1 Division of Cardiology, University of Parma, Parma University Hospital, 14 - 43126 Parma, Italy
2 Division of Cardiology, Parma University Hospital, 14 - 43126 Parma, Italy
†These authors contributed equally.
Abstract
Coronary microvascular dysfunction (CMD) comprises a wide spectrum of structural and/or functional abnormalities of coronary microcirculation that can lead to myocardial ischemia. Emerging evidence has indicated that CMD is a relevant cause of morbidity and mortality and is associated with a high risk of major adverse cardiovascular events (MACEs) and heart failure with preserved ejection fraction as well as poor quality of life. This review aims to elucidate briefly the pathogenesis and diagnostic modalities of CMD and to shed light on contemporary evidence on the prognostic impact of CMD. Finally, we will provide an overview of novel emerging therapeutic strategies for CMD.
Keywords
- coronary microvascular dysfunction
- major adverse cardiovascular events
- heart failure with preserved ejection fraction
- quality of life
Coronary microvascular dysfunction (CMD) refers to the inability of coronary microcirculation to dilate in response to increased myocardial oxygen demand due to a combination of structural and functional alterations [1]. CMD is an increasingly recognized mechanism of myocardial ischemia and a major determinant of ischemia with non-obstructive coronary artery (INOCA) disease [2, 3, 4] and chronic coronary syndrome (CCS) [5].
CCS encompasses a wide spectrum of clinical presentations characterized by an imbalance between myocardial oxygen supply and demand, ultimately resulting in myocardial ischemia. The underlying pathophysiology is complex, involving both structural and functional alterations in the coronary arteries and/or microcirculation [5].
Obstructive coronary artery disease (CAD) is the most common and well-known cause of myocardial ischemia. Moreover, CAD is characterized by the progressive accumulation of lipids and inflammatory cells within the coronary arteries, forming atherosclerotic lesions covered by a fibrous cap. Further, this gradual narrowing of the coronary arterial lumen can lead to myocardial hypoperfusion, particularly during physical exertion and emotional or other stresses [5].
Coronary vasomotor disorders represent the primary functional cause of CCS and
are characterized by transient hypercontraction of coronary vascular smooth
muscle cells. Endothelial dysfunction and increased vascular smooth muscle cell
reactivity underlie these disorders [2, 3]. Coronary artery spasms may occur at
the epicardial level, diagnosed when a
Interestingly, a recent meta-analysis involving 14,427 patients demonstrated that 23% of individuals with INOCA presented with both CMD and vasomotor disorders [8]. Notably, these functional alterations can also coexist with obstructive CAD. These findings highlight the importance of a comprehensive functional assessment of the coronary vasculature to identify the precise mechanisms underlying CCS and to facilitate the development of tailored medical therapies [9].
The condition involving chest pain in the absence of obstructive CAD has been recognized since the 1970s and was initially termed “cardiac syndrome X” [10]; however, the term CMD has recently been introduced. This condition is primarily associated with microvascular dysfunction, endothelial impairment, and heightened pain sensitivity. Notably, it is more prevalent in women than men [11].
While initially considered a neglected condition, the advent of non-invasive and invasive functional tests has exponentially broadened the ability to recognize CMD and delineate related clinical and biochemical features [3, 8].
Convincing evidence has suggested that CMD is not as benign as previously thought. Indeed, CMD confers a high risk of major adverse cardiovascular events (MACEs) [12] and heart failure with preserved ejection fraction (HFpEF) [13], while patients with CMD also often experience a poor quality of life (QOL) [14].
This review aims to provide a brief overview of the pathogenesis and invasive diagnosis of CMD and to elucidate contemporary evidence on the prognostic impact of CMD. Finally, we will discuss novel potential therapeutic strategies for personalized CMD treatments.
Although epicardial CAD is mainly responsible for myocardial ischemia, up to half of the subjects undergoing invasive coronary angiography for symptoms or non-invasive evidence of myocardial ischemia have non-obstructive CAD [5]. The spectrum of conditions underpinning myocardial ischemia and its symptomatic manifestation in the absence of obstructive CAD is generally referred to by the acronym INOCA [3]; meanwhile, a key mechanism underlying INOCA is CMD, characterized by a reduced coronary flow reserve (CFR) [3].
CMD can be classified into four categories based on clinical presentation: (1) CMD in the absence of myocardial disease and obstructive CAD; (2) CMD in myocardial disease; (3) CMD in obstructive CAD; (4) iatrogenic CMD [15].
Studies with adenosine and acetylcholine as vasodilator agents suggested that CMD was present in 60% of patients with non-obstructive CAD [16, 17, 18]. A further study found CMD in up to 40% of cases, although a transthoracic Doppler echocardiography seems to be characterized by low accuracy [19]. Conversely, positron emission tomography (PET) has shown the presence of CMD in 60% of cases, with no significant gender differences [20].
CMD has been shown to primarily arise from abnormalities in the coronary
microvascular circulation [2, 3]. The coronary microcirculation comprises a
network of vessels with a diameter of
From a pathophysiological standpoint, CMD results from a variable combination of microcirculatory functional and structural abnormalities [15] that can altogether lead to reduced CFR, augmented microvascular resistance, and paradoxical arteriolar vasoconstriction, which can be provoked during invasive provocative tests, such as acetylcholine infusion.
Several hypotheses have been proposed to explain the multifaceted mechanisms underlying CMD. Endothelial dysfunction is considered a key mediator of functional dysregulation affecting the microvasculature. Further, endothelial dysfunction involves a maladaptive shift towards a net vasoconstrictive state, promoted by a reduction in the bioavailability of vasodilatory agents such as prostaglandins, nitric oxide (NO), and endothelium-derived hyperpolarizing factors and through an increased release of constrictive agents such as endothelin-1 (ET-1), thromboxane A2, prostaglandin H2, and reactive oxygen species (ROS) [22, 23].
In addition, endothelium-independent mechanisms, including impaired relaxation of vascular smooth muscle cells jointly with enhanced RhoA/Rho kinase activity and increased vasoconstrictive mediators, contribute to the pathogenesis of CMD [24] and microvascular spasms [25].
Inflammation and neurohormonal disarrangements also play a pivotal role. A
pro-inflammatory state promotes oxidative stress and endothelial dysfunction
through a complex interplay involving interleukin-6, tissue necrosis factor
Lastly, structural changes also contribute to the pathogenesis of CMD. These include luminal narrowing of small resistance arterioles with hypertrophic inward remodeling, increased stiffness with perivascular fibrosis, and capillary rarefaction [28].
The limited resolution of coronary angiography has spurred the implementation of
invasive techniques that can test coronary microvascular status selectively. A
reduced CFR, the hallmark of CMD [3, 4], provides a unique opportunity to
evaluate both epicardial and microvascular compartments [12]. Two strategies are
currently available to measure CFR. The Doppler flow velocity method utilizes a
Doppler wire (ComboWire XT or FloWire, Philips Volcano Corporation, SAN DIEGO, CA, USA) to measure
coronary flow velocity (CFV) at rest and following a hyperemic stimulus.
Adenosine is the most used vasodilator agent in this context, even though
intracoronary papaverine is gaining popularity. The hyperemic to the resting CFV
ratio offers a reliable quantification of CFR with pathological values
Invasive function testing features the unique potential to categorize CMD into
structural and functional through the assessment of microvascular resistance
[32]. Two indices are available in clinical practice: The index of
microcirculatory resistance (IMR), which is a thermodilution-based index, defined
as the product of distal coronary pressure and Tmn during maximal hyperemia, with
normal values
A CMD diagnosis is associated with a wide spectrum of complications, including a high risk of MACEs and HFpEF and poor QOL, compared to healthy individuals, and that includes approximations of the complications associated with patients with obstructive CAD (Fig. 1).
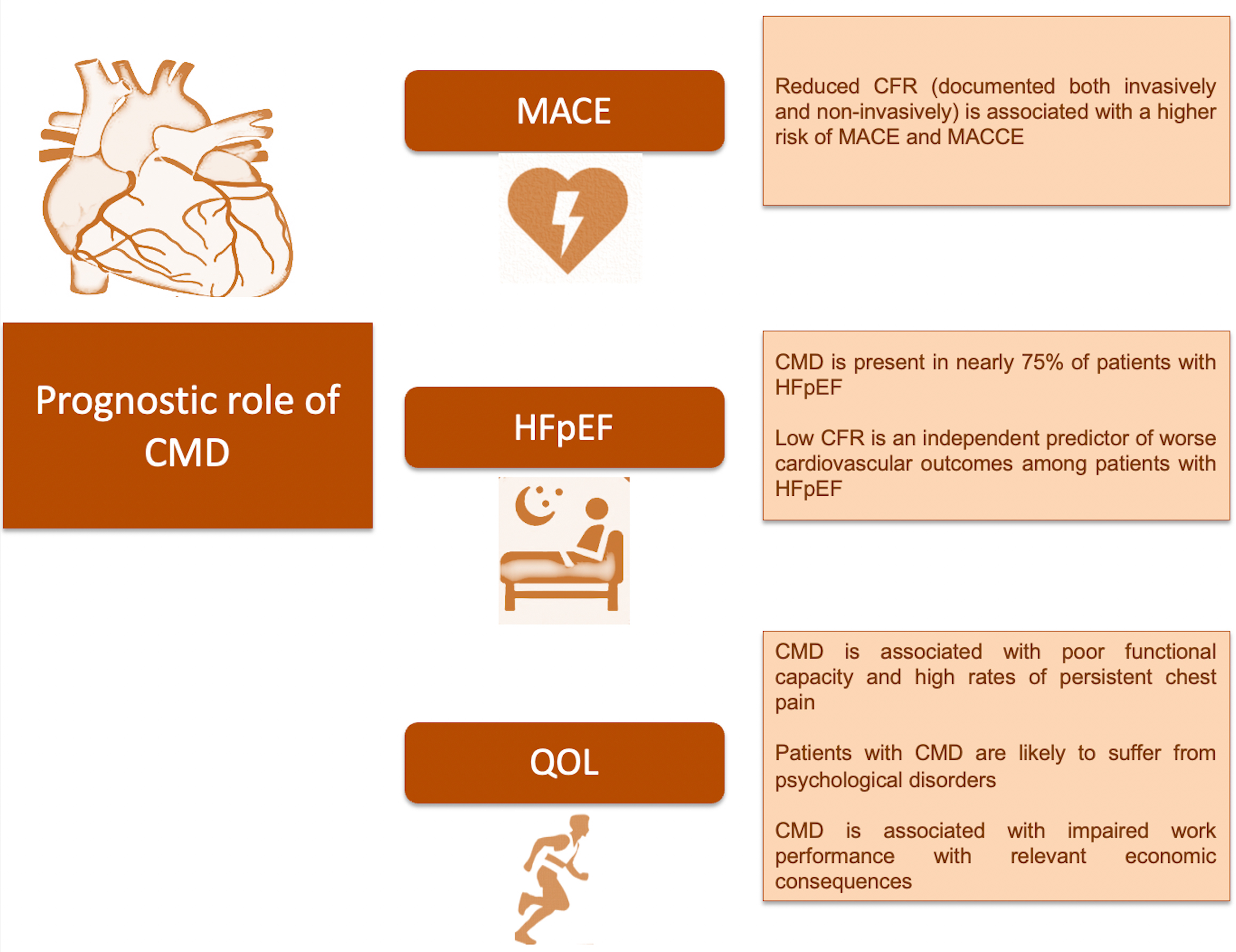 Fig. 1.
Fig. 1.
Overview of possible complications associated with CMD. Abbreviations: CFR, coronary flow reserve; CMD, coronary microvascular dysfunction; HFpEF, heart failure with preserved ejection fraction; MACE, major adverse cardiovascular event; MACCE, major adverse cardiovascular and cerebrovascular event; QOL, quality of life.
Patients presenting with angina and not obstructive CAD are generally reassured that their symptoms are noncardiac. However, in the presence of CMD, several studies have demonstrated that these patients are at increased risk of MACEs [12, 35]. Suwaidi et al. [36] showed that severe endothelial dysfunction, assessed through an impaired response to acetylcholine stimulus, is associated with increased cardiac events at a median follow-up of 28 months, confirmed in subsequent studies with longer follow-ups [37, 38].
The Women’s Ischemia Syndrome Evaluation (WISE) study demonstrated that female
subjects with symptoms and signs suggestive of ischemia but without obstructive
CAD present a 5-fold increased risk of MACEs compared with asymptomatic women
with no history of heart disease [39]; however, recent evidence has refuted these
findings, albeit an increased risk of MACEs (8.6% vs. 3.5%, p
Asymptomatic diabetic patients (n = 101) with impaired CFR in an adenosine
stress echocardiography presented an increased risk of MACEs (adj. HR 12.9, 95%
CI 3.9–43.2, p
In patients with suspected myocardial ischemia (n = 229), PET-derived abnormal
CFR was associated with increased risk of MACEs (adj. HR 1.60, 95% CI
1.00–2.57, p
Decreased CFR in a stress echocardiogram has been associated with markedly
increased risk in women (adj. HR 16.5, 95% CI 7.2–37.9, p
In the absence of obstructive coronary stenoses, abnormal non-invasive stress
tests in patients with stable CAD may indicate INOCA; however, this absence does
not identify patients with a higher risk of long-term cardiovascular events. A
recent analysis was conducted on 297 patients with a positive non-invasive test
and nonobstructive coronary stenoses (fractional flow reserve
The prognostic value of IMR was tested in this context. Liu et al. [50]
conducted a study including 151 consecutive patients with chest pain and
non-obstructive CAD who largely presented CMD (61.6%). Patients with CMD,
assessed through an impaired CFR (
The coronary microcirculatory function has recently been investigated with
microvascular resistance reserve (MMR) in 547 consecutive patients undergoing
systematic echocardiography and invasive physiological assessment for suspected
CAD. A study demonstrated that an impaired MMR
Finally, patients with CMD experienced a nearly 4-fold higher risk of overall mortality compared to healthy controls [52]: this finding suggests that reduced CFR might be a marker of systemic endothelial dysfunction [53] (Table 1, Ref. [20, 35, 36, 37, 38, 40, 41, 42, 44, 45, 46, 47, 48, 49, 50]).
| First author, year [Ref] | Sample size | Measure modality | Type of outcome | Rate of outcome |
| Non-invasive tests | ||||
| Herzog, 2009 [44] | 229 | Adenosine 13-N ammonia-PET | MACEs | 45.1% vs. 23.6% |
| Cortigiani, 2010 [46] | 1660 | Stress echocardiography | MACEs | 27.0% vs. 2.0%* |
| LAD CFR | 42.0% vs. 8.0%** | |||
| Ziadi 2011 [45] | 414 | Dypiridamole rubidium82-PET | MACEs | 24.0% vs. 9.0%# |
| Cortigiani, 2012 [47] | 4313 | CFR on LAD | Death | 39.0% vs. 7.0% |
| Murthy, 2014 [20] | 1218 | Vasodilator rubidium82-PET | MACEs | 8.6% vs. 3.5% |
| Dikic, 2015 [41] | 200 | Adenosine stress echocardiography | MACEs | 18.8% vs. 5.1% |
| Gan, 2017 [42] | 371 | Adenosine stress echocardiography | MACEs | 36.8% vs. 10.8% |
| Cortigiani, 2018 [48] | 375 | CFVR and LVCR | MACEs | 63.0% vs. 10%+ |
| Invasive tests | ||||
| Suwaidi, 2000 [36] | 157 | CBF | MACEs | 14.0% vs. 0.0% |
| Schächinger, 2000 [38] | 147 | CBF | MACEs | 11.0%*** |
| Britten, 2004 [37] | 120 | CFR | MACEs | 18.0% vs. 5.0% |
| Marks, 2004 [35] | 168 | CFR | Death | 20.0% vs. 7.0% |
| Lee, 2018 [40] | 631 | CFR | VOCO | 11.2% vs. 3.7% |
| Boerhout, 2022 [49] | 1102 | CFR + MR | MACEs | 11.7% vs. 5.5% |
| Liu, 2023 [50] | 151 | calMR | MACCEs | 40.9% vs. 13.8% |
Abbreviations: CMD, coronary microvascular dysfunction; VOCO, vessel-oriented clinical outcomes; calMR, coronary angiography-derived index of microvascular resistance; CBF, coronary blood flow; CFR, coronary flow reserve; CFVR, coronary flow velocity reserve; LAD, left anterior descending artery; LVCR, left ventricular contractile reserve; MACEs, major adverse cardiovascular events; MACCEs, major adverse cardiovascular and cerebrovascular events; MR, microvascular resistance; PET, positron emission tomography; *women; **men; ***overall population; #,+intergroups.
HFpEF is a clinical syndrome consisting of typical symptoms and signs of HF in
the presence of cardiac structural and/or functional abnormalities, which
generally lead to raised left ventricular filling pressures with preserved
systolic function [54]. In recent years, alongside a growing understanding of
this condition, a new paradigm has emerged that recognizes an important link with
microcirculatory dysfunction. Several studies have demonstrated a consistently
high prevalence of CMD among patients with HFpEF [13, 55, 56, 57], although the mutual
causative interplay of these two entities is still a matter of uncertainty.
Interestingly, HFpEF and CMD share similar risk factors (e.g., hypertension,
advanced age, female sex, chronic kidney disease, diabetes mellitus, obesity)
[58, 59], and the common denominator between these two conditions is believed to
be represented by endothelial dysfunction promoted by a comorbidity-driven
systemic pro-inflammatory and pro-oxidative state [60]. While endothelial
dysfunction may exist in both heart failure with reduced and preserved ejection
fractions, mounting evidence points toward its primary role in the pathogenesis
of HFpEF. Indeed, cardiac endothelium directly affects the contractility and
relaxation of underlying myocardial cells [61]. Consequently, inflamed
microvascular endothelium can contribute to diastolic dysfunction, the hallmark
of HFpEF, through reduced NO-bioavailability and the release of transforming
growth factor-
The presence of CMD is also an important prognostic factor in patients affected by HFpEF [65, 66, 67, 68].
The PROMIS-HFpEF (PRevalence Of MIcrovascular dySfunction in Heart Failure with
Preserved Ejection Fraction), the largest prospective study to date, showed a
high prevalence (75%) of CMD (defined as CFR
The majority of data regarding the prognostic role of CMD in patients with HFpEF
comes from cardiac magnetic resonance (CMR) imaging studies. Kato et al.
[71], in a retrospective study including 163 patients with HFpEF undergoing
vasodilator stress CMR, demonstrated that CFR was significantly lower in HFpEF
patients with adverse events compared with those without. Furthermore,
Kaplan–Meier analysis showed that at a median follow-up of 4.1 years, rates of
all-cause death and heart failure hospitalizations were significantly higher in
HFpEF patients with CFR
In conclusion, growing evidence strengthens the implementation of CMD as a useful prognostic marker for HFpEF. Moreover, given the frequent association between CMD and HFpEF, targeting endothelial dysfunction appears to be a promising therapeutic strategy for novel interventions. Nonetheless, it must be emphasized that despite the fascinating underlying pathophysiological hypothesis, more research is needed to clarify a cause-and-effect relationship between these two entities (Table 2, Ref. [13, 55, 56, 57, 65, 66, 68, 69, 70, 71, 72]).
| First author, year [Ref] | Population, comparison | CMD assessment method, CMD definition | Timeframe | Key findings |
| Sucato, 2015 [55] | n = 155 HFpEF, n = 131 non-HFpEF | Invasive coronary angiography, NA | NA | Significantly lower TFC and MBG of the three coronary arteries in HFpEF vs. non-HFpEF patients |
| Taqueti, 2018 [13] | n = 201 non-HFpEF, NA | Rb-82 PET-derived CFR, CFR |
Median follow-up of 4.1 years | Using a multivariate analysis, CMD was independently predictive of CV death, MI, and/or HFpEF hospitalization |
| Shah, 2018 [69] | n = 202 HFpEF, NA | TTE-derived CFR, CFR |
NA | Prevalence of CMD of 75% (95% CI: 69–81%) Poorer CFR was independently associated with markers of endothelial dysfunction and HF severity |
| Dryer, 2018 [56] | n = 30 HFpEF, n = 14 non-HFpEF | CFR and IMR through coronary pressure wire, CFR |
NA | Significantly lower mean CFR (2.55 IMR (26.7 |
| Yang, 2020 [65] | n = 162 HFpEF, NA | Endothelium-dependent: increase in CBF after acetylcholine administration Endothelium-independent: CFR through coronary Doppler wire, CFR |
Median follow-up of 12.5 years | Prevalence of CMD of 72% A trend toward worse mortality in endothelium-dependent CMD vs. preserved endothelial function Significantly worse mortality (adjusted HR 3.56, 95% CI 1.14–11.12, p = 0.03) in endothelium-independent CMD vs. CFR |
| Hage, 2020 [70] | n = 201 HFpEF, NA | TTE-derived CFR, CFR |
Median follow-up of 388 days | Significantly higher rate of CV death or HF hospitalization in HFpEF patients with CMD vs. non-CMD |
| Kato, 2021 [71] | n = 163 HFpEF, NA | CMR-derived CFR, CFR |
Median follow-up of 4.1 years | Significantly higher rate of all-cause death or HF hospitalization in HFpEF patients with CFR |
| Rush, 2021 [57] | n = 106 HFpEF, NA | Endothelium-independent: CFR and IMR through coronary pressure wire, CFR Endothelium-dependent: abnormal coronary vasoreactivity after acetylcholine CMR-derived MPRI, MPRI |
Median follow-up of 18 months | Prevalence of endothelium-independent CMD of 66%, endothelium-dependent CMD of 24%, and any CMD of 85% Prevalence of MPRI No significant difference in clinical outcomes between patients with and without CMD Significantly higher rate of death or hospitalization (p = 0.011, log-rank) in patients with MPRI |
| Ozcan, 2021 [66] | n = 40 HFpEF, n = 40 non-HFpEF | CFR and IMR through coronary pressure wire, CFR |
1 year | Trend toward higher prevalence of CMD in HFpEF vs. non-HFpEF patients (53% vs. 33%, p = 0.07) Significantly lower survival free of HF hospitalization in patients with abnormal CFR vs. those without |
| Mohammed, 2018 [69] | n = 22 HFpEF, n = 29 non-HFpEF | Endothelium-independent: CFR through coronary Doppler wire, CFR Endothelium-dependent: increase in CBF after acetylcholine |
NA | Significantly lower CFR (2.5 Significant inverse correlations between CFR and PAWP at rest (r = −0.31; p = 0.03) and peak exercise (r = −0.47, p = 0.001) |
| Arnold, 2022 [72] | n = 101 HFpEF, n = 43 non-HFpEF | CMR-derived MPR, MPR |
Median follow-up of 3.1 years | Significantly higher prevalence of CMD (70% vs. 48%, p = 0.014) in HFpEF vs. non-HFpEF patients In multivariable models, MPR was independently predictive of the composite of death or HF hospitalization in HFpEF patients |
| Mohammed, 2023 [68] | n = 137 HFpEF, NA | Pressure wire-free coronary angiography-derived IMR (caIMR), caIMR |
Median follow-up of 15 months | Prevalence of CMD of 64.2% Using a multivariate analysis, CMD was independently predictive of all-cause death or HF hospitalization |
Abbreviations: CFR, coronary flow reserve; CI, confidence interval; CMD, coronary microvascular dysfunction; CMR, cardiac magnetic resonance; CV, cardiovascular; HF, heart failure; HFpEF, heart failure with preserved ejection fraction; IMR, index of microcirculatory resistance; MBG, myocardial blush grade; MI, myocardial infarction; MPR, myocardial perfusion reserve; NA, not applicable; PET, positron emission tomography; Rb-82, rubidium 82; TFC, TIMI frame count; TTE, transthoracic Doppler echocardiography; TIMI, thrombolysis in myocardial infarction; CBF, coronary blood flow; MPRI, myocardial perfusione reserve index; PAWP, pulmonary arterial wedge pressure.
QOL refers to physical, psychological, emotional, and social features that are physiologically and socially constructed [73]. Assessing the QOL of an individual requires an extensive investigation: several functional tests and questionnaires have been implemented to explore all the QOL domains, including physical, mental, and social health.
Patients with CMD experience a worse QOL when compared to the general population, whereby all the QOL-related components are negatively affected.
The Duke Activity Status Index (DASI) questionnaire is the most adopted tool to
assess the functional capacity of an individual and is a reliable surrogate of
metabolic equivalents (METs) [74]. Several studies have documented poor physical
status among patients with CMD. The landmark WISE registry [75] estimated a poor
functional capacity (5.7
Comparisons between patients with CMD and obstructive CAD in terms of physical performance are controversial. On the one hand, the WISE registry [76] and the UK-based INOCA International survey [14] reported a slightly greater functional capacity in the group with INOCA and CMD. Conversely, Schumann et al. [77] noted a close link between INOCA and worse physical performance, especially in the group with CMD. Accordingly, in the study by Jespersen et al. [78], the risk of long-term angina was higher in the groups with non-obstructive CAD (64%) and normal coronary arteries (49%) than in patients with obstructive CAD (41%).
The burden of persistent chest pain in the context of CMD is relevant and poses
clinical and therapeutic concerns. Lamendola et al. [79], including 155
patients with cardiac syndrome X, reported that chest pain had remained unchanged
in 33% and had worsened in 14% of patients at a mean follow-up of 137
The assessment of functional capacity is of prognostic relevance. A functional
capacity
Patients with CMD are likely to suffer from psychological disorders. Several psychometric questionnaires have been implemented to deepen the relationship between CMD and psychiatric comorbidities.
A landmark study by Potts et al. [80] documented that nearly two-thirds of patients with non-obstructive CAD experience psychological illnesses, including anxiety, panic disorder, and major depression. In the WISE registry, almost 45% of women suffered from depression [75], while Gulati et al. [14] reported impaired mental health and outlook on life in nearly 70% of patients, and up to 30% of subjects in small studies experienced panic disorder [81, 82]. An elegant investigation by Altintas et al. [83] established that anxiety disorder, depression, and somatoform diseases are the most prevalent psychological comorbidities in the context of CMD.
Pathogenesis of psychological disorders in CMD is multifaceted. First, the lack of awareness about CMD and the relatively low penetration of invasive functional tests in clinical practice often results in uncertainty concerning the source of chest pain [84] and delayed diagnosis. According to the UK-based INOCA International survey, half of patients receive a proper diagnosis of CMD more than one year after the onset of chest pain [14]. Second, patients with CMD are often burdened by nociceptive abnormalities and increased pain sensitivity that might fuel anginal complaints. Autonomic cardiac control imbalance might subtend interindividual differences in ischemic and pain thresholds [85]. Third, patients with CMD are frequently undertreated due to the false perception that CMD is a benign condition, potentially aggravating microvascular function and myocardial ischemia. Fourth, the high burden of anginal attacks with an unpredictable onset breeds a continuous state of apprehension that discourages patients from participating in social activities [84]. Fifth, the decline in physical status significantly worsens work performance with a detrimental economic impact, which further increases the risk of psychological disorders [86].
Convincing evidence showed that the severity and duration of chest pain correlate with the risk of psychological disorders. Pioneeristic studies documented an association between cardiac syndrome X and the risk of prolonged chest pain, panic disorder, and anxiety [75, 81]. Accordingly, a close link between the persistent chest and the burden of anxiety, depression, and psychotropic medication use was observed in the WISE registry [75].
Conversely, the burden of psychological abnormalities is not influenced by the extent of CAD and cardiac injury. Valkamo et al. [86] and van Schalkwijk et al. [87] found a similar rate of chest pain and mental disorders between patients with and without CAD. In addition, patients with CMD experienced significantly greater levels of anxiety compared to those with sudden cardiac death, suggesting that the rate of psychological illnesses is not linked to the nature of cardiac disease [77].
Importantly, psychological disorders can play a potential pathogenic role in CMD via enhanced sympathetic activity, low-grade inflammation, and endothelial dysfunction [85]. Consistently, an elegant study by Vermeltfoort et al. [88] demonstrated a positive association between trait anxiety and the extent of myocardial ischemia among 20 patients with CMD assessed using myocardial perfusion scintigraphy. In addition, psychological comorbidities facilitate the onset of unhealthy behaviors and classical cardiovascular risk factors, as well as enhanced platelet reactivity and procoagulant state, promoting the development of accelerated CAD [85]. Accordingly, Rutledge et al. [89] showed that depression and anxiety predicted the occurrence of MACEs among a cohort of 489 women with non-obstructive CAD at a median follow-up of 5.9 years.
Up to 80% of patients with CMD state significant impairment in social activities, especially work performance [14]. After the onset of symptoms, nearly 75% of patients report missed days from work and fewer working hours per day. As outlined by the UK-based INOCA International survey [14], 47.5% retire earlier than expected from work, and nearly a third of patients are forced to change work due to significant physical impairment. Furthermore, physical, mental, and output limitations reduce work productivity [84]. These features culminate in significant economic consequences. Schumann et al. [77] estimated an annual economic impact of USD 21 billion due to INOCA-related premature exit from work, consistent with those of patients with obstructive CAD.
Finally, every 1 MET reduction in functional capacity led to 2.9
| First author, year [Ref] | Study population | Methodology | Baseline features | Main results |
| Potts, 1995 [80] | 46 patients with NOCAD and 53 healthy controls | Standardized interviews and rating scales at the time of angiography, after 1 year and 11.4 years later | 61% with psychiatric cases at angiography and 49% at 11.4 years | Levels of morbidity were significantly greater in patients with NOCAD Anxiety disorders were common, with panic disorder (15% of patients) the most common current diagnosis at final follow-up |
| Asbury, 2004 [82] | 100 females with CSX, 100 females with CHD, 100 healthy female volunteers | HAQ questionnaire HADS scale |
CSX patients were younger at the onset of symptoms (54 |
Syndrome X patients had higher levels of life interference (p |
| Handberg, 2013 [73] | Sub-study of the WISE registry (936 patients with NOCAD) | BDI, STAI, DASI questionnaires Diamond chest pain questionnaire |
Mean age 58 |
Persistent chest pain was associated with an increased rate of adverse events and relatively high rates of depression and anxiety with reduced functional capacity and impaired QOL over a median follow-up of 6 years |
| Altintas, 2014 [83] | 56 CSX patients and 53 CHD patients | Structured clinical interview for DSM-IV axis I disorders BDI, BAI questionnaires Short form 36 scale |
73.2% were women, 78.6% had a physical disease, 69.6% had experienced stressful life events | Depression was detected in 41% of the CSX group and 64% of the control group. Anxiety disorder was present in 64% of the CSX group. The somatoform disorder was determined in 24% of the CSX group |
| Schumann, 2021 [77] | 66 patients with INOCA underwent stress cardiac magnetic resonance | QOL questionnaires (SAQ, CAQ, WLQ) | 59% with definite or borderline CMD, 56.1% were women | INOCA patients reported: Lower SAQ scores, suggestive of worse symptoms than MI and stable CAD patients; higher CAQ scores, suggestive of greater cardiac anxiety than patients with sudden cardiac death; high WLQ scores, suggestive of significant work limitations |
| Gulati, 2023 [14] | 297 patients from the United Kingdom-based INOCA International | Self-reported physical, social, and mental health. DASI score |
60% with CMD, 91.2% were women | Functional capacity was poor after onset of symptoms (5.6 Adverse impact of symptoms on home life (80.5%), social life (80.1%), mental health (70.4%) In total, 75% of patients had reduced working activities, 47.5% retired early, and 38.4% applied for disability |
| van Schalkwijk, 2023 [87] | 373 patients enrolled in the IMR and THIO studies | Modified SAQ Mental Health Continuum-Short Form Fatigue Assessment scale PHQ-9 General anxiety disorder questionnaire PSS |
For INOCA: mean age of 62.74 years, 51% undertaking any alcohol use, and 69% being physically active | Compared to obstructive CAD patients, INOCA patients reported a better physical status lower smoking habits, obesity, and dyslipidemia, and similar rates of psychological distress and well-being |
| Humphreys, 2024 [84] | 17 patients with INOCA | Qualitative investigation on lived experiences with INOCA | 53% with CMD, 88.2% were women | Significant psychological impact on people living with INOCA Disappointment and lack of clarity over treatment and management pathways |
Abbreviations: BDI, Beck depression inventory; CAD, coronary artery disease; CHD, coronary heart disease; CMD, coronary microvascular dysfunction; CSX, cardiac syndrome X; DASI, Duke Activity Status Index; HADS, Hospital Anxiety and Depression scale; HAQ, health anxiety questionnaire; INOCA, ischemia with no-obstructive coronary arteries; METs, metabolic equivalents; MI, myocardial infarction; NOCAD, non-obstructive coronary artery disease; PHQ-9, patient health questionnaire-9; PSS, perceived stress scale; QOL, quality of life; SAQ, Seattle Angina Questionnaire; STAI, Spielberger Trait Anxiety Inventory; WISE, Women’s Ischemia Syndrome Evaluation; WLQ, Work Limitations Questionnaire; CAQ, cognitive avoidance questionnaire; IMR, index of microvascular resistance; DSM-IV, Diagnostic and Statistical Manual of Mental Disorders, 4th edition; BAI, Beck anxiety inventory; THIO, the heart inside out.
The heterogeneous pathophysiology underpinning CMD and the need for more pertinent randomized trials pose concerns about the optimal management of CMD [90]. Beta-blockers, renin–angiotensin system inhibitors, and statins are the cornerstone CMD therapies [5].
Beta-blockers exert vasodilatory properties and are the mainstay therapy for CMD without evidence of coronary artery spasm. Nebivolol might be the most effective drug in the context of CMD and has been proven in an elegant study enclosing 38 patients with cardiac syndrome X to mitigate the frequency of angina attacks while improving functional capacity compared to metoprolol [91].
Renin–angiotensin system inhibitors participate in several vascular protection and vasodilatory pathways. The addition of quinapril on top of optimal medical therapy was able to improve CFR and lower the rate of chest pain in a sub-analysis of the WISE registry involving 78 women with CMD [92].
Statins prevent endothelial dysfunction via downregulation of ROS production and inflammatory pathways. In an elegant study by Fábián et al. [93], the administration of simvastatin resulted in a 52% relative increase in brachial artery flow-mediated dilation compared to the placebo.
Recently, novel therapeutic strategies have been demonstrated to mitigate angina symptoms and/or to improve microvascular function.
ET-1 is a fascinating therapeutic target implicated in coronary vasoconstriction and endothelial dysfunction. Small clinical trials found improved myocardial perfusion after administration of ET-1 receptor antagonists [94, 95]. The large ongoing Precision Medicine with Zibotentan in Microvascular Angina (PRIZE) trial (ClinicalTrials.gov Identifier: NCT04097314) will address the potential role of zibotentan in improving functional capacity, assessed through treadmill exercise time, in a cohort of 356 patients with CMD.
Coronary sinus reducer implantation consists of the percutaneous deployment of a stainless-steel mesh in the coronary sinus. In recent studies, the putative ability to foster homogenous myocardial perfusion has been linked to high rates of angina relief and improvement in microcirculatory function [96]. The Coronary Sinus Reducer Objective Impact on Symptoms, MRI Ischaemia and Microvascular Resistance (ORBITA-COSMIC) trial enrolled 61 patients with CCS, angina, and evidence of myocardial ischemia unsuitable for coronary revascularization. At 6 months, coronary sinus reducer implantation reduced the number of daily angina episodes, with no evidence of improved myocardial perfusion [97]. In the INROAD (Index of Microcirculation Resistance Evaluation in Patients with Coronary Sinus Reducer Implantation) study, 21 patients with a history of coronary revascularization and refractory angina deemed not amenable for further revascularization underwent serial invasive coronary physiological assessment at 4-months. Here, the coronary sinus reducer lowered the rate of abnormal IMR by three times while significantly improving the CFR value and Seattle angina questionnaire summary score [98]. The ongoing COSIRA-2 (Efficacy of the Coronary Sinus Reducer in Patients With Refractory Angina II) phase 3 trial (Clinical Trial.gov Identifier: NCT05102019) will address the role of the coronary sinus reducer in patients with CMD.
Evidence of defective endothelial cell function has provided the foundation for testing regenerative therapy in CMD using CD34+ stem cells. Preclinical studies outlined the ability of CD34+ cells to drive endothelial proliferation and microvascular repair. The ESCaPE-CMD (Autologous CD34 cell therapy for the treatment of coronary microvascular dysfunction in patients with angina and nonobstructive coronary arteries) [99] and the IMPROvE-CED (Intracoronary CD34+ Cell Therapy for Treatment of Coronary Endothelial Dysfunction in Patients with Angina and Nonobstructive Coronary Arteries) [100] trials showed a significant reduction in angina episodes and need for nitrate therapy as well as an increase in CFR after intracoronary infusion of CD34+ stem cells. The ongoing FREEDOM (Clinical Trial.gov Identifier: NCT04614467) placebo-controlled trial will further clarify the role of CD34+ stem cells in patients with CMD and persistent angina.
The implementation of cardiac rehabilitation programs, when added to optimal
medical therapy, has significantly benefited the physical and mental health of
CMD patients. Leaf et al. [101] observed that a 12-week exercise
training program improved functional capacity and ischemic ST changes during
cardiopulmonary exercise training in patients with cardiac syndrome X. Later, a
pivotal study by Eriksson et al. [102] showed additional benefits of
physical training on endothelial function, neuroendocrine profile as well as
anginal burden and QOL. Moreover, exercise training has recently been associated
with improved CFV [103]. Indeed, high-intensity interval training (HIIT) has
notably emerged as an effective strategy, showing superior benefits in exercise
capacity, VO2 peak, endothelial function, cardiac function, and QOL compared
to conventional moderate-intensity continuous training [104]. HIIT involves
intermittent bouts of vigorous activity alternating with periods of active
recovery and should be tailored to the functional capacity of each individual.
Robust evidence supports the beneficial effects of HIIT protocols in patients
with CAD [104, 105]. Interestingly, a pilot study investigated the potential
benefits of an aerobic HIIT program consisting of treadmill exercises in a
4-minute
Cognitive-behavioral therapy is an emerging strategy for managing CMD, comprising a wide spectrum of interventions focused on stress control, optimized coping skills, relaxation techniques, and counseling programs. Cunningham et al. [107] found a significant improvement in terms of the burden of psychological disorders as well as physical status, anginal attacks, and exercise tolerance after a 12-week behavioral therapy in a cohort of nine women with cardiac syndrome X. The ongoing SAMCRO (Standardizing the Management of patients with Coronary Microvascular Dysfunction) study will investigate whether a multi-domain lifestyle intervention, consisting of dietary counseling, strict management of CV risk factors, tailored medical therapy based on the invasive assessment of CMD and coronary vasomotion, exercise training and psychological intervention, could help in improving individual’s QOL and rates of psychological disorders in a cohort of 120 patients with angina and non-obstructive CAD.
The advent of invasive functional tests has delineated two major endotypes (structural and functional) of CMD, which largely differ in pathophysiology and invasive physiological characteristics [3]. Abnormal microvascular resistance is the key feature of structural CMD, while an imbalance between vasodilatory and vasoconstrictive agents has been postulated in functional CMD [32]. Whether these endotypes could benefit from targeted therapy has never been investigated. The ongoing MINOSSE (platelet and endothelial activation in angina with non-obstructive coronary artery disease and microvascular dysfunction) study will explore potential differences in platelet function and biochemical microenvironment between structural vs. functional CMD endotypes. This study could help identify novel potential therapeutic targets for CMD.
Finally, novel diagnostic and therapeutic strategies capable of forecasting the natural history of patients with CMD (the onset of CAD, vasomotor disorders, and/or HFpEF) and preventing adverse events are warranted.
CMD is a condition frequently encountered in clinical practice caused by a variable combination of structural and functional abnormalities of coronary microcirculation. CMD is associated with a high risk of complications, including MACEs and HFpEF, as well as a poor QOL. Evidence on novel therapeutic strategies, including cardiac rehabilitation programs, HIIT protocols, and cognitive–behavioral therapy, are largely awaited to improve the prognosis of patients with CMD.
CAD, coronary artery disease; CCS, chronic coronary syndrome; CFR, coronary flow reserve; CFV, coronary flow velocity; CMD, coronary microvascular dysfunction; CMR, cardiac magnetic resonance; DASI, Duke Activity Status Index; ET-1, endothelin-1; HIIT, high-intensity interval training; hMR, hyperemic microvascular resistance; HFpEF, heart failure with preserved ejection fraction; IMR, index of microcirculatory resistance; INOCA, ischemia with non-obstructive coronary artery disease; MACEs, major adverse cardiovascular events; MACCE, major adverse cardiac and cerebro vascular events; METs, metabolic equivalents; MI, myocardial infarction; MRR, microvascular resistance reserve; NO, nitric oxide; QOL, quality of life; PET, positron emission tomography; ROS, reactive oxygen species.
FLG, GB, AD: design, conceptualization, writing-original draft. DD, MC, MDG, GD, RN: methodology, data research, drafting of the manuscript. GN: Substantial contributions to the conception or design of the work; supervision, writing-review and editing. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

