1 Department of Cardiology, The Affiliated Dongguan Songshan Lake Central Hospital, Guangdong Medical University, 523326 Dongguan, Guangdong, China
2 Department of Cardiology, Cangzhou Central Hospital, Hebei Medical University, 061001 Cangzhou, Hebei, China
†These authors contributed equally.
Abstract
The primary objective of this research was to determine the predictive value of the residual SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) score II (rSS-II) for long-term outcomes in individuals with complex coronary artery disease (CAD) and chronic renal insufficiency (CRI) who have undergone percutaneous coronary intervention (PCI).
A total of 1161 consecutive patients with complex CAD and CRI after PCI were retrospectively recruited from Cangzhou Central Hospital affiliated with Hebei Medical University between January 2014 and September 2017. The patients were stratified into three categories based on rSS-II tertiles: low rSS-II (n = 388), medium rSS-II (n = 389), and high rSS-II (n = 384). The primary endpoints were all-cause mortality (ACM) and cardiac mortality (CM), while the secondary endpoint was major adverse cardiovascular and cerebrovascular events (MACCEs), which included ACM, myocardial infarction, stroke, or unplanned revascularization. The discrimination, calibration, and clinical utility of the rSS-II for predicting long-term outcomes were examined.
The median follow-up period was 37 months (19 to 61 months). The Kaplan–Meier estimate rates of ACM (2.4% vs. 5.9% vs. 13.9%; p < 0.001) and CM (1.9% vs. 2.8% vs. 9.2%; p < 0.001) revealed significant differences among the three categories. Multivariate Cox regression analysis demonstrated that the rSS-II could independently predict ACM (hazard ratio: 1.08, 95% confidence interval: 1.04–1.12; p < 0.001) and CM (hazard ratio: 1.07, 95% confidence interval: 1.02–1.12; p = 0.009). The rSS-II performed satisfactorily in both discrimination (area under the curve for ACM and CM was 0.710 and 0.728, respectively) and calibration (Greenwood–Nam–D’ Agostino goodness-of-fit test for long-term outcomes; p > 0.05 for all). Additionally, decision curve analysis showed that the rSS-II had a high net benefit for long-term outcomes over threshold probabilities, indicating its superiority in daily practice.
The rSS-II is beneficial for predicting and stratifying the risk of long-term outcomes in individuals with complex CAD and CRI who have undergone PCI.
Keywords
- residual SYNTAX score II
- coronary artery disease
- chronic renal insufficiency
- percutaneous coronary intervention
Previous studies have validated the favorable effect of complete revascularization (CR) on the prognoses of patients with complex coronary artery disease (CAD) [1, 2, 3]. The COMPLETE trial, which compared CR versus culprit-only revascularization strategies for treating multivessel disease after early percutaneous coronary intervention (PCI) for ST-elevation myocardial infarction (STEMI), demonstrated that CR was better than culprit-lesion-only PCI in lowering the risk of adverse cardiovascular events in individuals affected by STEMI and multivessel CAD [4]. Additionally, renal function impairment has been associated with rapid plaque progression, potentially contributing to the vulnerability of coronary plaques and increasing the risk of CAD [5, 6]. Furthermore, the Grand Drug-Eluting Stent (Grand-DES) registry, which included five multicenter national Korean registries, reported that CR leads to improved clinical results in individuals with chronic kidney disease (CKD) compared to incomplete revascularization (IR) [7]. Therefore, the complexity and severity of coronary disease and renal function determine the PCI strategy for CR or IR in clinical settings. Hence, risk stratification and evaluation of residual disease should be performed for the long-term prognosis of individuals with complex CAD and chronic renal insufficiency (CRI) after PCI.
The development of the SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) score (SS), also known as the pre-PCI SS, was aimed at evaluating the anatomic complexity of angiographic stenoses and facilitating decision-making for the optimal revascularization approach in individuals with complex CAD [8]. However, previous research has revealed the association of SS with mortality and adverse cardiovascular events in acute coronary syndrome (ACS) [9, 10]. Despite its usefulness, the lack of clinical variables in SS has been identified as a limitation by European and United States revascularization guidelines [11, 12]. To overcome these limitations, the SYNTAX score II (SS-II) was developed, which combines anatomical factors (SS and unprotected left main coronary artery (ULMCA) disease) with six clinical variables (age, creatinine clearance, left ventricular ejection fraction (LVEF), sex, chronic obstructive pulmonary disease (COPD), and peripheral vascular disease). Compared to SS, SS-II has shown a gradually increasing predictive value for long-term mortality in individuals with complex CAD undergoing PCI [13]. Head et al. [1] reported that IR was associated with unfavorable clinical outcomes after PCI in a 3-year SYNTAX trial, and the post-PCI SYNTAX score, such as the residual SYNTAX score (rSS), was developed to evaluate the predictive value of IR after PCI in the Acute Catheterization and Urgent Intervention Triage Strategy (ACUITY) trial, quantifying the residual burden of anatomical coronary disease [2]. The high-rSS group was associated with an elevated risk of clinical comorbidity and a disease characterized by more anatomical complications compared to low-rSS groups [3]. Additionally, previous studies have shown that rSS is relevant to poorer clinical outcomes both during the in-hospital period and the subsequent long-term period after discharge [14, 15]. Recently, the post-PCI SS-II, such as the residual SS-II (rSS-II), has been validated for predicting long-term outcomes in individuals with ACS [16, 17]. However, no prior studies have evaluated the predictive value of rSS-II in patients with complex CAD and CRI. Therefore, the main objective of this research was to validate the prognostic value of rSS-II in individuals with complex CAD and CRI after PCI.
The design and methods of this study have been described in previous research
[15]. Briefly, 2529 consecutive patients with CAD and CRI who received PCI at
Cangzhou Central Hospital of Hebei Medical University were retrospectively
screened from January 2014 to September 2017. Patients were excluded if they had
previous coronary artery bypass grafting, staged PCI, or unplanned PCI during a
second hospitalization. Additionally, 1312 patients with no evidence of
three-vessel disease or left main disease involvement were excluded from the
study. Ultimately, 1161 individuals with complex CAD, defined as three-vessel
diseaseand/or left main disease (stenosis
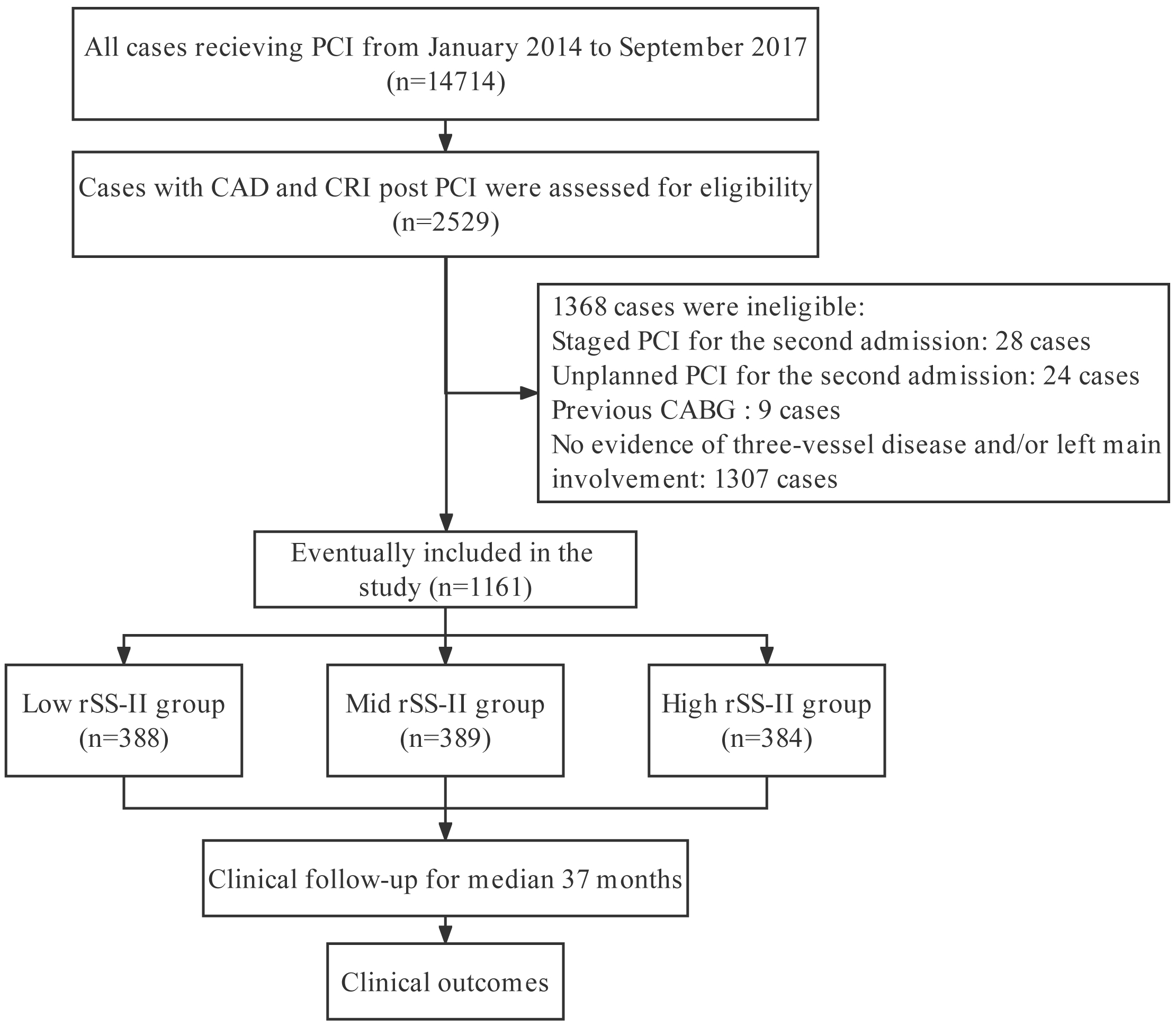 Fig. 1.
Fig. 1.
Flowchart of study. CABG, coronary artery bypass grafting; CAD, coronary artery disease; CRI, chronic renal insufficiency; PCI, percutaneous coronary intervention; rSS-II, residual SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) score II.
The calculation of rSS was performed using the SS online calculator, which considers obstructive coronary stenosis after PCI. IR was defined as an rSS value greater than 0. To evaluate the SS-II for PCI, two anatomical variables (SS and ULMCA disease) and six clinical variables (age, creatinine clearance, LVEF, sex, COPD, and peripheral vascular disease) were included using the online calculator available at http://www.syntaxscore.org [13]. The rSS-II was determined as the post-PCI SS-II, calculated using the PCI SS-II calculator based on the online calculator.
Follow-up assessments were conducted for all patients through telephone interviews or clinic visits, and clinical outcome data were verified by reviewing medical records. The primary endpoints of the study were all-cause mortality (ACM) and cardiac mortality (CM). In cases where a clear non-cardiac etiology was not identified, death was assumed to be due to a cardiac reason. The secondary endpoint was major adverse cardiovascular and cerebrovascular events (MACCEs), which was defined as a composite of ACM, myocardial infarction (MI), stroke, or unplanned revascularization. During the follow-up period, the fourth universal definition was used as the basis for defining MI [19].
The Kolmogorov-Smirnov test was used to assess the normal distribution of
continuous variables. All variables exhibited skewed distributions and were
therefore represented as median (interquartile range, IQR). Comparative analysis
of these variables was performed using the Kruskal-Wallis H test.
Categorical variables were presented in terms of counts and proportions (%), and
their comparative analysis was conducted using the Pearson
The validation of the rSS-II was conducted according to the strategy proposed by
Steyerberg and Vergouwe [20]. The discriminatory capacity of the rSS-II was assessed using the
area under the curve (AUC) of time-dependent receiver operating characteristic
(ROC) curves. The AUC values of the rSS-II were compared with those of the rSS
and SS-II based on a previous study [21]. Calibration of the rSS-II, rSS, and
SS-II was performed using the Greenwood-Nam-D’ Agostino goodness-of-fit test by
comparing the observed and predicted outcomes [22]. A calibration plot of the
predicted versus observed probabilities was developed, following recent
statistical recommendations for developing and validating risk scores [23]. The
calibrations of different models were compared at a median follow-up period of 37
months using the mean, median, and 90th percentile of absolute calibration error
(integrated calibration index (ICI), E50, and E90, respectively) [24]. Decision
curve analysis (DCA) was used to evaluate which model was most helpful in
identifying individuals with a higher risk of long-term outcomes at 37 months
[25]. Statistical significance was determined when a two-sided p-value
was
The median age of the patients was 67.0 (61.0–71.0) years, and the body mass
index (BMI) was 26.1 (24.8–27.3) kg/m2. Among the patients, 59.3% were
men. Table 1 displays the patient demographics and clinical characteristics
according to the tertiles of the rSS-II. Patients in the high-rSS-II category
were older and had lower baseline characteristics such as male gender, serum
creatinine, hemoglobin, eGFR, and LVEF, but had higher fasting glucose and total
cholesterol levels (all p
| Low (n = 388) | Medium (n = 389) | High (n = 384) | p-value | ||
| Age (years) | 66.0 (55.0–66.0) | 67.0 (62.0–71.0) | 70.0 (67.0–75.0) | ||
| Male (%) | 372 (95.9) | 216 (55.5) | 101 (26.3) | ||
| BMI (kg/m2) | 26.4 (25.2–27.6) | 26.1 (24.8–27.2) | 25.8 (24.6–27.1) | ||
| Risk factors (%) | |||||
| Hypertension | 255 (65.7) | 271 (69.7) | 277 (72.1) | 0.150 | |
| Dislipidemia | 160 (41.2) | 147 (37.8) | 161 (41.9) | 0.453 | |
| Diabetes | 78 (20.1) | 105 (27.0) | 122 (31.8) | 0.001 | |
| Smoking | 57 (14.7) | 46 (11.8) | 30 (7.8) | 0.011 | |
| Previous history (%) | |||||
| Myocardial infarction | 31 (8.0) | 35 (9.0) | 44 (11.5) | 0.239 | |
| PCI | 46 (11.9) | 43 (11.1) | 43 (11.2) | 0.932 | |
| Stroke | 36 (9.3) | 42 (10.8) | 51 (13.3) | 0.203 | |
| Initial presentation | |||||
| STEMI (%) | 112 (28.9) | 97 (24.9) | 119 (31.0) | 0.130 | |
| Heart rate (beats/min) | 73.0 (70.0–77.0) | 73.0 (70.0–77.0) | 73.0 (70.0–79.0) | 0.932 | |
| SBP (mmHg) | 133.0 (120.0–145.0) | 130.0 (124.0–149.0) | 132.0 (120.0–150.0) | 0.748 | |
| Renal and cardiac function | |||||
| eGFR (mL/min per 1.73 m2) | 83.3 (77.8–87.1) | 78.5 (71.1–84.1) | 68.0 (55.2–77.0) | ||
| LVEF (%) | 63.0 (60.0–64.0) | 62.0 (59.0–64.0) | 61.0 (54.0–64.0) | ||
| LVEDD (mm) | 48.0 (46.0–50.0) | 47.0 (45.0–50.0) | 47.0 (45.0–51.0) | 0.001 | |
| Laboratory data | |||||
| cTNI (ng/mL) | 0.30 (0.05–1.80) | 0.32 (0.05–1.34) | 0.45 (0.05–2.09) | 0.067 | |
| CK-MB (U/L) | 14.7 (12.0–23.3) | 14.9 (11.9–22.0) | 16.3 (12.1–30.4) | 0.111 | |
| Creatine (µmol/L) | 86.0 (83.0–92.0) | 83.0 (67.0–95.0) | 81.0 (70.0–105.0) | ||
| Fasting glucose (mmol/L) | 6.4 (5.3–8.4) | 6.7 (5.5–9.1) | 7.2 (5.8–10.0) | ||
| TC (mmol/L) | 4.4 (3.9–4.9) | 4.3 (3.9–5.0) | 4.5 (4.1–5.2) | 0.020 | |
| Hemoglobin (g/L) | 139.0 (130.0–149.0) | 131.0 (120.0–141.0) | 123.0 (113.0–132.0) | ||
Values are median (interquartile range) or n (%). BMI, body mass index; CK-MB, creatine kinase-MB; cTNI, cardiac troponin I; eGFR, estimated glomerular filtration rate; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention; rSS-II, residual SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) score II; SBP, systolic blood pressure; STEMI, ST-elevation myocardial infarction; TC, total cholesterol.
| Low (n = 388) | Medium (n = 389) | High (n = 384) | p-value | ||
| Diseased vessels (%) | |||||
| 3-vessel | 330 (85.1) | 339 (87.1) | 333 (86.7) | ||
| Left main | |||||
| Isolated | 2 (0.5) | 1 (0.3) | 0 | ||
| Plus-1-vessel | 6 (1.5) | 3 (0.8) | 1 (0.3) | ||
| Plus-2-vessel | 23 (5.9) | 22 (5.7) | 6 (1.6) | ||
| Plus-3-vessel | 27 (7.0) | 24 (6.2) | 44 (11.5) | ||
| Lesion anatomical characteristics | |||||
| Lesion length |
278 (71.7) | 283 (72.8) | 286 (74.5) | 0.672 | |
| Bifurcation or trifuraction | 64 (16.5) | 69 (17.7) | 45 (11.7) | 0.049 | |
| Aorto-ostial lesion | 3 (0.8) | 6 (1.5) | 10 (2.6) | 0.132 | |
| Heavy calcification | 16 (4.1) | 19 (4.9) | 28 (7.3) | 0.128 | |
| Severe tortuosity | 6 (1.6) | 12 (3.1) | 18 (4.7) | 0.042 | |
| Thrombus | 18 (4.6) | 26 (6.7) | 28 (7.3) | 0.277 | |
| CTO | 34 (8.8) | 30 (7.7) | 35 (9.1) | 0.768 | |
| Location of target vessels | |||||
| LM | 41 (10.6) | 21 (5.4) | 9 (2.3) | ||
| LAD | 229 (59.0) | 213 (54.8) | 158 (41.2) | ||
| LCX | 156 (40.2) | 131 (33.7) | 121 (31.5) | 0.031 | |
| RCA | 196 (50.5) | 183 (47.0) | 202 (52.6) | 0.295 | |
| Procedural characteristics | |||||
| Stent per patient | 2.0 (1.0–3.0) | 2.0 (1.0–3.0) | 2.0 (1.0–2.0) | ||
| Total length of the stent (mm) | 56.0 (35.0–86.0) | 50.0 (33.0–72.0) | 46.0 (29.0–66.0) | ||
| Stent length |
54 (13.9) | 42 (10.8) | 26 (6.8) | 0.005 | |
| CR (%) | 25 (6.4) | 10 (2.6) | 4 (1.0) | ||
| Mean stent diameter (mm) | 3.0 (2.8–3.2) | 2.9 (2.7–3.1) | 2.8 (2.7–3.0) | 0.001 | |
| Minimum stent diameter (mm) | 2.8 (2.5–3.0) | 2.8 (2.5–3.0) | 2.8 (2.5–3.0) | 0.488 | |
| Maximum stent diameter (mm) | 3.0 (3.0–3.5) | 3.0 (2.8–3.5) | 3.0 (2.8–3.5) | ||
Values are median (interquartile range) or n (%). CR, complete revascularization; CTO, chronic total occlusions; LAD, left anterior descending artery; LCX, left circumflex; LM, left main; RCA, right coronary artery.
The median follow-up time for the patients was 37 months (range: 19–61 months).
During this period, ACM, CM, and MACCE occurred in 57 (4.9%), 39 (3.4%), and
248 (21.4%) patients, respectively. The incidence of ACM and CM was
significantly higher in the high-rSS-II group compared to the medium- and
low-rSS-II groups (ACM: 13.9% vs. 5.9% vs. 2.4%, p
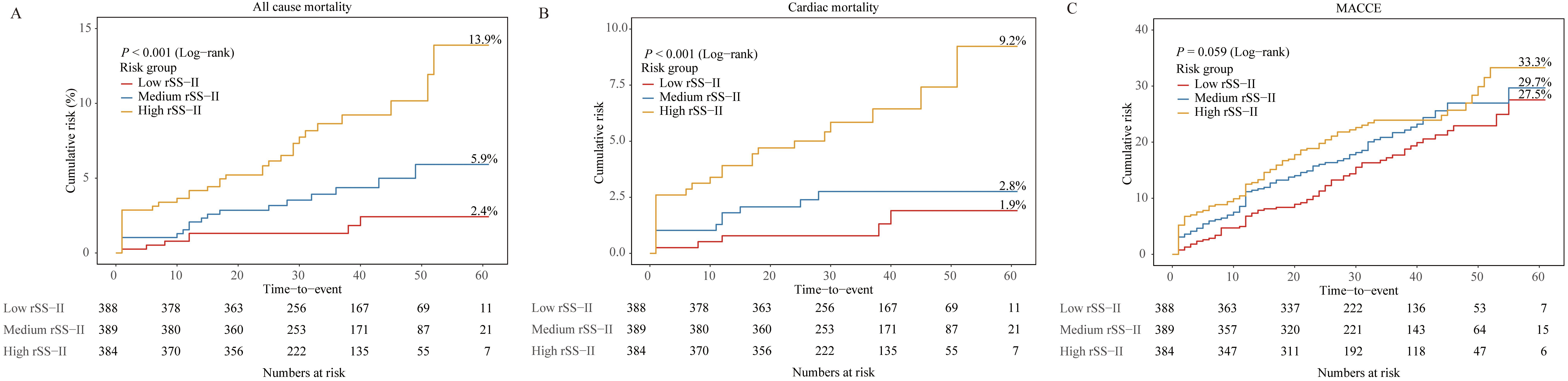 Fig. 2.
Fig. 2.
Kaplan-Meier cumulative risk curves for clinical outcomes according to the rSS-II tertiles. Kaplan-Meier cumulative risk curves for all-cause mortality (A), cardiac mortality (B), and MACCE (C). MACCE, major adverse cardiovascular and cerebrovascular event; SYNTAX, Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery; rSS-II, residual SYNTAX score II.
The univariate Cox regression analysis findings are presented in
Supplementary Table 1. The high-rSS-II group had significantly higher
risks for ACM (2.12- and 5.08-fold) and CM (2.57- and 5.09-fold) compared to the
medium- and low-rSS-II groups, respectively (all p
| Variables | HR (95% CI) | p-value | |
| All-cause mortality | |||
| Hypertension | 0.57 (0.33–0.96) | 0.035 | |
| Dislipidemia | 0.54 (0.29–0.99) | 0.047 | |
| rSS-II | 1.08 (1.04–1.12) | ||
| Cardiac mortality | |||
| Dislipidemia | 0.44 (0.21–0.92) | 0.029 | |
| Previous MI | 2.52 (1.11–5.70) | 0.027 | |
| rSS-II | 1.07 (1.02–1.12) | 0.009 | |
| MACCE | |||
| Previous PCI | 1.45 (1.01–2.07) | 0.042 | |
MACCE, major adverse cardiovascular and cerebrovascular event; MI, myocardial infarction; PCI, percutaneous coronary intervention; rSS-II, residual SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) score II; HR, hazard ratio.
The AUCs of time-dependent ROC at 37 months for rSS-II, rSS, and SS-II were
calculated and compared (Fig. 3). The discrimination capability of rSS-II was
acceptable for predicting ACM and CM. The AUCs of rSS-II for ACM (0.710 vs.
0.578, p = 0.004) and CM (0.728 vs. 0.585, p = 0.022) were
significantly higher than those of rSS. The AUCs of rSS-II had an increasing
trend compared to SS-II (AUC: 0.704 for ACM; 0.724 for CM), but the difference
was not statistically significant (all p
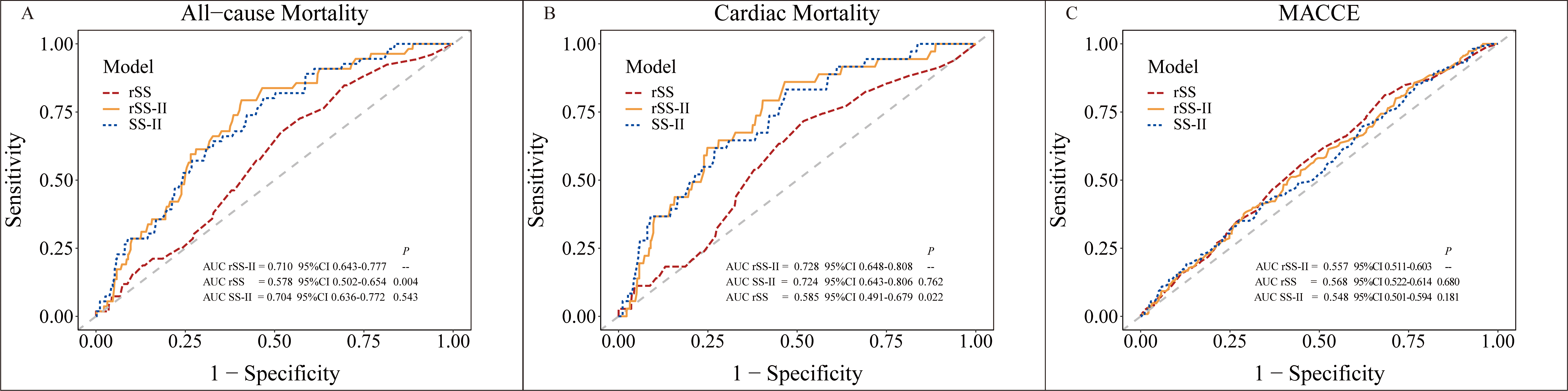 Fig. 3.
Fig. 3.
ROC curve for the discrimination capability of rSS-II compared with rSS and SS-II. Time-dependent receiver operating characteristic (ROC) curves for (A) all-cause mortality; (B) cardiac mortality; and (C) MACCE based on rSS, SS-II, and rSS-II are shown. AUC, the area under the curve; MACCE, major adverse cardiovascular and cerebrovascular event; SYNTAX, Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery; rSS-II, residual SYNTAX score II; rSS, residual SYNTAX score; SS-II, SYNTAX score II.
The calibration curve of rSS-II for the risk of clinical outcomes showed the best
agreement between prediction and observation (Fig. 4). The
Greenwood-Nam-D’Agostino test revealed that all three models were well-calibrated
for predicting ACM, CM, and MACCE (all p
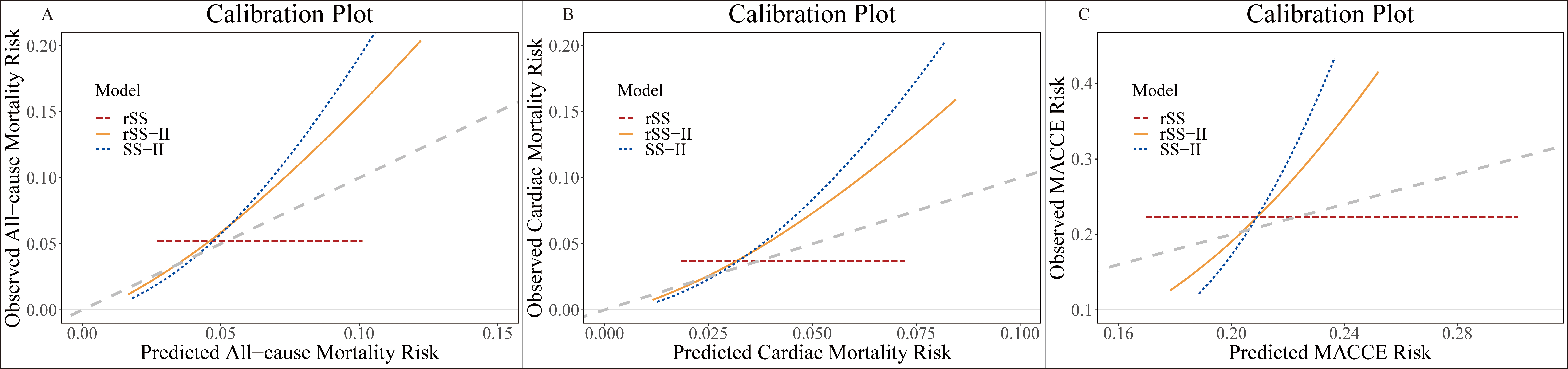 Fig. 4.
Fig. 4.
Calibration plot of predicted vs. observed outcomes. Calibration plots for (A) all-cause mortality, (B) cardiac mortality, and (C) MACCE based on rSS, SS-II, and rSS-II are shown. MACCE, major adverse cardiovascular and cerebrovascular event; SYNTAX, Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery; rSS-II, residual SYNTAX score II; rSS, residual SYNTAX score; SS-II, SYNTAX score II.
The net benefit of rSS, SS-II, and rSS-II at 37 months was evaluated using DCA for the examined prognosis probabilities (Supplementary Fig. 2). The rSS-II showed a high net benefit across a wide range of threshold probabilities for predicting ACM (1.1%–10.7% and 22.2%–28.3%) and CM (0.6%–8.0% and 17.9%–21.5%). However, SS-II had a limited range of threshold probabilities, and rSS showed a low net benefit. Additionally, rSS-II had an acceptable net benefit across a wide range of threshold probabilities for predicting MACCE (14.6%–22.7% and 32.6%–34.1%).
Several important findings were observed in this study: (1) rSS-II was able to predict long-term prognosis over a median follow-up time of 37 months in individuals with complex CAD and CRI after PCI. (2) Compared to rSS and SS-II, rSS-II showed improved calibration, higher net benefit for clinical performance, and better discriminative capacity. (3) rSS-II was able to independently predict long-term prognoses and effectively stratify patients based on their risks of ACM and CM after PCI in individuals with complex CAD and CRI.
Despite Généreux et al.’s [2] claim that post-PCI residual coronary stenosis is a nidus of new events, the treatment of non-culprit vessels and the timing of PCI have been questionable until recently. The SYNTAX trial showed that IR has a negative effect on long-term clinical outcomes and is a surrogate biomarker of significant coronary complexity and clinical comorbidity [26]. The COMPLETE trial reported that CR during the index hospitalization is linked to a clear clinical benefit compared to culprit-lesion-only PCI in individuals with STEMI and multivessel CAD [4]. Several studies have also shown that CR improves the prognosis of individuals receiving PCI for ACS with CKD [7, 26]. However, in the CKD population, Kim et al. [27] found no therapeutic benefit of angiographic CR compared to IR. In this study, first-generation DES was used by about 60% of patients, while second-generation DES has been reported to have a protective impact on clinical outcomes in CKD-affected individuals [28].
The Grand-DES investigation found that CKD-affected individuals who underwent multivessel PCI during the index hospitalization had a reduced incidence of death and cardiovascular events for up to three years after the procedure [7]. However, due to high adverse event rates and low procedural success rates, only a small proportion (3.4%) of patients received CR in this study. The findings of this research are consistent with those reported by the IR group of the trial, indicating that a higher residual disease burden, equivalent to a higher rSS-II, increases unfavorable cardiovascular outcomes in individuals with complex CAD and CRI after PCI.
A previous study demonstrated that rSS, which quantifies the residual burden of
anatomical coronary disease, evaluated the predictive value of IR after PCI [2].
Farooq et al. [3] indicated that rSS was capable of independently
predicting 5-year ACM in individuals affected by complex CAD after PCI using
drug-eluting stents in the SYNTAX trial. However, the application of rSS in
clinical practices is limited due to the absence of clinical variables, which has
led to the establishment and validation of rSS-II for predicting the prognosis of
individuals receiving PCI. Boukhris et al. [29] demonstrated that rSS-II
could anticipate midterm outcomes in patients with complex ACS. Bortnick
et al. [16] further suggested that rSS-II was associated with long-term ACM
using Cox regression analysis in a post-STEMI cohort from the Montefiore STEMI
registry (HR: 2.46, 95% CI: 1.51–3.99, p
In the SHINANO registry, Kashiwagi et al. [17] indicated that rSS-II
might be a crucial tool in predicting long-term mortality in individuals with ACS
and multivessel disease following PCI. The AUC of rSS-II for predicting ACM (AUC
= 0.82, 95% CI: 0.74–0.91) was significantly better compared to that of rSS
(AUC = 0.54, 95% CI: 0.1–0.67) (p
Discrimination, calibration, and clinical utility are well-established components of a predictive model. Steyerberg et al. [30] emphasized that decision-analytic measures should be reported prior to applying the prediction model for clinical decision-making. This approach can potentially assist physicians in evaluating the significance of information by using a risk assessment tool to compare potential risks and benefits. In this research, the advantage of rSS-II over rSS and SS-II for clinical applications was determined through DCA, given that rSS and SS-II were already being used in clinical research and practices. The rSS-II demonstrated acceptable net benefits across various threshold probabilities for long-term prognoses compared to rSS and SS-II.
There are certain limitations to this study. Firstly, it was a retrospective, single-center study. Therefore, further prospective studies and multicenter datasets are necessary to assess the generalizability and validity of rSS-II. Secondly, pre-PCI creatinine clearance and LVEF were used in the study. However, a previous study reported that creatinine clearance and left LVEF in SS II should be modified using PCI [29] and used as post-PCI in rSS-II. Hence, further studies including postoperative creatinine clearance and LVEF are needed to validate the performance of rSS-II in predicting the prognosis of individuals with complex CAD and CRI after PCI. Finally, rSS-II was validated only in the Chinese population. Therefore, additional studies with larger population samples are required to further validate its performance.
The present study provides favorable evidence for the precise use of rSS-II in predicting long-term clinical outcomes in individuals with complex CAD and CRI after PCI. Over a median follow-up of 37 months, rSS-II demonstrated good discriminatory power for risk prediction of ischemic outcomes. Furthermore, it can identify patients who may benefit from further revascularization.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
SYZ, YML, and LQY provided the conception of the idea for the study. SYZ, YML, and JFC contributed to the development of the methodology and wrote the manuscript. SYZ, YML, JFC, YCW, HTL, NG, YFW, and XFC were responsible for the interpretation of statistical results. LQY revised the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Ethics Committee of Cangzhou Central Hospital, Hebei Medical University had approved the study protocol (ID: 2017-006-01), which was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent could be successfully provided by all cases.
We are grateful to all of the study participants whose work made this study possible.
This work was supported by the Special Project for Clinical and Basic Sci & Tech Innovation of Guangdong Medical University (GDMULCJC2024112) and the Natural Science Foundation of Hebei Province, China (H2021110008).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM26962.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




