1 Department of Cardiology, Tianjin Medical University General Hospital, 300052 Tianjin, China
2 Department of Cardiology, Tianjin Chest Hospital, 300222 Tianjin, China
3 Department of Emergency, Qilu Hospital of Shandong University, 250012 Jinan, Shandong, China
†These authors contributed equally.
Abstract
Metabolic dysfunction significantly influences cardiovascular outcomes following ST-elevation myocardial infarction (STEMI). The triglyceride–glucose (TyG) index and triglyceride–glucose–body mass index (TyG–BMI) serve as surrogate markers of insulin resistance, whereas B-type natriuretic peptide (BNP) levels reflect cardiac dysfunction. However, the combined prognostic value of these biomarkers for predicting major adverse cardiovascular events (MACEs) in patients with STEMI remains underexplored.
We conducted a retrospective cohort study of 1177 consecutive patients with STEMI who underwent percutaneous coronary intervention between August 2018 and December 2023. Patients were stratified into four groups based on the TyG index (cutoff: 7.2), TyG–BMI (cutoff: 186), and BNP level (cutoff: 300 pg/mL). The primary endpoint was MACEs, defined as a composite of all-cause mortality, nonfatal myocardial infarction, ischemia-driven repeat revascularization, heart failure hospitalization, and cerebrovascular events. Cox proportional hazards models with progressive adjustment were employed to assess independent and combined prognostic significance.
A total of 483 patients (41.0%) experienced MACEs during a median follow-up of 461 days (interquartile range (IQR): 79–672). Patients with both an elevated TyG index (≥7.2) and a high BNP concentration (≥300 pg/mL) demonstrated the highest cardiovascular risk profile and a more than twofold increased MACE risk (hazard ratio (HR) 2.18, 95% confidence interval (CI): 1.57–3.03; p < 0.001) compared with the reference group (those with a low TyG index and low BNP concentration). Similarly, patients with elevated TyG–BMIs (≥186) and BNP levels had an 81% increased risk (HR 1.81, 95% CI: 1.30–2.51; p < 0.001). Meanwhile, the combined TyG index + BNP model demonstrated superior predictive accuracy (area under the curve (AUC): 0.67) compared with the individual biomarkers and the established Global Registry of Acute Coronary Events (GRACE) score (AUC: 0.58). Subgroup analyses revealed particularly pronounced associations in older patients, females, and those with hypertension.
The combination of the TyG index or TyG–BMI with BNP provides enhanced prognostic stratification for predicting MACEs in STEMI patients, offering superior discriminatory capacity compared with that of individual biomarkers. This integrated approach may facilitate personalized risk assessment and guide therapeutic decision-making in clinical practice.
Keywords
- ST-elevation myocardial infarction
- triglyceride–glucose index
- B-type natriuretic peptide
- major adverse cardiovascular events
- risk stratification
- insulin resistance
ST-elevation myocardial infarction (STEMI) represents the most severe form of acute coronary syndrome and is characterized by complete coronary artery occlusion and substantial myocardial necrosis. Despite significant advances in reperfusion strategies and evidence-based pharmacotherapy, STEMI patients continue to face a considerable risk of major adverse cardiovascular events (MACEs), with reported rates ranging from 10% to 20% annually following the index event [1, 2]. Accurate risk stratification remains paramount for optimizing therapeutic interventions and improving long-term cardiovascular outcomes.
Traditional risk assessment tools, including the Global Registry of Acute Coronary Events (GRACE) score and Thrombolysis in Myocardial Infarction (TIMI) risk score, primarily incorporate demographic, clinical, and procedural variables [3, 4]. However, these conventional models may not fully capture the complex pathophysiological processes underlying postinfarction cardiovascular risk, particularly the intricate interplay between metabolic dysfunction and cardiac stress responses.
Insulin resistance has emerged as a critical pathophysiological mechanism linking metabolic abnormalities to cardiovascular disease progression. Compared with the homeostatic model assessment of insulin resistance (HOMA-IR), the triglyceride–glucose (TyG) index serves as a reliable surrogate marker of insulin resistance with superior predictive capacity [5, 6]. Recent investigations have demonstrated significant associations between an elevated TyG index and adverse cardiovascular outcomes across diverse populations, including patients with acute coronary syndromes [7, 8]. Furthermore, the triglyceride–glucose–body mass index (TyG–BMI), which incorporates anthropometric parameters, may provide enhanced metabolic risk assessment by reflecting both insulin resistance and adiposity-related cardiovascular risk [9, 10].
However, the prognostic utility of single biomarkers remains limited. For instance, metabolic indicators such as the TyG index or TyG–BMI mainly reflect insulin resistance and obesity-related risk, whereas cardiac stress markers such as B-type natriuretic peptide (BNP) primarily capture the hemodynamic burden and ventricular dysfunction. Relying on a single dimension of risk information may fail to fully characterize the multifaceted pathophysiological processes after STEMI, thereby restricting predictive performance. In contrast, combined biomarker approaches integrate complementary mechanisms and provide a more comprehensive assessment, offering superior sensitivity and specificity in risk stratification and supporting more precise clinical decision-making.
In conjunction with metabolic risk assessment, BNP represents a well-established biomarker of cardiac dysfunction and hemodynamic stress. Elevated BNP levels reflect increased ventricular wall tension and volume overload and serve as powerful predictors of heart failure development and cardiovascular mortality following myocardial infarction [11, 12]. The prognostic utility of BNP has been consistently demonstrated across various cardiovascular conditions, with guideline recommendations supporting its clinical application for risk stratification and therapeutic monitoring [13].
The concept of integrated biomarker approaches for cardiovascular risk prediction has attracted considerable attention, as complex cardiovascular pathophysiology involves multiple interdependent mechanisms. The combination of metabolic markers with cardiac stress indicators may provide complementary prognostic information, potentially enhancing risk discrimination beyond individual biomarker assessment. However, the combined prognostic value of the TyG index, TyG–BMI, and BNP level for predicting MACEs in STEMI patients has not been comprehensively investigated.
Given the clinical importance of accurate risk stratification in STEMI management and the potential synergistic effects of metabolic and cardiac biomarkers, we hypothesized that the combination of the TyG index or TyG–BMI with BNP would provide superior prognostic discrimination for MACE prediction compared with individual biomarker assessment. Therefore, we conducted this comprehensive retrospective cohort study to (1) evaluate the individual prognostic significance of the TyG index, TyG–BMI, and BNP for MACE prediction in STEMI patients; (2) investigate the combined prognostic value of these biomarkers using systematic risk stratification approaches; (3) assess the incremental predictive capacity of integrated biomarker models compared to established risk scores; and (4) identify patient subgroups who may derive particular benefit from this combined biomarker approach.
We conducted a retrospective cohort analysis involving STEMI patients who were admitted to Tianjin Medical University General Hospital between August 2018 and December 2023. The study protocol was approved by the Ethics Committee of Tianjin Medical University General Hospital (approval number: IRB2023-YX-301-01/2023) and adhered to the principles outlined in the Declaration of Helsinki. Owing to the retrospective nature of the study, the requirement for informed consent was waived.
The inclusion criteria were adults (
At the time of admission, baseline demographic and clinical characteristics were comprehensively recorded. Current smoking status was defined as the daily consumption of at least one cigarette within the 30 days prior to hospitalization [15]. The diagnosis of diabetes mellitus was established either through a prior confirmed diagnosis or through the use of glucose-lowering medications. Hypertension was identified according to one of the following criteria: (1) a documented clinical diagnosis, (2) the use of antihypertensive medications before admission, or (3) a new diagnosis made during the index hospitalization based on repeated blood pressure readings exceeding 140/90 mmHg.
Sample size calculations were performed on the basis of Cox proportional hazards models, assuming a clinically meaningful hazard ratio of 1.5 with 80% power at a two-sided significance level of 0.05. Previous studies have reported a cumulative MACE incidence of 15% over a two-year follow-up period [16, 17]. The final required sample size, after accounting for potential follow-up losses, was 708 patients, and our cohort of 1177 patients ensured adequate statistical power.
Data, including demographic information, medical history, and clinical
parameters, were extracted from the patients’ electronic medical records.
Laboratory analyses, performed at admission, included metabolic indices (e.g.,
fasting blood glucose and lipid profiles), renal function, and cardiac biomarkers
(BNP and troponin). The TyG index was computed as TyG Index = ln(triglycerides
(mg/dL)
All patients underwent coronary angiography and subsequent percutaneous coronary intervention (PCI) per current guidelines [18]. The complexity of coronary lesions was assessed using the Synergy Between PCI With TAXUS and Cardiac Surgery (SYNTAX) scoring system by two experienced interventional cardiologists who were blinded to the patients’ clinical data. In cases of scoring discrepancies, a third cardiologist was involved to reach a consensus. The residual SYNTAX score (rSS) was then computed to quantify the untreated coronary disease burden after PCI. Both the initial SYNTAX score and the rSS have been shown to have prognostic value in previous studies [19]. Follow-up was conducted using electronic medical records and structured telephone interviews to ensure comprehensive event tracking. The MACE variable was defined as a composite of all-cause mortality, nonfatal myocardial infarction, ischemia-driven revascularization, hospitalization for heart failure, and cerebrovascular events.
The primary outcome of interest was MACE occurrence, which was analyzed using multivariable Cox regression models with restricted cubic splines (RCSs) to assess nonlinear associations. RCSs were employed to allow for flexible modeling of continuous variables, capturing potential nonlinear relationships between the biomarkers and the outcome of interest. The knots for the RCS were placed at the 10th, 50th, and 90th percentiles of each continuous variable to ensure a balanced representation across the range of data. This method was specifically chosen to account for potential nonlinear trends, which are often observed in medical outcomes, and to provide more accurate and clinically relevant hazard ratios.
The study population was stratified on the basis of cutoff values for the TyG
index (
The prognostic accuracy of these models was assessed using receiver operating characteristic (ROC) curve analysis to calculate the area under the curve (AUC), and the combined models were compared with individual biomarkers to establish risk scores. The AUC provides an aggregate measure of the model’s discriminative ability, and a comparison of the AUC values across the models was performed to determine whether the addition of biomarkers improved the prediction accuracy beyond traditional risk scores.
Model adjustments included demographic factors, clinical variables, coronary disease severity (SYNTAX score), and medical interventions, ensuring that potential confounders were accounted for in the analysis. To comprehensively assess the robustness of the associations, we constructed six progressively adjusted models as follows: Model 1 was the unadjusted model. Model 2 was adjusted for sex and age. Model 3 included the variables in Model 2, with the addition of heart rate, systolic blood pressure (SBP), diastolic blood pressure (DBP), current smoking status, hypertension, diabetes, stroke, left ventricular ejection fraction (LVEF), SYNTAX score, and rSS Model 4 was built upon Model 3 by further adjusting for the number of stents, antiplatelet therapy, statins, beta-blockers, angiotensin-converting enzyme inhibitor (ACEI)/angiotensin receptor blockers (ARBs)/angiotensin receptor and neprilysin inhibitor (ARNI), Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors, sodium-glucose cotransporter 2 (SGLT2) inhibitors, hemoglobin, platelet count, estimated glomerular filtration rate (eGFR), troponin T (TnT), and low-density lipoprotein cholesterol (LDL-C). Model 5 extended Model 4 by incorporating bootstrapping to enhance statistical robustness. Model 6 was performed using propensity score matching (PSM). A multivariable logistic regression model was applied to estimate the propensity score, adjusting for the covariates included in Model 4. Patients were matched 1:1 on the basis of their propensity scores using a greedy matching algorithm without replacement, with a caliper width set at 0.2 of the standard deviation of the log-transformed propensity score.
The proportional hazards assumption was verified using Schoenfeld residuals, and no significant violations were identified, indicating that the Cox regression model’s assumptions were met. Additionally, subgroup analyses were conducted to explore potential differences in risk prediction across subgroups on the basis of age, sex, diabetes status, and coronary complexity. These subgroup analyses help assess the heterogeneity of risk and evaluate the generalizability of the findings across different patient characteristics.
To further assess potential multicollinearity among the covariates in the multivariable models, collinearity diagnostics were performed using the variance inflation factor (VIF). A VIF value greater than 5 was considered indicative of significant multicollinearity.
All analyses were conducted using Stata version 16 (StataCorp, College Station,
TX, USA). Two-sided p-values
Between August 2018 and December 2023, our retrospective cohort study initially identified 1480 consecutive STEMI patients at Tianjin Medical University General Hospital. After excluding 76 patients due to inability to complete follow-up (all attributed to loss of contact, including invalid contact information or relocation), 1177 patients were enrolled for the final analysis. During a median follow-up period of 461 days (interquartile range: 79–672 days), 483 patients (41.0%) experienced MACEs. The distributions of individual MACE components, including all-cause mortality, nonfatal myocardial infarction, cerebrovascular events, heart failure hospitalization, and ischemia-induced revascularization, are summarized in Supplementary Table 1.
Baseline characteristics stratified by MACE occurrence are presented in
Supplementary Table 2. Patients who developed MACEs were significantly
older than those without MACEs (67 (59, 73) vs. 65 (54, 71) years, p
The MACE group exhibited greater coronary disease complexity, as evidenced by
significantly higher SYNTAX scores [22.0 (17.0, 27.5) vs. 16.0 (11.0, 21.5),
p
All the covariates demonstrated acceptable collinearity (VIF
The frequency distributions of the TyG index, TyG–BMI index, and BNP level are
illustrated in Supplementary Fig. 2. Multivariable restricted cubic
spline analysis revealed distinct relationships between these biomarkers and MACE
risk (Fig. 1A–C). Both the TyG index and BNP level exhibited nonlinear
relationships with MACE risk, with significant thresholds at a TyG index
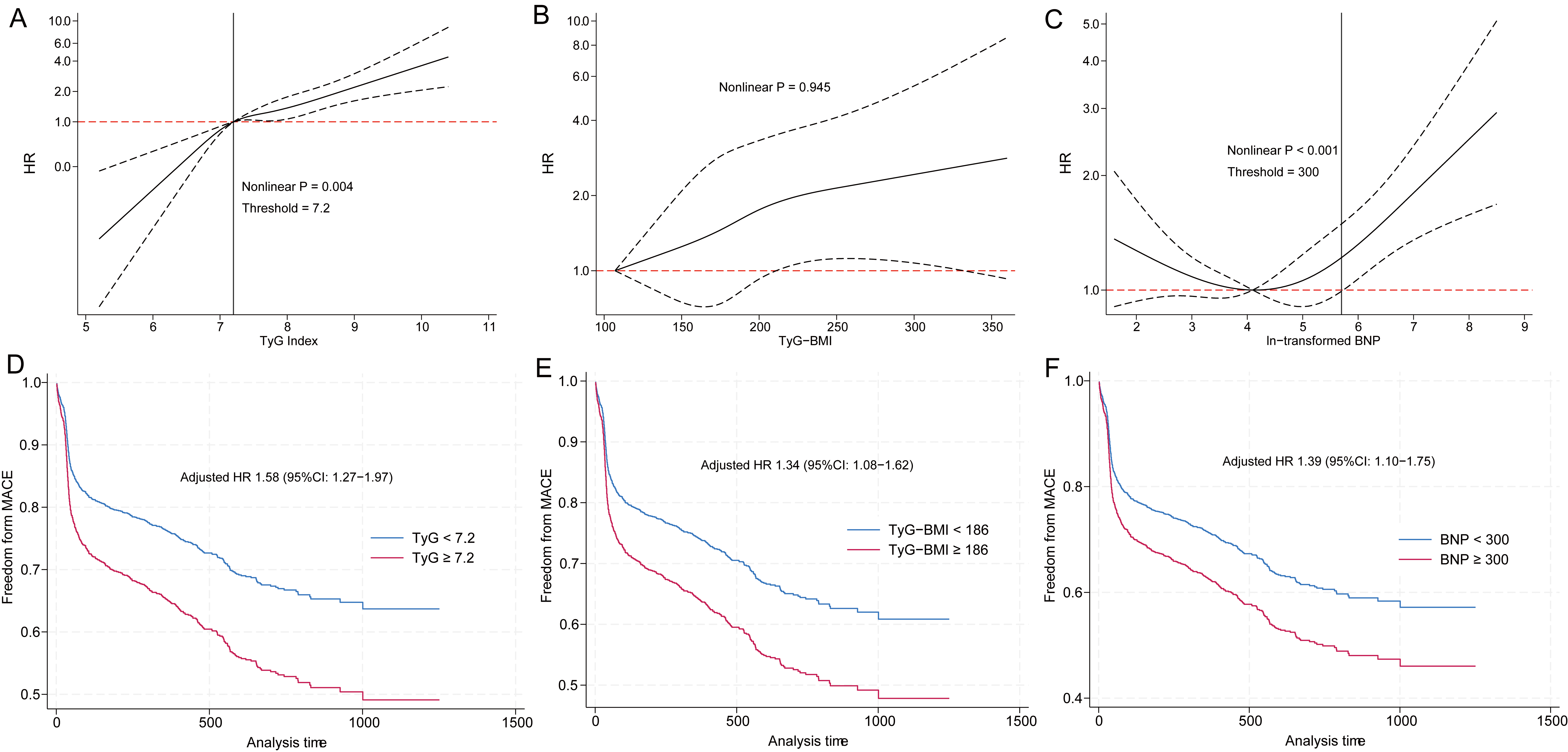 Fig. 1.
Fig. 1.
Relationships between the TyG index, TyG–BMI, and BNP level and
the risk of a MACE in patients with STEMI. (A) Multivariable RCS analysis
revealing the nonlinear relationship between the TyG index and MACE risk
(inflection point: 7.2). (B) Multivariable RCS analysis demonstrating the
predominantly linear association between TyG–BMI and MACE risk (optimal cutoff:
186). (C) Multivariable RCS analysis revealing the nonlinear relationship between
BNP and MACE risk (inflection point: 300). (D) Kaplan‒Meier curves stratified by
the TyG index (
Kaplan‒Meier survival curves with corresponding log-rank test results (Fig. 1D–F) illustrated differential MACE risk stratification by these biomarkers
according to their respective cutoff points. Compared with those with lower
values, those with an elevated TyG index (
On the basis of the established cutoff values, 1177 patients were stratified
into four groups according to their TyG index (
| TyG index |
TyG index |
p value | ||||
| BNP |
BNP |
BNP |
BNP | |||
| Age (years) | 66.0 (57.0, 72.0) | 70.0 (61.0, 76.0) | 64.0 (54.0, 70.0) | 69.0 (62.0, 75.0) | ||
| Female, n (%) | 63 (18.4%) | 26 (26.3%) | 122 (20.9%) | 54 (35.3%) | ||
| Heart rate (bpm) | 76.0 (67.0, 88.0) | 78.0 (70.0, 94.0) | 78.0 (68.0, 89.0) | 84.0 (73.0, 96.0) | ||
| SBP (mmHg) | 136.0 (122.0, 151.0) | 132.0 (117.0, 148.0) | 140.0 (124.0, 157.0) | 130.0 (113.0, 146.0) | ||
| DBP (mmHg) | 85.0 (75.0, 94.0) | 77.0 (68.0, 89.0) | 86.0 (77.0, 97.0) | 81.0 (71.0, 90.0) | ||
| Current smoking, n (%) | 174 (50.9%) | 40 (40.4%) | 272 (46.7%) | 63 (41.2%) | 0.120 | |
| Hypertension, n (%) | 210 (61.4%) | 59 (59.6%) | 423 (72.6%) | 106 (69.3%) | 0.001 | |
| Diabetes, n (%) | 41 (12.0%) | 20 (20.2%) | 204 (35.0%) | 83 (54.2%) | ||
| Stroke, n (%) | 46 (13.5%) | 15 (15.2%) | 70 (12.0%) | 28 (18.3%) | 0.220 | |
| Interventions | ||||||
| Stent, n (%) | 0.008 | |||||
| 0 | 28 (8.2%) | 10 (10.1%) | 53 (9.1%) | 14 (9.2%) | ||
| 1 | 208 (60.8%) | 49 (49.5%) | 355 (60.9%) | 70 (45.8%) | ||
| 106 (31.0%) | 40 (40.4%) | 175 (30.0%) | 69 (45.1%) | |||
| SYNTAX score | 18.5 (11.0, 22.5) | 19.0 (13.0, 25.0) | 18.0 (13.0, 24.0) | 20.5 (14.5, 28.0) | 0.001 | |
| rSS | 5.0 (2.0, 10.0) | 8.0 (3.0, 13.0) | 7.0 (2.0, 11.0) | 8.0 (3.0, 13.0) | ||
| LVEF (%) | 49.0 (43.0, 56.0) | 45.0 (40.0, 50.0) | 48.0 (43.0, 55.0) | 42.0 (36.0, 47.0) | ||
| Platelet (×109/L) | 219.0 (190.0, 255.0) | 217.0 (172.0, 280.0) | 224.0 (189.0, 269.0) | 224.0 (184.0, 265.0) | 0.586 | |
| Hemoglobin (g/L) | 144.0 (134.0, 156.0) | 129.0 (112.0, 141.0) | 148.0 (135.0, 159.0) | 133.0 (121.0, 148.0) | ||
| eGFR (mL/min/1.73 m2) | 100.7 (85.3, 117.2) | 93.8 (71.3, 112.6) | 97.0 (79.6, 115.4) | 83.6 (64.1, 104.9) | ||
| LDL-C (mmol/L) | 2.7 (2.2, 3.2) | 2.5 (2.0, 3.0) | 3.2 (2.6, 3.8) | 3.1 (2.5, 3.8) | ||
| TnT (ng/mL) | 0.2 (0.1, 1.4) | 1.4 (0.2, 3.1) | 0.2 (0.0, 1.3) | 1.2 (0.4, 3.1) | ||
| TyG index | 7.0 (6.7, 7.1) | 7.0 (6.7, 7.1) | 7.7 (7.5, 8.1) | 7.7 (7.5, 8.0) | ||
| BNP (pg/mL) | 54.7 (21.0, 125.0) | 553.0 (427.0, 978.0) | 48.2 (17.0, 103.0) | 669.0 (465.0, 1028.0) | ||
| P2Y12i, n (%) | ||||||
| Clopidogrel | 122 (35.7%) | 52 (52.5%) | 191 (32.8%) | 81 (52.9%) | ||
| Ticagrelor | 220 (64.3%) | 47 (47.5%) | 392 (67.2%) | 72 (47.1%) | ||
| Statin, n (%) | 0.002 | |||||
| Rosuvastatin | 313 (91.5%) | 78 (78.8%) | 525 (90.1%) | 140 (91.5%) | ||
| Atorvastatin | 29 (8.5%) | 21 (21.2%) | 58 (9.9%) | 13 (8.5%) | ||
| ACEI/ARB/ARNI, n (%) | 90 (26.3%) | 28 (28.3%) | 216 (37.0%) | 49 (32.0%) | 0.007 | |
| Beta blocker, n (%) | 195 (57.0%) | 49 (49.5%) | 376 (64.5%) | 82 (53.6%) | 0.004 | |
| PCSK9i, n (%) | 43 (12.6%) | 11 (11.1%) | 106 (18.2%) | 25 (16.3%) | 0.077 | |
| SGLT2i, n (%) | 27 (7.9%) | 15 (15.2%) | 136 (23.3%) | 49 (32.0%) | ||
Abbreviations: SBP, systolic blood pressure; DBP, diastolic blood pressure; SYNTAX, Synergy Between PCI With TAXUS and Cardiac Surgery score; rSS, residual SYNTAX score; LVEF, left ventricular ejection fraction; eGFR, estimated glomerular filtration rate; TNT, troponin T; BNP, B-type natriuretic peptide; LDL-C, low-density lipoprotein cholesterol; P2Y12i, P2Y12 receptor inhibitor; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; ARNI, angiotensin receptor and neprilysin inhibitor; PCSK9i, PCSK9 inhibitors; SGLT2i, sodium-glucose cotransporter 2 inhibitor.
Similarly, patients were stratified on the basis of TyG–BMI (
| TyG–BMI |
TyG–BMI |
p value | ||||
| BNP |
BNP |
BNP |
BNP | |||
| Age (years) | 67.0 (58.0, 73.0) | 70.0 (62.0, 76.0) | 62.0 (51.0, 69.0) | 68.0 (59.5, 74.5) | ||
| Female, n (%) | 100 (23.3%) | 45 (31.2%) | 85 (17.2%) | 35 (32.4%) | ||
| Heart rate (bpm) | 75.0 (65.0, 86.0) | 83.0 (70.0, 95.5) | 80.0 (70.0, 90.0) | 81.0 (73.0, 95.0) | ||
| SBP (mmHg) | 135.0 (119.0, 151.0) | 129.0 (113.5, 146.5) | 143.0 (126.0, 157.0) | 132.5 (113.5, 151.5) | ||
| DBP (mmHg) | 83.0 (73.0, 93.0) | 78.0 (69.5, 89.0) | 89.0 (78.0, 98.0) | 81.0 (70.0, 92.5) | ||
| Current smoking, n (%) | 198 (46.0%) | 57 (39.6%) | 248 (50.1%) | 46 (42.6%) | 0.110 | |
| Hypertension, n (%) | 266 (61.9%) | 89 (61.8%) | 367 (74.1%) | 76 (70.4%) | ||
| Diabetes, n (%) | 81 (18.8%) | 42 (29.2%) | 164 (33.1%) | 61 (56.5%) | ||
| Stroke, n (%) | 49 (11.4%) | 27 (18.8%) | 67 (13.5%) | 16 (14.8%) | 0.158 | |
| Interventions | ||||||
| Stent, n (%) | 0.004 | |||||
| 0 | 32 (7.4%) | 15 (10.4%) | 49 (9.9%) | 9 (8.3%) | ||
| 1 | 268 (62.3%) | 69 (47.9%) | 295 (59.6%) | 50 (46.3%) | ||
| 130 (30.2%) | 60 (41.7%) | 151 (30.5%) | 49 (45.4%) | |||
| SYNTAX score | 18.0 (12.0, 22.5) | 19.0 (13.0, 25.8) | 18.5 (12.5, 24.5) | 20.2 (15.0, 27.5) | 0.003 | |
| rSS | 5.0 (2.0, 11.0) | 8.0 (3.0, 12.0) | 7.0 (2.0, 11.0) | 9.0 (4.5, 14.0) | ||
| LVEF (%) | 48.0 (43.0, 55.0) | 42.0 (38.0, 48.0) | 48.0 (43.0, 55.0) | 43.0 (38.0, 48.5) | ||
| Platelet (×109/L) | 218.0 (189.0, 256.0) | 222.5 (172.5, 280.5) | 225.0 (191.0, 268.0) | 223.0 (182.5, 257.0) | 0.549 | |
| Hemoglobin (g/L) | 143.0 (131.0, 154.0) | 129.5 (116.0, 141.5) | 150.0 (139.0, 161.0) | 133.0 (120.0, 148.5) | ||
| eGFR (mL/min/1.73 m2) | 99.2 (83.3, 114.5) | 93.3 (75.0, 113.4) | 97.6 (81.2, 118.2) | 77.2 (59.0, 98.3) | ||
| LDL-C (mmol/L) | 2.8 (2.3, 3.4) | 2.7 (2.0, 3.3) | 3.1 (2.6, 3.9) | 3.1 (2.5, 3.9) | ||
| TnT (ng/mL) | 0.2 (0.0, 1.3) | 1.1 (0.3, 3.1) | 0.2 (0.1, 1.4) | 1.4 (0.4, 3.1) | ||
| TyG–BMI index | 167.7 (154.3, 177.5) | 164.4 (148.3, 173.9) | 211.6 (196.0, 234.2) | 206.5 (196.1, 223.1) | ||
| BNP (pg/mL) | 57.6 (21.7, 123.0) | 602.0 (441.0, 1027.5) | 42.0 (16.0, 99.7) | 636.5 (457.5, 959.5) | ||
| P2Y12i, n (%) | ||||||
| Clopidogrel | 169 (39.3%) | 79 (54.9%) | 144 (29.1%) | 54 (50.0%) | ||
| Ticagrelor | 261 (60.7%) | 65 (45.1%) | 351 (70.9%) | 54 (50.0%) | ||
| Statin, n (%) | 0.064 | |||||
| Rosuvastatin | 390 (90.7%) | 120 (83.3%) | 448 (90.5%) | 98 (90.7%) | ||
| Atorvastatin | 40 (9.3%) | 24 (16.7%) | 47 (9.5%) | 10 (9.3%) | ||
| ACEI/ARB/ARNI, n (%) | 120 (27.9%) | 32 (22.2%) | 186 (37.6%) | 45 (41.7%) | ||
| Beta blocker, n (%) | 235 (54.7%) | 74 (51.4%) | 336 (67.9%) | 57 (52.8%) | ||
| PCSK9i, n (%) | 59 (13.7%) | 11 (7.6%) | 90 (18.2%) | 25 (23.1%) | 0.002 | |
| SGLT2i, n (%) | 46 (10.7%) | 28 (19.4%) | 117 (23.6%) | 36 (33.3%) | ||
Abbreviations: SBP, systolic blood pressure; DBP, diastolic blood pressure; SYNTAX, SYNTAX score; rSS, residual SYNTAX score; LVEF, left ventricular ejection fraction; eGFR, estimated glomerular filtration rate; TNT, troponin T; BNP, B-type natriuretic peptide; LDL-C, low-density lipoprotein cholesterol; P2Y12i, P2Y12 receptor inhibitor; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; ARNI, angiotensin receptor and neprilysin inhibitor; PCSK9i, PCSK9 inhibitors; SGLT2i, sodium-glucose cotransporter 2 inhibitor.
The synergistic effects of the combined TyG index and BNP level on adverse cardiovascular outcomes across the five progressively adjusted models are shown in Table 3 and Fig. 2A. In the unadjusted analysis (Model 1), compared with the reference group (low TyG index-low BNP level), all three groups with at least one elevated biomarker exhibited a significantly higher risk. Following sequential adjustments for demographic characteristics, clinical parameters, procedural variables, and pharmacological interventions, elevated risk persisted across all groups.
| Index type | Model | Hazard ratio (95% CI) | p for trend | |||
| Low index + BNP |
Low index + BNP |
High index + BNP |
High index + BNP | |||
| TyG index (cutoff: 7.2) | Model 1 | Ref. | 2.13 (1.49–3.04)*** | 1.82 (1.43–2.32)*** | 3.35 (2.51–4.48)*** | |
| Sample size: | Model 2 | Ref. | 2.02 (1.41–2.38)*** | 1.86 (1.46–2.38)*** | 3.25 (2.43–4.36)*** | |
| TyG |
Model 3 | Ref. | 1.80 (1.25–2.60)** | 1.65 (1.29–2.11)*** | 2.38 (1.73–3.28)*** | |
| TyG |
Model 4 | Ref. | 1.57 (1.07–2.30)* | 1.66 (1.28–2.14)*** | 2.18 (1.57–3.03)*** | |
| TyG |
Model 5 | Ref. | 1.57 (1.03–2.39)* | 1.66 (1.28–2.15)*** | 2.18 (1.53–3.12)*** | |
| TyG |
Model 6 | Ref. | 2.15 (1.21–3.83)** | 1.63 (1.22–2.19)** | 2.47 (1.52–4.01)*** | 0.219 |
| TyG–BMI (cutoff: 186) | Model 1 | Ref. | 1.84 (1.38–2.46)*** | 1.42 (1.14–1.76)*** | 3.01 (2.26–4.02)*** | |
| Sample size: | Model 2 | Ref. | 1.76 (1.31–2.35)*** | 1.52 (1.22–1.90)*** | 2.99 (2.24–3.99)*** | |
| TyG–BMI |
Model 3 | Ref. | 1.55 (1.14–2.11)** | 1.39 (1.11–1.74)** | 2.14 (1.57–2.92)*** | |
| TyG–BMI |
Model 4 | Ref. | 1.45 (1.06–1.99)* | 1.35 (1.08–1.71)* | 1.81 (1.30–2.51)*** | 0.001 |
| TyG–BMI |
Model 5 | Ref. | 1.45 (1.03–2.05)* | 1.35 (1.07–1.72)* | 1.81 (1.26–2.60)** | 0.001 |
| TyG–BMI |
Model 6 | Ref. | 1.63 (1.04–2.57)* | 1.25 (0.96–1.62) | 1.77 (1.11–2.82)* | 0.292 |
Abbreviations: Model 1 is considered the unadjusted model. Model 2 is adjusted
for sex and age. Model 3 includes the variables of Model 2 with the addition of
heart rate, SBP, DBP, current smoking status, hypertension status, diabetes
status, stroke status, LVEF, SYNTAX score, and rSS. Model 4 builds upon Model 3
by further adjusting for the number of stents, antiplatelet therapy, statin,
beta-blockers, ACEI/ARBs/ARNI, PCSK9i, SGLT2i, hemoglobin, platelet count, eGFR,
TnT, and LDL-C. Model 5 is an extension of Model 4 with the addition of
bootstrapping for statistical robustness. Model 6 is the propensity score
matching (PSM) model. * denotes p
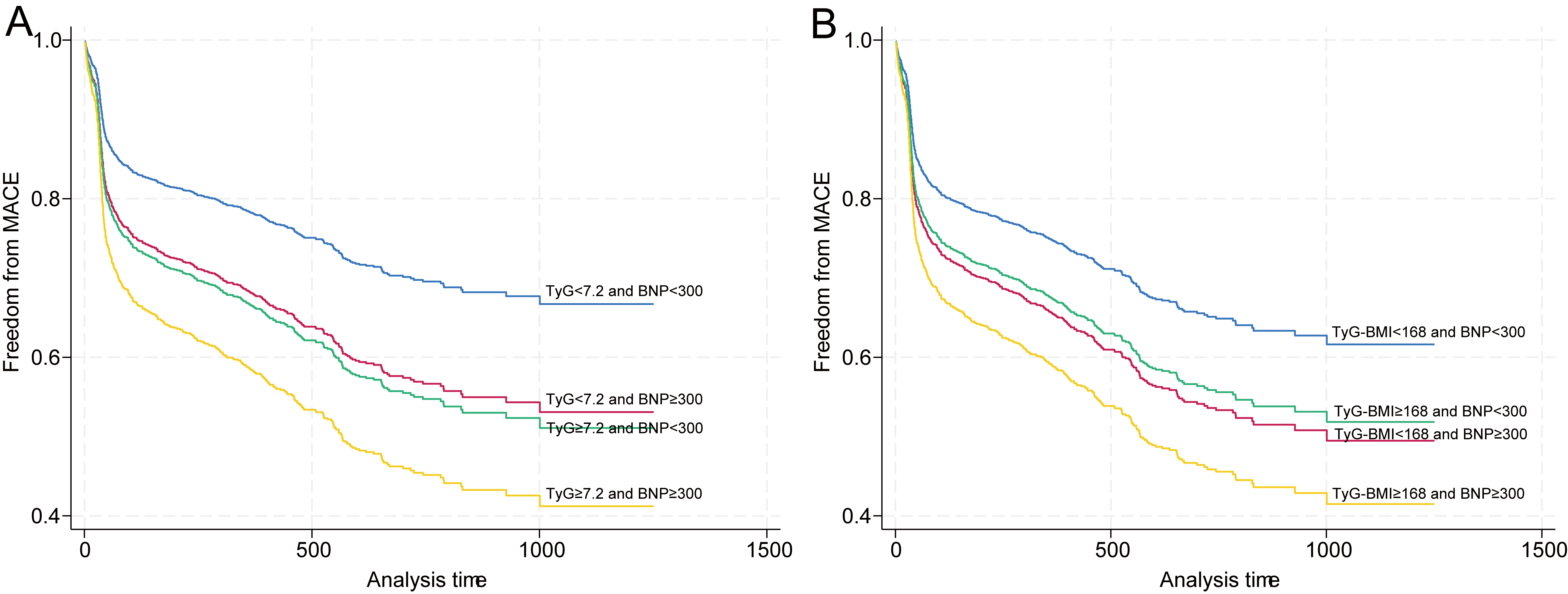 Fig. 2.
Fig. 2.
Synergistic effects of the combined TyG index, TyG–BMI, and BNP stratification on MACE incidence. (A) Kaplan‒Meier curves for four patient subgroups: low TyG index-low BNP (reference), low TyG index-high BNP (HR 1.57, 95% CI: 1.07–2.30), high TyG index-low BNP (HR 1.66, 95% CI: 1.28–2.14), and high TyG index-high BNP (HR 2.18, 95% CI: 1.57–3.03). (B) Kaplan‒Meier curves for four patient subgroups: low TyG–BMI-low BNP (reference), low TyG–BMI-high BNP (HR 1.45, 95% CI: 1.06–1.99), high TyG–BMI-low BNP (HR 1.35, 95% CI: 1.08–1.71), and high TyG–BMI-high BNP (HR 1.81, 95% CI: 1.30–2.51). Shaded areas around the survival curves represent 95% confidence intervals.
According to the fully adjusted model (Model 4), patients with a low TyG index
but elevated BNP levels had a 57% increased risk (HR 1.57, 95% CI: 1.07–2.30),
whereas those with a high TyG index and normal BNP levels had a 66% increased
risk (HR 1.66, 95% CI: 1.28–2.14). Most notably, patients with both an elevated
TyG index and elevated BNP levels presented the highest risk, with a more than
twofold increase (HR 2.18, 95% CI: 1.57–3.03). Bootstrap analysis (Model 5)
validated the robustness of these findings, with consistent hazard ratios and
maintained statistical significance. Additionally, the results of the propensity
score matching analysis (Model 6) revealed a good balance in baseline
characteristics across groups (Supplementary Tables 4,5). The results
remained consistent with those of the multivariable-adjusted models, with the
dual-high group (TyG index
TyG–BMI stratification revealed comparable risk elevation patterns (Table 3 and
Fig. 2B). In the fully adjusted model (Model 4), patients with low TyG–BMI but
elevated BNP levels had a 45% increased risk (HR 1.45, 95% CI: 1.06–1.99),
whereas those with high TyG–BMI and normal BNP levels had a 35% increased risk
(HR 1.35, 95% CI: 1.08–1.71). The dual high biomarker group exhibited an 81%
increased risk (HR 1.81, 95% CI: 1.30–2.51). Bootstrap validation (Model 5)
confirmed the association stability, with significant dose‒response relationships
(p for trend
Consistently, PSM analysis (Model 6) yielded similar results, with the dual-high
group (TyG–BMI
Subgroup analysis (Table 4) revealed significant interactions between metabolic
indices and BNP levels across various patient populations. With respect to the
TyG index (cutoff: 7.2), compared with the reference group (low TyG + BNP level
| Index type | Subgroup | Hazard ratio (95% CI) | p for trend | |||
| Low index + BNP |
Low index + BNP |
High index + BNP |
High index + BNP | |||
| TyG index (cutoff: 7.2) | Age | |||||
| Sample size: | No | Ref. | 1.31 (0.63–2.75) | 1.77 (1.19–2.65) | 1.13 (0.62–2.08) | 0.066 |
| TyG |
Yes | Ref. | 1.69 (1.06–2.72) | 1.51 (1.07–2.13) | 3.09 (2.07–4.60) | |
| TyG |
Female | |||||
| TyG |
No | Ref. | 1.69 (1.09–2.63) | 1.74 (1.31–2.33) | 2.1 (1.41–3.11) | |
| TyG |
Yes | Ref. | 1.61 (0.72–3.60) | 1.59 (0.89–2.84) | 2.9 (1.49–5.65) | 0.004 |
| Hypertension | ||||||
| No | Ref. | 1.49 (0.76–2.92) | 1.34 (0.82–2.19) | 1.5 (0.76–2.98) | 0.227 | |
| Yes | Ref. | 1.5 (0.92–2.42) | 1.74 (1.28–2.35) | 2.34 (1.59–3.44) | ||
| Diabetes | ||||||
| No | Ref. | 1.22 (0.76–1.94) | 1.77 (1.32–2.36) | 2.78 (1.85–4.18) | ||
| Yes | Ref. | 2.79 (1.33–5.86) | 1.29 (0.74–2.28) | 1.68 (0.89–3.18) | 0.343 | |
| SYNTAX Score | ||||||
| No | Ref. | 1.75 (1.00–3.06) | 1.99 (1.36–2.90) | 3.12 (1.90–5.12) | ||
| Yes | Ref. | 1.38 (0.79–2.40) | 1.37 (0.96–1.94) | 1.81 (1.16–2.83) | 0.013 | |
| SGLT2i | ||||||
| No | Ref. | 1.39 (0.90–2.14) | 1.66 (1.25–2.19) | 2.15 (1.47–3.14) | ||
| Yes | Ref. | 2.05 (0.84–5.02) | 0.95 (0.48–1.88) | 1.46 (0.67–3.21) | 0.642 | |
| TyG–BMI (cutoff: 186) | Age | |||||
| Sample size: | No | Ref. | 1.03 (0.54–1.95) | 1.37 (0.96–1.95) | 0.87 (0.47–1.60) | 0.344 |
| TyG–BMI |
Yes | Ref. | 1.64 (1.13–2.39) | 1.25 (0.92–1.71) | 2.98 (2.01–4.41) | |
| TyG–BMI |
Female | |||||
| TyG–BMI |
No | Ref. | 1.63 (1.12–2.36) | 1.45 (1.11–1.89) | 1.64 (1.09–2.48) | 0.004 |
| TyG–BMI |
Yes | Ref. | 1.23 (0.64–2.34) | 1.07 (0.65–1.76) | 2.48 (1.37–4.51) | 0.032 |
| Hypertension | ||||||
| No | Ref. | 1.64 (0.90–2.98) | 1.23 (0.76–1.98) | 1.08 (0.54–2.16) | 0.632 | |
| Yes | Ref. | 1.31 (0.89–1.91) | 1.33 (1.01–1.74) | 1.99 (1.36–2.92) | 0.001 | |
| Diabetes | ||||||
| No | Ref. | 1.33 (0.91–1.96) | 1.46 (1.10–1.93) | 2.23 (1.42–3.49) | ||
| Yes | Ref. | 1.5 (0.85–2.65) | 1.05 (0.69–1.62) | 1.63 (0.95–2.79) | 0.281 | |
| SYNTAX Score | ||||||
| No | Ref. | 1.76 (1.12–2.76) | 1.5 (1.08–2.09) | 1.98 (1.24–3.17) | 0.003 | |
| Yes | Ref. | 1.26 (0.79–1.98) | 1.17 (0.84–1.63) | 1.78 (1.10–2.88) | 0.066 | |
| SGLT2i | ||||||
| No | Ref. | 1.42 (1.00–2.02) | 1.24 (0.96–1.61) | 1.47 (0.99–2.19) | 0.043 | |
| Yes | Ref. | 1.51 (0.71–3.25) | 1.43 (0.79–2.59) | 2.76 (1.36–5.60) | 0.015 | |
Abbreviations: BNP, B-type natriuretic peptide; CI, confidence interval; SGLT2i,
sodium-glucose cotransporter 2 inhibitor; TyG, triglyceride–glucose index;
TyG–BMI, triglyceride–glucose–body mass index. Notes: All analyses were
performed using the fully adjusted model (Model 5 from Table 3).
p-values
Similarly, TyG–BMI (cutoff: 186) showed consistent patterns, with the highest
risk observed in elderly patients with high TyG–BMI and elevated BNP (HR 2.98;
95% CI: 2.01–4.41; p
These findings indicate that the prognostic value of the TyG and TyG–BMI indices is significantly enhanced when these indices are combined with BNP levels, particularly in high-risk populations, including elderly, female, hypertensive, and complex coronary disease patients.
To provide deeper clinical insight, we evaluated the hazard ratios for individual MACE components within these subgroups (Tables 5,6). Patients with elevated levels of both metabolic markers and BNP had significantly greater risks of adverse outcomes, including all-cause mortality, nonfatal myocardial infarction, cerebrovascular events, heart failure hospitalization, and ischemia-induced revascularization, further emphasizing the prognostic utility of combining TyG indices with BNP in high-risk patients.
| Outcome | TyG index |
TyG index | ||
| BNP |
BNP |
BNP |
BNP | |
| All-cause mortality | Ref. | 0.99 (0.19–5.28) | 1.16 (0.36–3.75) | 4.32 (1.30–14.4) |
| Nonfatal myocardial infarction | Ref. | 4.56 (1.34–15.5)* | 2.26 (0.95–5.37) | 3.03 (0.94–9.72) |
| Cerebrovascular event | Ref. | 1.20 (0.24–5.99) | 1.69 (0.72–3.99) | 3.74 (1.12–12.5)* |
| Heart failure hospitalization | Ref. | 0.74 (0.24–2.30) | 2.04 (1.00–4.17)* | 2.29 (0.99–5.26) |
| Ischemia-induced revascularization | Ref. | 1.69 (1.03–2.78)* | 1.53 (1.11–2.11)** | 1.52 (0.95–2.44) |
Abbreviations: BNP, B-type natriuretic peptide; CI, confidence interval; TyG,
triglyceride–glucose index. *p
| Outcome | TyG–BMI |
TyG–BMI | ||
| BNP |
BNP |
BNP |
BNP | |
| All-cause mortality | Ref. | 2.39 (0.81–7.06) | 0.79 (0.28–2.17) | 2.74 (0.91–8.21) |
| Nonfatal myocardial infarction | Ref. | 2.85 (1.06–7.62)* | 1.13 (0.56–2.27) | 1.32 (0.41–4.29) |
| Cerebrovascular event | Ref. | 0.79 (0.17–3.75) | 1.57 (0.71–3.47) | 5.02 (1.63–15.5)** |
| Heart failure hospitalization | Ref. | 1.23 (0.55–2.74) | 2.69 (1.37–5.27)** | 2.64 (1.14–6.07)* |
| Ischemia-induced revascularization | Ref. | 1.27 (0.81–1.99) | 1.32 (0.98–1.77) | 1.50 (0.95–2.37) |
Abbreviations: BNP, B-type natriuretic peptide; CI, confidence interval;
TyG–BMI, triglyceride–glucose–body mass index. *p
To evaluate the incremental predictive value of biomarker integration, receiver
operating characteristic analysis was performed to compare the discriminatory
performance of the combined models (TyG index + BNP and TyG–BMI + BNP) against
that of the individual biomarkers and the established GRACE score. The integrated
TyG index + BNP model demonstrated superior predictive accuracy, with an AUC of
0.67 (95% CI: 0.64–0.70; p
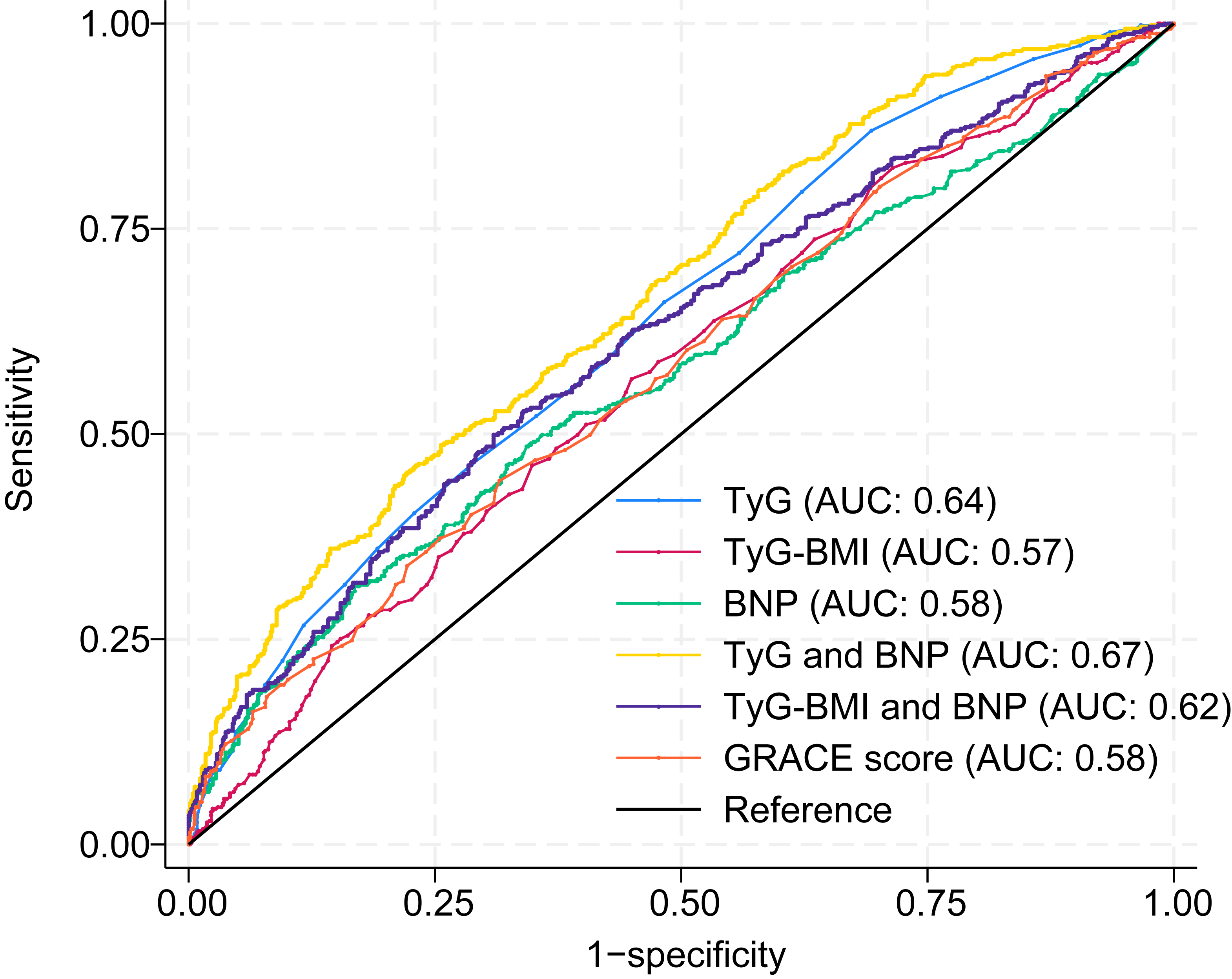 Fig. 3.
Fig. 3.
Discriminatory capacity of individual and combined biomarker
models for MACE prediction. Receiver operating characteristic curves comparing
the prognostic performance of the TyG index alone (AUC: 0.64, 95% CI:
0.60–0.67; p
In addition, sensitivity and specificity analyses were performed to further evaluate the discriminatory performance of each indicator (Table 7). The results revealed that the TyG index, TyG–BMI, BNP level, TyG + BNP level, TyG–BMI + BNP level, and GRACE score had sensitivities of 66%, 57%, 53%, 58%, 53%, and 47%, respectively, and specificities of 52%, 55%, 61%, 64%, 66%, and 65%, respectively. These findings further support the enhanced predictive value of combined biomarker models beyond conventional risk scores.
| Model | AUC | Sensitivity, % | Specificity, % |
| TyG index | 0.64, 95% CI: 0.60–0.67 | 66 | 52 |
| TyG–BMI | 0.57, 95% CI: 0.54–0.60 | 57 | 55 |
| BNP | 0.58, 95% CI: 0.54–0.61 | 53 | 61 |
| TyG index and BNP | 0.67, 95% CI: 0.64–0.70 | 58 | 64 |
| TyG–BMI and BNP | 0.62, 95% CI: 0.59–0.65 | 53 | 66 |
| GRACE score | 0.58, 95% CI: 0.56–0.62 | 47 | 65 |
Abbreviations: BNP, B-type natriuretic peptide; CI, confidence interval; TyG, triglyceride–glucose index; TyG–BMI, triglyceride–glucose–body mass index.
The predictive efficacy of the TyG index and TyG–BMI combined with BNP was
further evaluated across different subgroups. The results demonstrated enhanced
predictive performance in specific populations. In elderly patients (age
| Subgroup | AUC (95% CI) | ||
| TyG index and BNP | TyG–BMI and BNP | ||
| Age |
|||
| No | 0.66 (0.62, 0.71) | 0.59 (0.54, 0.64) | |
| Yes | 0.68 (0.64, 0.72) | 0.65 (0.61, 0.70) | |
| Female | |||
| No | 0.67 (0.63, 0.70) | 0.61 (0.57, 0.65) | |
| Yes | 0.67 (0.61, 0.74) | 0.65 (0.58, 0.71) | |
| Hypertension | |||
| No | 0.64 (0.58, 0.70) | 0.63 (0.57, 0.69) | |
| Yes | 0.68 (0.64, 0.72) | 0.61 (0.57, 0.65) | |
| Diabetes | |||
| No | 0.67 (0.63, 0.70) | 0.62 (0.58, 0.66) | |
| Yes | 0.64 (0.58, 0.70) | 0.56 (0.50, 0.62) | |
| SYNTAX Score |
|||
| No | 0.69 (0.65, 0.73) | 0.62 (0.58, 0.67) | |
| Yes | 0.63 (0.58, 0.68) | 0.60 (0.55, 0.66) | |
| SGLT2i | |||
| No | 0.68 (0.65, 0.71) | 0.63 (0.59, 0.66) | |
| Yes | 0.58 (0.51, 0.66) | 0.54 (0.46, 0.61) | |
Abbreviations: BNP, B-type natriuretic peptide; CI, confidence interval; TyG, triglyceride–glucose index; TyG–BMI, triglyceride–glucose–body mass index; SGLT2i, sodium-glucose cotransporter 2 inhibitor.
Our comprehensive retrospective cohort study of 1177 STEMI patients provides compelling evidence that the combination of metabolic indices (TyG index or TyG–BMI index) with BNP significantly enhances prognostic stratification for MACE prediction. The key findings demonstrate that patients with elevated levels of both metabolic markers and BNP exhibit substantially increased cardiovascular risk, with hazard ratios exceeding 2.0 for the highest-risk combinations. These results suggest that compared with conventional assessment methods, integrated biomarker approaches may offer superior risk discrimination.
The strong association between an elevated TyG index and adverse cardiovascular outcomes observed in our study aligns with growing evidence indicating that insulin resistance is a critical determinant of postinfarction prognosis. Insulin resistance promotes endothelial dysfunction, accelerated atherosclerosis, and prothrombotic states through multiple mechanisms, including increased oxidative stress, inflammatory cytokine activation, and altered lipid metabolism [20, 21]. The TyG index, as a simple and readily available surrogate marker of insulin resistance, offers practical advantages over more complex assessments, such as the hyperinsulinemic–euglycemic clamp technique [22]. Similarly, a previous clinical study first reported that higher TyG index values were significantly associated with increased MACE risk in STEMI patients, suggesting its potential as a valid predictor of outcomes after PCI [23]. These findings underscore the prognostic value of the TyG index and its utility in risk stratification following STEMI.
Our findings regarding the TyG–BMI index provide additional insights into the role of adiposity-related metabolic dysfunction in STEMI outcomes. Incorporating BMI into metabolic risk assessments may capture additional pathophysiological dimensions, including adipose tissue dysfunction, systemic inflammation, and altered adipokine profiles, that contribute to cardiovascular risk [24, 25]. The observed linear relationship between the TyG–BMI index and MACE risk, in contrast to the threshold effect seen for the TyG index, suggests potentially different underlying mechanisms and may inform clinical decision-making regarding risk stratification. These findings are consistent with those of Liu et al. [9], who likewise demonstrated that higher TyG–BMI values predict adverse outcomes in STEMI patients after PCI, reinforcing its prognostic relevance in the acute infarction setting.
In addition, by comparing integrated biomarker models, we observed that combining the TyG index with BNP level achieved better discriminatory performance than either the TyG–BMI or the conventional GRACE score did, underscoring the value of joint metabolic–cardiac biomarker assessment. Clinically, these results suggest that while the TyG–BMI can identify patients at elevated risk, greater predictive accuracy may be achieved through biomarker integration, which could assist in refining risk stratification and guiding secondary prevention strategies in real-world STEMI management.
The prognostic significance of elevated BNP levels in our STEMI cohort
corroborates the extensive literature demonstrating the utility of natriuretic
peptides for cardiovascular risk assessment. BNP elevation reflects multiple
pathophysiological processes, including ventricular dysfunction, increased wall
stress, and neurohormonal activation, all of which contribute to adverse
cardiovascular outcomes [26, 27]. The threshold effect observed at a BNP level
The most significant contribution of our study lies in demonstrating the synergistic prognostic value of combining metabolic and cardiac stress markers. Patients with both an elevated TyG index and elevated BNP levels had a more than twofold increased MACE risk, substantially exceeding the risk associated with either biomarker alone. These findings suggest that metabolic dysfunction and cardiac stress represent complementary pathophysiological domains that, when present simultaneously, confer particularly high cardiovascular risk.
The biological rationale for this synergistic effect may involve several interconnected mechanisms. Insulin resistance can exacerbate cardiac dysfunction through impaired myocardial glucose utilization, increased oxidative stress, and the promotion of myocardial fibrosis [30, 31]. Conversely, cardiac dysfunction may worsen insulin resistance through altered tissue perfusion, neurohormonal activation, and systemic inflammation [21, 32]. This bidirectional relationship may create a pathophysiological cycle that amplifies cardiovascular risk when both conditions coexist.
The superior discriminatory capacity of the combined biomarker models compared with the established GRACE score (AUC: 0.67 vs. 0.58) has important clinical implications. The GRACE score, while widely validated and recommended by guidelines, primarily incorporates demographic and clinical variables available at presentation [3]. Our findings suggest that the addition of readily available biomarkers may increase the accuracy of risk prediction, potentially enabling more personalized therapeutic approaches.
The subgroup analyses revealed particularly pronounced associations in elderly patients, women, and hypertensive patients, suggesting that certain populations may derive greater benefit from combined biomarker assessment. These findings may inform targeted risk stratification strategies and help identify patients who would benefit from intensive monitoring and aggressive therapeutic interventions.
The identification of high-risk patients through combined biomarker assessment may guide therapeutic decision-making in several domains. Patients with elevated metabolic indices may benefit from intensive glucose management, lipid-lowering therapy, and lifestyle interventions targeting insulin resistance [20, 33]. Those with elevated BNP levels may require closer monitoring for heart failure development and earlier initiation of guideline-directed medical therapy for left ventricular dysfunction [27].
Furthermore, emerging therapeutic approaches targeting metabolic dysfunction, such as SGLT2 inhibitors and Glucagon-Like Peptide-1 (GLP-1) receptor agonists, have demonstrated cardiovascular benefits in patients with and without diabetes [34, 35]. The identification of high-risk patients through metabolic biomarker assessment may help guide the selection of patients most likely to benefit from these novel therapeutic interventions.
Several limitations of our study warrant consideration. First, the retrospective design may introduce selection bias and limit the ability to establish causal relationships. Second, the single-center nature of our study may limit its generalizability to other populations and health care systems. Third, biomarker measurements were obtained at a single time point, and temporal changes in biomarker levels during follow-up were not assessed. Fourth, while we adjusted for multiple confounding variables, residual confounding from unmeasured factors cannot be completely excluded.
Additionally, the follow-up period, while adequate for detecting MACE occurrence, may not capture long-term cardiovascular outcomes. The composite nature of our primary endpoint, while clinically relevant, may obscure differences in individual outcome components. Finally, the cutoff values used for biomarker stratification were derived from our study population and may require validation in independent cohorts.
Our findings provide a foundation for several important research directions. Prospective validation studies in independent STEMI populations are needed to confirm the generalizability of our results. An investigation of optimal biomarker cutoff values across different populations and clinical settings may enhance the clinical utility of combined biomarker assessment.
The development of integrated risk prediction models incorporating multiple biomarker domains, clinical variables, and emerging risk factors represents an important area for future investigations. Additionally, studies examining the cost-effectiveness of biomarker-guided risk stratification strategies will be essential for informing clinical practice guidelines and health care policy decisions.
Finally, interventional studies examining whether biomarker-guided therapeutic approaches improve clinical outcomes compared with standard care would provide definitive evidence for the clinical utility of combined biomarker assessment in STEMI management.
Our study demonstrated that combining the TyG index or TyG–BMI with BNP level significantly enhanced MACE prediction in STEMI patients, with patients with both elevated metabolic markers and elevated BNP showing a more than twofold increase in cardiovascular risk. Compared with individual biomarkers and established risk scores, the integrated biomarker approach provides superior discriminatory capacity, supporting its potential utility for personalized risk stratification and therapeutic decision-making in clinical practice. Prospective validation studies are warranted to confirm these findings and establish the clinical utility of this combined biomarker strategy.
MACE, Major adverse cardiovascular event; STEMI, ST-elevation myocardial infarction; BMI, Body mass index; BNP, B-type natriuretic peptide; DBP, Diastolic blood pressure; GRACE, Global Registry of Acute Coronary Events; LDL-C, Low-density lipoprotein cholesterol; LVEF, Left ventricular ejection fraction; SBP, Systolic blood pressure; eGFR, Estimated glomerular filtration rate; TNT, Troponin T; PCI, Percutaneous coronary intervention; rSS, Residual SYNTAX score; SYNTAX score, SYNergy between PCI with TAXUS and the Cardiac Surgery score; ACEI, Angiotensin-converting enzyme inhibitor; ARB, Angiotensin receptor blocker; ARNI, Angiotensin receptor and neprilysin inhibitor; PCSK9i, Proprotein convertase subtilisin/kexin type 9 inhibitor; SGLT2i, Sodium-glucose cotransporter 2 inhibitor; ROC, Receiver operating characteristic; IQR, Interquartile range; HR, Hazard ratio; CI, Confidence interval; RCSs, Restricted cubic splines.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request, subject to appropriate ethical approvals and data sharing agreements.
SX and QY: Conceptualization, methodology, funding acquisition, project supervision. JH: Methodology, data curation, visualization, statistical analysis, writing—original draft preparation. JZ: Data curation, formal analysis, visualization, statistical validation. LL: Formal analysis, statistical validation, data interpretation. MC, YL, XY, SD, QW, and JC: Patient enrollment, data collection, clinical assessment, and follow-up coordination. JH and JZ contributed equally as first authors. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study protocol was approved by the Ethics Committee of Tianjin Medical University General Hospital (approval number: IRB2023-YX-301-01/2023) and adhered to the principles outlined in the Declaration of Helsinki. Due to the retrospective nature of the study, the requirement for informed consent was waived.
We thank the medical and nursing staff of the Cardiovascular Department at Tianjin Medical University General Hospital for their assistance in data collection and patient care. We also acknowledge the patients who participated in this study.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM44062.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



