1 The Xuzhou Clinical College of Xuzhou Medical University, 221000 Xuzhou, Jiangsu, China
2 Department of Cardiology, The Affiliated Shuyang Hospital of Xuzhou Medical University, 223600 Suqian, Jiangsu, China
3 Department of Cardiology, Xuzhou Central Hospital, 221002 Xuzhou, Jiangsu, China
Abstract
The HAVOC score is an emerging tool for estimating the risk of atrial fibrillation (AF), which has attracted growing interest. However, the use of the HAVOC score to predict in-hospital new-onset AF (NOAF) among patients with ST-segment elevation myocardial infarction (STEMI) remains unclear. Therefore, this study aimed to examine whether the HAVOC score is associated with NOAF during the index hospitalization following primary percutaneous coronary intervention (PCI) in patients with a STEMI.
We studied a consecutive cohort of patients presenting with STEMI from January 2023 to March 2025. After primary PCI, each participant underwent continuous electrocardiogram monitoring for at least 72 hours. The HAVOC score was calculated based on hypertension, age ≥75 years, valvular heart disease, peripheral vascular disease, obesity, and heart failure.
In total, 725 patients were analyzed, with a mean age of 63.37 ± 13.16 years; of whom 72.97% were male. During the hospital stay, 70 patients (9.66%) experienced NOAF. Multivariate logistic regression analysis showed that the HAVOC score (odds ratio (OR) = 1.42, 95% confidence interval (CI): 1.28–1.59) was independently associated with NOAF. Restricted cubic spline (RCS) analysis revealed a linear dose–response relationship between the HAVOC score and NOAF (p for overall <0.001). Integrating left ventricular ejection fraction (LVEF) and the presence of left anterior descending artery stenosis enhanced the discriminatory ability of the HAVOC score for identifying NOAF (net reclassification index [NRI] = 0.353, 95% CI: 0.114–0.592; p = 0.004) and improved integrated discrimination (0.024, 95% CI: 0.006–0.041; p = 0.008).
Higher HAVOC scores were independently linked to the occurrence of in-hospital NOAF among STEMI patients following PCI. NOAF risk increased with the HAVOC score, consistent with a linear dose–response across the score spectrum.
Keywords
- cardiovascular disease
- HAVOC score
- atrial fibrillation
- ST-segment elevation myocardial infarction
ST-segment elevation myocardial infarction (STEMI) is a common yet highly lethal presentation associated with cardiovascular disease [1]. Although there has been substantial progress in early recognition, reperfusion techniques, and secondary prevention, complications continue to occur both during the index admission and following hospital discharge [1, 2]. Atrial fibrillation (AF) is often precipitated by acute ischemic injury and necrosis of myocardial tissue during STEMI, which alter the electrophysiological properties of the heart [3, 4]. Previous studies have indicated that the incidence of new-onset AF (NOAF) in STEMI patients can be as high as 20% and is closely associated with poor prognosis [5, 6]. Currently, there is controversy regarding the optimal prevention and treatment strategies for NOAF in STEMI patients during their hospitalization [7]. Therefore, identifying the risk factors associated with NOAF, accurately stratifying high-risk patients, and formulating effective preventive and therapeutic approaches are crucial for improving the clinical outcomes of STEMI patients.
Tools designed to predict NOAF in the STEMI population are still scarce. The
HAVOC Score—comprising Hypertension, Age
This study retrospectively enrolled STEMI [15] patients who received treatment at the Xuzhou Central Hospital between January 2023 and March 2025. The inclusion criteria were as follows: (1) Successful completion of primary PCI (thrombolysis in myocardial infarction [TIMI]= 3). (2) All patients underwent continuous electrocardiogram (ECG) monitoring (telemetry) for at least 72 hours post-PCI. (3) Complete clinical data were available. The exclusion criteria were: (1) History of previous myocardial infarction (MI). (2) History of previous AF or atrial flutter. (3) Presence of thyroid dysfunction. (4) Presence of inflammatory diseases. (5) Presence of hematological diseases or malignant tumors. The study protocol was reviewed and approved by the Institutional Review Board (IRB) of the Xuzhou Central Hospital (XZXY-LK-20250317-0034), and all procedures were conducted in accordance with the ethical principles of the Declaration of Helsinki. This retrospective analysis involved no harm to the participants; therefore, the IRB authorized a waiver of informed consent. In total, 725 individuals were ultimately enrolled in the present study (Fig. 1).
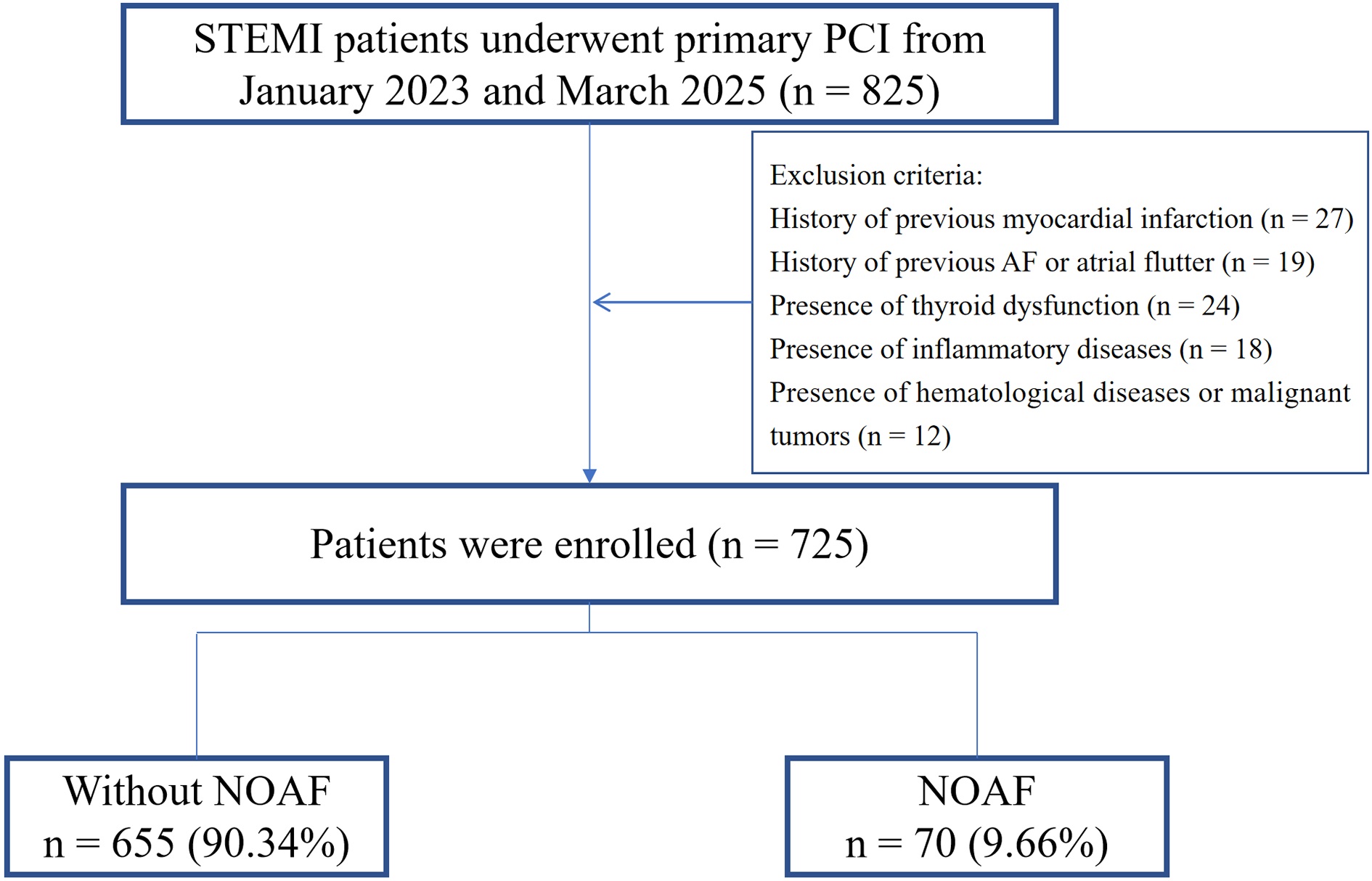 Fig. 1.
Fig. 1.
Study flowchart. STEMI, ST-segment elevation myocardial infarction; NOAF, new-onset atrial fibrillation; AF, atrial fibrillation; PCI, percutaneous coronary intervention.
All research data were systematically collected from the electronic medical
record system. The collected information included: Demographic characteristics
and the use of medications. Comorbidities including heart failure (HF),
hypertension, diabetes mellitus, peripheral vascular disease, and valvular heart
disease were recorded. Laboratory tests including electrolytes, lipid profile,
high-sensitivity C-reactive protein (hs-CRP), troponin I (TNI), and N-terminal
pro-brain natriuretic peptide (NT-proBNP) were included. Information about the
infarct-related artery (IRA) was determined based on the results of coronary
angiography. The HAVOC Score was calculated according to hypertension (2 points),
age
SPSS version 27.0 (IBM, Chicago, IL, USA) and R 4.4.3 (Lucent Technologies, New
Jersey, USA) were used to perform statistical analyses. The normality of
continuous variables was assessed using the Kolmogorov–Smirnov test. Continuous
data were expressed as mean
In total, 725 patients were analyzed, with a mean age of 63.37
| Variables | Total (n = 725) | Non-NOAF (n = 655) | NOAF (n = 70) | p | |
| Age, years | 63.37 |
62.14 |
74.90 |
||
| Male, n (%) | 529 (72.97) | 484 (73.89) | 45 (64.29) | 0.085 | |
| BMI, kg/m2 | 24.63 |
24.60 |
24.97 |
0.503 | |
| Heart rate, bpm | 79.40 |
79.18 |
81.50 |
0.185 | |
| SBP, mmHg | 126.68 |
126.78 |
125.73 |
0.755 | |
| DBP, mmHg | 78.75 |
78.97 |
76.71 |
0.186 | |
| Smoking, n (%) | 326 (44.97) | 297 (45.34) | 29 (41.43) | 0.531 | |
| HF, n (%) | 375 (51.72) | 320 (48.85) | 55 (78.57) | ||
| Valvular disease, n (%) | 121 (16.69) | 106 (16.18) | 15 (21.43) | 0.263 | |
| PVD, n (%) | 556 (76.69) | 501 (76.49) | 55 (78.57) | 0.695 | |
| Hypertension, n (%) | 300 (41.38) | 264 (40.31) | 36 (51.43) | 0.072 | |
| Diabetes, n (%) | 168 (23.17) | 148 (22.60) | 20 (28.57) | 0.260 | |
| Stroke, n (%) | 84 (11.59) | 75 (11.45) | 9 (12.86) | 0.727 | |
| HAVOC Score | 4.48 |
4.25 |
6.71 |
||
| TC, mmol/L | 4.41 |
4.43 |
4.26 |
0.191 | |
| Triglycerides, mmol/L | 1.47 |
1.46 |
1.63 |
0.155 | |
| LDL-C, mmol/L | 2.75 |
2.77 |
2.56 |
0.060 | |
| HDL-C, mmol/L | 1.05 |
1.06 |
1.00 |
0.080 | |
| TNI, ng/mL | 14.12 (3.01, 46.54) | 13.45 (2.87, 46.50) | 20.27 (7.86, 49.95) | 0.055 | |
| NT-proBNP, pg/mL | 2011.1 (1020.0, 4075.5) | 1913.2 (984.9, 3835.0) | 3779.6 (2280.5, 6881.7) | ||
| hs-CRP, mg/L | 26.40 (9.10, 71.00) | 25.60 (8.70, 67.15) | 33.60 (11.43, 113.50) | 0.046 | |
| Na+, mmol/L | 139.77 |
139.82 |
139.31 |
0.220 | |
| K+, mmol/L | 3.92 |
3.91 |
3.99 |
0.160 | |
| Ca+, mmol/L | 2.24 |
2.24 |
2.25 |
0.403 | |
| Aspirin, n (%) | 684 (94.34) | 619 (94.50) | 65 (92.86) | 0.768 | |
| P2Y12 inhibitors, n (%) | 712 (98.21) | 642 (98.02) | 70 (100.00) | 0.474 | |
| 603 (83.17) | 542 (82.75) | 61 (87.14) | 0.350 | ||
| Statins, n (%) | 697 (96.14) | 629 (96.03) | 68 (97.14) | 0.894 | |
| ACEI/ARB, n (%) | 396 (54.62) | 356 (54.35) | 40 (57.14) | 0.656 | |
| Left atrial diameter, mm | 39.30 |
39.20 |
40.31 |
0.181 | |
| LVEF, % | 51.44 |
51.74 |
48.70 |
0.007 | |
| Killip, n (%) | 0.004 | ||||
| I | 615 (84.83) | 565 (86.26) | 50 (71.43) | ||
| II | 48 (6.62) | 40 (6.11) | 8 (11.43) | ||
| III | 21 (2.90) | 15 (2.29) | 6 (8.57) | ||
| IV | 41 (5.66) | 35 (5.34) | 6 (8.57) | ||
| Length of stay, days | 5.56 |
5.51 |
6.03 |
0.203 | |
| LAD, n (%) | 364 (50.21) | 336 (51.30) | 28 (40.00) | 0.072 | |
| LCX, n (%) | 92 (12.69) | 81 (12.37) | 11 (15.71) | 0.424 | |
| RCA, n (%) | 249 (34.34) | 221 (33.74) | 28 (40.00) | 0.295 | |
| LM, n (%) | 20 (2.76) | 17 (2.60) | 3 (4.29) | 0.662 | |
BMI, body mass index; PVD, peripheral vascular disease; TC, total cholesterol; LVEF, left ventricular ejection fraction; SBP, systolic blood pressure; DBP, diastolic blood pressure; LAD, left anterior descending; LCX, left circumflex artery; RCA, right coronary artery; LM, left main; ACEI, angiotensin-converting-enzyme inhibitor; ARB, angiotensin II receptor blocker; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; hs-CRP, high sensitivity C-reactive protein; TNI, troponin I; NT-proBNP, N-terminal pro-B-type natriuretic peptide; HF, heart failure.
Univariate logistic regression analysis indicated that the HAVOC Score, HF, age,
LVEF, Killip class
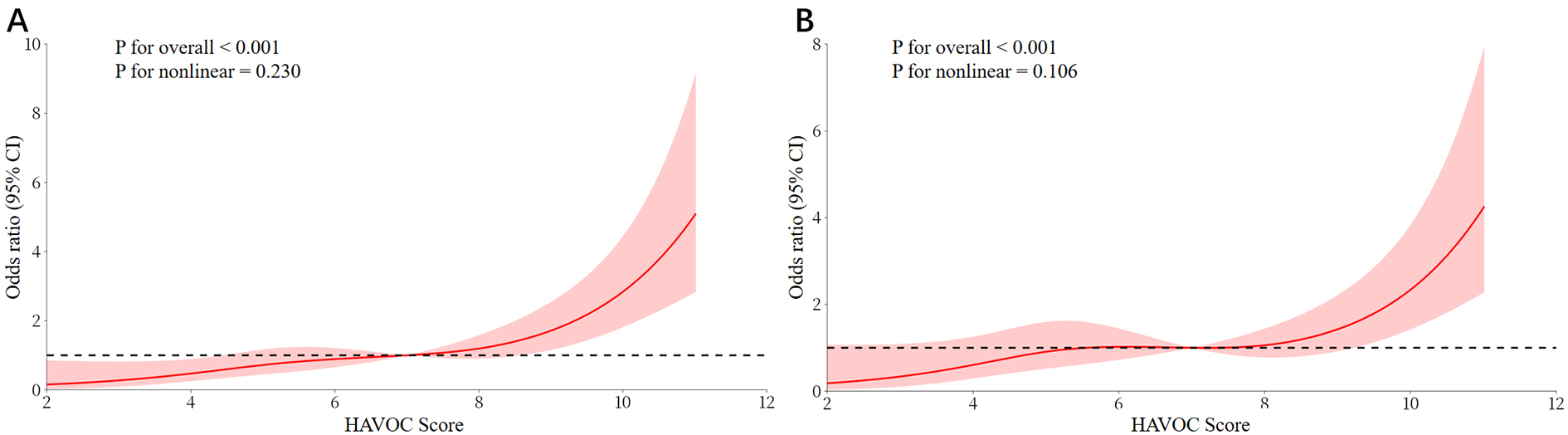 Fig. 2.
Fig. 2.
The RCS models for HAVOC score and NOAF. (A) Unadjusted dose-response curve showing the association between HAVOC score and risk of NOAF. (B) Adjusted dose-response curve after controlling for the risk factors.
| Variables | OR (95% CI) | p | |
| HAVOC Score | 0.35 (0.06) | 1.42 (1.28–1.59) | |
| LVEF, % | –0.04 (0.02) | 0.96 (0.93–1.00) | 0.031 |
| LAD, n (%) | –0.73 (0.27) | 0.48 (0.28–0.83) | 0.008 |
OR, odds ratio; CI, confidence interval; SE, standard error.
The ROC curve showed that the AUC of the HAVOC Score was 0.738 (95% CI:
0.677–0.798, p
| AUC | 95% CI | p | Cut-off | Sensitivity | Specificity | |
| HAVOC Score | 0.738 | 0.677–0.798 | 5.50 | 0.729 | 0.673 | |
| HAVOC Score + LVEF + LAD | 0.762 | 0.700–0.824 | - | 0.643 | 0.773 |
AUC, area under the curve; ROC, receiver operating characteristic.
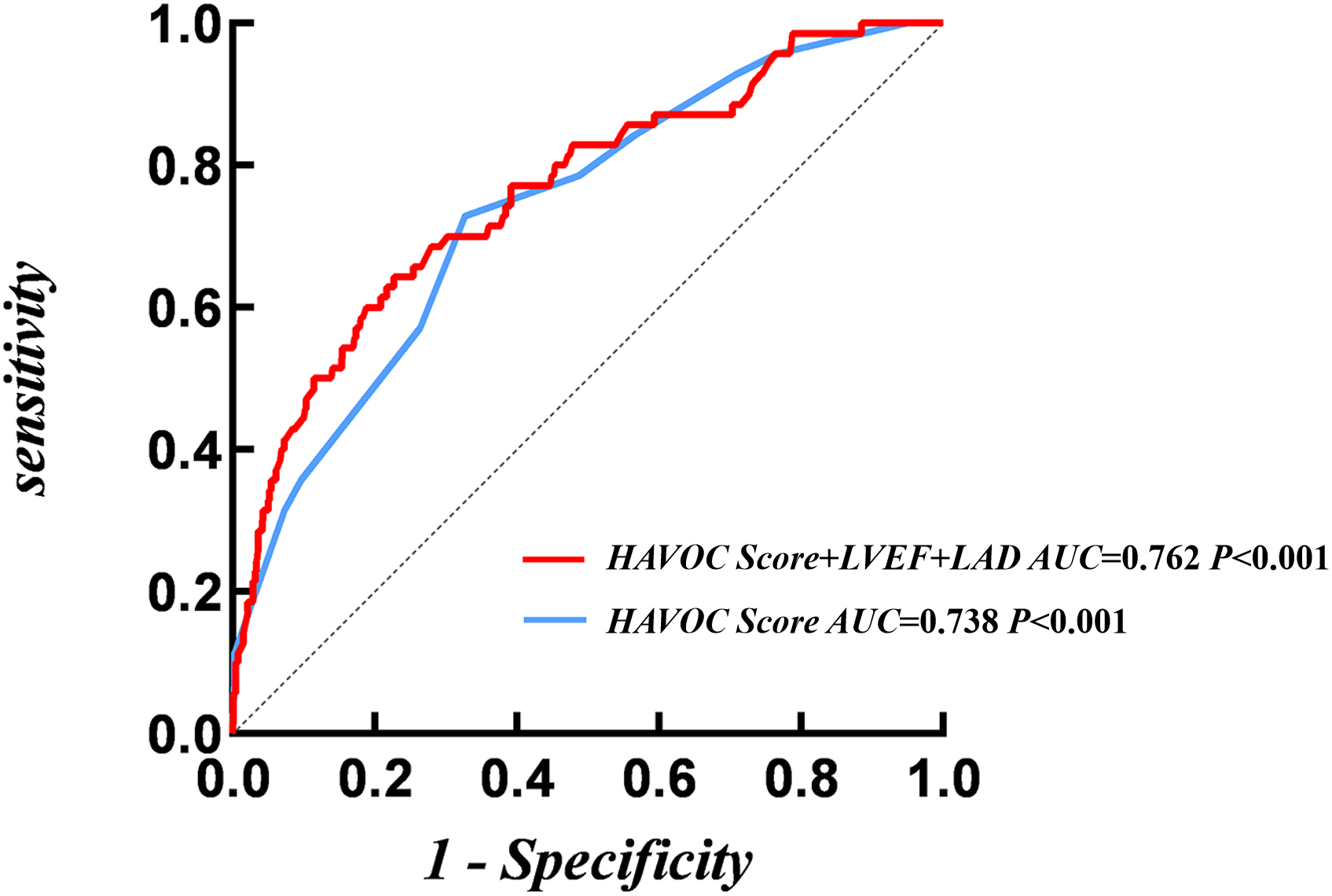 Fig. 3.
Fig. 3.
ROC analysis of HAVOC score for NOAF.
The results of IDI and NRI revealed that the NRI was 0.353 (95% CI: 0.114–0.592, p = 0.004), and the IDI was 0.024 (95% CI: 0.006–0.041, p = 0.008). These findings suggested that the new model could significantly enhance the discriminatory ability of the HAVOC Score for identifying NOAF (Table 4).
| NRI | IDI | |||
| Estimate (95% CI) | p | Estimate (95% CI) | p | |
| LVEF + LAD | Reference | - | Reference | - |
| HAVOC Score + LVEF + LAD | 0.353 (0.114–0.592) | 0.004 | 0.024 (0.006–0.041) | 0.008 |
IDI, integrated discrimination improvement.
Given the relatively small number of female participants, the subgroup analysis
was restricted to male patients. In the univariate regression analysis, Na+,
Ca+, HF, Killip class
| Variables | OR (95% CI) | p | |
| HAVOC Score | 0.27 (0.08) | 1.31 (1.12–1.53) | |
| LVEF, % | –0.06 (0.02) | 0.95 (0.90–0.99) | 0.016 |
| NT-proBNP, pg/mL | 0.92 (0.44) | 2.51 (1.06–5.99) | 0.038 |
LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal pro-brain natriuretic peptide.
This study is the first to evaluate the association between the HAVOC Score and in-hospital NOAF in STEMI patients undergoing PCI. We found that higher HAVOC values independently predicted NOAF, the risk increased in an approximately linear dose–response manner with increasing scores.
A recent report found that almost 70% of all AF patients have underlying CAD and that management of underlying CAD often results in a reduced burden of AF [17]. The impact of CAD on endothelial dysfunction can be another factor responsible for this effect as it results in inflammation which directly promotes AF [18, 19, 20]. NOAF following PCI in patients with STEMI represents a crucial clinical issue that significantly impacts patient outcomes [3, 4, 5, 6]. Although anticoagulant therapy has the potential to reduce the risk of death, it necessitates a careful balance against the risk of bleeding [7]. Despite existing studies having clearly defined the clinical characteristics and prognostic implications of NOAF, there is still a need for further exploration regarding the generalizability of risk models across different populations and the clinical translation of novel biomarkers.
The HAVOC Score, a novel assessment tool, includes multiple risk factors associated with AF. In patients with embolic stroke of undetermined source, a low HAVOC Score has been demonstrated to be associated with a low incidence of AF [8]. Among patients who have experienced cryptogenic stroke or transient ischemic attack, different HAVOC Scores can be utilized to determine varying levels of the risk of AF [9]. In another study focusing on patients with cryptogenic stroke, the HAVOC Score has proven capable of accurately identifying individuals at risk of AF during long-term monitoring [10]. Research on the HAVOC Score in patients with STEMI is still in its initial stage. Our study reveals that the HAVOC Score was independently associated with NOAF. In addition, we found a linear dose-response relationship between the HAVOC Score and NOAF. Hypertension can lead to structural and electrophysiological remodeling of the atrium through pressure overload [21]. After STEMI, myocardial ischemia further exacerbates the atrial load. In patients with hypertension, the process of atrial remodeling is accelerated, significantly increasing the risk of NOAF [22, 23]. It is well-known that myocardial fibrosis and autonomic nervous system dysfunction are the core mechanisms underlying the development of cardiovascular diseases in elderly patients [24]. In elderly patients, there is an increased apoptosis of atrial myocytes, and fibrous connective tissue replaces normal myocardium, resulting in a decrease of atrial conduction velocity and an increase in the dispersion of the refractory period [25, 26]. Valvular heart disease can cause diseases through hemodynamic disorders and abnormal atrial mechanical stress [27, 28]. Peripheral vascular disease is associated with systemic atherosclerosis and endothelial dysfunction, both of which are important risk factors promoting AF [29, 30, 31]. In obese patients, excessive accumulation of adipose tissue leads to insulin resistance and increased leptin levels, activating the RAS and the sympathetic nervous system, and inducing apoptosis and fibrosis of atrial myocytes. Inflammatory factors secreted by visceral fat directly participate in myocardial remodeling, leading to conduction heterogeneity [32, 33]. After STEMI, disorders of myocardial energy metabolism in obese patients may delay the repair process, reducing the tolerance of atrial myocytes to ischemic injury and significantly increasing the risk of NOAF [13]. HF has been proven in various diseases to be an important inducer of AF through atrial dilation and neurohumoral activation [34, 35]. In contrast to other diseases, STEMI patients often suffer from extremely severe myocardial ischemia, which can directly affect atrial function. During acute STEMI, ischemic injury can directly activate oxidative stress and the inflammatory response in atrial myocytes, inducing atrial arrhythmias [36]. The blood supply to the atrium is mainly provided by the right coronary artery (RCA) and the left circumflex artery (LCX) [37]. Therefore, when the culprit vessel is the RCA or LCX, it is often accompanied by atrial ischemia, increasing the risk of AF. In contrast, when the culprit lesion involves the LAD, the risk or extent of atrial ischemia may be lower, which may explain the lower incidence of NOAF observed in patients with LAD-related myocardial infarction. In our study, the LAD was demonstrated to be an independent protective factor for AF. The previous study has demonstrated a close association between C-reactive protein (CRP) levels and adverse outcomes in AF [38]. In a previous study, CRP was also identified as an independent risk factor for NOAF in patients with STEMI [39]. In contrast, although our study demonstrated that CRP levels were significantly elevated in the NOAF group, CRP was not an independent factor for NOAF after adjustment for confounding factors. This discrepancy may be partly explained by the inclusion of the HAVOC Score in our multivariable model, as several of its components are closely related to inflammation. The association between CRP and NOAF in STEMI patients remains controversial [40]. Therefore, further studies with more targeted and mechanism-based designs are warranted to clarify this relationship.
In our study, when the HAVOC Score is combined with LVEF and LAD, the results of IDI and NRI, suggest a significant enhancement in the ability to identify NOAF in STEMI patients after PCI. Impaired cardiac function and myocardial ischemia, which distinguish STEMI from other diseases, are key contributors to NOAF. Therefore, our study may provide more information beyond the HAVOC Score for the risk stratification of NOAF in STEMI patients. In future research, it may be advisable to consider incorporating LVEF and LAD into the HAVOC Score to develop and validate a scoring model for NOAF in STEMI patients, enabling more accurate identification of high-risk patients and achieving individualized risk stratification. This integrative approach may help clinicians to refine post-PCI management strategies, such as closer rhythm monitoring and optimization of cardioprotective therapy. In addition, to enhance clinical applicability, future studies could perform risk stratification based on the HAVOC Score, dividing patients into low-, moderate-, and high-risk groups. However, the larger, prospective cohort studies, are still needed to validate the corresponding cut-off values and the predictive performance of this stratification approach.
First, this is a single-center retrospective study, and there may be some unavoidable biases. Some potential confounders that may influence the risk of AF, such as infarct size and delay in reperfusion, were not controlled for; therefore, our findings should be interpreted in this context. Second, our study focused on the events of NOAF during the hospitalization period. Whether there are differences in the long-term prognosis of these patients and whether the HAVOC Score can predict NOAF in the long term requires further clarification. Third, this study was conducted in a single-center Chinese population, which may limit the generalizability of the findings to other ethnic or demographic groups. Fourth, our ascertainment of AF was based on the standard ECG and at least 72 hours of ECG monitoring, rather than continuous rhythm surveillance covering the entire hospital stay. This approach may have led to a non-differential misclassification of the outcome by potentially under-detecting cases of paroxysmal or subclinical AF.
Higher HAVOC Scores were independently associated with a greater risk of in-hospital NOAF among STEMI patients after PCI, showing an approximately linear dose–response relationship. These findings highlight the HAVOC Score as a practical tool for early identification of patients at increased risk for NOAF and may result in improved monitoring and preventive strategies in clinical practice.
The datasets used and analyzed during the present study are available from the corresponding author on reasonable request.
SZ, and WL made substantial contributions to the conception and design of the work. SZ, DC, XG and ZW made substantial contributions to the acquisition of data. SZ, XG, and WL revised the manuscript. SZ, and DC analyzed the data and produced the figures. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Institutional Review Board (IRB) of Xuzhou Central Hospital (Approval No. XZXY-LK-20250317-0034) and was conducted in strict accordance with the principles of the Declaration of Helsinki. Given that this was a retrospective study and posed no risk to patients, the requirement for informed consent was waived with the approval of the IRB.
Not applicable.
This work was partly supported by the Xuzhou Commission of Health (Grant No. XWKYHT20200056).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM47061.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



