1 Institute of Clinical Medical Sciences, China-Japan Friendship Hospital, Chinese Academy of Medical Sciences, Peking Union Medical College, 100029 Beijing, China
2 Department of Cardiology, China-Japan Friendship Hospital, 100029 Beijing, China
3 Department of Cardiology, The Third People's Hospital of Chengdu, Affiliated Hospital of Southwest Jiaotong University, 610031 Chengdu, Sichuan, China
†These authors contributed equally.
Abstract
Renal dysfunction is linked to both the complexity of coronary artery lesions and the prognosis of acute coronary syndrome (ACS). However, the nature of this intricate relationship remains unclear. Therefore, this study aimed to investigate the mechanisms through which coronary lesion complexity mediates the association between renal dysfunction and adverse cardiovascular outcomes in patients with ACS.
This analysis included 1400 ACS patients who underwent percutaneous coronary intervention (PCI). Renal function was assessed using the estimated glomerular filtration rate (eGFR), calculated according to the 2021 Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation, which incorporates both creatinine and cystatin C. Coronary lesion complexity was evaluated using the baseline SYNTAX score (bSS). The associations among eGFR, bSS, and major adverse cardiovascular events (MACEs) were examined using survival analysis, restricted cubic spline (RCS) analysis, and mediation analysis.
A total of 229 MACEs (16.4%) occurred over a median follow-up of 31.03 (27.34, 35.06) months, including 99 all-cause deaths (7.0%), 41 myocardial infarctions (2.9%), and 123 unplanned revascularizations (8.9%). After multivariate adjustment, both the eGFR and bSS significantly predicted MACEs across the total population and various subgroups. Mediation analysis showed that bSS mediated 16.49%, 14.76%, 12.87%, and 11.95% of the correlation between eGFR and MACEs in different adjusted models.
The relationship between renal dysfunction and MACEs in ACS patients is partially mediated by coronary lesion complexity. This finding underscores the importance of integrating kidney function assessments with coronary anatomical evaluations to develop individualized risk stratification strategies.
Keywords
- SYNTAX score
- estimating glomerular filtration rate
- cystatin C
- acute coronary syndrome
- mediation
Acute coronary syndrome (ACS) is a severe manifestation of coronary artery disease, characterized by increased morbidity and mortality [1]. Renal dysfunction is recognized as a significant risk factor in the progression and prognosis of atherosclerotic coronary vessel disease (ASCVD) [2, 3, 4, 5]. In the United States, the prevalence of chronic kidney disease (CKD) among patients undergoing percutaneous coronary intervention (PCI) is 30.5% for those with ST-elevation myocardial infarction (STEMI) and 42.9% for those with non-ST-elevation myocardial infarction (NSTEMI) [6]. Several studies have demonstrated that as the severity of renal impairment increases, the incidence of cardiovascular death and myocardial infarction significantly increases [7, 8, 9].
CKD leads to alterations in calcium and phosphorus metabolism [10], which, along with traditional metabolic risk factors such as diabetes mellitus, inflammation, and disorders of lipid metabolism, collectively accelerate the progression of atherosclerotic lesions [5, 11, 12, 13, 14]. Consequently, patients with CKD frequently exhibit complex and severe coronary artery lesions, characterized by multivessel disease and arterial calcification [15, 16, 17]. Among dialysis patients, the prevalence of coronary artery calcification ranges from 25% to 68.3% [18, 19, 20], with an annual progression rate between 20.8% and 27.6% [21, 22]. Previous studies have demonstrated that impaired renal function serves as an independent prognostic factor for baseline SYNTAX scores (bSS) [23]—which is a validated metric for determining the complexity of coronary artery disease [16, 17]. This complexity complicates revascularization procedures and may lead to adverse outcomes after PCI, including mortality and repeat revascularization in ACS.
In 2023, the American Heart Association (AHA) proposed a novel framework termed the cardiovascular-kidney-metabolic syndrome (CKM) to highlight the interconnections among metabolic risk factors, renal dysfunction and the cardiovascular system [13, 24]. However, there is currently a lack of studies exploring the pathway between CKD and coronary lesion complexity in the prognosis of ACS. Therefore, this study aimed to explore the role of the bSS in the relationship between estimated glomerular filtration rate (eGFR), calculated using the 2021 Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation incorporating both creatinine and cystatin C [25, 26], and adverse cardiovascular events in ACS patients.
A cohort of 1400 patients from the Third People’s Hospital of Chengdu, Sichuan, China, who underwent PCI between July 2018 and December 2020, were included in the study. Plasma creatinine and cystatin C measurements were recorded for each participant. The study excluded patients with: (1) prior coronary artery bypass grafting (CABG) surgery; (2) structural heart disease necessitating surgical or percutaneous treatment; (3) severe hepatic/respiratory dysfunction; (4) advanced malignancies with limited life expectancy; (5) in-hospital mortality; and (6) more than 10% of critical medical data missing.
Demographic characteristics, medical background, and key clinical parameters were extracted from electronic health records. Fasting venous blood samples were analyzed for lipid profiles, fasting blood glucose, homocysteine, renal biomarkers (serum creatinine, cystatin C), brain natriuretic peptide (BNP), and cardiac troponin T (cTnT) using standardized assays. The bSS was computed using an online calculator available at https://syntaxscore.org/. Preprocedural angiograms were independently assessed by two blinded evaluators, with discrepancies resolved by a third arbitrator. All data were meticulously entered into a specialized computer database, which was then rigorously checked for quality to ensure precision and reliability. eGFR values were calculated using the 2021 CKD-EPI equation, which integrates both creatinine and cystatin C measurements. This method yields a more accurate estimation of the glomerular filtration rate (GFR) than equations relying solely on creatinine [25].
Follow-up assessments were conducted at 1, 3, 6, and 12 months post-discharge, followed by annual evaluations via telephone interviews or clinic visits. Trained personnel prospectively documented clinical events throughout the follow-up period. The primary endpoint was major adverse cardiovascular events (MACEs), defined as a composite of all-cause mortality, nonfatal myocardial infarction (MI), or ischemia-driven unplanned revascularization. Secondary endpoints included all-cause mortality, cardiac death, MI, unplanned revascularization and nonfatal stroke. All endpoints underwent rigorous adjudication by an independent committee based on standardized criteria, including review of medical records and imaging data.
The sample size was calculated based on the expected incidence of MACEs over two
years (16%), with a target power of 0.8, a two-sided significance level
(
Continuous variables were presented as mean
The correlation between the bSS and eGFR was explored using Spearman’s
correlation analysis. Logistic regression analysis was adopted to analyze the
association between risk factors including eGFR and the angiographic severity of
coronary artery disease (CAD) (bSS
Mediation analysis was performed using the ‘mediation’ package in the R
Programming Language 4.4.1 (R Foundation for Statistical Computing, Vienna,
Austria). The analysis evaluated the mediating role of coronary artery lesion
complexity (bSS as a continuous variable) in the association between renal
dysfunction (eGFR as a continuous variable) and MACEs (time-to-event endpoint),
adjusted for multiple covariates in separate multivariable Cox regression models.
Bayesian methods were applied for the estimation of mediation effects. A
significant mediating effect was established when meeting all three criteria: (a)
statistically significant indirect effect (95% credible interval excluding
null); (b) significant total effect (p
Multiple confounding factors were adjusted in separate multivariable Cox
regression models, with variable selection guided by either statistical
significance (p
The final analytic cohort meeting the eligibility criteria included 1400
patients (Supplementary Fig. 1), with a mean age of 67.1
| Variable | Total population | No MACEs (n = 1171) | MACEs (n = 229) | p | |
| Age, years | 67.09 |
66.31 |
71.08 |
||
| Female, n (%) | 405 (28.9) | 336 (28.7) | 69 (30.1) | 0.661 | |
| BMI, kg/m2 | 24.39 |
24.47 |
24.02 |
0.036 | |
| Smoking, n (%) | 742 (53.0) | 626 (53.5) | 116 (50.7) | 0.437 | |
| Previous PCI, n (%) | 121 (8.6) | 94 (8.0) | 27 (11.8) | 0.064 | |
| COPD, n (%) | 44 (3.2) | 31 (2.7) | 13 (5.7) | 0.016 | |
| Hypertension, n (%) | 951 (67.9) | 786 (67.1) | 165 (72.1) | 0.144 | |
| DM, n (%) | 558 (39.9) | 444 (37.9) | 114 (49.8) | 0.001 | |
| Previous stroke, n (%) | 66 (4.7) | 54 (4.6) | 12 (5.2) | 0.681 | |
| SBP, mmHg | 133.14 |
133.16 |
133.05 |
0.942 | |
| Heart rate, bpm | 7.11 |
76.83 |
79.60 |
0.004 | |
| cTnT, pg/mL | 40.82 (11.73, 821.15) | 34.29 (10.95, 750.80) | 101.10 (16.15, 1094.00) | ||
| BNP, pg/mL | 124.65 (43.83, 328.49) | 117.90 (40.60, 328.49) | 213.40 (74.10, 630.20) | ||
| Serum creatinine, mg/dL | 0.87 (0.74, 1.04) | 0.87 (0.74, 1.02) | 0.89 (0.74, 1.21) | 0.005 | |
| Cystatin C, mg/dL | 1.14 (0.97, 1.41) | 1.13 (0.96, 1.36) | 1.27 (1.04, 1.73) | ||
| eGFR, mL/min/1.73 m2 | 75.62 (58.49, 91.24) | 77.13 (61.43, 92.34) | 65.02 (43.59, 84.75) | ||
| eGFR |
367 (25.2) | 282 (24.1) | 85 (37.1) | ||
| FBG, mmol/L | 6.96 |
6.92 |
7.17 |
0.285 | |
| TG, mmol/L | 1.52 (1.08, 2.13) | 1.55 (1.08, 2.17) | 1.44 (1.11, 1.89) | 0.203 | |
| TC, mmol/L | 4.37 (3.62, 5.15) | 4.36 (3.64, 5.18) | 4.44 (3.55, 5.10) | 0.760 | |
| HDL-C, mmol/L | 1.14 (0.96, 1.30) | 1.14 (0.96, 1.30) | 1.14 (0.95, 1.31) | 0.740 | |
| LDL-C, mmol/L | 2.67 (2.11, 3.24) | 2.67 (2.12, 3.24) | 2.68 (2.06, 3.25) | 0.829 | |
| Hcy, µmol/L | 14.15 (11.10, 17.2) | 13.90 (11.00, 16.72) | 15.60 (11.65, 19.95) | 0.001 | |
| LVEF | 54.99 |
55.47 |
52.54 |
||
| AMI, n (%) | 702 (50.1) | 568 (48.5) | 134 (58.5) | 0.006 | |
| Diagnosis, n (%) | 0.001 | ||||
| UA | 698 (49.9) | 603 (51.5) | 95 (41.5) | ||
| NSTEMI | 304 (21.7) | 233 (19.9) | 71 (31.0) | ||
| STEMI | 398 (28.4) | 335 (28.6) | 63 (27.5) | ||
| Aspirin, n (%) | 1365 (97.5) | 1150 (98.2) | 215 (93.9) | ||
| P2Y12 receptor inhibitor, n (%) | 1379 (98.5) | 1154 (98.5) | 225 (98.3) | 0.737 | |
| Statins, n (%) | 1372 (98.0) | 1148 (98.0) | 224 (97.8) | 0.828 | |
| 971 (69.4) | 815 (69.6) | 156 (68.1) | 0.658 | ||
| ACEI/ARB, n (%) | 638 (45.6) | 530 (45.3) | 108 (47.2) | 0.597 | |
| Diuretics, n (%) | 242 (17.3) | 172 (14.7) | 70 (30.6) | ||
| Insulin, n (%) | 147 (10.5) | 107 (9.1) | 40 (17.5) | ||
| bSS |
279 (19.9) | 201 (17.2) | 78 (34.1) | ||
| bSS | 13.00 (8.00, 20.00) | 12.00 (7.00, 19.00) | 19.00 (12.00, 26.50) | ||
COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; SBP, systolic blood pressure; FBG, fasting blood glucose; TG, triglyceride; TC, total cholesterol; Hcy, homocysteine; MACEs, major adverse cardiovascular events; PCI, percutaneous coronary intervention; BNP, brain natriuretic peptide; eGFR, estimated glomerular filtration rate; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; cTnT, cardiac troponin T; LVEF, left ventricular ejection fraction; NSTEMI, non-ST-elevation myocardial infarction; STEMI, ST-elevation myocardial infarction; AMI, acute myocardial infarction; UA, unstable angina; ACEI/ARB, angiotensin converting enzyme inhibitor/angiotensin receptor blocker; bSS, baseline SYNTAX score; BMI, body mass index.
Spearman’s correlation analysis identified a statistically significant negative
correlation between eGFR and bSS (r = –0.20, p
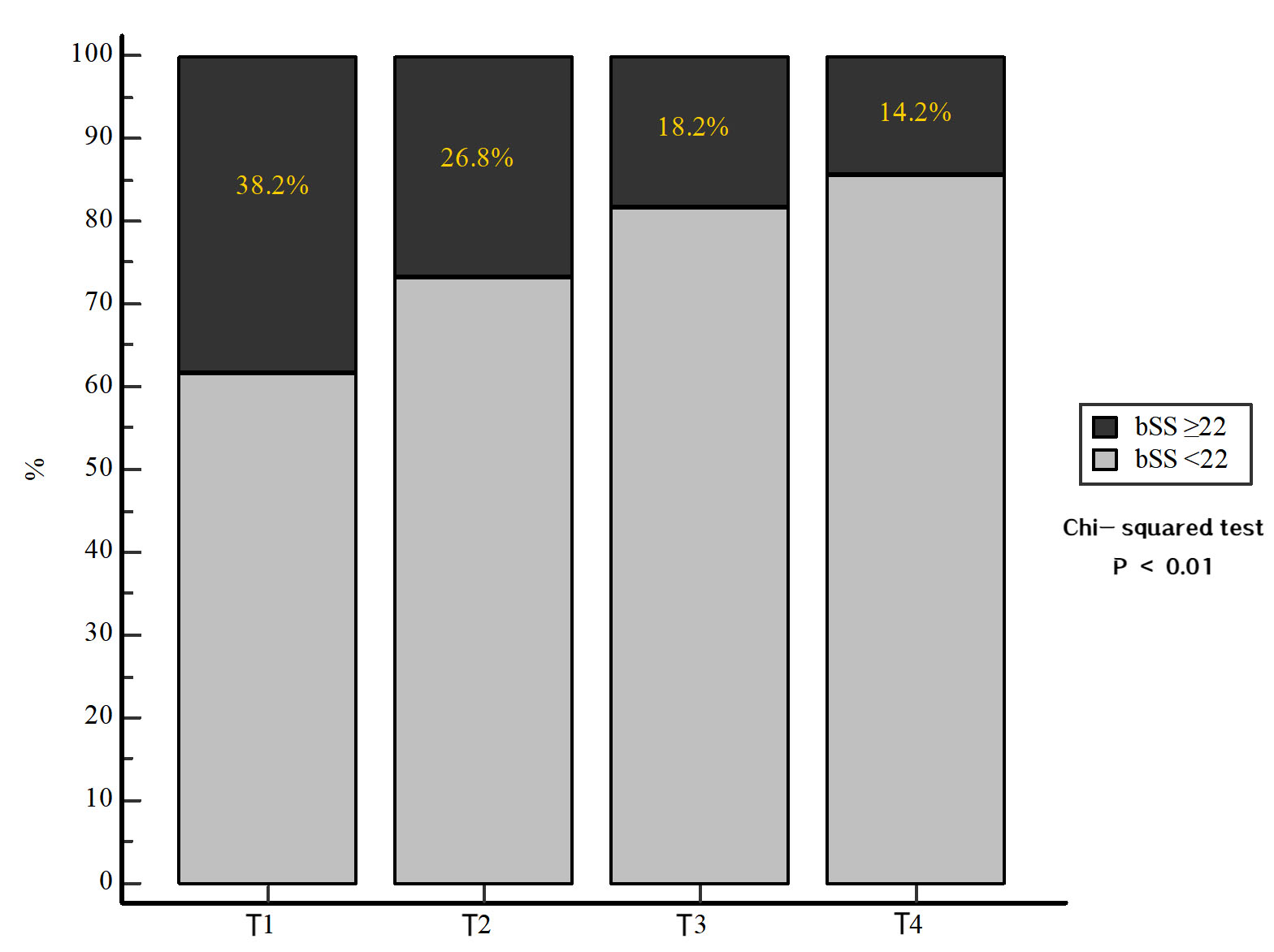 Fig. 1.
Fig. 1.
The proportion of complex lesions (bSS
Multivariate logistic regression analysis revealed that each 10-unit decrease in
eGFR was independently associated with an increased likelihood of complex lesions
(bSS
Over a median follow-up period of 31.03 (27.34, 35.06) months, 229 cases
(16.4%) of MACEs were documented, including 99 cases (7.0%) of all-cause death,
41 cases (2.9%) of MI, and 123 cases (8.9%) of unplanned revascularization. The
incidence of MACEs, all-cause death, and cardiac death increased with decreased
eGFR and elevated bSS (Fig. 2 and Supplementary Table 3,
Supplementary Figs. 4,5). Patients were further stratified into
four groups based on eGFR and bSS. Kaplan-Meier curves indicated that individuals
with both lower eGFR (
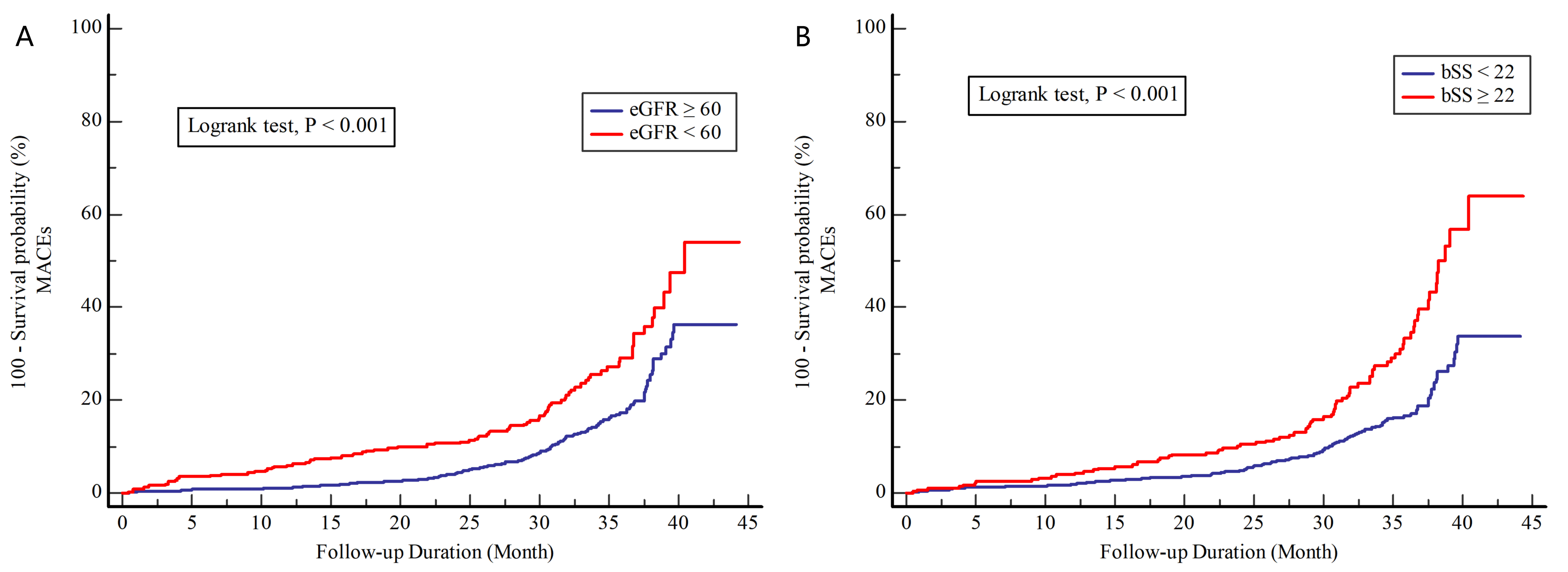 Fig. 2.
Fig. 2.
Cumulative incidence of MACEs stratified by eGFR (A) and bSS (B). bSS, baseline SYNTAX score; eGFR, estimated glomerular filtration rate; MACEs, major adverse cardiovascular events.
Univariate Cox regression showed that eGFR, age, BMI, AMI, DM, heart rate, bSS,
LVEF, diuretics, and insulin use were risk factors for MACEs, whereas aspirin use
was associated with a reduced risk (Supplementary Table 4). After
adjusting for multiple confounding factors, each 10-unit decrease in eGFR (Model
I: HR 1.125, 95% CI: 1.056–1.197, p
| MACEs | eGFR (Per 10-unit decrease) | bSS (Per 1-unit increase) | ||
| HR (95% CI) | p | HR (95% CI) | p | |
| Unadjusted Model | 1.190 (1.130–1.254) | 1.042 (1.029–1.054) | ||
| Adjusted Model I | 1.125 (1.056–1.197) | 1.037 (1.024–1.050) | ||
| Adjusted Model II | 1.095 (1.029–1.166) | 0.004 | 1.035 (1.021–1.049) | |
| Adjusted Model III | 1.093 (1.024–1.166) | 0.007 | 1.034 (1.020–1.048) | |
Model I was adjusted for age, sex, BMI, HTN, DM, smoking, previous PCI, TG, TC, LDL-C, HDL-C; Model II was adjusted for age, BMI, DM, Heart rate, diuretics, Isu, aspirin, AMI, LVEF; Model III was adjusted for Model I plus Model II. bSS, baseline SYNTAX score; eGFR, estimated glomerular filtration rate; MACEs, major adverse cardiovascular events; BMI, body mass index; HTN, hypertension; DM, diabetes mellitus; PCI, percutaneous coronary intervention; TG, triglyceride; TC, total cholesterol; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; HR, hazard ratio; AMI, acute myocardial infarction; LVEF, left ventricular ejection fraction.
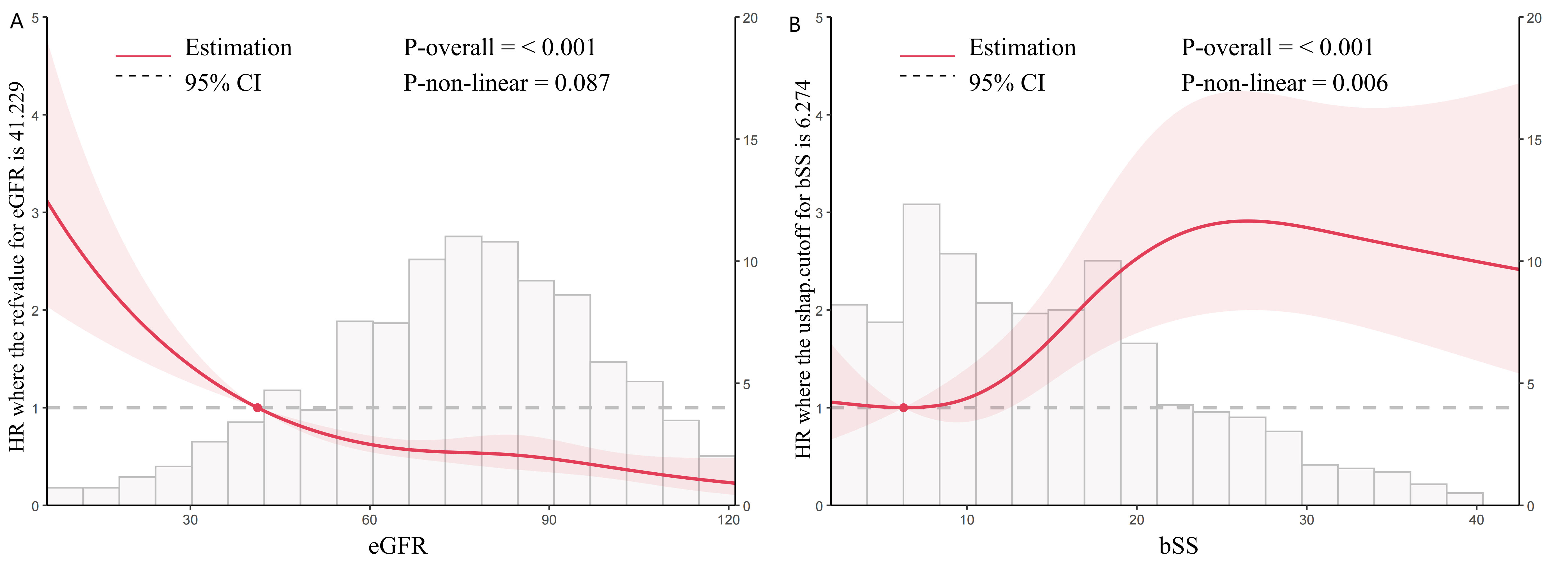 Fig. 3.
Fig. 3.
Dose-responsive relationship of the eGFR (A) and bSS (B) with MACEs. The RCS curves are derived from a Cox regression adjustment model, which includes factors such as age, sex, BMI, HTN, DM, smoking, previous PCI, TG, TC, LDL-C, HDL-C, HR, diuretics, Isu, aspirin, AMI, LVEF. RCS, restricted cubic spline; bSS, baseline SYNTAX score; eGFR, estimated glomerular filtration rate; MACEs, major adverse cardiovascular events; BMI, body mass index; HTN, hypertension; DM, diabetes mellitus; PCI, percutaneous coronary intervention; TG, triglyceride; TC, total cholesterol; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; HR, hazard ratio; AMI, acute myocardial infarction; LVEF, left ventricular ejection fraction.
Subgroup analyses were performed to determine if the predictive power of eGFR
and bSS was consistent across different demographic groups and comorbidities
(Fig. 4). After adjusting for various confounding variables, we found that both
eGFR and bSS were significant indicators of MACEs across various subgroups. In
the selected subgroup, no significant association was found with the risk of MACE
(all interaction p-values were
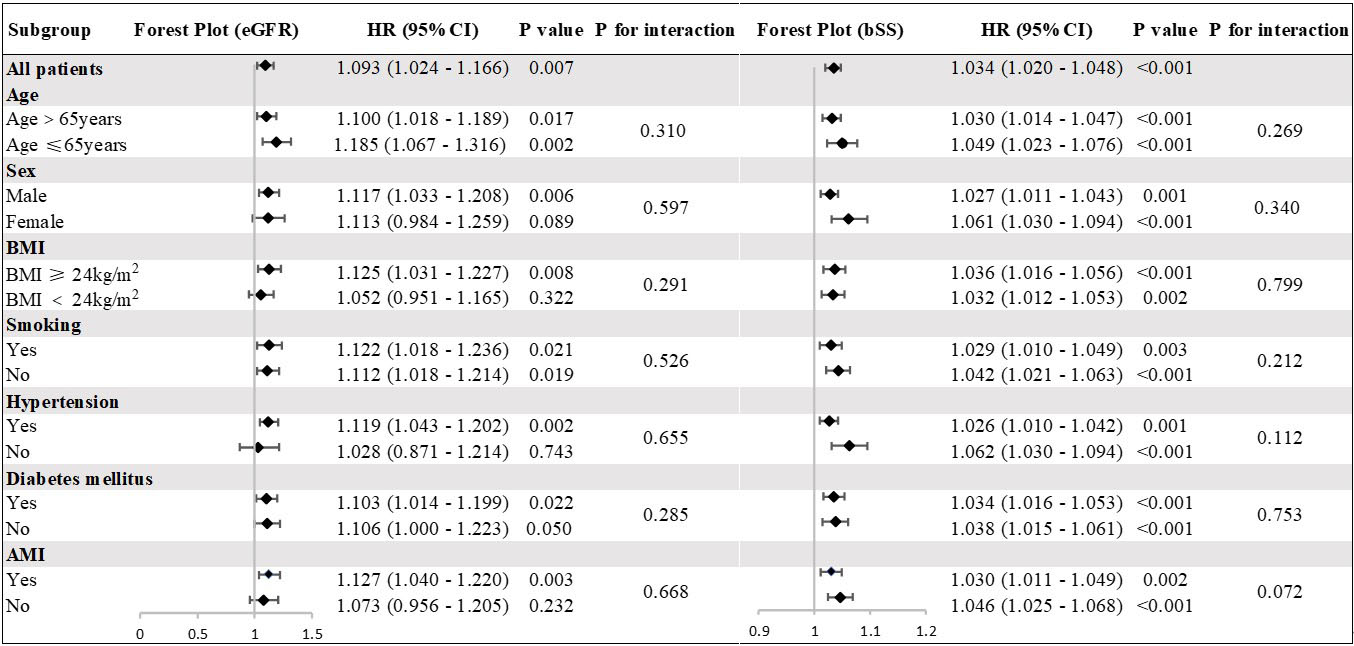 Fig. 4.
Fig. 4.
Subgroup Analyses for MACEs. The hazard ratio (HR) indicates that each 10-unit decrease in eGFR and each 1-unit increase in bSS correspond to an increased risk. All models were adjusted for age, sex, BMI, HTN, DM, smoking, previous PCI, TG, TC, LDL-C, HDL-C, Heart rate, diuretics, Isu, aspirin, AMI, LVEF. bSS, baseline SYNTAX score; eGFR, estimated glomerular filtration rate; MACEs, major adverse cardiovascular events; BMI, body mass index; HTN, hypertension; DM, diabetes mellitus; PCI, percutaneous coronary intervention; TG, triglyceride; TC, total cholesterol; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; AMI, acute myocardial infarction; LVEF, left ventricular ejection fraction.
As illustrated in Fig. 5, the mediation analysis demonstrated that the complexity of coronary artery lesions (bSS) mediated the relationship between renal function (represented by eGFR) and the occurrence of MACEs across various adjusted models. Specifically, bSS accounted for mediation proportions of 16.49%, 14.76%, 12.87%, and 11.95% in unadjusted Model, adjusted Model I, adjusted Model II, and adjusted Model III analyses, respectively. Furthermore, we tested the robustness of our findings by substituting eGFR values derived solely from creatinine. The mediation proportions were 20.71%, 20.30%, 17.20%, and 15.66% across the respective analyses (Supplementary Fig. 9). Similar mediating effects were noted when endpoint was defined as all-cause death (Supplementary Fig. 10).
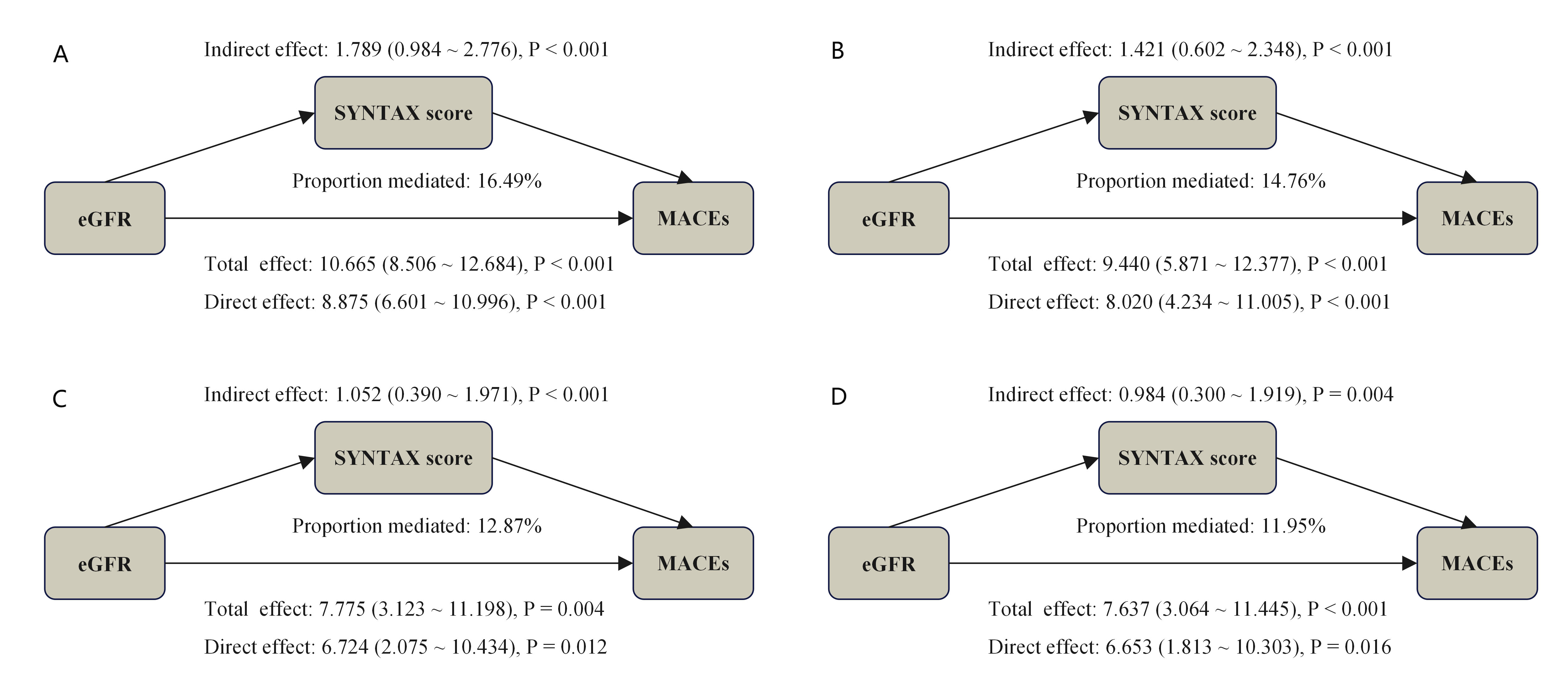 Fig. 5.
Fig. 5.
Causal mediation analysis quantified bSS-mediated pathways in eGFR-MACE associations across covariate-adjusted models. (A) represents the unadjusted Model; (B) represents the adjusted Model I; (C) represents the adjusted Model II; (D) represents the adjusted Model III. Model I was adjusted for age, sex, BMI, HTN, DM, smoking, previous PCI, TG, TC, LDL-C, HDL-C; Model II was adjusted for age, BMI, DM, Heart rate, diuretics, Isu, aspirin, AMI, LVEF; Model III was adjusted for Model I plus Model II. bSS, baseline SYNTAX score; eGFR, estimated glomerular filtration rate; MACE, major adverse cardiovascular event; BMI, body mass index; HTN, hypertension; DM, diabetes mellitus; PCI, percutaneous coronary intervention; TG, triglyceride; TC, total cholesterol; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; AMI, acute myocardial infarction; LVEF, left ventricular ejection fraction.
In this retrospective cohort study of 1400 patients with ACS followed for up to 31.03 months, a lower eGFR (calculated using the 2021 CKD-EPI equation with creatinine and cystatin C) and a higher bSS were significantly associated with an increased risk of MACEs after PCI. These associations remained significant after adjusting for established cardiovascular risk factors across models and subgroups. Moreover, the study suggested that an elevated bSS partially mediated the relationship between renal dysfunction and adverse cardiovascular outcomes in ACS patients.
Previous research has demonstrated that an extended duration of CKD,
particularly in patients progressing to dialysis, is associated with an increased
likelihood of complex coronary artery lesions (bSS
Furthermore, complex coronary artery lesions are linked to increased risks of perioperative complications, target lesion failure, recurrent MI, and in-stent restenosis post-PCI [29, 30], all of which collectively increase the incidence of adverse events. Our study revealed a significant positive dose-response relationship between elevated bSS and adverse cardiovascular events. Notably, a non-linear association was observed between bSS and MACE, whereas all-cause mortality exhibited a linear dose-response pattern. This discrepancy may be attributed to the composition of the composite MACE endpoint, in which unplanned revascularization accounted for 53.71% (123/229) of events. Theoretically, while increasing lesion complexity increases the likelihood of requiring late revascularization, patients with highly complex lesions may face technical barriers to revascularization and consequently opt for conservative management, potentially explaining the non-linear bSS-MACE relationship.
This study provides the first evidence that coronary lesion complexity partially mediates the association between renal dysfunction and adverse cardiovascular outcomes, shedding light on the pathophysiological interplay between CKD and coronary atherosclerosis. This mediation may be attributed to several factors. On one hand, CKD exacerbates calcium-phosphate metabolism disorders and inflammatory responses. These, along with associated metabolic risk factors such as obesity, diabetes, hypertension, and lipid metabolism disorders, collectively promote the progression of coronary atherosclerosis and vascular calcification [5, 11, 12, 13]. In addition, non-traditional risk factors also play a potential role in advancing atherosclerosis [5, 31]. For example, studies have revealed that enhanced tyrosine sulfation [31], as well as p-cresyl sulfate (PCS) [32], is associated with CKD-related atherosclerosis. Furthermore, a prospective cohort study (the CRUISE-MET study, NCT06383208) utilized lipidomics to investigate non-traditional lipid metabolites that promote the progression of coronary lesions in CKD patients [33]. Nevertheless, the mechanisms responsible for CKD-specific atherosclerosis are still not completely understood and warrant further study.
In summary, routine assessment of renal function using the 2021 CKD-EPI equation (incorporating creatinine and cystatin C) [25, 26] is critical for ASCVD risk management, and allows early identification of high-risk CKD populations. In high-risk populations, targeted use of cardiorenal protective agents—including finerenone [34], glucagon-like peptide-1 (GLP-1) receptor agonists [35], and sodium-glucose cotransporter-2 (SGLT-2) inhibitors [36]—may attenuate the progression of atherosclerosis [14, 37]. Furthermore, current ACS risk stratification tools (e.g., GRACE score) rely solely on creatinine-based renal estimates and omit coronary lesion complexity, limiting their utility in personalized care [38, 39, 40]. To address this gap, development of AI-enhanced tools integrating eGFR, bSS, and novel biomarkers [41] could better predict post-PCI MACE [39, 41]. Specifically for renal-impaired patients, combining renal function and coronary lesion assessments—guided by intravascular imaging or functional testing—may optimize revascularization strategies and clinical outcomes.
While this study provides valuable insights, it also presents certain limitations that warrant consideration. First, the single-center observational design limits causal inference between eGFR, bSS, and cardiovascular outcomes, and residual confounding may persist despite adjustment for known risk factors. Nevertheless, this limitation has been partially mitigated through subgroup analyses and sensitivity analyses. Secondly, although the 2021 CKD-EPI equation (combining creatinine and cystatin C) improved the accuracy for estimating GFR, reliance on a single in-hospital eGFR measurement may introduce bias due to fluctuations in short-term renal function. Future studies should incorporate repeated eGFR measurements during follow-up or use urinary biomarkers (e.g., albumin-to-creatinine ratio) to better capture trends in renal function [42]. Finally, since we exclusively used a Chinese cohort, validation across diverse ethnic populations is needed to confirm generalizability.
This study demonstrates that a lower eGFR and higher bSS are significant predictors of MACEs after PCI in patients with ACS. It also suggests that coronary lesion complexity partially mediate the relationship between renal dysfunction and adverse cardiovascular outcomes in ACS patients. Clinically, these findings underscore the importance of integrating assessments of kidney function with coronary anatomical evaluations to develop individualized treatment strategies for high-risk patients.
The datasets used and analyzed in the study are available from the corresponding author upon reasonable request.
QC and YKL drafted the manuscript, and were major contributors in the collection, analysis and interpretation of data. SQY, HMH, YYX, WW, LW, YXG, and LC were major contributors in the acquisition and interpretation of data and contributed to revision of the manuscript. SQX and JGZ designed the study and provided constructive suggestions for revisions of the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The ethics committee of the Third People’s Hospital of Chengdu approved this study (ethical approval number [2019S-67]). The study was carried out in full compliance with the Declaration of Helsinki. Informed consent was obtained from all participants in written.
The authors would like to thank all the participants.
This work was supported by National High Level Hospital Clinical Research Funding (2024-NHLHCRF-JBGS-WZ-06), Capital’s Founds for Health Improvement and Research (No. 2022-1-4062), National High Level Hospital Clinical Research Funding (2024-NHLHCRF-YS-01), Chinese Society of Cardiology’s Foundation (No. CSCF2021B02), National Key Clinical Specialty Construction Project (No. 2020-QTL-009), and Chengdu High-level Key Clinical Specialty Construction Project.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM38389.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





