1 Division of Cardiology, Beijing Anzhen Hospital, Capital Medical University, 100029 Beijing, China
2 Cardiometabolic Medicine Center, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, 100037 Beijing, China
†These authors contributed equally.
Abstract
Systolic blood pressure time in target range (SBP TTR) is a novel metric for blood pressure control. Previous studies have demonstrated an inverse association between SBP TTR and risks of cardiovascular events, but sex differences have never been reported. This study aims to investigate the sex-specific differences in the relationship using data from the Systolic Blood Pressure Intervention Trial (SPRINT).
This post hoc analysis included 8822 SPRINT participants with at least three follow-up systolic blood pressure (SBP) measurements within the first three months. SBP TTR was calculated using the Rosendaal method of linear interpolation. The primary endpoint was major adverse cardiovascular and cerebrovascular events (MACCE). Cox proportional hazards models and restricted cubic splines (RCS) were used to assess the association between SBP TTR and cardiovascular events.
Women accounted for 35.3% with a mean age of 68.6 ± 9.5 years, having a higher body mass index (p = 0.007) and a lower SBP TTR compared to men (p < 0.001). In the overall population and in women, each standard deviation (SD) increase in SBP TTR was associated with a reduced risk of MACCE (adjusted hazard ratio (HR) 0.89; 95% confidence interval (CI) 0.82–0.97; p = 0.007, and adjusted HR 0.85; 95% CI 0.74–0.99; p = 0.039, respectively) and acute decompensated heart failure (adjusted HR 0.86; 95% CI 0.73–0.99; p = 0.047, and adjusted HR 0.68; 95% CI 0.51–0.92; p = 0.011, respectively), while this was not observed in men. RCS indicated a similar trend in men only when SBP TTR exceeded 39%. Additional adjustments for mean SBP and SBP variability yielded similar outcomes.
The study demonstrates that in women, a higher SBP TTR is associated with a reduced risk of MACCE and acute decompensated heart failure, while in men, a similar trend is observed only when SBP TTR is higher, underscoring the necessity of considering sex differences in personalized blood pressure management strategies.
NCT01206062, https://www.clinicaltrials.gov/expert-search?term=NCT01206062.
Keywords
- hypertension
- systolic blood pressure
- sex differences
- time in target range
Cardiovascular disease (CVD) remains the leading cause of mortality worldwide, with hypertension identified as a central and modifiable risk factor [1]. Effective management of hypertension is essential to reduce cardiovascular risk [2]. While traditional measures of blood pressure control do not account for variability over time or the overall quality of blood pressure management, systolic blood pressure time in target range (SBP TTR) has emerged as a novel measure that incorporates average blood pressure levels, stability, and variability [3, 4]. SBP TTR, defined as the percentage of time during which systolic blood pressure is within a specified range, has been shown to be associated with both cardiovascular events and all-cause mortality in hypertensive individuals, highlighting its potential utility in predicting cardiovascular risk [5, 6].
The effect of sex differences on this association has not been thoroughly investigated, despite the evidence linking SBP TTR to cardiovascular outcomes. This is particularly important given the known differences between women and men in blood pressure regulation and cardiovascular risk [7]. Women, particularly in the postmenopausal period, face unique challenges in blood pressure management due to hormonal changes that can lead to increased arterial stiffness, endothelial dysfunction, and unfavorable lipid distribution [8, 9, 10]. These factors may contribute to a higher cardiovascular risk in older women compared to their male counterparts [11].
Given the potential sex differences in the relationship between SBP TTR and cardiovascular risk, it is crucial to investigate this association in a sex-stratified manner, which will enable the development of more precise and personalized management strategies to improve patient prognosis [12]. To investigate this issue, we conducted a post hoc analysis of data from the Systolic Blood Pressure Intervention Trial (SPRINT), a multicenter, randomized, controlled trial of individuals at high risk for cardiovascular events [13]. We sought to provide insight into the sex-specific relationship between SBP TTR and cardiovascular outcomes.
All datasets and materials relevant to this study have been made publicly accessible via the BioLINCC data retrieved from https://biolincc.nhlbi.nih.gov/home/.
SPRINT was a multicenter, open-label, randomized controlled trial designed for
individuals at increased cardiovascular risk, excluding those with a documented
history of diabetes mellitus or stroke. The trial sought to compare the
effectiveness of an aggressive systolic blood pressure management strategy
(
BP was measured by trained clinic staff. Values were obtained by averaging three seated measurements and were routinely measured at baseline, monthly for the initial three months, and every three months thereafter. SBP TTR is delineated as the proportion of time during the observation period wherein SBP values remain within the predefined target range. Systolic blood pressure (SBP) values throughout the observation period were derived using the linear interpolation method of Rosendaal which assumes a linear change between consecutive measurements to estimate SBP at each time point [15, 16]. As previously documented, the target range for the intensive group was defined as 110–130 mmHg with the upper limit aligning with current guideline recommendations, and the target range for the standard group was set at 120–140 mmHg [17, 18]. Ultimately, the percentage of time within this target range is calculated as the metric of SBP TTR. The calculation of SBP TTR, mean SBP, and SBP variability were based on all SBP data within the initial three months of the study.
Structured interviews were conducted quarterly with participants to ascertain the occurrence of potential events. The primary endpoint was major adverse cardiovascular and cerebrovascular events (MACCE), comprising stroke, non-fatal myocardial infarction, non-myocardial infarction acute coronary syndrome, cardiac death, and acute decompensated heart failure. Secondary endpoints encompassed the discrete constituents of MACCE.
Quantitative data following a normal distribution were characterized by the mean
8822 patients were included in the final analysis, with the exclusion of 539 participants due to the absence of a minimum of three follow-up SBP measurements within the initial three months or incomplete covariate information.
In the overall population, the mean age was 67.9
| Women (n = 3114) | Men (n = 5708) | |||||||||
| TTR | TTR | TTR | TTR | p value | TTR | TTR | TTR | TTR | p value | |
| 0% to |
14% to |
38% to |
64% to 100% | 0% to |
14% to |
38% to |
64% to 100% | |||
| (n = 762) | (n = 876) | (n = 774) | (n = 702) | (n = 1368) | (n = 1462) | (n = 1343) | (n = 1535) | |||
| Intensive arms | 210 (27.6) | 464 (53.0) | 465 (60.1) | 437 (62.3) | 288 (21.1) | 779 (53.3) | 773 (57.6) | 1005 (65.5) | ||
| Age, years | 69.2 |
68.5 |
68.4 |
68.3 |
0.275 | 67.5 |
68.0 |
67.8 |
67.1 |
0.048 |
| BMI, kg/m2 | 29.5 |
30.1 |
30.2 |
30.6 |
0.016 | 29.6 |
29.7 |
29.8 |
29.8 |
0.663 |
| White | 427 (56.0) | 486 (55.5) | 430 (55.6) | 400 (57.0) | 0.447 | 937 (68.5) | 1043 (71.3) | 985 (73.3) | 1105 (72.0) | 0.010 |
| Current smoking | 97 (12.7) | 104 (11.9) | 103 (13.3) | 91 (13.0) | 0.839 | 187 (13.7) | 182 (12.4) | 187 (13.9) | 184 (12.0) | 0.342 |
| History of CVD | 105 (13.8) | 140 (16.0) | 123 (15.9) | 106 (15.1) | 0.594 | 302 (22.1) | 366 (25.0) | 321 (23.9) | 308 (20.1) | 0.007 |
| 10-y ASCVD risk | 17 (16–19) | 17 (16–18) | 17 (16–18) | 17 (16–18) | 18 (16–20) | 18 (16–19) | 18 (16–19) | 17 (15–19) | ||
| Renal insufficiency1 | 247 (32.4) | 275 (31.4) | 241 (31.1) | 223 (31.8) | 0.954 | 316 (23.1) | 377 (25.8) | 343 (25.5) | 347 (22.6) | 0.096 |
| Baseline SBP, mmHg | 149.8 |
147.7 |
146.4 |
145.1 |
146.6 |
144.6 |
143.0 |
141.6 |
||
| Baseline DBP, mmHg | 80.0 |
80.0 |
79.9 |
79.7 |
0.961 | 81.2 |
80.7 |
80.1 |
79.7 |
0.002 |
| TTR, % | 0 (0–6) | 26 (20–32) | 50 (44–57) | 80 (71–92) | 0 (0–4) | 27 (21–32) | 51 (46–57) | 82 (72–96) | ||
Data are presented as mean
1eGFR
The antihypertensive intervention strategies, stratified by the number of
antihypertensive agents, showed no statistically significant differences between
men and women, either in total or when categorized by SBP TTR quartile of the
overall population. Notably, in patients who were administered a single type of
antihypertensive agent, women exhibited a lower SBP TTR compared with men (40%
There were 673 MACCE in the overall population after a follow-up period of 3.8 (3.3–4.4) years (Supplementary Table 4). The incidence rate of MACCE in the overall population was 20.7 per 1000 person-years (95% confidence interval [CI], 19.1–22.3), which was 17.4 per 1000 person-years (95% CI, 15.0–20.0) for women and 22.5 per 1000 person-years (95% CI, 20.5–24.6) for men (Table 2).
| IR1 (95% CI) | Unadjusted | Adjusted3 | ||||
| HR2 (95% CI) | p value | HR2 (95% CI) | p value | |||
| Major adverse cardiovascular and cerebrovascular events | ||||||
| Overall | 20.7 (19.1–22.3) | 0.84 (0.78–0.91) | 0.89 (0.82–0.97) | 0.007 | ||
| Women | 17.4 (15.0–20.0) | 0.83 (0.72–0.96) | 0.013 | 0.85 (0.74–0.99) | 0.039 | |
| Men | 22.5 (20.5–24.6) | 0.84 (0.77–0.92) | 0.91 (0.83–1.01) | 0.071 | ||
| Nonfatal myocardial infarction | ||||||
| Overall | 7.8 (6.9–8.8) | 0.84 (0.74–0.95) | 0.007 | 0.91 (0.80–1.04) | 0.167 | |
| Women | 6.6 (5.1–8.2) | 0.82 (0.65–1.04) | 0.103 | 0.90 (0.70–1.14) | 0.373 | |
| Men | 8.5 (7.3–9.8) | 0.84 (0.72–0.98) | 0.024 | 0.91 (0.78–1.07) | 0.270 | |
| Non-MI ACS | ||||||
| Overall | 2.5 (2.0–3.1) | 0.97 (0.78–1.20) | 0.785 | 0.95 (0.76–1.20) | 0.693 | |
| Women | 1.3 (0.8–2.2) | 1.24 (0.77–1.99) | 0.380 | 1.27 (0.77–2.07) | 0.348 | |
| Men | 3.1 (2.4–4.0) | 0.90 (0.70–1.14) | 0.381 | 0.89 (0.69–1.16) | 0.407 | |
| Stroke | ||||||
| Overall | 4.8 (4.1–5.6) | 0.71 (0.60–0.83) | 0.73 (0.61–0.88) | 0.001 | ||
| Women | 4.9 (3.7–6.4) | 0.71 (0.54–0.94) | 0.017 | 0.69 (0.51–0.92) | 0.013 | |
| Men | 4.7 (3.8–5.7) | 0.70 (0.57–0.87) | 0.001 | 0.77 (0.62–0.97) | 0.028 | |
| Acute decompensated heart failure | ||||||
| Overall | 6.0 (5.1–6.8) | 0.81 (0.70–0.94) | 0.005 | 0.86 (0.73–0.99) | 0.047 | |
| Women | 5.0 (3.8–6.4) | 0.67 (0.51–0.89) | 0.005 | 0.68 (0.51–0.92) | 0.011 | |
| Men | 6.5 (5.4–7.6) | 0.87 (0.73–1.03) | 0.100 | 0.94 (0.78–1.13) | 0.526 | |
| Cardiovascular death | ||||||
| Overall | 3.8 (3.1–4.5) | 0.86 (0.72–1.03) | 0.102 | 0.94 (0.78–1.13) | 0.517 | |
| Women | 2.9 (2.0–4.0) | 1.04 (0.75–1.45) | 0.809 | 1.16 (0.82–1.65) | 0.391 | |
| Men | 4.3 (3.4–5.2) | 0.79 (0.64–0.98) | 0.032 | 0.88 (0.70–1.11) | 0.277 | |
ACS, acute coronary syndrome; HR, hazard ratio; IR, incidence rate; MI, myocardial infarction; SD, standard deviation; SBP, systolic blood pressure; CI, confidence interval.
1IR per 1000 person-years;
2HR per one SD increase in time in target range;
3adjusted for age, race, treatment group, 10-year cardiovascular risk score, baseline SBP, body mass index, and renal insufficiency for women/men and additionally for sex for the overall population.
Each increment of one SD in SBP TTR was associated with a reduction in the MACCE
risk in the overall population and the women subgroup in the unadjusted model
(hazard ratio (HR), 0.84; 95%CI, 0.78–0.91; p
Restricted cubic splines were utilized to investigate the relationship, with the median SBP TTR of the respective population as the reference. As depicted in Fig. 1, an elevation in SBP TTR was inversely correlated with the risk of MACCE for the overall population and the women subgroup. However, for men, an inverse relationship between SBP TTR and MACCE risk was clearly observed only when SBP TTR exceeded 39%. Similar outcomes were observed in the relationship between SBP TTR and acute decompensated heart failure (Supplementary Fig. 2).
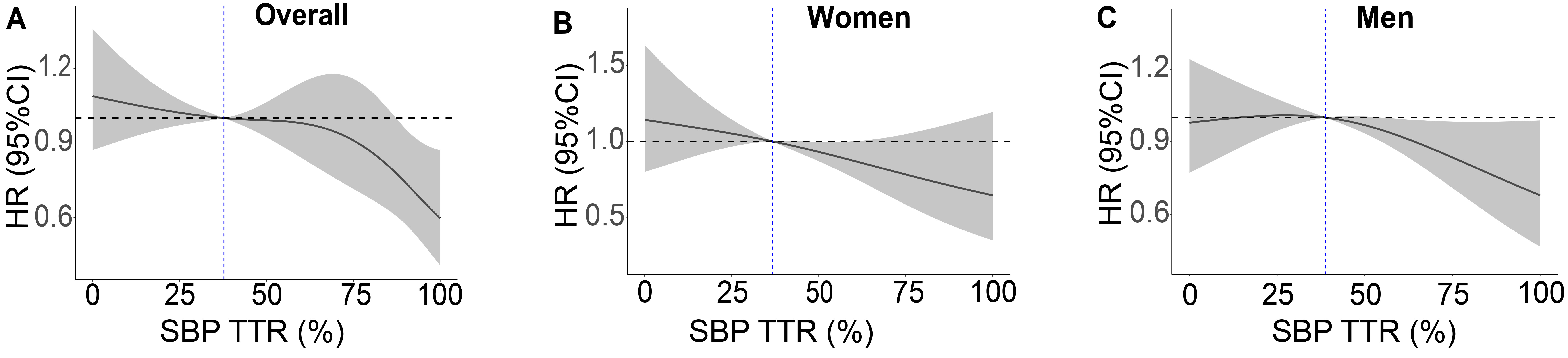 Fig. 1.
Fig. 1.
Relationship between systolic blood pressure time in target range (SBP TTR) and major adverse cardiovascular and cerebrovascular events in (A) the overall population and by sex: (B) women; (C) men. Restricted cubic splines were performed with the median SBP TTR as reference and the model was fully adjusted for age, race, treatment group, 10-year cardiovascular risk score, body mass index, renal insufficiency, and baseline SBP for women and men separately, with an additional adjustment for sex in the overall population.
To further explore this relationship, we stratified the participants via their respective SBP TTR quartiles: the overall population at 38% (14%–64%), women at 37% (14%–61%), and men at 39% (16%–67%). Elevated SBP TTR levels were correlative with a trend for reduced MACCE risk in the overall population and women, but in men, this trend was only observed when SBP TTR exceeded 39% (Fig. 2 and Supplementary Table 5).
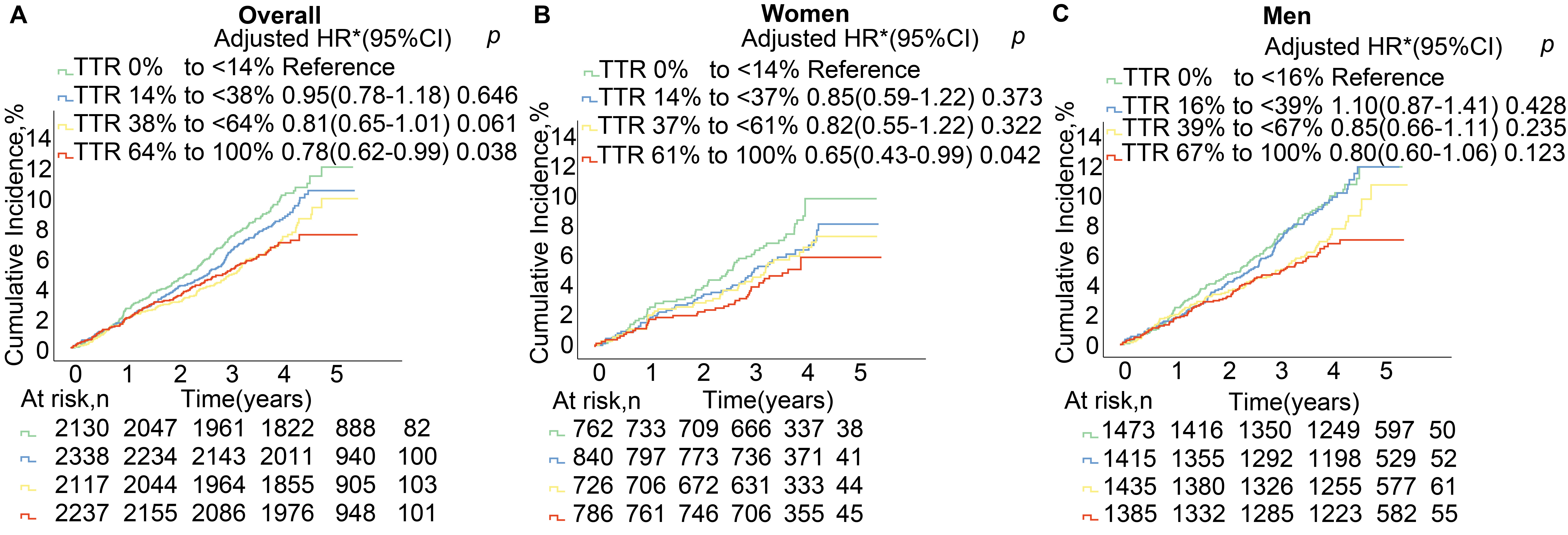 Fig. 2.
Fig. 2.
Systolic blood pressure time in target range (SBP TTR) categories and risk of major adverse cardiovascular and cerebrovascular events in (A) the overall population and by sex: (B) women; (C) men. Kaplan–Meier estimates and adjusted hazard ratios (adjusted HR; with 95% confidence intervals (CIs)) for age, race, treatment group, 10-year cardiovascular risk score, body mass index, renal insufficiency, and baseline SBP for women/men and additionally for sex for the overall population. To accurately reflect the impact of sex differences, our analysis utilized quartile groupings based on the SBP TTR for distinct populations: the overall population at 38% (14%–64%), women at 37% (14%–61%), and men at 39% (16%–67%).
To further validate the sex-specific independent predictive role of SBP TTR on MACCE risk, we additionally adjusted for mean SBP or SBP variability in the adjusted model. After additional adjustment for mean SBP, a reduction in the risk of MACCE was observed with a one SD increment in SBP TTR in the overall population (adjusted HR 0.90; 95% CI, 0.82–0.98; p = 0.016) and women (adjusted HR 0.84; 95% CI, 0.72–0.98; p = 0.030), while for men, the association remained non-significant (adjusted HR 0.93; 95% CI, 0.84–1.04; p = 0.189). Similar sex differences were also seen in the risk for acute decompensated heart failure (adjusted HR, 0.85; 95% CI, 0.72–0.999; p = 0.049 for overall, adjusted HR, 0.64; 95% CI, 0.47–0.87; p = 0.004 for women, and adjusted HR, 0.97; 95% CI, 0.79–1.18; p = 0.732 for men). Similar results were noted after additional adjustment for SBP variability (Supplementary Tables 6,7).
Our investigation has elucidated a sex-specific relationship between SBP TTR and cardiovascular event risk, indicating that an increment in SBP TTR is correlated with a reduced risk of MACCE and acute decompensated heart failure in the overall population and in women, while this relationship was not observed in men. In men, a similar trend was noted only when SBP TTR levels exceeded 39%. Furthermore, after additional adjustment for blood pressure variability and mean SBP, the sex-specific relationship between SBP TTR and cardiovascular event risk remained consistent.
Hypertension serves as a key factor in the pathogenesis of atherosclerosis via increasing vascular shear stress, which leads to arterial wall thickening, development of atherosclerosis, plaque vulnerability, and ultimately an elevated risk of cardiovascular events [19, 20, 21, 22]. SBP TTR serves as an indicator of effective blood pressure control, accounting for levels, stability, and duration of blood pressure. Higher SBP TTR signifies lower and more consistent levels of vascular shear stress, leading to a reduction in arterial medial thickening and preventing tears and fragmentation in the internal elastic lamina [23]. Moreover, higher levels of SBP TTR diminish the mechanical stimulation of the vascular wall, decreasing the activation of intracellular signaling pathways triggered by mechanical factors and stabilizing cellular functions [24]. Eventually, patients with hypertension benefit from more effective blood pressure control which reduces the risk of cardiovascular diseases.
A previous study has demonstrated an inverse relationship between SBP TTR and the risk of MACCE and acute decompensated heart failure in patients with hypertension, and our study has yielded similar results [17]. Notably, when first investigating sex-specific relationships, the trend of reduced MACCE and acute decompensated heart failure risk with increasing SBP TTR was observed only when SBP TTR exceeded 39%. These findings underscore the importance of implementing sex-specific and personalized blood pressure management strategies to improve cardiovascular outcomes.
Our study has elucidated a sex-specific relationship between SBP TTR and cardiovascular risk, which we hypothesize may be due to differences in sex hormones. Endogenous estrogen exerts cardioprotective effects by regulating nitric oxide (NO) biosynthesis and bioavailability, inhibiting the renin-angiotensin-aldosterone system (RAAS), modulating the sympathetic nervous system, and reducing endothelin-1 (ET-1) levels, thereby promoting vasodilation, inhibiting vascular remodeling, and decreasing arterial stiffness and cardiovascular risk [25, 26, 27, 28]. In postmenopausal women, estrogen deficiency also exacerbates endothelial dysfunction, promotes the rupture of arterial elastin, leads to collagen accumulation, and increases arterial stiffness [29]. In addition, estrogen deficiency is associated with the progression of metabolic syndrome, leading to adverse lipid distribution in elderly women [30]. Obesity, particularly a high BMI, is linked to adipose tissue dysfunction, elevated oxidative stress, activation of the RAAS system, and overactivity of the sympathetic nervous system, promoting chronic vascular inflammation and exacerbating cardiovascular risk [31]. These changes collectively result in greater arterial stiffness in postmenopausal women compared to age-matched men, with faster progression of vascular dysfunction, ultimately leading to higher cardiovascular risk.
In a large cross-sectional study involving 18,326 women, the proportion of
postmenopausal women was approximately 90% in the 54–55 age group (n = 2324),
96% in the 56–57 age group (n = 2448), and 98% in the 58–59 age group (n =
2621) [32]. This indicates that the women in our study primarily belong to a
postmenopausal population characterized by significantly reduced estrogen levels.
These women also exhibited higher BMI levels compared to men, potentially due to
adverse lipid distribution resulting from estrogen deficiency. The SBP TTR levels
in women were also lower than those in men, despite using a consistent number of
medications, indicating poorer blood pressure control in women. These findings
suggest that the women in our study have a higher risk of hypertension and
cardiovascular disease due to decreased estrogen levels. This presents greater
challenges for optimizing blood pressure management and improving cardiovascular
outcomes. Consequently, they may gain more significant benefits from effective
blood pressure control reflected by increased SBP TTR, compared to men. In
contrast, men only exhibit significant benefits after achieving higher SBP TTR
levels (SBP TTR
The findings of this study underscore the importance of considering sex-specific differences in clinical blood pressure management strategies. Based on the findings of this study, we recommend implementing sex-specific management strategies for hypertensive patients in clinical practice. For female patients, particularly postmenopausal women, higher SBP TTR levels are strongly associated with reduced risks of MACCE and acute decompensated heart failure. Therefore, prioritizing SBP TTR stability at elevated levels is advised. Pharmacologic choices should favor agents proven to enhance blood pressure stability, with regular monitoring to ensure sustained SBP TTR over time. For male patients, while elevated SBP TTR also provides protective benefits, the clinical focus should be placed on maintaining higher thresholds (e.g., above 39%). Implementing sex-specific follow-up intervals and blood pressure monitoring strategies may enhance SBP TTR control, thereby improving cardiovascular outcomes.
Our study has several limitations. First, the population was subject to rigorous blood pressure management, which limited fluctuations in blood pressure and converged the mean blood pressure and time in the target range. Second, the method for SBP TTR calculation is complex and the results are significantly affected by the selection of time periods for the analysis. Finally, this study is a post hoc analysis based on data from the SPRINT trial, a prospective randomized controlled trial that targeted a high cardiovascular risk population without a history of diabetes or stroke, which may have influenced the generalizability of the results to a large population.
In summary, there are significant sex-specific differences in the relationship between SBP TTR and the risk of cardiovascular events. In women, an elevation in SBP TTR is correlated with a reduced risk of MACCE and acute decompensated heart failure, while in men, a similar trend is observed only when SBP TTR reaches higher levels. These suggest that improving SBP TTR may be crucial in reducing cardiovascular event risk in patients with hypertension, and it is essential to consider sex differences and develop personalized blood pressure management strategies to optimize cardiovascular outcomes.
All datasets and materials relevant to this study have been made publicly accessible via the BioLINCC data retrieved at https://biolincc.nhlbi.nih.gov/home/. All SPRINT anonymized data can be found at the National Heart, Lung and Blood Institute (NHLBI) Biologic Specimen and Data Repository (https://biolincc.nhlbi.nih.gov/home/).
YZ: Conceptualization, Methodology, Formal analysis, Writing—original draft. WZ: Conceptualization, Methodology, Data curation, Formal analysis, Writing—review & editing. CJ: Conceptualization, Data curation, Writing—review & editing. WH, WG, and YY: Conceptualization, Writing—review & editing. XW: Conceptualization, Methodology, Funding acquisition, Writing—review & editing. CM: Conceptualization, Data curation, Investigation, Resources, Writing—review & editing. SN: Conceptualization, Methodology, Funding acquisition, Writing—review & editing, Project administration. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki. The research protocol was approved by each clinical site (IRB00014304), and all patients or their families/legal guardians provided signed informed consent.
All authors thank the SPRINT (Systolic Blood Pressure Intervention Trial) investigators for making the study data available for public use through the National Heart, Lung, and Blood Institute BioLINCC Biologic Specimen and Data Repository.
Dr. Xiao Wang was funded by National Key Research & Development Program of China (2022YFC2505600) and Beijing Municipal Natural Science Foundation Grant (JQ24039). Dr. Shaoping Nie was funded by Beijing Municipal Science & Technology Commission, China (Z221100003522027), Beijing Hospitals Authority Clinical Medicine Development of special funding support (ZLRK202318), and National Natural Science Foundation of China (82270258).
Dr. Shaoping Nie reports research grants to the Institution from Boston Scientific, Abbott, Jiangsu Hengrui Pharmaceuticals, China Resources Sanjiu Medical & Pharmaceuticals, East China Pharmaceuticals. Dr. Changsheng Ma reports honoraria from Bristol-Myers Squibb (BMS), Pfizer, Johnson & Johnson, Boehringer-Ingelheim (BI), Bayer and AstraZeneca for giving lectures. The other authors have no conflicts of interest to declare.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM26262.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


