1 Center for Cardiac Intensive Care, Beijing Anzhen Hospital, Capital Medical University, 100029 Beijing, China
Abstract
This study aimed to evaluate the clinical efficacy of in-line mechanical insufflation–exsufflation (IL-MIE) in airway secretion management in patients receiving invasive mechanical ventilation after cardiopulmonary bypass (CPB).
A total of 56 patients who underwent CPB and required invasive mechanical ventilation in the Cardiac Surgery Intensive Care Unit of Beijing Anzhen Hospital, Capital Medical University, between July 2015 and July 2020, were enrolled and divided into an IL-MIE group (n = 28) and a conventional suction (CS) group (n = 28). The IL-MIE group received automated secretion clearance every 30 min for 8 h, supplemented with CS as needed, whereas the CS group received standard CS treatment. General patient data, respiratory and hemodynamic parameters, ventilator settings, CS frequency, mechanical ventilation duration, and intensive care unit (ICU) length of stay were recorded during the 8 h intervention.
At 4 h and 8 h, the IL-MIE group exhibited significantly higher arterial oxygen partial pressure, oxygenation index, and static compliance and low plateau pressure (p < 0.05). Heart rate was significantly lower in the IL-MIE group at 4 h ((99.21 ± 13.87) vs. (89.32 ± 10.66); p < 0.01) and 8 h ((96.71 ± 14.47) vs. (89.61 ± 9.34); p = 0.033). The IL-MIE group required fewer CS interventions (0 (0, 1) vs. 4 (3, 4); p < 0.01) and had a shorter duration of mechanical ventilation (20 (16.75, 22) vs. 24 (18.75, 26.5); p = 0.029) than those in the CS group.
By mimicking physiological airway clearance, IL-MIE significantly improves oxygenation and lung compliance, reduces the duration of mechanical ventilation, and maintains hemodynamic stability during respiratory management in patients after CPB.
Keywords
- in-line mechanical insufflation–exsufflation
- catheter suction
- post-cardiopulmonary bypass
- invasive mechanical ventilation
Lung injury is a major complication after cardiopulmonary bypass (CPB) [1]. The primary causes of post-CPB pulmonary dysfunction include atelectasis, pulmonary ischemia-reperfusion injury, systemic inflammatory response syndrome, pulmonary microthrombus formation, and transfusion-related acute lung injury [2, 3]. The interruption of mechanical ventilation (MV) during CPB is associated with the development of microatelectasis, hydrostatic pulmonary edema, and surfactant diffusion abnormalities. Consequently, pulmonary function declines. Clinically, this manifests as alveolar collapse, ventilation/perfusion mismatch, reduced lung compliance, and mucus retention, which may further precipitate atelectasis and ventilator-associated pneumonia (VAP).
The inflammatory cascade triggered by CPB leads to pulmonary capillary endothelial injury, facilitating the extravasation of protein-rich exudates into the alveolar and interstitial spaces. Concurrently, ischemia-reperfusion injury impairs ciliary beat frequency and mucociliary transport rate, resulting in increased mucus viscosity and retention in the small airways. This process establishes a vicious cycle of “mucus plugging-alveolar collapse-elevated infection risk”. Therefore, effective airway clearance is essential for improving oxygenation and serves as a core strategy to interrupt the progression of pulmonary complications.
In current clinical practice, conventional suction (CS) is used to clear airway secretions. This technique employs negative pressure to directly remove secretions from the major airways but is associated with several limitations. Catheter insertion can induce hemodynamic fluctuations and cause mechanical trauma to the tracheal mucosa, thereby triggering localized inflammation. Repeated procedures further increase the risk of mucosal bleeding and compromise the epithelial barrier integrity [4, 5], thereby increasing the likelihood of VAP [6, 7]. Notably, CS primarily targets the main bronchi and upper airways and has limited efficacy in clearing deep-seated secretions, resulting in suboptimal clearance. Moreover, manual suctioning requires frequent interruptions of ventilation and manual operation, rendering it highly dependent on nursing procedures. Technical proficiency directly influences therapeutic outcomes, thereby introducing variability into clinical effectiveness.
In-line mechanical insufflation–exsufflation (IL-MIE) employs a mechanical simulation of the airflow dynamics observed during the physiological cough process, specifically the “insufflation–exsufflation” cycle. During the inspiratory phase, positive pressure is applied to ensure optimal alveolar expansion. Subsequently, during the expiratory phase, the system rapidly switches to high negative pressure, generating a high-velocity expiratory airflow. This airflow transfers kinetic energy to secretions, propelling them upward into the tracheobronchial tree and facilitating the mobilization of peripheral secretions toward the proximal airways [8]. This process can be performed without disconnecting the ventilator, thereby ensuring continuous lung-protective ventilation. Recent studies have investigated the therapeutic efficacy of MIE in critically ill patients receiving MV [9, 10, 11]. As a noninvasive modality, MIE demonstrates minimal hemodynamic impact compared with that of CS and is associated with low incidence rates of trauma and VAP [12]. No adverse safety events have been reported with its use [13]. Evidence confirms the significant clinical value of MIE in facilitating weaning in patients within the intensive care unit (ICU) [14], and its application in chronic noninvasive ventilation has been increasingly adopted [15].
During the early postoperative period after CPB, patients frequently exhibit significant hemodynamic fluctuations, necessitating invasive positive-pressure ventilation, stringent monitoring of the oxygenation index (OI), and intensive airway management. This study aimed to evaluate the clinical utility of IL-MIE in patients receiving invasive MV during the early postoperative phase after CPB.
This historical, single-center, observational study was conducted at Beijing
Anzhen Hospital, Capital Medical University, China. Patients aged between 18–75
years who underwent invasive MV after CPB cardiac surgery between July 2015 and
July 2020 were enrolled. Written informed consent was obtained from all patients
or their legal guardians. Patients were excluded if they had any of the following
contraindications: acute spinal shock (within 48 h after spinal cord injury,
based on neurological examination records); recent respiratory tract trauma or
surgery (within the past 4 weeks, confirmed by surgical records or bronchoscopy
reports); conditions including rib fractures, pneumothorax (based on chest
computed tomography (CT) imaging reports obtained within 24 h of admission), or
hemoptysis (
In the CS group, patients were administered 100% oxygen for 1 min before CS intervention, according to the ICU’s standard operating protocol. The observation period for the CS group was 8 h. CS was performed whenever patients exhibited symptoms of airway secretion accumulation, such as a sawtooth waveform on the ventilator’s volume–pressure curve, audible rhonchi on auscultation, increased peak airway pressure (volume-controlled ventilation mode), decreased tidal volume (pressure controlled ventilation mode), oxygen saturation below 95%, visible airway secretions in the respiratory circuit, acute respiratory distress, suspected aspiration of gastric contents or upper airway secretions.
The IL-MIE device (RC001-01A, CoughSync, Ruxin Technology Development Co., Ltd., Beijing, China) was used for 8 h, with automated secretion clearance performed every 30 min. Supplemental suctioning was performed using criteria identical to those in the CS group. No supplemental oxygen beyond the baseline requirement was provided to the IL-MIE group. The IL-MIE device operated 10 cycles per treatment, with an exsufflation pressure of –60 cmH2O. Accumulated airway secretions in the sputum collection cup were cleared regularly (Fig. 1).
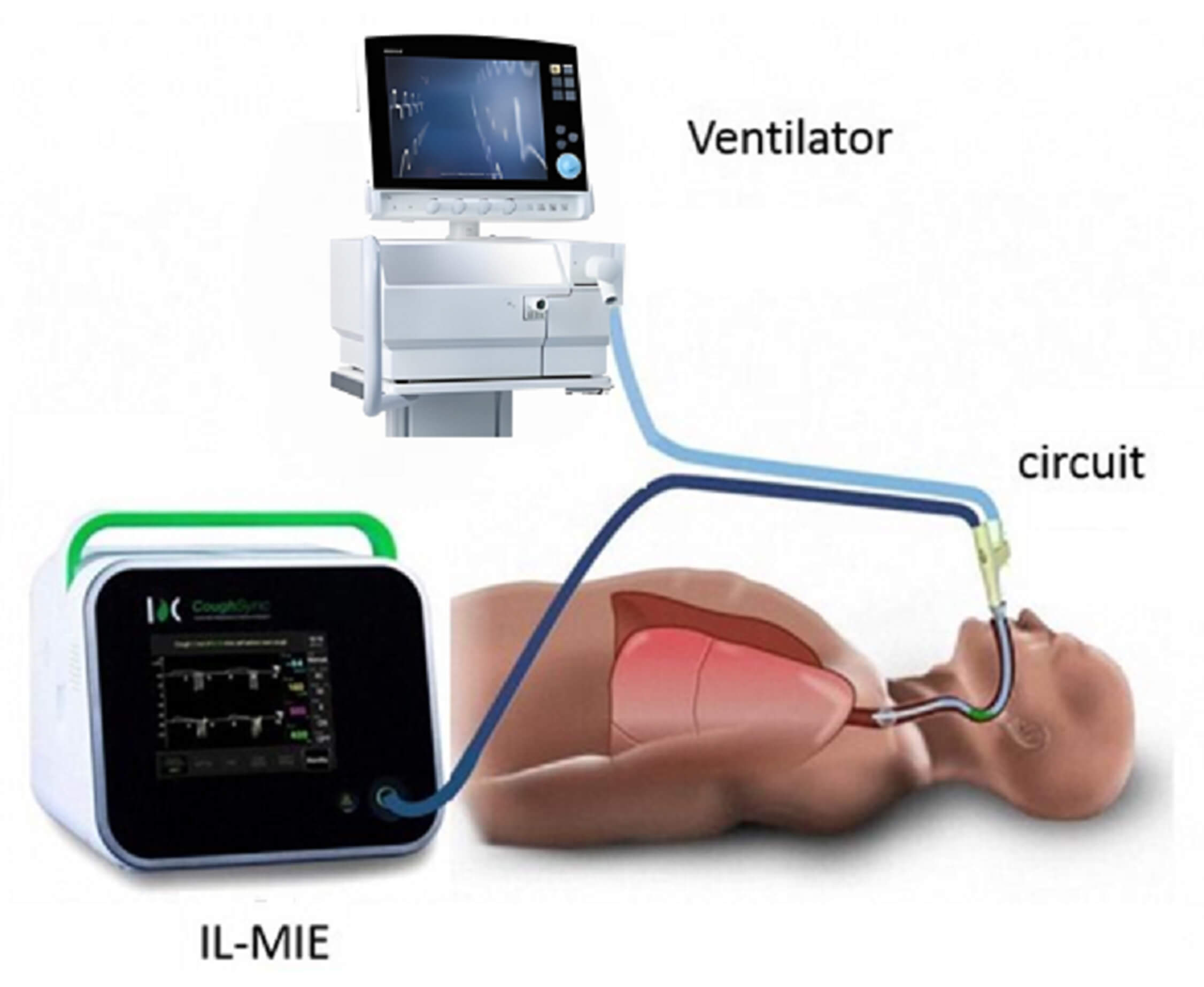 Fig. 1.
Fig. 1.
Setup and functioning of in-line mechanical insufflation–exsufflation (IL-MIE).
General information and relevant data were collected at 0, 4, and 8 h after treatment initiation. Respiratory-related clinical parameters included arterial oxygen partial pressure, OI, and arterial carbon dioxide partial pressure. The recorded ventilator parameters were tidal volume, peak inspiratory pressure, airway plateau pressure (Pplat), and static compliance (Cst). Circulation-related clinical indicators included heart rate (HR) and mean arterial pressure (MAP). Additionally, the frequency of endotracheal suction, duration of MV, and length of ICU stay were recorded during the observation period.
All analyses were performed using SPSS (version 25.0; IBM Corp., Armonk, NY,
USA). Categorical variables and frequencies were presented as percentages, and
continuous variables as “Mean
Fifty-six patients were enrolled in the study and stratified into the CS (n = 28) and IL-MIE groups (n = 28). One patient with severe ischemic heart disease was excluded. Normally distributed continuous data were analyzed using the t-test, whereas categorical data were analyzed using the chi-square test, specifically for ventilation modes; Fisher’s exact test was applied based on expected frequencies. The CS group comprised 35.7% men, whereas the IL-MIE group included 42.9% men. No significant differences were observed between the two groups in terms of age, body mass index (BMI), history of smoking or drinking, or comorbidities, including hypertension, diabetes, stroke, and chronic obstructive pulmonary disease. Additionally, no significant differences were observed in initial HR or MAP values. Regarding the type of surgery, valvular, aortic, and congenital heart surgeries accounted for 82.1%, 21.4%, and 14.3%, respectively, in the CS group. There were also no differences in ventilator modes between the two groups (Table 1).
| Characteristic | CS group (n = 28) | IL-MIE group (n = 28) | T/ |
p-value | |
| Men | 10 (35.7) | 12 (42.9) | 0.299 | 0.584 | |
| Age, years | 56.11 |
55.71 |
0.139 | 0.890 | |
| BMI, kg/m2 | 21.38 |
21.78 |
–0.567 | 0.573 | |
| Smoking | 12 (42.9) | 17 (60.7) | 1.788 | 0.181 | |
| Drinking | 16 (57.1) | 14 (50.0) | 0.287 | 0.592 | |
| Comorbid conditions | |||||
| Hypertension | 15 (53.6) | 19 (67.9) | 1.198 | 0.274 | |
| Diabetes | 15 (53.6) | 16 (57.1) | 0.072 | 0.788 | |
| Stroke | 5 (17.9) | 2 (7.1) | 1.469 | 0.225 | |
| COPD | 3 (10.7) | 4 (14.3) | 1.163 | 0.686 | |
| Type of surgery | |||||
| Valvular surgery | 23 (82.1) | 20 (71.4) | 0.902 | 0.342 | |
| Aortic surgery | 6 (21.4) | 5 (17.9) | 0.113 | 0.737 | |
| Congenital heart surgery | 4 (14.3) | 5 (17.9) | 0.132 | 0.716 | |
| Ventilator mode | |||||
| Volume-controlled | 25 (89.3) | 27 (96.4) | 0.611 | ||
| Pressure-controlled | 3 (10.7) | 1 (3.6) | 0.611 | ||
| HR, bpm | 80.57 |
78.21 |
1.089 | 0.281 | |
| MAP, mmHg | 80.79 |
81.57 |
–0.378 | 0.707 | |
IL-MIE, in-line mechanical insufflation–exsufflation; CS, conventional suction; BMI, body mass index; COPD, chronic obstructive pulmonary disease; HR, heart rate; MAP, mean arterial pressure.
Indicators of respiration and circulation were observed at 0, 4, and 8 h (Table 2). A repeated-measures ANOVA was used. At baseline, no significant differences
were observed in OI between the two groups. At 4 h, the mean OI value was
significantly lower in the CS group than in the IL-MIE group [(212.99
| CS group | IL-MIE group | F-value | p-value | ||
| PaO2, mmHg | 0 h | 124.07 |
136.61 |
2.425 | 0.125 |
| 4 h | 112.50 |
129.50 |
0.115 | 0.011 | |
| 8 h | 108.43 |
132.82 |
0.287 | 0.000 | |
| F-value | 6.073 | 1.773 | |||
| p-value | 0.004 | 0.180 | |||
| Main Effect | |||||
| Time (F, P) | 5.504, 0.007 | ||||
| Group (F, P) | 9.363, 0.003 | ||||
| Interaction (F, P) | 2.341, 0.106 | ||||
| OI, mmHg | 0 h | 213.25 |
236.90 |
2.523 | 0.118 |
| 4 h | 212.99 |
270.98 |
16.470 | 0.000 | |
| 8 h | 225.24 |
268.15 |
8.188 | 0.006 | |
| F-value | 0.845 | 8.176 | |||
| p-value | 0.435 | 0.001 | |||
| Main Effect | |||||
| Time (F, P) | 5.386, 0.006 | ||||
| Group (F, P) | 11.271, 0.001 | ||||
| Interaction (F, P) | 3.086, 0.050 | ||||
| PaCO2, mmHg | 0 h | 40.22 |
38.84 |
1.212 | 0.276 |
| 4 h | 39.82 |
38.82 |
1.168 | 0.285 | |
| 8 h | 39.22 |
38.60 |
0.495 | 0.485 | |
| F-value | 0.472 | 0.038 | |||
| p-value | 0.626 | 0.963 | |||
| Main Effect | |||||
| Time (F, P) | 0.356, 0.701 | ||||
| Group (F, P) | 3.194, 0.080 | ||||
| Interaction (F, P) | 0.128, 0.880 | ||||
| VT, mL | 0 h | 461.79 |
436.79 |
2.155 | 0.148 |
| 4 h | 462.50 |
442.14 |
1.333 | 0.253 | |
| 8 h | 463.21 |
448.21 |
0.770 | 0.384 | |
| F-value | 0.069 | 4.465 | |||
| p-value | 0.943 | 0.016 | |||
| Main Effect | |||||
| Time (F, P) | 2.819, 0.069 | ||||
| Group (F, P) | 1.389, 0.244 | ||||
| Interaction (F, P) | 1.715, 0.190 | ||||
| PIP, cmH2O | 0 h | 19.68 |
20.36 |
1.866 | 0.178 |
| 4 h | 20.16 |
20.07 |
0.043 | 0.837 | |
| 8 h | 20.07 |
20.39 |
0.406 | 0.527 | |
| F-value | 0.617 | 0.221 | |||
| p-value | 0.543 | 0.803 | |||
| Main Effect | |||||
| Time (F, P) | 0.172, 0.842 | ||||
| Group (F, P) | 1.124, 0.294 | ||||
| Interaction (F, P) | 0.579, 0.562 | ||||
| Pplat, cmH2O | 0 h | 12.71 |
12.54 |
0.266 | 0.608 |
| 4 h | 12.71 |
10.93 |
12.454 | 0.001 | |
| 8 h | 12.14 |
11.00 |
6.353 | 0.015 | |
| F-value | 0.959 | 7.934 | |||
| p-value | 0.390 | 0.001 | |||
| Main Effect | |||||
| Time (F, P) | 5.547, 0.005 | ||||
| Group (F, P) | 22.264, 0.000 | ||||
| Interaction (F, P) | 2.994, 0.049 | ||||
| Cst, mL/cmH2O | 0 h | 43.66 |
41.97 |
0.689 | 0.410 |
| 4 h | 40.13 |
46.12 |
4.794 | 0.033 | |
| 8 h | 44.69 |
51.84 |
6.060 | 0.017 | |
| F-value | 2.353 | 9.115 | |||
| p-value | 0.105 | 0.000 | |||
| Main Effect | |||||
| Time (F, P) | 7.628, 0.001 | ||||
| Group (F, P) | 4.263, 0.044 | ||||
| Interaction (F, P) | 4.705, 0.011 | ||||
| HR, bpm | 0 h | 89.14 |
86.75 |
0.567 | 0.455 |
| 4 h | 99.21 |
89.32 |
8.957 | 0.004 | |
| 8 h | 96.71 |
89.61 |
4.766 | 0.033 | |
| F-value | 4.783 | 0.453 | |||
| p-value | 0.012 | 0.683 | |||
| Main Effect | |||||
| Time (F, P) | 4.640, 0.012 | ||||
| Group (F, P) | 10.469, 0.002 | ||||
| Interaction (F, P) | 1.463, 0.236 | ||||
| MAP, mmHg | 0 h | 77.11 |
79.29 |
1.748 | 0.192 |
| 4 h | 76.71 |
77.89 |
0.522 | 0.473 | |
| 8 h | 76.57 |
74.54 |
1.778 | 0.188 | |
| F-value | 0.060 | 5.149 | |||
| p-value | 0.942 | 0.009 | |||
| Main Effect | |||||
| Time (F, P) | 2.895, 0.06 | ||||
| Group (F, P) | 0.215, 0.645 | ||||
| Interaction (F, P) | 1.942, 0.148 |
PaO2, arterial oxygen partial pressure; OI, oxygenation index; PaCO2,
arterial carbon dioxide partial pressure; VT, tidal volume; PIP, peak inspiratory
pressure; Pplat, airway plateau pressure; Cst, static compliance; HR, heart rate;
MAP, mean arterial pressure; IL-MIE, in-line mechanical
insufflation–exsufflation; CS, conventional suction. * p
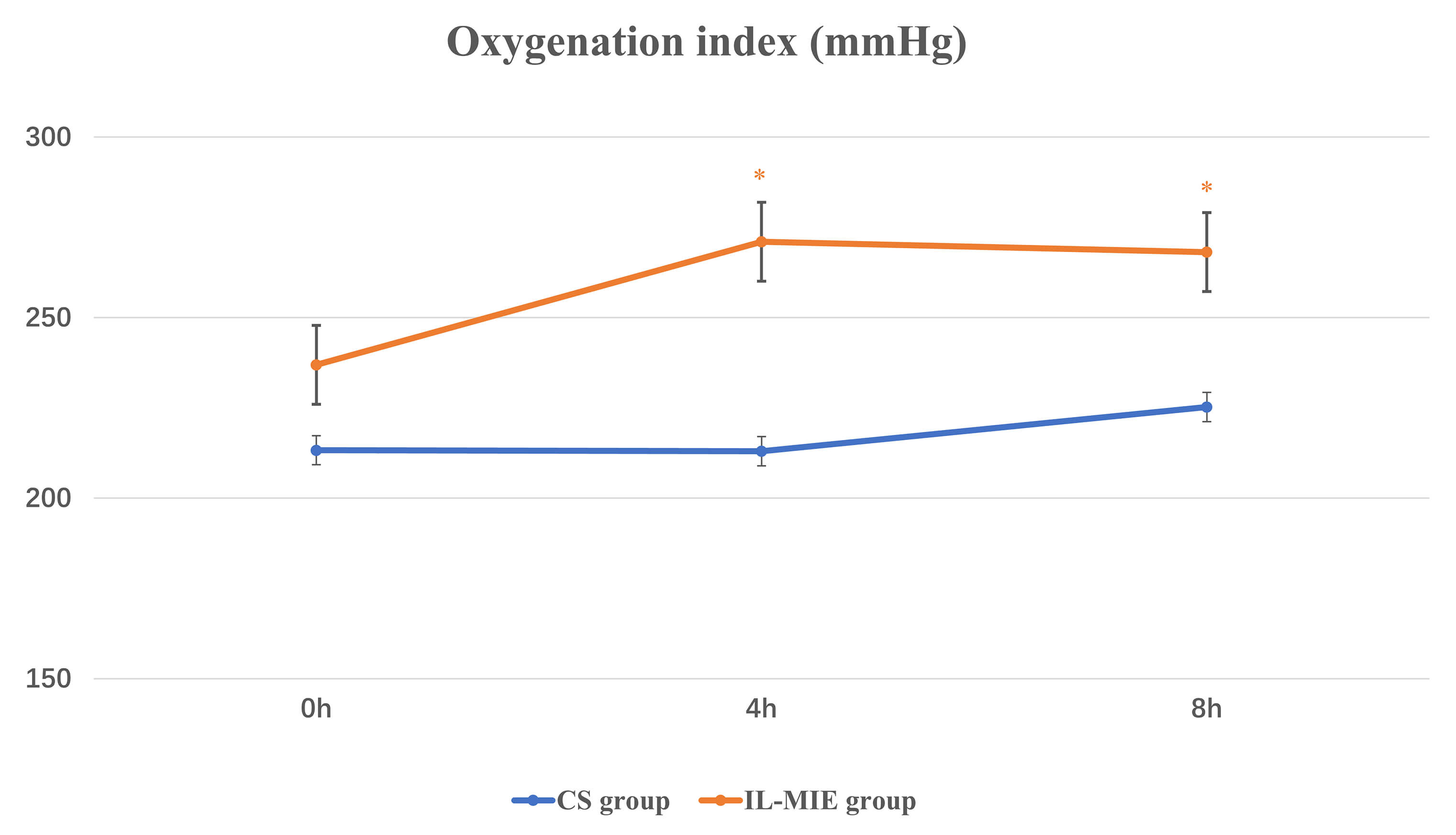 Fig. 2.
Fig. 2.
Oxygenation index. * p
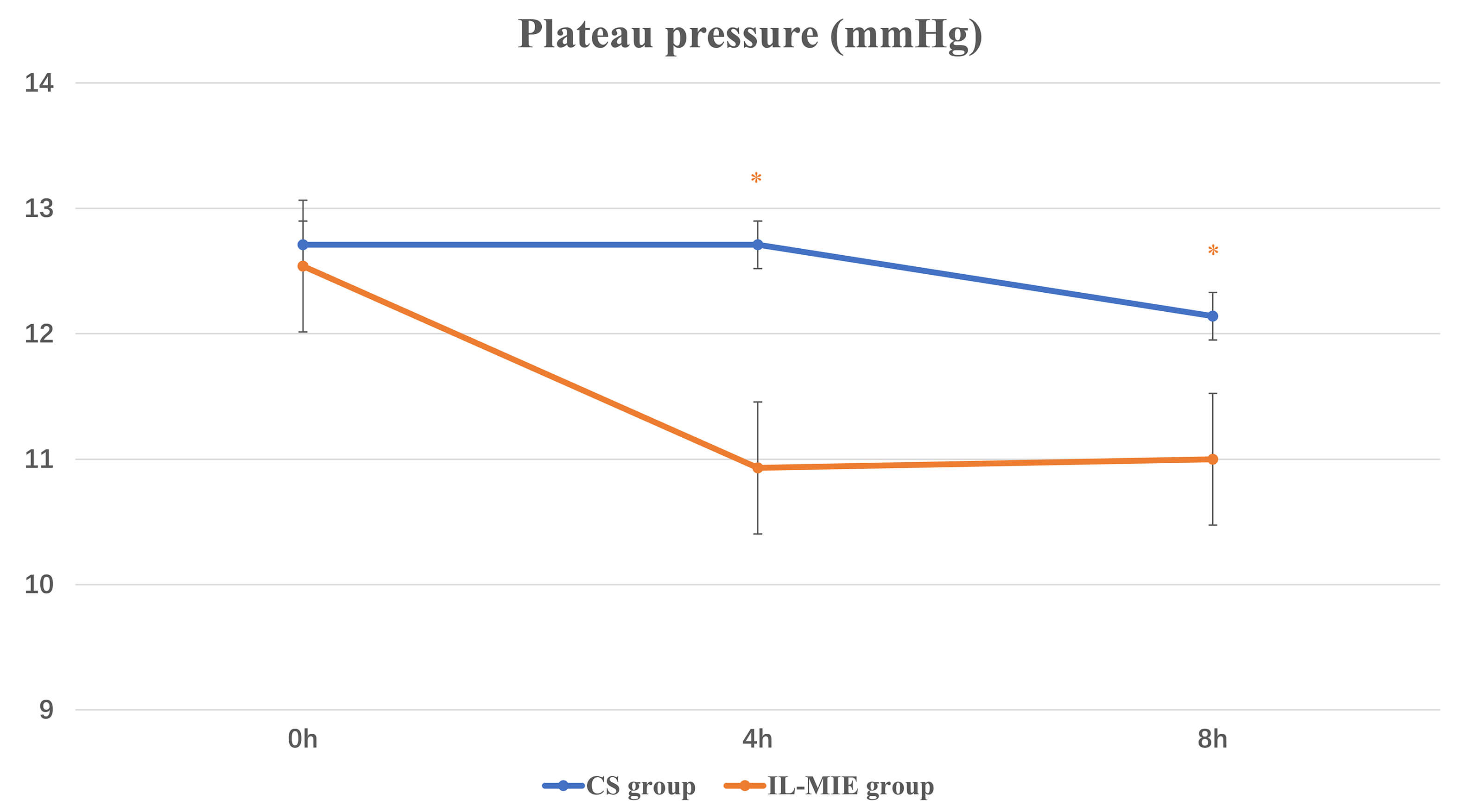 Fig. 3.
Fig. 3.
Plateau pressure. * p
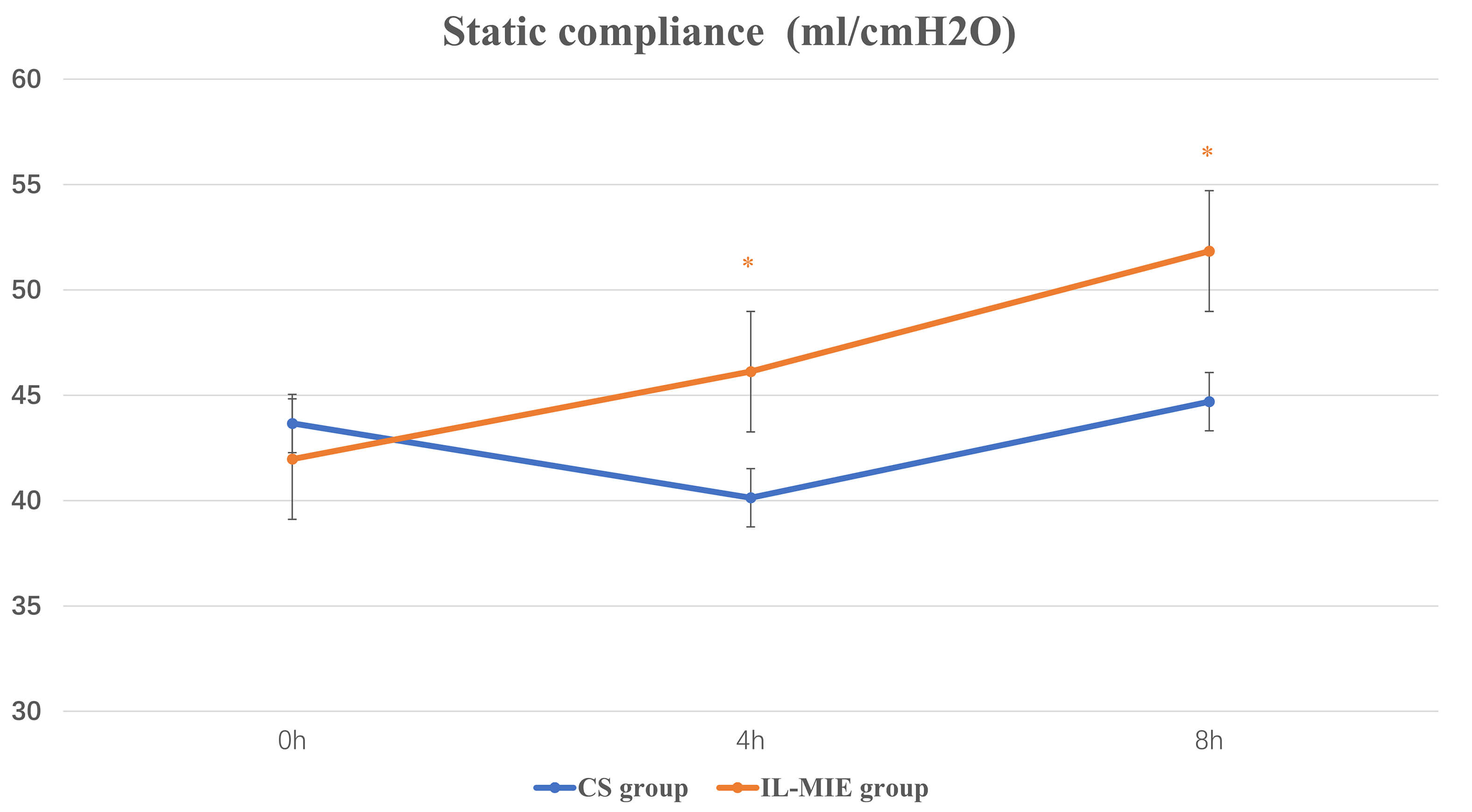 Fig. 4.
Fig. 4.
Static compliance. * p
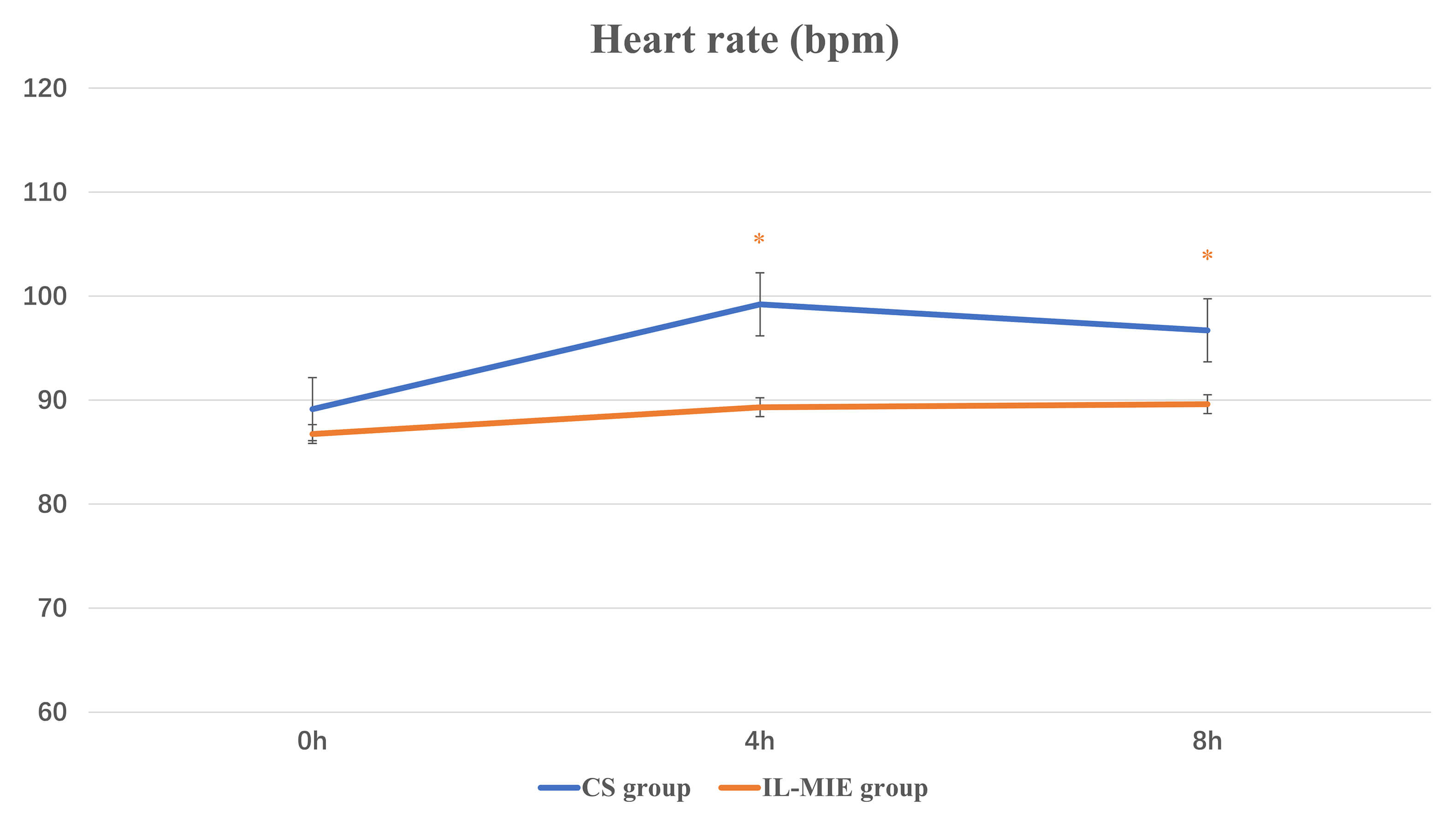 Fig. 5.
Fig. 5.
Heart rate. * p
In the IL-MIE group, the time to additional CS treatment was significantly
shorter than in the CS group [0 (0, 1) vs. 4 (3, 4), p
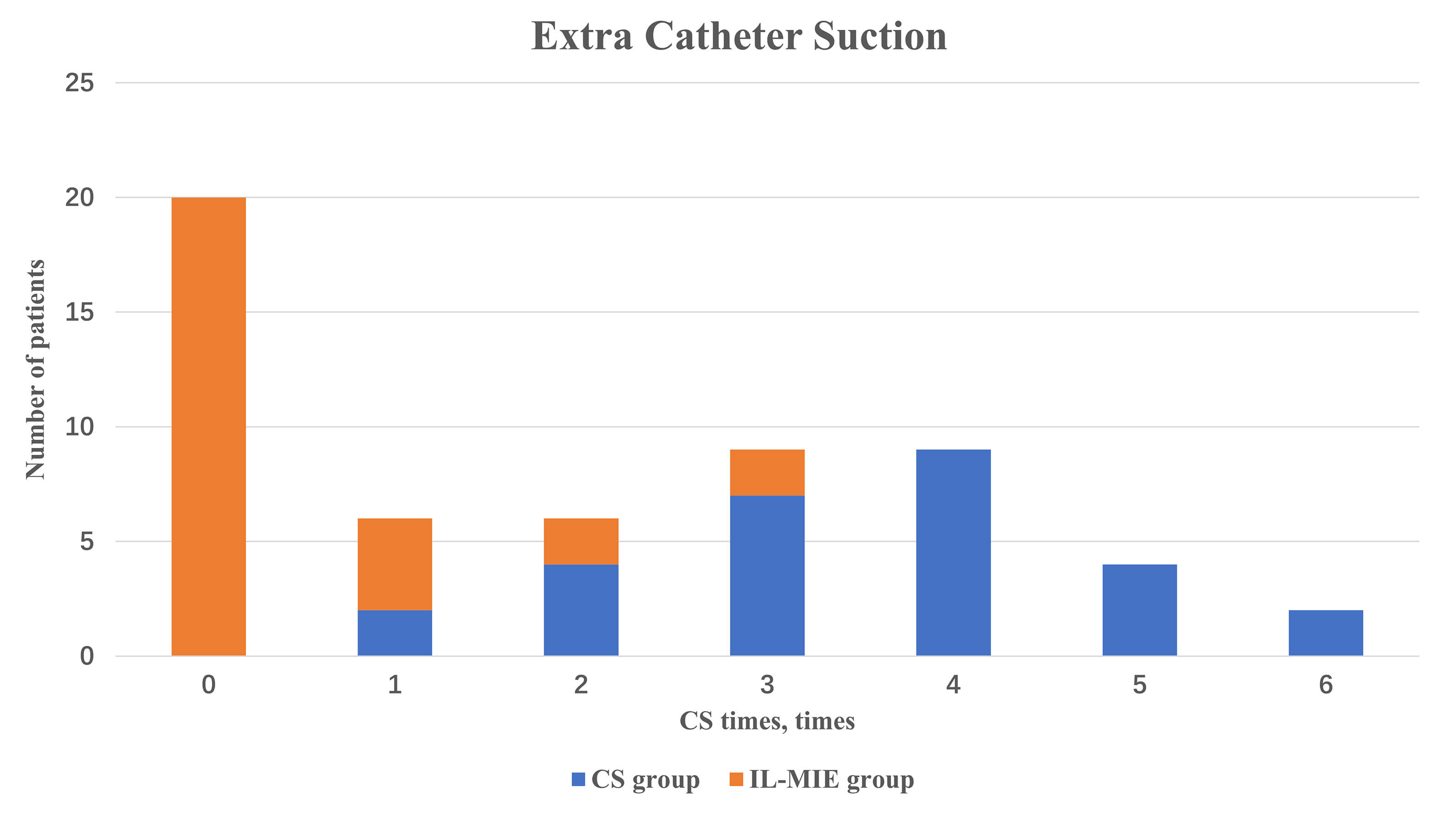 Fig. 6.
Fig. 6.
Extra catheter suction. IL-MIE, in-line mechanical insufflation–exsufflation; CS, conventional suction.
| CS group (n = 28) | IL-MIE group (n = 28) | U | p-value | |
| Extra CS, times | 4 (3, 4) | 0 (0, 1) | 32.000 | 0.000 |
| MV time, hours | 24 (18.75, 26.5) | 20 (16.75, 22) | 260.000 | 0.029 |
| VAP, % | 2 (7.14) | 0 (0) | 0.493 | |
| ICU stay, hours | 26.5 (20.75, 33) | 22 (20.75, 25.25) | 295.500 | 0.113 |
MV, mechanical ventilation; VAP, ventilator-associated pneumonia; IL-MIE, in-line mechanical insufflation–exsufflation; CS, conventional suction; ICU, intensive care unit.
An adjusted analysis for age, type of surgery, and chronic obstructive pulmonary disease status was conducted with respect to OI, duration of MV, and ICU length of stay. No statistically significant differences were observed in any subgroup (Table 4).
| OI (8 h), mmHg | MV time, hours | ICU stay, hours | ||
| Years | ||||
| 256.67 (221.67, 306.25) | 20.00 (17.00, 26.00) | 22.00 (20.50, 27.00) | ||
| 272.50 (220.00, 330.00) | 20.00 (16.00, 20.00) | 22.00 (20.00, 26.00) | ||
| p-value | 0.447 | 0.371 | 0.763 | |
| Types of surgery | ||||
| Valvular surgery | 272.72 (239.75, 315.13) | 19.00 (16.25, 20.00) | 22.00 (20.25, 24.75) | |
| Aortic surgery | 205.00 (183.33, 264.17) | 24.00 (18.00, 32.00) | 25.00 (19.50, 34.50) | |
| Congenital heart surgery | 273.33 (223.00, 335.30) | 20.00 (16.00, 23.00) | 24.00 (18.00, 39.50) | |
| p-value | 0.113 | 0.253 | 0.662 | |
| COPD | ||||
| Yes | 301.67 (210.83, 348.75) | 17.00 (16.00, 19.50) | 20.50 (16.25, 38.25) | |
| No | 260.34 (224.58, 306.88) | 20.00 (17.00, 22.00) | 23.00 (21.00, 25.75) | |
| p-value | 0.431 | 0.131 | 0.356 | |
OI, oxygenation index; MV, mechanical ventilation; COPD, chronic obstructive pulmonary disease; IL-MIE, in-line mechanical insufflation–exsufflation; CS, conventional suction.
The primary objectives of airway management in mechanically ventilated patients are to maintain airway patency, ensure adequate ventilation, and prevent pulmonary complications. Improper timing, duration, or technique of CS may lead to complications, such as airway mucosal injury, atelectasis, bronchospasm, hypoxemia, infection, hemodynamic instability, arrhythmias, and artificial airway obstruction. In ICU patients with excessive secretions, the frequent use of rigid suction catheters inevitably causes mucosal trauma, induces severe coughing, and exacerbates tissue hypoxia. In patients who underwent cardiac surgery, this may result in alveolar collapse, atelectasis, or hemodynamic fluctuations, thus failing to achieve an optimal balance between safety and efficacy. A meta-analysis by Jongerden et al. [16] encompassing 15 studies demonstrated that CS increased bacterial colonization in the airway and elevated the risk of VAP. Therefore, overcoming the limitations of rigid suction catheters, avoiding the interruption of the inspiratory phase during MV, minimizing hemodynamic instability, and improving weaning rates are critical for effective airway clearance in the ICU.
In this study, OI values were significantly higher in the IL-MIE group than in the CS group, and the duration of MV was markedly shorter. Notably, the CS group required 100% oxygen supplementation during each suction procedure, whereas the IL-MIE group did not require supplemental oxygen before or after treatment. Moreover, over the 8 h treatment period, OI values in the IL-MIE group were significantly improved compared with those in the CS group. Inadequate secretion clearance is a major risk factor for failed ventilator weaning in ICU patients [17, 18, 19]. CS often fails to reach the left main bronchus [20] and is less effective in clearing secretions beyond the first-generation bronchi. By contrast, physiological coughing can effectively clear secretions from the 7th–8th generation small bronchi [21], with comparable efficacy in both the left and right airways [22]. This suggests that IL-MIE may outperform CS in clearing secretions from the left lung and deeper small airways beyond the first-generation bronchi. Although OI represents indirect evidence, it serves as a key clinical indicator of improved pulmonary ventilation and gas exchange resulting from effective clearance of deep secretions.
The IL-MIE group exhibited significantly lower Pplat and higher lung compliance than the CS group. Reduced lung compliance decreases the effective tidal volume per unit time, impairing oxygenation and tissue perfusion, thereby adversely affecting the efficacy of MV. For instance, in patients with severe acute respiratory distress syndrome and poor alveolar compliance, alveolar volume often fails to normalize within a standard respiratory cycle post-suction, predisposing them to atelectasis. However, reducing suctioning frequency or duration may lead to inadequate secretion clearance, increased VAP incidence, and prolonged invasive ventilation. When IL-MIE is performed in a mode where the exsufflation volume does not exceed the patient’s tidal volume, it minimizes alveolar collapse, preserves lung compliance, and shortens the duration of invasive ventilation. A significant improvement in Cst was observed in the IL-MIE group after intervention. Increased compliance indirectly reflects alveolar recruitment and improved airway patency, which is consistent with the effects of secretion clearance.
There were no significant differences in VAP rates or ICU stay between the two groups. The development of VAP is a multifactorial process. The key target of IL-MIE is secretion accumulation and aspiration. Biofilm formation, oral bacterial colonization, and a decline in host immunity are also contributing factors to VAP. Similarly, many factors influence ICU stay, including multi-organ function, infection, and nutritional status. However, it is difficult to determine the impact of IL-MIE on long-term outcomes. In our study, the determination of both the 8 h treatment duration and 30 min intervention interval for IL-MIE was based on considerations of hemodynamic stability and clinical workflow. Future research could employ a randomized design to investigate whether increasing frequency or extending overall treatment duration might further reduce the need for CS, improve oxygenation, or affect long-term outcomes. Our study provides a foundational framework and reference for exploring this critical parameter, and determining the optimal regimen should be the key focus of subsequent investigations.
Our findings indicate that IL-MIE provides a safe and stable continuous, timed,
and automated airway clearance therapy. Compared with the CS group, no
significant increase in HR was observed in the IL-MIE group. Moreover, during the
observation period, mean HR values were lower in the IL-MIE group than in the CS
group at both 4 h and 8 h. However, MAP showed no significant intergroup
differences. This suggests that noninvasive secretion management with IL-MIE
promotes hemodynamic stability. IL-MIE is particularly valuable for maintaining
hemodynamic stability in patients after cardiac surgery. IL-MIE significantly
reduced or eliminated the need for CS in mechanically ventilated patients.
Specifically, 71.4% of IL-MIE-treated patients did not require CS, whereas the
remaining 21.4% experienced a
We conducted a secondary analysis of specific indicators in the IL-MIE group based on surgery type, age, and underlying pulmonary disease. However, no statistically significant differences were observed in the subgroup analysis. Future prospective studies with larger sample sizes or longer treatment durations may help minimize bias and validate these findings.
This study had several limitations. First, it was a single-center, historical, non-randomized controlled trial, which may introduce potential bias. The small sample size may have affected the generalizability of our results. Multicenter, randomized controlled trials with larger cohorts are needed to validate these findings. Second, the observation period was limited to 8 h, whereas post-cardiac surgery patients often require prolonged MV. In future studies, we will extend the observation period or increase follow-up data to evaluate potential long-term clinical benefits. Third, a direct assessment of secretion clearance efficacy via dry weight measurements was not performed. Cst reflects alveolar recruitment and improved airway patency, which align with the effects of secretion clearance. However, the ultimate goal of airway clearance is to improve oxygenation and ventilation efficiency and reduce ventilation duration, rather than merely removing secretions of a specific weight.
The IL-MIE achieves secretion clearance by closely mimicking physiological airway clearance mechanisms. It significantly improves oxygenation and lung compliance, shortens the duration of MV, and maintains hemodynamic stability during the respiratory management of patients after CPB. Its automated, continuous operation reduces clinical workload. Future multicenter, large-scale randomized controlled trials are necessary to further validate the efficacy of this technology.
CPB, cardiopulmonary bypass; MV, mechanical ventilation; VAP, ventilator-associated pneumonia; CS, conventional suction; IL-MIE, in-line mechanical insufflation–exsufflation; OI, oxygenation index; Pplat, airway plateau pressure; Cst, static compliance; HR, heart rate; MAP, mean arterial pressure.
All data generated during this study are included in this article. The datasets used and analyzed during the current study are available on reasonable request.
DH contributed to collecting and analyzing the patient data, drafting the manuscript. CL, MJ, and LW designed the research study. HW, XH, and LW provided help and advice on the research. HW contributed substantially to the acquisition and interpretation of clinical data, and participated in drafting the methodology section. XH contributed substantially to the analysis and interpretation of statistical data, participated in drafting the methodology section, and assisted in critical revision of the manuscript for important intellectual content. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the Declaration of Helsinki. The research protocol was approved by the Ethics Committee of Beijing Anzhen Hospital (Ethic Approval Number: 2025215x), and all of the participants provided signed informed consent.
We thank the physicians and nurses in the participating intensive care units for their support during patient selection and data collection.
This research was funded by: National Key Clinical College Construction Program (Critical Care Medicine), National Natural Science Foundation of China (No. 82170400), Beijing Natural Science Foundation (No. 7244327), and Beijing Nova Program (No. 20220484043).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






