1 Department of Cardiac Surgery, Città di Lecce Hospital, GVM Care & Research, 73100 Lecce, Italy
2 Department of Clinical and Experimental Medicine, Magna Graecia University, 88100 Catanzaro, Italy
3 Department of Cardiac Surgery, Paracelsus Medical University, 90419 Nuremberg, Germany
4 Department of Clinical and Experimental Medicine, University of Messina, 98124 Messina, Italy
5 Department of Medicine and Surgery, University of Enna, 94100 Enna, Italy
6 Interdisciplinary Center for Health Sciences, Scuola Superiore Sant’Anna, 56127 Pisa, Italy
7 Department of Cardiology, Città di Lecce Hospital, GVM Care & Research, 73100 Lecce, Italy
8 Department of Cardiac Surgery, Anthea Hospital, GVM Care & Research, 70124 Bari, Italy
†These authors contributed equally.
§These authors contributed equally.
Abstract
The tricuspid valve (TV) is a complex three-dimensional (3D) anatomical structure; however, current guidelines recommend tricuspid annulus (TA) measurements to be performed with two-dimensional (2D) echocardiography. The aim of this study was to compare TV measurements obtained with 2D and four-dimensional (4D) echocardiography for surgical planning.
All echocardiographic data of patients referred to our center for TV assessment were collected. Multimodality imaging data were reviewed, including 2D transthoracic echocardiography (TTE) integrated with information from 3D TTE. Measurements were also compared with those obtained using the 4D Auto Tricuspid Valve Quantification (TVQ) tool.
Overall, 11 patients (median age 72 [66–78] years, 18% female) were included in the study. Mild, moderate and severe tricuspid regurgitation (TR) was present in 6, 3 and 2 patients, respectively. Systolic pulmonary artery pressure was 35 ± 8 mmHg, inferior vena cava diameter 21 ± 4 mm, right atrial area 25 ± 9 cm2, 4D ejection fraction 45 ± 7%, 4D fractional area change 40 ± 6%, and tricuspid annular plane systolic excursion 21 [15–25] mm. 2D/4D right ventricular-basal diameter (RVD1) was significantly different (p < 0.005). Similarly, 2D/4D right ventricular diameter measured at the level of the left ventricular papillary muscles (RVD2) was significantly different (p < 0.012), as well as 2D/4D tricuspid annular diameter (p = 0.020). Despite these differences, a strong correlation between variables was observed (Spearman correlation coefficient >0.824). In evaluating the correlation between TR severity and analyzed variables, RVD1 was related to TR severity both at 2D and 4D echocardiography. Conversely, RVD2 and TA diameter were significantly associated with TR severity only at 4D echocardiography.
Our results suggest that specific patient subsets could benefit more from TA measurements using the 4D Auto TVQ tool to help identify the mechanisms responsible for TR, including candidates for left-sided valve surgery and patients in whom the indication for TV repair is unclear.
Keywords
- 4D echocardiography
- tricuspid valve
- tricuspid annulus
The tricuspid valve (TV) is a complex three-dimensional (3D) anatomical
structure, involving various components of the right heart. It is the largest
valve of the human heart, with a saddle-shaped elliptical annulus and an annular
area ranging from 9.72
Numerous conditions may impact valve function and size, particularly volume load, therefore the assessment for the presence of severe TR is challenging. In addition, echocardiographic quantification is not accurate as it is affected by right ventricular (RV) preload, afterload, and function.
The prevalence of severe TR is ~10–15% in patients undergoing
left-sided valve surgery. According to current guidelines, TV surgery should also
be considered in patients with mild or moderate TR with a dilated annulus
(
The aim of this study was to compare 2D vs 4D echocardiographic measurements of the TV using a four-dimensional (4D) Auto Tricuspid Valve Quantification (TVQ) tool (GE Healthcare Vingmed, Horten, Norway) in order to provide more accurate information for surgical planning of TV repair.
From January to April 2024, all echocardiographic data of patients referred to the Città di Lecce Hospital, GVM Care & Research for TV assessment were collected at baseline. All echocardiographic data sets were acquired using a GE Vivid E95 ultrasound system (GE Healthcare; Vingmed Ultrasound, Horten, Norway) equipped with an M5S probe (frequency range: 1.5–4.6 MHz; GE Healthcare; Vingmed Ultrasound, Horten, Norway) and the 4Vc-D matrix-array transducer (5692036, GE Healthcare; Vingmed Ultrasound, Horten, Norway). Video loops and images (2D–4D) were stored in the morning and analyzed in the afternoon during medical reporting by an expert cardiologist with 10 years of professional experience in echocardiography. All the measurements were performed again by trained fellows in echocardiography blinded to clinical data. Intraobserver variability was assessed by the same expert cardiologist one month later. While the interobserver variability was assessed by 2 independent blinded cardiologists (fellows in echocardiography) in a randomly selected subgroup of patients.
Exclusion criteria were active cancer, pregnant women, congenital heart disease,
atrial fibrillation, renal or hepatic failure, inadequate image quality.
Inclusion criteria were age
For the quantification of TV morphology, the novel 4D Auto TVQ tool was used. This tool is designed for clarifying the main mechanism (i.e., annulus dilation, leaflet tethering or mixed) in patients with functional tricuspid regurgitation (FTR) mostly if still unclear by 2DE. It’s useful too in patients with FTR who candidates for left-sided valve surgery are, device implantation or in patients who are considered for catheter-based interventional repair procedures. In these patients the quantification of TV morphology is mandatory, mostly when the indication to perform TV repair is unclear.
This tool enables a rapid semi-automated detection of the TV leaflet surface in only one systolic frame during the cardiac cycle. Quantitative TV measurements analysis included: annulus parameters (3D area, 2D area, perimeter, 4CH diameter, 2CH diameter, major diameter, minor diameter, sphericity index, excursion) and leaflets parameters (Coaptation point Height, Max Tenting Height, Tenting Volume) (Fig. 1).
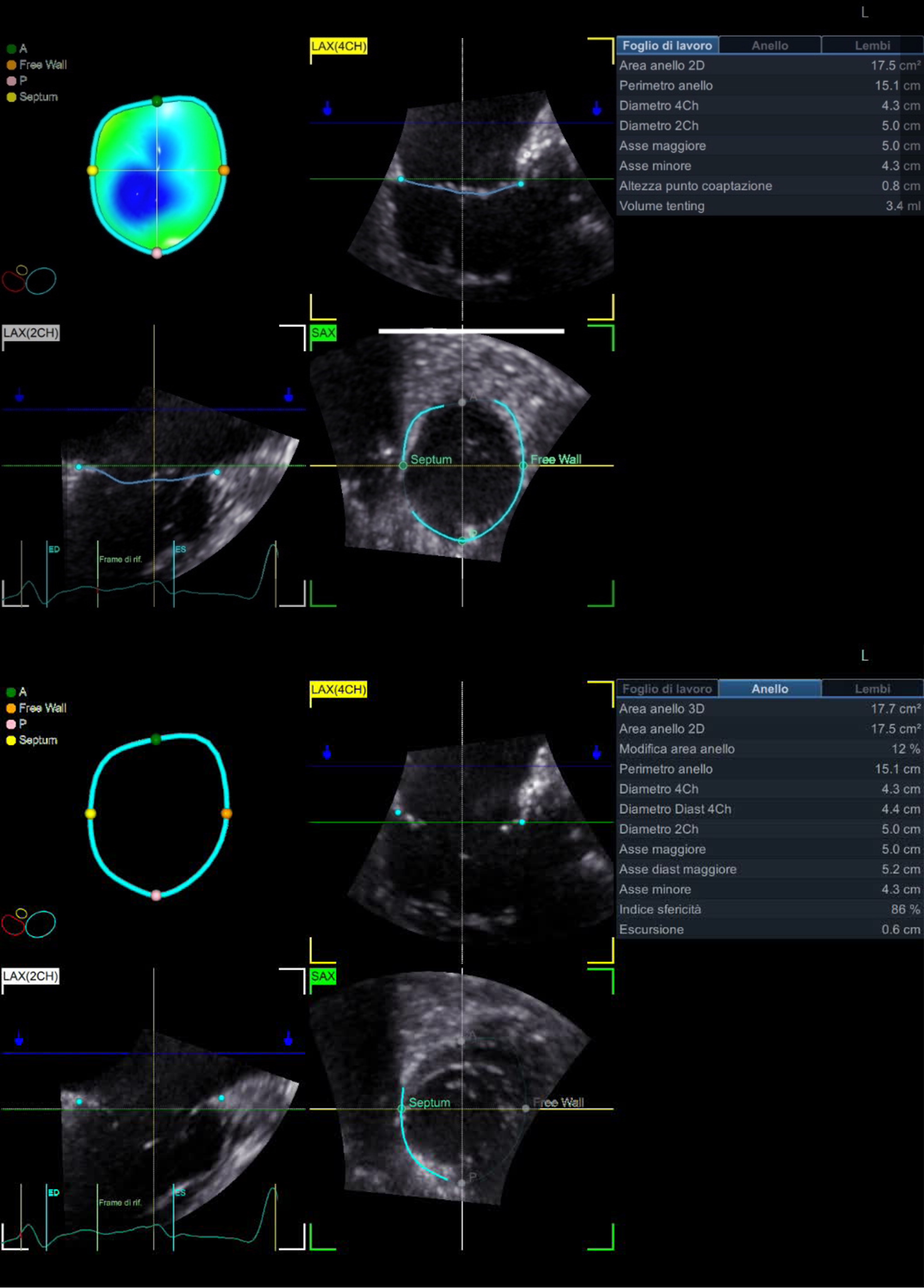 Fig. 1.
Fig. 1.
Tricuspid valve measurements using the 4D Auto TVQ analysis tool. Three-dimensional TV annulus model in patients with functional tricuspid regurgitation (FTR) seen en-face from the ventricular perspective after setting landmarks of TV on 4CH, 2CH and then on SAX. Quantitative analysis of the tricuspid annulus geometry: 2D area, 3D area, area change, perimeter, 4CH diameter, 4CH diastolic diameter, 2CH diameter, major axis, major diastolic axis, minor axis, sphericity index, excursion. Leaflets parameters: coaptation point height, max tenting height, tenting volume. 4D, four-dimensional; TVQ, Tricuspid Valve Quantification; TV, tricuspid valve; 4CH, 4 chamber; 2CH, 2 chamber; SAX, short-axis view; 2D, two-dimensional; 3D, three-dimensional; LAX (2CH), long axis 2 chamber.
It’s specifically designed for the quantification of tricuspid valve (TV) morphology, but it’s not yet validated against cardiac magnetic resonance imaging (MRI). It’s only verified against manual multiplanar reconstruction (MPR) measurements, anyway clinical studies are ongoing in this field.
The RV 3D model and worksheet were obtained using the 4D Auto right ventricle quantification (RVQ) tool. This tool is easily used by the operator for editing the contours after placement of six landmark points (two TA points and the RV apex point in the 4CH view, and the RV/LV posterior and anterior points plus the RV free wall point in the SAX mid papillary view). The worksheet and a time–volume curve were visualized at the results stage (Fig. 2).
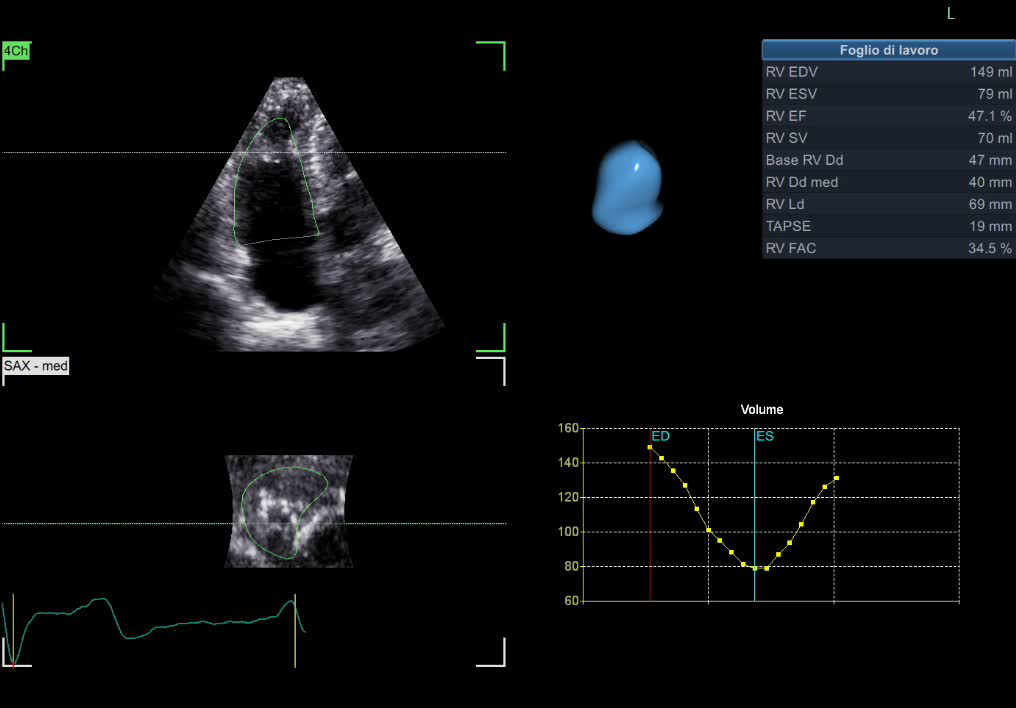 Fig. 2.
Fig. 2.
Right ventricle measurements using the 4D RVQ tool. Three-dimensional right ventricular model and worksheet after using 4D RVQ tool for quantification. The contours can easily be edited by the operator after placed six landmark points. The worksheet and a time–volume curve are visualized at the results stage. EDV, end-diastolic volume; ESV, end-systolic volume; EF, ejection fraction; SV, stroke volume; RV, right ventricular; Dd med, diameter (medium-level); Ld, diameter (lower); TAPSE, tricuspid annular plane systolic excursion; FAC, fractional area change; RVQ, right ventricle quantification.
Additional information on the mechanisms underlying TV dysfunction leading to altered global RV kinetics was derived from tissue Doppler and color Doppler data. The anterior and septal leaflets were visualized on the TTE apical 4-chamber view, whereas the posterior leaflet was identified more reliably in the apical 2-chamber view, since the plane passes through the lower part of the RV free wall adjacent to the diaphragm.
During the echocardiographic examination, several indices were obtained to evaluate TV function and morphology, including TR area allowing an estimation of TR severity and the tricuspid annular plane systolic excursion (TAPSE). Additionally, the peak systolic pressure gradient between the right atrium and right ventricle was measured as it is an important hemodynamic marker for evaluating pulmonary artery pressure and RV function. The assessment of RV size by means of 2D and 3D echocardiography included measurement of RV diameter and right atrial area using RV-focused apical 4-chamber views. RV size is best estimated at end-diastole [5]. Assessment of RV systolic function was also performed using several parameters, including TAPSE and fractional area change (FAC) on 2D echocardiography, which have been found to be significant markers of RV contractile function [6], and RV ejection fraction on 2D and 3D echocardiography, longitudinal strain and strain rate parameters.
All patients gave written informed consent for use of their clinical data, and data collection, analysis, publication and storage were approved by the ethics committee (BE-CdLH 1/24).
All variables were not normally distributed at the Shapiro-Wilk test, and data are reported as median and interquartile range. Differences in a same variable obtained using different methods (i.e., 2D/4D RV-basal diameter [RVD1], 2D/4D RV diameter measured at the level of the left ventricular papillary muscles [RVD2], and 2D/4D TA diameter) were detected by Wilcoxon test for dependent variables. The exact Fisher test has been applied where appropriate. Spearman correlation test was used to compute correlation analysis. Needed sample size was not computed due to this analysis was considered as Pivotal study.
Overall, 11 patients (median age 72 [66–78] years, 18% female) were included in the study. We included only 11 patients with good-quality echocardiographic images who underwent cardiac surgery in our center.
All patients had symptoms of dyspnea (NYHA [New York Heart Association] class II in 8 patients and NYHA class
III in 3 patients). Mild, moderate, and severe tricuspid regurgitation (TR) was
present in 6, 3 and 2 patients, respectively. The etiology for TR in all patients
was Secondary or Functional TR caused by dilatation of the TV annulus related to
right ventricular or right atrial remodeling. Systolic pulmonary artery pressure
(sPAP) was 35
Significant differences were observed between 2D and 4D measurements for RDV1
(right ventricular-basal diameter), RDV2 (right ventricular diameter measured at
the level of the left ventricular papillary muscles), and TA diameter (Table 1),
particularly for 2D vs. 4D RVD1 (2D RDV1 44 [40–55] vs 4D RDV1 47 [44–60];
p
| 2D | 4D | p-value | |
| RVD1 (mm) | 44 [40–55] | 47 [44–60] | 0.005 |
| RVD2 (mm) | 35 [30–40] | 35 [31–50] | 0.012 |
| TA diameter (mm) | 48 [42–56] | 44 [39–53] | 0.020 |
Values are reported as median [interquartile range].
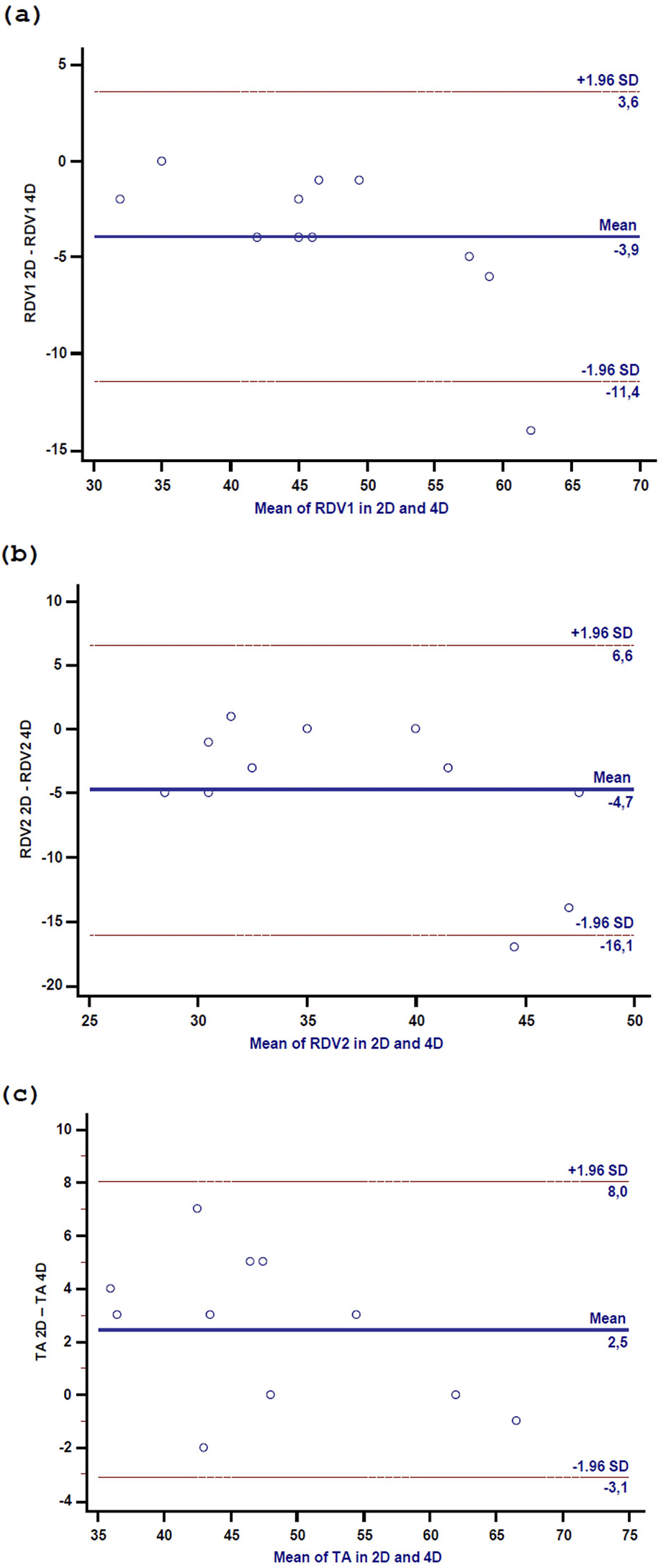 Fig. 3.
Fig. 3.
Scatter plot in 2D and 4D of RDV1 (a), RDV2 (b), and TA diameter (c). RVD1, right ventricular-basal diameter; RVD2, right ventricular diameter measured at the level of the left ventricular papillary muscles; TA, tricuspid annulus.
Similarly, a significant difference was found between 2D vs. 4D RVD2 (2D RDV2 35
[30–40] vs 4D RDV2 35 [31–50], p
| Spearman r | p-value | |
| RVD1 2D vs 4D | 0.94 | |
| RVD2 2D vs 4D | 0.82 | 0.002 |
| TA diameter 2D vs 4D | 0.92 |
| Spearman r | p-value | |
| RVD1 2D | 0.64 | 0.034 |
| RVD1 4D | 0.68 | 0.023 |
| RVD2 2D | 0.51 | 0.107 |
| RVD2 4D | 0.61 | 0.047 |
| TA diameter 2D | 0.52 | 0.098 |
| TA diameter 4D | 0.66 | 0.029 |
The TV is a complex anatomical structure, with a saddle-shaped elliptical annulus, making manual quantification on 2D echocardiography or 3D multi-planar reconstruction challenging. The tricuspid annulus is also a highly dynamic structure, with various changes in size and shape during cardiac cycle, which are difficult to fully characterize and measure frame-by-frame, unless the use of a novel semi-automated tracking tool, able to follow its changes in three dimensions. As the options for addressing tricuspid insufficiency continue to evolve, including both surgical and percutaneous intervention techniques, the meticulous analysis of annulus diameters and dimensions becomes increasingly essential [12]. This thorough assessment is crucial for planning both surgical and percutaneous procedures, as it significantly influences the procedural outcomes [13, 14].
Therefore, there is the need for a quantification TV tool, such as the 4D Auto
TVQ tool. In our pivotal study, we found a significant difference between
measurement of RV variables obtained with the 4D Auto TVQ tool vs 2D
echocardiography, confirming the importance of a more accurate assessment of the
TV. 2D echocardiography is the first-line imaging modality for TV evaluation,
though quantitative analysis of the TV apparatus is burdened by limited accuracy.
TA diameter by 2D echocardiography systematically underestimates the true largest
size of the annulus. In functional TR, TA enlargement occurs predominantly in the
antero-posterior direction, and the largest diameter is often not oriented in the
septo-lateral direction shown in the 4-chamber view [15, 16]. Only 3D
echocardiography allows to acquire the 3D TA area and perimeter mostly for its
complex geometry [17]. In clinical routine, echocardiography is the most
frequently used imaging technique to assess patients with heart valve diseases
[18]. Cardiac magnetic resonance (CMR) and cardiac computed tomography (CCT) are
new and emerging imaging modalities but less used in clinical practice for
three-dimensional visualization of the TA. Indeed, the limited access in some
medical centers which need specific imaging protocols for the right heart, and
the use of contrast media or radiation reduce their application mainly to
patients undergoing evaluation for TV surgery. In symptomatic patients with
severe functional TR, all these imaging variables—TA size, extent of leaflet
tenting, right atrial size and RV size and function—are carefully weighted for
choosing the best approach or device for valve repair. An antero-posterior TA
diameter
In reference to these guideline recommendations, a few aspects related to our
analysis should be underlined. First, TV surgery has a central role, given that
in current guidelines transcatheter procedures should be reserved only for
patients not amenable to surgery. In this respect, treatment of TV disease
differs greatly from treatment of other valvular pathologies, namely severe
aortic stenosis for which transcatheter procedures are preferred over
conventional surgery, particularly in some specific age groups (
In patients with TA dilatation not treated with annuloplasty and undergoing mitral valve surgery, TR increases by more than 2 grades compared to no or 1 grade increase in patients receiving concomitant tricuspid annuloplasty [22]. Contrary to current beliefs, TR does not disappear spontaneously after correction of left-sided valve disease. These data demonstrate that TA dilatation results in impairment of RV function with progressive and clinically relevant TR. Treatment of mitral lesions only reduces afterload, but it does not correct TA dilatation or affect RV preload or function.
Therefore, combined mitral valve repair and tricuspid annuloplasty should be considered in patients with TA dilatation also in the absence of significant TR. In a study conducted between 2003 and 2011, a strategy of routine treatment of moderate TR and/or significant TA dilatation at the time of mitral valve repair in patients with degenerative mitral regurgitation was associated with freedom from recurrent TR [23]. This above-mentioned study further supports the importance of a 4D echocardiographic tool for better surgical planning. Furthermore, the opportunity to quantify the right ventricle parameters (diameters and area of RV) using 4D Auto RVQ from the same data set of patients on which the 4D Auto TVQ analysis is done, is less time-consuming and provides more precise and reproducible results than the corresponding 2D parameters. The more precise characterization of right-heart structures allows to better understand the central role of RA enlargement in determining TA size in atrial fibrillation (AF) either in healthy subjects or in different etiologies of FTR.
Our study patients did not undergo cardiac surgery and we could not therefore compare echocardiographic values vs anatomical/surgical measurements. The impact of TR on long-term mortality of patients has been well-known for many years [24]. In a large population of over 5000 patients from almost 20 years ago, patients with moderate or severe TR showed a higher mortality than patients with mild TR despite of pulmonary artery pressure and left ventricular ejection fraction [24]. These findings strongly suggest that increasing TR severity without surgical correction is associated with worse survival in the long term. In addition, failure of the annuloplasty procedure performed during a concomitant intervention result in later TR progression. Significant TR (3+ or 4+) was identified in 12% of patients (11/91) due to failure of TV repair or progression of untreated TR [25]. These findings support the clinical relevance of our study, in that part of surgical failures was due to suboptimal planning of annuloplasty based on 2D measurements under various filling conditions. However, it can be argued that size of the tricuspid ring is established directly in the operating room, though this occurs in para-physiological conditions, i.e., with a still and open heart with a totally empty right ventricle. Our study does not aim at replacing this direct measurement, which represents the cardiac surgeon’s final decision-making criterion for choosing the most appropriate ring, but simply aims at comparing 2D measurements with those obtained using the new 4D Auto TVQ tool. Our pivotal analysis highlighted the differences between the two methods in the RVD1, RVD2 and TV diameters measurements, in a sample where is showed a higher association of 4D measures and TR severity than 2D and TR severity.
Many limitations to this retrospective study should be acknowledged. First, it was a single center, with a small number of patients. Multi-center studies on larger cohorts need to be recommended. Second, all echocardiographic datasets were acquired and measured by only one expert cardiologist and to achieve the interobserver variability cardiologists (fellows in echocardiography) with less experience were recruited. Third, our population was made by Caucasian volunteers only, which could limit the heterogeneity of our reference value. Fourth, our acquired TV measurements were mainly suitable to patients with high-quality echocardiographic images, which may restrict the generalizability of the results. However, currently there is no other commercially tool designed for TV quantification to compare our 3DE values. Moreover, we did not compare our measurements to CMR and CT results, but Muraru et al. [26] have tested the accuracy of their measurements obtained with 4D Auto-TVQ compared to cardiac CT but not to CMR imaging.
Further studies in this field with multi-center, larger cohorts of patients and control groups are warranted to validate these results.
TA enlargement is the most important mechanism leading to FTR and the first therapeutic target for surgical and interventional repair procedures. The primary imaging modality to image the TV is 2DE, but its accuracy for quantitative analysis of TV apparatus is severely limited. The 4D Auto TVQ is a novel and modern tool for TV quantification that could be used as a complement to standard guideline-recommended 2DE. Specific patient subsets may benefit more from 4D Auto TVQ, including patients with functional TR in whom measurement of TA diameter and leaflet tethering can help identify the main mechanism of TR or patients with functional TR who are going to left-sided valve surgery and in whom the indication for TV repair is still unclear. So, a better knowledge of TV apparatus and FTR pathophysiology provides the basis for performing a personalized treatment approach, more accurate surgical planning, and effective surgical procedures.
Datasets used and/or analyzed for this study are available from the corresponding author upon appropriate request.
GSa, GTa, VC, FC, GTr, OT, CF, GA, GN, GSp contributed to the study conception and design. Material preparation, data collection and analysis were performed by GSa, GTa, and VC. The first draft of the manuscript was written by GTa and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. All authors contributed to editorial changes in the manuscript. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Città di Lecce Hospital (internal board, BE-CdLH 1/24). All patients provided written informed consent for inclusion, data, or sample collection/use and/or publication of data results. The clinical ethical board has approved the use of patient’s clinical data, according to privacy rules.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Giuseppe Santarpino is serving as one of the Editorial Board members and Guest Editor of this journal. We declare that Giuseppe Santarpino had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Ferdinand Aurel Vogt.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



