1 Department of Pharmacy, China Aerospace Science & Industry Corporation 731 Hospital, 100074 Beijing, China
2 Evidence-Based Medicine Center, School of Basic Medical Sciences, Lanzhou University, 730000 Lanzhou, Gansu, China
3 Department of Pharmacy, Beijing Hospital, National Center of Gerontology, Institute of Geriatric Medicine, Chinese Academy of Medical Sciences, 100730 Beijing, China
4 Department of Cardiovascular Medicine, Beijing Hospital, National Center of Gerontology, Institute of Geriatric Medicine, Chinese Academy of Medical Sciences, 100730 Beijing, China
Abstract
Hypertension is one of the most prevalent disorders encountered in medical practice, yet effective pharmacotherapy options for resistant hypertension are limited. In this meta-analysis, we aimed to evaluate the efficacy and safety of aprocitentan in treating hypertension.
We searched PubMed, Embase, ClinicalTrials.gov, and the Cochrane Library databases from inception to June 3, 2024, for randomized controlled trials (RCTs) that compared the efficacy and safety between aprocitentan and placebo in treating hypertension. According to the dosage of aprocitentan, the study was divided into a low-dose group (10–12.5 mg), medium-dose group (25 mg), and high-dose group (50 mg).
This meta-analysis included five RCTs, which incorporated 1224 patients, and displayed that aprocitentan can reduce the mean sitting systolic blood pressure (msSBP) [(low dose subgroup: mean difference (MD): –3.85 mmHg; 95% confidence interval (CI): –7.47 to –0.23; p = 0.040; medium dose group: MD: –5.56 mmHg; 95% CI: –10.69 to –0.44; p = 0.030)], mean sitting diastolic blood pressure (msDBP) (low dose subgroup: MD: –3.95 mmHg; 95% CI: –4.06 to –3.85; p < 0.001; medium dose group: MD: –4.75 mmHg; 95% CI: –5.91 to –3.60; p < 0.001), 24-hour ambulatory systolic blood pressure (maSBP) (low dose group: MD: –4.18 mmHg; 95% CI: –4.32 to –4.04; p < 0.001; medium dose group: MD: –5.89 mmHg; 95% CI: –6.03 to –5.75; p < 0.001), and 24-hour ambulatory diastolic blood pressure (maDBP) (low dose group: MD: –4.33 mmHg; 95% CI: –4.42 to –4.24; p < 0.001; medium dose group: MD: –5.82 mmHg; 95% CI: –5.91 to –5.73; p < 0.001). In the high-dose group, there was no difference between the aprocitentan and placebo groups in the msSBP (MD: –4.83 mmHg; 95% CI: –11.44 to 1.79; p = 0.150). Meanwhile, the safety profile of aprocitentan was good, and no significant differences in the frequency of adverse events (AEs) and serious adverse events (SAEs) were observed compared to the placebo.
Aprocitentan significantly reduces blood pressure and has a good safety profile. However, it is worth noting that high doses of aprocitentan (50 mg) did not yield better blood pressure-lowering effects.
Keywords
- hypertension
- dual endothelin receptor antagonist
- aprocitentan
- meta-analysis
Hypertension is a major contributing factor to cardiovascular diseases and
deaths globally and has a prevalence in the total population of 30% to 40%
[1, 2]. The health losses and economic burdens caused by hypertension and its
associated complications may exceed the growth rate of the global population and
economy, making hypertension an ongoing significant global public health issue
[3]. Current guidelines recommend various classes of antihypertensive
medications, including calcium channel blockers (CCBs), angiotensin-converting
enzyme inhibitors (ACEIs), angiotensin II receptor blockers (ARBs), and
beta-blockers. Based on National Health and Nutrition Examination Survey (NHANES) data, “Just under 70% of drug-treated
hypertensive patients achieve blood pressure (BP) control below 140/90 mmHg, meaning over 30%
still have elevated BP, and just under 60% fail to meet the target of
One possible new target is the endothelin (ET) system, which exhibits increased activity under pathological conditions [5]. Endothelin-1 (ET-1) is a small peptide predominantly synthesized by vascular endothelial cells [6]. Additionally, ET-1 is a potent vasoconstrictor, a pathogenic factor in endothelial dysfunction, a growth factor, and a stimulant of aldosterone synthesis and catecholamine release [7]. ET-1 exerts its effects by acting on endothelin A (ETA) receptors in vascular smooth muscle cells and endothelin B (ETB) receptors in endothelial cells [8, 9, 10]. Overall, under physiological conditions, activation of ETA receptors leads to vasoconstriction, while activation of ETB receptors mediates vasodilation via nitric oxide release [10]. Studies have shown efficacy in blocking the ET-1 receptors in many models of hypertension, particularly under conditions of low-renin/salt-sensitive conditions [11, 12].
Aprocitentan is a potent and orally effective dual ET receptor antagonist that blocks the binding of ET-1 to ETA/ETB receptors [12]. Based on studies in rodent animal models, dual blockade of ETA/ETB receptors appears to carry a lower risk of fluid retention and vascular leakage compared to selective ETA blockade by excessive stimulation of ETB receptors, leading to non-selective vasodilation and vasopressin release [13]. Presently, the results of several randomized controlled trials (RCTs) have shown significant antihypertensive effects observed in clinical trials of aprocitentan, especially in patients with resistant hypertension.
This study aimed to conduct a meta-analysis, integrating the results of all published randomized controlled trials, to provide a more accurate assessment of the efficacy and safety of aprocitentan in the treatment of hypertension.
This systematic review and meta-analysis was performed according to the Cochrane Handbook and the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) [14, 15].
PubMed, Embase, the Cochrane Library, and ClinicalTrials.gov databases were searched from their inception to June 3, 2024, for articles using the following Medical Subject Headings or keywords: “aprocitentan”, “ACT–132577”, “Tryvio”, and “hypertension”. The detailed search strategy of all databases is presented in Supplementary Text 1. All published papers related to aprocitentan were searched. After completing the data extraction, we also updated the search to find the most recently published studies.
Trials that met the following criteria were included in this study: (1) the study was a RCT; (2) intervention was performed using aprocitentan; (3) changes in one or more of the following outcomes were measured: the changes were noted in the systolic blood pressure and diastolic blood pressure, while in the sitting position (mean sitting systolic blood pressure (msSBP), mean sitting diastolic blood pressure (msDBP)), changes were observed in the 24-hour ambulatory SBP (maSBP) and 24-hour ambulatory DBP (maDBP); (4) adverse events (AEs) or serious AEs (SAEs) were reported.
We excluded the studies presented as letters, case reports, reviews, conference abstracts, and articles with insufficient data. Additionally, animal experiments and studies that did not include relevant outcome indicators were excluded from the analysis.
All screening studies were performed using the Covidence software (Veritas Health Innovation, Melbourne, VIC, Australia; https://www.covidence.org/). Two authors (ML and XTG) independently screened the literature according to the titles and abstracts and excluded the studies that did not meet the inclusion criteria. The full text of the remaining articles was read to determine whether the study was eligible for inclusion in the analysis. When two authors disagreed, the decision was resolved by mutual consensus or adjudicated by a third author (YW).
The information we collected included the study’s title, design, patient characteristics, interventions, outcome indicators (including msSBP, msDBP, maSBP, maDBP, AEs, and SAEs), and duration of treatment.
Two authors (LZ and XTG) independently extracted data. Disagreements were resolved through discussion or decided by a third author (YTZ). In the case of multiple records pertaining to the same study (e.g., original fulltext publication, abstract, and post-analysis), we collected and analyzed all relevant data as a single study.
All included studies were assessed using the Cochrane Risk of Bias Tool 2 (ROB2) [16] and based on six domains: bias arising from the randomization process, bias due to deviations from the intended intervention, bias due to missing outcome data, bias in measurement of the outcome, bias in selection of the reported result, and other biases. Two authors (LZ and ML) performed the risk of bias; all disagreements were resolved through discussion or adjudicated by a third author (YW).
We performed this meta-analysis using the Review Manager (RevMan) 5.4.1 (Nordic
Cochrane Centre, Copenhagen, Denmark). Dichotomous outcomes were assessed by
risk ratios (RRs), while the mean difference (MD) was employed to express the
continuous outcomes data, with 95% confidence intervals (CIs). The
random-effects model was employed in this meta-analysis due to potential
heterogeneity among the included studies. I2 was used to assess heterogeneity
between included studies. An I2 value of
As none of the outcomes included more than 10 studies and small sample sizes per study, we did not explore sources of heterogeneity using meta-regression analysis. Sensitivity analysis was not planned. However, we used the magnitude of heterogeneity as one of the bases for the certainty of the evidence. We assessed the publication bias using the Egger test and funnel plots to determine if an outcome contained more than 10 studies.
A total of 248 studies were initially screened. Among them, 89 duplicate studies were automatically removed using EndNote software, and 113 were excluded based on their titles and abstracts. After reading the full texts, 41 studies were eliminated, leaving five studies [17, 18, 19, 20, 21] for inclusion in the final meta-analysis. The process of literature selection is shown in Fig. 1.
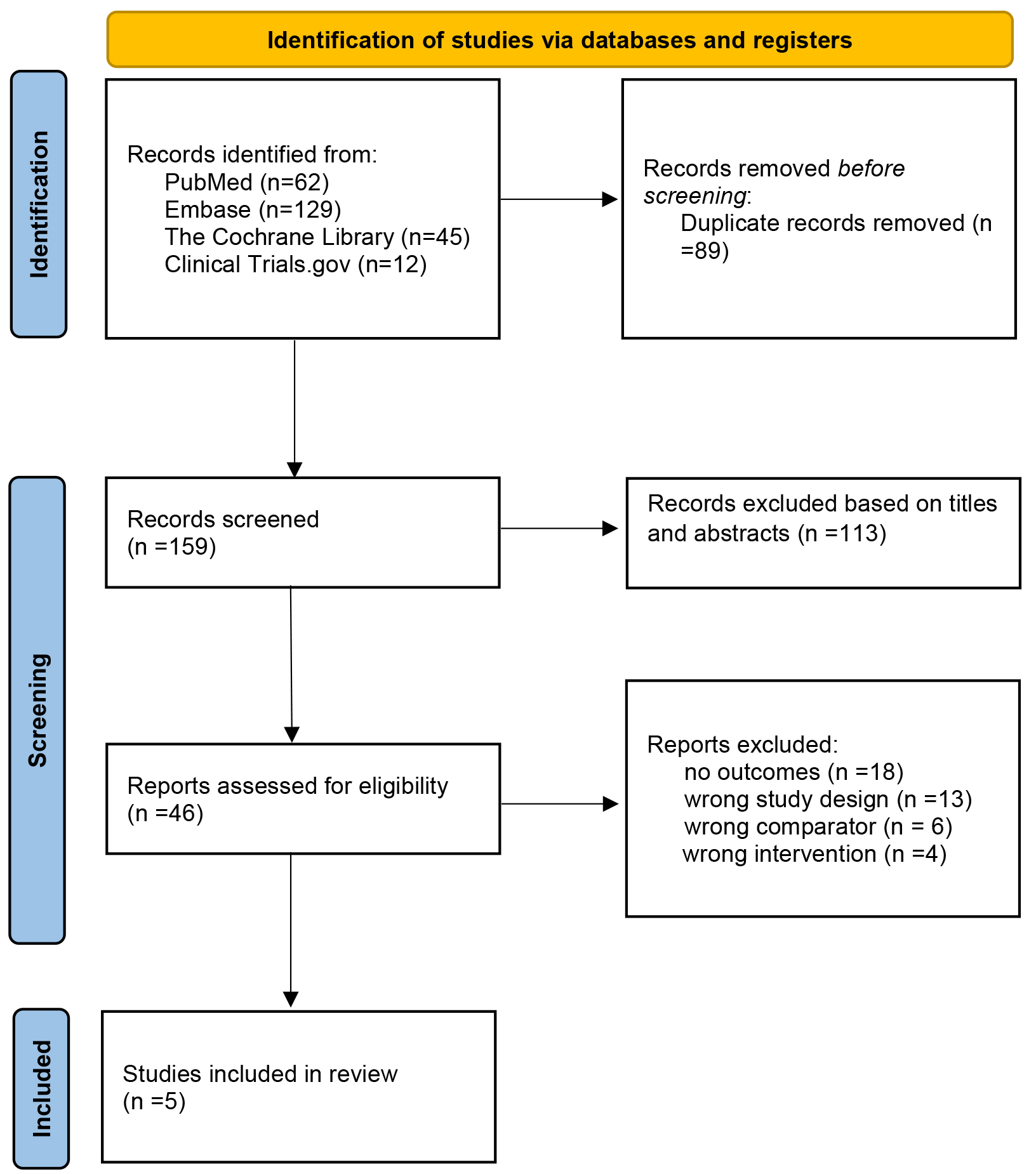 Fig. 1.
Fig. 1.
The Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) flowchart of the study screen.
The five included studies [17, 18, 19, 20, 21] comprised 1224 patients with hypertension. Of these, 865 patients received aprocitentan, and 359 received a placebo. The age range of the included patients was 21–73 years old, and the range dose of treatment was 10 mg–50 mg (divided into low dose group: 10–12.5 mg; medium-dose group: 25 mg; high dose group: 50 mg). The detailed characteristics of the included studies are shown in Table 1 (Ref. [17, 18, 19, 20, 21]).
| Study | Research design | Interventions | No. | Male | Mean age (y) | Duration of treatment |
| Schlaich et al. 2022 [17] | Multi-center, blinded, randomized, parallel-group, phase 3 trial | Aprocitentan 12.5 mg | 243 | 144 | 61.2 |
4 weeks |
| Aprocitentan 25 mg | 243 | 145 | 61.7 | |||
| Placebo | 244 | 145 | 62.2 | |||
| Gueneau de Mussy et al. 2021 [18] | Single-center, double-blind, randomized, placebo-controlled, two-way crossover study | Aprocitentan 10 mg | 8 | 8 | 28.9 | 9 days |
| Placebo | 8 | 8 | ||||
| Aprocitentan 25 mg | 7 | 7 | ||||
| Placebo | 7 | 7 | ||||
| Aprocitentan 50 mg | 8 | 8 | ||||
| Placebo | 8 | 8 | ||||
| Fontes et al. 2021 [19] | Single-center, double-blind, placebo controlled, randomized phase 1 study | Aprocitentan 25 mg | 16 | NR | NR | 10 days |
| Placebo | 4 | |||||
| Verweij et al. 2020 [20] | Randomized, double-blind, multicenter, placebo, and active comparator-controlled trial | Aprocitentan 10 mg | 82 | 51 | 55.3 |
8 weeks |
| Aprocitentan 25 mg | 82 | 45 | 55.1 | |||
| Aprocitentan 50 mg | 81 | 53 | 54.2 | |||
| Placebo | 82 | 55 | 53.5 | |||
| Sidharta et al. 2019 [21] | Double-blind, randomized, placebo-controlled, parallel-group | Aprocitentan 25 mg | 6 | NR | NR | 10 days |
| Placebo | 6 |
Note: NR means not report.
Table 2 (Ref. [17, 18, 19, 20, 21]) displays the bias assessment for the studies included in this meta-analysis. One study [17] was determined to present a low probability of bias. Collectively, the studies included in this meta-analysis exhibited high quality, suggesting a minimal risk of bias.
| Study | Bias arising from the randomization process | Bias due to deviations from the intended intervention | Bias due to missing outcome data | Bias in the outcome measurement | Bias in the selection of the reported results | Other risk of bias | Overall judgement |
| Schlaich et al. 2022 [17] | Low | Probably low | Low | Low | Probably low | Low | Probably low |
| Gueneau de Mussy et al. 2021 [18] | Low | Low | Low | Low | Probably low | Low | Low |
| Fontes et al. 2021 [19] | Low | Low | Low | Low | Probably low | Low | Low |
| Verweij et al. 2020 [20] | Low | Low | Low | Low | Probably low | Low | Low |
| Sidharta et al. 2019 [21] | Low | Low | Low | Low | Probably low | Low | Low |
The subgroup analysis was performed according to the different doses of
aprocitentan. Administering a low dose of aprocitentan treatment significantly
reduced the msSBP (MD: –3.85 mmHg; 95% CI: –7.47 to –0.23; I2 = 46%;
p = 0.040), msDBP (MD: –3.95 mmHg; 95% CI: –4.06 to –3.85; I2 =
0%; p
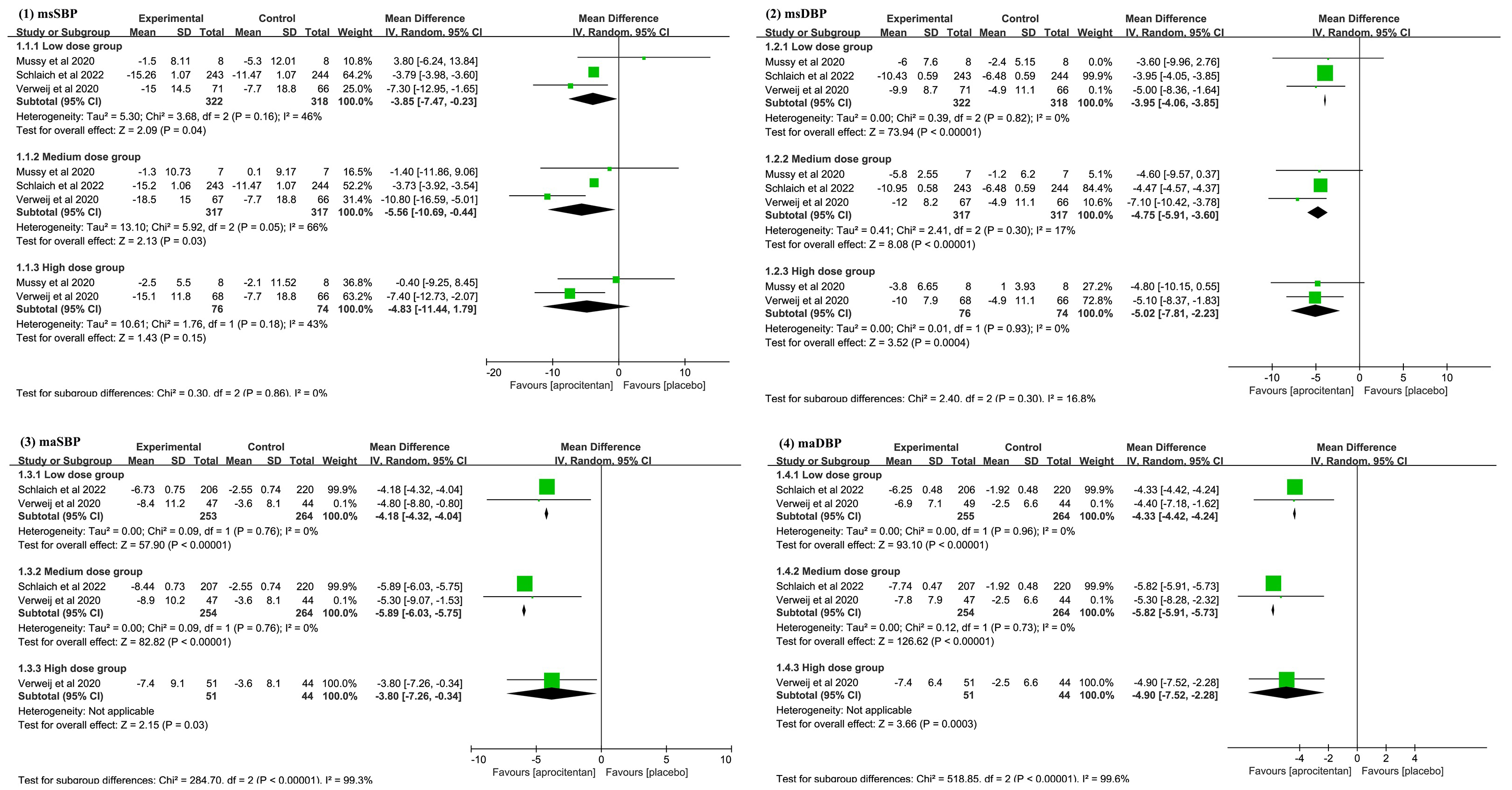 Fig. 2.
Fig. 2.
Meta-analysis results on: (1) mean sitting systolic blood pressure (msSBP); (2) mean sitting diastolic blood pressure (msDBP); (3) mean 24-hour ambulatory systolic blood pressure (maSBP); (4) mean 24-hour ambulatory diastolic blood pressure (maDBP). CI, confidence interval; IV, inverse variance.
All included studies [17, 18, 19, 20, 21] reported data on patients with SAEs, with three studies [18, 19, 21] indicating no occurrence of SAEs. Four studies [17, 19, 20, 21] reported data on patients with AEs. The pooled results showed that there was no statistically significant difference between aprocitentan and placebo groups in the incidence of AEs (RR: 1.45; 95% CI: 0.58 to 3.63; I2 = 80%; p = 0.510, Fig. 3) and SAEs (RR: 2.38; 95% CI: 0.77 to 7.41; I2 = 0%; p = 0.130, Fig. 3).
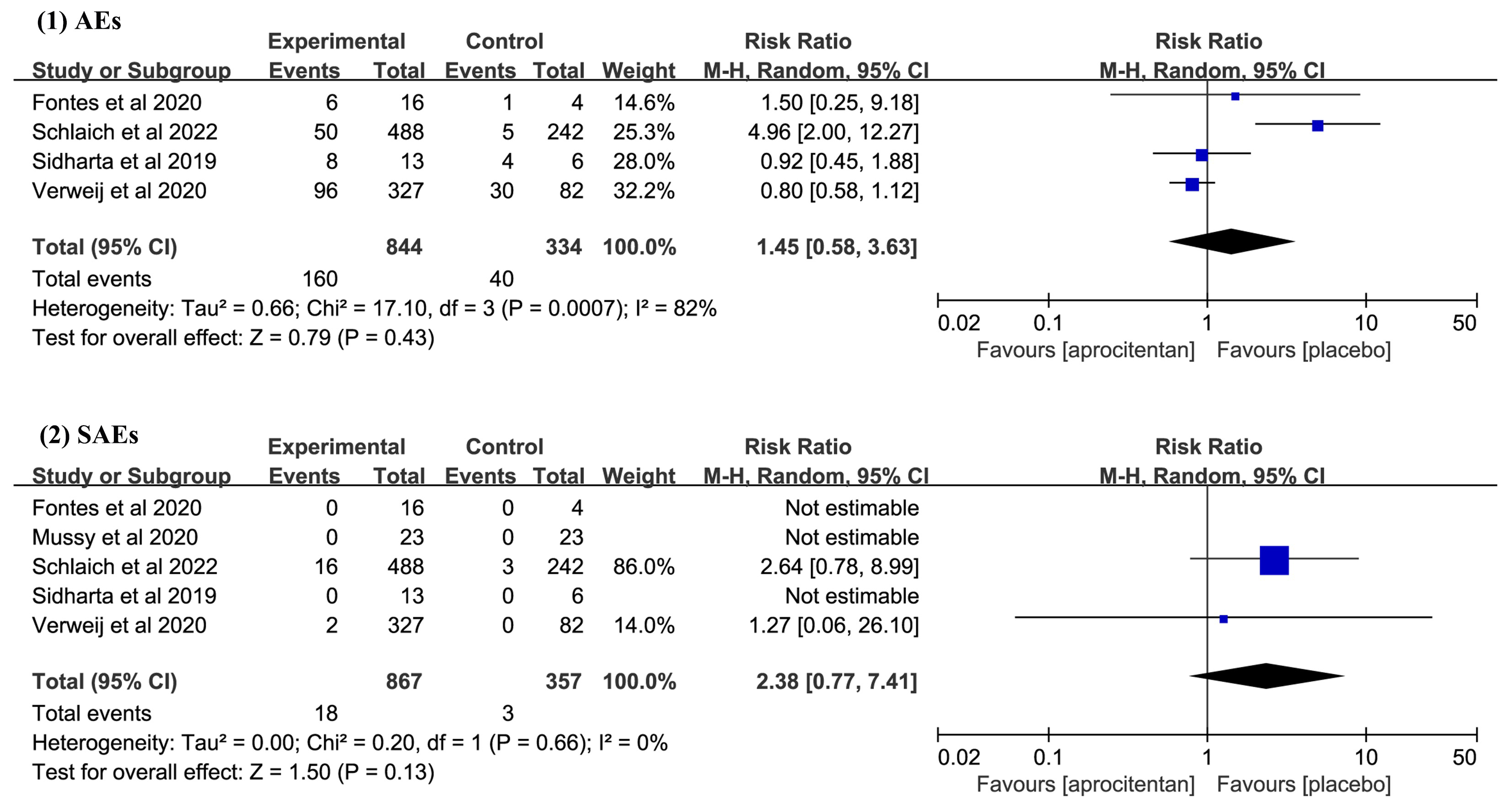 Fig. 3.
Fig. 3.
Meta-analysis results on the incidence of adverse events (AEs) and serious adverse events (SAEs). CI, confidence interval; M-H, Mantel-Haenszel.
This meta-analysis investigated the efficacy and safety of aprocitentan for
treating patients with hypertension. The main findings of this study were as
follows: (1) low (10–12.5 mg) and medium doses (25 mg) of aprocitentan can
significantly reduce msSBP, msDBP, maSBP, and maDBP, but a high aprocitentan
dosage (50 mg) did not significantly reduce msSBP relative to placebo treatment.
Notably, while the highest dose significantly reduced blood pressure
statistically, there was no clear incremental benefit over the medium or low
doses. This observation suggests a flattened dose-response curve, where higher
doses do not proportionally increase efficacy; (2) there were no significant
differences in the incidence of AEs and SAEs between both groups. In addition, it
is worth noting that a high dose of aprocitentan (
Treatment-resistant hypertension (TRH) refers to patients with elevated blood pressure, while 24-hour blood pressure is under treatment with three drugs, one being a diuretic [23], and it is currently estimated to be less than 10% in patients undergoing hypertension treatment [24]. However, the risk of cardiovascular events in patients with TRH is 47% [25]. Currently, the clinical application scope and therapeutic effect of traditional renin–angiotensin–aldosterone system (RAAS) antagonists and renal denervation for patients with TRH remain limited [26, 27]. Aprocitentan can significantly reduce blood pressure and has been shown to have additive effects alongside RAAS blockers [21]. Additionally, the mechanism of action of aprocitentan in blocking ET-1 may help lower blood pressure in patients with resistant hypertension and provide more effective cardiovascular protection [20, 28]. However, these findings have not been fully confirmed and require further investigation. Although clinical trials investigating the effect of aprocitentan on blood pressure are not numerous, they have shown encouraging results in decreasing blood pressure [17, 18, 20]. Especially for TRH, aprocitentan shows significant antihypertensive effects and has good safety in patients with chronic kidney disease and moderate liver dysfunction [21, 29]. Therefore, when hypertension cannot be effectively controlled using current treatment methods, aprocitentan represents a novel, effective, and well-tolerated treatment. An additional benefit of aprocitentan is its ability to reduce albuminuria, which may provide renal protection, especially in patients with hypertension and concomitant kidney disease [30].
Although our meta-analysis results indicate that aprocitentan did not cause any SAEs warranting special attention, notable adverse events such as peripheral edema and anemia were observed [17]. Aprocitentan, as a dual endothelin receptor antagonist, is likely to have drug-related adverse events due to its mechanism of action. Studies have demonstrated that ETA-selective receptor antagonists and dual ETA/ETB receptor antagonists can cause fluid retention, which is regarded to be a characteristic side effect of endothelin receptor antagonists (ERAs) [20, 31, 32, 33]. In addition, a mechanistic study in healthy subjects on a high sodium diet showed that aprocitentan induces a moderate (i.e., less than 1 kg) but statistically significant increase in body weight, which could suggest fluid retention [18]. Additionally, Schlaich et al. [17] found that aprocitentan may cause anemia. Therefore, when using aprocitentan, doctors should pay attention to whether patients develop fluid retention or anemia and intervene promptly to prevent the occurrence of related AEs such as peripheral edema, pulmonary edema, and heart failure [18, 34].
This study has several limitations. Firstly, although we searched a sufficient number of databases, there may still be studies included in geographically based databases that still need to be included. However, the very small number of studies not included will not affect the results of this study. Secondly, the included studies have brief treatment periods, and there is a lack of sufficient safety evaluation data for the long-term use of aprocitentan in treating hypertension. Thirdly, we performed a subgroup analysis, but several outcomes still had a high heterogeneity. This may be due to the differences in aprocitentan dosages, patient numbers, and treatment durations in the included studies. These factors increase the uncertainty of the clinical outcomes, potentially weakening the robustness of the analysis. Finally, this study strictly adhered to the inclusion and exclusion criteria, and ultimately, only five relatively small studies were included. Nevertheless, there were differences in the baseline levels among the included studies. One study [17] included patients who had received standardized treatment for hypertension, while others [18, 19, 20, 21] included patients who had not received other medication treatments. Further research and larger sample sizes are still needed to confirm the efficacy and safety of aprocitentan, such as longer follow-up periods, larger sample sizes, and multicenter studies.
The results of this study confirm that aprocitentan can significantly reduce blood pressure, as evidenced by significant effects on msSBP, msDBP, maSBP, and maDBP. Additionally, we did not observe any risks of AEs and SAEs following aprocitentan treatment. However, it is worth noting that these results lack support from large-scale studies, and it is necessary to conduct large RCTs in the future to confirm the efficacy and safety of aprocitentan.
The original data presented in the study are included in the article/supplementary material, further inquiries can be available from the corresponding author upon reasonable request.
YW and YTZ designed the research study; LZ and YW performed the research. XTG and ML screened studies. LZ and XTG extracted and analyzed the data. LZ and ML performed the quality assessment of the included studies. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This study was supported by the Fundamental Research Funds for the Central-level Scientific Research Institute (No. 2019XK320078, BJ-2019-092), National High Level Hospital Clinical Research Funding (BJ-2022–155), the National Key Research and Development Program of China (2020YFC2008100/2020YFC2008106).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM25909.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



