1 Department of Cardiology, Jiaxing University Master Degree Cultivation Base, Zhejiang Chinese Medical University, 310053 Hangzhou, Zhejiang, China
2 Department of Cardiology, The Affiliated Hospital of Jiaxing University, 314001 Jiaxing, Zhejiang, China
†These authors contributed equally.
Abstract
Emerging evidence has implicated the gut microbiota in the pathogenesis and progression of numerous cardiovascular diseases. Atherosclerosis is a major pathological process that leads to many severe cardiovascular complications. Meanwhile, atherosclerosis patients may experience local and systemic inflammatory responses, with structural changes in the intestinal microbiota and increased mucosal permeability. Currently, the role of gut microbiota-derived metabolites in atherosclerosis pathology is of great concern. Relevant findings have highlighted the potential direct or indirect impacts of gut microbiota on the metabolic health of the host via the production of various metabolites. Thus, this review places an emphasis on bile acids (BAs), metabolites derived from and regulated by the gut microbiota. BAs can delay the pathological processes associated with atherosclerosis, underscoring the significance of these metabolites as an early marker for disease progression risk. In addition, we explore the potential of BA-related gut metabolites as novel therapeutic targets for atherosclerosis, and propose several promising directions for future research.
Keywords
- gut microbiota
- bile acids
- atherosclerosis
Atherosclerosis is characterized by lipid accumulation and chronic inflammation
involving medium- and large-sized arteries, accompanied by the proliferation of
smooth muscle cells and fibromyosin [1]. It is a significant contributor to
coronary atherosclerotic heart disease (CHD) and stroke. Currently, the
prevalence of coronary heart disease, stroke, and other conditions has decreased,
owing to enormous research and various therapeutic options. However,
atherosclerosis remains a threat to human health. Pathologically, the debris of
inflammatory, smooth muscle, and massive necrotic cells, as well as lipids, may
accumulate and transform under the intima over a long period, eventually forming
plaques. Simultaneously, angina pectoris may occur when the luminal blood flow is
obstructed by
Documented evidence has emphasized the potential roles of certain intestinal microbiota and their metabolites in atherosclerosis development. Specifically, the dietary composition may disturb the intestinal microbiota structure, and the metabolites produced by certain bacterial strains may promote the progression of atherosclerosis [3]. Moreover, the intestinal microbiota may impact the host’s inflammatory response, altering the blood pressure and simultaneously accelerating atherosclerosis progression.
Among the numerous intestinal microbiota-derived metabolites, bile acids (BAs) emerge as particularly salient factors influencing atherosclerosis, and interventions targeting the BA receptor might delay plaque formation [4]. Hence, this review was designed to provide a comprehensive overview of the current state of research on the association between BAs and atherosclerosis, along with a critical evaluation of their potential for translation into novel therapeutic targets.
Acting as a major risk factor for numerous Cardiovascular Diseases (CVDs), atherosclerosis principally entails cholesterol accumulation and the recruitment of macrophages within the arterial wall, forming plaques [5]. Conversely, an inflammatory response constitutes the predominant mechanism in this context. Mounting evidence has confirmed that intestinal microbiota can modulate the inflammatory response and metabolite production by microbes, exerting a pathogenic role in atherosclerosis [6].
With an extremely complex microbial profile, the gastrointestinal tract contains an estimated 1014 microorganisms vital for human health. These may participate in organic matter digestion, production of various metabolites, modification of mucous membranes, pathogen defense, etc. Dysbiosis of intestinal microbiota represents a potential risk factor for various diseases, including atherosclerosis. Furthermore, atherosclerosis is associated with structural alterations in the intestinal microbiota. In a controlled investigation, an increase in the abundance of Enterobacteriaceae members and Streptococcus spp. was identified in the intestinal microbiome of atherosclerosis patients, accompanied by metabolic alterations that might promote disease progression [7]. A compositional assessment of atherosclerosis plaques indicated the presence of Actinobacteria spp. (including Collinsella spp.), Chryseomonas spp. and Helicobacter spp., as well as an increased abundance of the fungal genus Aspergillus, in individuals diagnosed with atherosclerosis compared to healthy individuals [8]. In summary, atherosclerosis advancement may be expedited owing to mutually reinforced alterations in the microbiota composition, induced by atherosclerosis and dysbiosis.
Atherosclerosis is recognized as a pathological process entailing chronic inflammation [9], constituting a fundamental component, and pervading most stages of atherosclerosis progression [10]. It serves as a common basis for atherosclerosis physiology and pathology, which primarily manifests as a substantial surge in the plasma levels of various pro-inflammatory cytokines. Recent evidence posits that imbalances in intestinal microbiota may heighten mucosal permeability, accelerating inflammation [11].
Patients with unstable plaques might have an altered microbiota structure
compared to those with stable plaques. Unstable plaques were also linked to
reduced fecal levels of Roseburia and an augmented theoretical capacity
of the microbiota to produce the pro-inflammatory peptidoglycan, along with a
diminished synthesis of anti-inflammatory carotenoids [12]. Such alterations
would facilitate the recruitment of inflammation-inducing factors by the lesion
and accelerate plaque transformation, as demonstrated by numerous studies. The
characterization of inflammatory vesicle complexes integrated immune receptor
responses from a range of stimuli, including cytokines, infections,
macromolecules, and reactive oxygen species [13]. These stimuli might enhance
endotoxemia, in turn exacerbating inflammatory responses and oxidative stress
within the vascular endothelium. Concurrently, the diminution of short-chain
fatty acid-producing bacteria, such as Roseburia, might impede
intestinal barrier function and augment wall permeability, thereby boosting the
translocation of microbiota-derived metabolites, including lipopolysaccharides
(LPS), into the bloodstream [14]. The levels of inflammatory markers
(Ultrasensitive C-reactive protein, Interleukin [IL]-6, and IL-1
In addition, the gut microbiota–immune system interaction is considered a critical component of the chronic inflammatory response observed in atherosclerosis [16]. Microbial metabolites can influence the differentiation and polarization of immune cells, such as a balance between the T helper cell subsets and the activation of macrophages, thereby shaping the inflammation-associated microenvironment of the vascular wall. Altered immune responses induced by gut microbial dysbiosis may thus represent a vital mechanistic link between the intestinal ecosystem and cardiovascular pathology [17].
Nonetheless, contemporary endeavors to treat atherosclerosis by suppressing the inflammatory response remain in the experimental phase, precluding the determination of a specific group of patients benefiting from anti-inflammatory therapy [18]. Consequently, the focus on the role of microbial metabolites in atherosclerosis has escalated.
A considerable body of empirical evidence corroborates the notion that gut microbiota exert systemic effects on the host as they enable the production of bioactive metabolites, such as short-chain fatty acids (SCFAs), BAs, and Trimethylamine-N-oxide (TMAO), which are particularly pronounced in the circulatory system [19]. In general, a majority of the bacterial metabolites typical of the human body promote well-being. However, disrupted gut microbiota can augment the precipitation of noxious metabolites, a phenomenon that may be pertinent to the pathological progression of atherosclerosis. Beyond traditional lipid metabolism, emerging microbe-derived metabolites have garnered increasing attention for their role in the pathogenesis and progression of atherosclerosis. Indole derivatives, such as indole-3-propionic acid (IPA) and indole-3-acetic acid (IAA), exert protective effects by activating the aryl hydrocarbon receptor and its related signaling pathways to modulate inflammation and endothelial function [20]. SCFAs, including acetate, propionate, and butyrate, act through GPR41/43 receptors and histone deacetylase (HDAC) inhibition to influence macrophage polarization, Treg differentiation, and vascular inflammatory responses, thereby enhancing plaque stability and maintaining metabolic homeostasis [21]. In contrast, recently, the microbe-derived metabolite imidazole propionate (ImP) was reported to be markedly elevated during the active stages of atherosclerosis, promoting inflammation and plaque progression via the imidazoline receptor (I1R), and is considered potentially pathogenic [22]. Moreover, BAs modulate cholesterol metabolism, inflammation-associated signaling, and endothelial function, thereby influencing the course of atherosclerosis and demonstrating a close association with disease prognosis.
BAs are synthesized in the liver through a series of cytochrome P450 (CYP)-catalyzed cholesterol oxidation reactions [23]. The primary BA may further combine with varying ratios of taurine or glycine [24] to form the conjugated BA (CBA). The CBA subsequently exits the hepatocytes into the gall bladder following meals, along with the flow of bile into the intestines, where it fulfills a predominant role in facilitating the absorption of dietary lipids and vitamins by the small intestines [25]. The recycling mechanism of the human body is a sophisticated process, termed “enterohepatic recycling”. It involves the reabsorption of a majority of the plasma protein-bound BAs through a multifaceted transport mechanism. Specifically, it occurs due to active transport within the distal ileum along with passive absorption throughout the intestine. The reabsorbed BAs are then recirculated to the liver through the portal vein. Such an enterohepatic cycle occurs 4–12 times daily [26]. The colon may absorb ~5%–10% of the BAs, where they are either biotransformed by the gut microbiota or excreted [25]. During this process, the gut microbiota principally modulates the BA pool by mediating a series of enzyme-catalyzed reactions. These include a portal reaction facilitated by the hydrolysis of the C-24 N-acyl bonds by which BAs are bound to taurine or glycine, catalyzed by bile salt hydrolase (BSH) in the lower small intestine and proximal colon to yield the secondary BAs (SBAs) [27], thereby generating free BAs in the liver. Gram-positive bacteria (e.g., Clostridium, Enterococcus, Bifidobacterium, and Lactobacillus), which are among the most abundant intestinal microbiota, exhibit the greatest diversity regarding BSH distribution among numerous other species. Recently, BSH expression was noticed to be exclusive to the Anaplasma phylum among the Gram-negative bacteria [28]. Prior studies have substantiated the involvement of BAs in diverse biological processes, such as modulating lipid metabolism, regulating glucose metabolism, facilitating antimicrobial effects, and suppressing cholesterol synthesis [29].
Animal foods, particularly those comprising offal, eggs, select seafoods, and
dairy products, contain notable cholesterol levels [30]. The conversion of
cholesterol to BAs in the liver involves 17 enzymes, situated within the
lysosomes, endoplasmic reticulum, mitochondria, and peroxisomes. Noticeably,
cholesterol 7
Atherosclerosis-triggered compositional alterations in fecal microbiota can modulate the circulating BA levels [32]. Human intestine-resident strains, including Pasteurella smithii, Clostridium difficile, and Enterococcus faecium, capable of synthesizing BSH have been identified. They can induce cholesterol accumulation within the macrophages (foam cell formation), promote the production of oxidized low-density lipoproteins by modulating BSH activity, and accelerate disease progression by augmenting the size and structural instability of atherosclerotic plaques. Similarly, atherosclerosis patients have exhibited an expanded proportion of certain bacterial strains within the gastrointestinal microbiota [33]. As a result, the alterations in gut microbiota may regulate BA levels by modulating BSH activity (Fig. 1).
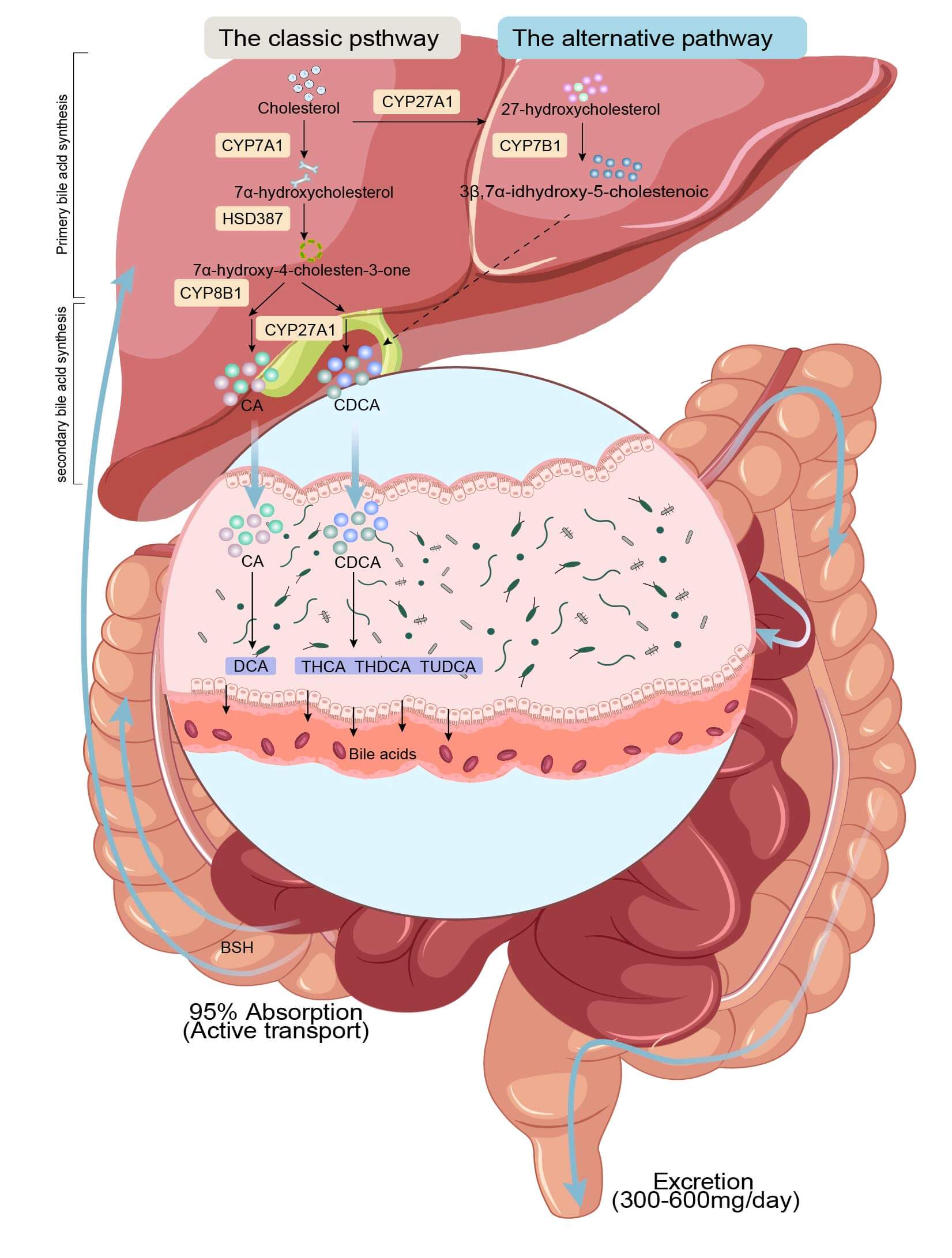 Fig. 1.
Fig. 1.
BA metabolism and enterohepatic circulation. In the liver, cholesterol undergoes a series of cytochrome P450 (CYP)-catalyzed reactions to form BAs, which may further facilitate the formation of CBA after integrating with taurine or glycine. CBA enters the intestines following the bile, where they form SBA. Most of the SBA dissociate under the catalytic action of BSH and return to the liver for enterohepatic recycling. A small fraction of SBA is excreted in the colon with feces. CDCA, Chenodeoxycholic acid; THCA, Trihydroxycholestane acid; THDCA, Taurohyodeoxycholic acid; TUDCA, Tauroursodeoxycholic acid; CA, Cholic acid; DCA, deoxycholic acid; BSH, bile salt hydrolase.
Certain gut microbiota-derived metabolites, such as TMAO and ImP, influence BA levels. Evidence suggests that TMAO can suppress BA biosynthesis, thereby accelerating the development and progression of atherosclerosis, and is considered a critical mechanism underlying TMAO-induced atherogenesis [34]. ImP, a novel microbial metabolite, has recently been implicated in altering the BA pool composition and has been closely associated with diseases such as diabetes, atherosclerosis, and gastrointestinal inflammation [35].
It is imperative to acknowledge the significance of the genetic factors of intestinal microbiota in producing metabolites. The hypothesis that host genetics contributes substantially to the structure of the intestinal microbiota has been advanced under the premise that such a structural element can regulate immune-related pathways and metabolism-related phenotypes [36]. A strong association was identified between the ABO blood group system-associated genes and FUT2 polymorphisms with several intestinal microbiota, principally the genera Collinsella, Bifidobacterium, and Enterococcus faecalis [37]. Meanwhile, a genome-wide analysis of the intestinal microbiota from 1812 subjects revealed that genetic factors accounted for ~10% of the variations in the microbiota [38]. Consequently, genetics is essential in BA production, given its indispensable ecological role in altering the structural composition of intestinal microbiota. Thus, it was hypothesized that genetics is also essential for BA production.
Several experiments have demonstrated that BAs can impede the progression of atherosclerosis in either a direct or indirect pattern (Table 1, Ref. [39, 40, 41, 42, 43, 44, 45]).
| Authors (year) | Models/cells | Main findings | Ref. |
| Zhou et al. (2024) | Mice | BAs can modulate platelet function by regulating the Syk, Akt, Erk1/2, and syntaxin-11 pathways, which are associated with NCK1. | [39] |
| Qi et al. (2025) | Mice | DCA functions as an inhibitor of platelet activation due to its interaction with TGR5. | [40] |
| Guo et al. (2016) | Mouse bone marrow-derived macrophages and mice | BAs can suppress the formation of NLRP3 inflammatory vesicles by acting through the TGR5-cAMP-PKA signaling axis. | [41] |
| Gilles Lambert et al. (2003) | Mice | FXR is the primary regulator of normal cholesterol metabolism, and genetic alterations affecting FXR function have the potential to promote atherosclerosis. | [42] |
| Yoyo T Y Li et al. (2007) | RASMC | FXR exerts a regulatory effect on VSMC inflammation by curtailing the migration of these cells. | [43] |
| Li et al. (2008) | Rats | FXR enhances eNOS expression. | [44] |
| He et al. (2006) | Rats | FXR serves to diminish the manifestation of the vasoconstrictor endothelin (ET)-1. | [45] |
FXR, Farnesoid-X-Receptor; TGR5, transmembrane G protein-coupled receptor-5; VSMC, vascular smooth muscle cell; eNOS, endothelial nitric oxide synthase.
Serum total BA (TBA) levels correlated directly with the extent of atherosclerotic lesions, with substantially reduced levels in patients with acute coronary syndrome (ACS), particularly in those with acute MI (AMI), than in the normal range [46]. Various SBAs, including DCA, lithocholic acid (LCA), and chenodeoxycholic acid (CDCA), can inactivate collagen-stimulated platelets determined by kinase analysis and by constructing a mouse atherosclerosis model. Furthermore, LCA and CDCA could suppress collagen, thrombin, U46619, and ADP-stimulated platelet aggregation in humans. DCA and CA impede collagen-stimulated platelet aggregation, while U46619 and ADP elicit such a response. Moreover, in another NCK1-knockout mouse model, BAs could act against platelet activation and aggregation, primarily by inhibiting the phosphorylation of NCK1-related Syk, Akt, ERK1/2, and syntaxin-11 [39], aligning with the documented anti-atherosclerosis effects of BAs. In addition, DCA could arrest platelet activation and thrombosis by interacting with the transmembrane G protein-coupled receptor-5 (TGR5) [40].
Vascular inflammation is recognized as a pivotal pathological mechanism for
atherosclerosis. Meanwhile, the nucleotide-binding, oligomerization, structural
domain-like receptor family containing pyrin structural domain 3 (NLRP3)
inflammatory vesicles assume a central role in the pathogenesis of vascular
inflammation [47]. It is primarily linked to the upregulation of adhesion
molecules facilitated by the enhanced processing and secretion of
pro-inflammatory cytokines (e.g., IL-1
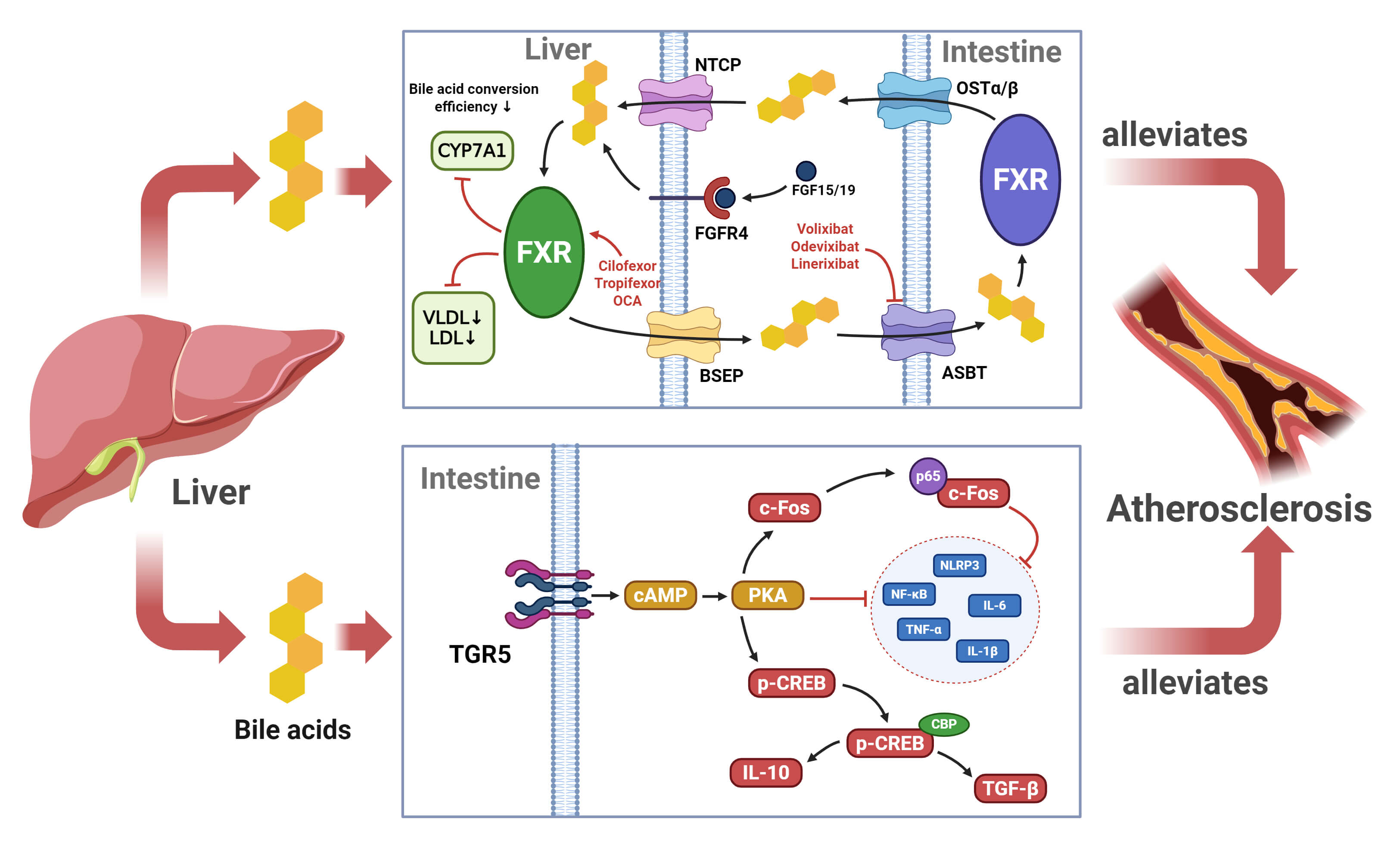 Fig. 2.
Fig. 2.
Mechanisms underlying the inhibitory effect of BA on
atherosclerosis progression. BAs serve as an anti-inflammatory to suppress
atherosclerosis progression by binding to TGR5 and FXR receptors to modulate the
inflammatory response and maintain lipid homeostasis. NTCP, Sodium Taurocholate
Cotransporting Polypeptide; OST
Lipid metabolism abnormalities and dyslipidemia have been recognized as risk
factors for many cardiovascular diseases, generally presenting elevated plasma
triglycerides (TGs) and low-density lipoprotein cholesterol (LDL-C).
Subsequently, a massive deposition of these lipids within blood vessels induces
local inflammation, vascular remodeling, and the formation of atherosclerotic
plaques [10]. Farnesoid-X-Receptor (FXR) is the most intensively studied BA
receptor [49], which is expressed in the pancreas, kidneys, vascular walls, and
various other tissue types, along with enhanced expression in the
gastrointestinal tract [50]. As a central mechanism by which FXR plays a role in
metabolic regulation, besides constituting a key biological pathway for
cholesterol catabolism, the FXR-mediated BA biosynthesis regulatory pathway can
also reveal the dual roles of this nuclear receptor in lipid metabolism
homeostasis through negative feedback. Specifically, it can regulate the
efficiency of cholesterol–BA conversion by inhibiting the rate-limiting enzyme
CYP7A1 [51], and also modulate BA conversion via signaling molecules such as
FGF19/15 to maintain a dynamic homeostasis during systemic lipid metabolism [52]
(Fig. 2). This pathophysiological mechanism serves as a theoretical basis for the
targeted therapy of metabolic syndrome or associated diseases. In a controlled
experiment, FXR knockout (FXR-/-) mice with substantially increased plasma
levels of apolipoprotein B-rich lipoproteins (i.e., non-HDL lipoproteins such as
VLDL and LDL) were detected [42]. These observations demonstrated that FXR can
reduce atherosclerosis-associated risk factors to a certain extent. In addition
to controlling the metabolism-related risk factors for atherosclerosis, FXR is
also expressed in different cell types of multiple blood vessels, including
coronary arteries and aortic vascular smooth muscle cells (VSMCs) [53]. For
instance, rat aortic smooth muscle cells were treated with the synthetic FXR
ligands GW4064 or 6
BAs signify a potentially auspicious risk indicator for cardiovascular events, which can forecast unfavorable outcomes in atherosclerosis patients to a certain extent.
A substantial corpus of evidence confirms the robust anti-atherosclerotic effects of BAs, with CHD representing a particularly adverse event during the advanced stage. The serum and fecal BA levels in 45 CHD patients and 35 normal subjects were determined, revealing significantly lower total BA concentrations in CHD patients than in normal subjects. Thus, the absence of BAs in the serum served as a predictor for CHD [56]. Furthermore, the present study found that the administration of statins markedly elevated the serum BA levels in CHD patients, supporting their therapeutic value. Nevertheless, a study on the relationship between plasma acylcarnitines and BA levels with CHD incidence revealed a positive correlation between DCA and the documented risk of CHD, as suggested by Wu et al. [57]. This observation may be attributed to the premise that DCA, as a secondary BA, is predominantly antagonistic to FXR in the presence of CDCA [57]. This mechanism may explain the divergent associations among various BAs and CHD observed in this investigation.
A more exhaustive examination of TBA has predominantly occurred in the context of MI. A survey encompassing 7438 patients diagnosed with coronary artery disease ascertained the initial correlation between TBA and MI. Patients afflicted with MI exhibited a conspicuously diminished TBA in comparison to age- and sex-matched subjects without MI [58]. An experimental model involving BA administration in a MI mouse specimen exemplified the cardioprotective impacts of BAs. The experiment involved administering FXR-overexpressing, adipose tissue-derived mesenchymal stromal cells into the mouse myocardium through intramyocardial injection. Compared with the control group, the experimental group exhibited a substantial enhancement in the left ventricular ejection fraction, a concurrent reduction in the end-systolic left ventricular internal or diastolic diameters, and MI size. In particular, the experimental group mice displayed a higher survival rate in comparison to the control group [59]. Meanwhile, in a separate analytical study on ACS, a mediation analysis was employed to evaluate the serum TBA levels of patients in isolation, unveiling a negative correlation between the TBA concentration and coronary artery disease severity in patients experiencing AMI. In addition, the TBA concentration was independently associated with all-cause mortality and cardiac-related death risk. Conversely, a decline in the TBA levels might indicate an elevated risk of MI-related mortality [46].
Atherosclerosis progression, accompanied by plaque hardening, may reduce the elasticity of the vessel wall [60]. This phenomenon is exemplified by the abdominal aorta among certain vessels characterized by elevated blood flow. The nonelastic vessel wall may not adequately counteract the wall tension engendered by the dynamic pulsation caused by blood flow, frequently precipitating localized verrucous dilatation, a condition clinically analogous to an aneurysm [61]. Despite their relatively limited prevalence relative to other cardiovascular conditions such as coronary heart disease and MI, aneurysms nevertheless pose a marked threat to atherosclerosis patients who are predisposed to rupture, triggering potentially severe consequences, including hemorrhage and high mortality. For instance, an experimental investigation was conducted by employing a 30-mouse abdominal aortic aneurysm (AAA) animal model to elucidate the functional role of BAs in aneurysms. Consequently, tauroursodeoxycholic acid (TUDCA) was found to curtail AAA formation in mice by impeding several signaling pathways [62]. While BAs harbor the potential to influence the prognosis of aneurysms, their efficacy as a diagnostic indicator for aneurysm development remains inconclusive.
Atherosclerosis of the intracranial large artery represents one of the most prevalent etiologies of ischemic stroke (IS) [63]. The underlying pathophysiological mechanisms encompass two primary pathways: atherosclerotic embolization within hemodynamically constricted vessels, culminating in suboptimal perfusion, thrombosis, and upstream embolism, in conjunction with plaque rupture or ulceration [64]. Building upon this hypothesis, atherosclerosis may serve as a potential pathophysiological IS mechanism, and IS prognosis may be predicted through the monitoring of atherosclerosis progression. In a randomized controlled trial (RCT), Monteiro-Cardoso et al. [65] observed that stroke-affected mice administered with CDCA had a significantly mitigated stroke infarct size, possibly due to the neuroprotective impacts of CDCA. Moreover, analogous observations were made by their research team in another prospective randomized case-control trial comprising 57 adult acute ischemic stroke patients and 54 age- and race-matched healthy individuals [65]. Another prospective epidemiological investigation of 7323 IS patients explored the correlation between serum TBA levels and the clinical outcomes of IS. The data confirmed a negative correlation between TBA levels at admission and post-admission prognosis. This finding substantiates the hypothesis that TBA serves as a prognostic IS biomarker [66].
Although BAs have considerable potential as prognostic indicators of various cardiovascular and cerebrovascular diseases, clinical trials remain relatively limited, with some findings being contradictory. For example, certain BAs, such as DCA, have demonstrated dual effects on atherosclerosis, raising concerns about their reliability over traditional lipid analyses. Moreover, BA levels are highly susceptible to dietary status, genetic background, infections, and dynamic changes in the gut microbiota, resulting in a lower stability than conventional biomarkers. Therefore, in-depth research and large-scale data validation are required to determine the feasibility of BAs as predictive indicators for atherosclerosis and related diseases.
A comprehensive review of the studies conducted on diverse populations, as well as clinical observations, reveals a correlation between gut microbiota dysbiosis and circulating BA levels, with susceptibility to atherosclerosis, highlighting BA-associated intestinal microbiota-derived metabolites as promising therapeutic targets for atherosclerosis (Table 2, Ref. [67, 68, 69, 70, 71, 72, 73, 74]; Fig. 3).
| Treatments | Interventions | Patients/models | Main findings | Ref. |
| Diet | High-fat diet + BA | Mice | The supplementation of a high-fat diet with a certain number of BAs reduces the risk of atherosclerosis. | [67] |
| Mediterranean diet | Metabolic Syndrome | The Mediterranean diet interferes with BA metabolism. | [69] | |
| Mediterranean diet | CHD | Mediterranean diet superior to traditional low-fat diet in the prevention of major adverse cardiovascular events in atherosclerosis patients. | [68] | |
| Probiotics | MVs isolated from Lactobacillus rhamnosus | Mice | Probiotic membrane vesicles (MVs) induce atherosclerotic plaque regression by modulating foamy macrophages. | [70] |
| Limosilactobacillus reuteri | Mice | Lactobacillus Royale alleviates vascular endothelial dysfunction. | [71] | |
| Lactobacillus vaginalis FN3, Bifidobacterium animalis subsp. Lactis F1–7, Enterococcus faecium WEFA23 | Mice | Probiotics inactivate the FXR-FGF15 signaling pathway to elevate intestinal progenitor BA levels. | [72] | |
| Prebiotics | Dietary fiber | Mice | Dietary fiber protects against MI. | [73] |
| FMT | Intestinal microbiota from WT mice | Mice | FMT for restoration of gut microbial homeostasis may be an effective therapeutic strategy in atherosclerosis. | [74] |
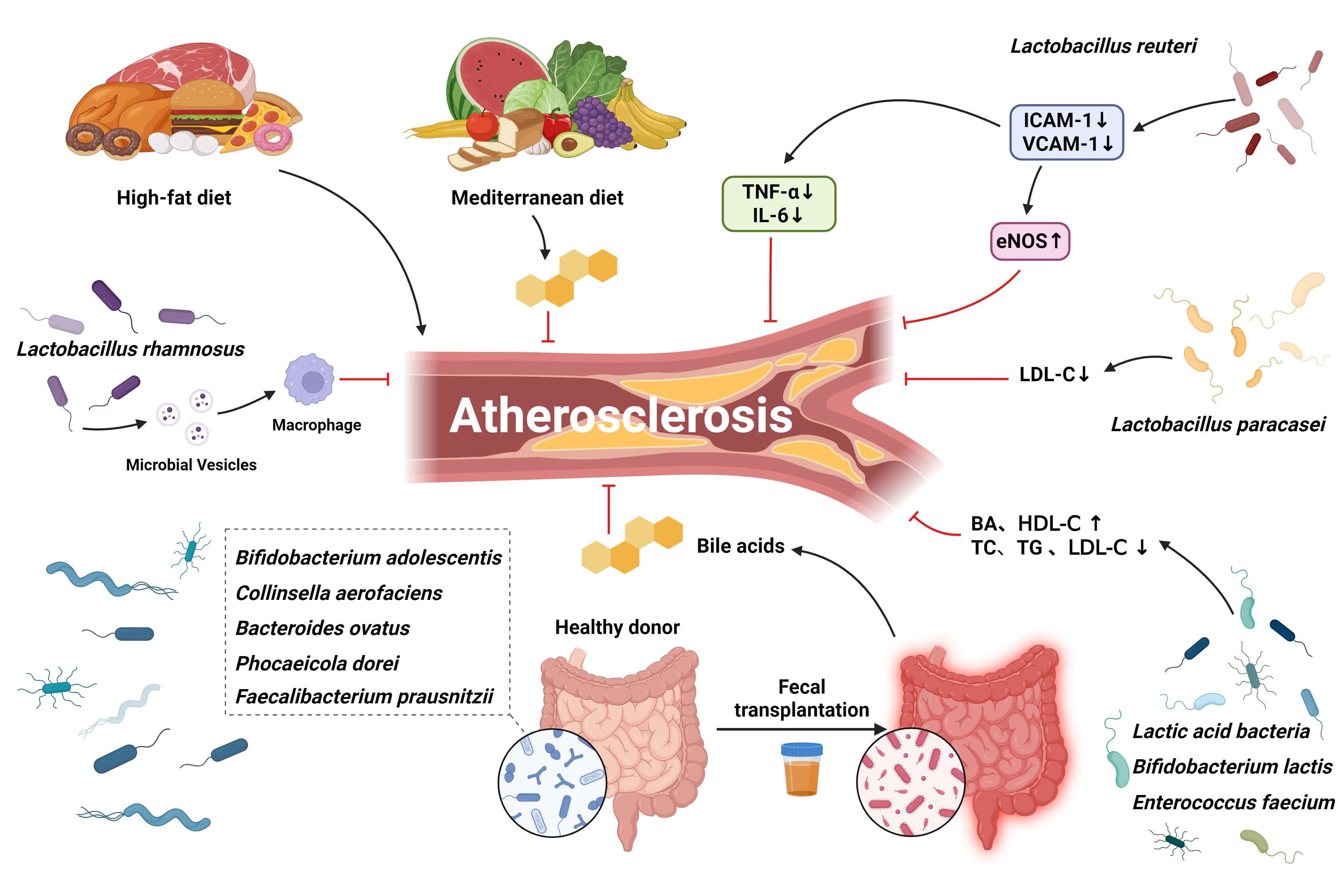 Fig. 3.
Fig. 3.
Potential therapeutic targets for BAs-associated atherosclerosis in the gut. The Mediterranean diet may promote the development of an optimal gut microbiota status and significantly reduce the prevalence of associated comorbidities, including atherosclerosis. Targeting gut microbiota, probiotics, prebiotics, and FMT may enhance Bas levels to some extent. Provides promising avenues for intervention in atherosclerosis. FMT, Fecal Microbiota Transplantation.
Plasma BA levels correlate robustly with dietary habits, underscoring the
pivotal role of nutrition in the etiology of numerous cardiovascular diseases and
the urgency for addressing this critical issue during medical inquiry. From the
synthesis of BAs to their biotransformation in the gastrointestinal tract,
numerous components exist that are prone to diet-based interventions. Given the
great fluctuation in intestinal microbiota composition throughout human life
[75], the application of such interventions precipitates more rapid alterations
in the levels of the associated metabolites [76]. Western diets, which frequently
comprise elevated saturated fatty acid levels, represent a remarkable contributor
to the prevalence of gut microbial dysbiosis, a condition characterized by the
altered composition of intestinal microbiota, and a concomitant suppression of BA
secretion. Several studies have revealed a positive correlation between diet and
the escalated risk of inflammation, particularly in the context of ulcerative
colitis and Crohn’s disease, prompting the recommendation of dietary
modifications as a potential therapeutic approach under such conditions [77].
Conversely, in a murine model on a high-fat diet, the incorporation of a specific
number of BAs impeded weight gain and substantially diminished the prevalence of
associated comorbidities, encompassing atherosclerosis [67]. However, the oral
administration of BAs is exclusive to patients diagnosed with genetic defects
resulting in a deficiency in primary BA synthesis [78]. Consequently, in the
realm of atherosclerosis, it represents a salient contemporary issue regarding
the investigation of the therapeutic potential of enhancing BA levels through
dietary interventions. In similar inquiries on heart failure (HF), the
Mediterranean diet—known for its numerous health benefits—has been endorsed
for HF patients as it can foster an optimal composition of intestinal microbiota
and curtail the prevalence of HF by
However, the specific impact of the Mediterranean diet on BAs and its mechanistic link to anti-atherosclerotic effects remains largely unexplored. Beyond its well-established benefits, specific dietary components such as fibers, polyphenols, and omega-3 fatty acids can modulate BA metabolism by regulating the gut microbiota, thereby influencing the development of atherosclerosis [83]. For example, dietary fibers can be fermented by gut microbes to produce SCFAs, which not only enhance intestinal barrier integrity but also indirectly affect BA synthesis and transformation, subsequently modulating the FXR and TGR5 signaling pathways [84]. Polyphenols, conversely, may alter the composition and proportion of the BA pool by suppressing the growth of pathogenic microbes while promoting the proliferation of beneficial bacteria, ultimately influencing inflammatory responses and cholesterol metabolism [85]. Collectively, these findings suggest that dietary components regulate the “gut microbiota–bile acid–signaling axis”, thereby playing a crucial role in maintaining metabolic homeostasis and reducing the risk of atherosclerosis.
A nutritious diet refers to the most straightforward and economical approach to address and manage atherosclerosis by positively affecting the composition of the gut microbiota. Nevertheless, there is a paucity of research on effective interventions for atherosclerosis via enhancing BA levels through dietary adjustment. Promising avenues for future exploration involve the testing of BAs, cholesterol, LDLs, HDLs, and other markers to tailor dietary regimens to individual patients.
Probiotics comprise live microorganisms that promote gastrointestinal health, which can modify the intestinal microbial composition and preserve homeostasis, eventually enhancing human health. They may facilitate the attenuation of atherosclerotic plaque softening, improvement in lipid parameters, curtailment of inflammatory responses, etc., eventually ameliorating the atherosclerosis-related parameters. For illustration, probiotic membrane vesicles isolated from Lactobacillus rhamnosus could induce atherosclerotic plaque regression by modulating foamy macrophages and exert a vasoprotective effect in mice with atherosclerosis [70]. Similarly, Lactobacillus reuteri, with anti-oxidant and anti-inflammatory properties, substantially mitigated the impacts of cell adhesion molecules (e.g., intercellular adhesion molecule-1 [ICAM-1]) and blood lipid levels [71]. Lactis F1-7 and Enterococcus faecalis WEFA23 regulated CYP7A1 expression in hyperlipidemic mice by inhibiting the FXR–FGF15 pathway, ultimately elevating the intestinal BA and HDL-C levels, while concomitantly suppressing serum TC, TG, and LDL-C levels [72]. Furthermore, substantial investigations involving animal models have demonstrated the capacity of probiotics to postpone the onset of atherosclerosis and safeguard blood vessels through diverse mechanisms. To validate their effectiveness in a clinical context, a 4-week interventional experiment on studying the effects of probiotics in patients with severe atherosclerosis was conducted, with a noteworthy enhancement in the abundance of beneficial gut strains, such as the Lactobacillus plantarum probiotic strain (DSM 9843) group, compared to the placebo group. The probiotic group exhibited a conspicuous augmentation in the diversity of beneficial intestinal strains [86]. Additionally, a prospective RCT in hyperlipidemic patients with comorbid early-onset atherosclerosis demonstrated that Lactobacillus paracasei TISTR 2593 considerably diminished LDL-C levels, contributing to anti-atherosclerosis by regulating the LDL-C levels [87]. Collectively, probiotic-based strategies continue to evince ambitious promise in the treatment of atherosclerosis, despite the dearth of studies on the impacts of probiotics in this group.
Another strategy to modulate gut homeostasis entails prebiotic administration. Prebiotics consist of indigestible carbohydrates that selectively regulate the proliferation, composition, and functionality of the gut microbiota [88], thereby improving host health to a certain extent. A series of experiments discovered that prebiotics enhanced intestinal permeability and alleviated metabolic endotoxemia, while concomitantly diminishing inflammatory responses in an obese mouse model [89]. Furthermore, in another study, MI-induced mice were administered prebiotics composed of dietary fibers, thereby remarkably improving cardiac function, diminishing infarct size, and preventing adverse remodeling post-MI. Subsequent metabolomics revealed that high-fiber diets elevated the BA levels, underscoring the protective role of dietary fibers in MI [73]. Extensive research on the prebiotic inulin has revealed its capacity to stimulate the proliferation of beneficial intestinal microbiota, optimize their diversity, and functionality. Concurrently, it can facilitate the mitigation of probiotics-related adverse effects [90], which, however, show no effect on ameliorating atherosclerosis. In an RCT on mice with hypercholesterolemia, inulin did not demonstrate any remarkable impact on plasma cholesterol levels. Additionally, it was impossible to prevent or delay atherosclerosis progression, despite increasing the proportion of specific bacterial genera and elevating the cecal SCFA levels [91]. Following previous inquiries, another investigation discovered that inulin can trigger a substantial escalation in total plasma cholesterol levels, while concomitantly expediting the progression of atherosclerosis in hypercholesterolemic E3L mice [92]. Cautiously, diverse prebiotics elicited contrasting effects on atherosclerosis, requiring further research to decipher the precise underlying etiologies and mechanisms.
Although some preliminary studies have demonstrated the theoretical anti-atherosclerotic potential of probiotics and prebiotics, clinical evidence and practical applications remain limited, and their effects are still debated. For example, animal experiments have shown that inulin may exert protective effects against atherosclerosis by promoting the production of SCFAs, thereby improving lipid metabolism and the inflammatory status [93]. However, inulin may exacerbate the progression of atherosclerosis in mouse models on a high-fat diet, possibly due to excessive fermentation leading to the accumulation of metabolites such as SBAs or other microbial byproducts, as well as causing microbiota dysbiosis [92]. Thus, the effects of prebiotics may be double-edged, influenced by host metabolic background, gut microbial composition, and dosage. Future research should aim to identify specific prebiotic formulations that provide consistent cardiovascular benefits across populations. For instance, galacto-oligosaccharides and fructo-oligosaccharides in certain clinical studies have shown the potential to improve lipid profiles and inflammatory markers [94]. These findings suggest that interventions targeting atherosclerosis should consider individual selection of prebiotic types and dosages based on host microbial characteristics, to maximize therapeutic benefits while minimizing potential risks.
FMT has recently been established as an available therapeutic option for diseases related to intestinal ecological dysregulation, mainly by transplanting intestinal microorganisms from healthy individuals to patients with certain intestinal disorders. FMT can combat enteric conditions such as Clostridioides difficile infection and inflammatory bowel syndrome [95], as well as several nonintestinal conditions such as neurological conditions [96], autism [97], obesity [98], and diabetes mellitus [99]. FMT was performed on patients with metabolic syndrome, resulting in the increased diversity of fecal microbiota, prominently associated with improved insulin sensitivity [100]. These positive features were confirmed in a subsequent RCT [101], illustrating the therapeutic potential of FMT against metabolic syndrome. Furthermore, in a secondary analysis of this double-blind RCT, obese patients with metabolic disorders were treated with FMT capsules donated by a lean donor. Consequently, after the successful colonization of the recipients with FMT, the evolution of glucose intolerance was delayed, as described previously. Additionally, the increase in BA metabolism induced by gut bacteria, comprising Bifidobacterium adolescentum, Collinsella aerofaciens, Anabaena ovale, Phocaeicola dorei, and Faecalibacterium prausnitzii, was positively associated with intestinal microbiota-derived BAs, which might be a potential therapeutic option for metabolic syndrome [102].
In light of these findings, FMT appears to possess therapeutic potential against atherosclerosis. In a seminal experiment, FMT was performed on atherosclerosis-prone mice, termed C1q/TNF-related protein 9 knockout (CTRP9-KO) mice. Transplantation of fecal matter from wild-type mice diminished atherosclerotic lesions in the carotid arteries of the CTRP9-KO mice, retarding the occurrence of atherosclerosis. Building upon these revelations, subsequent experiments by the same research group sought to promote atherosclerosis progression in wild-type mice through fecal transplantation from CTRP9-KO mice. In successive experiments, it was observed that the lesions promoted atherosclerosis propagation in the wild-type mice. The investigators concluded that the restoration of homeostasis in intestinal microbiota by FMT may serve as an effective strategy for atherosclerosis [74].
Indeed, FMT is efficient for treating atherosclerosis in animal models. Its clinical application still faces significant ethical and practical challenges. First, donor variability is a major limiting factor influencing the efficacy of FMT, as differences in the composition and functions of donor-derived microbiota may lead to substantial heterogeneity in clinical outcomes among the recipients [103]. Second, FMT carries potential safety concerns, including the transmission of pathogens, the transfer of antimicrobial resistance-associated genes, and the risk of adverse immune reactions. The U.S. Food and Drug Administration reported a case of death due to severe bacterial infection post-FMT, and issued a safety warning in 2019 [104]. In addition, FMT lacks long-term follow-up clinical evidence, and its efficacy and safety in cardiovascular diseases have not yet been systematically validated [105]. Consequently, while FMT shows promise in potentially transforming the therapeutic approach for numerous cardiovascular diseases, including atherosclerosis, further rigorous investigations are imperative to ascertain its practical clinical significance, particularly in addressing safety concerns. Moreover, before FMT can be considered as an intervention for atherosclerosis, it is essential to establish stringent donor screening criteria and long-term safety evaluation systems, as well as to explore standardized and controllable alternative approaches (such as synthetic microbial consortia or specific functional probiotic combinations) to minimize potential risks and enhance clinical feasibility.
The structure of the intestinal microbiota or related metabolites has exhibited a profound association with atherosclerosis, as demonstrated by multiple human clinical trials. However, a majority of these analyses consist of typical correlations, hindering the establishment of a causal relationship between atherosclerosis and intestinal microbiota. Consequently, future research should prioritize prospective studies integrating pertinent metabolomic data, clinical data, and dietary factors to investigate the causal relationship among changes in intestinal microbiota.
In essence, the benefits of BAs for atherosclerosis can be categorized into two distinct clinical applications. The role of BAs in predicting atherosclerosis prognosis and as a potential therapeutic target involves their production in the liver, which plays a functional role through the transformation of intestinal microbiota. Variations in BA levels potentially mirror modifications in the composition of intestinal microbiota and the extent of ecological dysfunction. BAs also exhibit correlations with cholesterol and other lipids, encompassing LDLs, HDLs, and related compounds, all of which are of particular relevance to understanding the occurrence of atherosclerosis and patient survival. Hence, BAs serve as a valuable tool for healthcare professionals, assisting in the identification of high-risk patients while ensuring prompt follow-up testing and intervention. Notably, these factors are also intimately associated with adverse events and survival rates in atherosclerosis patients. Furthermore, treatment outcomes may be improved by the clinical screening of high-risk patients, along with follow-up testing and timely intervention. Nevertheless, there is a paucity of relevant studies; moreover, those available are mainly on Caucasians from Europe and the United States, with an absence of ethnic diversity. Thus, to ascertain the viability of BAs as a prognostic monitoring indicator and therapeutic target for atherosclerosis, there is an urgent need for further cohort analyses encompassing a more extensive array of ethnic types. In light of the preceding findings, pertinent interventions are feasible to elevate the levels of plasma BAs to impede deterioration in atherosclerosis.
Future research should also aim to define the optimal BA-targeted intervention strategies for the different subtypes of atherosclerosis, such as stable vs. unstable plaques. It is crucial to further elucidate whether distinct BA receptor pathways (e.g., FXR, TGR5) or specific microbiota-derived BA profiles exert beneficial or detrimental effects in varied patient populations. In addition, it should be recognized that various intervention modalities—including dietary modification, probiotics, pharmacological agents, or fecal microbiota transplantation—may yield heterogeneous outcomes across individuals, underscoring the necessity of developing personalized therapeutic approaches.
Additional research leveraging corresponding methodologies is pending, despite the presence of prior investigations that have employed gut microbial genome sequencing to identify the genera associated with BAs, TMAO, SCFA, and analogous metabolites. Nevertheless, there remains a substantial gap between theoretical underpinnings and their practical implementation in clinical settings. Conversely, the utilization of metabolites derived from gut microbes in blood, urine, and feces to direct targeted interventions demonstrates an enhanced clinical transformative value. Overall, these efforts can provide new directions for identifying novel therapeutic targets, optimizing risk stratification, and developing personalized intervention strategies, thereby reducing the disease burden associated with atherosclerosis.
To sum up, intestinal microbiota correlates intimately with various forms of cardiovascular diseases. BAs, one of the byproducts of intestinal microbiota-catalyzed biotransformation, have emerged as promising candidates for elucidating the mechanisms underlying the eliciting of atherosclerosis by gut microbiota. Correspondingly, these observations offer novel concepts for the subsequent development of interventions targeting the intestinal microbiota for atherosclerosis treatment. From a clinical perspective, these findings highlight the potential value of gut microbiota and BA-related biomarkers in risk stratification, prevention, and management of atherosclerosis. They also open new avenues for therapeutic intervention, examples of which include dietary modifications, probiotics, prebiotics, and FMT. The modulation of intestinal microbiota composition and the targeting of elevated BA levels represent promising avenues for the treatment of atherosclerosis. For researchers, these advances underscore the necessity of conducting in-depth mechanistic studies, large-scale multiethnic cohort investigations, and well-designed clinical trials to validate causal relationships and evaluate therapeutic efficacy. In the future, additional microbiota-derived metabolites associated with atherosclerosis are expected to be identified, which will further advance the development of safer and more effective microbiota- and BA-targeted therapeutic strategies, ultimately helping to reduce the global burden of cardiovascular diseases.
HLH and LJX designed the study. LJX drafted the manuscript. HJY, HHZ and ZJ collected the data. YHZ and KXL analyzed the data. HLH and CJH provided help and advice on the research, including planning and execution. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This study was supported by the Zhejiang Provincial Natural Science Foundation of China under Grant No. LQ23H020001 and National Natural Science Foundation of China (No. 82300363). Zhejiang Province clinical key specialty construction project (2024-ZJZK-001).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



