1 Department of Cardiology, General Hospital Paphos, State Health Services Organization, 8026 Paphos, Cyprus
Abstract
Transcatheter aortic valve implantation (TAVI) is a minimally invasive procedure to treat severe aortic stenosis in select patients. Patients who have undergone TAVI are at high risk of infective endocarditis (IE), especially during the first year post-operation. Early diagnosis of IE is essential to initiate targeted antibiotic therapy and/or surgical intervention. However, the early detection of IE following TAVI poses significant diagnostic challenges. Current imaging techniques, including echocardiography, nuclear imaging, and magnetic resonance imaging, have varying degrees of sensitivity and specificity, each with inherent limitations. Nuclear imaging modalities, such as positron emission tomography/computed tomography using 18F-fluorodeoxyglucose (18F-FDG PET/CT) and white blood cell single photon emission computed tomography/computed tomography (WBC SPECT/CT), have shown promise in early IE detection, particularly due to the ability of these methods to identify metabolic and anatomical abnormalities. However, false-positive results related to post-operative inflammation complicate data interpretation, and limited data exist for using these methods in very early IE detection post-TAVI. Intracardiac echocardiography (ICE) offers enhanced visualization of prosthetic valve leaflets, but the invasive nature of ICE restricts its widespread use. Whole-body imaging, such as 18F-FDG PET/CT, facilitates the identification of distant lesions and systemic complications, aiding diagnosis and treatment decisions. Diagnosing IE after TAVI is especially challenging within the first 60 days post-procedure, a critical period when imaging findings may be inconclusive due to false negatives or limited availability of advanced modalities. This review underscores the diagnostic complexity of very early and early (0–60 days) IE post-TAVI, emphasizing the need for a multimodal imaging approach to overcome the limitations of individual modalities. Nonetheless, early antimicrobial therapy should be considered even without definitive imaging findings, highlighting the importance of clinical vigilance in managing this challenging condition.
Keywords
- very early post-TAVI infective endocarditis
- early post-TAVI infective endocarditis
- post-TAVI infective endocarditis
Transcatheter aortic valve implantation (TAVI) has emerged as a vital, minimally
invasive intervention for severe symptomatic aortic stenosis, particularly in
older patients (aged
Infective endocarditis (IE) refers to the inflammation of the endocardium, which is the inner lining of the heart. The intact, healthy endocardium is typically resistant to bacterial seeding [7]. However, the development of infectious endocarditis necessitates initial endocardial injury, followed by bacteremia [8]. Among the serious complications associated with TAVI, prosthetic valve endocarditis (PVE) poses a considerable risk, with studies revealing a one-year incidence of IE ranging from 0.2% (in the PARTNER 3 trial) to 3.1% (in a Danish cohort of 509 patients) [9, 10]. Both patient-related and procedural-related risk factors significantly contribute to the development of IE following TAVI [11].
The timing of IE post-TAVI, concerning the procedure, can be categorized as very early (within 30 days), early (within 30–60 days), intermediate (between 60 and 365 days), or late (after 365 days) [12]. Nonetheless, accurate diagnosis is crucial, as it dictates the initiation of targeted antibiotic therapy and potential surgical intervention [13]. However, diagnosing IE in TAVI patients can be particularly challenging due to the elusive nature of vegetation and the need for advanced imaging techniques [14]. The European Society of Cardiology (ESC) has modified diagnostic criteria incorporating additional imaging modalities to aid this endeavor [15, 16]. Meanwhile, imaging techniques such as transthoracic (TTE) and transesophageal echocardiography (TEE/TOE) remain the cornerstone for initial evaluation. However, additional modalities, including computed tomography (CT), cardiac computed tomography angiography (CTA), magnetic resonance imaging (MRI), and nuclear imaging such as positron emission tomography/computed tomography using 18F-fluorodeoxyglucose (18F-FDG PET/CT) and single photon emission tomography/computed tomography with white blood cell (WBC SPECT/CT) labeling, are becoming increasingly utilized. Furthermore, intracardiac echocardiography (ICE) is emerging as a valuable tool in this context. Despite modifications, diagnosing very early and early IE after TAVI remains challenging.
Given the elevated in-hospital and 30-day mortality rates associated with IE, largely attributable to advanced age and multiple comorbidities, early suspicion and diagnosis are paramount [17]. Antimicrobial therapy for IE post-TAVI is similar to that of PVE. Therefore, in cases where PVE is highly suspected, it is vital to initiate appropriate antibiotic treatment without delay [18]. Additionally, when complications arise from IE, surgical intervention is regarded as the optimal management strategy [15].
This review focuses on the diagnostic challenges of very early and early onset IE following TAVI, moving beyond a mere outline of diagnostic and therapeutic strategies. It emphasizes the critical role of specific imaging modalities in detecting IE in patients who have recently undergone TAVI, while addressing the current limitations inherent with these approaches.
IE is primarily diagnosed based on the modified major and minor Duke criteria.
The major criteria include positive blood cultures and evidence of endocardial
involvement with echocardiography; meanwhile, the minor criteria encompass
predisposing heart conditions or injection drug use, fever with a temperature
Patients with IE post-TAVI may exhibit vague complaints and nonspecific symptoms, leading to delays in diagnosis and appropriate management [21]. Fever (71.7%) and heart failure (58.5%), along with nonspecific symptoms such as malaise and fatigue, are among the most common symptoms of IE post-TAVI [22, 23]. Specific symptoms resulting from septic emboli include neurological symptoms; meanwhile, the less commonly observed symptoms include cutaneous stigmata, local chills, loss of appetite, macrophage activation syndrome, and limb ischemia due to septic emboli [23]. Although the modified Duke criteria demonstrate high sensitivity for diagnosing native valve endocarditis (NVE), the sensitivity of these criteria is reduced in cases of PVE [24]. In a comparative study of Duke criteria and those of the ESC, which incorporate multimodal imaging, the Duke criteria exhibited a sensitivity of only 50%. In contrast, the modified ESC criteria exhibited a sensitivity of 100% [25]. A summary of the modified ESC criteria can be found in Appendix Table A1, adapted from Delgado et al. [15].
TAVI entails an elevated risk of IE. The incidence of IE post-TAVI ranges from 0.3 to 1.9 per 100 patient–years, with one recent study reporting a lower incidence of IE post-TAVI at approximately 3–10 per 1000 patient–years [15, 26]. This risk is particularly elevated in the first year post-procedure, with a notable increase within the initial 3 months [27, 28]. Both patient-related and procedural-related risk factors significantly contribute to developing IE following TAVI [11].
Patient-related factors, such as younger age, male sex, and comorbidities, including diabetes mellitus, chronic obstructive pulmonary disease, chronic kidney disease, peripheral arterial disease, and an elevated body mass index, all contribute to increased risk [29, 30]. Indeed, a recent meta-analysis comprising over 68,000 patients revealed that older age was linked to a significantly reduced risk of IE post-TAVI [31]. One proposed explanation is that younger patients undergoing TAVI exhibit more pronounced comorbidities compared to their older counterparts [28]. Meanwhile, the lower occurrence of IE post-TAVI in females has been attributed to the protective effects of estrogen on the vascular endothelium [32]. However, further studies are needed to examine the underlying mechanisms thoroughly. While most studies identify male sex as a risk factor for IE post-TAVI, a recent retrospective cohort study showed a similar occurrence between the two genders [29].
Procedural-related factors, such as orotracheal or nasotracheal intubation, heighten the risk of IE post-TAVI due to potential bacteremia following intubation [33]. Additional procedural-related risk factors include residual aortic regurgitation, significant paravalvular leak, and high-degree atrioventricular block post-TAVI, necessitating pacemaker implantation [14, 24, 34].
It is also important to thoroughly discuss the risk factors specific to very early IE post-TAVI since this clinical entity is more challenging to diagnose early, and limited literature is available on the subject. A recent retrospective observational study suggests that female gender is a risk factor, potentially because women undergoing TAVI are often older, frailer, and more immunosuppressed [35, 36, 37, 38]. Furthermore, self-expanding valves have been identified as a risk factor, likely due to their larger stent frame and the fact that their delivery systems often come into direct contact with the skin before valve deployment. This contrasts with balloon-expandable valves, which are always inserted within an external introducer sheath. Sepsis remains a risk factor as a periprocedural complication. Additionally, the occurrence of stroke may increase risk, as this condition requires extensive nursing care and leads to longer hospital stays, thereby heightening exposure to potential pathogens [38].
Recent studies have shown that mortality rates for IE post-TAVI are higher compared with NVE and SAVR-IE. More specifically, 1-year mortality rates for IE post-TAVI range from 40% to 45.6%, while the rates for SAVR-IE range from 23.1% to 32.8%, and for NVE, the rates are approximately 15% to 30% [39, 40, 41, 42, 43]. It is important to highlight that very early IE is associated with the highest mortality rate among patients with IE post-TAVI [38]. However, the mortality rate of IE can also be influenced by microorganisms, with certain organisms being associated with higher mortality rates compared to others [44, 45].
Indeed, Gram-positive bacteria are frequently implicated in most reports detailing IE post-TAVI [46]. According to a recent systematic review and meta-analysis, Streptococcus spp. (25.3%), Staphylococcus spp. (25.3%), and Enterococcus spp. (24.1%) were the most commonly identified pathogens causing IE after TAVI [47]. Notably, Staphylococcus aureus and Enterococcus faecalis were the dominant species, comprising 60% and 65.8% of cases, respectively [48]. Interestingly, Staphylococcus aureus, most commonly methicillin-resistant, represented the primary pathogen in very early IE post-TAVI, followed by enterococci organisms as the second most common pathogens [38]. The predominance of Staphylococcus aureus in very early IE post-TAVI is due to its natural colonization of the skin, its role as a healthcare-associated pathogen, procedural contamination risks, inadequate prophylactic antibiotic coverage, and its biofilm-forming ability on prosthetic surfaces. Therefore, addressing this issue may involve improving perioperative aseptic protocols, re-evaluating antibiotic prophylaxis to include coverage for MRSA, and adopting strategies to minimize procedural contamination [38].
Before we thoroughly analyze the imaging techniques, it is worth mentioning that transcatheter valve prostheses contain leaflets with a higher metal quantity in the stent frame compared to surgical valves [32]. However, no vegetation may be detected in 38–60% of cases [14]. Vegetations are typically located in the stent frame of the transcatheter valve, rather than on the valve leaflets, in 12% of cases. This rate increases to 19% in the presence of some self-expanding valve systems with a longer stent frame that occupies the ascending aorta [49]. Additionally, vegetations are found outside the transcatheter valve in about one-third of cases, primarily at the mitral valve level [14]. Table 1 summarizes the imaging techniques, their indications, strengths, and challenges for diagnosing infective endocarditis post-TAVI.
| Imaging technique | Indications | Strengths | Challenges |
| TTE/TOE | Primary imaging modality for diagnosing IE post-TAVI. | ||
| Cardiac CT | Assessing complications, such as abscesses or fistulas, in IE post-TAVI. | ||
| CTA | Evaluation of vegetations, mycotic aneurysms, abscesses, and paravalvular leakage. | ||
| 18F-FDG PET/CT(A) | Detecting early-stage infection, monitoring metabolic activity in IE post-TAVI. | ||
| (99mTc-HMPAO) SPECT/CT | When FDG-PET/CT is unavailable or unreliable due to early post-operative inflammatory changes. | ||
| Superior specificity in diagnosing PVE compared to 18F-FDG PET/CT. | |||
| MRI | Assessing valve function and detecting abscesses or thrombus post-TAVI. | ||
| ICE | Detection of early or very early-stage vegetations and complications post-TAVI. | ||
99mTc-HMPAO, technetium-99m-hexamethylpropyleneamineoxime-labeled leukocytes; MRI, magnetic resonance imaging; ICE, intracardiac echocardiography; 18F-FDG PET/CT, positron emission tomography/computed tomography using 18F-fluorodeoxyglucose; TAVI, transcatheter aortic valve implantation; TTE, transthoracic echocardiography; TOE, transesophageal echocardiography; TEE, transesophageal echocardiography; PVE, prosthetic valve endocarditis; IE, infective endocarditis; CTA, computed tomography angiography; SPECT, single photon emission computed tomography.
Echocardiography serves as the primary imaging modality for diagnosing IE post-TAVI, regardless of the timing of the disease, whether it is very early, early, intermediate, or late. Meanwhile, for NVE, TTE demonstrates a sensitivity of approximately 71% and a specificity of 80% [50]. In contrast, TEE offers superior diagnostic accuracy, with sensitivities ranging from 87% to 100% and specificities between 91% and 100% [51, 52, 53]. However, in cases of PVE, the sensitivity of TTE diminishes to 36–69%, while TEE maintains a higher sensitivity, reported between 86% and 100% [51, 54]. However, using TTE and TEE to detect small vegetations or paravalvular abscesses is challenging due to the shadowing effect and reflectance of prosthesis material. Moreover, detecting incipient vegetation in the free space between the transcatheter and native aortic valve, amidst extensive calcifications and prosthetic material, is particularly challenging using TTE and TEE, if even possible [55]. TTE and TEE may also be unable to differentiate between vegetations and fibrous strands or thrombi [28]. Fig. 1a,b illustrates the concepts mentioned above.
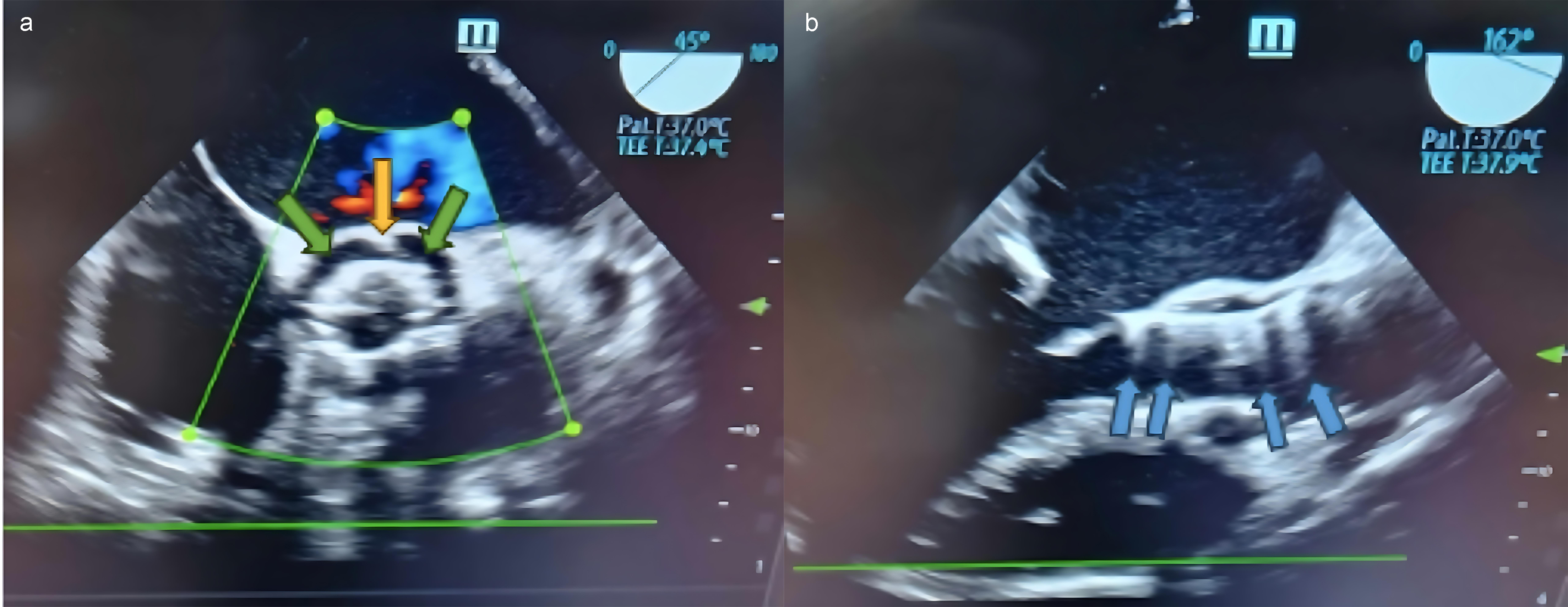 Fig. 1.
Fig. 1.
Infective endocarditis of a prosthetic aortic valve. (a) Mid-esophageal aortic valve short-axis view of vegetation at the 12 o’clock position (yellow arrow) in the stent frame of the transcatheter valve, along with a paravalvular leak (green arrows). (b) Mid-esophageal aortic valve long-axis view. Same patient. Blue arrows show the shadowing effect and the reflectance of the prosthetic valve. TEE, transesophageal echocardiography.
Despite these challenges, while TTE is recommended as the initial imaging procedure for all patients suspected of having NVE or PVE, it does not hold the status of the gold standard [56]. In all cases of alleged PVE, TEE remains essential for valvular hemodynamics assessment and the potential detection of vegetation, abscess, or fistula. To conclude, TEE is strongly recommended in patients with an inconclusive TTE, in those with a negative TTE and a high suspicion of IE, in all patients with clinical suspicion when a prosthetic valve or an intracardiac device is present, as well as in patients with a positive TTE to document local complications [15]. It is worth mentioning that echocardiography is necessary in any case of bacteremia with Staphylococcus aureus [15]. Moreover, the echocardiographic examination must be repeated after five to seven days in case of unconfirmed but highly suggestive PVE [56].
Cardiac CT is another imaging modality that can assist in diagnosing IE
following TAVI, regardless of the timing of the disease, by providing superior
visualization of its complications. Cardiac CT is superior to TEE in accurately
diagnosing perivalvular and periprosthetic complications of IE. Moreover, cardiac
CT may be useful in identifying these crucial complications in cases where a
paravalvular abscess, pseudoaneurysm, or fistulae is suspected with equivocal
findings from TEE [57, 58]. Nonetheless, TEE remains the gold standard for
detecting valvular lesions, particularly small vegetations (
CTA can be utilized as a diagnostic imaging technique for assessing IE post-TAVI, irrespective of the timing of onset, enabling the visualization of features such as vegetations, mycotic aneurysms, abscesses, paravalvular leakage, and valve dehiscence. However, recognizing the inherent challenges when interpreting valvular abnormalities on CTA remains crucial since artifacts from prosthetic stent materials may pose challenges in accurately assessing valvular irregularities, potentially resulting in false-negative outcomes. Furthermore, post-TAVI leaflet thrombosis, also known as hypoattenuating leaflet thickening (HALT), may occur even in patients receiving anticoagulation therapy, adding another layer of complexity to the interpretation. Indeed, the possibility of misinterpreting HALT as vegetation must be considered, as it can result in false-positive CTA findings [60].
It is also important to acknowledge that contrast-induced nephropathy is typically reversible, often presenting as a mild reduction in the glomerular filtration rate (GFR), which improves within three to seven days; most patients return to or near their baseline estimated GFR. However, individuals with advanced renal failure may require temporary dialysis following contrast administration [61]. Therefore, performing CTA can contribute to the diagnosis in cases where echocardiography provides equivocal findings.
18F-FDG PET/CT was incorporated into the ESC modified criteria to enhance sensitivity [15]. The most recent meta-analysis found that this imaging method had a sensitivity of 86% [62]. Thus, abnormal FDG uptake can be observed before the manifestation of infectious damage in echocardiography by assessing metabolic tissue activity. This underscores the capability of 18F-FDG PET/CT to detect infection before significant damage has occurred [63]. Increased metabolic activity in the heart can mimic the patterns seen in IE due to the inflammatory response following TAVI. This heightened metabolic activity can potentially lead to false-positive interpretations on 18F-FDG PET/CT scans [63]. While the ESC guidelines recommend performing 18F-FDG PET/CT three months after cardiac surgery to reduce the risk of false-positive results related to the post-operative inflammatory process [64], limited data exist for very early IE post-TAVI. Indeed, recent data suggest that 18F-FDG PET/CT can be accurately used at least one month after TAVI to diagnose IE post-TAVI, owing to the distinct FDG uptake patterns observed during this period. Specifically, circumferential or hemi-circumferential uptake represents a normal post-TAVI pattern, whereas focal or multifocal uptake strongly suggests definite IE–TAVI, enhancing diagnostic specificity [65].
Furthermore, 18F-FDG PET/CT imaging presents technical challenges, including adequate patient preparation. This typically involves fasting at least 6 hours before the procedure and heparin administration to minimize false-positive results and optimize imaging quality [66]. Additionally, it is crucial to ensure that the patient has proper blood glucose control, as elevated levels can interfere with the accuracy of the FDG uptake assessment. Moreover, PET/CT has limitations in detecting small foci during inflammation, which may result in false-negative findings, particularly in early-stage infections. Studies have also highlighted that PET imaging may not be as effective in detecting localized infection in patients with low-grade inflammatory responses [67].
Combining PET/CT acquisition with CT angiography (PET/CTA) enables the detection of both metabolic findings (such as 18F-FDG uptake distribution and intensity) and anatomical findings (related to infective endocarditis lesions) within a single imaging procedure [60, 68].
WBC SPECT/CT is an alternative nuclear imaging technique for diagnosing IE, when PET/CT is unavailable and in inexperienced centers, with a sensitivity of 64–90% [15]. WBC SPECT/CT, specifically using technetium-99m-hexamethylpropyleneamineoxime-labeled leukocytes (99mTc-HMPAO), is a highly specific imaging modality for diagnosing infections, including IE, even within the first month after TAVI. Moreover, WBC SPECT/CT can effectively differentiate between normal post-surgical inflammatory uptake and abnormal patterns indicative of infection. Studies have demonstrated its specificity—up to 100%—in early post-operative settings when other imaging methods, such as 18F-FDG PET/CT, may be less reliable due to nonspecific inflammatory changes. WBC SPECT/CT can be used to diagnose both intracardiac and extracardiac infections [69].
Thus, WBC SPECT/CT has emerged as a valuable tool in reducing the number of misdiagnosed cases of IE, previously classified as “possible IE” by the modified Duke criteria, with a significant decrease of 27% [70]. This imaging technique demonstrates high specificity in identifying active infective processes in perivalvular regions, and, as mentioned, shows superior specificity in diagnosing PVE compared to 18F-FDG PET [71]. However, it is essential to recognize that WBC accumulation may be diminished in certain conditions, such as in drained or encapsulated abscesses or non-pyogenic bacterial infections. This highlights the importance of using WBC SPECT/CT in the appropriate clinical context to maximize diagnostic accuracy [72].
While MRI is a common modality in diagnosing stroke and embolic events, its role in diagnosing IE is limited [15]. Especially in PVE, the value of MRI is low due to artifact interference. Nevertheless, in cases with high clinical suspicion of IE, cardiac MRI can provide valuable diagnostic and prognostic information by depicting features such as antegrade and retrograde dissemination, paravalvular tissue extension, and subendocardial and vascular endothelial involvement for delayed contrast-enhanced images [73]. Despite its potential, cardiac MRI is not a replacement for echocardiography but a complementary tool in specific scenarios. Additionally, the diagnostic potential of MRI is constrained by a lower spatial resolution than cardiac CT, and signal voids from certain prostheses may hinder the precise evaluation of the prosthetic valve anatomy and function [74].
ICE is a valuable tool for diagnosing IE post-TAVI, particularly in the early and very early stages. ICE presents a distinct advantage by directly visualizing prosthetic valve leaflets on the endocardial surface, overcoming challenges posed by acoustic shadowing from metallic stent frames. While TTE and TEE are commonly used, ICE provides real-time, high-resolution imaging, offering greater sensitivity and specificity when evaluating valvular and perivalvular structures [75]. In cases of early IE post-TAVI, ICE is particularly useful for identifying vegetations, abscesses, and complications such as valve perforations or pseudoaneurysms. Vegetations, a hallmark of IE, appear as oscillating or non-oscillating masses on the valve leaflets and are often better visualized using TEE or ICE compared to TTE, especially in the context of complex intracardiac structures, such as prosthetic valves [75].
Despite this, the major drawback lies in the invasive nature of the procedure. Typically performed via venous access to mitigate vascular complications, ICE becomes more complex in aortic valve prosthesis endocarditis, necessitating arterial access and elevating the risk of vascular complications. Additionally, the retrograde introduction of the ICE catheter across the aortic arch introduces potential risks such as aortic wall injury, dissection, or stroke. While theoretically possible, there have been no reported instances of embolization of vegetation by the ICE catheter, despite its proximity to the infected valve endocardial surface [76, 77].
In summary, ICE offers superior imaging for detecting and assessing the complications of IE early in the disease process. Moreover, ICE allows clinicians to accurately monitor patients and intervene promptly when other imaging modalities are limited.
Identifying distant lesions represents a minor diagnostic criterion, crucial for achieving a more conclusive, definite, or rejected IE diagnosis, and plays a significant role in guiding treatment decisions. Whole-body and brain CT scans offer valuable insights into systemic complications of IE, including septic emboli [78, 79]. MRI, boasting higher sensitivity than CT, excels in detecting neurological lesions, thereby improving the diagnosis of neurological complications in IE patients [79]. Moreover, whole-body 18F-FDG PET/CT imaging proves invaluable in suspected or confirmed IE cases, facilitating the identification of distant lesions and mycotic aneurysms and pinpointing the site of infection entry [60, 80].
Based on the aforementioned imaging techniques, a proposed algorithm for their use in diagnosing IE post-TAVI is presented in Fig. 2.
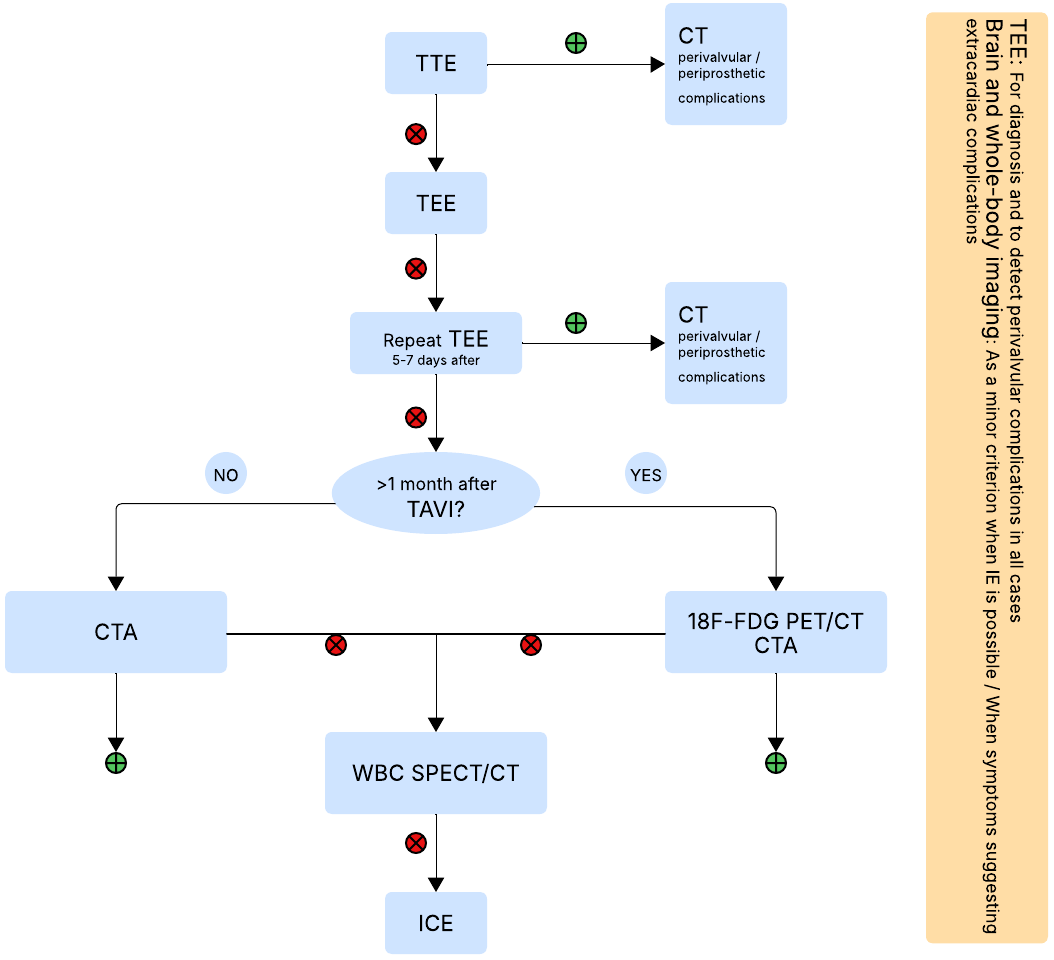 Fig. 2.
Fig. 2.
Proposed algorithm for diagnosis of IE post-TAVI. CT, computed tomography; 18F-FDG, 18F-fluorodeoxyglucose; PET, positron emission tomography; WBC SPECT, white blood cell single photon emission tomography; TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; IE, infective endocarditis; TAVI, transcatheter aortic valve implantation; ICE, intracardiac echocardiography; CTA, computed tomography angiography. Fig. 2 was created using draw.io.
The treatment approach for IE is multifaceted, involving both antimicrobial therapy and, in many cases, surgical intervention.
In suspected cases of IE, empirical antibiotic therapy should be initiated
promptly, even before blood culture results are available, to cover the most
likely pathogens. Considering the microbiological profile, initial empirical
treatment should encompass effective agents against methicillin-resistant
staphylococci and enterococci. Subsequently, therapy should be tailored to the
identified pathogens once blood cultures and sensitivities are available. The PVE
antibiotic treatment duration should last longer (
Surgery is necessary when complications such as heart failure, uncontrolled infection, established embolism, or a high risk of embolism are present. The timing of surgical intervention is crucial and should be individualized based on the clinical condition of each patient: emergency surgery within 24 hours, urgent surgery within 3–5 days, and non-urgent surgery within the same hospital admission. The decision to proceed with surgery in IE post-TAVI patients should be individualized, balancing the surgical risks and the prognosis of medical treatment alone [15]. Despite notably high in-hospital mortality rates, very few individuals with indications for surgery undergo the procedure. This reluctance may be attributed to the significant surgical risks associated with this older population, which often presents with multiple comorbidities [38]. Nevertheless, current evidence suggests that surgical treatment may provide up to a 20% survival advantage in the first year [81].
Diagnosing IE following TAVI presents challenges. The complexity of diagnosing infective endocarditis post-TAVI increases significantly when the condition manifests early after the TAVI procedure (0–60 days). Despite advanced imaging techniques and their diagnostic sensitivity, a diagnosis of IE in the initial diagnostic examination may remain possible rather than certain. This is due to falsely negative imaging findings presented by each modality, as well as the limited availability or use of certain imaging techniques.
This review addresses critical gaps in the diagnostic landscape of TAVI-related IE, focusing on the very early and early onset of this complication. While previous studies have primarily centered on clinical outcomes and management strategies, our work uniquely emphasizes the diagnostic challenges associated with the early detection of IE [82]. By highlighting the limitations of current imaging modalities, we advocate for a multimodal imaging approach that synthesizes recent advancements in the field. This perspective enriches the understanding of TAVI-related IE and offers practical insights for clinicians. Future research should continue to explore innovative imaging techniques and their integration into clinical practice to enhance early detection and improve patient outcomes in this rapidly evolving area.
In conclusion, early detection of IE following TAVI is vital, as delayed diagnosis can dramatically worsen patient outcomes. Although imaging techniques play a central role, their sensitivity is limited, particularly in the early stages of the disease. Therefore, maintaining a high index of clinical suspicion, prompting microbiological testing, and initiating broad-spectrum empirical antibiotic therapy, including coverage for MRSA, is essential for effective management. Enhancing diagnostic approaches and refining treatment protocols are critical steps toward reducing mortality and improving recovery rates in this high-risk patient population.
NH, DM, TEP, VD, VG, CS, AM contributed to the review and are responsible for the article content. All authors contributed to the conception and editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
See Appendix Table A1.
| Major criteria | ||
| 1. Blood cultures positive for IE: | ||
| A. Typical microorganisms consistent with IE from two separate blood cultures: | ||
| B. Microorganisms consistent with IE from continuously positive blood cultures: | ||
| C. Single positive blood culture for C. burnetii or phase I IgG antibody titer | ||
| 2. Imaging positive for IE: | ||
| Minor criteria | ||
| 1. Predisposing conditions (i.e., predisposing heart condition at high or intermediate risk of IE or PWIDs). | ||
| 2. Fevers with a temperature | ||
| 3. Embolic vascular dissemination (including those asymptomatic detected by imaging only): | ||
| 4. Immunological phenomena: | ||
| 5. Microbiological evidence: | ||
| IE classification | ||
| Definite ΙΕ: | ||
| 2 major criteria, or | ||
| 1 major criterion and at least 3 minor criteria, or | ||
| 5 minor criteria. | ||
| Possible ΙΕ: | ||
| 1 major criterion and 1 or 2 minor criteria, or | ||
| 3–4 minor criteria. | ||
| Rejected ΙΕ: | ||
| None of the criteria for definite or possible are met at admission with or without a firm alternative diagnosis. | ||
18F-FDG-PET/CT, positron emission tomography/computed tomography using 18F-fluorodeoxyglucose; CT(A), computed tomography (angiography); HACEK, Haemophilus, Aggregatibacter, Cardiobacterium, Eikenella, and Kingella; IE, infective endocarditis; Ig, immunoglobulin; PWIDs, people who inject drugs; TOE, transesophageal echocardiography; TTE, transthoracic echocardiography; WBC SPECT/CT, white blood cell single-photon emission computed tomography/computed tomography.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


