1 Department of Cardiology, First Clinical Medical College, China Three Gorges University, 443002 Yichang, Hubei, China
2 Hubei Key Laboratory of Ischemic Cardiovascular Disease, 443003 Yichang, Hubei, China
3 Hubei Provincial Clinical Research Center for Ischemic Cardiovascular Disease, 443003 Yichang, Hubei, China
4 Department of Traditional Chinese Medicine, Xianning Central Hospital, The First Affiliated Hospital of Hubei University of Science and Technology, 437000 Xianning, Hubei, China
5 Division of Cardiology, Department of Internal Medicine, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, 430030 Wuhan, Hubei, China
†These authors contributed equally.
Abstract
Atrial fibrillation (AF) is a common cardiac arrhythmia strongly associated with an imbalance between T helper 17 (Th17) cells and regulatory T cells (Treg). Secreted phosphoprotein 1 (SPP1), an immune signaling molecule implicated in AF pathogenesis, may shift the Th17/Treg cell balance in non-valvular AF (NVAF). This study aimed to explore the regulatory effects of SPP1 on the balance of Th17 and Treg cells in NVAF.
Venous blood samples were collected from 58 patients with NVAF (observation group) and 58 age- and sex- matched healthy controls (control group). The serum concentrations of SPP1, along with the percentages of Treg and Th17 cells, and the levels of their associated cytokines, were measured. Correlation analysis was employed to evaluate the association between serum SPP1 levels and the Treg/Th17 cell ratio. In parallel, an experimental rat model of AF was established to investigate the expression of SPP1, related inflammatory factors, and fibrin within the left atrial tissue.
NVAF patients showed significantly higher serum levels of SPP1 and certain inflammatory cytokines (interleukin (IL)-17A and IL-23) than the controls. NVAF patients exhibited increased Th17 cells and elevated collagen I levels. Meanwhile, Treg cell frequency and IL-10 levels were significantly reduced compared to controls. Consequently, the Treg/Th17 ratio was significantly lower in NVAF patients. Notably, a significant inverse correlation was identified between serum SPP1 concentrations and the Treg/Th17 ratio. Consistent results were also obtained in animal models of AF, further supporting these findings.
Our findings suggest that elevated SPP1 levels disrupt the Treg/Th17 cell balance in NVAF patients, promoting inflammation and fibrosis. These findings indicate that SPP1 represents a promising therapeutic target for the prevention and management of NVAF.
Keywords
- secreted phosphoprotein 1
- Treg/Th17
- non-valvular atrial fibrillation
- inflammation
- fibrosis
Non-valvular atrial fibrillation (NVAF) is a common tachyarrhythmia strongly
associated with myocardial fibrosis driven by chronic inflammation [1]. Although
atrial fibrillation (AF) has traditionally been regarded as a cardiac rhythm
disorder resulting from atrial myocyte remodeling, emerging evidence increasingly
implicates immune dysregulation in its pathogenesis [2, 3]. Regulatory T cells
(Treg) and T helper 17 cells (Th17) are key CD4+ T cells subsets that
differentiate from naive CD4+ T cells and critically regulate cellular
immunity [4]. As Treg cells suppress immunity while Th17 cells promote
inflammation, the Treg/Th17 ratio serves as a biomarker of inflammatory status
and a predictor of diseases such as atherosclerosis [5, 6]. Given its chronic
inflammatory nature, AF involves complex interactions among various immune cells
and inflammatory cytokines [7, 8]. Disruption of circulating Treg/Th17
homeostasis has also been reported in patients with AF and rheumatoid arthritis
[9]. Moreover, a decreased Treg/Th17 ratio reflects a pro-inflammatory state and
predicts an increased risk of AF following off-pump coronary artery bypass
grafting [10]. Secreted phosphoprotein 1 (SPP1), also known as osteopontin, is an
extracellular matrix protein implicated in various pathological processes,
including calcification, fibrosis, and inflammation [11, 12, 13]. Recent research
has underscored the potential involvement of SPP1 in the pathogenesis of AF. SPP1
in atrial fibroblasts has been reported to promote atrial fibrosis through the
Akt/glycogen synthase kinase-3 (GSK)-3
Fifty-eight patients with non-valvular AF treated at the first clinical medical
college of China Three Gorges University and Tongji Hospital between January 2023
and December 2024 were included in the observation group. The inclusion criteria
were as follows: (1) electrocardiograph (ECG) showing absence of P waves, f-waves
at 350–600 bpm, and irregularly irregular QRS complexes, as defined by current
guidelines [18]; and (2) documented AF duration
Sixteen specific pathogen-free (SPF) Sprague-Dawley (SD) rats, aged 6 to 8 weeks
and weighing between 200 and 220 grams, were supplied by the Animal Experiment
Center of Three Gorges University. An AF animal model was established in eight SD
rats through daily intravenous injection of a mixture of acetylcholine and
calcium chloride (ACH-CaCl2, 1 mg
Five milliliters of fasting venous blood were collected from each patient and
processed using density gradient centrifugation. At room temperature, the blood
was combined with an equal volume of pre-warmed phosphate-buffered saline (PBS)
and lymphocyte separation medium in a 50 mL centrifuge tube, followed by
centrifugation at 1700 rpm for 15 minutes. After centrifugation, the sample was
separated into four distinct layers. The mononuclear cell layer was meticulously
transferred to a 15 mL tube and subsequently washed with PBS, using a volume 2–5
times greater than that of the cell layer, and centrifuged again (1000 rpm, 10
min, room temperature). After discarding the supernatant, the cells were
resuspended in PBS to achieve a PBMC concentration of 2
For this analysis, microcentrifuge tubes for antibody incubation were divided into five groups: one for blank control, three for single-staining of T cells (CD4+, CD25+, and CD127–, respectively), and one for triple-staining of Treg cells (CD4+CD25+CD127–). The antibodies employed for subsequent staining included anti-human CD4-FITC (Ebioscience, RRID: AB_1272074, San Diego, CA, USA), anti-human CD25-PE (Ebiosciences, RRID: AB_2744720, San Diego, CA, USA), and anti-human CD127-PE-CY7 (Ebiosciences, RRID: AB_2043801, San Diego, CA, USA). Subsequently, 5 µL of the appropriate antibodies were added to the tubes for each group, with a mixture of the three antibodies used for the Treg cell triple-staining tube. Each tube contained 100 µL of cell suspension (isolated PBMCs). The mixture was incubated at 4 °C in the dark for 30 minutes, then ice-cold PBS (4 °C) was added, and the suspension was centrifuged at 1500 rpm for 5 minutes to form a cell pellet. Following the removal of the supernatant, the cells were washed twice with 2 mL of ice-cold PBS and resuspended in 300 µL PBS. The prepared samples were then analyzed using a flow cytometer (Beckman Kurt Technology, model: CytoFLEX, Brea, CA, USA).
The isolated PBMCs were placed in 24-well plates and stimulated for 6 h at 37
°C with 5% CO2 (ESCO, CLM-1708-8-NF, Singapore) in the presence of
a stimulation cocktail containing PMA (25 ng/mL, Sigma, CAS No.: 108-65-6, St.
Louis, MO, USA), ionomycin (1 µg/mL, Sigma, CAS No.: 56092-82-1, St.
Louis, MO, USA), monensin (1.4 µg/mL, MCE, CAS No.: 22373-78-0,
Monmouth Junction, NJ, USA), and brefeldin A (3 µg/mL, MCE, CAS
No.: 20350-15-6, Monmouth Junction, NJ, USA). Subsequently, the PBMCs were
collected, washed with PBS at 37 °C, centrifuged at 1500 rpm for 5
minutes at room temperature, and the supernatant was discarded. The cells were
then resuspended in PBS to achieve a concentration of 2
The transcription factor Foxp3 is specifically involved in the
proliferation and differentiation of regulatory T cells, whereas RORC
functions as a key transcription factor for Th17 cells. Total RNA of PBMCs was
extracted utilizing the TRIzol method. Subsequently, reverse transcription of RNA
to cDNA was performed using a cDNA synthesis kit (Sigma, CAS No.: 11483188001,
USA), followed by RT-PCR using the SYBR premix Kit (Takara company, Kusatsu,
Shiga, Japan). PCR conditions were as follows: 95 °C for 2 minutes, 95
°C for 15 seconds
Blood samples were collected in standard tubes, allowed to clot at room temperature for 30 minutes and then centrifuged at 3000 rpm for 20 minutes. The resulting supernatant serum was then separated and stored at –80 °C until further analysis. The levels of SPP1 (Shanghai Jianglai Biotechnology Co., Ltd., Cat. No.: JL10368, Shanghai, China), collagen I (Shanghai Jianglai Biotechnology Co., Ltd., Cat. No.: JL47278, Shanghai, China), IL-10 (Xinboseng Biotechnology Co., Ltd., Cat. No.: EHC009.96, Shanghai, China), IL-17A (Xinboseng Biotechnology Co., Ltd., Cat. No.: EHC170.96, Shanghai, China), and IL-23 (Xinboseng Biotechnology Co., Ltd., Cat. No.: EHC171.96, Wuhan, China) were measured following the manufacturer’s ELISA kit instructions.
The levels of SPP1 (Shanghai Jianglai Biotechnology Co., Ltd., Cat. No.: JL14517, Shanghai, China), collagen I (Wuhan Jilide Biotechnology Co., Ltd., Cat. No.: J23744, Wuhan, China), IL-10 (Wuhan Jilide Biotechnology Co., Ltd., Cat. No.: J22449, Wuhan, China), IL-17A (Wuhan Jilide Biotechnology Co., Ltd., Cat. No.: J23469, Wuhan, China), and IL-23 (Wuhan Jilide Biotechnology Co., Ltd., Cat. No.: J22374, Wuhan, China) in the left atrium of rats was detected using ELISA following the manufacturer’s guidelines.
The left atrial tissue samples were embedded in paraffin and sectioned at 5 µm thickness. Masson’s trichrome staining was employed to assess the extent of fibrosis in the left atrium. These sections were examined under a microscope, and the measurements were taken using Image Pro-Plus software (Media Cybernetics Inc., Silver Spring, MD, USA).
HDL-C, LDL-C, TC, TG and FBG were measured by the clinical laboratory department of Tongji Hospital.
Statistical analyses were conducted utilizing SPSS version 22.0 (IBM Corp.,
Armonk, NY, USA). Continuous variables are expressed as mean
Baseline characteristics showed no significant differences between the
observation and control groups in terms of age, sex, hypertension, coronary
artery disease, smoking status, fasting glucose, serum creatinine, or lipid
profile parameters (p
| Observation group (n = 58) | Control group (n = 58) | t/ |
p | |
| Gender, Male (%) | 34 (58.6) | 37 (63.8) | 0.327 | 0.568 |
| Age (years) | 67.8 |
66.4 |
1.087 | 0.279 |
| Hypertension, n (%) | 42 (72.4) | 38 (65.5) | 0.644 | 0.422 |
| Coronary heart disease, n (%) | 35 (60.3) | 37 (63.8) | 0.146 | 0.702 |
| Ejection fraction (%) | 47.35 |
60.13 |
8.259 | |
| Left atrial diameter (mm) | 40.13 |
32.52 |
7.681 | |
| Left ventricular end diastolic diameter (mm) | 44.23 |
32.52 |
13.141 | |
| Smoking history, n (%) | 19 (32.8) | 21 (36.2) | 0.153 | 0.696 |
| Fasting blood glucose (mmol/L) | 4.5 |
4.6 |
–1.523 | 0.130 |
| Serum creatinine (umol/L) | 76.1 |
79.5 |
1.162 | 0.248 |
| Total cholesterol (mmol/L) | 4.2 |
4.1 |
0.512 | 0.609 |
| Triglycerides (mmol/L) | 1.6 |
1.5 |
0.716 | 0.475 |
| High-density lipoprotein (mmol/L) | 1.3 |
1.4 |
–1.523 | 0.130 |
| Low-density lipoprotein (mmol/L) | 2.3 |
2.2 |
0.716 | 0.475 |
The proportions of CD4+CD25+CD127– Treg cells and
CD3+CD8–IL17+ Th17 cells in PBMCs were determined. Compared to
the control group, the observation group exhibited a significant reduction in the
proportion of CD4+CD25+CD127– Treg cells (p
| Control group (n = 58) | Observation group (n = 58) | t | p | |
| CD4+CD25+CD127– Treg (%) | 5.9 |
3.9 |
13.359 | |
| CD3+CD8–IL17+ Th17 (%) | 1.1 |
2.4 |
–13.730 | |
| Treg/Th17 | 5.3 |
1.6 |
52.326 |
*p
In the observation group, RORC mRNA was significantly
up-regulated, whereas Foxp3 mRNA was down-regulated (p
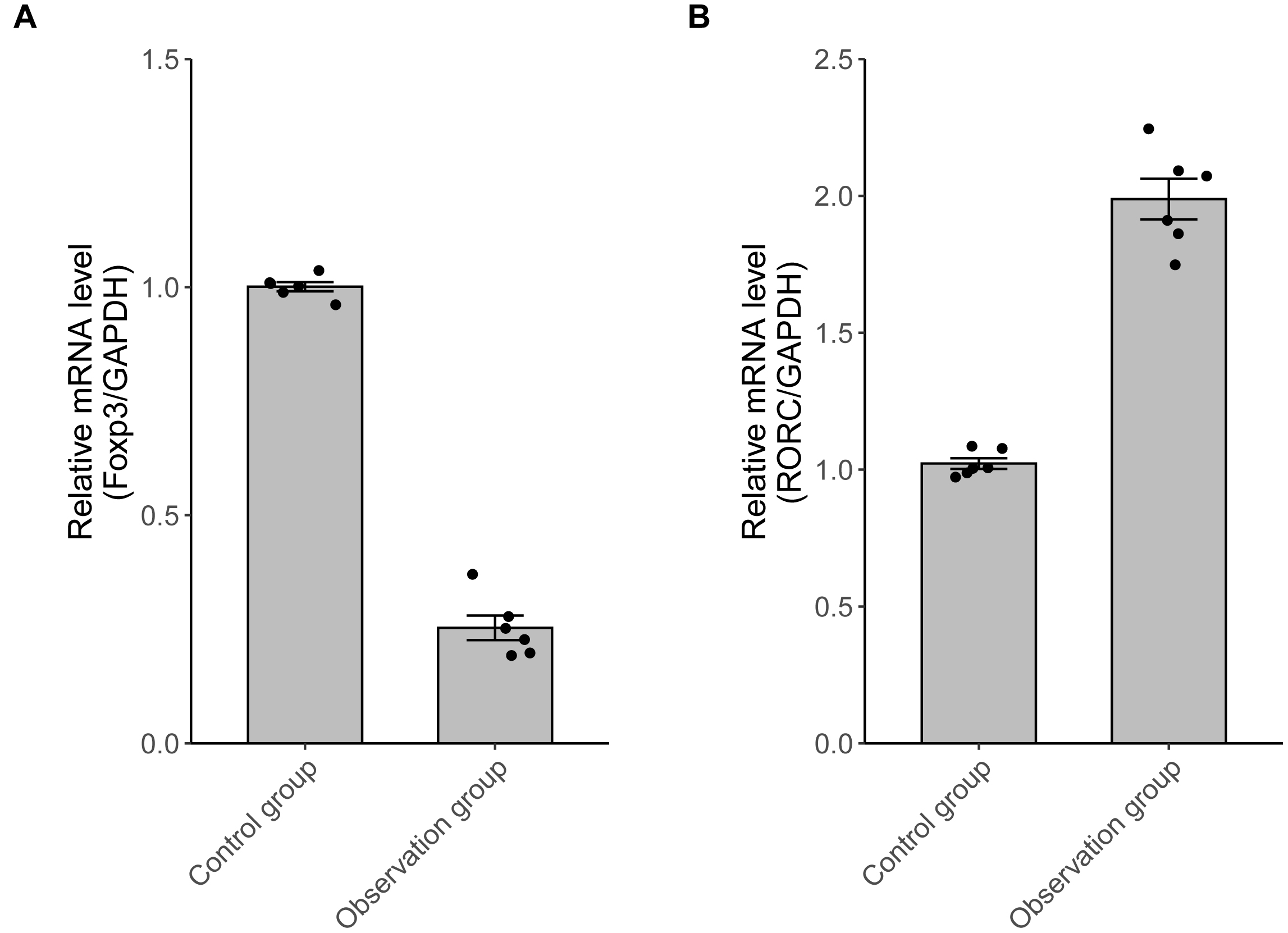 Fig. 1.
Fig. 1.
The Foxp3 and RORC mRNA expressions. (A) In
the observation group, Foxp3 mRNA was lower (n = 6, p
Serum levels of SPP1, collagen I, IL-10, IL-17A, and IL-23 were measured using
ELISA. Compared to the control group, the observation group exhibited a
significant increase in SPP1 and collagen I concentrations (p
| Control group (n = 58) | Observation group (n = 58) | t | p | |
| SPP1 (ng/mL) | 6.7 |
16.3 |
–20.277 | |
| Collagen I (ug/mL) | 1.1 |
1.9 |
16.898 | |
| IL-10 (pg/mL) | 421.3 |
199.6 |
42.687 | |
| IL-17A (pg/mL) | 17.4 |
49.8 |
–29.652 | |
| IL-23 (pg/mL) | 37.6 |
77.5 |
–33.135 |
*p
After tail-vein injection, all eight rats exhibited typical AF waveforms: f
waves replaced P waves and R-R intervals were irregular. The duration of AF in
all rats exceeded 10 seconds (18.92
| Normal group (n = 8) | Atrial fibrillation group (n = 8) | t | p | |
| SPP1 (pg/mgprot) | 2.4 |
9.1 |
–15.321 | |
| IL-10 (pg/mgprot) | 64.7 |
32.8 |
20.190 | |
| IL-17A (pg/mgprot) | 57.7 |
103.4 |
15.738 | |
| IL-23 (pg/mgprot) | 26.4 |
49.5 |
13.356 |
*p
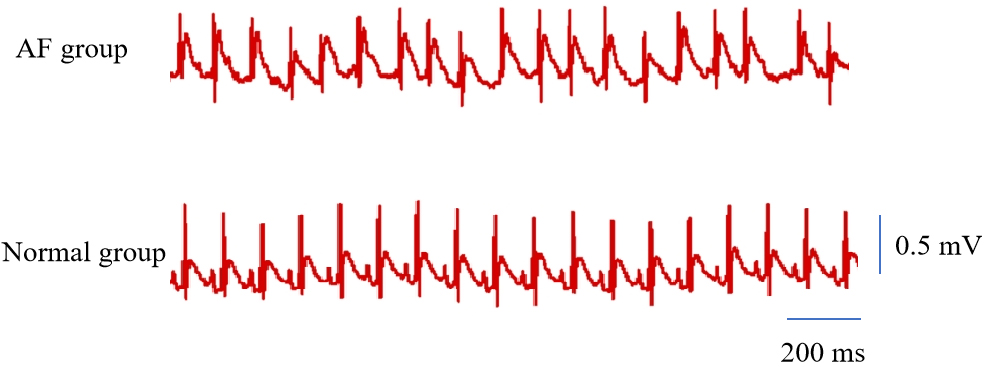 Fig. 2.
Fig. 2.
A typical ECG of AF. AF, atrial fibrillation; ECG, electrocardiograph.
Atrial fibrosis was assessed using Masson’s trichrome staining. Our findings indicate that left atrial fibrosis was significantly more extensive in the AF group than that of the control group (Fig. 3).
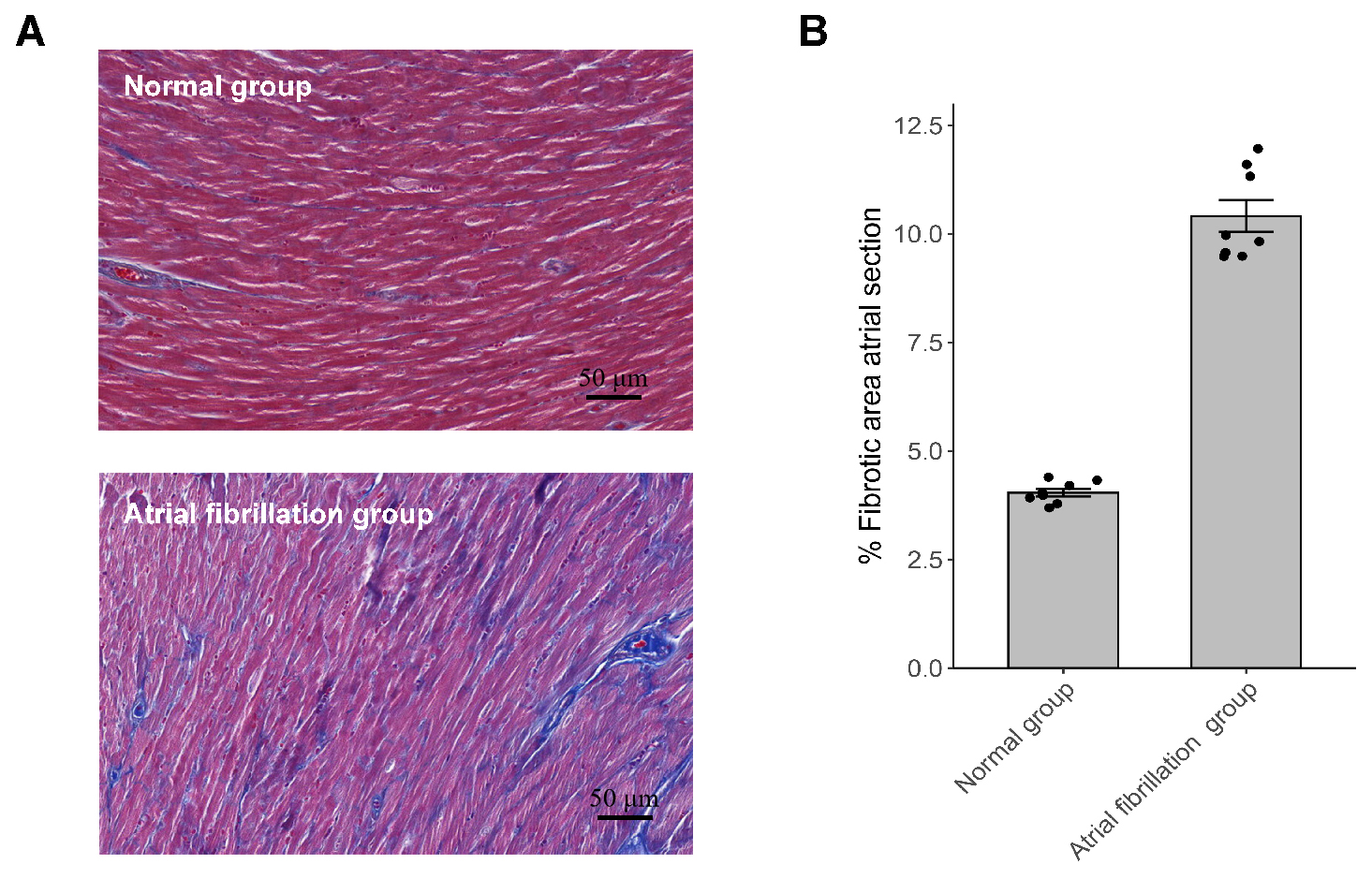 Fig. 3.
Fig. 3.
Atrial fibrosis was assessed by Masson staining. (A)
Representative images of Masson staining of the left atrium (magnification,
Correlation analysis revealed a statistically significant inverse relationship
between SPP1 and the Treg/Th17 ratio (n = 58, r = –0.655, p
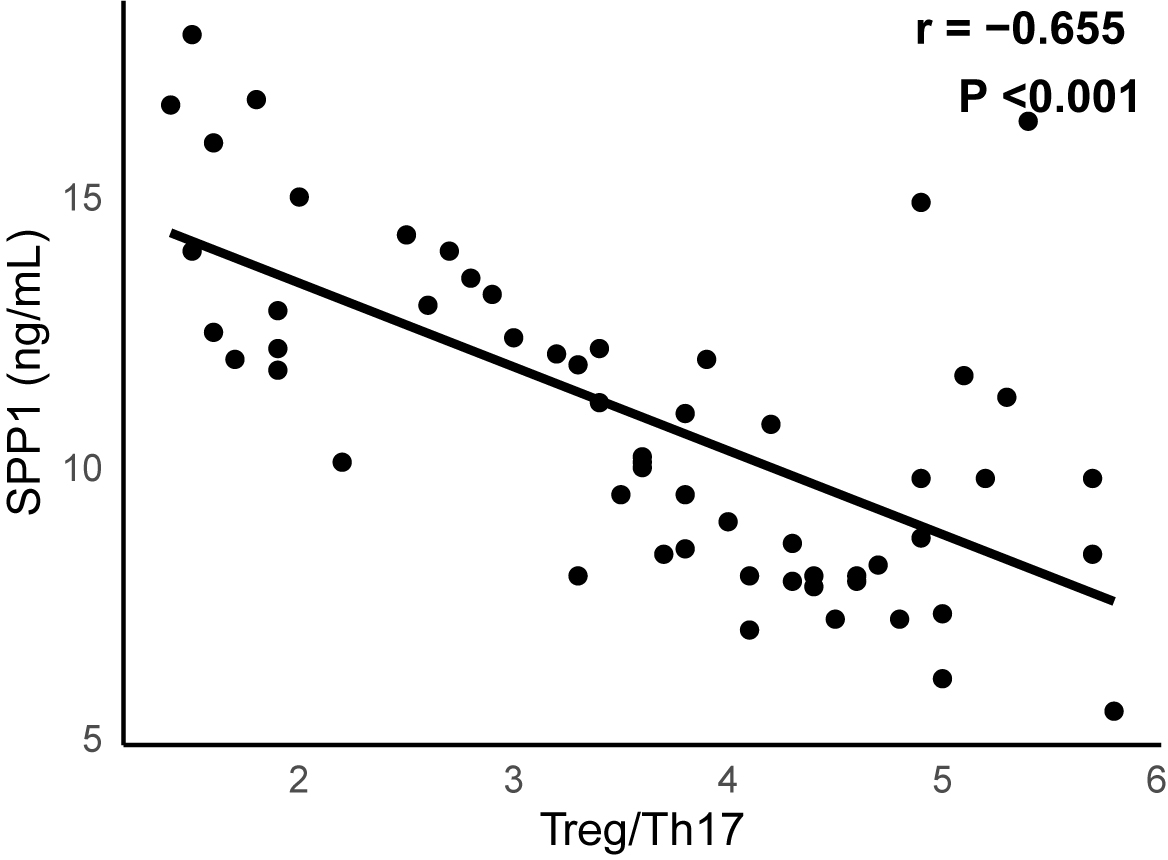 Fig. 4.
Fig. 4.
A significant negative correlation between SPP1 and Treg/Th17 (n
= 58, r = –0.655, p
SPP1, also known as osteopontin, has gained recognition for its role in immune modulation and fibrotic remodeling in various cardiovascular diseases [19, 20]. In this study, we identified significantly elevated serum SPP1 levels in patients with NVAF, accompanied by a marked reduction in Treg cells and an increase in Th17 cells. As a result, the Treg/Th17 ratio was markedly reduced in NVAF patients. These immunological changes correlated with elevated pro-inflammatory cytokines (IL-17A and IL-23) and reduced anti-inflammatory markers (IL-10). Similar findings were observed in animal models of AF, further supporting these observations.
Furthermore, we observed a strong inverse correlation between serum SPP1 levels and the Treg/Th17 ratio, suggesting that SPP1 may exert regulatory effects on immune cell differentiation in patients with NVAF. A similar Treg/Th17 imbalance has been reported in patients with inflammatory lung injury [21]. However, Chen et al. [22] observed elevated SPP1 levels along with an increased Th17/Treg ratio and, unexpectedly, a concomitant rise in Treg percentage. Given the evidence of reduced Treg cells in AF patients, this discrepancy may be attributed to differences in study populations, or disease stages.
The fibrotic and inflammatory roles of SPP1 have been well-documented in various
cardiovascular contexts. Recent research indicates that SPP1 may facilitate
atrial fibrosis through activation of the Akt/GSK-3
A recent study suggests that SPP1 plays a crucial role in immune regulation
[23]. Elevated SPP1 expression has been shown to influence CD4+ T cell
differentiation, promoting the Th17 phenotype while inhibiting Treg development,
primarily through stabilization of HIF-1
An imbalanced Treg/Th17 ratio has been previously associated with inflammatory cardiovascular diseases such as atherosclerosis and viral myocarditis [26, 27]. Treg cells typically exert protective effects by suppressing immune activation through IL-10 secretion, whereas Th17 cells promote inflammation and tissue injury [28, 29]. Our results are consistent with this paradigm, as both AF patients and AF rats demonstrated reduced anti-inflammatory markers alongside increased pro-inflammatory markers.
However, this study also has several limitations. First, the sample size was limited, and a causal relationship between SPP1 and the Treg/Th17 imbalance has not been established. Second, the specific molecular mechanisms by which SPP1 modulates T cell differentiation in AF remain to be elucidated. Future mechanistic studies are needed to elucidate the role of SPP1 in AF pathogenesis and to evaluate its potential as a therapeutic target.
In conclusion, our study demonstrates that serum SPP1 levels are inversely correlated with the Treg/Th17 ratio in NVAF. Elevated SPP1 levels in AF are associated with both immune imbalance and fibrotic remodeling, suggesting a dual role in disease progression. These findings provide novel insights into the immunoinflammatory mechanisms underlying AF, highlighting SPP1 as a promising biomarker and potential therapeutic target.
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
CJY, YFH, and BF helped in the design of the work and drafting the manuscript. ZXF, YHL, and JYW helped with critical suggestions for the trial design and statistical analyses. All authors have participated sufficiently in the work. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of the Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology (No. 2023-1223), and patients had previously signed a consent form for the use of their medical records for research. All animal experiments received authorization from the institutional ethics committees of China Three Gorges University (2024050L).
Not applicable.
This research was supported by the National Science and Technology Major Project of China (Grant No. 2024ZD0524600), the National Natural Science Foundation of China (No. 82371597), the Natural Science Foundation of Hubei Province (No. 2023AFB609) and Health Commission of Hubei Province Project (No. WJ2023M150).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




