1 German Centre for Heart and Circulatory Research (DZHK), Partner Site Berlin, 10785 Berlin, Germany
Abstract
Ventricular pump function, which is determined by myocyte contractility, preload and afterload, and, additionally, also significantly influenced by heart rhythm, synchrony of intraventricular contraction and ventricular interdependence, explains the difficulties in establishing the contribution of myocardial contractile dysfunction to the development and progression of heart failure. Estimating myocardial contractility is one of the most difficult challenges because the most commonly used clinical measurements of cardiac performance cannot differentiate contractility changes from alterations in ventricular loading conditions. Under both physiological and pathological conditions, there is also a permanent complex interaction between myocardial contractility, ventricular anatomy and hemodynamic loading conditions. All this explains why no single parameter can alone reveal the real picture of ventricular dysfunction. Over time there has been increasing recognition that a load-independent contractility parameter cannot truly exist, because loading itself changes the myofilament force-generating capacity. Because the use of a single parameter is inadequate, it is necessary to perform multiparametric evaluations and also apply integrative approaches using parameter combinations which include details about ventricular loading conditions. This is particularly important for evaluating the highly afterload-sensitive right ventricular function. In this regard, the existence of certain reluctance particularly to the implementation of non-invasively obtainable parameter combinations in the routine clinical praxis should be reconsidered in the future. Among the non-invasive approaches used to evaluate ventricular function in connection with its current loading conditions, assessment of the relationship between ventricular contraction (e.g., myocardial displacement or deformation) and pressure overload, or the relationship between ejection volume (or ejection velocity) and pressure overload, as well as the relationship between ventricular dilation and pressure overload, were found useful for therapeutic decision-making. In the future, it will be unavoidable to take the load dependency of ventricular function much more into consideration. A solid basis for achieving this goal will be obtainable by intensifying the clinical research necessary to provide more evidence for the practical importance of this largely unsolved problem.
Keywords
- load dependency of ventricular function
- left ventricle
- right ventricle
- echocardiography
- pulmonary hypertension
- ventricular remodeling
Ventricular pump function, which provides the necessary blood supply under a wide range of circumstances to the tissues all over the body, depends in addition to the myocyte contractility, also crucially on the existing loading conditions (i.e., preload and afterload). Furthermore, myocardial pump function is also significantly influenced by heart rhythm, synchrony of intraventricular contraction and ventricular interdependence [1].
Under clinical conditions, evaluation of myocardial contractility is the most
difficult challenge because the most commonly used clinical measurements of
overall cardiac performance do not differentiate contractility changes from
alterations in loading conditions [1]. Under both physiological and
pathological conditions, there is a permanent complex interaction between
myocardial contractility, ventricular anatomy and hemodynamic loading conditions.
During the last 3 decades there has been increasing recognition that a
“load-independent index of contractility” cannot truly exist, because loading
itself changes myofilament Ca
This article gives an overview of the current knowledge of the relationship between the ventricular pump function and its loading conditions, as well as of the diagnostic tools, with a particular focus on non-invasive approaches, aiming at distinguishing secondary (overloading induced) myocardial morphological and functional alterations from primary myocardial damages. Special attention is also focused on differences between the left and right ventricle (LV and RV, respectively) sensitivity to pressure and volume overloading. Overall, the review aimed to provide an updated theoretical and practical basis for those engaged in this demanding and still current topic due to the new challenges which have arisen especially with the increasing use of temporary or durable mechanical circulatory support devices.
Heart failure (HF), a clinical syndrome with different aetiologies and
pathophysiology rather than a specific disease, arises from the disability of the
heart to pump adequate amount of blood to meet the demands of the body at rest
and during physiological effort, without abnormally high cardiac filling
pressures [3, 4, 5]. Such inability can result from a complex interplay between
intrinsic cardiac abnormalities and extracardiac factors that impair and limit
ventricular pump function. Since the early 1990s HF was therefore usually defined
as “a pathophysiological state in which an abnormality of cardiac function is
responsible for failure of the heart to pump blood at a rate commensurate with
metabolic requirements or to do so only from an elevated filling pressure”
[3, 4]. Patients who meet this definition of HF are a very heterogeneous
group with regard to the underlying pathomechanisms for the pathological
reduction of the cardiac output (CO) and/or increase of the filling pressure in
both or only in one of the two ventricles (depending on the individual
etiopathogenetic particularities of the HF syndrome). More recently, these
complex definitions, although accurate in principle, were considered less
suitable for the everyday practice because all the requirements can often not be
verified in outpatient care, and they also do not apply to all subgroups of
patients with HF [6]. Therefore, a recently proposed “universal HF definition
and classification”, which aimed to facilitate the evaluation of HF patients,
defined HF as a clinical syndrome with symptoms and/or signs caused by a
structural and/or functional cardiac abnormality corroborated by elevated
natriuretic peptide levels and/or objective evidence of pulmonary or systemic
congestion [6]. However, particularly in chronic HF accompanied by signs
and symptoms of pulmonary and/or peripheral congestion, further stratification of
patients into those with LV and/or RV systolic dysfunction and those with
predominantly diastolic dysfunction will often be necessary due to the existence
of relevant therapeutic and prognostic differences between these subsets of HF
patients [3, 4, 5, 7]. Somewhat surprisingly, in the recently proposed “universal HF
definition and classification”, right-sided HF is mentioned only very briefly in
a small chapter entitled: “Other syndromes related to heart failure”
[6]. As Bozkurt et al. [6] underline, they did not
specify left- or right-sided HF in their new definition and classification given
that in advanced HF biventricular failure is common, and right HF can also be
recognized as part of the above definition when patients present with symptoms or
signs caused by a cardiac abnormality and have elevated natriuretic peptide
levels or objective evidence of cardiogenic systemic or pulmonary congestion.
However, the revised classification of HF according to LV ejection fraction
(LVEF) which includes HF with reduced LVEF (HFrEF, defined as HF with LVEF
Optimal cardiac function is based on an ordered sequence of mechanical events orchestrated by electrical timing, which involves the interdependent work of both ventricles. Although the LV and RV differ greatly in their size, geometry, architecture, and function, the balance in their outputs must be maintained under equilibrium conditions and also be rapidly restored during or after transitions from one flow rate to another [9, 10]. Ventricular ejection is dependent on myocardial contractility, preload and afterload. Additionally, ventricular pump function is substantially influenced by heart rhythm, cardiac valve function (i.e., valve alterations and dysfunction which affect the loading conditions of the heart), synchrony of intra-ventricular and inter-ventricular contraction, ventricular interdependence and pericardial constraint [10, 11, 12, 13].
Both LV and RV failure occur most often as a consequence of myocardial injury of
various causes and/or hemodynamic overloading. In initial stages of ventricular
dysfunction, the ongoing increasing stretching of the myocardial fibers by the
increased intraventricular end-diastolic pressure (EDP) initiates an adaptive
rise in cardiomyocyte contraction force (Frank-Starling law of the heart) and a
myocardial hypertrophy. Neuro-hormonal activation (i.e., sympathetic activation)
by the low CO, aimed to maintain the blood supply to the vital organs by raising
the systemic vascular resistance (SVR) and also the renal retention of salt and
water (i.e., renin-angiotensin-aldosterone system activation), also act initially
as adaptive responses. Persistent ventricular overloading-induced excessive
myocyte stretch and hypertrophy initiate pathological remodeling processes with
progressive increase in ventricular wall stiffness and reduction in pump function
[14, 15]. Spherical ventricular dilation (i.e., most characteristic
remodeling-induced morphologic alteration) increases the systolic wall stress
which in turn reduces the efficiency of ventricular myocardial contraction [16].
With the ongoing exacerbation of its geometry alterations the ventricle must
develop progressively higher wall tension to preserve the same systolic pressure
and, therefore, the dilation itself enhances the mechanical energy expenditure of
the failing ventricle. Simultaneously, the progressive increase of the
ventricular wall tension, associated with a corresponding reduction of the CO,
exacerbates the reduction of the myocardial oxygen supply by impairing the
coronary blood flow [15, 16]. Ventricular dilation and remodeling also cause size
and geometry alterations of the atrioventricular valve ring which facilitates the
development and progression of functional ventricular-atrial regurgitation,
inducing ventricular volume overloading with additional reduction of forward
stroke volume (
The pathophysiology of diastolic heart failure (DHF) is characterized by a low CO that results typically from a ventricle with thick walls but a small cavity (increased mass/volume ratio) [17, 18]. If the LV is stiff, the slow relaxation in early diastole and the increased resistance to filling in late diastole induce an increase in diastolic pressures associated with reduction of the stroke volume (SV) [18]. The low CO manifests as fatigue, while the high EDP, which is transmitted backwards through the pulmonary veins to the pulmonary capillaries, induces dyspnea under slight physical stress or even at rest [18]. Like in systolic HF, these pathophysiological abnormalities trigger neuro-hormonal activation and increase in the pulmonary vascular resistance (PVR) leading to pressure overload-induced RV dysfunction. In earlier stages, symptoms may be unmasked by exercise because patients with DHF are unable to augment their SV by increasing their ventricular EDV via the Frank-Starling mechanism [18]. These patients often have an exaggerated response of systolic blood pressure to exercise. Mechanisms contributing to abnormal LV diastolic properties include stiff large arteries, hypertension, myocardial ischemia (particularly in patients with coronary microvascular dysfunction without functionally significant epicardial coronary stenoses), diabetes, and intrinsic myocardial changes with or without associated hypertrophy [18, 19].
Certain anatomical and functional particularities of the LV and RV must be considered when assessing their systolic and diastolic function. The most important particularities of the RV are the varying degrees of intensity, spatial direction and timing of regional myocardial contraction, the fact that its different anatomical regions (i.e., inlet, infundibulum and apex) play a different role in blood ejection, as well as its characteristic responses to hemodynamic overloading [20, 21, 22]. In addition, the diversity in myocardial mechanics (including its timing), even among the normal right ventricles, complicates the evaluation of both normal and impaired RV function. Because the thicker sub-endocardial RV layer is composed of preferentially longitudinally arranged myocytes, whereas in the thinner subepicardium (approximately 25% of wall thickness) the myofibers are arranged circumferentially, overall, the RV myocytes are mainly oriented in the longitudinal direction [23]. Due to the different orientations of myocardial fibers in the inflow and outflow part, these two essential parts of the RV are contracting perpendicular to each other: the RV inflow longitudinally and the RV outflow circumferentially [22]. As a result, whereas the normal LV myocardial shortening occurs symmetrically in the transverse and longitudinal planes, the normal RV contraction pattern is mainly characterized by longitudinal shortening [24, 25]. Despite this, chronic pressure and volume overload-induced hypertrophy of circumferential fibers can increase the relative contribution of circumferential myocardial contraction to the global RV systolic function [25, 26]. RV myocardial longitudinal shortening also correlates strongly with RV ejection fraction (RVEF), whereas transverse shortening and RVEF do not correlate [25].
Functioning predominantly as a volume pump, the compliant thin-walled RV tolerates better volume than pressure overload and is more sensitive to afterload changes than the LV [9]. As a consequence of the distinctly high afterload sensitivity of RV pump function, both reductions in ventricular ejection and maladaptive ventricular dilation occur much earlier in the pressure overloaded RV than in the pressure overloaded LV [26, 27].
RV diastolic function also differs considerably from that of the LV. The thin RV walls and the concave interventricular septum (IVS) are relatively distensible conferring the RV a higher compliance which allows greater changes in RV volume, associated with only small changes in the diastolic pressure [25]. Thus, unlike the LV, the RV may dilate significantly in response to acute pressure or volume overload even without a decrease in myocardial contractility [27].
Although both systolic and diastolic myocardial dysfunction are always involved in the pathogenesis of advanced HF due to primary impaired LV function, a particularity of LV dysfunction is that in half of cases, diastolic dysfunction has proved to be the major cause of HF [28, 29]. Before the use of the term HFpEF, this complex clinical syndrome dominated by signs and symptoms of HF despite the absence of relevant LVEF reduction was designated as diastolic HF, and accordingly, HFpEF was originally also considered as a disorder caused solely by abnormalities in LV diastolic function [15, 29]. Meanwhile, there are strong indications that HFpEF should be considered as part of a systemic syndrome involving multiple organ systems, likely triggered by inflammation and with an important contribution of ageing, genetic predisposition, lifestyle factors, and multiple comorbidities [29, 30]. Basic mechanisms affecting the myocardium in HFpEF include myocyte alterations (hypertrophy, diastolic and systolic dysfunction, energetic abnormalities), interstitial fibrosis, inflammation, increased oxidative stress, endothelial dysfunction, as well as reduced density and impaired autoregulation of the microcirculation [7]. The major cardiovascular pathophysiological processes involved in HFpEF incorporate increased systemic vascular resistance, increased conduit arterial stiffness, abnormal ventricular-arterial coupling, reduced LV long-axis systolic function, slowed early diastolic relaxation, reduced LV compliance with increased end-diastolic stiffness, reduced left atrial (LA) reservoir and contractile function, impaired RV function, and chronotropic incompetence [7]. Despite its limitations for predicting cardiac functional reserve and symptoms, the diagnosis of LV failure is still based on LVEF, although in fact, a preserved LVEF has no diagnostic role for HFpEF except to exclude HFrEF [7]. Thus, LVEF enables effective separation of HFrEF and HFpEF patients, but has limited capacity to further stratify HF patients [31]. The LVEF value can estimate global function but does not indicate LV volume or SV [7].
In HF caused initially by LV dysfunction, the rising filling pressures in the LV and LA transmitted to the pulmonary vessels increase the filling pressures in the post-capillary pulmonary circulation and thereby also the PVR. The resulting pulmonary hypertension (PH) associated with increasing RV pressure-overloading will be followed by progressive RV dysfunction.
Left-sided HF-related pulmonary hypertension (PH World Heath Organization [WHO] type 2) is the most common cause of chronic RV failure. Unlike patients with RV failure related to pulmonary arterial hypertension (PAH type 1, precapillary pulmonary hypertension without elevated left-sided heart filling pressures) who benefit particularly from selective pulmonary vasodilatation therapy, those with RV failure related to PH type 2 can often not benefit from such a therapy, which may even aggravate the congestion in the pulmonary circulation if the RV output improvement cannot be immediately balanced by the failing LV.
Given the distinctly high afterload sensitivity of the RV pump function, pressure overload is the predominant pathophysiological mechanism in RV failure and pressure loading resulting from high resistance against blood flow from the pulmonary artery to the LA as well as pulmonary valve stenosis are the major causes for right-sided HF [32]. Other causes of right-sided HF include RV ischemia and infarction, primary cardiomyopathies with mainly RV involvement (e.g., arrhythmogenic right ventricular cardiomyopathy), and cardiac lesions associated with congenital heart diseases. Fig. 1 provides an overview of the pathophysiological mechanisms involved in the development of RV failure in the presence of high resistance to blood flow in the pulmonary circulation.
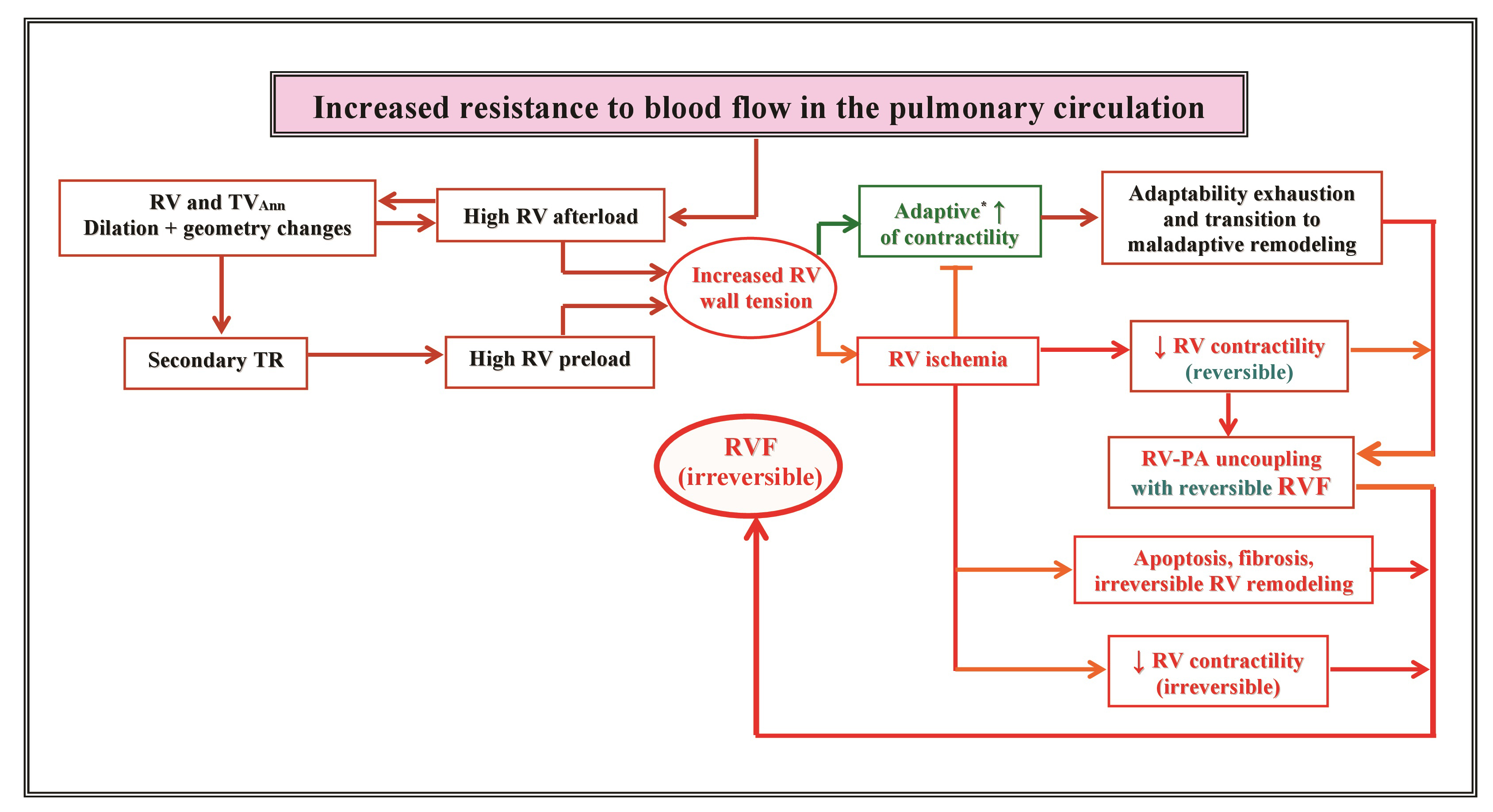 Fig. 1.
Fig. 1.Overview of pathophysiological mechanisms involved in the
development of right ventricular failure secondary to persistent increased
resistance to blood flow in the pulmonary ciculation. RV, right ventricle;
TV
It is well established that pressure and volume overload affect ventricular size, geometry and pump function to an extent that depends largely on the intensity and duration of ventricular overloading, as well as on the structural and functional particularities of the LV and RV regarding their resistance and responses (i.e., adaptive or maladaptive) to hemodynamic overloading [9, 26, 33]. Experimental studies revealed that, in pressure overload and volume overload-induced HF, the pathological myocardial remodeling differs not only structurally and functionally but is also associated with distinct proteomic alterations [33].
Pressure overload-induced LV remodeling processes and myocardial dysfunction are
a major cause for HF particularly in elderly patients with arterial hypertension
(AH) and/or severe aortic stenosis (AS) [34, 35]. For a long time, it has been
controversial whether the myocardium preserves normal systolic function in
pressure overload LV hypertrophy (LVH) [34]. This resulted in part from the fact
that most whole-heart studies incorporated endocardial measurements (e.g., LVEF)
that reflect LV chamber function, whereas most experimental studies utilized
myocardial or myofibril function [34, 36]. Experimental studies on LV myocardium
with pressure overload-induced hypertrophy revealed consistently depressed
cardiomyocyte contractility [34, 37]. By contrast, most studies involving
the entire LV indicated that the functional state of hypertrophied ventricles
evaluated by LVEF measurements remains long time unaltered before the LVEF and
other functional indexes become abnormal due to myocardial exhaustion and
decompensation [34]. However, the use of LV midwall stress-shortening data
strongly indicated that myocardial contractile function in AH-related LVH can be
depressed also in the presence of a normal EF [34, 38, 39, 40]. Comparing patients with
massive AH-related pressure overload LVH but undoubtedly normal LVEF with healthy
persons (LVEF: 69
The adaptive responses of the LV myocardium to pressure overload appear to be
both highly relevant and strikingly reversible even at advanced ages. An
evaluation of 80 patients (mean age 80
As time passes, pressure overload-induced LV hypertrophy will be accompanied by progressive diastolic dysfunction due to delayed relaxation and interstitial fibrosis-related increased LV chamber stiffness [46]. In a study of AS patients who have not undergone valve surgery, the mitral E/e’ ratio (i.e., ratio of early diastolic mitral inflow velocity to early diastolic mitral annulus velocity) was the most predictive parameter of clinical events among clinical and imaging measurements [47]. In other studies, the E/e’ ratio was an important independent predictor of early, midterm, and late mortality after AV replacement [46, 48].
LV volume overload occurs more often in response to mitral regurgitation (MR) or
aortic regurgitation (AR) where a relevant part of the ejected blood is not
delivered to the systemic circulation, but instead is either delivered back to
the LA or returned to the LV, respectively. The chronic volume
overload results in LV chamber enlargement with eccentric myocardial hypertrophy
which allows the ventricle to counteract, at least in part, the negative impact
of the regurgitation on the
LV volume overload is the major pathognomonic feature of chronic AR. The degree of volume overload is determined by the magnitude of the AR, which is related to the size of the regurgitant orifice, the aorta-ventricular pressure gradient, and the diastolic time [21]. However, the AR-related LV volume overload induced by the simultaneous increase of both the SV and the regurgitant volume (RegV) associated with concomitant progression the LV eccentric myocardial remodeling lead inevitably also to an increase in LV myocardial wall tension, and thus also to an additional pressure overloading. The progressive increase of the LV end-diastolic volume (LVEDV) can also induce dilation and geometry change of the mitral valve ring associated with MR of different degrees which aggravates LV dysfunction by aggravation of the volume overload [21].
The adaptation of the RV to pressure overload is based on its intrinsic myocardial contractility, the duration, progression rate (e.g., chronic steadily increase or acutely occurring massive increase in the resistance to blood flow in the pulmonary circulation) and severity of the pressure overloading, as well as the adaptability of the RV myocardium to sustain abnormally high wall stress [49, 50]. The initial adaptive responses to persistent pressure overload, which are mainly achieved by an increase in myocardial mass (i.e., adaptive hypertrophy) and contractility, are enabled by an upregulation of subcellular organelles (e.g., sarcolemma, sarcoplasmic reticulum, myofibrils, and mitochondria), which aim to minimize the wall stress for the RV exposed to an abnormally high workload (homeometric adaptation) [51, 52]. In this early adaptive state, RV–pulmonary artery (PA) coupling, the CO and the RVEF, as well as the exercise capacity are maintained [51]. However, myocardial hypertrophy leads to increased RV diastolic pressure, which indicates that the increased RV contractile function occurs at the cost of an alteration of its diastolic function [51]. The aggravation of pressure overloading finally exhausts the homeometric adaptation and induces a transition of the RV alterations to a heterometric adaptation where the RV dilates, and uncoupling arises because its contractility fails to match the excessively high afterload [51].
There are different stages of adaptive and maladaptive RV remodeling processes. Throughout maladaptive RV remodeling, there are further stages of reversible and irreversible RV failure (RVF). Thus, adaptive and maladaptive phenotypes are not completely different responses, but rather parts of a sequence of ventricular responses to pressure overloading [53]. Currently, the mechanisms behind the transition toward RVF are still incompletely understood.
Several mechanisms and myocardial structural changes have been associated with either adaptive or maladaptive RV phenotypes (e.g., sympathetic hyperactivity, metabolic shift from oxidative metabolism toward glycolysis, capillary rarefaction, and fibrosis). Experimental data suggest a larger contribution of interstitial fibrosis to total stiffness in end-stage RV failure, whereas cardiomyocyte stiffening may play a larger role in earlier stages of pressure overload-induced RV structural and functional alterations [54]. Prolonged activation of adaptive mechanisms, particularly in association with a progressive increase of pressure overloading, finally leads to severe RV systolic dysfunction and diastolic stiffness, followed by irreversible RVF. Given that RV adaptation to pressure overload is quite variable among patients, the progression to right-sided heart failure remains difficult to predict [53].
After full exhaustion of its adaptive capacities to overcome the excessively
high resistance in the pulmonary circulation, the RV responses to the
continuously high afterload exhibit more often a transition to pathological
myocardial hypertrophy associated with a down-regulation of subcellular adaptive
activities as well as ischemia due to reduced capillary density [52, 53, 54].
Oxidative stress, Ca
RVF caused by ventricular pressure overloading is ultimately the consequence of both systolic and diastolic RV dysfunction. Nevertheless, whereas RV systolic dysfunction was steadily found to be an independent predictor of mortality in PH, for RV diastolic dysfunction such correlation was not found in all studies [56, 57]. Given that RV diastole consists of several phases, it cannot be characterized by one single parameter and, therefore, the evaluation of RV diastolic function is much more demanding [9].
Because of its anatomical and physiological particularities, the RV tolerates
volume overload better, and for much longer periods of time, than pressure
overload [58]. Whereas in children the most common causes of RV volume overload
are congenital heart diseases, in adults, tricuspid and/or pulmonary
regurgitation in the presence of various cardiac pathologies are the major
triggers of RV chronic volume overload [59]. In the early stages of volume
overloading, the RV increases its contractile function through the Frank-Starling
law, which can effectively compensate for the altered hemodynamic conditions.
This adaptive response was also confirmed by the evidence of an increase in RV
longitudinal shortening in the presence of relevant volume overload [59]. Chronic
volume overload may ultimately lead to RV systolic dysfunction and increased
morbidity and mortality, particularly in the presence of superimposed pressure
overload and/or marked RV enlargement, which argues for corrective interventions
before significant RV dilatation ensues [58, 60]. RV volume overload
leads also to simultaneous LV dysfunction, without intrinsic alteration in
myocardial contractility, primarily due to LV underfilling secondary to septal
displacement and changes in LV geometry rather than due to a decreased
Ideally, an index of contractility should be sensitive to changes in myocardial contractile state but indifferent to loading conditions [61]. Already more than 3 decades ago it was proven that the EF, which was long considered the most useful single hemodynamic parameter for the assessment of ventricular dysfunction, is highly load dependent and, therefore, its usefulness as a measure of ventricular function is limited [61, 62]. Because the EF reflects the ventricular contractile function in relation to loading conditions, it cannot be considered as an index of contractility [9]. Accordingly, patients with identical EF values can have very different levels of myocardial contractility and ventricular dysfunction [9]. Even with unchanged contractility an increase of the afterload can relevantly reduce the EF, whereas a reduction of the afterload increases the EF [63, 64, 65, 66, 67]. LVEF can be normal in patients with LV hypertrophy associated with small cavity size, even in the presence of LV systolic dysfunction [65]. Regarding the highly load-sensitive RV, because there is no single morphological or functional cardiac parameter which can alone reveal the overall appearance of RV morphological and functional alterations, it is necessary to conduct multiparametric assessments and to use integrative approaches utilizing parameter combinations which also include data referring to the RV actual loading conditions [9, 68, 69, 70].
There are several clinical presentations of LV dysfunction where the commonly
used volumetric EF calculation: LVEF (%) = [(EDV – ESV)/EDV)]
In the presence of MR, the difference between the EDV and ESV does not reflect
anymore the
Although the LVEF reflects the LV contractile function also in the presence of
AR, it is important to take into consideration that an increase of the
aortic RegV in the detriment of the delivered blood volume into the systemic
circulation will not change the LVEF as long as the amount of blood ejected into
the aorta (i.e.,
In patients with AS, LVEF reduction does not clearly suggest impaired myocardial
contractility, if the hypertrophied left ventricle can develop supernormal
systolic pressures without relevant changes of its cavity size and geometry
[77, 78]. This can explain the frequently detected EF improvement after AV replacement [78]. Given the increasing prevalence of AS with
advancing age, as well as its more frequent association with atherosclerosis and
AH in these patients, the impact of reduced systemic arterial compliance (SAC) on
the LV afterload in patients with “degenerative” AS was more intensively
investigated during the last two decades [79, 80, 81, 82]. Meanwhile there is conclusive
evidence that in AS patients older than 60 years, particularly in those with low
flow AS associated with reduced SAC, the LV faces a double load (i.e., valvular
and arterial) and, therefore, AS cannot be viewed in all patients as an isolated
disease of the AV [79, 80, 81]. In patients with AS, reduced SAC has a major influence
on the occurrence of LV systolic and diastolic dysfunction [81]. Relevant AS and
reduced SAC showed additive effects in increasing afterload and deteriorating LV
function [80, 82]. The total SAC can be indirectly calculated with the equation:
SAC = SVi/PP, where SVi is the SV index, and PP the brachial pulse pressure
(i.e., the difference between systolic and diastolic arterial blood pressure).
Whereas SVi/PP
Because LVEF reduction is associated with poor prognosis in patients with relevant AS, a progressive EF reduction is considered as a strong indicator for necessary intervention [83]. However, in patients without additional cardiac co-morbidities, several adaptive mechanisms which act to preserve the EF can delay the fall in EF during the transition period toward LV failure. This was confirmed by the use of STE, which appeared more reliable than the conventional echocardiography for early identification of patients with AS at great risk for LV failure [84, 85].
A more recent attempt to improve the diagnostic and prognostic value of the LVEF
revealed that the EF up to the time of maximal ventricular fiber shortening named
as: “First-Phase Ejection Fraction” (EF
Due to the load dependency of all the measurements which are routinely used in daily practice for evaluation of LV systolic function it is necessary to consider this important problem in the interpretation of measured values [21, 63]. However, the evaluation of systolic function in relation to its loading conditions requires integrative approaches using several LV anatomical and functional parameters in combination with hemodynamic parameters which usually necessitate both invasively and noninvasively obtained measurements. However, such demanding integrative approaches which include also invasively obtained hemodynamic parameters are hardly applicable in the outpatients. The introduction of new completely non-invasive methods for quantifying the LV myocardial work (MW), which take into account the loading conditions during myocardial deformation and involve combining non-invasively obtained LV pressure curves with STE-derived strain measurements, has resulted in rising interest for noninvasive integrated approaches [86, 87, 88, 89, 90, 91]. The pressure curve can be obtained by associating peripheral systolic blood pressure (SBP) with Echo-derived cardiac event times including isovolumic contraction (IVC), systolic ejection, and isovolumic relaxation (IVR) [88, 90]. With software support, by integrating the STE-derived myocardial longitudinal strain measurements with the pressure curves, it is possible to generate a non-invasive LV pressure-strain loop, which enables the quantification of MW [88, 90].
MW consists of 4 distinct components: the global work index (GWI), the global
constructive work (GCW), the global wasted work (GWW), and the global work
efficiency (GWE) [87, 88, 89, 91]. Each of these components provides different
information about LV mechanics. The GWI quantifies the indexed total work
performed by the LV throughout the entire mechanical systole including both IVC
and IVR (corresponds to the myocardial energy translated into mechanical energy
between MV closure and opening) [87]. The GCW is the LV work that contributes to
LV ejection and is performed by contraction with and without myocyte shortening
(i.e., positive work during the IVC and ejection, respectively), and by active
relaxation (negative work implying energy-dependence) with and without myocyte
lengthening during the IVR and the early diastolic LV filling, respectively [87].
Thus, the GCW quantifies the energy consumed by the myocardium that contributes
effectively to the generation of the CO by facilitating LV ejection. On the other
hand, GWW represents the negative work that does not contribute to LV
ejection [87]. It includes the negative segmental work during IVC and ejection
where the myocardium undergoes lengthening as well as positive segmental work
during IVR when the myocardium undergoes shortening. It quantifies the energy
consumed by the myocardium that is wasted and does not contribute to CO. Finally,
the GWE is the ratio between constructive work and total (constructive and
wasted) work, which reflects the net percentage of performed MW which is actually
translated into CO and can be calculate with the formula: GWE = [GCW/(GCW + GWW)]
In AS patients, the estimation of LV pressure from brachial artery blood pressure measurements is limited due to the presence of AV obstruction, which creates a higher peak intraventricular pressure compared with the peripheral SBP. To address this limitation, Fortuni et al. [90] introduced a method in which they combined the mean gradient over the AV measured by echocardiography with arterial SBP, creating a new parameter for the noninvasive estimation of the LV pressure in patients with AS. GWI and GLS tend to vary over time in AS patients. Initially, as long as the AS is compensated and LV contractile function is preserved, both GWI and GCW increase, whereas absolute GLS can appear incorrectly low due to its afterload dependency [91]. Later, in decompensated stages, when LV contractile function becomes impaired, both GWI and GCW decrease, symptoms of heart failure occur irrespective of the underlying GLS [91]. Even in asymptomatic AS patients, lower GWI values appeared to be a marker of decompensation and was also found associated with increased mortality [91, 92].
In AR, which is characterized by an increase in both pre- and afterload, initially, the RegV reduces the final SV which leaves the LV and increases the LV enddiastolic volume. The elevated preload triggers LV adaptation according to the Frank-Starling law which leads to an increase in SV that tends to compensate for the volume overload. The elevated preload triggers LV adaptation according to the Frank-Starling law which leads to an increase in SV that tends to compensate for the volume overload. In the further course of the AR, sustained high LV wall tension progressively leads to increased afterload. A recent study which included patients with moderate or severe chronic AR and preserved LVEF has revealed that before surgical intervention, both GWI and GCW were elevated in relation to the severity of AR [93]. A recent study which included patients with moderate or severe chronic AR preserved LVEF has revealed that before surgical intervention, both GWI and GCW were elevated in relation to the severity of AR [93]. After the elimination of the AR, the GWI, GCW, and GWE decreased significantly, while GWW remained unchanged [93]. However, in 28% of the patients, the GWI remained abnormally high, suggesting reduced LV reverse remodeling in the presence of irreversible myocardial damage [93].
Patients with severe secondary MR revealed significantly altered GWI and GCW values [94]. However, those alterations appeared to be associated with better GWE and better preserved (i.e., less impaired) GWW values, which suggest the existence of potential benefits on myocardial energetics caused by the additional low impedance alternative for a partial emptying of the LV into the LA [94]. This could explain the finding that not only the altered GWI and GCW, but also better GWE and better preserved (i.e., less impaired) GWW values appeared independently associated with worse long-term survival in those patients [94]. The latter may also suggest that in very advanced stages of LV remodeling and dysfunction, associated with severe secondary MR, the elimination of MR (by MV repair or replacement) could also offset the potential benefits on myocardial energetics provided before that therapy by the low impedance LA leak through the incompetent MV, could trigger the development of post-interventional acute life-threatening LV decompensation. It is well known that the postoperative necessity of a ventricular assist devce (VAD) implantation or heart transplantation in patients with advanced LV failure who underwent MV surgery for severe secondary MR is not an absolute rarity [95]. Table 1 (Ref. [21, 63, 64, 65, 66, 72, 73, 74, 75, 79, 80, 81, 82, 83, 86, 87, 88, 89, 90, 91]) provides an overview of major Echo-derived combined parameters and indices for evaluation of LV myocardial responses to pressure and/or volume overloading.
| Parameters | Calculation | Particularities and clinical usefulness |
| MR-corrected LVEF [21, 63, 64, 65, 66, 72, 73, 74, 75] | LVEF (%) = [ |
- Avoids overvaluation of LV pump function in the presence of mitral regurgitation (MR) because the difference between the measured EDV and ESV is not anymore only the blood volume rejected into the aorta, but in fact becomes the sum of |
| - | ||
| EDV = end-diastolic volume | ||
| RegV = regurgitant volume | ||
| RegF = regurgitant fraction | ||
| First-phase ejection fraction (EF |
EF |
- Can predict cardiac worsening in patients with |
| V |
- Appeared helpful for optimal timing of AV replacement in asymptomatic patients with severe AV narrowing. | |
| Total systemic arterial compliance (SAC) [79, 80, 81, 82] | SAC = SVi/PP | - In older patients with AS, reduced SAC ( |
| SVi = stroke volume index | - SAC appears useful for differentiation between the relative contribution of AS and arterial stiffness on LV pressure overloading. | |
| PP = brachial pulse pressure* | ||
| “Valvulo-arterial impedance” (Z |
Z |
- Z |
| SAP = systolic arterial pressure | - Z | |
| MG |
||
| Energy loss index (ELI) [80] | ELI = [(EOA × A |
- Useful for a better classification of patients with AS, based on both the severity of AV narrowing (severe AS defined as ELI |
| EOA = the effective orifice area of the AV | - ELI, together with SAC and Z | |
| A |
||
| Global work index (GWI) [86, 87, 88, 89, 90, 91] | Calculated by software from the global longitudinal strain (GLS), blood pressure values, and PW Doppler-derived time measurements of valvular events. | - GWI quantifies the indexed total work performed by the LV throughout the entire mechanical systole including both IVC and IVR (corresponds to the myocardial energy translated into mechanical energy between MV closure and opening). |
| - This new parameter has the advantage of incorporating information on afterload, through the interpretation of strain in relation to dynamic non-invasive LV pressure. | ||
| Global constructive work (GCW) [87, 88, 89, 90, 91] | It is calculated by the same software, using some of the measurements which were also used for calculation of GWI | - GCW is the LV work that contributes to LV ejection and is performed by shortening (positive work) during IVC and systole or by lengthening (negative work) in IVR. |
| - It quantifies the energy consumed by the myocardium that effectively contributes to cardiac output by facilitating LV ejection. | ||
| Global wasted work (GWW) [87, 88, 89, 90, 91] | It is calculated also by the same software, using some of the measurements which were used for calculation of GWI | - Represents the LV work that does not contribute to ejection. |
| - It quantifies the energy consumed by the myocardium that is wasted and does not contribute to the cardiac output (CO). | ||
| Global work efficiency (GWE) [87, 88, 89, 90, 91] | GWE (%) = [GCW/(GCW + GWW)] × 100 | - GWE is the ratio between constructive work and total (constructive and wasted) work, which reflects the net percentage of the performed myocardial work, which is actually translated into CO. |
MR, mitral regurgitation; LV, left ventricle; LVEF, left ventricular ejection fraction; ESV, end-systolic volume; PW, pulsed-wave; AV, aortic valve; AS, aortic stenosis; IVC, isovolumic contraction; IVR, isovolumic relaxation; BSA, body surface area; MV, mitral valve. * PP, difference between systolic and diastolic arterial blood pressure.
The presence of a distinctively high sensitivity of ventricular size, geometry and function to pressure overloading associated with a relatively high tolerance to volume overloading are major particularities of the RV with a relevant impact on the reliability of its evaluation [9, 50, 96, 97, 98, 99]. Therefore, the evaluation of the RV and LV differ not only in terms of the preferred techniques and measured parameters but even more importantly in the interpretation of the measurements in context with their particularities and the instantaneous hemodynamic loading conditions [100, 101].
Assessment of the RV in relation to its actual loading conditions and prediction of both RV reverse remodeling and functional improvement in case of a reduction of its hemodynamic overloading is particularly helpful in decision-making before heart transplantation (HTx) and VAD implantation, as well as for optimal timing of lung transplant listing for patients with refractory end-stage precapillary PH [66, 102].
End-stage HF involves both ventricles, even if its initial cause was left-sided heart disease. Although LV assist devices (LVADs) provide better survival and quality of life than biventricular assist devices, it must also be considered that RVF associated with increased morbidity (more often renal, hepatic or multi-organ failure) and mortality can occur in about 25% of LVAD recipients, even if LVAD implantation is later followed by a complementary RV assist device (RVAD) implantation [103, 104]. Therefore, patients who require a long-term biventricular assist device, as well as those who require a temporary RVAD in addition to the LVAD should be identified already before surgery or at the latest intraoperatively [102, 105]. In the meantime, it has become obvious that an Echo can be a cornerstone for major decision-making processes prior to, as well as during, VAD implantation surgery [105, 106]. Those patients who had an unfavorable clinical course of RV function after LVAD implantation, revealed already pre-operatively significant differences in Echo-derived parameter values related to right atrial (RA) and RV size, geometry and function [106, 107, 108, 109, 110]. Nevertheless, such anatomical and functional RV alterations in LVAD candidates could not be recognized in all studies as significant risk factors for RVF during LVAD support [100, 101, 102]. The vast majority of Echo-derived variables identified as risk factors for RVF were found alone not able to predict neither RVF nor provide freedom from RVF after LVAD implantation [101, 106].
RVF induced by myocardial pressure overloading is the main cause of death in patients with severe PAH (i.e., mortality rate up to 40% in PAH patients with acute RHF) and chronic thromboembolic PH [26, 109]. With the continuous prolongation of waiting-times for lung transplantation (LTx), timely prediction of no longer reversible RVF in LTx candidates is crucial for the optimal timing of listing procedures, and therefore, the identification of significant prognostic predictors is a major goal [110, 111, 112].
Two-dimensional echocardiography (2D-Echo) is the main working tool for routine
clinical evaluation of the RV anatomy and function. However, the utility of RV
volume measurements necessary for the calculation of the EF appeared unreliable
[100, 101]. Accordingly, RVEF calculations derived from 2D-Echo measurements are
no longer recommended, neither for scientific research nor for clinical use
[25, 56]. Because 3D-Echo provides better reproducibility and higher accuracy of
RV volume measurements, it can replace 2D-Echo for these measurements [56, 64].
However, 2D-Echo remains particularly useful for the assessment of RV and RA size
and function, detection and quantification of tricuspid regurgitation (TR),
as well as for the measurement of the pressure gradient between the RV and the RA
(
Although a large number of non-invasively and invasively derived individual parameters were tested over the time for their diagnostic and prognostic value in different pathological conditions with relevant involvement of the right-sided heart, there is still controversy about the reliability of the individual parameters used in the clinical praxis. The main reason for this controversy lies in the fact that all right-sided anatomical and functional parameters are load-dependent [21, 100]. Given the particularly high load dependency of RV size, geometry and pump function, it is necessary to consider this fundamental aspect in the interpretation of all collected data related to the evaluation of the right-sided heart. Because no single parameter, regardless the method (e.g., Echo, MRI or CT) used for its measurement, can alone reveal the overall picture of RV dysfunction, it is necessary to carry out multiparametric assessments and to also apply integrative approaches using combinations of parameters which should always also include details about the RV hemodynamic loading conditions [21, 68, 113]. In the past few years, the potential usefulness of various parameters and indices of ventricular myocardium adaptability to load referring to different concepts like ratios of functional parameters and load, RV-PA coupling, or indices reflecting the ability of the RV to overcome increased PVR was also investigated [27, 49, 68]. Over the past few years, several indices of load adaptability referring to different concepts like simple ratio of function and load, RV-PA coupling, or indices assessing the ability of the RV to overcome increased PVR have been proposed [27, 49, 68]. There was a general tendency to create parameter combinations and indices from measurements obtained either by invasive or non-invasive examinations.
The best known index obtained from invasive measurements is the right heart
catheterization derived stroke work index, which is calculated by the
formula: right heart catheterization (RHC) derived RV stroke work index (RVSWi
Most of the tested integrative approaches based only on Echo-derived parameters
appeared more or less limited by the lack of more precise hemodynamic data.
Nevertheless, these approaches have the advantage of being particularly suited
for close monitoring of outpatients. Undoubtedly, the most optimal integrative
approaches would necessitate both non-invasively obtained RV measurements
(derived from different Echo techniques, CT or MRI) and invasively obtained
hemodynamic details during RHC, because most of the tested approaches based only
on Echo-derived parameters appeared limited due to the lack of more precise
hemodynamic data [15, 16, 21]. Thus, with the exception of the above mentioned
RVFWLS, the RHC-derived RVSWi was found more useful than the currently used
Echo-derived parameters for the evaluation of the RV in end-stage congestive HF
[113, 114]. The correlation between the RV stroke work index (SWI
During the last few years, several complex Echo-derived indexes suggested as possible surrogates for the RHC-derived RVSWi, as well as different Echo-derived composite variables which incorporate either RV myocardial displacement and load, or velocity of myocardial shortening and load, were tested for their usefulness for both the evaluation of RV remodeling and dysfunction, and the prediction of impending severe RV failure [27, 49, 113, 114, 115, 116, 117, 118].
A simplified composite Echo-derived index was proposed as a surrogate for the
SWI
During the last decade, certain Echo-derived composite indices which include either longitudinal movement of a RV myocardial component and load (i.e., TAPSE/systolic PAP and TAPSE/PVR) or velocity of myocardial shortening (i.e., velocity of deformation) and load (i.e., afterload-corrected peak systolic longitudinal strain rate) appeared also useful for assessment of RV contractile function [5, 8, 121, 122, 123, 124, 125, 126, 127].
The TAPSE/systolic PAP ratio is a simplified approach to assess RV contraction by plotting longitudinal myocardial shortening vs. the force generated for overcoming the imposed load [121, 122, 123, 124]. This parameter can facilitate therapeutic decision-making processes and prognostic assessments in patients with RV dysfunction, and, based on its high correlation with invasively evaluated RV systolic elastance/arterial elastance TAPSE/systolic PAP was also proposed as an index of RV-PA coupling [124]. Several studies demonstrated a high reproducibility of the necessary measurements and this index appeared able to predict mortality in patients with HF due to primary impaired LV function and also in patients with severe PAH [68, 125]. However, as mentioned above the significant predictive value of mortality in patients with HF originating from primary impaired LV function was not confirmed by all studies [119].
The RV ejection efficiency (RVEe), defined as RVEe = TAPSE/PVR, is another composite variable which was proposed as a non-invasive index of RV-PA coupling [12]. Thus, using TAPSE as a surrogate for RV ejection and the Echo-derived PVR as a surrogate for the RHC-derived PVR, the RVEe is easily calculable with the formula: PVR = TR peak velocity/RV outflow tract velocity-time integral. The calculation of the RVEe might be appropriate for the assessment of RV systolic function. However, further studies will be necessary to determine whether the Echo-derived RVEe can be indeed useful for the assessment of RV function. A limitation of this index is its decreasing reliability with the aggravation of the TR in patients with high afterload-induced severe RV dilation. It is well-known that advanced TR is a confounding factor that can affect the use of TAPSE for assessing RV function [100, 102, 126, 127].
The ratio of SV/RV end-systolic volume (SV/RVESV) was also recommended as a surrogate for the RV-PA coupling, defined as the ratio between ventricular maximal elastance and arterial elastance (i.e. Emax/Ea), which could therefore be useful in the evaluation of myocardial contractility corrected for afterload. The major limitations of SV/RVESV are the fundamentally wrong suppositions that the relationship between the RV end-systolic pressure and RVESV is linear and crosses the origin, and that, also for the RV, the end-systolic elastance (Ees) coincides with Emax [128]. Another weakness of SV/RVESV is the inaccurate measurement of RV volumes with 2D-Echo. Given that the 2D-Echo-derived RV area measurements are much more reliable than the RV volume measurements, and one study [68] has already confirmed the potential usefulness of the RV area change/RV end-systolic area ratio as a prognostic marker in patients with severe PAH, this simple approach could be more useful in a clinical setting.
The “afterload-corrected peak systolic global longitudinal strain rate”
(GLSR), based on the relationship between RV myocardial shortening-velocity and
RV load, which is calculated by multiplying the measured systolic GLSR value
with the
A different approach for the assessment of RV adaption to pathologically
elevated afterload is provided by the Echo-derived “RV load-adaptation index”
(LAI
 Fig. 2.
Fig. 2.Right ventricular adaptation to increasing afterload. PVR,
pulmonary vascular resistance; RV, right ventricle; EDV
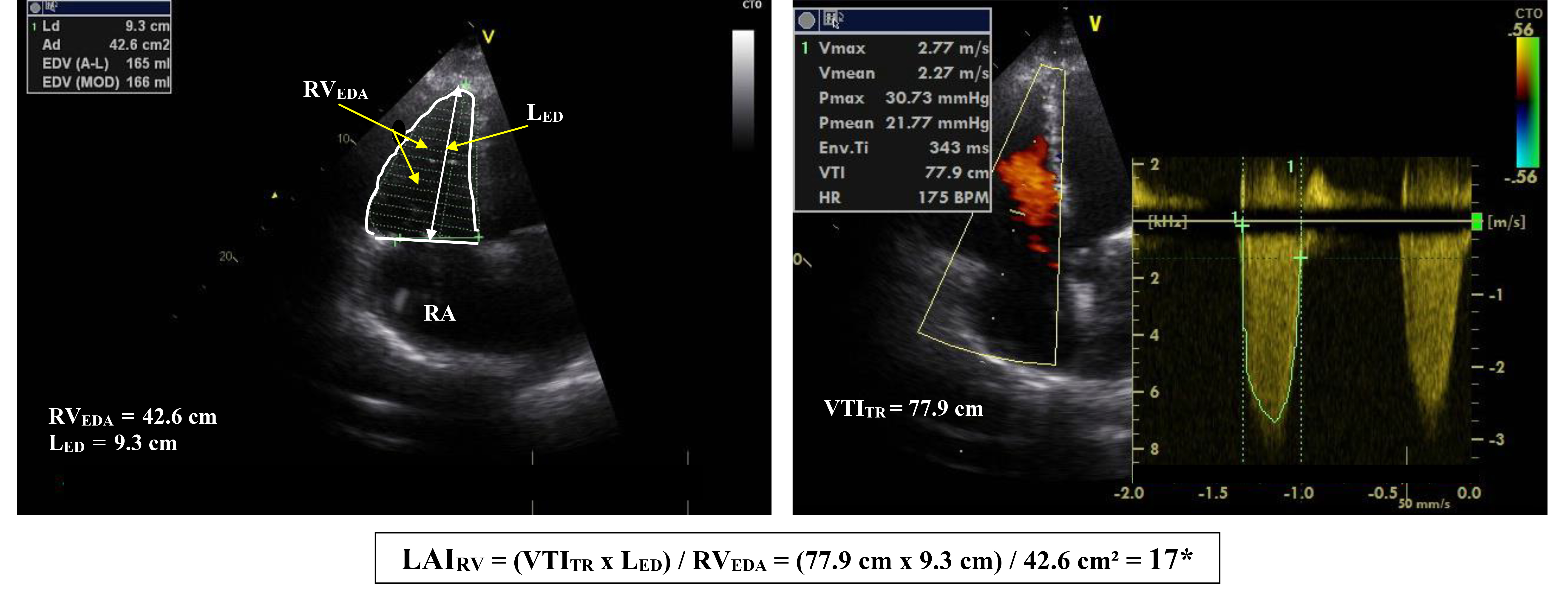 Fig. 3.
Fig. 3.Calculation of the right ventricular “load adaptation index”
from 2D-Echo-derived measurements in a patient with advanced heart failure due to
primary impaired left ventricular function. RV
Recently, the Echo-derived RV global work efficiency (RV-GWE) was found useful for predicting the risk of early RHF after LVAD implantation [130]. In this small study, the predictive value of the RV global work efficiency was superior to that of both the TAPSE/systolic PAP and the RVFW longitudinal strain/sPAP ratio.
More recently, 3D-STE data were used to assess the relationship between RV remodeling and afterload (i.e., RV end-systolic volume index and systolic PAP) [131]. Regression analysis between the systolic PAP and RV end-systolic volume index appeared to be able to distinguish adapted, adapted-remodeled and adverse-remodeled RV from one another [131].
Table 2 (Ref. [5, 8, 12, 27, 49, 68, 117, 118, 119, 120, 121, 122, 123, 124, 128, 130]) provided an overview of major Echo-derived combined parameters and indices for evaluation of RV myocardial responses to pressure and/or volume overloading.
| Parameters | Calculation | Particularities and clinical usefulness |
| RV contraction-pressure index (RVCPI) [117, 119, 120] | RVCPI = TAPSE × ΔP |
- The RVCPI revealed a close correlation with the RHC-derived SWI |
| TAPSE = tricuspid annular peak systolic excursion | - Was found to be an independent predictor of early RVF after LVAD implantation. | |
| ΔP |
- In a multivariate analysis, a low RVCPI was identified as the best predictor of outcome, whereas neither TAPSE/sPAP nor FAC | |
| RV stroke work (RVSW) [118] | RVSW = 4 × [TRj peak velocity] |
- By incorporating the SV and load, it revealed a strong correlation with the RHC-derived SWi |
| TRj = tricuspid regurgitation jet | - Direct calculation of the RV stroke volume can be challenging due to the difficulties to counteract the angle dependency of systolic flow VTI measurements along the PVA. | |
| PVA = pulmonary valve area | ||
| TAPSE/sPAP [121, 122, 123, 124] | Calculated from conventional echo-derived measurements. | - Easily obtainable useful parameter for estimation of RV performance by assessing the relationship between longitudinal displacement and load, which correlates well with the invasively evaluated RV systolic elastance/arterial elastance. |
| - Was found useful for prediction of cardiovascular mortality in patients with HF induced by LV dysfunction, as well as in patients with advanced PAH, but this usefulness could not be confirmed in all studies. | ||
| RV ejection efficiency (RVEe) [12] | RVEe = TAPSE/PVR | - A limitation is its decreasing reliability with the aggravation of the TR. |
| Echo-derived PVR = TR peak velocity/VTI |
- Is clinical usefulness is currently not established. | |
| SV/RVESV ratio [128] | Stroke volume/RV end-systolic volume | - Is considered as a surrogate for the RV-PA coupling which is defined as Emax/Ea (i.e., ratio of end-systolic ventricular elastance and arterial elastance). It facilitates the estimation of myocardial contractility corrected for afterload. |
| - Its limitations are the incorrect inherent assumptions that the relationship between the RV end-systolic pressure and RV endsystolic volume is linear and crosses the origin and that Ees coincides with Emax, as well as its as the not very reliable calculation of RV volumes from 2-dimensional echocardiography-derived parameters. | ||
| RVArea change/ESA [68] | RV area change/RV end-systolic systolic area | - It was proposed as a surrogate for RV-PA coupling. One study has already confirmed its potential usefulness for the prognostic assessment of patients with advanced PAH. |
| Afterload-corrected peak GLSR [5, 8, 27, 49] | Afterload corrected peak GLSR = GLSR × ΔP |
- Easily obtainable and reproducible combined variable reflecting the relation between RV myocardial velocity of shortening and RV loading conditions. This combined parameter was found more useful for the assessment of RV contractile function than the peak GLSR as an individual parameter. |
| - Has the advantage of also including the impact of TR on RV function. | ||
| - Revealed a high ability to predict in LVAD candidates the development or aggravation of RHF after LVAD implantation, as well as a high predictive value for imminent worsening of RV function in potential lung transplant candidates with precapillary PH. | ||
| RV load-adaptation index (LAI |
LAI |
- Simple and highly reproducible parameter which reflects the relationship between RV hemodynamic load and RV dilation which appeared preoperatively highly predictive for RV function after implantation of a LVAD. |
| - Appeared highly predictive value for imminent RV failure in lung transplant candidates with pre-capillary PH. | ||
| RV global work efficiency (GWE) [130] | GWE (%) = [GCW/(GCW + GWW)] × 100 | - Was found useful for predicting the risk of early RHF after LVAD implantation. |
| - Its predictive value was found superior to that of both the TAPSE/sPAP and the RVFW longitudinal strain/sPAP ratio. |
RV, right ventricle; RHC, right heart catheterization; SWI, stroke work index;
sPAP, systolic pulmonary arterial pressure; FAC, fractional area change; VTI,
velocity-time integral; SV, stroke volume; HF, heart failure; PAH, pulmonary
arterial hypertension; TR, tricuspid regurgitation; PA, pulmonary artery; Ees,
end-systolic elastance; PH, pulmonary hypertension; RHF, right heart failure;
RVFW, RV free wall; GCW, global constructive work; GWW, global wasted work; RA, right atrium; RVF, RV failure; ESA, end-systolic area; LVAD, left ventricular assist device; SWi
Assessment of ventricular dysfunction and prediction of its further course, selection of the most appropriate therapeutic approaches, as well as monitoring of therapy results, are crucial to the successful management of patients with HF. However, despite the important progress achieved in medical technology with a corresponding improvement of diagnostic methodologies (particularly for cardiac imaging) there are still substantial challenges, particularly those related to the load dependency of ventricular morphology and myocardial contractile function. The latter fact explains why no single parameter is able to alone reveal the real picture of ventricular dysfunction.
Given that the use of a single parameter is inadequate, it is necessary to perform multiparametric evaluations and to also apply integrative approaches using parameter combinations which include details about the ventricular loading conditions. This is particularly important for the evaluation of RV dysfunction because of its remarkably high sensitivity to supernormal afterload. In this regard, the existence of certain reluctances towards the implementation of such parameter combinations in the routine clinical praxis is difficult to understand.
Among the non-invasive attempts to evaluate ventricular function in connection with its current loading conditions, the relationship between ventricular contraction (e.g., myocardial displacement or deformation) and pressure overload, the relationship between ejection volume or ejection velocity and pressure overload, as well as the relationship between ventricular dilation and pressure overload were found useful for therapeutic decision making [21, 27, 68, 69]. However, based on the available evidence, currently it is not possible to establish a reliable hierarchy of combined parameters based on their reliability for evaluation of the LV and RV in relation to their loading conditions. Given the crucial impact of interconnected myocardial remodeling and contractility on the ability of a ventricle to overcome hemodynamic overload, it would be more beneficial to use combined parameters reflecting both the relationship between pump function and afterload, and the relationship between ventricular overloading and remodeling responses for clinical decision-making (particularly in the case of severe RV dysfunction). The latter approach would also have the advantage of diminishing the negative impact of TR on the reliability of parameters which reflect the relationship between pump function and afterload by the concurrent use of the RV load adaptation index whose reliability even increases with the progression of TR [27, 100, 102].
In the future it will be necessary to pay more attention to the load dependency of ventricular pump function and to take into consideration its impact on the evaluation of the severity and the prognostic relevance of myocardial dysfunction. Further sustained efforts to provide more evidence for its practical importance by intensifying the clinical research in this field could be a solid basis for achieving this goal.
The single author was responsible for the entire preparation of this manuscript.
Not applicable.
Not applicable.
This research received no external funding.
The author declares no conflict of interest. Michael Dandel is serving as one of the Editorial Board members and Guest editors of this journal. We declare that Michael Dandel had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Massimo Iacoviello.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



