1 Cardiology Division, Department of Medical Sciences, “Città della Salute e della Scienza” Hospital, Univerisity of Turin, 10116 Turin, Italy
2 Department of Medicine and Surgery, University of Insubria, 21100 Varese, Italy
3 Cardiology Unit, Department of Translational Medical Sciences, Monaldi Hospital, University of Campania “Luigi Vanvitelli”, 80131 Naples, Italy
4 Institute of Cardiology, IRCCS Azienda Ospedaliero Universitaria di Bologna, 40138 Bologna, Italy
5 Fondazione Policlinico Universitario Agostino Gemelli IRCCS, University “Cattolica del Sacro Cuore”, 00168 Rome, Italy
6 Cardiology and Arrhythmology Clinic, Department of Clinical, Special and Dental Sciences, Marche Polytechnic University, Marche University Hospital, 60126 Ancona, Italy
7 Cardiology Division, “Vito Fazzi” Hospital, 73100 Lecce, Italy
8 Department of Heart and Vessels, Ospedale di Circolo, 21100 Varese, Italy
9 Department of Cardiothoracic, Vascular Medicine and Intensive Care, Dell'Angelo Hospital, 30174 Mestre-Venice, Italy
10 Electrophysiology and Cardiac Pacing Unit, Cardiology Division, Monaldi Hospital, 80131 Naples, Italy
11 Cardiology Division, Department of Biomedical, Metabolic and Neural Sciences, University of Modena and Reggio Emilia, Policlinico di Modena, 41124 Modena, Italy
†These authors contributed equally.
§AIAC: Italian Association of Arrhythmology and Cardiac Pacing.
Abstract
Atrial high-rate episodes (AHREs) and subclinical atrial fibrillation (AF) are frequently registered in asymptomatic patients with cardiac implantable electronic devices (CIEDs) and insertable cardiac monitors (ICMs). While an increased risk of thromboembolic events (e.g., stroke) and benefits from anticoagulation have been widely assessed in the setting of clinical AF, concerns persist about optimal clinical management of subclinical AF/AHREs. As a matter of fact, an optimal threshold of subclinical episodes’ duration to predict stroke risk is still lacking and recently published randomized clinical trials assessing the impact of anticoagulation on thromboembolic events in this specific setting have shown contrasting results. The aim of this review is to summarize current evidence regarding classification and clinical impact of subclinical AF/AHREs and to discuss the latest evidence regarding the potential benefit of anticoagulation in this setting, highlighting which clinical questions are still unanswered.
Keywords
- atrial high-rate episode
- subclinical atrial fibrillation
- cardiac implantable electronic devices
- thromboembolic risk
- cognitive impairment
- anticoagulation
Atrial fibrillation (AF) is the most common sustained cardiac arrythmia in the adult population [1, 2]. The lifetime risk of AF in European individuals is estimated as 1 in 3, with an increasing incidence starting from the age of 50 years in males and 60 years in females, reaching a cumulative incidence of roughly 30% by the age of 90 years. Considering progressive ageing of the population, AF is estimated to affect more than 17.9 million people in Europe by 2030. The link between AF and increased incidence of thromboembolic events, namely transient ischemic attack (TIA), overt ischemic stroke, peripheral embolism and silent embolic lesions, has been assessed [3]. Independently from these events, AF is associated with a 30% increased risk of cognitive decline and dementia [4, 5], whose pathophysiology still needs to be fully clarified.
Considering its clinical impact and increasing prevalence, AF has become a prominent public health issue, prompting the need for a rapid diagnosis and correct clinical management. While diagnosis is straightforward in symptomatic patients, identification of asymptomatic patients is often achieved during rhythm monitoring after cerebrovascular accidents (CVAs) [6], questioning the need for AF screening, or occasionally at surface electrocardiogram (ECG). Furthermore, the increasing number of patients with cardiac implantable electronic devices (CIEDs) capable of atrial rhythm monitoring, as well as insertable cardiac monitors (ICMs), has led to frequent detection of atrial high-rate episodes (AHREs) or subclinical AF [7]. When these episodes are not associated with surface ECG documentation, there is uncertainty about correct clinical management and anticoagulation.
In the present review, we discuss the epidemiological and clinical impact of atrial fibrillation, providing insights into the latest research regarding the pathophysiological link between AF, cognitive decline and dementia. Subsequently, we focus on subclinical AF and AHREs with regard to their definition and impact on thromboembolic risk. Eventually, we summarize the latest evidence concerning use of anticoagulants in this specific setting, highlighting clinical issues that persist unsolved.
Despite being frequently encountered in clinical practice, confusion in terminology is still widespread when it comes to classifying AF. According to the latest guidelines [8], the definition of clinical AF implies the recording of a 12-lead surface ECG or at least 30 second single-lead tracing, documenting irregular R-R intervals and the absence of P waves. Depending on symptoms, clinical AF can be distinguished by being symptomatic or asymptomatic. The real proportion of asymptomatic patients is difficult to assess, varying from 10 to 40% between studies, depending on patients’ features, duration of follow-up and modality of screening [9, 10, 11]. However, asymptomatic AF is more frequent in male patients and when arrhythmia is persistent [9, 12].
On the other hand, the definition of AHRE and subclinical AF has been extremely
heterogeneous in literature, both in terms of atrial rate and episode duration
cut-offs, starting from any atrial tachyarrhythmia with an atrial rate
According to the European Society of Cardiology (ESC) guidelines [8], the
definition of AHRE implies the presence of an atrial tachyarrhythmia with an
atrial rate
According to guidelines [8] it is the single-episode duration to be considered in AHRE definition. However, it is important to introduce the concept of “AHRE/subclinical AF burden”, which is defined as the overall time spent in AHRE during a certain period, usually 24 hours. Guidelines suggest that both elements should be considered when trying to predict thromboembolic risk, as the dynamic entity of AHRE implies a progressive increase in episodes’ duration and daily or monthly burden during follow-up, as well as progression to clinical AF [15]. Despite the lack of a specific single episode duration to predict a thromboembolic event, the latest guidelines suggest that a single episode duration of 24 hours should be the cut off to consider use of oral anticoagulants (OACs), particularly in the presence of a high monthly burden [8].
Cardioembolic ischemic stroke is the most dreaded complication of AF. It can be AF’s first clinical manifestation in otherwise asymptomatic patients, while AF is detected in 25–30% of patients with embolic stroke of unknown source (ESUS), rising questions on the need for AF screening [16]. Despite diffusion of effective acute treatments, stroke is still associated with a dramatic increase in the risk of developing dementia, and cognitive impairments are found in nearly 70% of stroke survivors [17].
In the latest years, it has been demonstrated that AF is associated with a significant increase (30%) in the risk of developing cognitive decline/dementia independently from stroke or TIA, even in patients receiving OACs [5]. Despite overlapping risk factors, the relationship between AF and cognitive impairment persists after adjustment for these variables [18]. However, controlled trials are essential to prove the existence of a causal relationship between AF and cognitive impairment. The presence of causality seems to be favored by a possible biological gradient between arrhythmic burden and dementia, as suggested by the Rotterdam study [19], in which there was a correlation between quantitative exposure to AF and new-onset dementia in young patients. These results are consistent with those of the ARIC study, in which persistent, rather than paroxysmal, AF was associated with lower cognitive performances assessed through validated scores [20]. However, it remains to be established what the minimal amount of arrhythmic burden is, which is associated with cognitive decline and whether OAC therapy is efficient in preventing it independently from stroke [21]. Interestingly, the two trials designed to assess the potential benefit of OAC therapy on stroke risk in patients with AHREs deal with this issue. The ARTESIA trial [22] incorporates a cognitive substudy in which patients are periodically evaluated through cognitive assessment scales, and cognitive function changes are included in the secondary outcomes of the NOAH-AFNET 6 trial [23].
Several mechanisms have been proposed to explain the link between AF and cognitive decline in the absence of CVAs. First, silent cerebral lesions (SCLs) due to micro-embolic events are found in a high proportion of patients with AF and a negative anamnesis for stroke or TIA undergoing cerebral imaging, such as magnetic resonance. When clinically silent lesions are represented by large non-cortical and cortical infarction, they correlate with cognitive decline [24]. Another potential mechanism is represented by microbleeds [25], which are more frequently encountered in patients with AF compared to those in sinus rhythm. Their presence has been linked to an increased risk of mortality, intracranial hemorrhage and stroke [26].
More recently, researchers have been concentrating on the hemodynamic consequences of AF [27], especially focusing on cerebral perfusion. Using phase contrast MRI, Gardarsdottir at al. [28] demonstrated a reduction of cerebral blood flow and estimated brain tissue perfusion in patients with AF. Interestingly, cerebral blood flow reduction was reversible after 10 weeks from effective cardioversion, with a documented increase in tissue perfusion assessed through both MRI and arterial spin labeling [29]. Similarly, a small prospective study demonstrated a significant increase in cerebral blood flow assessed with phase-contrast MRI after successful ablation with maintenance of sinus rhythm beyond a blanking period of three months [30].
Based on data from two closed lump models simulating AF and sinus rhythm,
Saglietto et al. [31] proposed that beat-to beat variability during AF
results in the alternation of micro-hypertensive and micro-hypoperfusion events
in distal cerebral circulation. It has been hypothesized that the observed
high-variability in hemodynamic parameters could result in microbleeds and
infarctions, and therefore in the development of cognitive decline. Impact of
beat-to-beat variability in cerebral perfusion has been validated in
vivo using spatially resolved near-infrared spectroscopy (SR-NIRS) [31], which
showed that both hypoperfusive and hyperperfusive events at the microcirculatory
level were reduced after restoration of sinus rhythm through electrical
cardioversion (p
When SCLs are found, they are usually located at the subcortical level and involve the white matter. Considering lenticulostriate arteries (LSAs) are the main blood supply of this area, it was hypothesized that these terminal vessels could be particularly exposed to extreme hemodynamic events determined by AF’s “irregularly irregular” rhythm. Computational studies evaluating the effects of irregular AF rhythm, compared to sinus rhythm, on wall shear stress and intraluminal pressure along these vessels [32]. Results showed that the irregular AF rhythm exposes LSAs to both an increased range of wall shear stress, and to a wider range of intraluminal pressure along their course. The excessive oscillations from shear stress in both directions have been associated with both a pro-atherogenic effect [33] and acute complications, such as plaque erosion and rupture [34]. Similarly, oscillations in intraluminal pressure during hypertensive states can lead to brain barrier damage and accelerate lypohyalinosis, resulting in lacunar stroke; on the other hand, reduction of intraluminal pressure can provoke hypoperfusion and subsequent ischemia [35]. Coherently with these observations, a cognitive benefit from rhythm control would be expected. The AFFIRM [36] and EAST AFNET 4 [37] trials compared rate and rhythm control strategies in patients with AF. Neither AFFIRM (in which rate control was pursued through drug therapy) nor EAST AFNET 4 (in which catheter ablation was allowed) showed a benefit in cognitive functions in the rhythm control group. However, in the AFFIRM trial only 63% of patients in the rhythm control group were in sinus rhythm at 5-year follow-up [38]. Furthermore, more recent studies and metanalyses have shown an advantage of rhythm over rate control on dementia outcome (subdistribution hazard ratio (sHR) 0.86, 95% confidence interval (CI) 0.80–0.93 and hazard ratio (HR) 0.60, 95% CI 0.42–0.88 respectively) [39, 40]. Overall, evidence is still conflicting and relies on retrospective studies with possible selection bias. Furthermore, SCLs are a potential complication of catheter ablation itself, mostly represented by small, cortical lesions [41]. Randomized clinical trials (RCTs) are necessary to clear contrasting evidence; however, they are difficult to organize, considering difficulties in early cognitive decline assessment and the need for long-term follow-up.
It is difficult to define true prevalence of AHREs in patients carrying a CIED. Despite definitions introduced by guidelines throughout the years, criteria to identify AHRE remain heterogeneous between studies. Furthermore, the prevalence of AHREs not only depends on the chosen cut offs in terms of atrial rate and duration, but also on population features, indication for implantation of devices, arrhythmia recognition algorithms and duration of follow-up (Table 1, Ref. [6, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56]). Overall, AHREs are quite common in the CIED population, and episodes lasting more than 5 minutes are found in a proportion of patients varying between 10 and 68% [7, 42]. When considering only studies excluding patients with previous clinical AF, overall prevalence of AHRE is lower (approximately 30%) [43, 57, 58, 59].
| Author and year | Number of patients | Type of device | AHRE/Subclinical AF definition | Exclusion if clinical AF documented | Mean follow-up | Patients with AHRE/Subclinical AF |
| Caldwell et al. 2009 [44] | 162 | CRT | Any mode-switch occurrence on the device with an atrial rate |
NA | 14.1 months | 16.6% |
| Bertini et al. 2010 [45] | 393 | ICD, CRT | Any AT with an atrial rate |
No | 16 months | 21.3% |
| Petrač et al. 2012 [46] | 308 | Dual chamber PM | Any AT with an atrial rate |
Yes | 36 months | 24.6% |
| Healey et al. 2012 [ASSERT] [42] | 2580 | Dual chamber PM, CRT, ICD | Any AT with an atrial rate |
Yes (if lasting more than 5 minutes) | 30 months | 10.1% |
| Witt et al. 2015 [43] | 394 | CRT | Any AT (according to manufacturer-specific nominal settings) lasting for at least than 6 minutes. | Yes | 50 months | 20.0% |
| Martin et al. 2015 [47] | 2718 | ICD, CRT-D | Any AT with an atrial rate |
Yes (permanent) | 23 months | 21.0% |
| Kim et al. 2016 [48] | 880 | PM, ICD and CRT | Any AT with an atrial rate |
Yes | 52.2 months | 13.8% |
| Van Gelder et al. 2017 [ASSERT] [49] | 2455 | Dual chamber PM, ICD | Any AT with an atrial rate |
Yes | 30 months | 36.3% |
| Amara et al. 2017 [SETAM] [50] | 595 | PM | Any AT with an atrial rate |
Yes | 12.8 months | 25% |
| Kawakami et al. 2017 [51] | 343 | Dual chamber PM | Any AT with an atrial rate |
Yes (permanent and persistent) | 52 months | 48.1% |
| Kaplan et al. 2019 [52] | 21,768 | Dual chamber PM, ICD and CRT | AT/AF lasting for at least 6 minutes. | No | NA | 22.7% |
| Li et al. 2019 [53] | 594 | PM, ICD, CRT | Any AT with an atrial rate |
Yes | 50.4 months | 29.4% |
| Miyazawa et al. 2019 [54] | 856 | Dual chamber PM, ICD, CRT | Any AT with an atrial rate |
No | 48.2 months | 14.6% |
| Nakano et al. 2019 [55] | 348 | Dual chamber PM, ICD, CRT | Any AT with an atrial rate |
Yes | 65 months | 21.5% |
| Nishinarita et al. 2019 [56] | 104 | Dual chamber PM | Any AT with an atrial rate |
Yes | 65 months | 32.6% |
| Lu et al. 2021 [6] | 355 | PM | Any AT with an atrial rate |
Yes | 42.1 months | 45.6% |
AF, atrial fibrillation; AHREs, atrial high-rate episodes; AT, atrial tachyarrhythmia; CIED, cardiac implantable electronic device; CRT-P/D, cardiac resynchronization therapy-pacemaker/defibrillator; EGM, electrogram; ICD, implantable cardioverter defibrillator; PM, pacemaker; NA, not available.
AHREs and subclinical AF represent a dynamic entity. In the Asymptomatic Atrial
Fibrillation and Stroke Evaluation in Pacemaker Patients and the Atrial
Fibrillation Reduction Atrial Pacing Trial (ASSERT) [42], which identified AHREs
as any atrial tachyarrhythmia (atrial rate
Patients with AHREs are at higher risk of developing clinical AF. In a
metanalysis considering 2892 patients from ASSERT and Ancillary Mode Selection
Trial (MOST AHREs), AHREs were associated with a 5.7 times (95% CI 4.0–8.0,
p
Ischemic stroke, which can be the first clinical manifestation of AF, plays a detrimental contribution to its morbidity and mortality. It is estimated that nearly 30% of ischemic strokes are related to AF; this is the reason why long-term cardiac rhythm monitoring should be implemented to detect arrhythmia in patients with cryptogenetic stroke [64]. Furthermore, cardioembolic strokes are usually multifocal and involve large cerebral territories, resulting in significant neurologic sequelae [65].
AF-related stroke risk is not mitigated in asymptomatic patients. When comparing symptomatic and asymptomatic patients (12%) in the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) study, no significant difference was found in terms of mortality and major events after adjusting for baseline features [66]. Similarly, in a sub-study of the Prevention of Thromboembolic Events—European Registry (PREFER) in AF Registry there was no difference in the incidence of ischemic stroke or TIA between symptomatic and asymptomatic patients. Despite these observations, prescription of anticoagulants is lower in this particular subset of patients [9].
Considering the well-assessed link between stroke and clinical AF, which is independent from symptoms, one of the main interests regarding AHRE/subclinical AF was assessing its possible relationship with increased thromboembolic risk and the subsequent need for anticoagulation.
In the Registry of Atrial Tachycardia and Atrial Fibrillation Episodes (RATE Registry) [43], very short AHREs (defined as AHRE in which the onset and offset of the arrhythmic event were within the same electrogram (EGM), lasting between 15 and 20 seconds) did not correlate with an increase in adverse clinical events, including stroke, at a follow up of nearly 2 years. However, despite lack of uniformity in AHRE definition, numerous studies considering longer episodes have highlighted that patients with AHREs display an increased thromboembolic risk, whose entity varies across study groups [42, 45, 47, 49, 50, 51, 52, 54, 55, 63, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76].
When considering data from more than 10,000 patients’ using the Italian Clinical
Service Project, PANORAMA and TRENDS [76], AHRE burden resulted as an independent
predictor of stroke after adjusting for the CHA2DS2VASc score. After testing
different cut offs, a 1-hour AHRE burden was associated with a significant
increase in the risk of ischemic stroke (2.11; 95% CI 1.22–3.64, p =
0.008). However, absolute stroke risk in the AHRE population was low (0.39%
annual rate). A metanalysis [77] considering various cut offs in terms of AHRE
rate, duration, and burden, has shown that patients with subclinical AF lasting
more than the study-specific cut off had a 2.4-fold increase (95% CI 1.8–3.3,
p
In a recent metanalysis including more than 61,000 patients with CIEDs and insertable loop recorders (ILRs), AHREs lasting more than 30 seconds as well as day-level cumulative duration lasting more than 24 hours were associated with a significant increase in the risk of stroke and systemic embolism (HR, 4.41; 95% CI 2.32–8.39); the increase in stroke risk persisted across longer single episodes’ cut off duration (5-minutes, 6-hours and 24-hours thresholds), while no association was found between episodes shorter than 30 seconds and thromboembolic events [70]. Furthermore, both linear and non-linear meta-regression did not suggest an increase in the risk of stroke or systemic embolism considering progressively longer AHRE thresholds. Overall, stroke risk in patients with subclinical AF was lower than clinical AF, especially when considering historical cohorts [78].
In the assessment of AHRE-related thromboembolic risk, clinical information
should be taken into consideration as well. In a study by Botto et al.
[75], patients were stratified not only on the basis of subclinical AF daily
burden (
A recently published multiple cut-off diagnostic metanalysis [79] aiming at defining an optimal threshold for AHRE duration to predict thromboembolic events, identified an extremely short duration threshold when considering both single episode duration and daily burden (0.07 minutes and 1.4 minutes per day, respectively). Furthermore, it confirmed that thromboembolic events are uncommon in the CIED population (3.0%, 95% CI 2.2–4.0). Finally, studies have failed to demonstrate a clear temporal association between AHREs and thromboembolic events. Considering data from the ASSERT trial [49], which enrolled 2,580 patients with CIED and no history of AF, only 8% of patients registered AHREs in the 30 days preceding stroke or other thromboembolic events; AHRE was only present in 1 patient at the time of the event. In a sub-group analysis of TRENDS [80] considering 40 patients with CIEDs, half of patients with stroke had experienced at least an episode of AHRE before the clinical event, but nearly 45% of them did not have any episodes in the 30 days before the clinical event. Therefore, it is unsolved whether AHRE should be considered in a binary or continuous manner. Altogether, these data suggest that AHRE could be interpreted as a marker of stroke risk, rather than its direct cause, and that relationship between AHREs and thromboembolic risk could be independent from episodes’ duration. Furthermore, besides assessing the link between AHREs and thromboembolic events, it would be necessary to deal with the potential neurocognitive impact of AHREs and its long-term implications.
Interestingly, AHREs show a complex yet strict connection with atrial cardiomyopathy (ACM). ACM has been defined as “any complex of structural, architectural, contractile or electrophysiological changes affecting the atria with the potential to produce clinically relevant manifestations” [61]. ACM refers to a mixture of structural, functional, and electrical alterations in the atria which can be triggered by cardiovascular risk factors, as well as cardiac and extracardiac comorbidities (heart failure, neuromuscular disorders) [81]. In recent years, increasing interest in this clinical entity derived from evidence that alterations in atrial contractility and progressive fibrosis could result in an increase in the thromboembolic risk independently from the presence of AF [82]. Establishing the independent contribution of ACM to stroke and other embolic manifestations is complex, considering the mutual relationship between AHREs/AF and ACM. AHRE may be a signal of progressive atrial electrical derangement, however, the correlation between AHRE burden and ACM extension must be further investigated [70]. Even though it has been proven that patients with ACM have a higher thromboembolic risk independent from the presence of AHRE/AF, the Atrial Cardiopathy and Antithrombotic Drugs in Prevention After Cryptogenic Stroke (ARCADIA) trial [83] enrolling patients with a history of cryptogenic stroke, ACM (defined on the basis of ECG P wave abnormalities, echocardiographic left atrium enlargement and elevated NT-proBNP levels) and no history of AF at the time of enrolment, was prematurely interrupted due to futility for benefit of OAC therapy (apixaban 5 mg or 2.5 mg) vs aspirin in stroke recurrence (HR, 1.00 [95% CI 0.64–1.55]). Currently, ACM does not represent an indication to start anticoagulation [8].
Despite being frequently encountered in clinical practice [7], management of AHREs is still a matter of debate, especially when addressing the potential need for anticoagulation. Even though it has been established that AHREs are associated with an increase in thromboembolic risk, incidence of systemic embolism is not comparable to that of clinical AF [77]. Furthermore, a definite threshold of a single episode duration or burden to distinguish between innocent bystanders and episodes with a significant impact on stroke risk has not been established [79].
When deciding whether to start an OAC or not, clinical AF
is considered in a binary way (presence or absence of arrhythmia). Independent
from the arrhythmic pattern (paroxysmal, persistent, or permanent AF), the start
of anticoagulants (preferably direct anticoagulant oral agents, DOACs) relies on
the thromboembolic risk profile, assessed through validated risk scores (mainly
CHA2DS2VASc score), without distinguishing between symptomatic and asymptomatic
patients [7]. An annual absolute risk for stroke
Despite the absence of a clear linear relationship between AHREs/subclinical AF
and stroke risk, the approach suggested by the latest guidelines support
consideration of these events in a continuous manner. AHRE duration, both in
terms of single episodes and daily burden, coupled with the individual risk
profile defined through CHA2DS2VASc score, should be considered when deciding
whether to start anticoagulants or not. According to both ESC [8] and AHA [64]
(American Heart Association) guidelines, starting anticoagulation therapy
requires shared decision-making, taking into consideration AHRE duration, monthly
burden and ischemic risk profile. In a subanalysis of the ASSERT trial [49],
adjudicated AHREs lasting more than 24 hours correlated with a significant
increase in the risk of thromboembolic events, including ischemic stroke
(adjusted HR, 3.24 [95% CI 1.51–6.95]; p = 0.003). Based on these
observations, guidelines state that anticoagulation therapy may be considered in
patients with a high risk of stroke (CHA2DS2VASc
When the duration of AHREs is limited (
Absence of clear indications and cut offs on if and when to start
anticoagulation therapy in patients with CIEDs and detection of AHREs results in
great heterogeneity in clinical practice. Perception of thromboembolic risk
related to AHREs is variable, and prescription of OACs depends on the clinical
scenario, with high prescription rates in patients with previous stroke [85].
Furthermore, balancing between thrombotic and hemorrhagic risk is pivotal when
considering anticoagulation in fragile patients who are at increased risk of
bleeding complications [86, 87]. In a cohort study [73] including data of
patients with CIEDs from the Veterans Health Administration coupled with remote
monitoring information about daily subclinical AF burden, there was great
heterogeneity in OAC prescription after subclinical AF detection. Overall,
treatment rates were low (30%), even when considering patients with long
episodes (
Recommendations of the latest guidelines on the use of OACs in patients with AHRE have been formulated awaiting for results of two large RCTs specifically addressing this issue: the NOAH AFNET 6 [23] (Non–Vitamin K Antagonist Oral Anticoagulants in Patients with Atrial High Rate Episodes) trial and the ARTESIA [88] (Apixaban for the Reduction of Thrombo-Embolism in Patients with Device-Detected Subclinical Atrial Fibrillation) trial (Fig. 1).
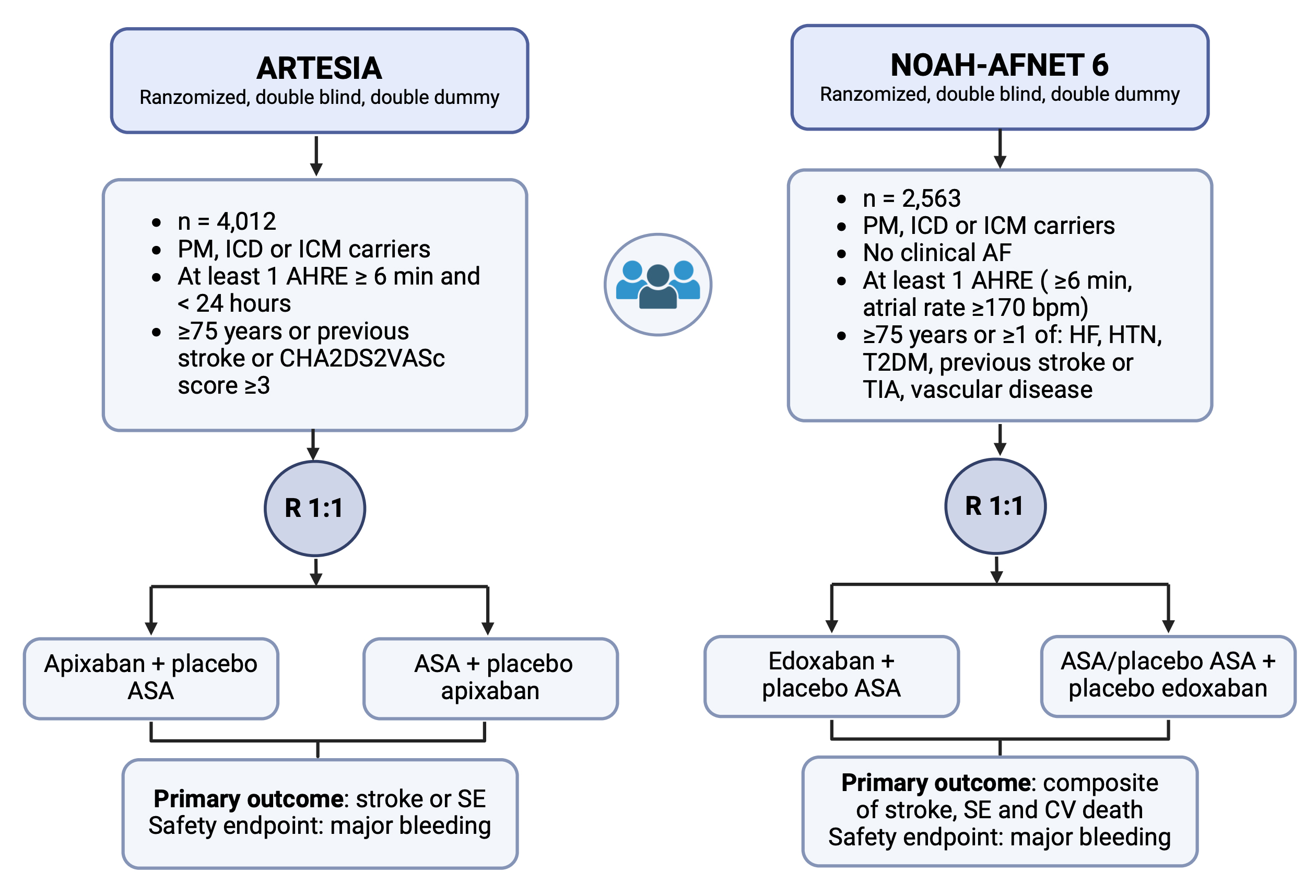 Fig. 1.
Fig. 1.
A visual comparison between RCTs: ARTESIA and NOAH AFNET 6. Details of the two RCTs assessing the potential benefit of OAC (apixaban and edoxaban, respectively) versus aspirin or placebo on stroke prevention in patients with at least one episode of AHRE lasting more than 6 minutes. AHRE, atrial high-rate episodes; ASA, aspirin; CV, cardiovascular; HF, heart failure; HTN, hypertension; ICD, implantable cardioverter defibrillator; ICM, insertable cardiac monitor; OACs, oral anticoagulants; PM, pacemaker; SE, systemic embolism; T2DM, type 2 diabetes mellitus; TIA, transient ischemic attack; AF, atrial fibrillation; RCTs, randomized clinical trials.
NOAH AFNET 6 is an event-driven, double-blind, double-dummy RCT which enrolled
2536 patients with CIEDs and ICMs, aged 65 years or older who had at least a risk
factor for stroke on top of AHREs with a duration
ARTESIA is a double-blind, double-dummy RCT which enrolled 4012 patients with
CIEDs and ICMs with episodes of subclinical AF lasting from 6 minutes to 24
hours. Any patient displaying AHREs longer than 24 hours or developing clinical
AF was excluded from analysis and started on open label OACs. Patients were
randomly assessed to receive either apixaban 5 mg twice daily (or 2.5 mg when
indicated according to guidelines) or aspirin (81 mg daily). The primary efficacy
outcome was the incidence of stroke or systemic embolism, while the primary
safety outcome was major bleeding, defined according to ISTH criteria. Compared
to a general population of patients with subclinical AF, those enrolled in the
trial were less likely to have experienced a previous stroke or TIA. Mean
CHA2DS2VASc score was 3.9
After being long awaited, results of RCTs on the use of OACs in patients with AHREs, despite appearing as contrasting, suggest a solution to complex clinical questions which have been long unanswered. Due to early interruption, NOAH-AFNET 6 failed to demonstrate a benefit in the primary outcomes for patients with AHREs receiving edoxaban. However, the inclusion as part of the primary outcome of cardiovascular death, which strongly depends on patients’ comorbidities and underlying cardiac diseases and is only partly related to stroke, together with the selection of a population with lower thromboembolic risk compared to the ARTESIA trial, could have decreased the chance of detecting the benefit of anticoagulants [90]. Conversely, in the ARTESIA trial, patients receiving apixaban experienced a reduction in the risk of stroke and systemic embolism, at the cost of an increase in bleeding events. Even though such a result in safety outcomes was predictable, it remained to be established whether patients with AHRE have a net clinical benefit from receiving DOACs. Investigators highlighted how AHRE-related strokes, despite infrequent, were associated with permanent disability and neurologic sequalae in a significant proportion of patients (nearly 50%). On the contrary, bleeding events, even though more common in patients receiving both OACs, were frequently manageable with conservative measures and transfusions, without threatening survival or requiring urgent invasive procedures.
A recently published metanalysis [91] demonstrated that results of the two RCTs are consistent, showing a significant reduction in ischemic stroke in patients on OAC (RR 0.68, 95% CI 0.5–0.92, I2 = 0%), at the cost of an increase in major bleeding. No impact on cardiovascular death and all-cause mortality was found. However, patients with long AHREs (lasting at least 24 hours) were underrepresented in RCTs, as they were excluded from ARTESIA and represent a minority in NOAH-AFNET 6.
Taking all these aspects into consideration, the two RCTs offer evidence-based information for individualized decision-making for the use of DOACs in patients with AHRE. Anticoagulants have proved to be effective in reducing the risk of stroke, which is frequently disabling and fatal also in patients with subclinical AF, at the cost of an increased number of bleeding events, which were managed conservatively in more than 90% of cases [90]. Considering that stroke is perceived as a worse outcome than death in analyses evaluating patients’ preferences and perception [92], we suggest that a very careful, individually-based decision-making process, with patients’ education on the risk-benefit ratio of anticoagulation, as well as consideration of individual preferences, is essential when deciding whether to start an anticoagulation therapy in patients with AHREs. We believe that patients with device-detected subclinical AF should be conscious that OACs can consistently reduce the risk of disabling or fatal stroke, with limited and manageable adverse bleeding events. However, the decision to start OAC therapy must not obscure the need for comorbidities and bleeding risk factor assessment and modification. Considering that a significant proportion of patients with device-detected subclinical AF progress to clinical AF, especially when single episodes last more than 24 hours [23], and that progression results in an increased risk of stroke, periodical surveillance, and re-assessment of arrhythmic burden, with closer follow-ups or remote monitoring, is essential. In summary, considering NOAH AFNET 6 trial’s underpower to detect a benefit of OACs on stroke incidence, evidence suggests that OACs can be beneficial in patients with AHREs who have additional risk factors for stroke.
As previously discussed, AHREs seem to be a marker of risk rather than a direct cause of stroke [80], and a duration threshold to predict thromboembolic events has not been identified [79]. The ARTESIA and NOAH AFNET 6 trials enrolled patients with AHREs lasting more than 6 minutes, without assessing an eventual correlation between AHREs’ duration and benefits from receiving OACs. Therefore, we are waiting for subgroup analysis to define whether a cut-off of AHRE duration or any other characterization at baseline could help identify patients who are likely to experience a greater clinical benefit from the start of anticoagulation therapy (Fig. 2).
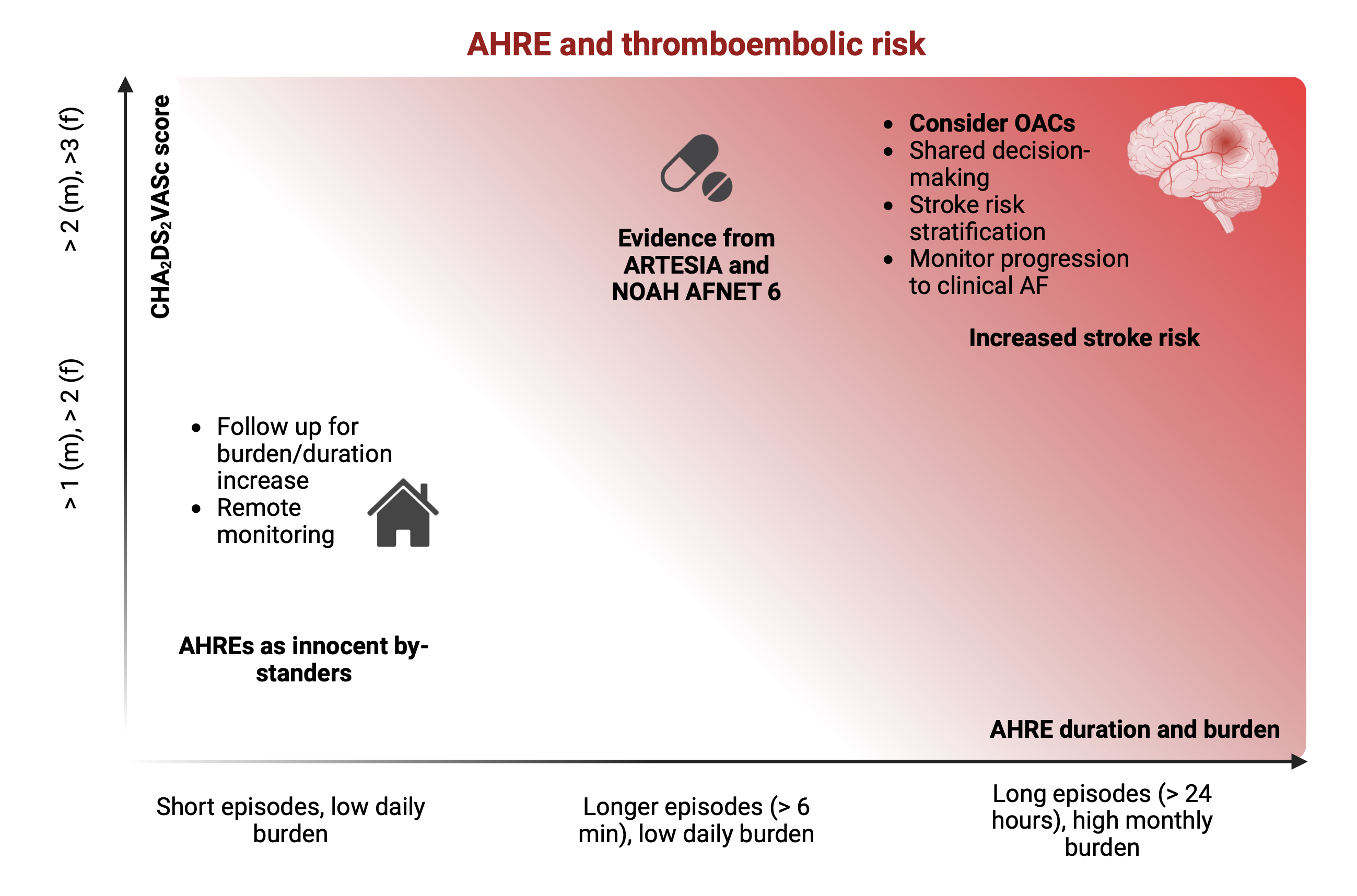 Fig. 2.
Fig. 2.
Relationship between AHRE and thromboembolic risk. Even though
a cut off duration for AHRE to predict thromboembolic events has not been
identified, studies have shown that patients with either longer single-episode
duration (especially when
Atrial fibrillation is deemed to become a major public health issue in the coming years, and its well-established relationship with cognitive decline urges an effort to unveil underlying mechanisms and limit its incidence. Despite recent evidence regarding the impact of the irregular AF rhythm on cerebral hemodynamics as a potential mechanism of silent cerebral lesions, prevention of overt stroke and systemic embolism still represents the backbone of clinical management. Notwithstanding the increase in thromboembolic risk in patients with subclinical AF, overall incidence of stroke is lower than in patients with clinical AF. Studies have failed to identify a unanimous cut off for subclinical arrhythmic events to predict thromboembolic risk, while demonstration of a temporal relationship with stroke is lacking. Overall, evidence suggests that AHREs could represent a marker of risk rather than a direct cause of stroke. Recently published randomized clinical trials exploring the effect of OACs on prevention of thromboembolic events in patients with AHREs (lasting more than 6 minutes) have shown a benefit from the use of OACs on stroke risk, despite an expected increase in major but not fatal bleeding events, which could be managed conservatively in more than 90% of cases. Albeit infrequent, AHRE-related strokes are frequently associated with permanent neurologic sequalae, suggesting a net clinical benefit could derive from use of OACs despite an increase in bleeding events. Further studies are needed to clear these aspects and to establish whether a cut-off of AHRE duration could help identify patients who may benefit from OACs.
Conceptualization MA, GB, RDP, CGB; investigation CGB, RDP, MC, GP, MV, VR, ST, AD, MZ, ML; writing—original draft preparation, CGB, MA, RDP, GP, ST; writing—review and editing MA, GB, VR, MZ, MC, AD, MV, ML; project administration, MA; supervision, GB, MA; all authors read, revised and approved the final version of the paper. All authors significantly contributed to the paper, according to the International Committee of Medical Journal Editors (ICMJE) guidelines. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The author declares no conflict of interest. Roberto De Ponti, Vincenzo Russo and Matteo Anselmino are serving as one of the Guest editors of this journal. Giuseppe Boriani is serving as Editor-in-Chief of this journal. We declare that Roberto De Ponti, Vincenzo Russo, Matteo Anselmino and Giuseppe Boriani had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Bernard Belhassen.
MA is consultant for Johnson & Johnson and Boston Scientific, clinical proctor for Medtronic, and has received educational grants from Abbott; RDP has received honoraria for lecture and scientific collaboration from Biosense Webster and Medtronic; MZ received speaker’s fees from Abbott and Boston Scientific, Biotronik; MC received speaker’s fees from Abbott and Biosense Webster; GB reported speaker’s fees of small amount from Bayer, Boston, Boehringer, Daiichi-Sankyo, Janssen, Sanofi outside the submitted work. None of the above-mentioned conflicts relates to the topic of this manuscript.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


