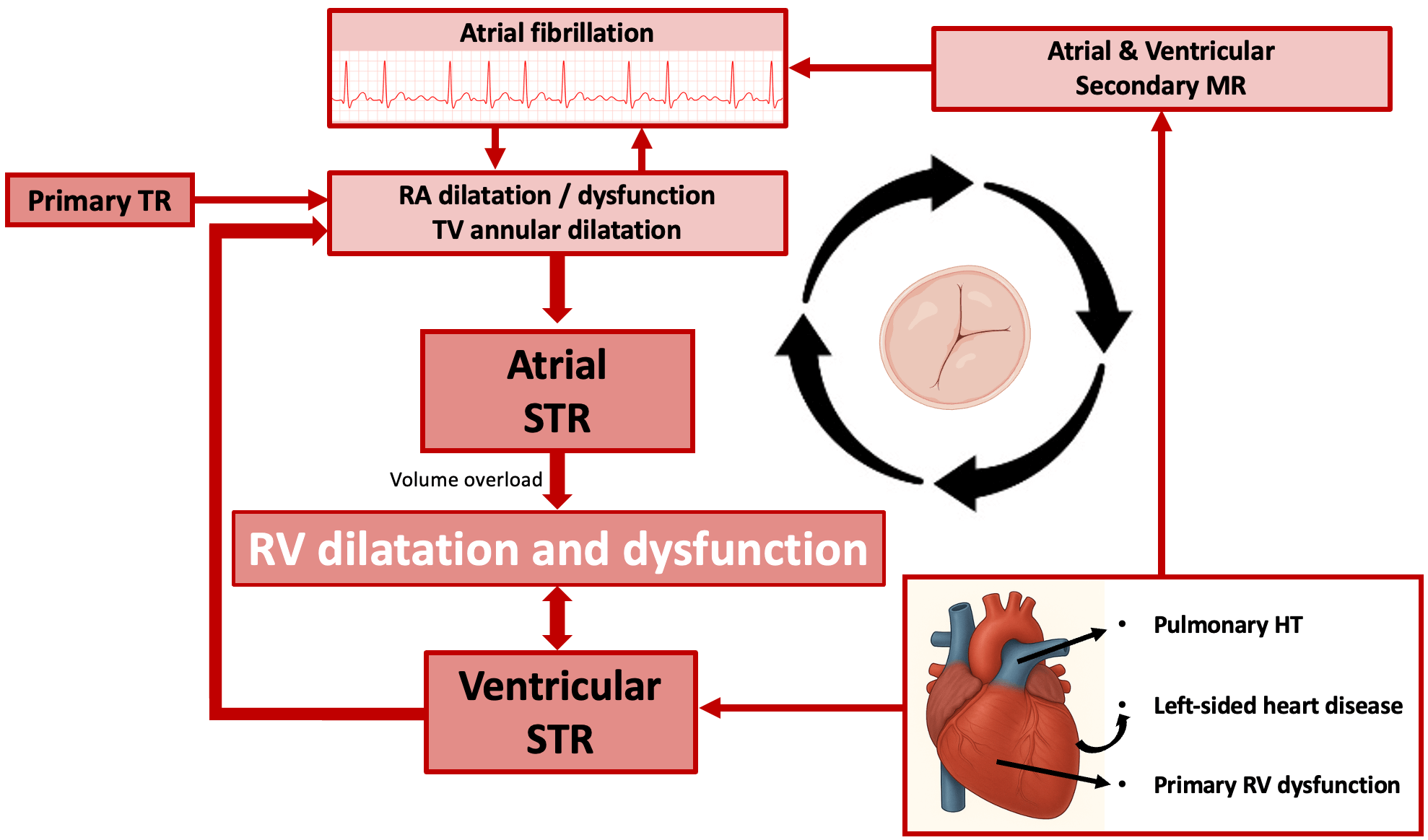 Fig. 2.
Fig. 2.
Vicious circle of right ventricular dilatation and dysfunction in patients diagnosed with atrial and ventricular secondary tricuspid regurgitation. Abbreviations: HT, hypertension; MR, mitral regurgitation; RA, right atrium; RV, right ventricle; STR, secondary tricuspid regurgitation; TR, tricuspid regurgitation; TV, tricuspid valve.
Left-sided heart disease may also contribute substantially to the development of TR and subsequently to RV dysfunction (Fig. 2). In addition to left-sided valvular heart disease and to LV dysfunction, TR may also result from heart failure with preserved left ventricular ejection fraction (LVEF) (HFpEF), as a result of increased LV filling pressure, exercise-induced congestion, and, later, of pulmonary vascular disease. VSTR is a common phenotype in this setting. RV adaptation to the level of the pulmonary pressure (RV to pulmonary artery coupling (RVPAC)) is an independent prognostic factor in HFpEF [27]. However, atrial myopathy may also contribute to the development of STR [28]. Among patients with ASTR, the combination of AF and HFpEF is frequent and associated with poor outcomes [29].
The clinical presentation of RV dysfunction or failure is primarily related to systemic venous congestion and, in advanced cases, to low cardiac output. Venous congestion typically manifests as peripheral edema, jugular vein distention, hepatojugular reflux, ascites, painful hepatomegaly, and nocturia; in addition, as a result of congestion in the gastrointestinal tract, nausea and loss of appetite may occur. Low cardiac output induces fatigue, weakness, shortness of breath, chest pain/discomfort, dizziness and fainting. A S3 gallop may be heard upon auscultation. Weight gain as a result of fluid retention often indicates worsening heart failure and, in very advanced cases, jaundice and cachexia may occur. A prominent jugular V wave, pansystolic murmur at the lower left sternal border with inspiratory increase, and pulsatile liver are hallmarks of severe TR.
The assessment of RV function in patients with TR is particularly challenging as a result of the unique anatomy of the RV and its sensitivity to loading conditions. The main challenges result from the following: (1) The RV is a crescent-shaped structure with a broad base and a triangular apex, and includes outlet, apex, and inlet portions. When RV volume and pressure overload occur, as in significant TR, the RV loses its triangular shape and becomes more elliptical. Hence, geometrical assumptions are unreliable, making the assessment and interpretation of RV ejection fraction (RVEF) particularly challenging. Moreover, these anatomical features of the RV prevent accurate assessment of global contractility using a single index. (2) Loading conditions may also affect the assessment of RV function. RV radial function and TA motion are usually accentuated in the early and compensated stages of severe TR, and may eventually result in overestimation of RV performance. (3) Non-invasive (i.e., echocardiography-derived) measurement of pulmonary arterial pressure can be misleading when TR severely alters the pressure dynamics between the right heart chambers, reducing the reliability and accuracy of TR maximal velocity for prediction of pulmonary systolic pressure. This effect may also significantly impact the assessment of RV–pulmonary artery coupling. Therefore, caution is needed to prevent underestimation of pulmonary pressure using echocardiography. (4) TR may develop after cardiac surgery, particularly that involving the mitral valve, and RV longitudinal function is typically reduced in this setting, even when overall RV function is preserved. RV longitudinal function parameters should be used with caution in this setting.
As a result of the abovementioned limitations, no single measure offers perfect diagnostic and prognostic accuracy for assessment of RV function in the setting of TR and a multiparametric evaluation is usually recommended [12].
Mitral regurgitation frequently coexists with TR. Mitral regurgitation may occur as a primary left-sided valvular abnormality or may develop secondary to left-sided cardiac pathology (e.g., dilated cardiomyopathy or ischemic heart disease); both etiologies can contribute to VSTR. Alternatively, mitral regurgitation and TR may occur concomitantly as part of a secondary atrial mechanism driven by atrial fibrillation or heart failure with preserved ejection fraction. The presence, etiology, and severity of mitral regurgitation can influence TR severity and RV function, and vice versa, and must therefore be systematically integrated into the comprehensive echocardiographic and clinical evaluation. Following mitral valve intervention, progression of TR is commonly observed and has been associated with adverse long-term outcomes [30, 31]. Consequently, current guidelines recommend concomitant TV surgery in patients with at least moderate TR undergoing left-sided valve procedures [12]. The effect of a TV intervention on mitral regurgitation is less well defined. Cannata et al. [32] reported that the severity of mitral regurgitation varied considerably after transcatheter TV intervention, with mitral regurgitation remaining stable in 61% of patients, worsening in 10%, and improving in 30%. Additional studies are required to clarify the bidirectional interaction between mitral regurgitation and TR and its therapeutic implications.
Echocardiography is the first-line imaging modality for assessing TV anatomy, the size and function of the RA and RV, and quantifying TR severity [33, 34, 35, 36]. However, echocardiographic evaluation of the right heart remains challenging due to the abovementioned limitations, including the complex crescentic three-dimensional geometry of the RV and the hemodynamic impact of TR itself [34, 35]. Fortunately, advances in three-dimensional echocardiography and other imaging modalities have enhanced our ability to assess RV function. Fig. 3 provides an overview of parameters for RV function assessment using the different imaging modalities.
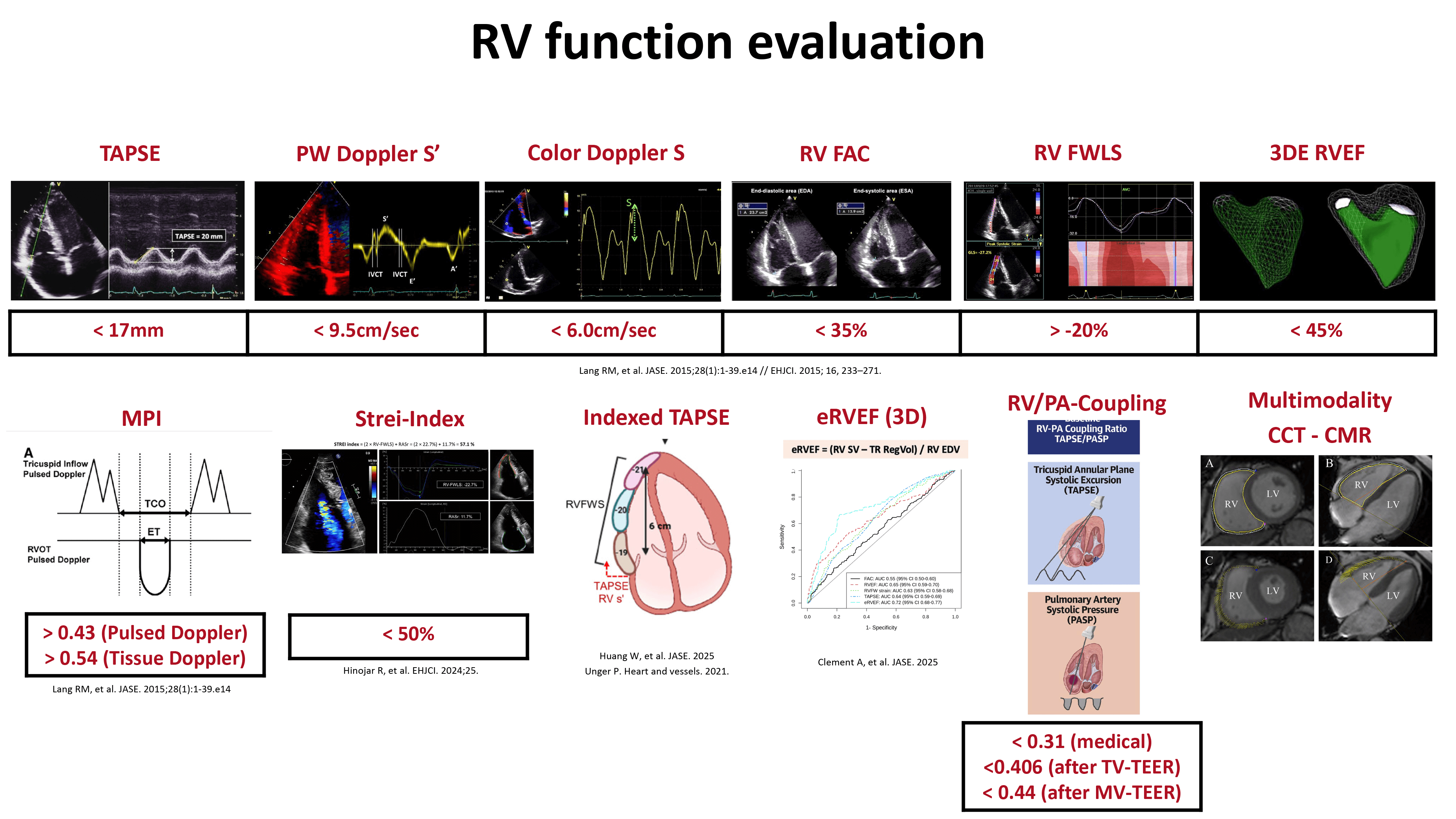 Fig. 3.
Fig. 3.
Assessment of right ventricular function using different parameters from echocardiography, cardiac magnetic resonance imaging, or cardiac computed tomography. Abbreviations: 3DE, three-dimensional echocardiography; CCT, cardiac computed tomography; CMR, cardiac magnetic resonance; EF, ejection fraction; eRVEF, effective RVEF; FAC, fractional area change; FWLS, free wall longitudinal strain; MPI, myocardial performance index; MV, mitral valve; PA, pulmonary artery; PW, pulsed wave Doppler; RV, right ventricle; RVEF, right ventricular ejection fraction; TAPSE, tricuspid annular plane systolic excursion; TEER, transcatheter edge-to-edge repair; TV, tricuspid valve.
The echocardiographic assessment should start with a qualitative evaluation of RV anatomy using multiple acoustic windows, including the parasternal long-axis and RV inflow views, an RV-focused apical four-chamber view, and the subcostal four-chamber view. A multiparametric evaluation then provides a comprehensive assessment of RV systolic function. Table 1 provides an overview of the many echocardiographic parameters available, with their advantages and limitations. Ideally, several of the echocardiographic parameters should be used, including tricuspid annular plane systolic excursion (TAPSE), RV fractional area change (FAC), tissue Doppler-derived systolic velocity of the lateral TA (S′), RV global longitudinal strain (GLS), RV free wall longitudinal strain (FWLS), and three-dimensional echocardiographic RVEF [34, 37].
| Echocardiographic parameter | Recommended method | Advantages | Limitations |
| Global RV function | |||
| Pulsed wave Doppler RIMP (Tei-index) | RIMP = (TCO – ET)/ET | ||
| Tissue Doppler RIMP | RIMP = (TCO – ET)/ET | ||
| FAC | Percentage change of end-diastolic and end-systolic area of the RV, measured on an RV-focused apical view FAC (%) = 100 |
||
| Myocardial work | Using left-sided software for the evaluation of myocardial work on the RV, using RV GLS and the invasively acquired systolic and diastolic pulmonary artery pressures | ||
| Volumetric assessment | Percentage change of end-diastolic and end-systolic volume of the RV, measured by 3DE | ||
| RVEF = (RVEDV – RVESV)/RV EDV |
|||
| 3DE RV ejection fraction | Dedicated 3DE-software calculation of the RV EF | ||
| Effective 3DE RVEF | Takes into account the volume overload | ||
| eRVEF = (RV forward SV)/(RVEDV) | |||
| Longitudinal RV function | |||
| TAPSE | Longitudinal lateral annular excursion M-mode, measured between end-diastole and peak systole | ||
| Pulsed wave tissue Doppler S′ | Peak systolic velocity of the lateral tricuspid annulus by pulsed wave tissue Doppler imaging | ||
| Global longitudinal strain | Longitudinal speckle tracking derived strain, averaged over the six segments of the RV | ||
| Free wall longitudinal strain | Longitudinal speckle tracking derived strain, averaged over the three segments of the lateral free wall of the RV | ||
| RV-PA-coupling | |||
| TAPSE/sPAP | The ratio between TAPSE and echocardiographic estimated sPAP | ||
| RVFWLS/sPAP | The ratio between RVFWLS and echocardiographic estimated sPAP | ||
| Novel methods | |||
| Forward SV/RV ESV | Ratio between the calculated forward SV and 3DE-aquired RV ESV | ||
| RV free wall longitudinal fractional shortening | Indexed TAPSE to be used as a surrogate for RV free wall longitudinal strain = TAPSE/RVdiastolic length |
||
Abbreviations: 2DE, two-dimensional echocardiography; 3DE, three-dimensional echocardiography; CMR, cardiac magnetic resonance; EDA, end-diastolic area; EDV, end-diastolic volume; EF, ejection fraction; eRVEF, estimated right ventricular ejection fraction; ESA, end-systolic area; ESV, end-systolic volume; ET, ejection time; FAC, fractional area change; FWLS, free wall longitudinal strain; GLS, global longitudinal strain; RA, right atrium; RIMP, Right Ventricular Index of Myocardial Performance; RV, right ventricle; RVFWLS, right ventricular free wall longitudinal strain; sPAP, systolic pulmonary artery pressure; SV, stroke volume; TAPSE, tricuspid annular plane systolic excursion; TCO, total contraction time; TR, tricuspid regurgitation.
New measures have been developed to optimize RV function assessment by
eliminating some of the limitations of conventional parameters. In this context,
the effective RVEF accounts for the regurgitant volume in the presence of TR by
calculating the ratio between the RV forward stroke volume and the RV
end-diastolic volume [38]. In addition, measures assessing the RVPAC account for
pulmonary arterial afterload, offering a more physiologically integrated
assessment of RV function than conventional deformation or volumetric parameters
alone, and reflecting the adequacy of RV adaptation to pressure overload [39, 40]. RV function and pulmonary pressure are closely linked because PHT can impair
RV function and, conversely, severe RV dysfunction can prevent the generation of
elevated pulmonary pressures. RVPAC is traditionally defined as the ratio of
end-systolic ventricular elastance to arterial elastance as measured using
invasive right heart catheterization. However, echocardiography provides a
non-invasive surrogate by calculating the ratio of a longitudinal parameter
(TAPSE or RVFWLS) to the systolic pulmonary arterial pressure (sPAP) estimated
by echocardiography. This load-adjusted metric may enhance risk stratification
and guide timing of intervention in conditions where afterload is elevated and
conventional measures may underestimate RV dysfunction. In patients with
significant TR, a reduced TAPSE/sPAP ratio consistently emerges as a marker of
RV–pulmonary arterial uncoupling and adverse prognosis. The optimal prognostic
threshold is context-dependent, with reported values of approximately 0.31 in STR
[41, 42], 0.40 in isolated functional TR [43], 0.406 in transcatheter TR
intervention cohorts [44], and up to 0.49 in a mixed TR population [45]. A
threshold of 0.31 mm/mmHg is most frequently associated with excess mortality in
patients with STR. Taken together, these data support a continuum in which
TAPSE/sPAP values less than ~0.3–0.4 mm/mmHg indicate clinically
relevant RV–pulmonary artery (PA) uncoupling and identify higher-risk patients,
whereas higher values are more compatible with preserved coupling. Furthermore,
the pulmonary artery pulsatility index is a hemodynamic measure of RVPAC,
calculated as (sPAP – pulmonary artery diastolic pressure) divided by right
atrial pressure. In patients with significant TR, available data suggest a
largely continuous inverse relationship between the pulsatility index and
outcome, without a universally accepted TR-specific cutoff value. In patients
undergoing transcatheter TV intervention, who were stratified according to
pulsatility index values (
Most Doppler methods used to quantify left-sided valvular regurgitation can be
used to assess TR severity. This includes jet size, vena contracta width, and
proximal convergence analysis. However, some characteristics of TR jets are
inherently different from mitral regurgitation jets. Importantly, except in
severe PHT, the TR jet is usually characterized by a lower pressure gradient (and
thus lower velocity) than in mitral regurgitation, as a result of the lower RV
systolic pressures. This difference may significantly impact jet analysis [48].
Jet flow, and consequently color Doppler jet area, is mainly driven by
conservation of momentum (flow
Cardiac magnetic resonance (CMR) imaging is currently the gold standard for quantifying RV size and systolic function due to its high spatial resolution and independence from geometric assumptions [51]. Moreover, CMR provides detailed tissue characterization using late gadolinium enhancement and extracellular volume mapping, identifying myocardial scar/fibrosis or infiltrative processes that impact RV function [52]. Recent advances in CMR enable strain-imaging by CMR-derived feature tracking during post-processing, which appears to be an early marker of RV dysfunction [53]. However, despite its well-established advantages in providing detailed and reproducible assessments of RV function, CMR imaging has limitations that hinder widespread clinical adoption. These include limited availability, prolonged acquisition and post-processing times, and higher costs compared to other imaging modalities. A recent survey by the European Association of Cardiovascular Imaging revealed the magnitude of the impact of these limitations: only 7.25% of respondents reported using CMR as the primary imaging modality for assessment of RV function [54].
Cardiac computed tomography (CCT) is not routinely used for the assessment of RV function because of its limited availability, exposure to ionizing radiation, and need for iodinated contrast agents, in particular in patients with significant TR, which can lead to end-organ damage including cardio-renal syndrome. However, CCT offers high three-dimensional spatial resolution, enabling unlimited multiplanar reformats and providing a detailed visualization of the entire right heart, including the RV outflow tract and pulmonary arteries, with an accurate quantification of RV volumes and TA dimensions [55, 56]. A detailed anatomical assessment of the right heart structures, in particular the TA and the surrounding tissues, is crucial when planning transcatheter TV interventions. This requirement has led to increased use of CCT in symptomatic patients with significant TR. In addition to providing good anatomical resolution, RV volumes and EF values derived from CCT have been shown to be strongly correlated with those obtained using CMR. In a study by Tanaka et al. [57], CCT was used to evaluate RVEF in symptomatic patients with severe TR who were undergoing transcatheter TV repair. CCT-derived RV functional parameters provided additional prognostic information beyond that of conventional echocardiographic indices, highlighting the potential role of CCT in the comprehensive pre-procedural evaluation and risk stratification of this patient population.
Historically, nuclear imaging techniques have been the most widely used method for assessing RV function. These techniques provide accurate quantification of RV volumes and ejection fraction, derived from differences in end-diastole and end-systole radionuclide count densities, thereby eliminating the need for the geometric assumptions inherent to other imaging modalities [58, 59]. First-pass and equilibrium radionuclide ventriculography techniques have been extensively validated for this purpose [60, 61]. However, despite some diagnostic robustness, nuclear methods are limited by relatively low temporal resolution and risks associated with exposure to ionizing radiation. In the current era dominated by 3D echocardiography and CMR, the main advantage of nuclear imaging is its ability to provide additional insights into myocardial perfusion and metabolic activity [62].
Echo Doppler is the primary imaging technique for assessing TR and guiding
management decisions. However, massive or torrential TR may hinder the
non-invasive assessment of sPAP, because the Bernoulli equation is not applicable
in such conditions and right atrial pressure is markedly elevated. This effect
leads mainly to an underestimation of sPAP when compared with invasive
measurements [63, 64]. In a series of 243 patients undergoing transcatheter TV
repair, RV systolic pressure values obtained non-invasively and by right heart
catheterization were discordant in 23% of the patients, and a discordant pattern
with invasive sPAP
RV dysfunction in the setting of significant TR independently predicts poor
outcomes, including all-cause mortality, heart failure–related hospitalizations,
and reduced functional capacity. However, large-scale studies validating the
prognostic implications of RV dysfunction in this context are limited [17]. Table 2 (Ref. [9, 18, 38, 41, 44, 53, 57, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92]), provides
an overview of the studies that have assessed the association of different RV
function parameters with outcome. In medically treated patients with significant
TR, approximately two-thirds exhibit RV dysfunction, which is commonly defined as
a TAPSE
| Parameter for RV-function assessment | Population | Outcome |
| Conventional echocardiographic parameters: TAPSE, S′, FAC | ||
| Dietz MF, et al. [82] | 1311 patients with significant ( |
|
| Dietz MF., et al. [68] | 1292 patients with significant ( |
|
| Zornitzki L., et al. [83] | 1143 patients with significant ( |
|
| Galloo X., et al. [9] | 278 patients with significant TR undergoing TV surgery | |
| Vogelhuber J., et al. [69] | 262 patients with symptomatic TR undergoing TEER | |
| Rodríguez Torres DJ., et al. [18] | 70 patients undergoing cardiac and TV surgery | |
| 2D Speckle tracking echocardiography: RV GLS and RV FWLS | ||
| Prihadi E., et al. [71] | 896 patients with significant ( |
|
| Ogawa M., et al. [84] | 53 patients with severe atrial secondary TR associated with atrial fibrillation | |
| Ogawa M., et al. [72] | 377 patients with severe secondary TR | |
| Curtis E., et al. [73] | 262 consecutive patients undergoing echocardiography and right-heart catheterization on the same day. | |
| Hinojar R., et al. [74] | 151 patients with severe secondary TR and no formal indication for TV intervention. | |
| Ancona F., et al. [85] | 79 consecutive patients with severe TR undergoing isolated TV surgery | |
| Kim M., et al. [86] | 115 patients with severe secondary TR who underwent isolated TV surgery | |
| 3D Echocardiography | ||
| Tomaselli M., et al. [87] | 554 patients with moderate and severe secondary TR, under medical treatment | |
| Ladányi Z., et al. [75] | 205 consecutive adult patients referred for echocardiography with secondary TR | |
| Badano L., et al. [88] | 758 patients with moderate-to-severe secondary TR | |
| Tomaselli M., et al. [76] | 513 patients with moderate and severe secondary TR | |
| Formula: EROA corrected for PISA | ||
| EROAc = 6.28 |
||
Clement A., et al. [38] Formula eRVEF = RV forward SV/RV ESV |
513 patients with first echocardiographic diagnosis of mild to severe secondary TR | |
| Orban M., et al. [89] | 75 patients with severe TR undergoing TV-TEER | |
| Multi-modality imaging | ||
| 1. Cardiac magnetic resonance | ||
| Hinojar R., et al. [77] | 75 patients with significant TR ( |
|
| Romano S., et al. [53] | 544 consecutive patients with severe secondary TR undergoing CMR | |
| Park JB., et al. [90] | 75 patients with severe secondary TR | |
| Kresoja KP., et al. [70] | 79 patients with severe TR undergoing TTVR | |
| 2. Cardiac Computed Tomography | ||
| Tanaka T., et al. [57] | 157 symptomatic patients with TR who underwent CCT before TTVR | |
| Kirchner J., et al. [91] | 100 patients with severe TR undergoing TTVR | |
| Novel measures of RV function | ||
| 1. Echocardiographic RV-PA coupling | ||
| 1.1. TAPSE/sPAP | ||
| Fortuni F., et al. [41] | 1149 patients with |
|
| Brener M., et al. [44] | 444 patients undergoing transcatheter TV intervention | |
| Stolz L., et al. [67] | 848 patients who underwent TV-TEER | |
| Sugiura A., et al. [92] | 206 patients who underwent TV-TEER | |
| 1.2. TAPSE/RVFWLS | ||
| Ancona F., et al. [78] | 250 consecutive patients with severe TR | |
| 1.3. 3DE-derivde RV-PA-coupling | ||
| Gavazzoni M., et al. [79] | 108 patients with moderate or severe secondary TR | |
| Formula = RV forward SV/RV ESV | ||
| 2. STREI-index | ||
| Hinojar R., et al. [80] | 176 consecutive patients with isolated |
|
| Formula = [2 |
||
| 3. RV Contractile reserve | ||
| Utsunomiya H., et al. [81] | 36 patients with severe secondary TR | |
Abbreviations: 2D, two-dimensional; 3D, three-dimensional; CT, cardiac computed tomography; CMR, cardiac magnetic resonance imaging; EF, ejection fraction; EROA, effective regurgitant orifice area; eRVEF, effective right ventricular ejection fraction; ESV, end-systolic volume; FAC, fractional area change; FWLS, free wall longitudinal strain; GLS, global longitudinal strain; HF, heart failure; PA, pulmonary artery; PISA, proximal isovelocity surface area; RASr, right atrial reservoir strain; ROC, receiver operating characteristic; RV, right ventricle; S’, tissue Doppler imaging S’; sPAP, systolic pulmonary artery pressure; SV, stroke volume; TAPSE, tricuspid annular plane systolic excursion; TEER, transcatheter edge-to-edge repair; TTVR, transcatheter tricuspid valve repair; TV, tricuspid valve.
Among patients with STR, RVFWLS identifies RV dysfunction in approximately 85% of patients (versus 72% by TAPSE, and 49% by FAC) [71]. RVFWLS independently predicts all-cause mortality and provides additional prognostic value beyond that of TAPSE, FAC, and TR severity [71, 72, 73, 74]. Reduced 3DE-derived RVEF is associated with higher mortality and cardiac death across various cardiovascular disease cohorts [8, 93]. In STR, 3DE reveals significant RV remodeling and different contraction patterns as TR severity increases, with a decline in longitudinal shortening, whereas radial and anteroposterior contractions remain stable. Radial shortening, in turn, correlates with prognosis [75]. Furthermore, assessment of effective RVEF (which takes regurgitant volume into account) using 3DE has a stronger association with mortality and heart failure hospitalization than standard 3DE-derived RVEF [76].
Multimodality imaging further improves prognostic accuracy. In prospective CMR
studies of severe TR, effective RVEF and feature-tracking-derived RVFWLS
independently predicted death, in addition to clinical and other imaging risk
factors [53, 77]. Similarly, CT-derived RVEF (
Decision-making in the management of TR involves the assessment of various factors, including patient-related, anatomical, hemodynamic, and RV function aspects. Patient-related factors encompass comorbidities, age and life expectancy, quality of life, and rehabilitation capacity. Anatomical factors involve the determination of primary vs secondary vs cardiac implantable electronic device-related-TR, leaflet morphology, and the location of the jet. Because echocardiography often underestimates pulmonary pressures in the presence of significant TR, invasive assessment of pulmonary arterial pressure as an estimate of RV afterload and of pulmonary vascular resistance to rule out precapillary PHT is mandatory. Fig. 4 shows an algorithm guiding clinical decision making for medical therapy or surgical TV intervention, based on the assessment of pulmonary pressure and RV function.
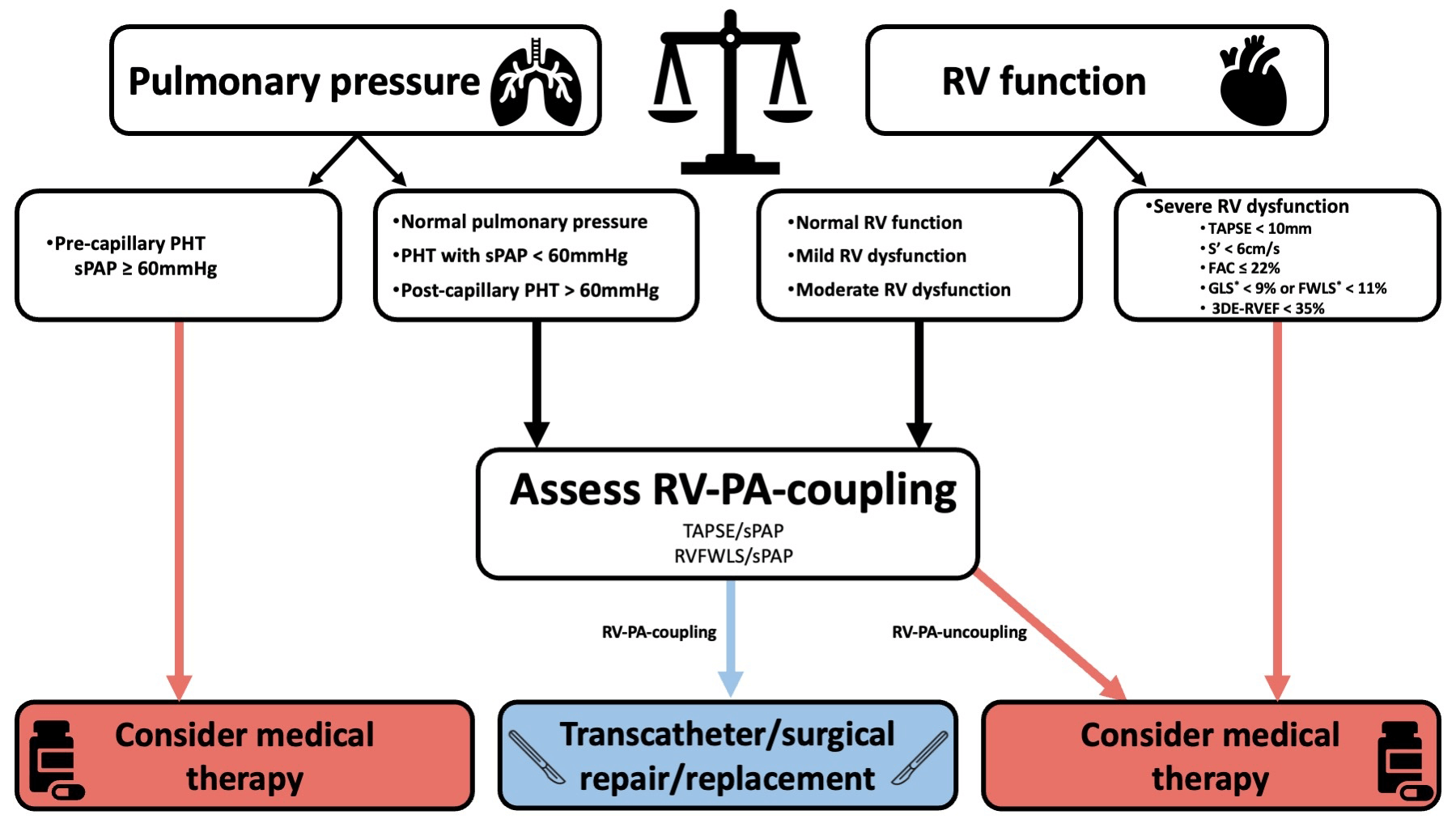 Fig. 4.
Fig. 4.
Proposed algorithm for clinical decision making integrating the assessment of pulmonary pressure and RV function. *expressed as absolute (i.e., positive) values. The exact cutoffs to define RV-PA-uncoupling may vary according to the selected patient population (these values are discussed in chapter 5 - Echocardiographic Assessment of RV Function). Abbreviations: 3DE, three-dimensional echocardiography; FAC, fractional area change; FWLS, free wall longitudinal strain; GLS, global longitudinal strain; PHT, pulmonary hypertension; RV, right ventricle; RVEF, right ventricular ejection fraction; RV-PA-coupling, right ventricular to pulmonary artery coupling; sPAP, systolic pulmonary arterial pressure; TAPSE, tricuspid annular plane systolic excursion.
Elevated pre-capillary pulmonary arterial systolic pressure
| RV function parameter | RV dysfunction | Severe RV dysfunction |
| TAPSE (mm) | ||
| RV TDI s’ (cm/s) | ||
| RV GLS* (%) | ||
| RV FWLS* (%) | ||
| 3D RVEF (%) | ||
| FAC (%) |
* Values are expressed as absolute (i.e., positive) values.
Abbreviations: 3D, three-dimensional; EF, ejection fraction; FAC, fractional area change; FWLS, free wall longitudinal strain; GLS, global longitudinal strain; RV, right ventricule; TAPSE, tricuspid annular plane systolic excursion; TDI, tissue Doppler imaging.
Assessing procedural risk and determining the optimal timing for referral of patients with significant TR remain challenges in clinical practice for. Despite advances in our understanding of RV and TR pathophysiology and growing experience with TV surgery, in-hospital mortality remains approximately 10% for isolated TV surgical intervention [96, 97]. Current recommendations state that patient evaluation and TV intervention should be performed in a euvolemic state, as right-sided or biventricular congestion has been associated with poorer outcomes, including lower procedural success rates and reduced survival [98]. Conventional surgical risk models, such as the European System for Cardiac Operative Risk Evaluation (EuroSCORE) [99, 100] and the Society of Thoracic Surgeons (STS) score [101], have been widely used in this context. However, the original validation cohorts of these models included a limited proportion of patients with significant TR. Consequently, these models demonstrate limited predictive accuracy in this population. Recently, several research groups have developed TR-specific risk scores for patients with severe TR who are managed medically, surgically, or with transcatheter interventions. In patients with severe TR managed conservatively, dedicated risk scores have been developed and demonstrated prognostic value; both incorporated RV function as a key variable [102, 103]. Several other risk models have been evaluated and shown to be predictive for those undergoing surgical or transcatheter TV intervention. These models include the Model for End-Stage Liver Disease (MELD) score, TRI-SCORE, TRIVALVE score, and the dedicated STS Adult Cardiac Tricuspid Valve Surgery Risk Calculator [104, 105, 106, 107, 108]. Notably, among these models, only the TRI-SCORE includes an assessment of RV function (Table 4), highlighting the limited integration of this critical parameter into existing risk stratification tools for this population. The TRI-SCORE was initially developed to predict in-hospital mortality in patients undergoing isolated TV surgery and demonstrated superior prognostic performance compared to the EuroSCORE I and II (area under the curve 81.7% vs. 66.8% and 62.9%, respectively). Based on the total score, patients can be categorized into low-, intermediate-, and high-risk strata, each associated with markedly different surgical mortality rates [105]. The TRI-SCORE was subsequently evaluated in the TRIGISTRY, a multicenter registry of 2414 patients with severe isolated secondary TR. Survival declined progressively with increasing TRI-SCORE values, irrespective of the therapeutic strategy. Moreover, among patients with low or intermediate risk, early and successful surgical or transcatheter intervention was associated with superior 2-year survival compared to conservative management [106]. More recently, the TRI-SCORE has also been validated across multiple cohorts undergoing transcatheter TV repair, further supporting its applicability and predictive value in this population [109].
| TRI-SCORE | |
| Risk factors | Scoring |
| Age |
1 |
| NYHA functional class III or IV | 1 |
| Right-sided heart failure signs | 2 |
| Daily dose of furosemide |
2 |
| Glomerular filtration rate |
2 |
| Elevated total bilirubin | 2 |
| Left ventricular ejection fraction |
1 |
| Moderate/severe right ventricular dysfunction | 1 |
| FINAL TRI-SCORE | 12 |
Abbreviations: NYHA, New York Heart Association function class.
RV function is a critical determinant and prognostic marker in patients with significant TR. Its careful assessment is mandatory for optimizing the management strategy. However, while several imaging and hemodynamic parameters have been used to characterize RV function in patients with TR, there is currently a lack of clear cutoff values and prospective validation. Further advances in the assessment of RV function will substantially improve this field, including progress in imaging technology, the integration of artificial intelligence into the daily clinical workflow, and new biomarker discovery.
Integration of novel echocardiographic parameters such as RV myocardial work [110] and segmental strain analysis holds promise for improved RV characterization and earlier detection of subclinical RV dysfunction. Additionally, CCT, particularly with newer, high-resolution, low-radiation protocols, is emerging as a viable modality for RV assessment in patients undergoing structural heart interventions [55, 111]. Furthermore, alternative metrics, such as fast-SENC intramyocardial strain, a unique CMR modality that measures intramyocardial RV contraction in 1 heartbeat per image plane, have been shown to detect subclinical RV dysfunction well before changes in RVEF; this measurement needs further clinical validation [112].
New artificial intelligence-driven algorithms are showing potential for automating and improving RV functional analysis across multiple imaging modalities. Deep learning models can segment the RV with high precision, enabling consistent quantification of RV volumes, ejection fraction, and advanced imaging modalities such as RV speckle-tracking imaging, while minimizing interobserver variability and enhancing clinical efficiency [113, 114]. Furthermore, integrating multi-parametric data from CMR, including late gadolinium enhancement, T1/T2 mapping, and feature tracking, into machine learning models may facilitate phenotypic classification and risk stratification beyond conventional metrics [115, 116].
Alongside imaging advancements, novel circulating biomarkers, such as soluble ST2 and GDF-15 [117], galectin-3 [118], and extracellular vesicle profiles, are being investigated for their ability to reflect subclinical RV myocardial remodeling and fibrosis.
The convergence of AI-enhanced imaging and biomarker-based precision phenotyping is expected to transform the assessment of RV function. This convergence will enable the earlier identification of maladaptive remodeling, more accurate risk stratification, and improved timing and appropriate choice of interventions in patients with valvular heart disease.
AHA/ACC, American Heart Association/American College of Cardiology; CCT, Cardiac computed tomography; CMR, Cardiac magnetic resonance imaging; EF, Ejection fraction; ESC, European Society of Cardiology; FAC, Fractional area change; FWLS, Free wall longitudinal strain; GLS, Global longitudinal strain; LV, Left ventricle/left ventricular; RA, Right atrium; RV, Right ventricle/right ventricular; RVPAC, Right ventricular to pulmonary artery coupling; S′, tissue Doppler-derived systolic velocity of the lateral tricuspid annulus; sPAP, systolic pulmonary arterial systolic pressure; TAPSE, Tricuspid annular plane systolic excursion; TR, Tricuspid regurgitation; TV, Tricuspid valve.
XG and PU both designed the outlines of the review, performed the research, analyzed the data and contributed both equally to writing the manuscript. PU supervised and revised the manuscript. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors take full responsibility for the final published version.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




