1 Langfang Core Laboratory of Precision Treatment of coronary artery disease, Langfang People's Hospital, 065000 Langfang, Hebei, China
Abstract
To investigate the effect of dual antiplatelet therapy (DAPT) guided by platelet function testing (PFT) on the prognosis of patients with acute coronary syndrome (ACS) at a high risk for ischemia and bleeding who underwent percutaneous coronary intervention (PCI).
A retrospective analysis was conducted on 1816 patients with ACS and a dual high risk who underwent PCI at a single center from March 2017 to November 2022. Patients were stratified into the guided DAPT group (n = 712) and standard DAPT group (n = 1104) according to whether the patient received PFT. All patients received oral DAPT for a duration of 12 months post-PCI. The deadline for the endpoint was within 12 months of receiving PCI. The primary endpoint was the number of net clinical adverse events (NACEs) that occurred during follow-up, including the composite endpoint of major adverse cardiovascular and cerebrovascular events (MACCEs) and bleeding, as defined by the bleeding academic research consortium (BARC) (type 3 or greater).
Compared with the standard DAPT group, the guided DAPT group exhibited a lower incidence of NACEs (4.8% vs. 8.7%; p = 0.001), MACCEs (3.9% vs. 6.7%; p = 0.017), cardiac death (0.4% vs. 1.5%; p = 0.038), and stroke (0.6% vs. 2.5%; p = 0.005) during follow-up. Cox regression analysis revealed that the incidence of NACEs (hazard ratio (HR): 0.543, 95% confidence interval (CI): 0.363–0.812; p = 0.003), MACCEs (HR: 0.570, 95% CI: 0.364–0.893; p = 0.014), cardiac death (HR: 0.249, 95% CI: 0.072–0.866; p = 0.029), and stroke (HR: 0.174, 95% CI: 0.060–0.501; p = 0.001) in the guided DAPT group was 0.543, 0.570, 0.249, and 0.174 times, respectively, that in the standard DAPT group.
For patients with ACS who are at high risk in the East Asian population, the primary recommendation is to use PFT to guide DAPT within 12 months after PCI, which can reduce the incidence of NACEs, primarily by lowering the rate of MACCEs.
Keywords
- dual antiplatelet therapy
- platelet function testing
- dual high-risk
- net clinical adverse events
- acute coronary syndrome
According to current guidelines, acute coronary syndrome (ACS) patients undergoing percutaneous coronary intervention (PCI) should receive dual antiplatelet therapy (DAPT) for a minimum of 12 months, with a preference for potent P2Y12 receptor inhibitors [1, 2, 3]. These agents are key for thrombosis prevention and reducing cardiovascular risk. However, the protection that DAPT provides against thrombotic events comes at the cost of an elevated bleeding risk. Therefore, risk stratification and personalized DAPT strategies for patients undergoing PCI will help maximize net clinical benefit [4]. With the continuous development of precision medicine, related research has increasingly focused on methods for assessing ischemic and bleeding risks in ACS patients [4, 5, 6, 7] to help the identify ACS patients with high-risk features as well as to formulate precise DAPT strategies. In order to stratify the high risk, multiple guidelines and study have defined high-risk populations for ischemia or bleeding: the definition of high-risk in the 2023 European Society of Cardiology (ESC) guidelines, the Academic Research Consortium for High Bleeding Risk (ARC-HBR) criteria, and the derivation and validation of the predicting bleeding complications in patients undergoing stent implantation and subsequent dual antiplatelet therapy (PRECISE-DAPT) score, etc. [5]. Potent P2Y12 receptor inhibitors or extended duration of DAPT may be a therapeutic consideration for patients with high risk ischemic features, whereas clopidogrel or shortened DAPT duration may be appropriate for patients with high bleeding risk. However, for patients with dual high-risks, determining the optimal DAPT strategy based on risk assessment alone is not straightforward because there is a greater need to balance ischemic and bleeding risk in these patients. Currently, the optimal DAPT strategy within 12 months after PCI for dual high-risk ACS patients has not been firmly established.
Despite advancements in defining antiplatelet strategies for the chronic phase beyond one year post-PCI, a critical evidence gap remains regarding the optimal antiplatelet regimen during the first year following PCI, specifically in patients at dual high-risk. While clinical trials, such as the OPT-BIRISK (optimal antiplatelet therapy for high bleeding and ischemic risk) trial has focused on determining the strategy after the standard DAPT period [5], there is a distinct lack of randomized or large-scale data guiding the initial strategy within this first year. This gap is particularly relevant for dual high-risk patients, in whom the balance between preventing thrombotic events and mitigating bleeding complications is most delicate.
Platelet function testing (PFT) can individualize the responses to platelet activation and aggregation capacity, ensuring that patients with ACS receive adequate platelet inhibition during long-term treatment. Recognizing that PFT serves as a predictor for ischemic and bleeding events in post-PCI patients, researchers have used PFT to adjust antiplatelet therapy. The 2020 ESC guidelines for non-ST-segment elevation ACS (NSTE-ACS) state that de-escalation is presented as a viable alternative in patients with ACS who are unsuitable for intensive therapy. This de-escalation may be based on clinical judgment or guided by PFT or genetic testing, depending on the risk profiles of patients and the feasibility of testing, although it is only a class IIb recommendation [3]. For patients requiring DAPT escalation, platelet function may be oversuppressed, thereby requiring adjustment under guidance. The platelet function monitoring to adjust antiplatelet therapy in elderly patients stented for an acute coronary syndrome (ANTARCTIC) trial, which adopted a mixed strategy of adjusting therapy (either escalation or de-escalation) based on PFT results in 877 ACS patients aged 75 and above after PCI. However, this trial failed to demonstrate that the PFT-guided strategy significantly reduced the net adverse clinical events [8]. This negative outcome suggests that the utility of a PFT-guided strategy remains uncertain. Consequently, there is currently no clear consensus regarding the precise implementation of PFT in clinical care. Key questions include whether it holds greater value in guiding de-escalation rather than escalation, or in selected high-risk subgroups. A meta-analysis showed that PFT-guided medication adjustment, compared to empirical medication adjustment, could reduce the risks of cardiac death, recurrent myocardial infarction, in-stent thrombosis, stroke, and minor bleeding; however, no differences in all-cause mortality and major bleeding were found [9]. The current studies have not identified the optimal population for PFT. Therefore, the reason for this study which was the investigation and outcomes of PFT in dual high risk patients because even in other populations with different risk profiles prove its efficacy and safety.
This single-centre retrospective cohort study included patients with dual high-risk ACS who underwent PCI at the Cardiology Department of Langfang People’s Hospital from March 2017 to November 2022. All patients with ACS included in this study met the relevant diagnostic criteria outlined in the ESC guidelines [1, 3]. Clinical Presentation: acute chest discomfort (or chest pain-equivalent signs/symptoms)-pain, pressure, tightness, heaviness, or burning. Electrocardiogram (ECG): STEMI (persistent ST-segment elevation (or ST-segment elevation equivalents) on ECG); NSTE-ACS (transient ST-segment elevation, persistent or transient ST-segment depression, and T wave abnormalities, including hyperacute T waves, T wave inversion, biphasic T waves, flat T waves, and pseudo-normalization of T waves. Alternatively, the ECG may be normal). Biomarkers: STEMI/non-ST-elevation myocardial infarction (NSTEMI) -a significant rise and/or fall of cTn, with at least one value above the 99th percentile upper reference limit. The patients met the OPT-BIRISK trial-defined criteria for the dual risk of ischemia and bleeding (Table 1) [7]. The study population was categorized into two groups according to whether they received PFT-guided DAPT: the guided DAPT group and standard DAPT group.
| Dual high risk | High ischaemia risk | High bleeding risk |
| (at least one of the criteria must be met) | (at least one of the criteria must be met) | (at least one of the criteria must be met) |
| (1) age |
(1) multivessel coronary disease; | (1) female sex; |
| (2) age of 65–74 years with high ischemia or high bleeding risks; | (2) total stent length |
(2) iron deficiency anaemia; |
| (3) age |
(3) presence of thrombotic lesions; | (3) history of haemorrhagic or ischemic stroke; |
| (4) bifurcation lesions requiring double stent treatment (Medina classification 0, 1, 1 or 1, 1, 1); | (4) diabetes requiring medication (oral hypoglycaemic therapy or subcutaneous insulin injection); | |
| (5) lesions in the left anterior descending artery ( |
(5) chronic kidney disease, defined as estimated glomerular filtration rate | |
| (6) calcified lesions requiring rotational atherectomy; | ||
| (7) acute coronary syndrome with positive troponin levels; | ||
| (8) diagnosis of vascular disease, including previous myocardial infarction, ischemic stroke, peripheral artery disease (PAD), or coronary atherosclerotic heart disease (CAD)/PAD vascular reconstruction; | ||
| (9) recurrent myocardial infarction, coronary revascularization, stent thrombosis, or stroke occurring within 9 months prior to percutaneous coronary intervention; | ||
| (10) diabetes requiring medication (oral hypoglycaemic therapy or subcutaneous insulin injection); | ||
| (11) chronic kidney disease, defined as estimated glomerular filtration rate |
The inclusion criteria were as follows: (1) age
The case report form (CRF) includes patient baseline and prognostic data. These data included general information, diagnosis, and treatment of patients during hospitalization, including age, sex, body mass index (BMI), primary PCI, history of chronic diseases, laboratory examination, culprit artery, and PCI details. Baseline patient data were collected from electronic medical records, while the grouping and prognostic data for CRF were acquired via telephone follow-ups and outpatient visits. For patients in the guided DAPT group, PFT and medication adjustments at 3 months after PCI were completed during outpatient visits. After completing the CRF for each patient, the relevant data were entered into SPSS software version 26.0 (IBM Corp., Armonk, NY, USA) by two individuals to ensure accuracy of the data. The follow-up period was 12 months after PCI.
In the guided DAPT group, fasting venous blood was obtained from the elbow at 3
months after PCI. 3 mL of venous blood was collected into a coagulation vacuum
tube, thoroughly mixed for anticoagulation, and tested within 3 hours at room
temperature. Blood was placed in a tube containing sodium citrate anticoagulant
(20231208A, Shenzhen Kangnaige Biological Technology Co., Ltd., Shenzhen, Guangdong, China)
and centrifuged at 150
At 3 months after PCI, the DAPT regimen for patients in the guided DAPT group was adjusted based on PFT results as follows (decision tree: Supplementary Material 2):
(1) For patients whose DAPT regimen within 3 months after PCI was aspirin (100
mg quaque die (QD)) + clopidogrel (75 mg QD): if MAR(ADP) was
(2) For patients whose DAPT regimen within 3 months after PCI was aspirin (100
mg QD) + ticagrelor (60 mg BID): if MAR(ADP) was
(3) For patients whose DAPT regimen within 3 months after PCI was aspirin (100
mg QD) + ticagrelor (90 mg BID): if MAR(ADP) was
For patients in the standard DAPT group, the DAPT regimen within 3 months after PCI was aspirin (100 mg QD), clopidogrel (75 mg QD), ticagrelor (60 mg BID), and ticagrelor (90 mg BID). At 3 months, adjustments to the DAPT regimen were made based on the clinical judgment of the physician.
A minimum 12-month duration of DAPT was implemented in all patients.
In this study, the primary endpoint was the net clinical adverse events (NACE) during the follow-up period, which included a composite endpoint of major adverse cardiovascular and cerebrovascular events (MACCE) and bleeding academic research consortium (BARC) bleeding (type 3 or greater). The secondary endpoint was MACCE, which included a composite endpoint of cardiac death, myocardial infarction, ischemia-driven revascularization, and stroke. The safety endpoint was BARC bleeding, including BARC bleeding (type 3 or greater) and BARC bleeding (type 1 or 2).
The department trained data clerks to follow up on all patients. Visits were scheduled at 2 weeks, 3 months, 6 months, and 1 year after PCI. For patients who did not attend outpatient follow-ups, telephone follow-ups were conducted to track the endpoints. For patients in the guided DAPT group, if adjustments to the DAPT regimen were made at 3 months after PCI, outpatient follow-ups were still required when PFT was performed. The endpoint deadline was within 12 months after PCI.
Statistical analysis was performed using IBM SPSS Statistics (version 26.0). Normally distributed
continuous data were expressed as mean
This study screened participants among 3307 ACS patients who underwent PCI.
After applying the inclusion and exclusion criteria, 1822 dual high-risk patients
were enrolled. During the 12-month follow-up period, two patients in the guided
DAPT group were lost to follow-up, resulting in the final inclusion of 712
patients. In the standard DAPT group, four patients were lost to follow-up,
leading to the final inclusion of 1104 patients. The mean follow-up duration in
this study was 40.70
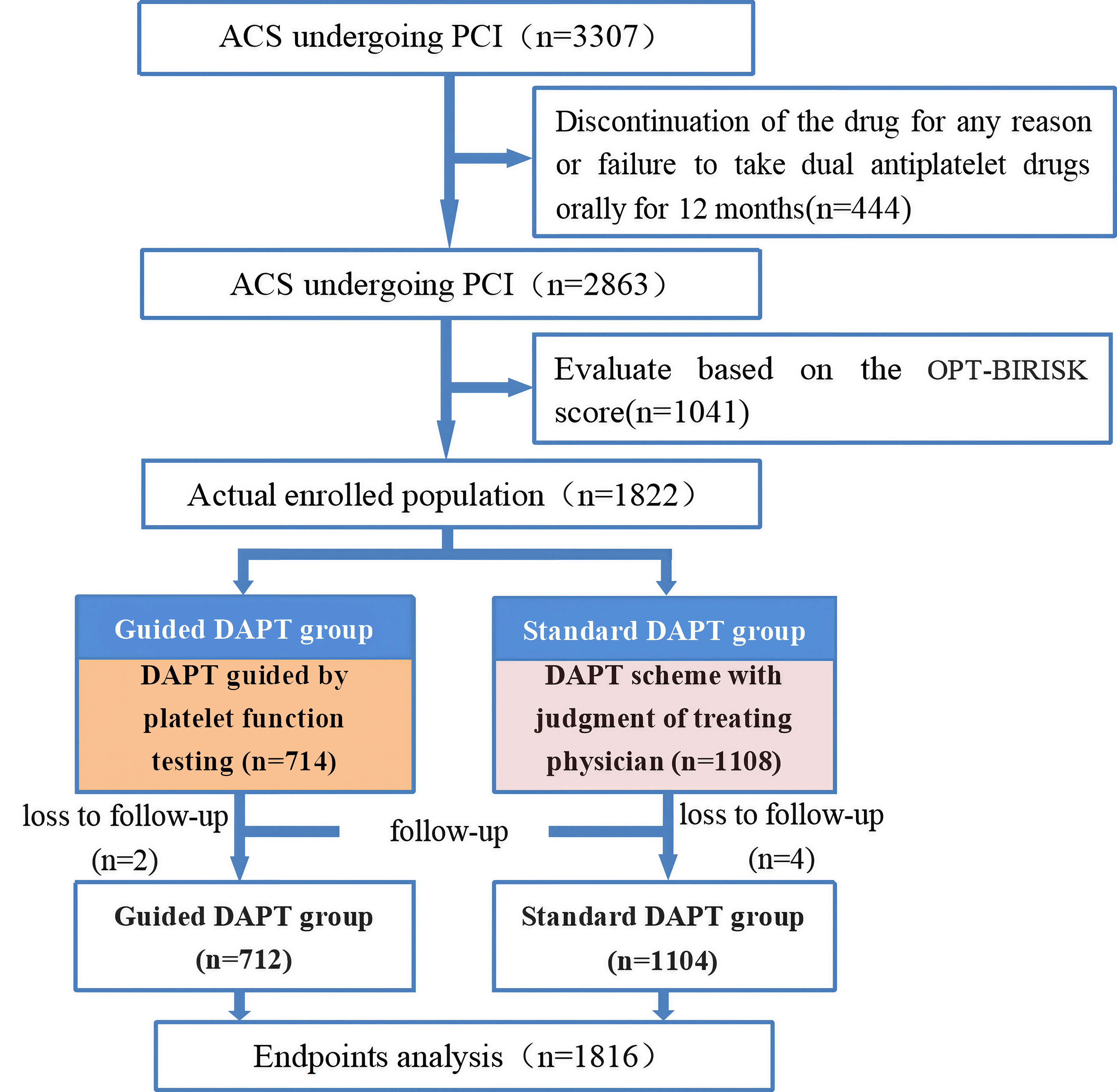 Fig. 1.
Fig. 1.
Flowchart. OPT-BIRISK, optimal antiplatelett herapy for high bleeding and ischemic RISK patients; ACS, acute coronary syndrome; PCI, percutaneous coronary intervention; DAPT, dual antiplatelet therapy.
No statistically significant differences between the two groups were detected in
terms of age, female sex, BMI, presentation, hypertension, type 2 diabetes,
cerebrovascular disease, previous myocardial infarction, atrial fibrillation,
smoking, family history of coronary atherosclerotic heart disease (CAD),
OPT-BIRISK, optimal antiplatelet antiplatelet therapy for Chinese patients with
coronary artery disease (OPT-CAD), ARC-HBR and ticagrelor at discharge
(all p
| Characteristic | Guided DAPT group (n = 712) | Standard DAPT group (n = 1104) | t/ |
p value | |
| Age (years; m |
63.51 |
63.80 |
0.638 | 0.524 | |
| Female (n, %) | 331 (46.5%) | 468 (42.4%) | 2.949 | 0.086 | |
| BMI (kg/m2; m |
25.76 |
25.86 |
0.723 | 0.470 | |
| Presentation (n, %) | |||||
| UAP | 411 (57.7%) | 512 (46.4%) | 29.139 | ||
| NSTEMI | 50 (7.0%) | 61 (5.5%) | |||
| STEMI | 251 (35.3%) | 531 (48.1%) | |||
| Primary PCI (n, %) | 180 (25.3%) | 457 (41.4%) | 49.354 | ||
| Medical history (n, %) | |||||
| Hypertension | 527 (74.0%) | 782 (70.8%) | 2.180 | 0.140 | |
| Type 2 diabetes | 250 (35.1%) | 362 (32.8%) | 1.045 | 0.307 | |
| Cerebrovascular disease | 125 (17.6%) | 187 (16.9%) | 0.116 | 0.733 | |
| OMI | 56 (7.9%) | 70 (6.3%) | 1.558 | 0.212 | |
| Atrial fibrillation | 16 (2.2%) | 33 (3.0%) | 0.908 | 0.341 | |
| Current smoker | 298 (41.9%) | 451 (40.9%) | 0.180 | 0.672 | |
| CAD family history | 30 (4.2%) | 48 (4.3%) | 0.019 | 0.890 | |
| High ischaemia risk of OPT-BIRISK (n, %) | 698 (98.0%) | 1093 (99.0%) | 2.999 | 0.083 | |
| OPT-CAD (n, %) | |||||
| Low risk | 227 (31.9%) | 389 (35.2%) | 2.805 | 0.246 | |
| Medium risk | 430 (60.4%) | 623 (56.4%) | |||
| High risk | 55 (7.7%) | 92 (8.3%) | |||
| ARC-HBR (n, %) | 186 (26.1%) | 278 (25.2%) | 0.202 | 0.653 | |
| Ticagrelor at discharge (n, %) | 327 (45.9%) | 541 (49.0%) | 1.642 | 0.200 | |
| Switch of P2Y12 inhibitors | 218 (30.6%) | - | - | - | |
| Types of P2Y12 inhibitors 3–12 months after PCI (n, %) | |||||
| Clopidogrel 75 mg | 446 (62.6%) | 692 (62.7%) | 17.158 | ||
| Ticagrelor 90 mg | 224 (31.5%) | 387 (35.1%) | |||
| Ticagrelor 60 mg | 42 (5.9%) | 25 (2.3%) | |||
M, mean; SD, standard deviation; BMI, body mass index; UAP, unstable angina
pectoris (a. p
There were no statistically significant differences in baseline laboratory
parameters, including white blood cell count, hemoglobin, platelet count,
fibrinogen, eGFR, uric acid, fasting blood glucose, and triglycerides, between
the two groups (all p
| Characteristic | Guided DAPT group (n = 712) | Standard DAPT group (n = 1104) | t value | p value |
| WBC (×109/L) | 7.40 |
7.54 |
1.230 | 0.219 |
| HGB (g/L) | 134.73 |
134.04 |
0.936 | 0.350 |
| PLT (×109/L) | 234.30 |
233.29 |
0.375 | 0.707 |
| FIB (g/L) | 3.44 |
3.44 |
0.081 | 0.935 |
| eGFR | 92.72 |
92.14 |
0.739 | 0.460 |
| UA (µmol/L) | 320.75 |
317.46 |
0.780 | 0.435 |
| FBG (mmol/L) | 6.95 |
7.12 |
1.467 | 0.142 |
| TC (mmol/L) | 4.92 |
4.67 |
3.089 | |
| TG (mmol/L) | 1.95 |
1.88 |
1.345 | 0.179 |
| LDL-C (mmol/L) | 2.93 |
2.80 |
3.065 | 0.002 |
| HDL-C (mmol/L) | 1.13 |
1.09 |
2.882 | 0.004 |
WBC, white blood cell; HGB, hemoglobin; PLT platelet; FIB, fibrinogen; eGFR, estimated glomerular filtration rate; UA, uric acid; FBG, fasting blood glucose; TC, total cholesterol; TG, triglyceride; LDL-C, low density lipoprotein-cholesterol; HDL-C, high density lipoprotein-cholesterol.
No statistically significant differences between the two groups were detected in
terms of the culprit artery, chronic total occlusion, in-stent restenosis,
proximal segment of the left anterior descending artery, and number of vessels
with lesions (all p
| Characteristic | Guided DAPT group (n = 712) | Standard DAPT group (n = 1104) | t/ |
p value | |
| Culprit artery (n, %) | |||||
| LM | 15 (2.1%) | 33 (3.0%) | 1.310 | 0.252 | |
| LAD | 637 (89.5%) | 994 (90.0%) | 0.154 | 0.695 | |
| LCX | 506 (71.1%) | 780 (70.7%) | 0.036 | 0.849 | |
| RCA | 555 (77.9%) | 856 (77.5%) | 0.043 | 0.836 | |
| Ostial lesion (n, %) | 231 (32.4%) | 295 (26.7%) | 6.890 | 0.009 | |
| Diffused lesion (n, %) | 378 (53.1%) | 512 (46.4%) | 7.806 | 0.005 | |
| CTO (n, %) | 7 (1.0%) | 24 (2.2%) | 3.658 | 0.056 | |
| ISR (n, %) | 19 (2.7%) | 18 (1.6%) | 2.337 | 0.126 | |
| Small vessel (n, %) | 52 (7.3%) | 178 (16.1%) | 30.441 | ||
| LADp (n, %) | 133 (18.7%) | 190 (17.2%) | 0.639 | 0.424 | |
| Complete RV (n, %) | 168 (23.6%) | 398 (36.1%) | 31.30 | ||
| Number of lesion vessel (n; m |
2.42 |
2.43 |
0.434 | 0.664 | |
| Number of stents (n; m |
1.46 |
1.37 |
3.053 | 0.002 | |
LM, left main; LAD, left anterior descending; LCX, left circumflex coronary artery; RCA, right coronary artery; CTO, chronic total occlusion; ISR, In-stent restenosis; LADp, proximal segment of left anterior descending; RV, revascularization.
The results showed that, compared to the standard DAPT group, the guided DAPT
group had significantly lower incidence of NACE, BARC bleeding (type 3 or
greater), cardiac death, and stroke within 0–12 months after PCI (p =
0.018, p = 0.029, p = 0.038, and p = 0.007,
respectively). During the follow-up period, there were no statistically
significant differences in the incidence of MACCE, myocardial infarction, target
vessel revascularization, BARC bleeding, or BARC bleeding (type 1 or 2) between
the two groups (all p
| Characteristic | Guided DAPT group (n = 712) | Standard DAPT group (n = 1104) | HR | 95% CI | p | |
| Endpoints within 0–12 months | ||||||
| NACE (n, %) | 43 (6.0%) | 102 (9.3%) | 0.651 | 0.456–0.930 | 0.018 | |
| MACCE (n, %) | 38 (5.3%) | 79 (7.2%) | 0.766 | 0.520–1.129 | 0.178 | |
| Cardiac death (n, %) | 3 (0.4%) | 17 (1.5%) | 0.273 | 0.080–0.932 | 0.038 | |
| MI (n, %) | 16 (2.3%) | 24 (2.2%) | 1.033 | 0.549–1.944 | 0.921 | |
| TVR (n, %) | 23 (3.3%) | 25 (2.3%) | 1.427 | 0.810–2.515 | 0.218 | |
| Stroke (n, %) | 5 (0.7%) | 29 (2.7%) | 0.269 | 0.104–0.694 | 0.007 | |
| BARC bleeding events (n, %) | 131 (18.4%) | 183 (16.6%) | 1.116 | 0.892–1.397 | 0.337 | |
| Type 3 or greater | 6 (0.8%) | 25 (2.3%) | 0.370 | 0.152–0.902 | 0.029 | |
| Type 1 or 2 | 126 (17.7%) | 167 (15.1%) | 1.179 | 0.936–1.486 | 0.163 | |
| Endpoints within 3–12 months | ||||||
| NACE (n, %) | 34 (4.8%) | 96 (8.7%) | 0.525 | 0.353–0.779 | 0.001 | |
| MACCE (n, %) | 28 (3.9%) | 74 (6.7%) | 0.588 | 0.380–0.908 | 0.017 | |
| Cardiac death (n, %) | 3 (0.4%) | 17 (1.5%) | 0.273 | 0.080–0.932 | 0.038 | |
| MI (n, %) | 11 (1.5%) | 22 (2.0%) | 0.798 | 0.387–1.648 | 0.543 | |
| TVR (n, %) | 19 (2.7%) | 22 (2.0%) | 1.407 | 0.760–2.605 | 0.277 | |
| Stroke (n, %) | 4 (0.6%) | 28 (2.5%) | 0.224 | 0.079–0.638 | 0.005 | |
| BARC bleeding events (n, %) | 83 (11.7%) | 105 (9.5%) | 1.239 | 0.929–1.652 | 0.145 | |
| Type 3 or greater | 6 (0.8%) | 22 (1.9%) | 0.418 | 0.170–1.032 | 0.059 | |
| Type 1 or 2 | 77 (10.8%) | 90 (8.2%) | 1.347 | 0.994–1.826 | 0.055 | |
NACE, net adverse clinical event; MACCE, major adverse cardiovascular and cerebrovascular event; MI, myocardial infarction; TVR, target vessel revascularization; BARC, bleeding academic research consortium.
The results showed that, compared to the standard DAPT group, the guided DAPT group had significantly lower incidence of NACE, MACCE, cardiac death, and stroke within 3–12 months after PCI (p = 0.001, p = 0.017, p = 0.038 and p = 0.005, respectively). There were no statistically significant differences in the incidence of myocardial infarction, target vessel revascularization, BARC bleeding, BARC bleeding (type 3 or greater), or BARC bleeding (type 1 or 2) within 3–12 months after PCI between the two groups (p = 0.077) (Table 5).
Kaplan–Meier survival curves showed that within 3 months after PCI, there were
no significant differences in the incidence of NACE, MACCE, and BARC bleeding
(type 3 or greater) between the two groups (all p
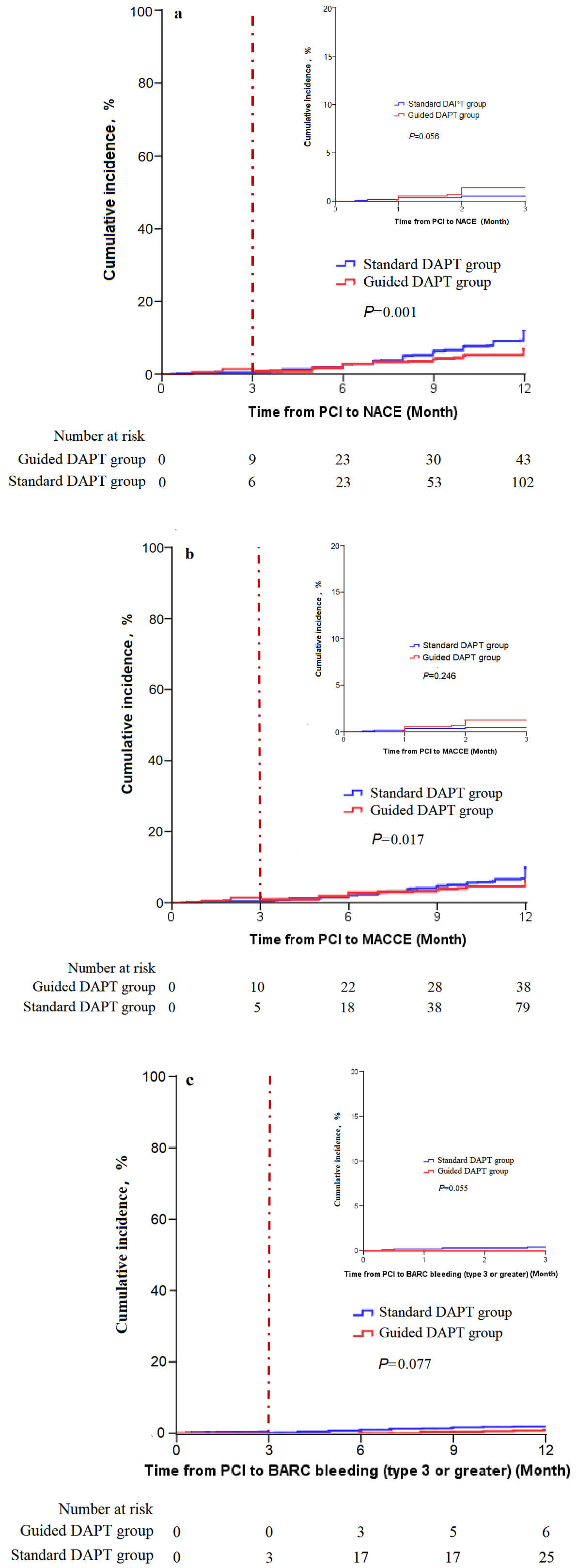 Fig. 2.
Fig. 2.
Cumulative incidence. (a) NACEs cumulative incidence; NACEs, net clinical adverse events, including the composite endpoint of major adverse cardiovascular and cerebrovascular events (MACCEs) and BARC bleeding (type 3 or greater). Cutoff point: 3rd month after PCI. (b) MACCEs cumulative incidence; MACCEs, major adverse cardiovascular and cerebrovascular, including the composite endpoint of cardiac death, myocardial infarction, ischemia-driven revascularization, and stroke. Cutoff point: 3rd month after PCI. (c) BARC bleeding (type 3 or greater) cumulative incidence. Cutoff point: 3rd month after PCI.
Regarding NACE and MACCE within 3–12 months after PCI, variables with statistically significant differences in baseline characteristics (presentation, primary PCI, total cholesterol, low-density lipoprotein, high-density lipoprotein, ostial lesions, diffuse lesions, small vessel disease, complete revascularization, and number of stents) as well as variables that may significantly impact the outcome measures (age, male, BMI, hypertension, type 2 diabetes, cerebrovascular disease, history of myocardial infarction, smoking history, and proximal left anterior descending artery lesions) were included as independent variables in the Cox regression analysis. The results showed that the risks of NACE and MACCE in the guided DAPT group were 0.543 times (hazard ratio [HR]: 0.543, 95% confidence interval [CI]: 0.363–0.812, p = 0.003) and 0.570 times (HR: 0.570, 95% CI: 0.364–0.893, p = 0.014), respectively, compared to the standard DAPT group. The risk of NACE and MACCE in patients with a history of cerebrovascular disease was 1.544 times (HR: 1.544, 95% CI: 1.025–2.327, p = 0.038) and 1.821 times (HR: 1.821, 95% CI: 1.155–2.870, p = 0.010) higher than that in patients without such a history. For each additional stent implanted, the risk of MACCE increased to 1.345 times the original risk (HR: 1.345, 95% CI: 1.013–1.786, p = 0.040) (Table 6).
| Endpoints | Variables | Quotient | Wald | HR | 95% CI | p | |
| B | SE | ||||||
| NACE | Guided DAPT group | –0.611 | 0.206 | 8.835 | 0.543 | 0.363–0.812 | 0.003 |
| Male | 0.357 | 0.198 | 3.273 | 1.430 | 0.971–2.105 | 0.070 | |
| Age | 0.017 | 0.010 | 2.778 | 1.018 | 0.997–1.039 | 0.096 | |
| BMI | 0.035 | 0.033 | 1.154 | 1.036 | 0.972–1.104 | 0.283 | |
| UAP | 0.138 | 0.236 | 0.339 | 1.148 | 0.722–1.825 | 0.560 | |
| Primary PCI | 0.217 | 0.260 | 0.699 | 1.243 | 0.746–2.070 | 0.403 | |
| Hypertension | 0.097 | 0.209 | 0.214 | 1.101 | 0.731–1.658 | 0.644 | |
| Type 2 diabetes | –0.201 | 0.201 | 1.001 | 0.818 | 0.551–1.213 | 0.317 | |
| Cerebrovascular disease | 0.435 | 0.209 | 4.323 | 1.544 | 1.025–2.327 | 0.038 | |
| OMI | –0.356 | 0.422 | 0.713 | 0.700 | 0.306–1.602 | 0.399 | |
| Current smoker | 0.224 | 0.185 | 1.466 | 1.251 | 0.871–1.799 | 0.226 | |
| TC | –0.102 | 0.138 | 0.554 | 0.903 | 0.689–1.182 | 0.457 | |
| LDL-C | 0.208 | 0.181 | 1.329 | 1.232 | 0.864–1.756 | 0.249 | |
| HDL-C | 0.039 | 0.364 | 0.011 | 1.040 | 0.509–2.124 | 0.915 | |
| Ostial lesion | –0.350 | 0.212 | 2.709 | 0.705 | 0.465–1.069 | 0.100 | |
| Diffused lesion | –0.142 | 0.180 | 0.621 | 0.868 | 0.610–1.235 | 0.431 | |
| Small vessel | 0.075 | 0.263 | 0.082 | 1.078 | 0.644–1.805 | 0.775 | |
| Complete RV | –0.209 | 0.216 | 0.940 | 0.811 | 0.531–1.238 | 0.332 | |
| LADp | 0.178 | 0.219 | 0.662 | 1.195 | 0.778–1.833 | 0.416 | |
| Number of stents | 0.204 | 0.132 | 2.376 | 1.226 | 0.946–1.589 | 0.123 | |
| MACCE | Guided DAPT group | –0.563 | 0.229 | 6.034 | 0.570 | 0.364–0.893 | 0.014 |
| Male | 0.367 | 0.226 | 2.627 | 1.443 | 0.926–2.250 | 0.105 | |
| Age | 0.012 | 0.012 | 0.992 | 1.012 | 0.989–1.036 | 0.319 | |
| BMI | 0.039 | 0.036 | 1.125 | 1.039 | 0.968–1.116 | 0.289 | |
| UAP | –0.123 | 0.264 | 0.218 | 0.884 | 0.527–1.484 | 0.641 | |
| Primary PCI | 0.016 | 0.297 | 0.003 | 1.016 | 0.567–1.820 | 0.958 | |
| Hypertension | –0.049 | 0.232 | 0.045 | 0.952 | 0.604–1.499 | 0.831 | |
| Type 2 diabetes | –0.295 | 0.233 | 1.610 | 0.744 | 0.472–1.175 | 0.205 | |
| Cerebrovascular disease | 0.599 | 0.232 | 6.669 | 1.821 | 1.155–2.870 | 0.010 | |
| OMI | –0.090 | 0.427 | 0.044 | 0.914 | 0.396–2.111 | 0.834 | |
| Current smoker | 0.221 | 0.211 | 1.090 | 1.247 | 0.824–1.886 | 0.296 | |
| TC | –0.146 | 0.154 | 0.906 | 0.864 | 0.639–1.167 | 0.341 | |
| LDL-C | 0.247 | 0.201 | 1.513 | 1.280 | 0.864–1.898 | 0.219 | |
| HDL-C | 0.297 | 0.389 | 0.581 | 1.346 | 0.627–2.887 | 0.446 | |
| Ostial lesion | –0.389 | 0.243 | 2.557 | 0.678 | 0.421–1.092 | 0.110 | |
| Diffused lesion | –0.159 | 0.205 | 0.603 | 0.853 | 0.570–1.275 | 0.438 | |
| Small vessel | 0.005 | 0.313 | 0.000 | 1.005 | 0.545–1.854 | 0.987 | |
| Complete RV | –0.133 | 0.246 | 0.293 | 0.875 | 0.540–1.418 | 0.588 | |
| LADp | 0.204 | 0.244 | 0.697 | 1.226 | 0.760–1.978 | 0.404 | |
| Number of stents | 0.296 | 0.145 | 4.202 | 1.345 | 1.013–1.786 | 0.040 | |
| Cardiac death | Guided DAPT group | –1.389 | 0.635 | 4.784 | 0.249 | 0.072–0.866 | 0.029 |
| Male | 0.581 | 0.492 | 1.397 | 1.788 | 0.682–4.687 | 0.237 | |
| Age | 0.052 | 0.029 | 3.136 | 1.053 | 0.994–1.116 | 0.077 | |
| Primary PCI | –0.594 | 0.569 | 1.091 | 0.552 | 0.181–1.684 | 0.296 | |
| Type 2 diabetes | –0.511 | 0.569 | 0.805 | 0.600 | 0.197–1.830 | 0.370 | |
| Cerebrovascular disease | 0.704 | 0.494 | 2.031 | 2.021 | 0.768–5.321 | 0.154 | |
| Complete RV | 0.218 | 0.539 | 0.163 | 1.243 | 0.432–3.575 | 0.686 | |
| Number of stents | 0.547 | 0.275 | 3.963 | 1.728 | 1.009–2.960 | 0.047 | |
| Stroke | Guided DAPT group | –1.751 | 0.541 | 10.483 | 0.174 | 0.060–0.501 | 0.001 |
| Male | 0.377 | 0.388 | 0.948 | 1.459 | 0.682–3.118 | 0.330 | |
| Age | –0.024 | 0.021 | 1.350 | 0.976 | 0.938–1.017 | 0.245 | |
| Primary PCI | –0.325 | 0.443 | 0.536 | 0.723 | 0.303–1.724 | 0.464 | |
| Type 2 diabetes | –0.760 | 0.439 | 2.996 | 0.468 | 0.198–1.106 | 0.083 | |
| Cerebrovascular disease | 1.498 | 0.364 | 16.982 | 4.473 | 2.194–9.120 | ||
| Complete RV | –0.900 | 0.527 | 2.918 | 0.407 | 0.145–1.142 | 0.088 | |
| Number of stents | 0.521 | 0.231 | 5.079 | 1.683 | 1.070–2.647 | 0.024 | |
| BARC bleeding (type 3 or greater) | Guided DAPT group | –0.620 | 0.476 | 1.695 | 0.538 | 0.212–1.368 | 0.193 |
| Male | 0.213 | 0.407 | 0.273 | 1.237 | 0.557–2.748 | 0.602 | |
| Age | 0.047 | 0.023 | 4.012 | 1.048 | 1.001–1.097 | 0.045 | |
| Primary PCI | 0.973 | 0.451 | 4.642 | 2.645 | 1.092–6.406 | 0.031 | |
| Type 2 diabetes | 0.418 | 0.413 | 1.022 | 1.519 | 0.675–3.415 | 0.312 | |
| Cerebrovascular disease | –0.194 | 0.546 | 0.126 | 0.824 | 0.283–2.403 | 0.723 | |
| Complete RV | –0.480 | 0.461 | 1.083 | 0.619 | 0.250–1.529 | 0.298 | |
| Number of stents | –0.065 | 0.347 | 0.035 | 0.938 | 0.475–1.851 | 0.853 | |
RV, revascularization; LADp, proximal segment of left anterior descending.
Regarding cardiac death and stroke within 3–12 months after PCI, variables that
may significantly impact the outcome measures (age, male, primary PCI, type 2
diabetes, cerebrovascular disease, complete revascularization, and number of
stents) were included as independent variables in the Cox regression analysis.
The results showed that the risks of cardiac death and stroke in the guided DAPT
group were 0.249 times (HR: 0.249, 95% CI: 0.072–0.866, p = 0.029) and
0.174 times (HR: 0.174, 95% CI: 0.060–0.501, p = 0.001), respectively,
compared to the standard DAPT group. For each additional stent implanted, the
risk of cardiac death increased to 1.728 times the original risk (HR: 1.728, 95%
CI: 1.009–2.960, p = 0.047). The risk of stroke in patients with a
history of cerebrovascular disease was 4.473 times higher than that in patients
without such a history (HR: 4.473, 95% CI: 2.194–9.120, p
Regarding BARC bleeding (type 3 or greater) within 3–12 months after PCI, variables that may significantly impact the outcome measures (age, male, primary PCI, type 2 diabetes, cerebrovascular disease, complete revascularization, and number of stents) were included as independent variables in the Cox regression analysis. For each additional age, the risk of BARC bleeding (type 3 or greater) increased to 1.048 times the original risk (HR: 1.048, 95% CI: 1.001–1.097, p = 0.045). The risk of BARC bleeding (type 3 or greater) in patients undergoing primary PCI was 2.645 times higher than that in patients not undergoing primary PCI (HR: 2.645, 95% CI: 1.092–6.406, p = 0.031) (Table 6).
To verify the robustness of the results, this study conducted a subgroup
analysis based on gender, age, presentation, primary PCI, cerebrovascular
disease, current smoker, OPT-CAD, ARC-HBR, ostial lesion, diffused lesion, small
vessel, complete revascularization, number of stents, and post-October 2020 era.
In the subgroups of
This study included a certain proportion of patients lost to follow-up, with two cases in the guided DAPT group and four cases in the standard DAPT group. A sensitivity analysis was conducted to assess the potential impact of loss to follow-up on the results. The NACE analysis was repeated on two hypothetical datasets: one assuming that all patients lost to follow-up experienced endpoints and the other assuming that none of the patients lost to follow-up experienced endpoints. The results from both hypothetical datasets revealed that the incidence of NACE in the guided DAPT group was significantly lower than that in the standard DAPT group (p = 0.001 and p = 0.001), indicating that the study results are relatively robust (Fig. 3).
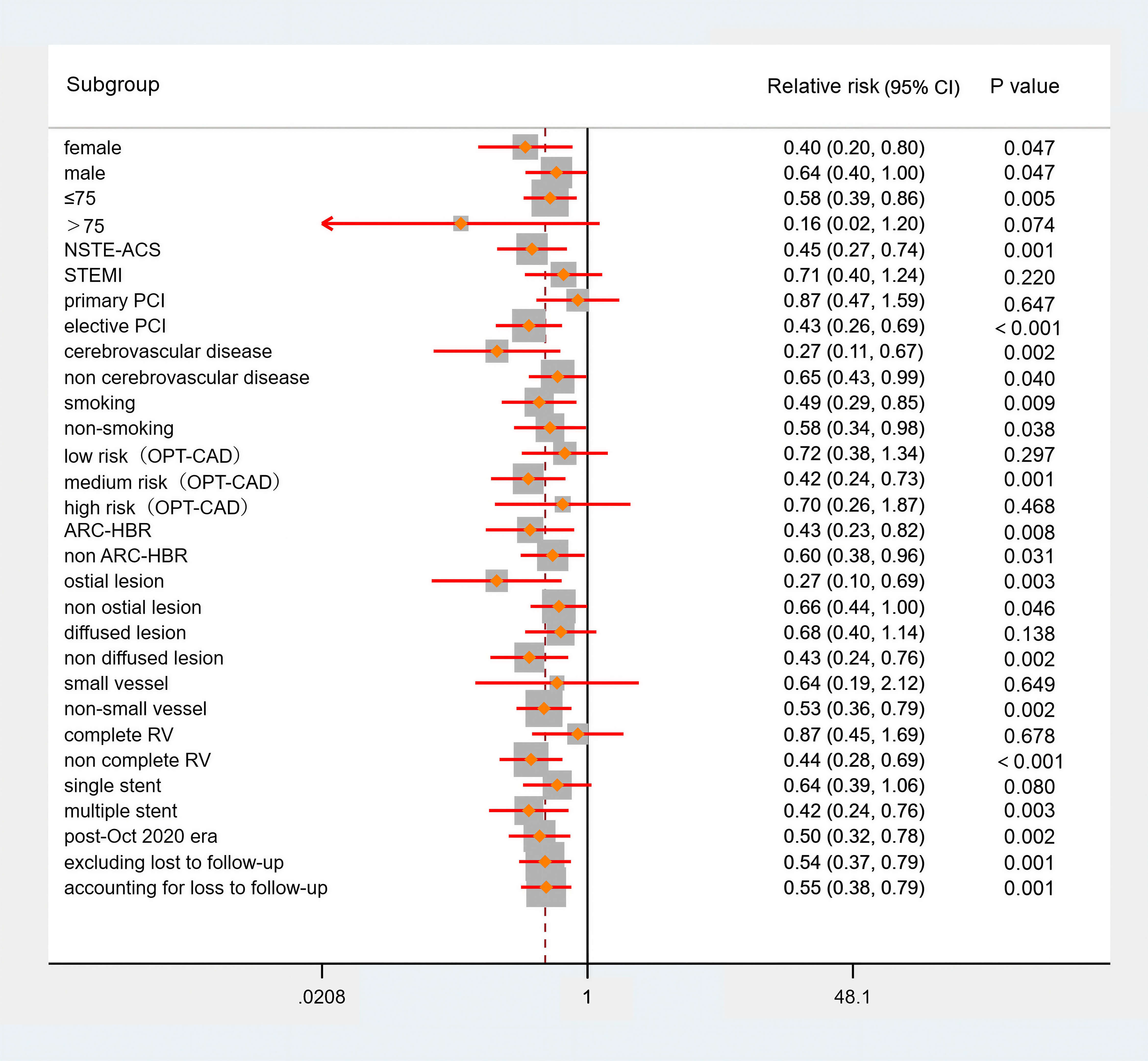 Fig. 3.
Fig. 3.
Sensitivity analysis (NACE). NSTE-ACS, non-ST-segment elevation acute coronary syndrome; OPT-CAD, optimal antiplatelet antiplatelet therapy for Chinese patients with coronary artery disease.
This was a real-world, single-centre cohort study that retrospectively analysed 1816 ACS patients at a dual high risk who underwent PCI. The conclusions are as follows. (1) A significant proportion of ACS patients undergoing PCI exhibit the dual high-risk features, accounting for approximately 55.09% (1822/3307). (2) The PFT-guided DAPT scheme selection is recommended for patients with dual high-risk ACS who have undergone PCI. Compared to DAPT regimens subjectively chosen by clinicians based on experience, this approach can significantly benefit from NACE (a composite endpoint of MACCE and BARC bleeding (type 3 or greater)) and MACCE.
For ACS patients undergoing PCI, current guidelines recommend using a 12-month combination of aspirin and potent P2Y12 receptor inhibitors for DAPT. Although intensified or prolonged DAPT treatment can reduce the risk of ischemia, it can increase bleeding events, which is a predictive factor for poor prognosis. The risk of ischemia in patients with ACS is time-dependent and gradually decreases over time. Therefore, in clinical practice, efforts are being made to balance bleeding and ischemia risks through unguided de-escalation strategies, in order to achieve personalized treatment [10]. For patients at a dual high risk for ischemia and bleeding, there is an even greater need to balance ischemic and bleeding risks. In this study, the prevalence of dual high-risk ACS patients was approximately 60%, notably higher than the 32% reported using the “evaluating the performance of the can rapid risk stratification of unstable angina patients suppress adverse outcomes with early implementation of the ACC/AHA guidelines (CRUSADE)”-“global registry of acute coronary events (GRACE)” risk combinations [11]. This result differs significantly from our findings. A likely explanation for this discrepancy is the divergent methodological approaches used to define dual high-risk status. Additionally, while the aforementioned study focused on ACS patients, our study exclusively included ACS patients who underwent PCI. This suggests that the coronary artery disease severity in our cohort may be greater, potentially explaining the higher proportion of dual high-risk patients. Similarly, a study applying the OPT-BIRISK criteria reported a comparable dual high-risk prevalence of 58.8% (4146/7049) in ACS patients who underwent PCI, which is closely aligned with our findings [12]. However, current evidence offers little guidance on DAPT regimens for dual high-risk patients. In 2020, the ESC guidelines proposed a preliminary antiplatelet drug transition model based on ischemic and bleeding risk. Bleeding risk was assessed in NSTE-ACS patients who underwent PCI. For high bleeding risk patients, de-escalation to aspirin monotherapy is recommended at the 3-month post-PCI. For remarkably high bleeding risk patients, de-escalation to clopidogrel monotherapy is suggested at the 1-month post-PCI. For low bleeding risk patients, further assessment of high ischemic risk is required to develop a more precise antiplatelet strategy [3]. However, this model lacks discussion on antiplatelet regimens for dual high-risk patients, highlighting the gap in evidence in the guidelines. For dual high-risk patients, an additional tool beyond the current risk scoring systems is required to assist clinicians in selecting DAPT regimens. However, ischemic and bleeding events in ACS patients undergoing PCI result from the synergistic effects of multiple factors, including age and comorbidities such as diabetes, hypertension, atrial fibrillation, heart failure, complex CAD, procedural factors, and anticoagulants or nonsteroidal anti-inflammatory drugs [3, 5, 13, 14, 15]. In recent years, studies on PFT-guided DAPT for patients with ACS have not resulted in definitive guidelines [3, 8, 9]. PFT can individually reflect platelet activation and aggregation capacity, ensuring adequate platelet inhibition during the long-term treatment of patients with ACS. Previous studies identified high platelet reactivity as an independent risk factor for MACCE within 12 months after PCI. Conversely, it was negatively correlated with bleeding events [16, 17]. In the ANTARCTIC study, adjustments were made for DAPT escalation or de-escalation based on PFT [8, 18]. Compared to medication adjustments based on experience, PFT-guided medication adjustments can reduce the risks of cardiac death, myocardial infarction, in-stent thrombosis, and stroke, as well as minor bleeding risks [9]. However, other studies suggested that PFT-guided DAPT does not reduce the occurrence of ischemic and bleeding events [19, 20]. A potential explanation is that PFT alone has a limited role in guiding DAPT. In contrast, risk-scoring systems for patients with ACS encompass various high-risk factors beyond PFT. Therefore, it is important to determine how PFT can be combined with risk scores to guide the selection of DAPT regimens selection. In other diseases, combining scoring systems with biomarkers to guide diagnosis and treatment is common, such as combining the results of the Wells and Geneva scores with D-dimer levels to assist in the identification of pulmonary embolism [21].
A study has shown that the association between low platelet reactivity and bleeding events is weaker than the association between high platelet reactivity and thrombotic complications [22]. This may be because the optimal cutoff values for platelet reactivity related to high bleeding and ischemic risks are not yet clearly defined [23, 24]. However, because the ischemic risk in ACS patients gradually decreases after PCI while the bleeding risk increases. Consequently, the bleeding risk surpasses the ischemic risk at one month post-ACS onset, after which both risks tend to stabilize [25]. Since bleeding and ischemic risks vary at different stages following PCI, the optimal therapeutic window for platelet reactivity may differ. The most convenient approach is to perform PFT to guide the DAPT scheme selection when a patient’s ischemic and bleeding risks begin to stabilize, which is also the optimal time for medication transition. In this study, PFT was conducted at 3 months after PCI to guide the DAPT strategy selection. Most patients in this study underwent outpatient follow-up at 3 months after PCI; however, evidence confirms that potent P2Y12 inhibitors maintain a critical advantage in preventing early-phase ischemic events within the first month of ACS [26]. This suggests that although the bleeding risk begins to exceed the ischemic risk at 1 month after PCI, the ischemic risk remains relatively high, and transitioning medications at this stage may increase the occurrence of ischemic events, particularly when de-escalating antiplatelet drugs are used. Despite the continuing increase in the risk of in-stent thrombosis within six months post-PCI with newer-generation drug-eluting stents, in-stent thrombosis has been shown not to significantly differ between 3 and 6 months after PCI [27]. Therefore, in our study, we chose to adjust antiplatelet therapy and perform PFT at 3 months after PCI. In addition, because the distribution of cytochrome P450 family 2 subfamily C member 19 (CYP2C19) alleles varies among African, European, and East Asian populations and these alleles can influence platelet reactivity [28]. There is a need to identify the optimal window for platelet reactivity across ethnic groups, especially considering the ‘East Asian paradox’. Owing to the inclusion of East Asian populations in this study, there may be certain limitations in the generalizability of the results.
In real-world practice, the choice of P2Y12 receptor inhibitors for post-PCI patients typically considers three aspects: the assessment results of the ischemia and bleeding risks, the complexity of lesions during PCI, and whether the patient has respiratory distress and gout caused by ticagrelor. Given that the population included in this study were all at double high risk, and the complexity of lesions was also included in the assessment of ischemic risk, the use of ticagrelor in this study was based on the subjective habits and experience of different clinical doctors. An analysis was performed to assess the potential confounding effect of ticagrelor use at discharge, which revealed no significant difference between the two groups (p = 0.200). De-escalation strategies for DAPT include switching to a less potent P2Y12 receptor inhibitor, dosage reduction, and shortening the duration of DAPT. Clearly, De-escalation ticagrelor from the standard dose (90 mg BID) to a lower dose (60 mg BID) represents one of these regimens. Previous pharmacodynamic and pharmacokinetic study have demonstrated that in patients with a history of myocardial infarction, the antiplatelet effect of low-dose ticagrelor is comparable to that of the 90 mg twice daily regimen [3]. Thus, reducing ticagrelor from the standard dose to the lower dose. Its essence is a “response-guided de-escalation”. Rationale for reducing platelet related from 90 mg to 60 mg is a continuation of strong platelet inhibition. And it is applicable to patients who still have significant ischemic risk (such as complex lesions, diabetes, previous myocardial infarction), but cannot tolerate the risk of bleeding at a dose of 90 mg. The strategy of switching from ticagrelor 90 mg to clopidogrel 75 mg is primarily suitable for patients with bleeding risk outweighing ischemic risk, those intolerant to ticagrelor, and those who prefer or require clopidogrel due to personal preference or economic considerations.
The results of this study indicate that PFT-guided DAPT strategy selection can
benefit patients in terms of NACE, MACCE, cardiac death, and stroke without
increasing the risk of BARC bleeding (type 3 or greater). The results in this
dual high-risk ACS population are corroborated by other studies of PFT-guided
DAPT [20, 29]. The “Guided de-escalation of antiplatelet treatment in patients
with acute coronary syndrome undergoing percutaneous coronary intervention
(TROPICAL-ACS)” study analysis showed that for acute myocardial infarction
patients post-PCI, after 7 days of prasugrel and clopidogrel treatment, those
with low platelet reactivity,who de-escalated to clopidogrel had a lower
incidence of the primary endpoints (cardiac death, myocardial infarction, stroke,
and BARC
Although the prognostic impact of guided DAPT in dual high-risk ACS patients has
not been established, similar study have been conducted [8]. The ANTARCTIC study
was a randomized controlled trial (RCT) conducted across 35 centres in France.
This trial enrolled 877 elderly (age
Cox regression analysis revealed that in addition to guided DAPT being a
protective factor for NACE, MACCE, cardiac death, and stroke, cerebrovascular
disease, age, and number of stents were identified as risk factors for ischemic
events. Primary PCI is a risk factor for bleeding. These findings are consistent
with those of multiple studies and ischemic or bleeding risk scoring systems [4, 5, 7, 32, 33, 34, 35]. These factors directly increase the risk of NACE. The ACS
guidelines, developed by the ESC in 2023, propose criteria for high ischemic
risk, including complex CAD [32]. The OPT-CAD, patterns of non-adherence to antiplatelet regimens in stented patients-coronary artery thrombosis event (PARIS-CTE), and DAPT scoring
systems also include multiple risk factors, such as smoking [4, 5]. This study
adopted the risk evaluation system from the OPT-BIRISK trial, which is more
suitable for the Chinese ACS population. Compared with other scoring systems, it
incorporates PCI-related factors. This approach provides a comprehensive risk
profile by simultaneously evaluating both ischemic and bleeding in the ACS
patients undergoing PCI [7]. Since the population of the OPT-BIRISK trial was
Chinese, it was more appropriate to evaluate the Chinese population in this
study. The risk assessments in this study were conducted during patient
hospitalization, and no dynamic assessment was implemented at 3 months after PCI.
However, the OPT-BIRISK trial included indicators such as GFR and creatinine
level, which can be improved with standardized treatment. Improvements in these
indicators might lead to changes in the dual high-risk assessment results for
ischemia and bleeding at 3 months after PCI. In this study, the decision not to
perform dynamic assessments was primarily based on the low proportion of patients
with eGFR
(1) This was a cohort study, and physicians subjectively determined whether to
use PFT to guide the selection of DAPT protocol samples. In addition, we began
conducting PFT in October 2020. The enrollment times of the patients in the two
groups were not entirely synchronised, resulting in a selection bias in the study
population. Moreover, owing to the limitation of not conducting random grouping
in the cohort study, there were certain confounding factors in this research. To
address potential confounding, baseline characteristics were thoroughly analyzed,
and a Cox regression model was employed to control for their influence on the
outcomes. (2) Currently, there are advantages and disadvantages to the assessment
systems for the risk of ischemia and bleeding. This study adopted the definition
of dual high risk established in the OPT-BIRISK trial. Although it is the most
suitable risk assessment system for this study, it has the inherent limitation of
not comprehensively assessing risks. The OPT-BIRISK criteria are trial-specific
and their generalisability is questionable. (3) The current analysis was
performed on East-Asian patients, a population with distinct ischemic and
bleeding risk profiles compared to other ethnic groups. (4) LTA was used in this
study for PFT. However, the cutoff values for high and low platelet reactivity
obtained using different PFT methods are inconsistent, and the conversion of
platelet reactivity between different testing methods is currently unclear.
Therefore, further research is needed to explore whether other PFT methods could
benefit ACS patients at a dual high risk for ischaemia and bleeding. (5) Since
this was a real-world study, patients in the control group received various DAPT
regimens. Future studies should compare PFT-guided DAPT regimens with
de-escalation, escalation, extension, and shortening of DAPT to provide more
evidence for PFT-guided DAPT and to refine antiplatelet therapy strategies. (6)
At present, there is no recognized clinical cut-off value for the treatment of
platelet dysfunction worldwide. So each laboratory should develop a cutoff value
related to clinical outcomes based on their own treatment (medication) situation,
testing methods, and clinical practice. The normal range of ADP for platelet
function testing in our center is 52–84%, and the safe range for ADP treatment
is 30–50%. At the same time, drawing on relevant research [22, 36], we have set
the following standards: When the MAR(ADP)
The primary recommendation for patients with ACS with dual high risk among the East-Asian population is to use PFT to guide DAPT within 12 months after PCI, as it can reduce the incidence of NACE, primarily by lowering MACCE. However, the conclusions of this study need to be further verified in RCTs with a larger sample size.
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
ZX: Formulation or evolution of overarching research goals and aims; ML: Development or design of methodology; ML, YL and JW: Application of statistical or analysis of study data; ML, XS, CL, XW and ZZ: Data curation; ML: Writing the initial draft; ZX and ZZ: Critical review. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The studies involving human participants were reviewed and approved by the Ethics Committee of Clinical Investigation, Langfang People’s Hospital (2025-YXLW-002). Based on the retrospective study design of this study, the ethics committee believes that informed consent can be exempted. This study was conducted in accordance with the Declaration of Helsinki.
Not applicable.
This study was supported by the Medical Science Research Project of Hebei (no. 20232053).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM41544.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



