1 Department of Nursing, Zhongshan Hospital Fudan University, 200032 Shanghai, China
2 Cardiac Intensive Care Center, Zhongshan Hospital Fudan University, 200032 Shanghai, China
3 Department of Nursing, Zhongshan Hospital Fudan University Xiamen Branch, 361015 Xiamen, Fujian, China
4 Department of Biostatistics, Zhongshan Hospital Fudan University, 200032 Shanghai, China
5 Department of Critical Care Medicine, Zhongshan Hospital Fudan University, 200032 Shanghai, China
6 Shanghai Key Laboratory of Lung Inflammation and Injury, Zhongshan Hospital Fudan University, 20032 Shanghai, China
†These authors contributed equally.
Abstract
Pleural effusion (PE) commonly occurs in cardiac surgery patients, often requiring tube drainage. This study aimed to investigate associations between PE drainage trajectories and clinical outcomes in patients undergoing cardiac surgery.
Patients who underwent cardiac surgery and subsequent tube drainage during hospitalization in the intensive care unit, due to substantial PE, were enrolled. PE drainage volumes were recorded daily. The relationships between PE drainage and poor outcome or mortality risks were examined using logistic regression analysis. Latent class growth analysis (LCGA) was used to classify PE trajectories, and the characteristics of each latent class were compared.
In total, 386 patients were enrolled over 3 years, of whom 113 (29.3%) developed poor outcomes. These patients had significantly higher average PE drainage volumes on days 2–4 (1.7 vs. 1.2 mL/kg/day; p = 0.002) and days 5–7 (0.9 vs. 0 mL/kg/day; p < 0.001). Average PE drainage volumes during the first 2–4 and 5–7 days were associated with poor outcomes (odds ratio (OR) = 1.10 (95% confidence interval (CI): 1.02–1.20); p = 0.014 and 1.19 (95% CI: 1.08–1.32); p < 0.001, respectively). LCGA identified three distinct PE drainage trajectory classes: persistently high (Class 1, n = 39), gradually declining from high to low (Class 2, n = 128), and persistently low (Class 3, n = 219). Among these, Class 1 had the highest mortality and poor outcome risks.
A trend in PE formation demonstrated a strong correlation with mortality and poor outcomes in patients who underwent cardiac surgery. Patients with persistently high PE drainage volumes required close monitoring and attention.
Keywords
- pleural effusion
- thoracentesis
- mortality
- cardiac surgical procedures
Pleural effusion (PE) is a highly prevalent complication in patients following cardiac surgery, with some medical institutions reporting incidences exceeding 60% [1, 2, 3]. PE is classified into early and late categories based on the time of onset, with early PE occurring within the first postoperative month and accounting for the majority of PE cases after cardiac surgery [4]. PE etiology in this population is multifactorial, including pleural inflammatory reactions induced by cardiopulmonary bypass and surgical trauma, coagulopathy secondary to hemodilution, hypoalbuminemia, and most crucially, elevated capillary hydrostatic pressure resulting from cardiac congestion or suboptimal volume management [2, 4, 5, 6]. Moreover, PE is a significant factor contributing to intensive care unit (ICU) re-admission and poor prognosis outcomes [1, 7].
Minimal pulmonary effusion generally does not warrant further clinical intervention. Conversely, significant pulmonary effusion may induce hypoxia, necessitating thoracentesis to ensure effective drainage [8, 9, 10, 11]. This intervention serves two purposes: it alleviates pulmonary compression and provides an indirect way to assess hydrostatic equilibrium. For the majority of patients, maximum PE volumes are usually drained on the first day. Nevertheless, persistent PE production may require repeated daily drainage.
At present, no studies have analyzed the quantitative relationship between early daily PE drainage trajectories and clinical outcomes. We hypothesize that PE drainage volumes after cardiac surgery are associated with clinical prognosis outcomes, and patients with different PE drainage trajectories have different clinical outcomes. To verify this, we conducted this study.
This study was part of a cohort study investigating risk factors after cardiac
surgery (approved by the ethics committee of Zhongshan Hospital, Fudan
University: B2019-075R). From January 2019 to June 2022, in our 40-bed cardiac
surgical ICU, patients who underwent thoracentesis (either concurrent bilateral
or unilateral) and catheter drainage for large, early PE volumes were included.
Inclusion criteria were as follows: (1) undergoing elective cardiac surgery; (2)
complete clinical data. Patients were excluded if they met any of the following
criteria: (1) aged
Generally, we identified potential patients with large PE volumes using chest radiographs and then assessed volumes using bedside ultrasound. When PE was detected, clinicians first assessed whether it was due to active surgical bleeding which required surgical intervention. Drainage was only performed after excluding the surgical cause of bleeding. Thoracentesis and catheter placement procedures followed in-house protocols [13]. Drainage catheters were either single-lumen central venous (Model CS-24301-E, 16 Ga; Arrow International, Žďár nad Sázavou, Czech Republic; Lot 7175240491; CE 2797) or specialized pigtail drainage catheters (Model DC-0620, 6/8 Fr; Danong Medical, Weifang, China; NMPA Reg. No. 20173140217; Lot C324291204). For patients receiving respiratory support with positive end-expiratory pressure effects, such as high-flow oxygen therapy and non-invasive or invasive mechanical ventilation, the drainage volume was not restricted [14]. Otherwise, drainage volume on the first day was limited to 1000 mL. The daily PE drainage volume was recorded by measuring the amount of bloody fluid in the drainage bag at 6:00 am each day.
We recorded daily PE drainage volumes for the first 7 consecutive days and
converted units to mL/kg/day. Additionally, average PE drainage volumes were
separately calculated for the first 2–4 and 5–7 days. If the patient had an
indwelled drain for
The primary endpoint was a composite endpoint defined as either hospital mortality or postoperative hospitalization exceeding 30 days (poor outcomes). Secondary outcomes included hospital mortality, length of ICU stay, and length of hospital stay.
The normality of continuous variables was assessed using the Kolmogorov-Smirnov
test. Data were presented as the mean
During the 3-year study period, 15,928 patients were admitted to our ICU after cardiac surgery, and 386 were included in the study. The cohort comprised of 236 (61.1%) males with a median age of 65 years (IQR = 56–71). Among patients, 40 (10.4%) died during hospitalization, 113 (29.3%) had poor outcomes, 83 (21.5%) underwent tracheostomy, and 80 (20.7%) required RRT support. Median ICU and hospital stays were 8 (IQR = 5–18) and 14 days (IQR = 10–27), respectively. Patients with poor outcomes had higher EuroSCOREs, lower preoperative left ventricular ejection fraction scores, higher preoperative creatinine and NT-proBNP levels before surgery, and also higher APACHE II scores, NT-proBNP levels, TB levels, and lower hemoglobin levels on the day of thoracentesis (Table 1).
| Overall | No poor outcome | Poor outcome | p value | ||
| (n = 386) | (n = 273) | (n = 113) | |||
| Preoperative condition | |||||
| Age, years | 65 [56–71] | 65 [55–71] | 65 [57–71] | 0.888 | |
| Men, n (%) | 236 (61.1) | 158 (57.9) | 78 (69.0) | 0.041 | |
| BMI, kg/m2 | 23 [21–25] | 23 [21–25] | 23 [21–25] | 0.782 | |
| Hypertension, n (%) | 189 (49.0) | 132 (48.4) | 57 (50.4) | 0.708 | |
| Diabetes mellitus, n (%) | 63 (16.3) | 44 (16.1) | 19 (16.8) | 0.866 | |
| CKD, n (%) | 24 (6.2) | 17 (6.2) | 7 (6.2) | 0.990 | |
| EuroSCORE | 5 [3–7] | 5 [3–7] | 6 [4–8] | 0.005 | |
| LVEF, % | 62 [51–65] | 62 [56–65] | 60 [45–65] | 0.010 | |
| Creatinine, µmol/L | 91 [73–121] | 88 [72–112] | 99 [79–138] | 0.002 | |
| NT-proBNP, ng/mL | 1154 [378–3123] | 918 [332–2616] | 1896 [661–4046] | ||
| Postoperative condition | |||||
| APACHE II score | 9 [7–14] | 9 [7–13] | 11 [8–16] | ||
| LVEF, % | 58 [49–62] | 58 [50–62] | 58 [46–63] | 0.755 | |
| NT-proBNP, ng/mL | 3431 [1471–7975] | 2895 [1325–6427] | 5292 [2441–12,044] | ||
| TB, µmol/L | 20 [13–32] | 18 [12–30] | 22 [15–41] | 0.003 | |
| Albumin, g/L | 35 [32–38] | 35 [32–38] | 35 [31–37] | 0.190 | |
| ALT, U/L | 21 [13–40] | 20 [13–35] | 25 [12–54] | 0.241 | |
| AST, U/L | 37 [22–64] | 36 [23–58] | 38 [20–91] | 0.629 | |
| Creatinine, µmol/L | 110 [83–171] | 108 [85–154] | 127 [82–200] | 0.147 | |
| Hemoglobin, g/L | 86 [76–99] | 88 [78–100] | 83 [74–98] | 0.008 | |
| Platelet, 109/L | 107 [73–156] | 106 [76–146] | 109 [62–178] | 0.853 | |
| Lactate, mmol/L | 1.6 [1.3–2.6] | 1.6 [1.3–2.5] | 1.7 [1.3–2.7] | 0.378 | |
| Procalcitonin, ng/mL | 0.74 [0.31–2.09] | 0.66 [0.30, 1.62] | 1.16 [0.32, 2.89] | 0.009 | |
| WBC, 109/L | 10.6 [8.2–13.6] | 10.5 [8.3–13.6] | 10.9 [8.0–13.8] | 0.772 | |
| pH | 7.44 [7.41–7.47] | 7.44 [7.41–7.46] | 7.44 [7.39–7.48] | 0.902 | |
| PaCO2, mmHg | 39 [35–43] | 39 [35–43] | 37 [34–42] | 0.039 | |
| PaO2, mmHg | 105 [81–147] | 103 [80–143] | 113 [84–151] | 0.173 | |
| Warfarin, n (%) | 304 (78.8) | 223 (81.7) | 81 (71.7) | 0.027 | |
| Aspirin, n (%) | 104 (26.9) | 73 (26.7) | 31 (27.4) | 0.886 | |
| Clinical outcomes | |||||
| Tracheotomy, n (%) | 83 (21.5) | 19 (7.0) | 64 (56.6) | ||
| RRT, n (%) | 80 (20.7) | 31 (11.4) | 49 (43.4) | ||
| NIV, n (%) | 142 (36.8) | 97 (35.5) | 45 (39.8) | 0.426 | |
| Hospital mortality, n (%) | 40 (10.4) | 0 (0.0) | 40 (35.4) | ||
| Length of ICU stay, day | 8 [5–18] | 6 [4–9] | 24 [13–39] | ||
| Length of hospital stay, day | 14 [10–27] | 12 [8–17] | 40 [32–56] | ||
| Pleural effusion drainage | |||||
| Day 1, mL/kg/day | 11.1 [7.2–16.4] | 10.6 [7.1–15.7] | 11.8 [7.3–17.3] | 0.151 | |
| Day 2, mL/kg/day | 2.0 [0.5–4.1] | 1.8 [0.5–4.1] | 2.2 [0.7–4.2] | 0.146 | |
| Day 3, mL/kg/day | 0.3 [0.0–1] | 0.3 [0–0.9] | 0.6 [0.1–1.4] | ||
| Day 4, mL/kg/day | 0.1 [0.0–0.9] | 0.0 [0.0–0.6] | 0.5 [0.0–1.5] | ||
| Day 5, mL/kg/day | 0.0 [0–0.6] | 0.0 [0.0–0.3] | 0.3 [0.0–1.1] | ||
| Day 6, mL/kg/day | 0.0 [0.0–0.2] | 0.0 [0.0–0.0] | 0.1 [0.0–1.0] | ||
| Day 7, mL/kg/day | 0.0 [0.0–0.1] | 0.0 [0.0–0.0] | 0.1 [0.0–0.8] | ||
| Day 2 to 4 mL/kg/day | 1.4 [0.4–3.1] | 1.2 [0.4–2.6] | 1.7 [1.0–3.8] | 0.002 | |
| Day 5 to 7, mL/kg/day | 0.0 [0.0–1.3] | 0.0 [0.0–0.4] | 0.9 [0.0–2.8] | ||
Values are median (IQR) or number of patients (n).
Poor outcome, defined as either hospital mortality or postoperative hospitalization exceeding 30 days. BMI, body mass index; CKD, chronic kidney disease; LVEF, left ventricular ejection fraction; EuroSCORE, European System for Cardiac Operative Risk Evaluation; APACHE II score, Acute Physiology and Chronic Health Evaluation II score; NT-proBNP, N-terminal pro-brain natriuretic peptide; TB, total bilirubin; ALT, alanine transaminase; AST, aspartate aminotransferase; WBC, white blood cell; RRT, renal replacement therapy; NIV, non-invasive ventilation; ICU, intensive care unit; IQR, interquartile ranges.
Most PE drainage processes (350 patients, 90.7%) were initiated within 48 h
after cardiac surgery, with 279 patients (72.3%) being unilateral (left side:
144 and right side: 135). The median drainage volume on day 1 was 11.1
(7.2–16.4) mL/kg/day, with no significant differences between outcome groups. On
day 2, the volume decreased to 2.0 (0.5–4.1) mL/kg/day, but differences remained
non-significant. From day 3 onwards, daily drainage volumes were significantly
higher in patients with poor outcomes (p = 0.001) (Table 1). Patients
had higher average drainage volumes in the first 2–4 days (1.7 vs. 1.2
mL/kg/day, p = 0.002) and the first 5–7 days (0.9 vs. 0 mL/kg/day,
p
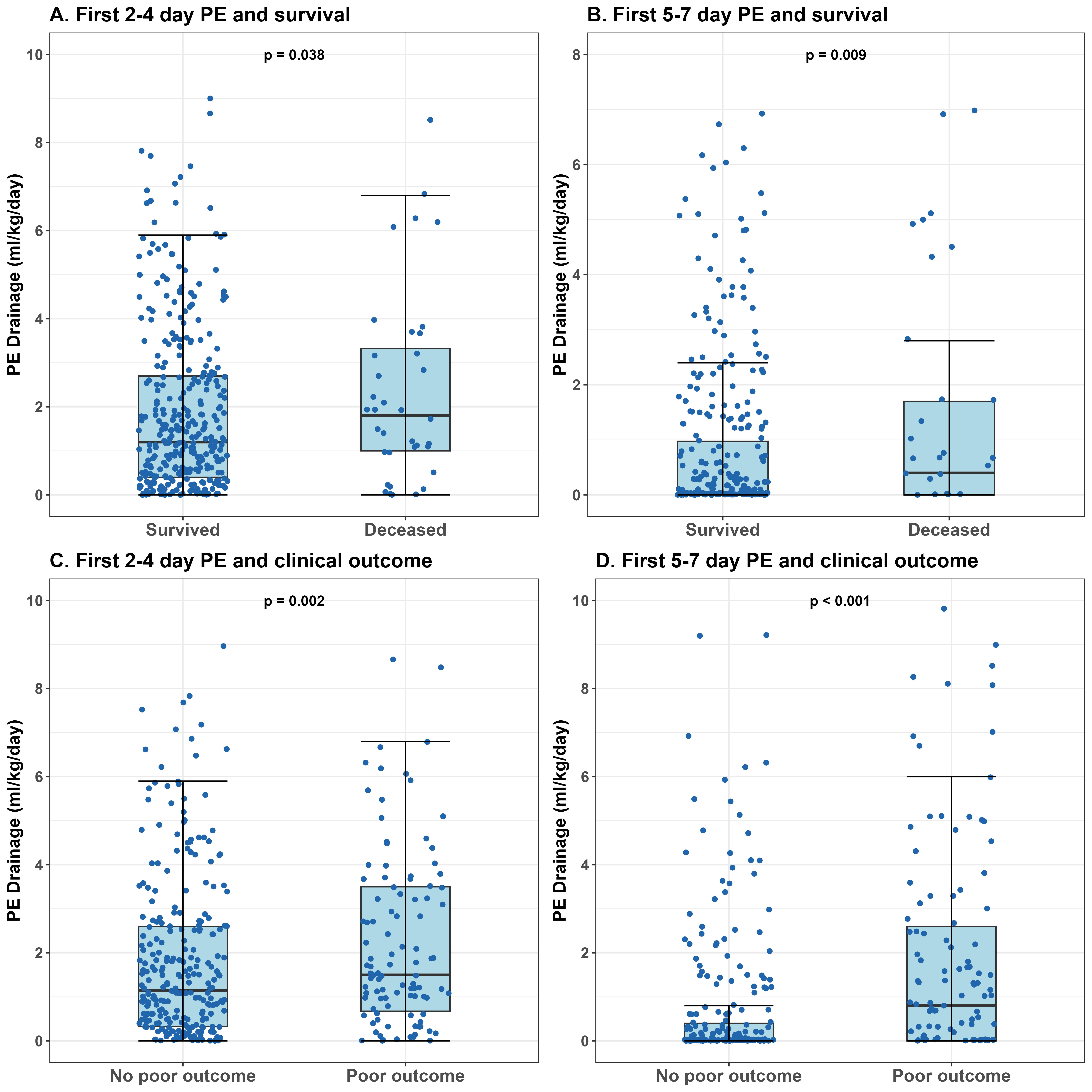 Fig. 1.
Fig. 1.
PE drainage volumes versus patient survival and clinical
outcomes. (A) Significantly higher PE drainage volumes were observed in deceased
patients compared to survivors during days 2–4 (p = 0.038). (B)
Significantly higher PE drainage volumes were observed in deceased patients
compared to survivors during days 5–7 (p = 0.009). (C) Significantly
higher PE drainage volumes were observed in patients with poor outcomes compared
to those without during days 2–4 (p = 0.002). (D) Significantly higher
PE drainage volumes were observed in patients with poor outcomes compared to
those without during days 5–7 (p
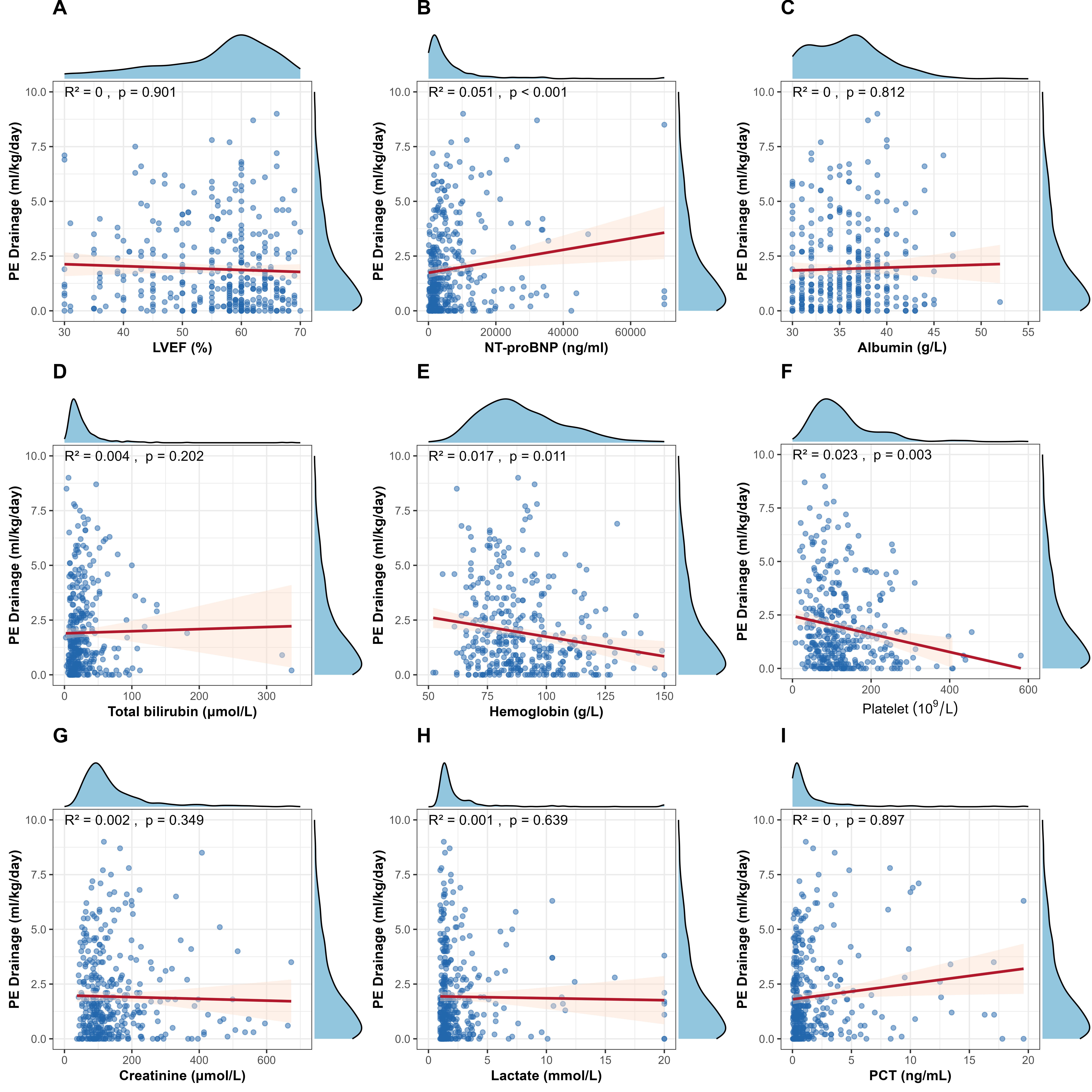 Fig. 2.
Fig. 2.
PE drainage volumes versus laboratory tests on the day of
thoracentesis. (A) PE drainage (mL/kg/day) versus LVEF (%) showed no
significant correlation (R2 = 0, p = 0.901). (B) PE drainage versus
NT-proBNP (ng/mL) showed a weak but statistically significant positive
correlation (R2 = 0.051, p
The first day’s PE drainage volumes showed no significant associations with
hospital mortality (adjusted odds ratio (OR) = 1.03 (95% confidence interval
(CI): 0.99–1.07), p = 0.170) or poor outcomes (adjusted OR = 1.00 (95%
CI: 0.98–1.03), p = 0.743) (Table 2). However, the average PE drainage
volume in the first 2–4 days showed a stronger, significant association with
poor outcomes (adjusted OR = 1.10 (95% CI: 1.02–1.20), p = 0.014)
(Table 2). This association was further increased in the first 5–7 days
(mortality: adjusted OR = 1.13 (95% CI: 1.01–1.26), p = 0.036 and poor
outcomes: adjusted OR = 1.19 (95% CI: 1.08–1.32), p
| Clinical outcomes | Pleural effusion | Crude OR | p value | Adjusted OR | p value |
| Death | Day 1 | 1.04 [1.01–1.08] | 0.023 | 1.03 [0.99–1.07] | 0.170 |
| Day 2 to 4 | 1.13 [1.04–0.23] | 0.006 | 1.09 [0.96–1.22] | 0.066 | |
| Day 5 to 7 | 1.17 [1.06–0.29] | 0.002 | 1.13 [1.01–1.26] | 0.036 | |
| Poor outcome | Day 1 | 1.02 [1.00–1.05] | 0.092 | 1.00 [0.98–1.03] | 0.743 |
| Day 2 to 4 | 1.15 [1.06–1.24] | 1.10 [1.02–1.20] | 0.014 | ||
| Day 5 to 7 | 1.25 [1.13–1.38] | 1.19 [1.08–1.32] |
Data are presented as true value (95% CI). OR, odds ratio.
Based on daily PE volumes from days 2 to 7 (after thoracentesis and adequate drainage), PE drainage trajectories were generated. LCGA was used to identify three distinct PE drainage trajectory classes: Class 1 (persistently high levels, n = 39), Class 2 (gradually declining from high to low levels, n = 128), and Class 3 (persistently low levels, n = 219) (Fig. 3). NT-proBNP levels decreased progressively from Class 1 to Class 3 (Table 3). Class 1 patients had the highest hospital mortality (20.5%) and poor outcome rates (53.8%), and the longest ICU (median (IQR) = 15 (9–27 days)) and hospital (IQR = 27 (16–40 days)) stays (Table 3). Although Class 2 patients had slightly higher incidence of poor outcomes than Class 3 patients, the differences were not statistically significant (p = 0.306).
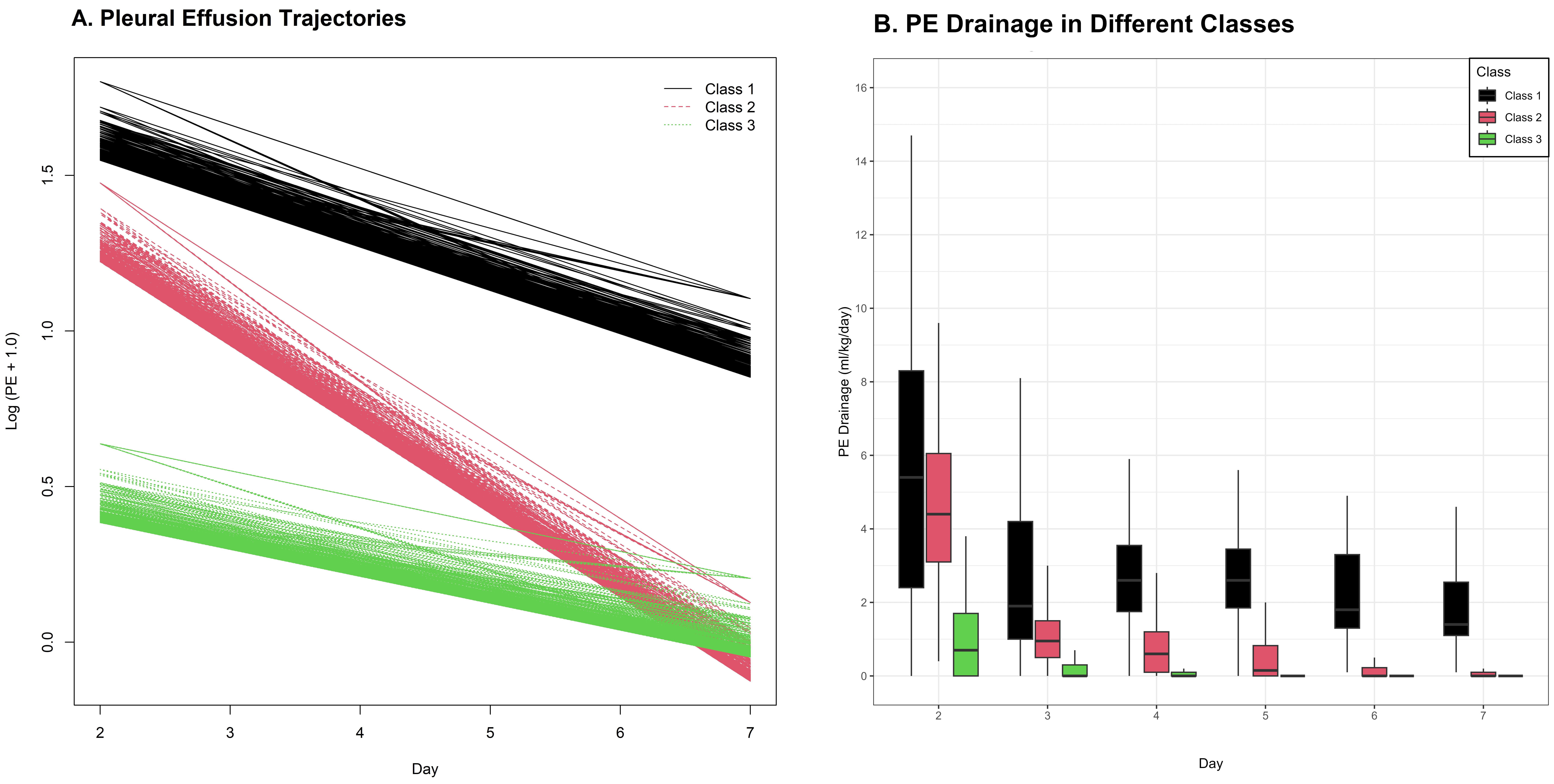 Fig. 3.
Fig. 3.
Three distinct PE drainage trajectory classes. (A) Three distinct PE drainage trajectory classes identified by latent class growth analysis (LCGA). (B) Box plots of daily PE drainage volume (mL/kg) stratified by trajectory class.
| Class 1 | Class 2 | Class 3 | p value | ||
| (n = 39) | (n = 128) | (n = 219) | |||
| Pleural effusion drainage | |||||
| Day 1, mL/kg/day | 20.0 [11.0, 25.6] | 14.3 [9.7, 19.6] | 8.8 [6.1, 12.9] | ||
| Day 2, mL/kg/day | 6.3 [2.5, 10.5] | 4.5 [3.2, 6.1] | 0.7 [0.0, 1.7] | ||
| Day 3, mL/kg/day | 1.9 [1.0, 4.2] | 1.0 [0.5, 1.5] | 0.0 [0.0, 0.3] | ||
| Day 4, mL/kg/day | 2.6 [1.8, 3.6] | 0.6 [0.1, 1.2] | 0.0 [0.0, 0.1] | ||
| Day 5, mL/kg/day | 2.6 [1.9, 3.5] | 0.2 [0, 0.83] | 0.0 [0.0, 0.0] | ||
| Day 6, mL/kg/day | 1.8 [1.3, 3.3] | 0.0 [0.0, 0.2] | 0.0 [0.0, 0.0] | ||
| Day 7, mL/kg/day | 1.4 [1.1, 2.6] | 0.0 [0.0, 0.0] | 0.0 [0.0, 0.0] | ||
| Day 2 to 4 mL/kg/day | 5.8 [4.2, 10.8] | 2.8 [2.1, 4.5] | 0.5 [0.1, 1.1] | ||
| Day 5 to 7, mL/kg/day | 5.5 [4.3, 8.1] | 0.3 [0.0, 1.4] | 0.0 [0.0, 0.1] | ||
| Lab examinations (on the day of thoracentesis) | |||||
| NT-proBNP, ng/mL | 5616 [2954, 11,650] | 4089 [1647, 8666] | 2799 [1326, 6706] | 0.002 | |
| TB, µmol/L | 28 [14, 44] | 21 [14, 29] | 18 [12, 32] | 0.059 | |
| Albumin, g/L | 35 [33, 38] | 36 [32, 38] | 35 [32, 38] | 0.545 | |
| ALT, U/L | 18 [12, 28] | 20 [12, 40] | 22 [14, 42] | 0.279 | |
| AST, U/L | 39 [21, 72] | 36 [22, 68] | 36 [23, 60] | 0.988 | |
| Creatinine, µmol/L | 107 [84, 181] | 109 [81, 165] | 112 [86, 171] | 0.503 | |
| Hemoglobin, g/L | 85 [75, 95] | 84 [75, 94] | 88 [79, 104] | 0.011 | |
| Platelet, 109/L | 90 [55, 138] | 102 [70, 141] | 113 [77, 164] | 0.046 | |
| Lactate, mmol/L | 1.5 [1.4, 3.1] | 1.7 [1.3, 2.8] | 1.6 [1.3, 2.4] | 0.573 | |
| Procalcitonin, ng/mL | 0.86 [0.38, 2.30] | 0.61 [0.30, 1.77] | 0.85 [0.31, 2.42] | 0.183 | |
| WBC, 109/L | 9.7 [7.6, 13.7] | 10.6 [8.6, 13.3] | 10.6 [8.3, 14.5] | 0.612 | |
| Clinical outcomes | |||||
| Hospital mortality, n (%) | 8 (20.5) | 14 (10.9) | 18 (8.2) | 0.065 | |
| Length of hospital stay, day | 27 [16, 40] | 14 [10, 27] | 13 [8, 24] | ||
| Length of ICU stay, day | 15 [9, 27] | 8 [5, 16] | 7 [4, 15] | ||
| Poor outcome, n (%) | 21 (53.8) | 38 (29.7) | 54 (24.7) | 0.001 | |
Values are median (IQR) or number of patients (n).
In this study, we screened more than 15,000 patients, of which 386 had PE with at least one puncture and drainage issue. The most significant conclusion was that increased continuous thoracic drainage was significantly associated with poor patient outcomes after cardiac surgery. To our knowledge, this is the first study to explore the relationship between PE drainage volume (and associated dynamic trajectories) with prognoses in patients undergoing cardiac surgery.
We evaluated associations between PE dynamics and patient outcomes from two perspectives. First, regarding pleural fluid production, higher mean daily PE volumes during postoperative days 2–4 and 5–7 were significantly associated with worse clinical outcomes, indicating that increased fluid production correlated with a poorer prognosis. Second, an analysis of PE production trends revealed that a downward trajectory—whether rapid (resulting in persistently low levels) or gradual—was associated with improved outcomes. In contrast, patients with persistently high PE volumes over 1 week had higher mortality rates and a greater incidence of composite endpoint events. Correlation analyses between PE production rates and cardiac function indices suggested that both the magnitude and duration of postoperative PE potentially reflected underlying cardiac function. In patients with impaired cardiac function, prolonged and elevated PE may have indicated either a lack of therapeutic benefit from the surgical intervention or a sustained procedure-related injury. Notably, these differences in PE dynamics became apparent by postoperative day 3 following thoracentesis and were predictive of subsequent clinical outcomes.
Despite limited studies on PE trajectory, several studies have examined PE causes after cardiac surgery. In Labidi et al. [4], patients with very early PEs were hemorrhagic, had higher neutrophil counts, and elevated lactate dehydrogenase levels, and the authors suggested that early effusions were possibly due to pleura trauma, whereas late effusions were more likely due to immune-inflammatory processes related to postcardiac injury syndrome. Pleurotomy can impair the pleura to resorb pleural fluid and may also lead to inflammation contributing to PE formation, consistent with previous findings showing that pleural injury and immunological perturbations were two important PE formation mechanisms after cardiac surgery [15].
Although pleural injury is known to contribute to PE following cardiac surgery, interindividual differences in PE formation rates and duration may also be influenced by other factors. We hypothesize that variations in postoperative cardiac function, particularly left ventricular performance, may have key roles. In heart failure, the primary mechanism underpinning pleural fluid accumulation involves transudation from the pulmonary interstitium into the pleural space. Elevated left atrial and left ventricular end-diastolic pressures are retrogradely transmitted to the alveolar capillaries, promoting fluid leakage and subsequent PE formation. Elevated hydrostatic pressure in these capillaries increases interstitial fluid in the lungs [16]. The fluid then moves from the pulmonary interstitial space across the visceral pleura into the pleural space along a pressure gradient. Pleural fluid accumulates when its formation rate exceeds parietal lymphatic capacity to absorb it.
In this study, we hypothesized that in early PE stages, cardiac insufficiency or
excessive circulation load may have triggered PE generation mechanisms. This
caused different PE generation rates in different patients from day 3 after
pleural puncture and drainage. One observation that could support this hypothesis
are NT-proBNP levels. In recent years, circulating natriuretic peptide levels
have become useful adjunctive tools in heart failure (HF) diagnostics [17]. Both
BNPs and NT-proBNPs are secreted almost exclusively from ventricles in response
to pressure and volume overload. Overall, as BNP or NT-proBNP levels increase,
the likelihood of HF also increases. In our study, we did not test for BNP in
pleural fluid, but collected serum NT-proBNP data. NT-proBNP levels were high in
the “persistently high” trajectory group (median (IQR) = 5616 (2954–11,650
ng/mL)) and were associated with mean daily pleural fluid production on days
2–4. Although our analysis revealed a statistically significant correlation
between PE drainage volume and serum NT-proBNP levels (R2 = 0.051,
p
In our study, another indicator closely related to PE generation was platelet levels. Similar to NT-proBNP, platelets were inversely correlated with average daily PE volumes from 2 to 4 days. Although a statistically significant but weak inverse correlation was observed (R2 = 0.023, p = 0.003), suggesting that platelet levels may partially reflect disease severity and surgical trauma intensity in PE patients, the minimal explained variance indicates that other pathophysiological factors likely play more predominant roles in determining drainage volumes. In the “persistently high” trajectory group, platelet levels were significantly lower, and levels were highest in the “persistently low” control group. Cardiopulmonary bypass (CPB) used in cardiac surgery may decrease platelet counts in initial phases [18, 19, 20]. Postoperative thrombocytopenia may also occur due to CPB hemodilution and mechanical damage to platelets [21]. In Keles et al. [22], pump and cross-clamp times were negatively correlated with platelet counts. Beyond baseline fibrinolysis and inflammatory stimuli associated with blood exposure to cardiopulmonary bypass circuits, high-dose systemic heparin exposure may further increase heparin induced thrombocytopenia perioperative risks [23, 24]. For patients with normal preoperative platelet counts, postoperative decreases may not be necessarily related to cardiac function, but lower platelet counts mean that patients may have higher bleeding risks. In our study, at the time of PE, patients with lower platelet counts had greater pleural fluid production. Therefore, platelet counts may be contributing factors to PE generation and amplify PE effects caused by decreased cardiac function and pleural injury.
This study had several limitations. First, as a single-center, retrospective observational study, the generalizability of our findings may be limited, and the decision to perform thoracentesis and catheter drainage, based on attending physician judgement, may have introduced selection bias. Second, we analyzed PE drainage volumes for only the first 7 days after cardiac surgery, without assessing the long-term impact on patient outcomes or the biochemical/cellular composition of drained PE volumes, which may have highlighted underlying pathophysiology mechanisms. Finally, despite adjusting for potential confounders in logistic regression analysis, residual confounding factors may still have existed due to the observational nature of the study.
PE formation trends showed a strong correlation with mortality and poor outcomes in patients who underwent cardiac surgery. Patients with persistently high PE drainage volumes represent a population that require close monitoring and attention.
The datasets generated and analyzed during the current study are not publicly available due to restrictions imposed by the hospital’s institutional policies. However, they can be made available from the corresponding author upon reasonable request.
JZ, JCL, GWT, YX—Conception and design; YX, GWT—Provision of study material or patients; JZ, JCL, JLL, GWT, MHL—Collection and assembly of data; JZ, JCL, JG, KL, GWT—Data analysis and interpretation; all authors—Manuscript writing. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Zhongshan Hospital, Fudan University (B2019-075R). Individual consent for this retrospective analysis was waived.
Thanks to all the research participates who provided data for this study.
This article was supported by the Science and Technology Commission of Shanghai Municipality (20DZ2261200).
The authors declare no conflict of interest. Guo-wei Tu is serving as one of the Editorial Board members , Ming-hao Luo and Guo-wei Tu are serving as Guest Editors of this journal. We declare that Ming-hao Luo and Guo-wei Tu had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Michael Dandel.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



