1 Department of Physiology, Faculty of Medicine, Mashhad University of Medical Sciences, 91779 48564 Mashhad, Iran
2 Applied Biomedical Research Center, Mashhad University of Medical Sciences, 91779 48564 Mashhad, Iran
3 Department of Pediatrics, Mashhad University of Medical Sciences, 91779 48564 Mashhad, Iran
Abstract
Background: Exercise training could be essential in preventing
pathological cardiac remodeling in diabetes. Therefore, the effects of
moderate-intensity continuous training (MICT) and high-intensity interval
training (HIIT) singly or plus metformin on diabetes-induced cardiomyopathy were
investigated in this study. Methods: Forty-nine Wistar rats (male) were
recruited. Seven groups of animals were treated for six weeks as control,
diabetes, MICT (15 m/min, 40 min/day), HIIT (20 m/min, 40 min/day), metformin
(300 mg/kg), HIIT+metformin (Met-HIIT), and MICT+metformin (Met-MICT). The
metformin was orally administered with an intragastrical needle, and the
exercised rats were trained (5 days/week) with a motorized treadmill. Metabolic
parameters, echocardiographic indices, histopathology evaluation, and assessment
of gene expression connected with cardiac fibrosis, hypertrophy, mitochondrial
performance, and intracellular calcium homeostasis were investigated.
Results: Our results demonstrated that all the interventions prevented
weight loss and enhanced heart weight/body weight ratio and fasting plasma
glucose in diabetic rats. Both types of exercise and their metformin combinations
improved diabetic animals’ echocardiography indices by enhancing heart rate,
fractional shortening (FS), ejection fraction (EF) and reducing end-systolic and
end-diastolic diameter of left ventricular (LVESD and LVEDD). Gene expression of
atrial natriuretic peptide (ANP), brain natriuretic
peptide (BNP), transforming growth factor
(TGF)-
Keywords
- diabetes
- cardiomyopathy
- exercise
- HIIT
- MICT
- hypertrophy
- fibrosis
One of the most fatal complications of diabetes is diabetic cardiomyopathy (DCM)
[1]. DCM is defined as cardiac muscle dysfunction caused by diabetes, independent
of hypertension and atherosclerosis [2]. It is estimated that 40 to 50 percent of
diabetic patients suffer from heart disorders [3]. Although the exact induced
mechanisms of DCM are unknown, the inflammation process, apoptosis, hypertrophy,
and fibrosis contribute to the development of cardiac remodeling in diabetes [2].
Oxidative stress is caused by chronic hyperglycemia (the main complication of
diabetes) and can lead to ventricular contractile dysfunction [4]. The elevation
of reactive oxygen species (ROS) might activate the
renin-angiotensin-aldosterone-system and signaling pathways like transforming
growth factor beta (TGF)-
Physical activity as a thrifty and non-pharmacological co-treatment is recommended as part of the rehabilitation for diabetic patients with cardiac disorders. Exercise can improve cardiac function by decreasing cardiac risk factors and myocardial damage in diabetes [11]. Furthermore, exercise could enhance mitochondrial biogenesis, ATP production, and cardiomyocyte contractility [12]. Physical activity has been shown to promote angiogenesis and vascular performance by attenuating oxidative stress and inflammatory processes [13]. The impact of exercise on diabetes-induced cardiovascular disorder might be related to the modalities, duration, and intensity level of training programs. The evidence showed that low and moderate-intensity exercise had been indicated to improve the metabolism of glucose and cellular apoptosis in the heart, which results in enhanced cardiac function [11].
Moreover, a cardiotonic and protective role for high-intensity exercise training
has been demonstrated [11]. Both moderate-intensity continuous training (MICT)
and high-intensity interval training (HIIT) modalities of exercise have been
shown to decrease the glycosylated form of hemoglobin and fasting blood glucose
(FBS) in patients with diabetes [13, 14]. However, HIIT training showed better
outcomes in reducing FBS and ameliorating hyposensitivity to insulin in adults
with diabetes [15, 16]. Furthermore, HIIT attenuated glucose and fatty acid
metabolism, the respiratory capacity of mitochondria, and ventricular
mechano-energetic coupling in cardiac tissue. At the same time, the MICT exercise
could not improve this parameter [17, 18]. Exercise training in diabetes improves
myocardial fibrosis by decreasing myocardial collagens and restoring
fibrosis-related gene expression of matrix metalloproteinases [6]. Significant
protective effects of exercise training in DCM have been revealed by improving
PGC-1
Male ten-week-old Wistar rats (weight of 250
Rats (n = 49) were randomly split up into seven groups (n = 7) (Table 1).
Animals recruited to a single intact control group (control; injected sterile
saline, 1 mL/kg, i.p.) and six diabetic animal groups as follows: diabetic
control group (Diabetes), diabetes treated with metformin (300 mg/kg)
(Metformin), diabetes trained with HIIT exercise (HIIT), diabetes trained with
MICT exercise (MICT), diabetes treated with metformin and trained with HIIT
exercise (Met-HIIT), diabetes treated with metformin and trained with MICT
exercise (Met-MICT). Treatment protocols were started after confirmation of
diabetes induction (FBS
| Groups (number of rats = 7) | Treatment protocols |
| Group I (Control) | —– |
| Group II (Diabetes) | —– |
| Group III (Metformin) | Metformin (300 mg/kg) |
| Group IV (HIIT) | HIIT (High-intensity interval training) |
| Group V (MICT) | MICT (Moderate-intensity continuous training) |
| Group VI (Met-HIIT) | HIIT exercise + Metformin (300 mg/kg) |
| Group VII (Met-MICT) | MICT exercise + Metformin (300 mg/kg) |
Diabetes + metformin (Metformin), diabetes + HIIT exercise training (HIIT), diabetes + MICT exercise training (MICT), diabetes + metformin + HIIT exercise training (Met-HIIT), diabetes + metformin + MICT exercise training (Met-MICT). HIIT, high-intensity interval training; MICT, moderate-intensity continuous training.
Echocardiography was performed to survey heart function using a neonatal echocardiographic device (12-MHz linear probe). At first, the rats underwent light anesthesia with the intraperitoneal injection of low dose xylazine + ketamine/(2 mg/kg and 10 mg/kg respectively, i.p.). The left ventricular end-systolic diameter (LVESD), left ventricular end-diastolic diameter (LVEDD), and heart rate (HR) indices of the left ventricle in the animals were measured. Also, according to the previous study, fractional shortening (FS) and ejection fraction (EF) indices were obtained through the standard formulas [24].
After preparing and processing cardiac tissue, the Masson trichrome staining was applied to identify fibrosis. A double-blinded researcher assessed the images with a light microscope (Nikon Eclipse E200, Tokyo, Japan). Two examiners, blinded to the animal groups, analyzed ten randomly selected fields on each slide for seven rats per group; the blue color stain identified collagen fibers. Cardiac fibrosis was determined according to data from ten randomly selected high-power fields (400X) for each tissue section. The image J software (Version: 1.53f51, NIH, Bethesda, MD, USA) was chosen to evaluate collagen percentage [21, 24].
To perform RNA extraction, the cardiac tissue was homogenized with Trizol (Yekta
Tajhiz Azma Co, Tehran, Iran). The quality and purity of the harvested RNA were
detected using a nanodrop 2000 (Thermo Scientific, Waltham, MA, USA). The cDNA
was synthesized with the easy cDNA kit (Parstous, Mashhad, Iran) using a
BioRad C1000 thermal cycler (Bio-Rad, Hercules, CA, USA). The quantitative
real-time polymerase chain reaction (qRT-PCR) was conducted using the Light
Cycler System (Roche Diagnostics, Mannheim, Germany) and Ampliqon Real Q Plus 2x
Master Mix Green (Ampliqon, Odense, Denmark) to investigate the gene expressions.
| Gene | Primer sequence 5 |
| Beta-Actin | F-CCCGCGAGTACAACCTTCT |
| R-CCATCACACCCTGGTGCCTA | |
| Collagen | F-TGCCGTGACCTCAAGATGTG |
| R-TCTGACCTGTCTCCATGTTGC | |
| TGF- |
F-GCTACCATGCCAACTTCTGTCT |
| R-CCTACCACCCCAGCCTCTG | |
| ANP | F-CTCCATCACCAAGGGCTTCTTC |
| R-ATCTGTGTTGGACACCGCACTG | |
| BNP | F-CCAGAACAATCCACGATGCAG |
| R-TTGTAGGGCCTTGGTCCTTTG | |
| PGC1- |
F-CGCAGGTCGAATGAAACTGAC |
| R-GTGGAAGCAGGGTCAAAATCG | |
| AMPK | F-CCCTTGAAGCGAGCAACTATC |
| R-AGCATCATAGGAGGGGTCTTC | |
| SERCA | F-ACGAGACGCTCAAGTTTGTGG |
| R-GCTAACAACGCACATGCAC | |
| RyR | F-CGAATCAGTGAACGCCAAGG |
| R-CCTGCTCGGTCAGCTCTAAG |
TGF-
Acquired data are represented as mean
The data showed that the body weight (BW) was significantly decreased in
diabetic animals. In contrast, the ratio of heart weight to body weight (HW/BW)
and FBS increased dramatically in the diabetic group compared to the control rats
(p
| Groups | BW (g) | HW/BW (mg/g) | FBS (mg/dL) |
| Control | 320 |
3.06 |
104 |
| Diabetes | 230 |
5.24 |
512 |
| HIIT | 255 |
3.73 |
236 |
| MICT | 245 |
4.07 |
352 |
| Met-HIIT | 234 |
4.07 |
166 |
| Met-MICT | 270 |
3.51 |
197 |
| Metformin | 259 |
3.63 |
190 |
Data are represented as mean
HR in the diabetes group (351
| Group | HR (bpm) | LVEDD (mm) | LVESD (mm) | EF (%) | FS (%) |
| Control | 474 |
5.16 |
4.05 |
81.2 |
46.6 |
| Diabetes | 351 |
6.33 |
6.07 |
52.2 |
33.5 |
| HIIT | 417 |
5.96 |
5.51 |
85.3 |
57.9 |
| MICT | 393 |
5.99 |
5.76 |
65.8 |
39.9 |
| Met-HIIT | 404 |
5.28 |
4.89 |
73.2 |
44.1 |
| Met-MICT | 403 |
5.35 |
5.12 |
68.6 |
41.1 |
| Metformin | 394 |
5.47 |
5.27 |
71.8 |
41.5 |
Data are represented as mean
The ventricular muscle had typical structures in the control animals. The heart
tissue of the diabetic rats had prominent fibrosis without regular patterns that
it demonstrated with interstitial collagen structure. The data showed that in the
diabetic group, there was a significant collagen deposition compared to control
rats in the left ventricular tissue (p
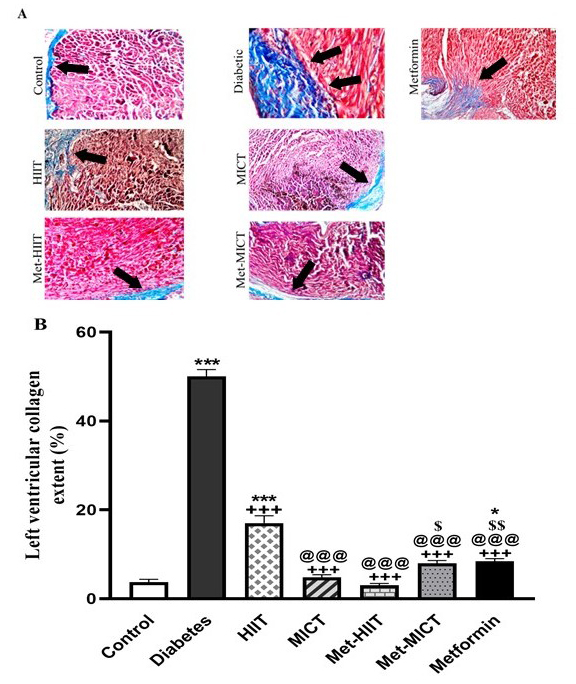 Fig. 1.
Fig. 1. Comparison of the cardiac pathological changes
in different groups of study. (A) Representative images of cardiac tissue
sections stained with the Masson trichrome. The arrow indicates myocardial
fibrosis stained in blue. (B) Quantitative analysis of fibrosis area. The data
are expressed as mean
The expression of ANP and BNP was upregulated in the diabetes
group compared to the control group (p
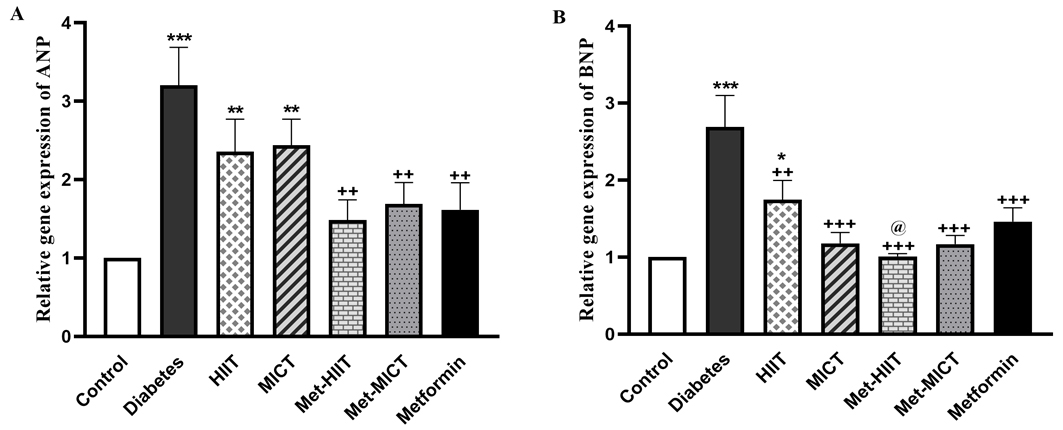 Fig. 2.
Fig. 2. Comparison of the ANP (A) and BNP (B) gene
expression in different groups of study. The data are shown as mean
The relative TGF-
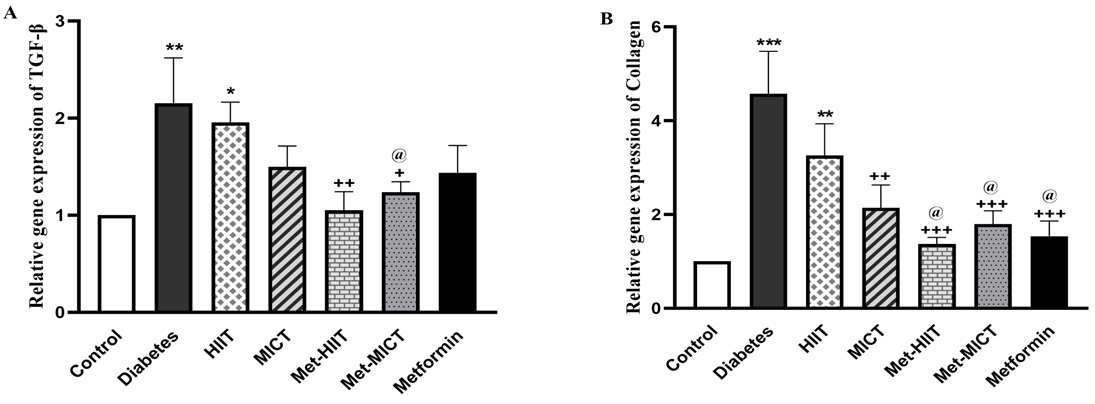 Fig. 3.
Fig. 3.Comparison of the gene expression of
TGF-
The expression of PGC-1
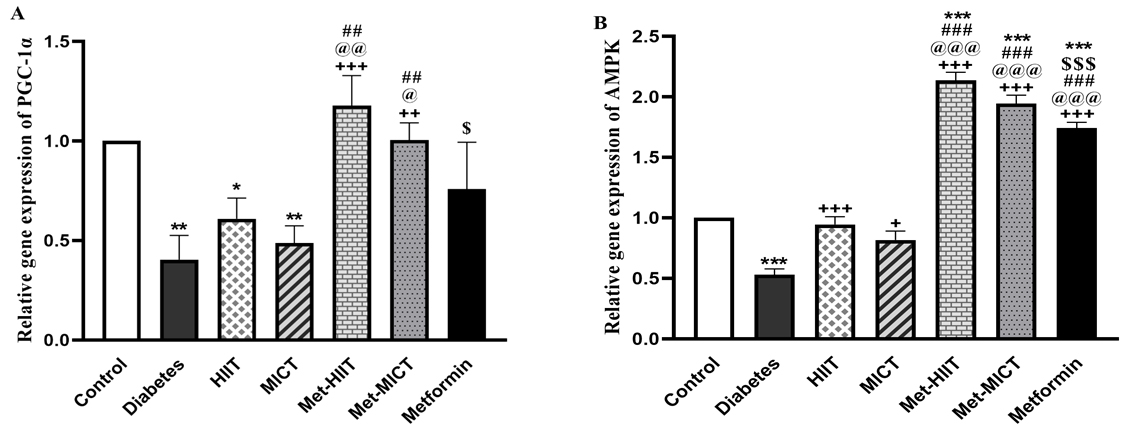 Fig. 4.
Fig. 4.Comparison of the gene expression of PGC-1
The gene expression of RyR was not significantly changed in the
diabetes vs control group. Also, the upregulation of the RyR gene
expression in all the treatment groups was insignificant compared to the diabetes
group. However, it is worth noting that the Met-HIIT group had the most increase
in the RyR gene expression compared to the diabetic ones. The expression
of SERCA was reduced in the diabetes group compared to the control group
(p
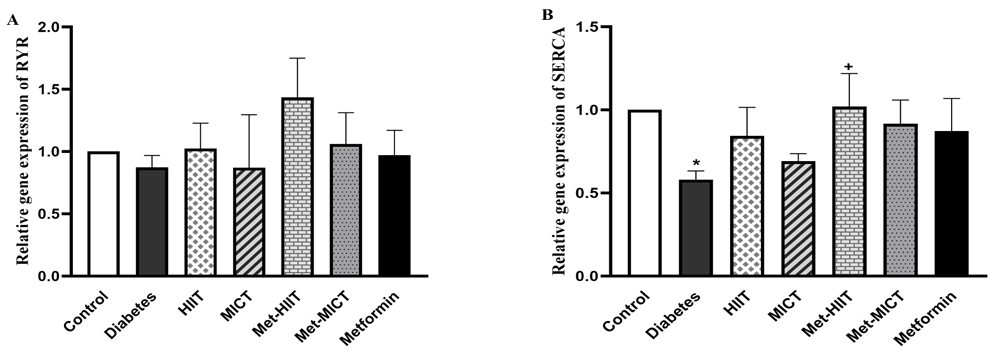 Fig. 5.
Fig. 5.Comparison of the gene expression of RyR (A) and
SERCA (B) in different groups of study. The data are expressed as mean
Fig. 6 shows a significant relationship between the expression of the
ANP gene as a hypertrophic marker and the TGF-
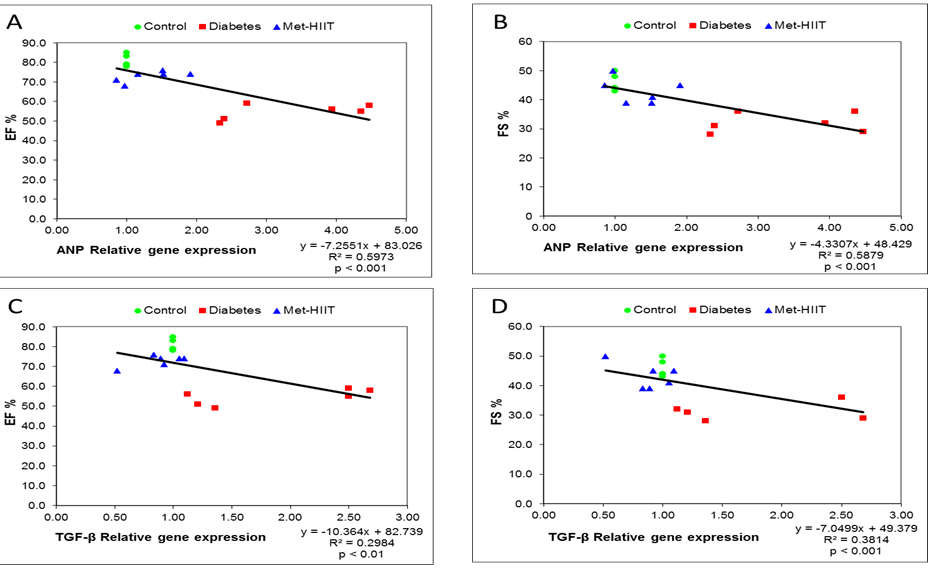 Fig. 6.
Fig. 6.Scatterplots of ANP (A,B) and
TGF-
Although cardiomyopathy caused by diabetes does not have a precise and proven
mechanism, pathological cardiac remodeling caused by inflammatory processes,
fibrosis, and hypertrophy could be a principal cause of the development of this
disorder [2]. The pathological cardiac remodeling due to fibrosis is induced by
extracellular matrix accumulation and upregulation of TGF-
Nevertheless, we studied the impact of two exercise modalities in preventing cardiac complications following diabetes. Metformin, a pharmacological diabetic treatment with a cardioprotective effect, was considered a positive control [24]. We observed that exercise with or without metformin and improving hyperglycemia inhibited fibrosis and hypertrophy by modifying the expression of target genes contributing to this process. Echocardiographic and histological data also confirmed this alleviation. Likewise, the improvement in the expression of genes that contributed to mitochondrial function and homeostasis of intracellular calcium after exercise treatment was in favor of the positive effect of exercise in inhibiting pathological cardiac remodeling in the course of diabetes.
As in previous similar experimental and clinical investigations, FBS levels in diabetic rats were significantly reduced following exercise training, although this reduction was more pronounced in the Met-HIIT group [23, 26, 27, 28, 29]. However, another study indicated the beneficial effect of MICT in lowering blood sugar in diabetic rats [13]. Exercise training might restore hyperglycemia by enhancing the muscle blood flow and ameliorating mitochondrial function. Additionally, its mechanism may be related to enhancing insulin sensitivity, which might improve glucose uptake [22, 29, 30].
Cardiac fibrosis might disturb myocardial adjustment and cardiac contraction,
which leads to cardiomyopathy development and heart failure [6]. Here, we
revealed that the exercise alleviated the expression of TGF-
Pathological hypertrophy following cardiac fibrosis is associated with the over-expression of fetal hypertrophic genes that enhance the lengths or widths of cardiomyocytes and develop DCM [3]. Improvement of cardiac hypertrophy by reversing ANP and BNP gene expression has been indicated after exercise conditioning in diabetic animals [33]. In this regard, we observed that in the Met-HIIT group, the expression of these genes decreased more and became closer to the control group. The changes in the echocardiographic data mirrored this. Lowered expression of the hypertrophy hallmark genes, in addition to restoring the echocardiographic indices of LVEDD and LVESD in exercise groups, indicate that exercise training might prevent DCM by improving cardiac hypertrophy in the course of diabetes.
Nevertheless, exercise conditioning has been demonstrated to fail to boost the cardiac hypertrophy of diabetic db/db mice in a study, but metformin administration reversed cardiac hypertrophy. Consequently, cardiac hypertrophy in the early stages of cardiomyopathy is more related to glucose metabolism abnormality [34]. It is worth noting that in this work, exercise training prevented weight loss and decreased HW/BW index in diabetic animals. It has been shown that exercise might improve glucose metabolism and prevent diabetic ketoacidosis and weight loss by increasing the skeletal muscle and liver response to insulin and reducing fat oxidation [16, 29]. The effectiveness of exercise training may improve cardiac performance by attenuating fibrosis and hypertrophy in the heart, which results in the prohibition of cardiac disturbance induced by diabetes. It also can be a reasonable justification for these changes, as mentioned by previous studies [6, 13, 16].
Mitochondrial dysfunction in myocardial cells is another complication caused by
diabetes. The level of PGC-1
Modulating RyR and SERCA mRNA expression and their protein levels following endurance exercise protocols, which were reduced in diabetic rats, may also improve systolic and diastolic dysfunction in diabetic cardiomyopathy [10, 37]. Our data did not show a significant change in the expression levels of the RyR gene in all of the treated groups; meanwhile, in the Met-HIIT treated animals, there was an increase in the expression level of the SERCA gene. This upregulation could be attributed to the impact of exercise conditioning in enhancing cardiac performance in terms of echocardiographic parameters EF and FS.
Moreover, we showed a significant correlation between the gene expression level
of ANP as an index of hypertrophy in cardiac tissue and
TGF-
It should be mentioned that our findings cannot suggest which type of exercise is more effective for diabetes management. According to the effectiveness of exercise type, some parameters, including EF and FS in the echocardiography and the expression of the AMPK gene, were improved more in the HIIT-exercised rats than the MICT one, at the same time, reduced the percentage of collagen content in the MICT-exercised animals than the HIIT-exercised group. Controversial findings were also mentioned in previous studies [29, 38].
Considering that the progression of fibrosis and hypertrophy are the most critical mechanisms in the development of cardiomyopathy and diastolic dysfunction in diabetic patients, it seems that recommending physical exercises in addition to common diabetes treatments can prevent or delay pathological remodeling of the heart.
As a limitation, the induction of diabetes in animals could not mimic all features of cardiomyopathy in diabetic patients. In addition, maximal oxygen consumption (VO2 max) was not determined in this study, and a performance test evaluated exercise intensity. Although in the present experiment, HIIT exercise resulted in a better outcome for ameliorating cardiomyopathy, however for diabetic patients, the exercise intensity and duration should be determined individually based on pathological condition.
Furthermore, some clinical echocardiographic indices could not be evaluated in the rodents. Since our results were obtained from experimental animal studies, which differ from those of humans in some aspects, more evidence is needed to apply such findings in clinics. Additionally, complementary molecular investigations such as western blot and immunohistochemistry could be considered in future studies for better elucidation of DCM signaling pathways.
The data showed that exercise training, especially in combination with metformin, in addition to improving hyperglycemia, prevents cardiomyopathy in diabetic rats by attenuating cardiac hypertrophy and fibrosis and maintaining mitochondrial function and intracellular calcium homeostasis (Fig. 7). These effects were observed more prominently in the Met-HIIT group, related to the type of exercise training protocols. Our findings increase our understanding of the benefits of physical activity on diabetes-induced cardiovascular disease and provide a practical target for DCM prevention. These results showed that exercise, especially with an anti-diabetic drug such as metformin, can be included in the rehabilitation therapy of diabetic patients with cardiovascular complications.
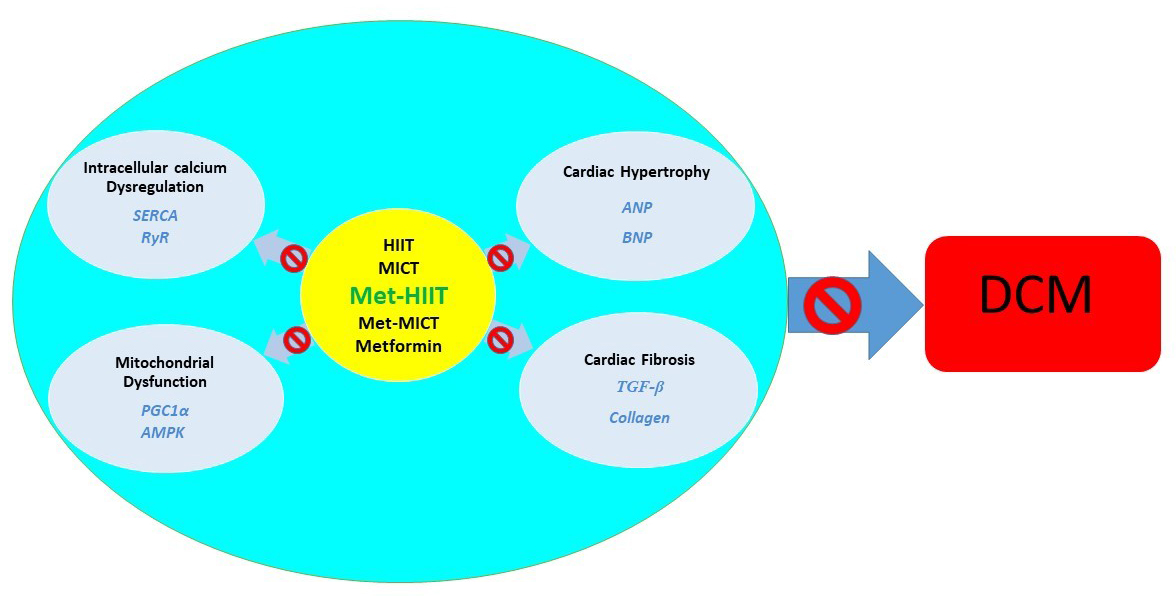 Fig. 7.
Fig. 7.The impact of exercise
training on diabetic cardiomyopathy mechanisms. TGF-
Data will be made available on request, and the corresponding author can be contacted if needed.
SS and MF performed the experiments, and MM designed the study, contributed to data acquisition, analysed and interpreted the data, revised the manuscript and supervised the project advancement. ZME, ZG, and SN assisted in data collecting and interpreting. SS, MF and ZME made the manuscript draft, and MM, ZG and SN critically reviewed it. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Mashhad University of Medical Sciences Ethics Committee approved the experiments’ protocols. The experimental process was promoted according to the caring standard for laboratory animals (Approval No. IR.MUMS.MEDICAL.REC.1399.224).
The authors would like to express their acknowledgment to the Mashhad University of Medical Sciences Research Affairs for their support. This manuscript is extracted from Sadegh Shabab’s Ph.D. thesis in Medical Physiology. Dr. Narges Kasraie made edits and revisions.
Mashhad University of Medical Sciences Research Affairs; Grant No. 981872.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







