1 Department of Cardiology, Angiology and Intensive Care Medicine, Deutsches Herzzentrum der Charité, 13353 Berlin, Germany
2 Charité – Universitätsmedizin Berlin, Freie Universität Berlin and Humboldt-Universität zu Berlin, 10117 Berlin, Germany
3 DZHK (German Centre for Cardiovascular Research), Partner Site Berlin, 10785 Berlin, Germany
4 Division of Cardiology, Department of Medicine and Geriatrics, United Christian Hospital, Hong Kong, China
5 Division of Cardiovascular Imaging, Department of Cardiology, University Hospital Ramón y Cajal, 28034 Madrid, Spain
6 Philips Clinical Science, Philips Healthcare, 22335 Hamburg, Germany
7 Pie Medical Imaging BV, Product Management, 6227 AK Maastricht, The Netherlands
8 BIH Biomedical Innovation Academy, BIH, Berlin Institute of Health at Charité – Universitätsmedizin Berlin, 10117 Berlin, Germany
Abstract
Magnetic resonance imaging (MRI) allows for the assessment of myocardial strain and identification of heart failure (HF) patients with reduced (HFrEF), mildly reduced (HFmrEF), or preserved (HFpEF) left ventricular ejection fraction (LVEF). The cardiovascular angiographic analysis system magnetic resonance (Caas MR) strain (Pie Medical Imaging) has recently been implemented in the IntelliSpace Portal Suite (Philips Healthcare) to assess the global longitudinal strain (GLS), global circumferential strain (GCS), and global radial strain (GRS). However, standard values for this software across different HF entities, as well as normal values, have yet to be established. Thus, this study aimed to establish reference values for the GLS, GCS, and GRS using the Caas MR strain in healthy individuals and HF patients, to assess the ability of these parameters to differentiate between HF subtypes, and to compare CAAS-derived strain values with those obtained using CVI42 software.
Using a 1.5 T Philips Achieva scanner, we analyzed 19 healthy volunteers and 56 HF patients (HFpEF, n = 19; HFmrEF, n = 20; and HFrEF, n = 17) using the feature tracking post-processing software Caas MR Strain. GLS, GCS, and GRS were quantified using 4-chamber-view, 2-chamber-view, and short-axis (SAX) cine images. All volunteers and patients were evaluated by CVI42 to analyze inter-vendor reliability with a validated software.
Mean GLS, GCS, and GRS by Caas MR Strain were significantly different for healthy volunteers compared to HF patients (GLS –15.8 ± 1.9% vs. –11.7 ± 3.0%, p < 0.001; GCS –17.0 ± 2.6% vs. –11.4 ± 3.3%, p < 0.001; GRS 27.3 ± 6.2% vs. 14.5 ± 5.5%, p < 0.001). The upper limit of the 99% confidence interval for healthy volunteers was –14.6% for GLS, –15.3% for GCS and the lower limit of the 99% CI for GRS was 23.1%. GLS, GRS, and GCS by Caas MR Strain were significantly different among HF entities (p < 0.001). Intervendor comparison showed very good agreement for GLS and GRS between Caas MR Strain and CVI42 (GLS r = 0.86, p < 0.001; GCS r = 0.83, p < 0.001; GRS r = 0.76, p < 0.001).
Magnetic resonance imaging assessment of left ventricular myocardial strain using Caas MR Strain software reliably identifies HF patients. Discrimination between the different HF entities is potentially feasible by GLS, GCS, and GRS. Intervendor agreement was most robust for GLS and GCS, but less robust for GRS. For practical clinical use, we propose cut-off values for GLS above –15%, GCS above –15%, and GRS below 23% to define pathological findings.
Keywords
- cardiac magnetic resonance
- strain
- feature tracking
- deformation imaging
- healthy
- heart failure
- patients
Heart failure (HF) is a complex cardiovascular disorder characterized by impaired cardiac function and increased morbidity and mortality worldwide [1, 2, 3]. Accurate assessment of myocardial deformation in human subjects has emerged as a valuable tool in evaluating cardiac function and identifying early abnormalities in patients with HF [2, 4, 5, 6].
Myocardial strain assessment by cardiac magnetic resonance (CMR) imaging is a useful tool to measure global and regional myocardial function and deformation quantitatively in CMR imaging [5]. It has been shown to offer insights regarding the patient’s prognosis, both in acute and chronic HF [6, 7, 8, 9, 10]. Left ventricular deformation can be quantified in three dimensions: longitudinal strain, circumferential strain, and radial strain [11]. Among the various imaging techniques available, feature tracking (FT-MRI) strain analysis has gained significant attention due to its ability to quantify myocardial deformation using standard cine CMR sequences [5, 12]. After manual or automated segmentation of the myocardium in end-diastole, tracking of distinct pixel patterns (around 15 mm2) throughout the cardiac cycle for the entire myocardium reveals the myocardium deformation field from which the strain in three main axes are computed: longitudinal, radial, and circumferential strain [13]. Previous studies have demonstrated the potential of FT-MRI strain analysis in discriminating between healthy individuals and patients with HF [14, 15, 16]. This technique has been proven to be as reliable as acquisition-based strain-measurements. Post-processing solutions that allow for FT-MRI strain analysis based on standard steady-state free precession (SSFP) sequences are now offered by many vendors [17, 18]. However, universal standards for the interpretation of FT-MRI results do not currently exist, and different vendors and methods for analysis can significantly affect deformation values [18, 19, 20].
The cardiovascular angiographic analysis system magnetic resonance (Caas MR) Strain (Pie Medical Imaging) has recently been implemented in the IntelliSpace Portal Suite (Philips Medical Systems Nederland B.B., Best, The Netherlands) to assess global longitudinal (GLS), circumferential (GCS), and radial strain (GRS). It relies only on a two-chamber (2CH) and a four-chamber (4CH) view to calculate GLS and uses a full stack short axis sequence to assess GCS and GRS. However, normal values for these measurements have not yet been established.
Although previous publications have reported myocardial strain parameters using the present patient cohort, this study provides novel insights by establishing reference values for GLS, GCS and GRS using the Caas MR Strain software and reporting the first direct comparison with the widely used CVI42 platform. This study also proposes potentially clinically applicable cut-off values for pathological strain patterns in HF patients, aiming to support decision-making in routine practice. The aims of this study were threefold: to establish cutoff values for GLS, GCS, and GRS that distinguish healthy volunteers from HF patients, to evaluate the discriminatory power of these parameters in different HF subgroups, and to validate the comparability and agreement between software solutions from different vendors.
This study was performed at two centers in Berlin, Germany, the
Charité–University Medicine Berlin and the German Heart Centre Berlin, in
the years 2017 and 2018. Data from this cohort have been previously published by
our research group in studies addressing related aspects of HF phenotyping and
tissue characterization [14, 15, 21]. We included previously reported demographic
data to support the current original strain analysis using Caas MR Strain. We
prospectively identified 19 healthy volunteers without cardiovascular
comorbidities or regional wall motion abnormalities and 56 HF patients,
presenting with symptoms of HF and an increased N-terminal pro b-type natriuretic
peptide (NT-proBNP) (
After informed consent was obtained in all HF patients and volunteers, CMR was
performed on a 1.5 Tesla (T) CMR scanner (Achieva, Philips Healthcare, Best, The
Netherlands) using a five-element phased array receiver coil. Patients and
volunteers were placed in the supine position, and images were acquired during
breath holds of 10 to 15 seconds by using vector electrocardiogram gating. A
rapid balanced SSFP sequence with a repetition time (TR) = 3.3 ms, echo time (TE)
= 1.6 ms, flip angle = 60°, voxel size = 1.8
All image analyses were performed according to consensus recommendations [23]. After careful scanning for artifacts independently by two experienced readers (European Association of Cardiovascular Imaging (EACVI) level III), cine images were analyzed using commercially available software (IntelliSpace Portal V 12.1, Philips Medical Systems Nederland BV, Best, The Netherlands; Caas MR Strain, Pie Medical Imaging, Maastricht, The Netherlands). All volunteers and patients were further analyzed by CVI42 (CVI42 version 5.13.7, Circle Cardiovascular Imaging Inc., Calgary, Alberta, Canada). Caas MR Strain uses feature tracking to detect the ventricular deformation patterns. Feature tracking is based on a block-matching approach. It first identifies anatomic features in the CMR image along the myocardial boundaries, then defines regions of interest around these locations and finally tracks them along the cardiac cycle by looking for the most similar region as illustrated in Fig. 1. The strain algorithm in Caas MR Solutions represents the strain values of the whole myocardium. The type of strain calculated is the Lagrangian strain.
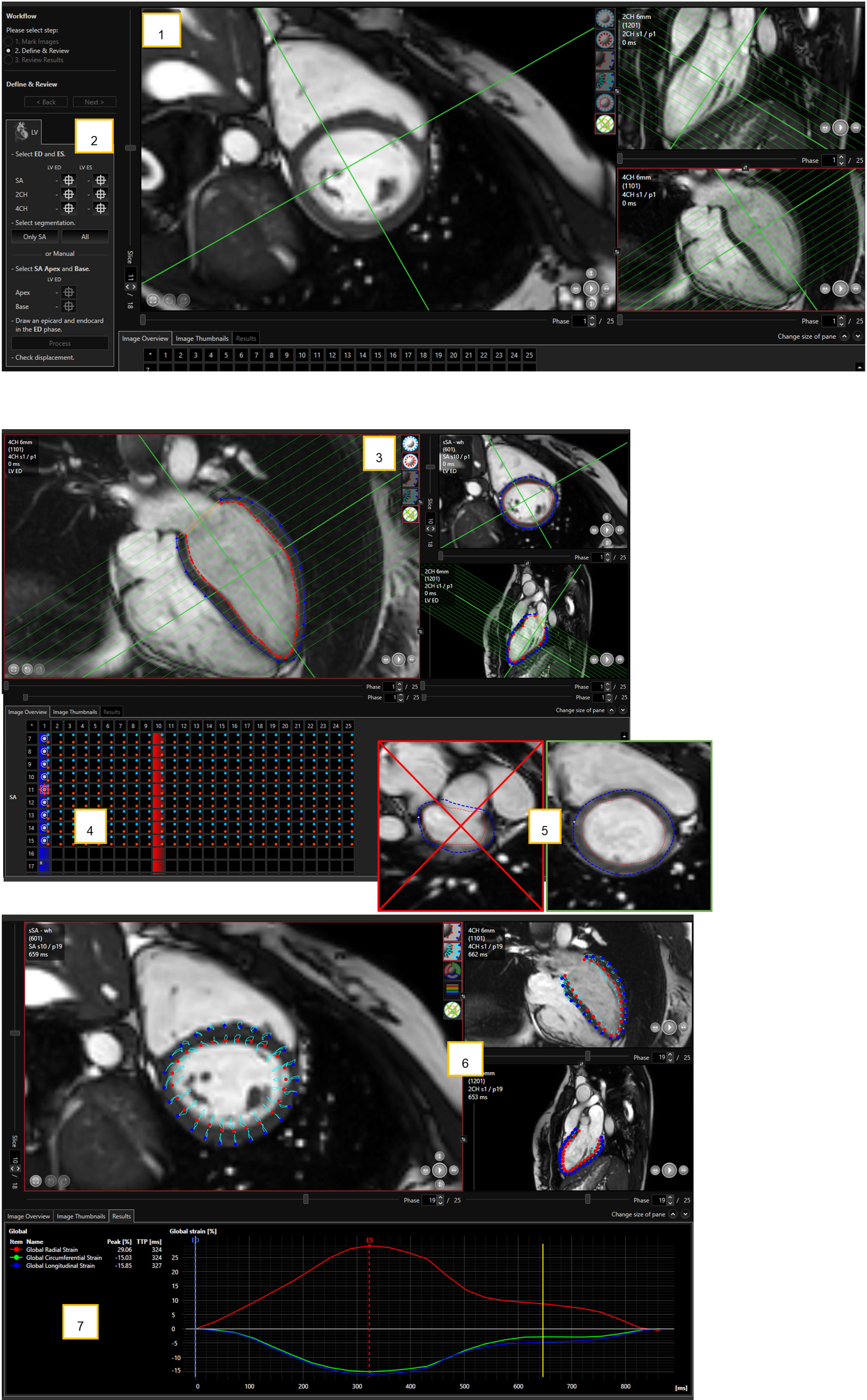 Fig. 1.
Fig. 1.
Semi-automatic left ventricular segmentation and strain analysis using Caas MR Strain. (1,2) Example of semi-automatic segmentation by Caas MR Strain. (1) Short-axis (SAX, left) and long-axis (2CH and 4CH, right) SSFP cine images were loaded directly from Philips Intellispace to the Caas MR Strain. The green lines indicate the intersection lines in the short and long axis. (2) Users can choose between automatic segmentation or manually define endo- and epicardial contours. Image processing was performed using Caas MR Strain software (Pie Medical Imaging, Maastricht, The Netherlands). (3–5) Example of a semi-automatic strain analysis by Caas MR Strain. (3) After automatic segmentation, endo- (red dotted line) and epicardial (blue dotted line) borders can be visually assessed and corrected if necessary. (4) The basal and apical slice for the short axis (SAX) stack can be defined manually. (5) SAX slices in the diastolic or systolic phases containing the left ventricular outflow tract (LVOT) should be excluded from the analysis. Caas MR Strain, Pie Medical Imaging, Maastricht, The Netherlands. (6,7) Example of a semi-automatic strain analysis by Caas MR Strain. (6) Deformation is automatically derived from feature tracking which can be assessed visually (light blue lines), for endo- (red dotted line) and epicardial (blue dotted line) borders. (7) Global strain values (not shown) and segmental values are given for user defined segments (not shown) or the American Heart Association (AHA) 17-segment model. Caas MR, cardiovascular angiographic analysis system magnetic resonance; 2CH, two-chamber; 4CH, four-chamber; SSFP, standard steady-state free precession.
LV function and volumes were quantified in a whole SAX stack according to the recommendation of the Society for Cardiovascular Magnetic Resonance (SCMR) with papillary muscles excluded from the LV volume for both vendors [23]. In the strain analysis, if the left ventricular outflow tract (LVOT) was seen in diastolic and/or systolic phases, SAX slices were completely excluded. Endo- and epicardial contours were automatically drawn in the end-diastolic and end-systolic phases, individually checked, and manually adjusted if necessary. The entire SAX was used to evaluate both segmental and global circumferential (GCS) and radial strain (GRS) by Caas MR Strain and CVI42. 2CH and 4CH were used by Caas MR Strain and 2CH, 3CH, and 4CH by CVI42 to assess segmental and global longitudinal strain (GLS). Left ventricular segmentation was based on the 17-segment model from the American Heart Association (AHA) excluding the apex (Segment 17) [24]. An example of an illustration of a strain analysis by Caas MR Strain is shown in Fig. 1.
Continuous variables are presented as mean
We identified 19 healthy volunteers without cardiovascular comorbidities or
regional wall motion abnormalities and 56 HF patients [a total of 75
participants, 27 female (36%), mean age 68
| Healthy volunteers (n = 19) | HF patients (n = 56) | Test (statistic) | p value | |
| Female, n (%) | 9 (47) | 18 (32) | 0.232 | |
| Age (years) | 63 |
69 |
t = 2.31 | 0.024* |
| Height (cm) | 171 |
170 |
t = –0.17 | 0.868 |
| Weight (kg) | 74 |
80 |
t = 1.62 | 0.109 |
| BMI (kg/m2) | 25 |
28 |
t = 1.98 | 0.051 |
| BSA (m2) | 2 |
2 |
t = 1.11 | 0.269 |
| LVEDV (mL) | 144 |
192 |
t = 2.87 | 0.005* |
| LVEDVi (mL/m2) | 76 |
98 |
t = 2.85 | 0.006* |
| LVESV (mL) | 53 |
110 |
t = 3.97 | |
| LVESVi (mL/m2) | 28 |
55 |
t = 3.96 | |
| LVM (g) | 85 |
115 |
t = 2.98 | 0.004* |
| LVMi (g/m2) | 44 |
59 |
t = 3.40 | 0.001* |
| LVEF (%) | 64 |
46 |
t = –6.08 | |
| SV (mL) | 91 |
82 |
t = –1.90 | 0.062 |
| SVi (mL/m2) | 48 |
43 |
t = –2.77 | 0.007* |
Values are presented as n (%) for categorical variables and as mean
Note: Demographic data from this cohort have been partially reported in previous publications by our research group [14, 15, 21]. These data are reproduced here to provide context for the present original analysis of myocardial strain using Caas MR.
Mean GLS by Caas MR Strain was –15.8
| Healthy (normal strain) | Heart failure (impaired strain) | |
| GLS (Caas MR Strain) | ||
| GCS (Caas MR Strain) | ||
| GRS (Caas MR Strain) |
LV, left ventricle; GLS, global longitudinal strain; GCS, global circumferential strain; GRS, global radial strain; Caas MR Strain, Pie Medical Imaging, Maastricht.
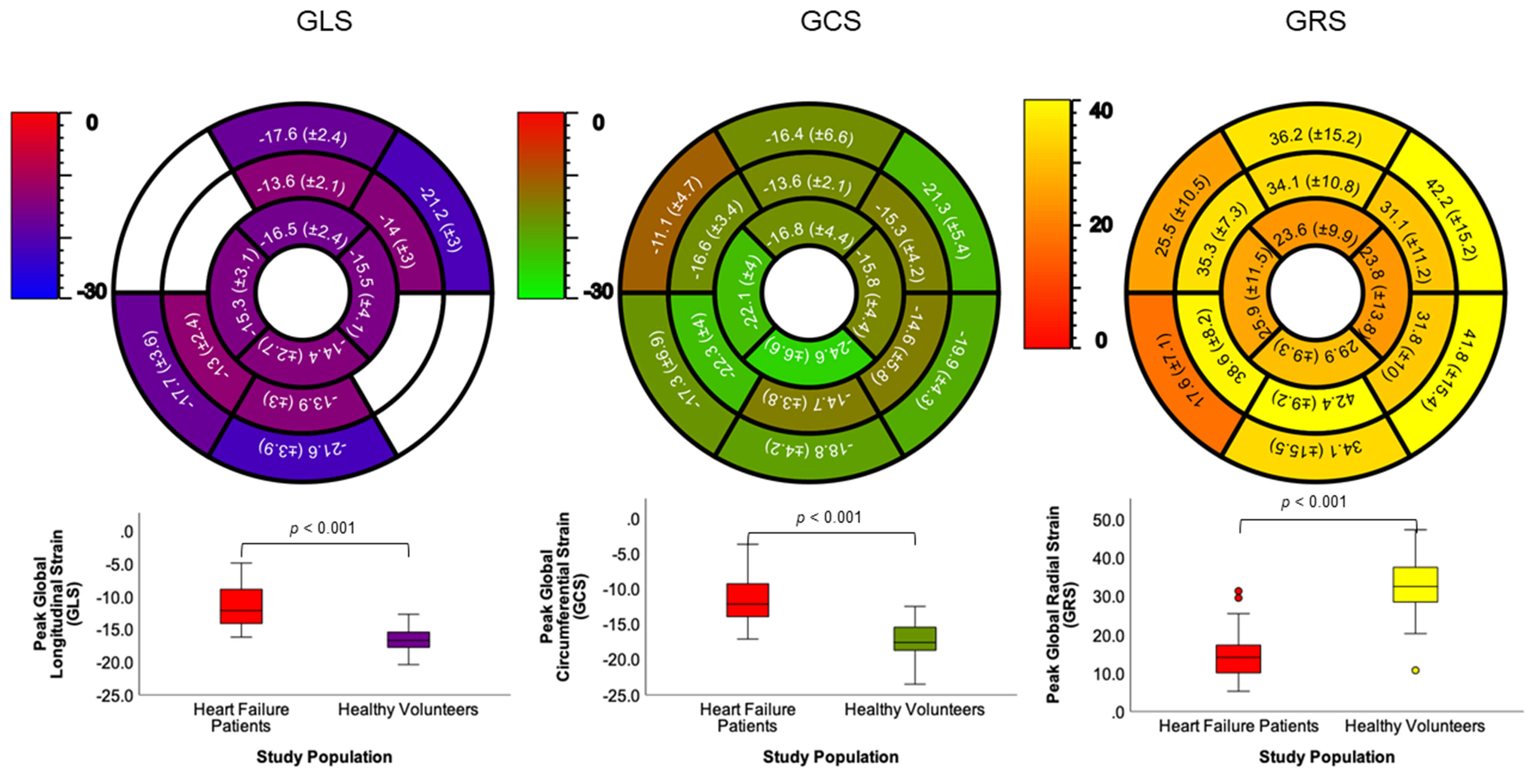 Fig. 2.
Fig. 2.
Normal values for healthy volunteers. Global and segmental
normal values for GLS (left), GCS (middle) and GRS (right). Boxplots below show
significant differences between healthy volunteers and HF patients; level
p
There was a statistically significant difference for GLS among HF patients with
different HF entities, p
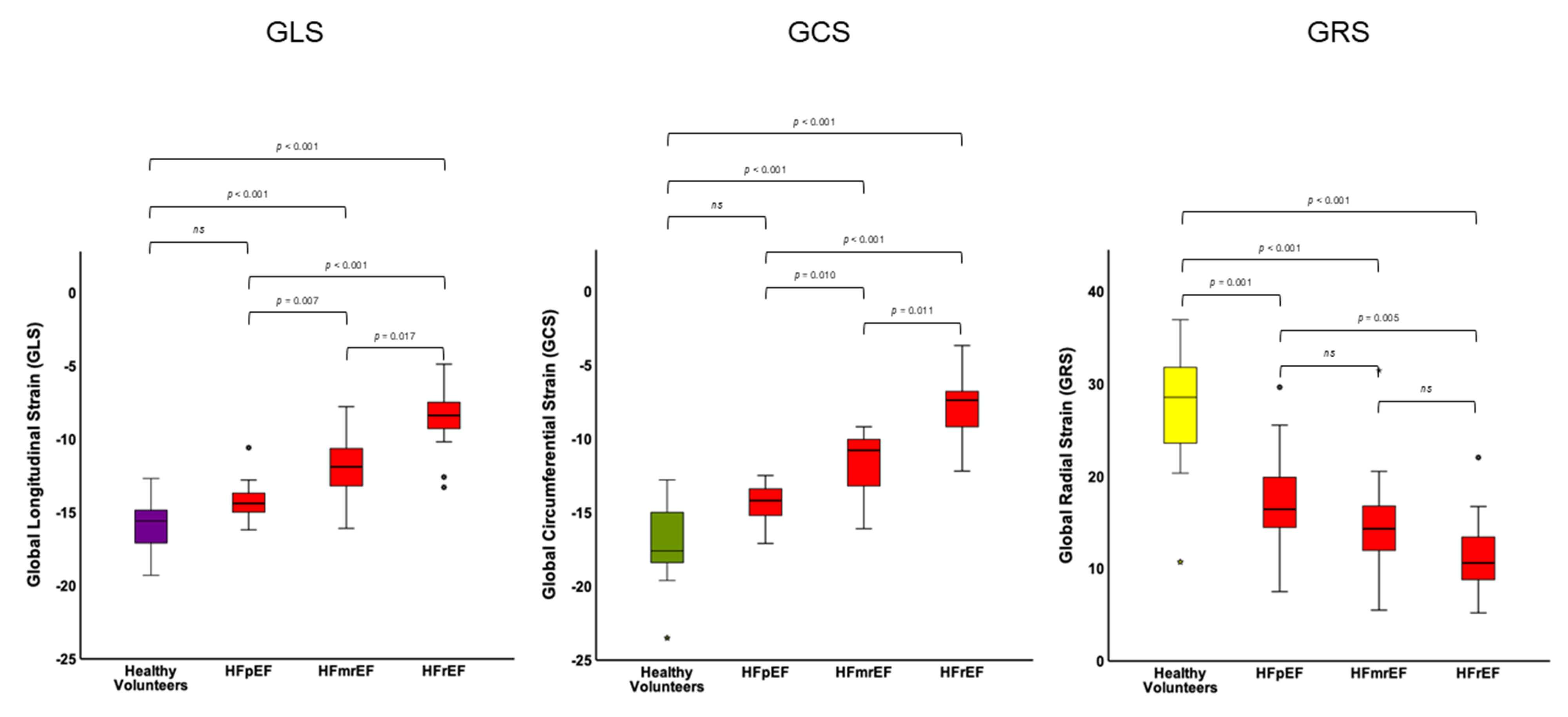 Fig. 3.
Fig. 3.
Strain values for HF entities volunteers. Global values for GLS
(left), GCS (middle) and GRS (right) for HFpEF, HFmrEF and HFrEF patients and
healthy volunteers. Significance level p
A high level of agreement was found when all participants were analyzed by two
experienced readers (both EACVI level III) to assess inter-rater reliability of
strain values. The average measure ICC for GLS by Caas MR Strain was 0.991 with a
95% CI from 0.986 to 0.994 (F = 110.319, p
Vendor-specific strain values for healthy volunteers and HF patients are
presented in Table 3. After careful evaluation of any systemic bias or outliers
in the data using Bland-Altman plots, computation of the Pearson correlation
coefficient showed a significant positive correlation for GLS for Caas MR Strain
versus CVI42 [r = 0.86, p
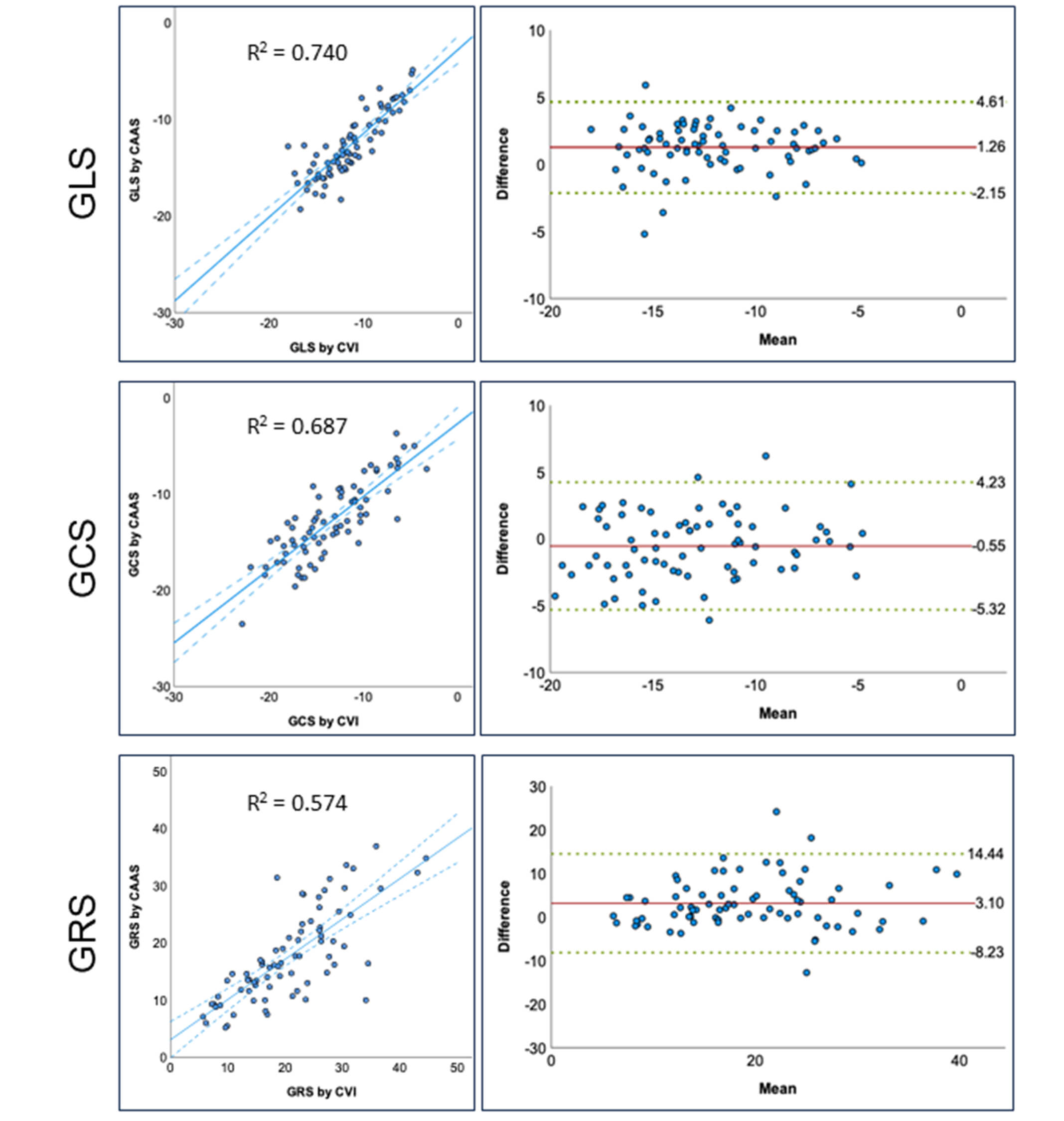 Fig. 4.
Fig. 4.
Inter-vendor comparison for GLS, GCS and GRS. Scatterplots and Bland-Altman analysis comparing Caas Strain MR with CVI42. GLS, global longitudinal strain; GCS, global circumferential strain; GRS, global radial strain; Caas MR Strain, Pie Medical Imaging, Maastricht, The Netherlands; CVI42, Circle Cardiovascular Imaging Inc., Calgary, Canada.
| Volunteers (n = 19) | HFpEF (n = 19) | HFmrEF (n = 20) | HFrEF (n = 17) | |
| GLS (Caas MR Strain) | –15.8 |
–14.3 |
–11.8 |
–8.5 |
| GLS (CVI4) | –14.9 |
–12.8 |
–10.5 |
–7.2 |
| GCS (Caas MR Strain) | –17.0 |
–14.4 |
–11.7 |
–7.8 |
| GCS (CVI4) | –17.4 |
–15.9 |
–11.7 |
–8 |
| GRS (Caas MR Strain) | 27.3 |
17.0 |
14.9 |
11.2 |
| GRS (CVI4) | 28.9 |
25.4 |
17.2 |
11 |
Values are mean
Caas MR Strain, Pie Medical Imaging, Maastricht, The Netherlands; CVI42, Circle Cardiovascular Imaging Inc., Calgary, Canada.
* Comparisons between HF subgroups and healthy volunteers are statistically
significant (p
Kruskal–Wallis test results (
To the best of our knowledge, this study is among the first to demonstrate that
calculation of GLS using only two long axis (LAX) acquisitions, 2CH and 4CH, is
feasible and shows very good agreement with CVI42-based GLS assessment
(r = 0.86, p
Our findings suggest that FT-MRI strain analysis using Caas MR Strain may help differentiate between healthy individuals and patients with HF. In this study, we were able to propose cutoff values for GLS above –14.6%, for GCS above –15.3% and GRS below 23.1% to define pathological findings. Given the higher SD for GRS, the values seem less reliable. This is in line with previous findings suggesting increased reproducibility for GLS and GCS but less for global radial strain measurements [19, 25, 26].
A recent study from our research group identified a GLS cut-off point
(
While in our study, the classification of HF was conducted based on an imaging perspective according to LVEF and FT-MRI strain parameters, the classified categories can generally be defined as different stages or severity of clinical HF, such as the classification systems used by the New York Heart Association (NYHA). In future studies, the combination of strain analysis with established clinical classifications may improve phenotyping and aid clinical decision-making.
The lower GLS observed in our study, compared to the literature, likely reflects
both physiological and methodological factors. First, our control group was
relatively older (mean age 63
In patients with advanced HF, myocardial thinning and impaired wall motion may reduce the accuracy of endocardial and epicardial border definition, which could decrease the accuracy of FT-MRI strain measurements. While previous studies have shown good agreement between FT and reference techniques (tagging or Strain-Encoded (SENC)) [12, 19], strain accuracy can be reduced as ventricular remodeling increases [25, 26]. In our study, contours were carefully reviewed and manually corrected by two experienced readers (EACVI level III), but this variability remains an inherent limitation of FT-based analysis in abnormal myocardium.
A strength of our study is the reproducibility of myocardial strain assessment. The excellent inter-rater ICC values observed for GLS, GCS, and GRS reflect low ROI-dependent variability, thereby providing quantitative error estimates for accuracy-dependent changes. This methodological robustness further supports the assumption of a high reliability of strain values measured by this novel tool.
Our study only reports values for 1.5 T and for images acquired with the use of acceleration technique SENSE, which is widely adopted in clinical practice. Therefore, although our results are specific to 1.5 T scanners and acquisitions using SENSE acceleration, they may still be cautiously extrapolated to other settings, as previous studies have suggested minimal influence of field strength and acceleration techniques on strain values [19]. Since strain rate has been shown to be of incremental value in HF patients, future analysis should ideally include these time-dependent parameters [14]. Future directions of strain analysis hold the potential for including prospective cohorts for prognostic evaluation and assessing large populations using artificial intelligence [30, 31].
Segmental strain analysis was also performed and is shown in Fig. 2. Although our primary objective focused on global strain values, segmental strain may provide additional diagnostic information. However, segmental strain is generally associated with higher variability and lower reproducibility, which limits its immediate applicability in clinical practice. Future studies with larger cohorts should explore the diagnostic potential of segmental strain values in different HF populations.
This study has several limitations. We did not report age or gender-specific
values, which could be beneficial since it has been shown that age is a potential
confounder and younger patients are reported to have more pronounced myocardial
deformation values [13, 29]. However, since we chose to study a group of HF
patients, our values from relatively old volunteers (mean age 63
CMR imaging assessment of left ventricular myocardial strain using Caas MR Strain software reliably identifies HF patients. Discrimination between HF subgroups by GLS, GCS, and GRS showed statistically significant trends. However, given the limited sample size, these findings should be considered exploratory and require confirmation in larger studies. Inter-vendor agreement was most robust for GLS and GCS, but less robust for GRS. For practical clinical use, we propose cut-off values for GLS above –15%, GCS above –15%, and GRS below 23% to better define pathological changes.
Patients signed informed consent regarding publishing their data.
All data will be available from the corresponding author upon reasonable request.
MB, DH, H-DD, and SK conceived and designed the study. MB, DH, SK acquired clinical data. SC and KJW reviewed all scans and performed blinded strain analysis. KJW executed the statistical analysis. KJW and ICS drafted the manuscript. GV assisted with methodology and figure design. PD, KC, RT, SW, JV, REB, NM and CS revised and amended critical parts of the manuscript and approved the final version of this manuscript. All authors contributed to the interpretation of the data, revised and amended critical parts of the manuscript and approved the final version of this manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study protocol, inclusion of study participants, data access, and usage fully comply with the Declaration of Helsinki. All studies were reviewed and approved by the Charité–Universitätsmedizin Berlin Ethics Committee (Approval number: EA4/112/16) complied with the Declaration of Helsinki and was registered at the German Register for Clinical Studies (DRKS) (registration number: DRKS00015615). Informed consent was obtained from all individual participants included in the study.
We thank Corinna Else, Alireza Khasheei and Mandy Domning for their assistance.
The authors declare that this study received funding from Philips Healthcare, DZHK (German Centre for Cardiovascular Research), Partner Site Berlin, Germany and Myocardial Solutions. This work was partially funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)-SFB-1470-B06. The funders were not involved in the study design, collection, analysis, interpretation of data, and the writing of this article or the decision to submit it for publication. ICS received funding from the European Association of Cardiovascular Imaging (EACVI Training Grant 2025).
Sebastian Kelle reports grants and other support by the DZHK (German Center for Cardiovascular Research), Partner Site Berlin, Philips Healthcare, BioVentrix, Berlin-Chemie, Merck/Bayer, Novartis, AstraZeneca, Siemens and Myocardial Solutions outside of the submitted work. SK was also on the advisory board for Merck/Bayer, BioVentrix, and Myocardial Solutions. All other authors declare that they have no relationships relevant to the contents of this paper to disclose. Christian Stehning is an employee of Philips Healthcare or has a financial relationship with the company. Gaston Vogel is an employee of Pie Medical Imaging BV or has a financial relationship with the company. However, the company had no role in the handling or conduct of the study. The authors had full access to all data in the study and take full responsibility for the integrity of the data and the accuracy of the data analysis.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




