1 School of Nursing, Bengbu Medical University, 233030 Bengbu, Anhui, China
2 Department of Cardiac Surgery, The First Affiliated Hospital of Bengbu Medical University, 233000 Bengbu, Anhui, China
3 Clinical Medical School, Zhengzhou University, 450001 Zhengzhou, Henan, China
Abstract
To systematically evaluate risk factors for stress-induced hyperglycemia in patients without diabetes after cardiac surgery.
Databases including CNKI, WanFang data, VIP, SinoMed, PubMed, Web of Science, Embase, and the Cochrane Library were searched using computer retrieval. The data were subjected to an in-depth meta-analysis using RevMan 5.4 and Stata 15.0 software.
This study involved 11,645 postoperative cardiac surgery patients, including 8 case-control studies and 3 cohort studies, over which 18 risk factors were identified. The results of the meta-analysis indicated that statistically significant risk factors included age >65 years [odds ratios (OR) (95% CI ) = 3.47 (2.61–4.32)], female gender [OR (95%) = 1.54 (1.34–1.76)], combined heart valve and coronary artery bypass surgery [OR (95%) = 1.82 (1.23–2.70)], ejection fraction <40% [OR (95%) = 1.38 (1.17–1.63)], history of heart surgery [OR (95%) = 1.30 (1.06–1.59)], myocardial infarction [OR (95%) = 1.17 (1.05–1.31)], hyperlipidemia [OR (95%) = 0.76 (0.67–0.86)], hypertension [OR (95%) = 1.12 (1.03–1.22)], anticoagulant medication [OR (95%) = 0.77 (0.65–0.90)], cardiopulmonary bypass time >2 hours [OR (95%) = 20.26 (17.03–23.48)] and history of cardiopulmonary bypass [OR (95%) = 1.24 (1.09–1.41)].
Current evidence suggests that there are key risk factors for postoperative stress hyperglycemia in patients without diabetes who have undergone cardiac surgery. These factors can help identify patients at a high risk of perioperative stress hyperglycemia during cardiac surgery. This evidence provides a basis for healthcare professionals to develop predictive management strategies for perioperative stress hyperglycemia in patients without diabetes. However, more high-quality studies are required to address the limitations of the current research.
CRD42024479215, https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=479215.
Keywords
- cardiac surgery
- stress-induced hyperglycemia
- risk factors
- systematic review and meta-analysis
Stress hyperglycemia (SHG) refers to a temporary increase in
blood glucose levels under high-stress conditions, such as major trauma, severe
infection, or cardiovascular incidents, in patients with no prior history of
diabetes [1, 2]. According to the American Diabetes Association (ADA) consensus,
SHG is defined as a fasting blood glucose level
A comprehensive literature search was conducted using several databases, including CNKI, Wanfang Data, VIP, SinoMed, PubMed, Web of Science, Embase, and the Cochrane Library. The search period was extended to December 1, 2023. The included literature consisted of works in Chinese, English, and other languages, as well as grey literature, employing a combination of subject and free terms. The references of the included studies were also traced. The Chinese search terms included “off-pump coronary artery bypass”, “cardiac surgery”, “stress hyperglycemia”, “hyperglycemia”, “stress-induced hyperglycemia”, “risk factors”, and “influencing factors”. The English search terms included (“cardiac operation” OR “cardiac surgical procedures”) OR (“coronary artery bypass, off-pump”) AND (“hyperglycemia” OR “irritable hyperglycemia” OR “stress hyperglycemia”).
2.2.1.1 Diagnosis and Measurement of Stress Hyperglycemia
The review Stress Hyperglycemia, published in the
prestigious journal The Lancet [3], mentioned that SHG typically refers
to transient hyperglycemia occurring during illness, usually in patients without
a prior history of diabetes. The ADA defines hospital-related
hyperglycemia as fasting blood glucose
2.2.1.2 Study Types
Case-control studies and Cohort studies.
2.2.1.3 Postoperative Monitoring Timing
The first 24–48 hours [15] after surgery are a high-risk period for stress-induced hyperglycemia. Therefore, frequent blood glucose monitoring is necessary, especially when insulin or other interventions are being used.
2.2.1.4 Monitoring Methods
① Capillary Blood Glucose Monitoring: Blood is drawn from the fingertip and tested immediately using a portable glucometer. This method is convenient and quick, but may be affected by local circulation conditions. ② Venous Plasma Glucose Measurement: Blood is drawn from a vein, and the glucose concentration in the plasma is measured in a laboratory. This method is more accurate than capillary blood glucose testing but slower, and is typically used for diagnosis and precise monitoring. ③ Continuous Glucose Monitoring (CGM): A subcutaneous sensor is used to monitor blood glucose levels in real time, providing continuous tracking of glucose fluctuations. This is particularly useful for postoperative monitoring of stress-induced hyperglycemia, especially in critically ill patients who require close observation.
2.2.1.5 Study Subjects
Patients without diabetes who underwent cardiac surgery, regardless of race, nationality, or disease duration.
2.2.1.6 Exposure Factors
Exposure factors included general risk factors potentially associated with the patient during the perioperative period, preoperative comorbidities, perioperative medications, and certain intraoperative risks related to extracorporeal circulation.
2.2.1.7 Outcome Measure
The occurrence of SHG after cardiac surgery.
① Newcastle-Ottawa Scale (NOS) Score Below 6 [16].
② Studies that were duplicates or suspected of being duplicate reports
were excluded. ③ Studies with incomplete, insufficient, or erroneous
data were excluded. ④ Exclusion criteria included the following:
impaired fasting glucose (IFG) patients (fasting blood glucose 5.6–6.9 mmol/L,
with a 2 hour postprandial blood glucose
The NOS [16] is a tool specifically designed to assess the quality of non-randomized studies, including case-control and cohort studies. Given the potential biases and confounding factors inherent in non-randomized studies, it is essential to evaluate their quality. The NOS helps researchers to systematically and quantitatively assess whether the design, execution, and interpretation of results in these studies are scientifically sound and valid. The NOS evaluates studies across three key domains: ① Selection: Assesses whether the selection of cases or controls in the study is appropriate. ② Comparability: Evaluates how well the study controls confounding factors between the study and control groups to ensure that the results are comparable. ③ Outcome/Exposure: Examines whether the measurement of exposure or outcomes is accurate, consistently applied across study and control groups, and considers issues such as non-response rates. The Selection domain is scored out of 4 points, Comparability out of 2 points, and Outcome/Exposure out of 3 points, with a total score ranging from 0 to 9. Higher scores indicate better study quality and a lower risk of bias.
A NOS score of
Two researchers independently performed literature screening and data collection. After completing independent data extraction, the two sets of extracted data were compared with particular attention to key variables (such as sample size and region). For any discrepancies, the original literature was reviewed to verify which set of data was correct; if discrepancies arose, discussions were held to reach a consensus, and third-party consultation was sought if necessary. The initial screening involved reviewing titles to exclude irrelevant articles, followed by abstract and full-text reviews to determine inclusion. When required, the original authors were contacted for additional data. The study primarily included basic information about the research participants, such as the first author, region, and publication date, as well as demographic data, including sample size and age. It also included the number of patients and main factors for bias risk assessment. The outcome measures and related data were reviewed independently by two researchers. If disagreements occurred, a third-party expert was consulted, or multiple discussions were held until a consensus was reached.
Data were analyzed that the Revman 5.4 software (Cochrane Collaboration, Oxford, Oxon, UK), developed by the UK Cochrane
Collaboration, and the Stata 15.0 software (Statacorp, college station, TX, USA), created by the US Computer Resource
Center. Count data are expressed as odds ratios (OR) and continuous data as mean
differences (MD), with each effect presented as a point estimate and a 95%
confidence interval (CI). Heterogeneity was assessed using Chi-squared
(
This study has been registered in the National Institute for Health Research (NIHR) PROSPERO database, registration number CRD42024479215 (https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=479215).
A total of 4470 relevant articles were retrieved from the CNKI (n = 141), VIP (n = 2), WanFang Data (n = 144), SinoMed (n = 118), PubMed (n = 1724), Web of Science (n = 1013), Embase (n = 779), and Cochrane Library (n = 549) databases. This study included 405 articles in Chinese and 4065 articles in English. The articles were imported into EndNote and duplicates were removed. After the initial screening of titles and abstracts, 3543 articles were selected for further review. Ultimately, 11 articles were included in this study, comprising eight case-control studies [7, 11, 13, 17, 18, 19, 20, 21] and three cohort studies [12, 22, 23]. These studies involved 11,645 patients who underwent cardiac surgery-related procedures. The detailed literature screening process is illustrated in Fig. 1.
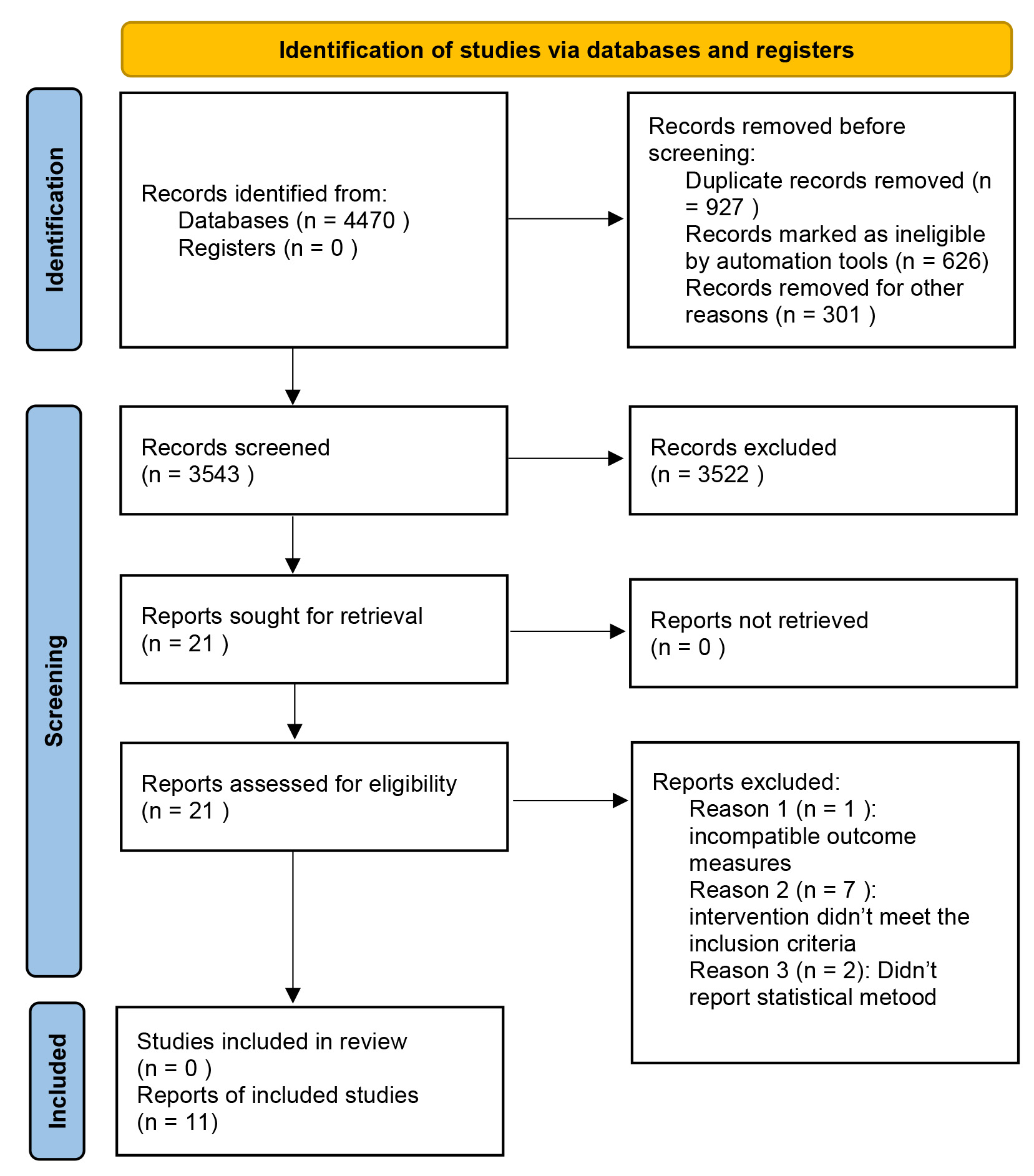 Fig. 1.
Fig. 1.
Literature screening flow chart.
A total of 4470 relevant articles were retrieved from the following databases: CNKI (n = 141), VIP (n = 2), WanFang Data (n = 144), SinoMed (n = 118), PubMed (n = 1724), Web of Science (n = 1013), Embase (n = 779), and Cochrane Library (n = 549). This study included 405 articles in Chinese and 4065 articles in English. The articles were imported into EndNote and duplicates were removed. After the initial screening of titles and abstracts, 3543 articles were selected for further review. Ultimately, 11 articles were included in this study, comprising eight case-control studies [7, 11, 13, 17, 18, 19, 20, 21] and three cohort studies [12, 22, 23]. These studies involved 11,645 patients who underwent cardiac surgery-related procedures. The detailed literature screening process is illustrated in Table 1 (Ref. [7, 11, 12, 13, 17, 18, 19, 20, 21, 22, 23]), Table 2 (Ref. [12, 22, 23]), Table 3 (Ref. [7, 11, 13, 17, 18, 19, 20, 21]).
| Include in references | Country | Study type | Age (years) | Researching spells | Sample size | Type of operation | Number of cases | Incidence | Risk factors |
| Amit A. Prasad et al. 2007 [7] | USA | case control study | 66–70 | 2004–2006 | 162 | Cardiac surgery | 35 | 21.6% | ①②⑤⑧⑩⑪⑫⑬⑭⑮⑰ |
| Vikaesh Moorthy et al. 2019 [11] | Singapore | case control study | 46.5–69.7 | 2008–2010 | 1602 | CABG | 898 | 56.0% | ②⑤⑩⑫⑭⑮⑰⑱ |
| Rajesh Garg et al. 2013 [12] | USA | cohort study | 50.9–78.3 | 2004–2009 | 3658 | Valve surgery and CABG | 1453 | 40.0% | ①②③④⑤⑥⑦⑧⑨⑩⑰ |
| Russell E. Anderson et al. 2005 [22] | Sweden | cohort study | 64–69 | 2004 | 45 | CABG | 23 | 51.1% | ①②⑤⑦⑧⑩⑪⑯ |
| Roma Y. Gianchandani et al. 2015 [23] | USA | case control study | 55–67 | 2015 | 61 | Valve surgery and CABG | 49 | 80.3% | - |
| Jiang Jiduan 2016 [17] | China | case control study | 46.3–63.9 | 2014–2015 | 69 | Valve surgery | 27 | 39.1% | ②⑰ |
| Xiaojue Li et al. 2023 [13] | China | cohort study | 54–67 | 2011–2014 | 5450 | CABG | 4342 | 79.7% | ②③④⑥⑦⑧⑨⑩⑫⑬⑭⑮⑯⑱ |
| Utkan Sevuk et al. 2014 [18] | Turkey | case control study | 50.2–72 | 2011–2014 | 200 | CABG | 100 | 50.0% | ②③ |
| Yubin Chen et al. 2023 [19] | China | case control study | 39.4–64.3 | 2016–2020 | 203 | Type A aortic dissection | 86 | 42.4% | ②③⑤⑩ |
| Shen Minwei et al. 2019 [20] | China | case control study | 36–58 | 2016–2017 | 100 | Type A aortic dissection | 31 | 31.0% | ② |
| Zhang Yan et al. 2010 [21] | China | case control study | 25–75 | 2006–2008 | 95 | Valve surgery and CABG | 41 | 43.1% | - |
① age
| Incorporate into study | Study population selection | Comparability | Ending | Total points | |||||
| (4 points) | (2 points) | (3 points) | |||||||
| Exposure group representativeness | Non-exposed group selection | Identification of exposure groups | Outcome events before the study begins | Comparability of exposed and unexposed groups | Outcome event evaluation | Adequacy of follow-up | Integrity of follow-up | ||
| Rajesh Garg et al. 2013 [12] | 1 | 1 | 1 | 0 | 2 | 1 | 0 | 1 | 7 |
| Russell E. Anderson et al. 2005 [22] | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 0 | 8 |
| Roma Y. Gianchandani et al. 2015 [23] | 1 | 0 | 1 | 1 | 2 | 1 | 1 | 0 | 7 |
| Incorporate into study | Study population selection | Comparability | Ending | Total points | |||||
| (4 points) | (2 points) | (3 points) | |||||||
| Whether cases are properly defined and diagnosed | Case representation | Contrast selection | Definition of contrast | Comparability of cases and controls | Methods of investigation and assessment of exposure | Case and control investigation methods | Non response rate | ||
| Amit A. Prasad et al. 2007 [7] | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 0 | 8 |
| Vikaesh Moorthy et al. 2019 [11] | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 0 | 8 |
| Xiaojue Li et al. 2023 [13] | 1 | 1 | 1 | 0 | 2 | 1 | 0 | 0 | 6 |
| Jiang Ji duan 2016 [17] | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 0 | 8 |
| Utkan Sevuk et al. 2014 [18] | 1 | 1 | 1 | 0 | 2 | 1 | 1 | 0 | 7 |
| Yubin Chen et al. 2023 [19] | 1 | 1 | 1 | 1 | 2 | 1 | 0 | 1 | 8 |
| Shen Minwei et al. 2019 [20] | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 0 | 8 |
| Zhang Yan et al. 2010 [21] | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 0 | 8 |
3.3.1.1 Age
Three studies [7, 12, 22] reported on the relationship between age
3.3.1.2 Gender and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Nine studies [7, 11, 12, 13, 17, 18, 19, 20, 22] reported on the relationship between
gender and SHG in patients without diabetes after cardiac surgery. The
heterogeneity test revealed high heterogeneity among the studies (I2 = 83%,
p
3.3.1.3 Smoking and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Four studies [12, 13, 18, 19] examined the relationship between smoking and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 29%, p = 0.24). The pooled analysis using a fixed-effect model revealed that smoking had no significant impact on the occurrence of SHG in patients without diabetes after cardiac surgery, with no statistically significant difference [WMD = 0.97, 95% CI (0.89, 1.06), Z = 0.62, p = 0.53].
3.3.1.4 Combined Valve and Coronary Artery Bypass Surgery and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Two studies [12, 13] reported on the relationship between combined valve and coronary artery bypass surgery and SHG in patients without diabetes after cardiac surgery. The heterogeneity test revealed high heterogeneity among the studies (I2 = 87%, p = 0.006). The pooled analysis using a random-effect model indicated that combined valve and bypass surgery significantly impacts SHG in patients without diabetes after cardiac surgery, with a statistically significant difference [WMD = 1.82, 95% CI (1.23, 2.70), Z = 3.01, p = 0.003].
3.3.1.5 Ejection Fraction
Two studies [11, 13] investigated the relationship between ejection fraction
3.3.1.6 History of Cardiac Surgery and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Four studies [7, 11, 12, 22] reported on the relationship between a history of cardiac surgery and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 7%, p = 0.36). The pooled analysis using a fixed-effect model demonstrated that a history of cardiac surgery significantly impacts SHG in patients without diabetes after cardiac surgery, with a statistically significant difference [WMD = 1.30, 95% CI (1.06, 1.59), Z = 2.48, p = 0.01].
3.3.2.1 History of Cerebrovascular Disease and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Two studies [12, 13] reported on the relationship between a history of cerebrovascular disease and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 0%, p = 0.85). A pooled analysis using a fixed-effect model demonstrated that a preoperative history of cerebrovascular disease does not significantly impact SHG in patients without diabetes after cardiac surgery, with no statistically significant difference [WMD = 1.08, 95% CI (0.93, 1.26), Z = 0.98, p = 0.33].
3.3.2.2 Myocardial Infarction and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Three studies [12, 13, 22] reported on the relationship between myocardial infarction and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 12%, p = 0.32). The pooled analysis using a fixed-effect model revealed that a history of myocardial infarction significantly impacts SHG in patients without diabetes after cardiac surgery, with a statistically significant difference [WMD = 1.17, 95% CI (1.05, 1.31), Z = 2.76, p = 0.006].
3.3.2.3 Hyperlipidemia and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Four studies [7, 12, 13, 22] examined the relationship between hyperlipidemia and
SHG in patients without diabetes after cardiac surgery. The heterogeneity test
revealed high heterogeneity among the studies (I2 = 90%, p
3.3.2.4 History of Peripheral Vascular Disease and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Two studies [12, 13] reported on the relationship between a history of peripheral vascular disease and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed high heterogeneity among the studies (I2 = 59%, p = 0.12). The pooled analysis using a random-effect model demonstrated that a preoperative history of peripheral vascular disease does not significantly impact SHG in patients without diabetes after cardiac surgery, with no statistically significant difference [WMD = 1.21, 95% CI (0.88, 1.66), Z = 1.18, p = 0.24].
3.3.2.5 Hypertension and its Relationship with SHG in Patients without Diabetes Patients after Cardiac Surgery
Six studies [7, 11, 12, 13, 19, 22] reported on the relationship between hypertension and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 19%, p = 0.29). A pooled analysis using a fixed-effect model revealed that hypertension significantly impacts SHG in patients without diabetes after cardiac surgery, with a statistically significant difference [WMD = 1.12, 95% CI (1.03, 1.22), Z = 2.68, p = 0.007].
3.3.2.6 Unstable Angina and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Two studies [7, 22] reported on the relationship between unstable angina and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 0%, p = 0.80). The pooled analysis using a fixed-effect model demonstrated that unstable angina does not significantly impact SHG in patients without diabetes after cardiac surgery, with no statistically significant difference [WMD = 1.19, 95% CI (0.59, 2.41), Z = 0.48, p = 0.63].
3.3.3.1 Perioperative Use of Anticoagulants and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Two studies [7, 13] reported a relationship between the perioperative use of anticoagulants and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 0%, p = 0.60). The pooled analysis using a fixed-effect model revealed that perioperative use of anticoagulants significantly impacts SHG in patients without diabetes after cardiac surgery, with a statistically significant difference [WMD = 0.77, 95% CI (0.65, 0.90), Z = 3.30, p = 0.001].
3.3.3.2 Perioperative Use of Lipid-lowering Drugs and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Two studies [7, 13] examined the relationship between the perioperative use of lipid-lowering drugs and SHG in non-diabetic patients after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 0%, p = 0.91). The pooled analysis using a fixed-effect model demonstrated that perioperative use of lipid-lowering drugs does not significantly impact SHG in patients without diabetes after cardiac surgery, with no statistically significant difference [WMD = 0.88, 95% CI (0.73, 1.05), Z = 1.46, p = 0.14].
3.3.3.3 Perioperative Use of Beta-blockers and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Two studies [7, 13] reported a relationship between the perioperative use of beta-blockers and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed low heterogeneity among the studies (I2 = 0%, p = 0.89). The pooled analysis using a fixed-effect model indicated that perioperative use of beta-blockers does not significantly impact SHG in patients without diabetes after cardiac surgery, with no statistically significant difference [WMD = 0.81, 95% CI (0.64, 1.03), Z = 1.70, p = 0.09].
3.3.3.4 Perioperative Use of Angiotensin-Converting Enzyme (ACE) Inhibitors and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Three studies [7, 11, 13] reported on the relationship between perioperative use of ACE inhibitors and SHG in patients without diabetes after cardiac surgery. The heterogeneity test showed high heterogeneity among the studies (I2 = 89%, p = 0.0001). Sensitivity analysis identified Xiaojue Li et al. [13] as the source of heterogeneity. After excluding this study, heterogeneity decreased to 74%. The pooled analysis using a random-effect model showed that perioperative use of ACE inhibitors does not significantly impact SHG in patients without diabetes after cardiac surgery, with no statistically significant difference [WMD = 1.58, 95% CI (0.74, 3.38), Z = 1.19, p = 0.23].
3.3.4.1 Cardiopulmonary Bypass and its Relationship with SHG in Patients without Diabetes after Cardiac Surgery
Two studies [13, 22] reported the relationship between cardiopulmonary bypass (CPB) and SHG in patients without diabetes after cardiac surgery. The heterogeneity test indicated high heterogeneity among the studies (I2 = 0%, p = 0.76). Pooled analysis using a random-effects model showed that the use of CPB during cardiac surgery did not significantly affect the incidence of SHG in patients without diabetes postoperatively [WMD = 1.24, 95% CI (1.09, 1.41), Z = 3.24, p = 0.001].
3.3.4.2 CPB Duration
Three studies [7, 12, 17] investigated the relationship between CPB duration
exceeding 2 hours and SHG in patients without diabetes after cardiac surgery. The
heterogeneity test showed moderate heterogeneity among the studies (I2 =
47%, p = 0.17). The pooled analysis using a fixed-effects model
demonstrated that the duration of cardiopulmonary bypass
| Risk factor | Number of articles included | I2 (%) | p | OR | 95% CI | Effect model | |
| General risk factors for patients | |||||||
| Age |
3 [7, 12, 22] | 0% | 3.47 | 2.61, 4.32 | fixed effect model | ||
| Female | 8 [7, 11, 13, 17, 18, 19, 20, 22] | 41% | 1.54 | 1.34, 1.76 | fixed effect model | ||
| Smoking history | 4 [12, 13, 18, 19] | 29% | 0.53 | 0.97 | 0.89, 1.06 | fixed effect model | |
| Heart valve surgery combined with bypass surgery | 2 [12, 13] | 87% | 0.003 | 1.82 | 1.23, 2.70 | random effect model | |
| Ejection fraction |
2 [11, 13] | 0% | 0.0002 | 1.38 | 1.17, 1.63 | fixed effect model | |
| History of cardiac surgery | 4 [7, 11, 12, 22] | 7% | 0.01 | 1.30 | 1.06, 1.59 | fixed effect model | |
| Risk factors for preoperative comorbidities | |||||||
| Cerebrovascular disease history | 2 [12, 13] | 0% | 0.33 | 1.08 | 0.93, 1.26 | fixed effect model | |
| Myocardial infarction | 3 [12, 13, 22] | 12% | 0.006 | 1.17 | 1.05, 1.31 | fixed effect model | |
| Hyperlipemia | 3 [7, 13, 22] | 0% | 0.76 | 0.67, 0.86 | fixed effect model | ||
| Peripheral vascular disease | 2 [12, 13] | 59% | 0.24 | 1.21 | 0.88, 1.66 | random effect model | |
| Hypertension | 6 [7, 11, 12, 13, 19, 22] | 19% | 0.007 | 1.12 | 1.03, 1.22 | fixed effect model | |
| Unstable angina | 2 [7, 22] | 0% | 0.63 | 1.19 | 0.59, 2.41 | fixed effect model | |
| Risk factors for preoperative drug use in patients | |||||||
| Anticoagulant drug | 2 [7, 13] | 0% | 0.001 | 0.77 | 0.65, 0.90 | fixed effect model | |
| Lipid-regulating drug | 2 [7, 13] | 0% | 0.14 | 0.88 | 0.73, 1.05 | fixed effect model | |
| 2 [7, 13] | 0% | 0.09 | 0.81 | 0.64, 1.03 | fixed effect model | ||
| Renin angiotensin converting enzyme inhibitors | 2 [7, 11] | 74% | 0.23 | 1.58 | 0.74, 3.38 | random effect model | |
| Risk factors associated with cardiopulmonary bypass | |||||||
| Extracorporeal circulation | 2 [13, 22] | 0% | 0.001 | 1.24 | 1.09, 1.41 | fixed effect model | |
| Cardiopulmonary bypass time |
3 [7, 12, 17] | 47% | 20.26 | 17.03, 23.48 | fixed effect model | ||
Sensitivity analysis revealed changes in heterogeneity before and after
excluding individual studies. Hyperlipidemia [before excluding a single study
[12] (p = 0.66, I2 = 90%); after excluding the study (p
The study included a total of 11 published papers [7, 11, 12, 13, 17, 18, 19, 20, 21, 22, 23]. Begg’s test was conducted, and the results indicated a low risk of publication bias among the included studies (Z = –2.65, p = 0.0127).
Sources of heterogeneity in the meta-analysis results: Clinical characteristics and disease severity. The severity of the disease in the study subjects, along with the presence of other chronic conditions, may lead to varying outcomes, contributing to heterogeneity. Different study designs can introduce heterogeneity, as each type of study has inherent biases and limitations. Some low-quality studies may lead to publication bias, further contributing to heterogeneity. Methods to reduce sources of heterogeneity include strict inclusion and exclusion criteria: clearly defined, stringent criteria should be applied to ensure consistency in key characteristics of the study population, thereby minimizing variations in outcomes due to sample heterogeneity. Sensitivity and subgroup analysis: When significant heterogeneity is present among the influencing factors, sensitivity and subgroup analyses can be conducted by categorizing and grouping the data. By sequentially excluding studies involving specific influencing factors, the sources of heterogeneity can be identified, thereby reducing heterogeneity and uncovering the effects within specific populations.
Patients in the SHG group were more likely to be over 65 years old than those in
the non-SHG group, making age an independent risk factor for SHG after cardiac
surgery, which aligns with the findings of Xie Huihui [24]. This study showed a
significant association between age and SHG in non-diabetic patients after
cardiac surgery, with the severity of SHG increasing with age
[25]. Elderly patients often have weakened immune responses and
poorer stress responses due to underlying conditions and declining organ
function; however, the impact of sex on stress hyperglycemia remains
controversial. This study found that female sex is a relevant risk factor for SHG
in non-diabetic patients after cardiac surgery, suggesting that older women with
poorer cardiac function are more prone to SHG. The release of stress hormones and
insulin resistance in response to surgical stress can lead to SHG. Although the
mechanism related to female hormones requires further investigation, this study
underscores the importance of early prediction and management of SHG in female
patients during the perioperative period [26, 27]. Research by ME Ertorer
et al. [28] on 494 ICU patients with coronary artery disease confirmed
sex differences in SHG. Sharma et al. [29] also demonstrated a
sex-related association with SHG levels. Stress conditions prolong the elevation
of hormone levels, contributing to SHG in patients without diabetes post-cardiac
surgery. The traumatic nature of surgery, particularly complex procedures like
combined valve and coronary artery bypass surgeries, increases the risk of SHG
compared to standard cardiac surgeries [30]. Patients with a history of cardiac
surgery often experience physiological and metabolic changes, including SHG,
consistent with the results of Christos Kourek et al. [31]. In patients
with a history of heart surgery [32], unique cardiovascular characteristics lead
to a more pronounced response to SHG risk factors. Reduced ejection fraction is a
significant risk factor for SHG, as declining cardiac function leads to decreased
myocardial contractility, a sharp drop in cardiac output, worsening
microcirculation, and further exacerbation of myocardial injury, which is
consistent with the findings of Stalikas N et al. [33]. This
meta-analysis also identified hypertension as a risk factor for SHG in patients
post-cardiac surgery. Izzo R et al. [34] and Conen D [35] found that at
admission, systolic blood pressure is an independent risk factor for SHG.
Retrospective studies [29] have shown that prehypertensive individuals have
higher fasting blood glucose levels than those with ideal blood pressure,
indicating that while abnormal blood pressure may not directly cause glucose
abnormalities, it does affect blood glucose levels. A study [36] has shown that
effective blood pressure control in hypertensive patients helps reduce the
incidence of stress-induced hyperglycemia and the development of new-onset
diabetes. These mechanisms include hypertension and significant fluctuations in
blood pressure, which lead to insulin resistance, sympathetic nervous system
activation, Renin-angiotensin-aldosterone system (RAAS) activation, oxidative
stress, and inflammation [17], ultimately resulting in stress-induced
hyperglycemia [37]. After acute myocardial infarction, the sympathetic nervous
system activation increases the levels of glucagon, growth hormones,
glucocorticoids, and catecholamines. Research has shown that glucagon is a key
factor in gluconeogenesis, promoting hepatic glycogen breakdown, and inducing
hyperglycemia. These hormones and cytokines also have complex feedback
mechanisms, such as tumor necrosis factor (TNF)-
Current evidence suggests that age
The dataset from this study has been provided as part of the submitted manuscript.
MLZ, YKW and ZYZ designed the research study. MLZ and YKW performed the research. NNZ and LLW provided help and advice on the Systematic review and meta-analysis. HH and FX analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM25485.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

