1 Department of Traditional Chinese Medicine, Jinan Maternity and Child Care Hospital Affiliated to Shandong First Medical University, 250000 Jinan, Shandong, China
2 Department of Gerontology, The First Affiliated Hospital of Shandong First Medical University (Shandong Provincial Qianfoshan Hospital), 250014 Jinan, Shandong, China
3 Department of Traditional Chinese Medicine, Qilu Hospital of Shandong University, 250012 Jinan, Shandong, China
Abstract
Diabetic cardiomyopathy is an insidious and fatal disease, imposing major
financial and social burdens on affected individuals. Among the various methods
proposed for the treatment of diabetic cardiomyopathy (DCM), treatments with
natural products have achieved promising results due to their high efficiency and
minimal side-effects. Literature was searched, analyzed, and collected using
databases, including PubMed, Web of Science, Excerpt Medica, Science Direct, and
Springer. In this study, we reviewed the DCM-related studies on 72 representative
natural products. These natural products have been confirmed to be applicable in
the therapeutic intervention of DCM, acting through various mechanisms such as
the amelioration of metabolic abnormalities, protecting the mitochondrial
structure and function, anti-oxidant stress, anti-inflammatory, anti-fibrosis,
regulation of Ca
Graphical Abstract
Keywords
- diabetic cardiomyopathy (DCM)
- natural products
- pharmacology
- mechanism
- efficacy
- review
Diabetic cardiomyopathy (DCM) is one of the most prevalent cardiovascular complications of diabetes mellitus (DM), which arises from the effects of type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) on the myocardium [1]. This disease process was first described by Rubler et al. in 1972 [2]. DCM is a specific cardiac manifestation in patients with diabetes, as a secondary effect of metabolic damage. It is characterized by gradual heart failure (HF) and detrimental cardiac remodeling (such as fibrosis, and diastolic and systolic dysfunction) [3]. The onset of DCM is insidious and often asymptomatic in the early stages of the disease. There is no efficient and specific methodology for DCM diagnosis at present, and one factor is the absence of symptoms [4]. Despite the presence of initial symptoms of DCM, such as mild left ventricular (LV) stiffness, slight decline in compliance, and diastolic dysfunction, patients frequently overlook them [5]. The myocardial interstitial fibrosis appears to be the initial detectable stage of DCM, and it is currently diagnosed mostly using cardiac magnetic resonance [6]. Once DCM is diagnosed, it is typically classified into two stages: in the early stages, left ventricular hypertrophy (LVH) and impaired diastolic function are present, while in the late stages, myocardial fibrosis, systolic dysfunction, and overt HF are present [7]. Changes in cardiac function in early stage DCM are reversible, especially when LVH has not yet occurred [6]. However, once systolic insufficiency occurs in patients with DCM, the prognosis becomes significantly worse. In late stage, changes of the metabolism with abnormal neurohumoral activation, and development of myocardial fibrosis could promote the coronary micro-circulation, then leading to diastolic function and systolic dysfunction in DCM [8].
The pathogenesis of DCM is likely to be complex and multi-factorial, has not yet
been completely elucidated. Hyperglycemia (HG), insulin resistance, high free
fatty acids (FFA), mitochondrial dysfunction, oxidative stress, myocardial
inflammation, endothelial dysfunction, and calcium homeostasis are the basis of
the pathogenesis of DCM, and these factors (independently or jointly) affect the
occurrence and development of DCM. The heart is a primary target organ of the DM
pathology, as HG is linked to an increased risk of diabetic cardiac events. Blood
sugar control is the most fundamental measure in the treatment of DCM. The
insulin deficiency and/or insulin resistance is the starting point of the series
of reactions leading to impaired cardiac function in DCM, which is consistent
with the pathogenesis of most diabetic complications. The healthy heart is
metabolically flexible and can draw energy from diverse substrates. Fatty acid
(FA) oxidation serves as the primary source of energy for the normal adult heart,
accounting for approximately 60% [9]. HG and insulin deficiency and/or insulin
resistance can lead to loss of metabolic flexibility in the heart. Cardiomyocytes
are subject to a metabolic shift caused by HG and insulin deficiency and/or
insulin resistance, which results in higher FA intake and
Myocardial inflammation is a heterogeneous process, partially contributes to
structural and metabolic changes in the DM heart [13]. The chronic inflammatory
response will appear in myocardial tissue, throughout the whole process of DCM.
Endothelial dysfunction (ED) is involved in the pathological process of DCM, by
promoting impaired myocardial metabolism, intracellular Ca
DCM’s pathogenesis and clinical features have been well-studied in the past decade, but there are still few effective approaches for prevention and treatment [16]. At present, there is no effective drug for the treatment of DCM in clinical practice, so studying the research and development of effective therapeutic drugs is very necessary. Natural products continue to be a promising source of scaffolds with a wide range of structural diversity and bioactivity, that have the potential to be developed directly or used as starting points for optimizing novel drugs [17]. In recent years, natural products have been shown to be successful as anti-diabetic agents both in vitro and in vivo, as well as clinical trials [18, 19]. Considering the need for clinical treatment and scientific research focused on DCM, it is necessary to develop new drugs with high efficacy, few side-effects, and low prices. Natural products have been extensively discussed for their therapeutic effects, indicating their great potential for treating DCM. Over the past decade, a large number of studies have considered the use of natural products for the intervention of DCM, but the value of these research results still needs to be explored and sorted out. The purpose of this paper is to review the recent research progress concerning natural products and their underlying mechanisms of action, in order to provide a comprehensive introduction to the potential of natural products for the treatment of DCM.
We searched the databases PubMed, Web of Science, Excerpt Medica, Science Direct, and Springer for the period 2012 to 2022 regarding the use of natural products to treat DCM, using the following search terms: (“natural products” OR “effective constituents” OR “polysaccharides” OR “alkaloids” OR “flavonoids” OR “terpenoids” OR “phenylpropanoids” OR “quinones” OR “sterides” OR “glycosides”) AND (“diabetic cardiomyopathy” OR “DCM”).
This review excluded studies that were found to have significant methodological errors or lack scientific value. To help our classification efforts, studies that focused on mixtures of various compounds or crude extracts were also excluded from this study, in addition to those focused on natural products with poorly defined chemical structures; for example, Polysaccharides such as Astragalus polysaccharides and Lycium barbarum polysaccharides have also been shown to treat DCM in vitro and in vivo. However, since the chemical structure is unclear, we did not include them in the study. In total, 72 natural compounds were identified and grouped, based on their structural characteristics, into five categories: Flavonoids, terpenoids, alkaloids, quinones, and others. In the following, the different types of natural products are classified and introduced according to their relative quantities. Fig. 1 shows the numbers of the different types of natural products.
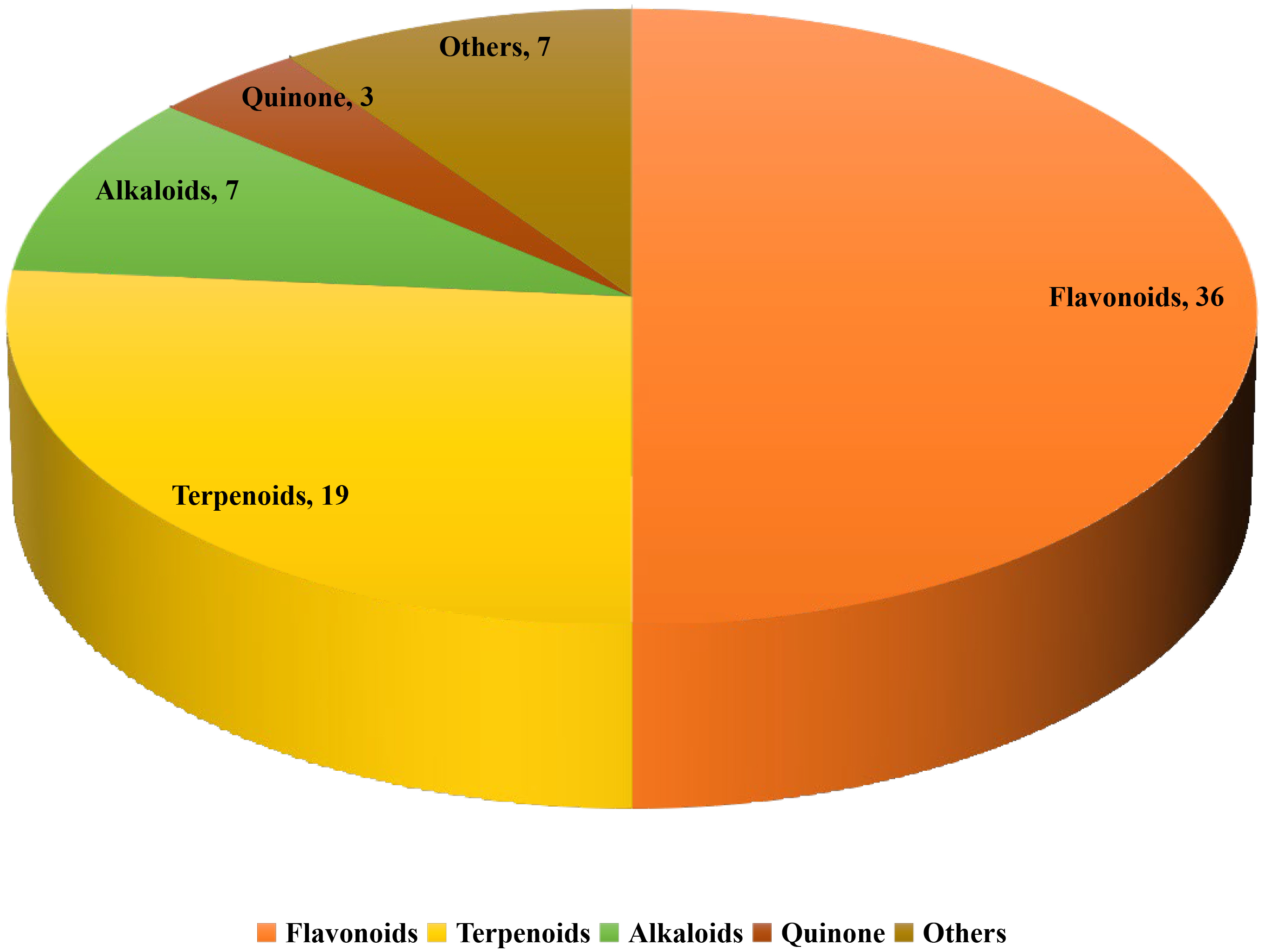 Fig. 1.
Fig. 1.Distribution of different sub-classes of natural products.
As one of the most diverse families of bioactive phytochemicals, flavonoids include over 9000 different compounds [20]. Flavonoid class compounds are naturally occurring poly-phenolic phytochemicals which are abundantly found among phytochemicals. Generally, the structure of flavonoids includes a basic C6–C3–C6 skeleton structure. There are many flavonoid sub-classes, such as flavonols, flavones, dihydroflavones, dihydroflavonols, chalcones, isoflavones, and biflavones. Flavonoids are often considered as breakthrough compounds for the development of new drugs, and have been widely studied for their effects in protecting the heart against diabetes-induced myocardial injury [21]. Flavonoids have the potential to alleviate DCM due to their anti-hyperglycemic, anti-oxidant, anti-inflammatory, and anti-apoptotic agents. A total of 36 flavonoids have been shown to possess effective therapeutic intervention effects on DCM, including 12 flavonols, 9 flavones, 4 dihydroflavones, 4 dihydroflavonols, 4 chalcones, 2 isoflavones, and 1 biflavone. Table 1 (Ref. [22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89]) provides the basic information and mechanisms of 36 flavonoids from recent studies on DCM, while Fig. 2 shows the chemical structures of the 36 flavonoids.
| Number | Flavonoid Subclass | Compounds | Molecular formula | Weight (g/mol) | Resources | Animal/Cell model | Dosage (mg/kg/d; µm/24 h) | Dosing cycle | Target/Pathways/Mechanism | Effects | Reference |
| 1 | Flavonol | Rutin | C |
610.5 | Ruta graveolens L., Scphora japonica L., etc. | STZ-induced diabetic rats | 8 | 12 days | / | Ameliorated metabolic abnormalities, anti-oxidative stress, anti-inflammatory, decreased myocardial apoptosis | [22] |
| STZ-induced diabetic rats | 50 | 24 weeks | / | Anti-inflammatory, anti-oxidative stress | [23] | ||||||
| STZ-induced diabetic rats | 50 (injected once a week) | 7 weeks | / | Anti-oxidative stress, attenuates cardiac remodeling, improve left ventricular and myocardial dysfunction | [24] | ||||||
| HFD + STZ-induced diabetic ApoE knockout mice | 60 | 6 weeks | Up-regulated Akt and MAPK signaling pathway | Decreased myocardial fibrosis, increased myocardial capillary density and decreased apoptosis, reduced ectopic lipid deposition, anti-oxidative stress | [25] | ||||||
| Alloxan-induced diabetic rats | 100 | 4 weeks | / | Decreased myocardial fibrosis, inhibition of metabolic acidosis | [26] | ||||||
| STZ-induced diabetic rats | 5 and 40 | 4 weeks | / | Suppression of tTG | [27] | ||||||
| HG-induced H9c2 myoblast cells | 2, 10, and 50 | 24 h | / | Decreased myocardial apoptosis, inhibits endoplasmic reticulum stress | [28] | ||||||
| 2 | Flavonol | Troxerutin | C |
742.7 | Robinia pseudoacacia L. | STZ-induced diabetic rats | 150 | 4 weeks | Activation of the AKT/IRS/JNK signaling pathway | Regulate glucose and lipid metabolism disorders, reduces levels of ROS | [29] |
| 3 | Flavonol | Quercetin | C |
302.23 | Psidii guajavae L., Allium cepa L., Camellia sinensis (L.) Kuntze, Malus pumila Mill., etc. | HC-induced diabetic rats | 0.005 | 4 weeks | / | Anti-oxidant stress, protected against diastolic dysfunction, prevent cholesterol accumulation and ATP reduction | [30] |
| HFD + STZ-induced diabetic mice | 100 | 4 months | / | Anti-inflammatory, regulated glycerophospholipid metabolism, ameliorate cardiac dysfunction, decreased myocardial fibrosis | [31] | ||||||
| Zucker Diabetic Fatty rats (fa/fa) | 20 | 6 weeks | Attenuated pro-hypertrophic gene transcription-regulated HDAC4/MEF2 signaling pathway, mitigation of pro-hypertrophic NFAT/calcineurin network | Improved diastolic dysfunction, reduced LV collagen content, reduced LV mass thickness and increased the internal diameter of LV | [32] | ||||||
| High-fat feeding + STZ-induced diabetic rats; HG-induced H9c2 cardiomyocytes | 160 | 6 months | Activation of Nrf-2 signaling pathway | Inhibited myocardial fibrosis, reduced the accumulation of ROS, inhibited pyroptosis | [33] | ||||||
| STZ-induced diabetic rats | 10 and 30 | 28 days | / | Anti-oxidative stress, decreased cardiomyocyte apoptosis | [34] | ||||||
| TAC-induced congestive heart failure mice | 50 | 15 days | Promote the desuccinylation of IDH2 through SIRT5 | Maintain mitochondrial homeostasis, anti-inflammatory, decreased myocardial fibrosis | [35] | ||||||
| 4 | Dihydroflavone | Naringenin | C |
272.25 | Citrus |
STZ-induced diabetic mice | 25 and 75 | 4 weeks | Activation of PPARs. | Anti-cardiac hypertrophy | [36] |
| HG-H9c2 cells | 0.1, 1, and 10 | 48 h | Activation of EETs/PPARs | Anti-cardiac hypertrophy | [37] | ||||||
| STZ-induced diabetic mice; HG- H9c2 cells | 25, 50, and 75; 10 | 63 days; 2 h | Inhibition of NF- |
Anti-oxidative stress, anti-inflammatory, decreased myocardial fibrosis and alleviated cardiomyocyte apoptosis | [38] | ||||||
| 5 | Dihydroflavone | Naringin | C |
580.5 | Citrus |
HS + HFD + STZ-induced diabetic rats; H9c2 cardiac cells | 25, 50 and 100; 80 | 6 weeks; 2 h | Up-regulated KATP channels, inhibition of the NF- |
Decreased cardiomyocyte apoptosis, anti-oxidative stress | [39] |
| Diabetic db/db mice | 20, 40 and 60 | 4 weeks | / | Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis | [40] | ||||||
| STZ-induced diabetic rats | 25, 50 and 100 | 8 weeks | / | Anti-oxidative stress, decreased cardiac apoptosis | [41] | ||||||
| 6 | Flavonol | Icariin | C |
676.7 | Epimedium brevicornu Maxim. | Diabetic db/db mice; primary cardiomyocyte obtained from the ventricles of newborn C57 mice | 30 | 16 weeks | Activation of Apelin/SIRT3 signaling pathway | Rescued the impaired mitochondria, decreased cardiac apoptosis | [42] |
| STZ + HG/high-fat diet-induced diabetic rats | 60 and 120 | 12 weeks | Suppression of TGF-β1/Smad signaling pathway | Improved glucose tolerance and insulin sensitivity, inhibited ECM accumulation, decreased myocardial fibrosis | [43] | ||||||
| STZ + HG/high-fat diet-induced diabetic rats | 30 and 60 | 8 weeks | Suppression of NOS3/PDE5-sGC-cGMP-PKG signaling pathway | Improves myocardial functions, decreased myocardial fibrosis, improve Ca |
[44] | ||||||
| 7 | Flavonol | Icariside II | C |
514.5 | Epimedium pubescens, Epimedium grandiflorum | STZ-induced diabetic rats | 5 | 8 weeks | Activation the Akt/NOS/NF- |
Anti-inflammatory, anti-oxidative stress, decreases cardiac apoptosis | [45] |
| 8 | Flavonol | (−)-Epigallo-catechin-3-gallate | C |
458.4 | Tea | STZ-induced diabetic rats/HG-induced H9c2 cardiac cells | 100; 20 | 6 weeks; 24 h | Stimulating the SIRT1 signaling pathway | Attenuated cardiac dysfunction, decreased myocardial fibrosis, decreased myocardial apoptosis, anti-oxidative stress | [46] |
| HFD + STZ-induced diabetic rats | 40 and 80 | 8 weeks | Regulation of AMPK/mTOR signaling pathway, repression of the TGF-β/MMPs signaling pathway | Activation of autophagy, decreased myocardial fibrosis | [47] | ||||||
| STZ-nicotinamide-induced diabetic rats | 2 (on alternate days) | 1 month | / | Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, decreased cardiac apoptosis | [48] | ||||||
| 9 | Dihydroflavonol | (-)-Epicatechin | C |
290.27 | Tea | Hyperglycemia-induced cardiac fibroblasts | 1 | 48 h | Regulation of Smad/TGF-β1 signaling pathway | Decreased myocardial fibrosis | [49] |
| 10 | Flavone | Scutellarin | C |
462.4 | Scutellaria baicalensis Georgi | STZ-induced diabetic mice | 10 and 20 | 4 weeks | / | Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis | [50] |
| HFD + STZ-induced diabetic rats | 10 and 20 | 6 weeks | Regulation of Nrf-2/Keap1 signaling pathway and TLR4/MyD88/NF- |
Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, decreased cardiac apoptosis | [51] | ||||||
| HS + HFD + STZ-induced diabetic rats | 100 and 200 | 8 weeks | / | Activation of autophagy, decreased cardiac apoptosis | [52] | ||||||
| 11 | Dihydroflavonol | Dihydromyricetin | C |
320.25 | Ampelopsis grossedentata Hand.-Mazz. | STZ-induced diabetic rats | 100 | 2 weeks | Suppression of miR-34a | Activation of autophagy, mitigates cardiac dysfunction | [53] |
| STZ-induced diabetic mice | 100 | 14 weeks | Activation of AMPK/ULK1 signaling pathway | Anti-inflammatory, anti-oxidative stress, decreased cardiac apoptosis, improved mitochondrial function, restored cardiac autophagy | [54] | ||||||
| STZ-induced diabetic mice | 250 | 12 weeks | Activation of SIRT3 | Improved cardiac dysfunction; ameliorated cardiac hypertrophy, anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, decreased cardiac necroptosis | [55] | ||||||
| 12 | Flavone | Luteolin | C |
286.24 | Capsicum annuum L., Lonicera japonica Thunb., Perilla frutescens (L.) Britton, etc. | STZ-induced diabetic mice; HG-induced H9C3 cells | 20; 5 and 10 | 15 weeks; 24 h | Inhibition of NF- |
Anti-oxidant stress, anti-inflammatory, decreased myocardial fibrosis and hypertrophy | [56] |
| STZ-induced diabetic mice and db/db mice; HG and high-insulin-induced primary neonatal rat cardiomyocytes | 20; 1 µM | 12 weeks; 48 h | Up-regulated phosphorylated protein AMPK and AKT/GSK-3 signaling pathway | Prevented cardiac hypertrophy, decreased myocardial fibrosis, ameliorate cardiac dysfunction | [57] | ||||||
| STZ-induced diabetic rats | 200 | 8 weeks | / | Anti-oxidative stress, inhibits left ventricular dysfunction and remodeling | [58] | ||||||
| 13 | Flavonol | Kaempferol | C |
286.24 | Tea, Citrus |
STZ-induced diabetic mice; HG-induced H9c2 cells | 10; 2.5 | 8 weeks; 1 h | Inhibition of NF- |
Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, decreased cardiac apoptosis | [59] |
| STZ-induced diabetic rats | 50 | 8 weeks | Activation of SIRT1 signaling pathway, down-regulated TGF-β1, up-regulated Nrf-2, and suppression of NF- |
Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, inhibits cardiomyocytes intrinsic cell death | [60] | ||||||
| 14 | Flavone | Genistein | C |
270.24 | Glycine max (L.) Merr., Pueraria montana var. lobata (Ohwi) Maesen & S. M. Almeida | STZ-induced diabetic rats | 300 | 24 weeks | / | Decreased myocardial fibrosis and hypertrophy | [61] |
| STZ-induced diabetic rats | 5 and 25 | 4 weeks | Suppression of the TGF-β1/Smad3 signaling pathway | Decreased myocardial fibrosis and hypertrophy | [62] | ||||||
| 15 | Chalcone | Phloretin | C |
274.27 | Malus pumila Mill., Litchi chinensis Sonn. | STZ-induced diabetic mice; HG-induced H9c2 cells | 20 | 56 days; 24 h | Restored SIRT1 expression | Decreased myocardial fibrosis, anti-inflammatory | [63] |
| STZ-induced diabetic mice; HG-induced H9c2 cells | 10 (on two days); 10 | 8 weeks; 1 h | Activation of Keap1/Nrf-2 signaling pathway | Anti-oxidative stress, decreased myocardial fibrosis | [64] | ||||||
| 16 | Dihydroflavonol | Silymarin | C |
482.4 | Silybum marianum (L.) Gaertn. | STZ-induced diabetic rats; primary cardiac fibroblasts | 25, 50, and 100; 100 mmol/L | 5 weeks; 24 h | Inhibition of TGF-β1/Smad signaling pathway | Decreased myocardial fibrosis and collagen deposition | [65] |
| Alloxan-induced diabetic rats | 120 | 10 days | / | Decreased cardiac apoptosis | [66] | ||||||
| 17 | Flavonol | Fisetin | C |
286.24 | Acacia greggii A.Gray, Vachellia farnesiana (L.) Wight & Arn., etc. | STZ-induced diabetic rats | 2.5 | 6 weeks | / | Anti-inflammatory, anti-oxidative stress, decreased cardiac apoptosis, ameliorates hyperglycemia and dyslipidemia | [67] |
| STZ-induced diabetic rats | 2.5 | 12 weeks | / | Anti-inflammatory, anti-oxidative stress | [68] | ||||||
| 18 | Isoflavone | Puerarin | C |
416.4 | Pueraria montana var. lobata (Ohwi) Maesen & S. M. Almeida | STZ-induced diabetic rats; HG-induced H9c2 cells | 50, 100, and 200; (10 |
8 weeks; 12 h | Inhibition of NF- |
Anti-inflammatory, decreased myocardial fibrosis | [69] |
| STZ-induced diabetic rats | 50, 100, 200 | 6 weeks | / | Regulated lipid metabolism disorder, anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, preserved the myocardial integrity, inhibited pyroptosis | [70] | ||||||
| 19 | Chalcone | Aspalathin | C |
452.4 | Aspalathus linearis | Diabetic db/db mice; HG-induced H9c2 cells | 13 and 130; 1µM | 6 weeks; 6 h | Activation of Nrf-2 | Anti-inflammatory, anti-oxidative stress, decreased cardiac apoptosis | [71] |
| HG-induced H9c2 cells | 6 µM | 6 h | / | Anti-inflammatory, anti-oxidative stress, decreased cardiac apoptosis | [72] | ||||||
| 20 | Dihydroflavone | Liquiritin | C |
418.4 | Glycyrrhiza uralensis Fisch. | High fructose-induced diabetic mice; cardiomyocytes from the experimental mice | 10 and 20; 0–32 µM | 10 weeks; 24 h | Suppression of NF- |
Anti-inflammatory, decreased myocardial fibrosis | [73] |
| 21 | Dihydroflavone | Liquiritigenin | C |
256.25 | Glycyrrhiza uralensis Fisch. | High fructose-induced mice as the experimental mice; fructose-induced H9c2 cells | 4, 8 and 16; 10 and 20 | 10 weeks; 24 h | Inhibition of NF- |
Anti-inflammatory, decreased myocardial fibrosis | [74] |
| 22 | Chalcone | Isoliquiritigenin | C |
256.25 | Glycyrrhiza uralensis Fisch. | STZ-induced diabetic mice; HG-induced H9c2 cells | 10 and 20; 2.5, 5, 10, 20, and 40 | 12 weeks; 24 h | Inhibition of MAPKs signaling pathway, induction of the Nrf-2 signaling pathway | Suppression HG-induced hypertrophy, anti-inflammatory, decreased myocardial fibrosis, decreased cardiac apoptosis | [75] |
| 23 | Isoflavone | Daidzein | C |
254.24 | Glycine max (L.) Merr. | STZ-induced diabetic rats | 25, 50, and 100 | 4 weeks | / | Anti-oxidative stress | [76] |
| 24 | Flavone | Apigenin | C |
270.24 | Apium graveolens L., Verbena officinalis L., etc. | STZ-induced diabetic mice; HG-induced H9c2 cells | 100; 25 | 7 months; 24 h | Inhibition of NF- |
Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, decreased cardiac apoptosis | [77] |
| 25 | Flavonol | Myricitrin | C |
464.4 | Morella rubra Lour. | STZ-induced diabetic mice; AGEs-induced H9c2 cells | 75, 150, and 300; 3.12, 6.25, 12.5, 25, 50, and 100 µg/mL | 8 weeks; 12 h | Activation of the PI3K/Akt signaling pathway and Nrf-2/ARE signaling pathway | Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, decreased cardiac apoptosis, attenuates hypertrophic | [78] |
| 26 | Flavone | Nobiletin | C |
402.4 | Citrus |
STZ-induced diabetic mice | 50 | 11 weeks | Suppression of JNK, P38, and NF- |
ameliorated oxidative stress, inflammatory status and apoptosis, decreased myocardial fibrosis | [79] |
| 27 | Flavonol | Myricetin | C |
318.23 | Morella rubra Lour. | STZ-induced diabetic mice | 200 | 6 months | Inhibition of I |
Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, decreased cardiac apoptosis | [80] |
| 28 | Flavone | Baicalein | C |
270.24 | Scutellaria baicalensis Georgi | HS + HFD + STZ-induced diabetic rats | 100 and 200 | 16 weeks | Activation of PI3K/Akt signaling pathway | Anti-inflammatory, anti-oxidative stress | [81] |
| 29 | Flavone | Sciadopitysin | C |
580.5 | Ginkgo biloba L. | HG-induced Human cardiomyocyte line AC16 | 0, 1, 5, 10, and 20 µM | 24 h | Activation of PI3K/PKB/GSK‐3β signaling pathway | Anti-oxidative stress, decreased cardiac apoptosis | [82] |
| 30 | Flavonol | Spiraeoside | C |
464.4 | Spiraea salicifolia L. | HG‐induced human cardiomyocytes; HG-induced AC16 cells | 1, 5, 10, and 20 µM | 0, 24, and 48 h | Activation of PI3K/Akt/Nrf-2 pathway | Anti-oxidative stress, decreased cardiac apoptosis | [83] |
| 31 | Flavone | Chrysin | C |
254.24 | Oroxylum indicum (L.) Kurz, Pinus monticola Dougl., honey, etc. | STZ-induced diabetic rats | 60 | 4 weeks | Inhibition of AGE-RAGE axis; activation of PPAR-γ | Anti-inflammatory, anti-oxidative stress | [84] |
| 32 | Biflavone | Kolaviron | C |
588.5 | Garcinia kola Heckel | STZ-induced diabetic rats | 200 | 28 days | / | Anti-inflammatory, anti-oxidative stress | [85] |
| 33 | Flavonol | Galangin | C |
270.24 | Alpinia officinarum Hance | STZ-induced diabetic rats | 15 mg/kg/d | 6 weeks | / | Anti-inflammatory, anti-oxidative stress, decreased cardiac apoptosis, ameliorate dyslipidemia and myocardial injury, prevents DNA damage | [86] |
| 34 | Dihydroflavonol | Taxifolin | C |
304.25 | Silybum marianum (L.) Gaertn., Tamarindus officinalis Gaertn. and Larix gmelinii (Rupr.) Kuzen. | STZ-induced diabetic mice; HG-induced H9c2 cells | 25, 50 and 100 | 4 weeks | / | Anti-oxidative stress, decreased cardiac apoptosis, improved diastolic dysfunction, ameliorated myocardium structure abnormality | [87] |
| 35 | Flavone | Wogonin | C |
284.26 | Scutellaria baicalensis Georgi | STZ-induced diabetic mice; HG-primary NRVMs | 10; 10 | 16 weeks; 12 h | / | Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis, decreased cardiac apoptosis | [88] |
| 36 | Chalcone | Hydroxysafflor yellow A | C |
612.5 | Crocus sativus L. | HFD + STZ-induced diabetic mice | 60 | 12 weeks; 25 h | / | Anti-oxidative stress | [89] |
AGE, advanced glycation end-product; AKT, protein kinase B; ARE , antioxidant
response element; cGMP, cyclic guanosine monophosphate; EETs, epoxyeicosatrienoic
acids; GSK-3
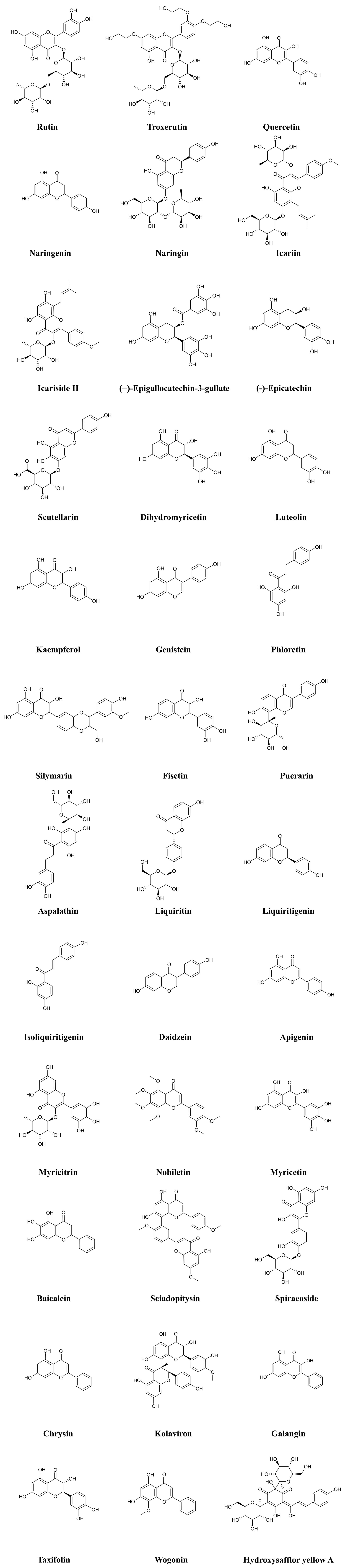 Fig. 2.
Fig. 2.Chemical structures of considered flavonoids.
Rutin, as a natural flavonoid compound, is found naturally in common foods. It is especially abundant in Ruta graveolens L., Scphora japonica L., and so on. Rutin may represent a potential therapeutic agent for DM and its complications in the flavonoid family. As early as 1948, there were reports of rutin treating DM complications [90]. Rutin substantially improved cardiac function and structure in DCM, but the mechanism and effect are complex and multi-faceted.
Metabolic disorders, oxidative stress, and inflammatory reactions are involved
in the occurrence and development of DCM, which interact to induce myocardial
injury. The risk of oxidative stress and inflammatory responses is increased by
metabolic disorders, and oxidative stress and chronic inflammation can lead to
the development of metabolic diseases [91]. Studies have shown that rutin is
effective in the treatment of DCM, through ameliorating myocardium metabolic
abnormalities, oxidative stress, inflammation, and cellular apoptosis, in
streptozotocin (STZ)-induced diabetic rats [22]. Inflammation and oxidative
stress often complement and reinforce each other to form a vicious cycle. Rutin
improves DCM by exerting both antioxidant stress and anti-inflammatory effects,
and its cardioprotective effects were mediated by alterations in tumor necrosis
factor
Alloxan is a synthetic pyrimidine derivative first synthesized in the 19th century, which causes necrosis by a selective, toxic effect in certain cells [92]. Fibrosis, the excess and unsuitable accumulation of extracellular matrix in various tissues, is a common occurrence in patients with advanced DM [93]. Rutin has been proven to be potential therapeutic target against alloxan-induced diabetic kidney disease (DKD) and DCM in experimental rats, through the prevention of metabolic acidosis and fibrosis [26]. The cardiovascular disease (CVD) and kidney disease are closely inter-related [94]. DKD is highly correlated with DCM in terms of consistency of initial etiology and similarity of underlying pathological mechanisms, which may be a key factor in the remarkable efficacy of natural products in treating both diseases simultaneously.Treating one disease while benefiting other diseases may also be the advantage of rutin, which has reference significance for the study of its role in diabetic complications.
Tissue transglutaminase (tTG) belongs to the transglutaminase family, members of which have a diverse array of enzymatic and non-enzymatic functions. The inhibition of tTG has been reported to benefit CVD, by decreasing myocardial fibrosis and reducing cardiomyocyte hypertrophy [95]. Rutin may inhibit the expression of tTG and regulate the progression of myocardial injury and fibrosis in STZ-induced DCM rats [27]. However, the pathway mechanism underlying the above processes remains unclear, as tTG has not been studied much in the context of DCM.
The ER is an organelle that is specialized in protein folding and trafficking. Through its stress and calcium handling, proteins co-operate to keep the myocardial cell properly functioning. Endoplasmic reticulum stress (ERS) serves an important role in the course of DCM’s pathological progression, which can cause cell dysfunction and apoptosis [96]. Rutin alleviated HG-induced myocardial cell dysfunction and apoptosis by inhibiting ERS [28].
Troxerutin, a derivative of the naturally occurring bioflavonoid rutin. The
c-Jun N-terminal protein kinases (JNKs) form one sub-family of the
mitogen-activated protein kinases (MAPK) group, mediate eukaryotic cell responses
to a wide range of abiotic and biotic stress insults [97]. As a critical node for
the insulin signal regulation mechanism, insulin receptor substrate (IRS) is
essential for the prevention and treatment of DM. Insulin resistance has been
linked to modifications in protein kinase B (PKB, also known as AKT)
phosphorylation. Troxerutin appears to protect against DCM through inhibition of
nuclear factor kappa B (NF-
Quercetin is one of the widely existing flavonoids, which is abundant in nature, and the quantity of quercetin in onion is the highest [98]. The cardioprotective function of quercetin seems to focus more on two aspects in the context of DCM: regulation of lipid metabolism disorder series reactions and intervention of abnormal cell death.
Abnormal energy metabolism plays a significant role in the occurrence and development of CVDs, and cardiac energy metabolism regulation is a new frontier in CVD treatment [99]. In the context of lipid metabolism disorder, the accumulation of lipid in the myocardium causes cardiac lipotoxicity and induces cardiac dysfunction. Quercetin attenuated cardiac diastolic dysfunction, up-regulated intracellular anti-oxidant stress mechanisms, prevented cardiac cholesterol accumulation, and decreased the increase in myocyte density resulting from high cholesterol [30]. In addition, quercetin may ameliorate cardiac dysfunction and fibrosis by reducing glycerophospholipid metabolism dysregulation [31]. A connection exists between lipid metabolism disorder and pathological myocardial hypertrophy. Quercetin ameliorated pro-hypertrophic signaling pathways regulating the hypertrophic response in the cardiomyocyte, which provoked the inhibition of pro-hypertrophic signals in Zucker Diabetic Fatty rats (fa/fa) [32].
Myocardial cell death is a crucial factor in the development and progression of different etiological cardiomyopathies [100]. Pyroptosis has been observed in different heart cell types in DCM, including cardiomyocytes, endothelial cells, and fibroblasts [101]. Quercetin inhibits the progression of cell pyroptosis, thereby alleviating DCM, and its mechanism of action is related to the activation of the nuclear factor erythroid 2-related factor 2 (Nrf-2) signaling pathway [33]. Myocardial apoptosis plays a vital role in the pathogenesis of CVD in the DM. Mitochondrial pathways of apoptosis are inhibited by Quercetin, which prevents the death of cardiomyocytes [34].
The silent information regulator 5 (SIRT5), as a representive of the Sirtuin family, is valued for its role in myocardial injury in diabetes. Quercetin may promote the desuccinylation of isocitrate dehydrogenase 2 (IDH2) through SIRT5, thus maintaining mitochondrial homeostasis, protecting cardiomyocytes from inflammatory conditions and improving myocardial fibrosis, and thus reduce the incidence of HF [35].
Naringenin is found mainly in citrus fruits (e.g., grapefruit) and others, such as tomatoes and cherries. Naringenin has emerged as an important natural phytochemical with potential for the treatment or prevention of various disorders, such as obesity, diabetes, cardiac diseases, and metabolic syndrome [102].
Cardiac hypertrophy is an adaptive response to stimulation, but pathological cardiac hypertrophy usually develops into HF. Naringenin improved cardiac hypertrophy in vivo, which may be related to up-regulation of the expression of cytochrome P450 2J3 (CYP2J3), elevated levels of epoxyeicosatrienoic acids (EETs), and the activation of peroxisome proliferator-activated receptors (PPARs) [36]. Cell experiments have also confirmed that EETs and PPARs function together, which may contribute to the anti-hypertrophic effect of naringenin in vitro under HG conditions [37]. EETs and PPARs seem to be effective signaling pathways for naringenin intervention in DCM, but relevant studies still need to be further enriched.
Naringenin can regulate the Nrf-2 and NF-
Naringin is a natural polyphenol bioflavonoid, is the same as Naringenin mainly found in citrus fruits. Naringin could significantly alleviate various physical and chemical stimuli induced cardiovascular disorders such as DCM, ischemic heart diseases, oxidative stress-induced cardiac injury and diet-induced cardiovascular dysfunctions [103].
NF-
The precise regulation of calcium homeostasis in cardiomyocytes is the key to
maintaining the systolic function of the heart. Treatment of DCM with naringin
protected cardiomyocytes by reducing diastolic Ca
Furthermore, by mitigating mitochondrial oxidative stress-induced injuries and inhibiting the ERS-mediated apoptotic pathway, naringin may provide protection against diabetes-induced myocardial damage [41].
Icariin, a major flavonoid extracted from Epimedium brevicornu Maxim, has presented a wide range of pharmacological activities. icariin and icariside II (its bioactive form), have been found to have preventive and therapeutic effects on DCM in pre-clinical studies.
Mitochondrial dysfunction generates more ROS and disrupts the oxidative phosphorylation process which, in turn, leads to myocardial oxidative stress damage. Icariin’s cardioprotective effect against DCM is mediated by activation of the Apelin/Silent information regulator 3 (SIRT3) signaling pathway, which prevents mitochondrial dysfunction [42].
Icariin is a promising natural product in anti-fibrotic and myocardial
amelioration. Transforming growth factor-
Icariside II is the main pharmacological metabolite of icariin in vivo.
Treatment with icariside II improved DCM through antioxidative stress,
antiinflammatory, and anti-apoptotic effects. Thus, the above mechanism is
mediated by the AKT/NOS/NF-
Catechins are a class of phenolic active compound extracted from edible plants such as tea. Standardized green tea extract, which is rich in catechins, prevented the initial myocardial damage in diabetic hearts from developing into DCM [45]. There are four main types of green tea catechins: (-)-epigallocatechin-3-gallate (EGCG), which accounts for approximately 60% of the green tea catechin content; (-)-epigallocatechin (EGC) (19%); (-)-epicatechin-3-gallate (ECG) (13.6%); and (-)-epicatechin (EC) (6.4%) [106].
EGCG is the major polyphenolic compound present in green tea, which attenuated
cardiac dysfunction, reduced myocardial infarct size and myocardial fibrosis, and
decreased apoptosis and oxidative stress by stimulating the silent information
regulator 1 (SIRT1) signaling pathway [46]. AMP activated protein kinase (AMPK)
is a highly conserved metabolic master regulator, mammalian target of rapamycin
(mTOR) is a serine/threonine protease, and the AMPK/mTOR signaling pathway
involved in both plays a leading role in the regulation of autophagy. EGCG
attenuated myocardial fibrosis in DCM, and its underlying mechanisms were
associated with activation of autophagy through supression of
TGF-
Emerging evidence supports a beneficial action of the potential impact of EC on
the development/progression of DCM. The cardiac fibroblasts cultured in HG
acquired a profibrotic phenotype, which was blocked by EC. The underlying
mechanism was likely mediated by the effects of the G-protein coupled estrogen
receptor (GPER) on the Smad/TGF-
Scutellarin is an herbal flavonoid glucuronide, extracted from
Scutellaria baicalensis Georgi, with multiple pharmacological
activities. Scutellarin has a series of effects such as anti-inflammation,
anti-oxidative stress, improve heart function and inhibit myocardial fibrosis
level. The specific mechanism includes inhibition of the activation of
nucleotide-binding oligomerization domain-like receptor with a pyrin domain 3
(NLRP3) and NF-
Autophagy and apoptosis are often associated in the pathological process of DCM. Scutellarin can promote the autophagy signaling pathway by up-regulating autophagy-related factors (Beclin-1 and LC3-II) and inhibit the apoptotic signal pathway by down-regulating apoptosis-related factors (caspase-3, caspase-8, caspase-9, caspase-12, Bax, and Cyt-C), thereby relieving DCM [52]. The complex interplay between apoptosis and autophagy further inspired a treatment concept for CVD through balanced switching between the two responses [109]. However, the relationship between the intervention of scutellarin in apoptosis and autophagy has not yet been revealed.
Dihydromyricetin is an important plant flavonoid, extracted from vine tea (Ampelopsis grossedentata Hand-Mazz.), which has attracted great attention for its health-beneficial activities. Excessive or insufficient autophagy has been described as a contributing factor to many pathological conditions. Targeting specific microRNA (miRNA) for autophagy modulation may provide reliable promising therapeutic strategies for DCM. By reducing the expression of miR-34a, dihydromyricetin restores impaired autophagy and thus alleviates DCM [53]. Unc-51-like autophagy activating kinase 1 (ULK1)—one of the key elements of the autophagy activator complex—together with AMPK kinases, guarantee the precise function of autophagy. The autophagy regulation mechanism of dihydromyricetin is realized through AMPK/ULK1 signaling pathway activation [54].
Dihydromyricetin has the potential to be used in the treatment of DCM, as it reduced inflammation, anti-oxidative stress, improved cardiac dysfunction, ameliorated cardiac hypertrophy, inhibited myocardial fibrosis and suppressed necroptosis. The above effects were realized through SIRT3 signaling pathway activation [55].
Luteolin is a common flavonoid present in many types of plants, such as flowers,
fruits, vegetables, medicinal herbs, and spices. Luteolin has displayed a wide
range of pharmacological properties, including anti-oxidant, anti-microbial,
anti-inflammatory, chemopreventive, chemotherapeutic, cardioprotective,
anti-diabetic, and neuroprotective activities [110]. Luteolin has a significant
pharmacological effect in terms of DCM prevention and treatment. It can
significantly reduce the inflammatory phenotype and anti-oxidative stress, as
well as preventing myocardial fibrosis, cardiac hypertrophy, and dysfunction. The
mechanisms involved include activation of the Nrf-2 signaling pathway and
inhibition of the NF-
HF is one of the pathological features of DCM and the final outcome of its development. The results of one study demonstrated that luteolin attenuates myocardial oxidation, thereby inhibiting the progression of LV dysfunction in mice model of HF [58].
Kaempferol is a flavonoid aglycone found naturally in many plants, such as
beans, bee pollen, broccoli, capers, cauliflower, cabbage, endive, fennel, and
garlic [112]. Kaempferol acts as a potential therapeutic agent for the treatment
of DCM, as it can prevent diabetes-induced inflammation, oxidative stress,
myocardial fibrosis, and apoptosis, mechanically linked to the inhibition of
NF-
Genistein is the natural isoflavone with a comprehensive range of
pharmacological properties, such as anti-oxidant stress, anti-inflammatory,
anti-bacterial, and anti-viral activities, as well as effects on diabetes and
lipid metabolism [113]. Genistein improved the damage of diabetic myocardium by
virtue of its anti-inflammatory and anti-oxidant effects. Its cardioprotective
effect seems to be mediated by inhibiting the activities of TNF-
Phloretin is one of the best-known and abundant dihydrochalcones, having significant pharmacological activity. SIRT1-mediated deacetylation has a significant impact on several biological processes, which include cellular senescence, apoptosis, glucose metabolism, lipid metabolism, oxidative stress, and inflammation [114]. Phloretin protected against HG-induced inflammation and fibrosis in H9c2 cell, by regulating the expression of SIRT1 [63]. In addition, phloretin acts as a promising natural agent through increased Nrf-2 expression and dissociation of the Keap1/Nrf-2 complex, suppressing HG-induced cardiomyocyte oxidation and fibrotic injury [64].
Silymarin is obtained from Silybum marianum (L.) Gaertn., which has
principally been used over the centuries to treat liver disease. Studies have
revealed other therapeutic effects of silymarin in terms of cardioprotection,
neuroprotection, immune modulation, and cancer [115]. The therapeutic effect of
silymarin on DCM has been newly discovered in recent years. Administration of
silymarin attenuated myocardial fibrosis and collagen deposition through
decreased p-Smad2/3 and TGF-
Fisetin is a flavonoid with significant biological activity, which is found in many fruits and vegetables such as strawberries, persimmons, apples, onions, grapes, and cucumbers. Fisetin might be worth considering the therapeutic potential of fisetin for human DCM, which attenuates the development of DCM by ameliorating oxidative stress, inflammation, and apoptosis [67]. Protein kinase R (PKR) is a key inducer of inflammation, oxidative stress, insulin resistance, and glucose homeostasis in DM. Fisetin can preserve cardiac function and prevent further cardiac damage in diabetes through anti-inflammatory, improving cardiac glucose metabolism, suppression of FAs oxidation, anti-fibrotic, and anti-apoptotic effects. The above role may be related to suppression of PKR [68].
Puerarin is the most important phytoestrogen extracted from Pueraria
montana var. lobata (Ohwi) Maesen & S. M. Almeida, and is widely used as a
clinical auxiliary drug for the treatment of metabolic disorders and CVD.
Puerarin may have promising therapeutic potential for DCM, with related to the
attenuation of inflammation and fibrotic. Further evidence comes from the result
that puerarin significantly inhibited the production of pro-inflammatory
cytokines by blocking NF-
Aspalathin is abundantly present in Aspalathus lineari, a plant from South African often used as a herbal tea. It increases glucose oxidation and modulates fatty acid utilization, producing a favorable substrate shift in H9c2 cells. Such a favorable shift may be of importance in the protection of the myocardium against cell apoptosis [71]. Related mechanisms include maintaining cellular homeostasis, modulating anti-inflammatory and anti-oxidative stress, and protecting the myocardium against HG-induced apoptosis through activation of Nrf-2 [72].
Liquiritigenin, liquiritin, and isoliquiritigenin are natural flavonoids
distributed in Glycyrrhizae Radix et Rhizoma, which has been widely used
as a herbal medicine for centuries in China. Liquiritin may be a promising
candidate for the treatment of diabetes-related myocardial fibrosis, which had a
protective effect against myocardial fibrosis through the suppression of
NF-
Daidzein is an isoflavone extract from soy, and the role of it in diabetic cardiac complications has been well studied and proved. Daidzein has therapeutic potential against diabetes-related cardiac complications, which may reduce glucotoxicity-induced cardiac mechanical dysfunction [116]. Daidzein prevented the progression of DCM through an anti-oxidative mechanism by inhibiting the activation of NADPH oxidase 4 (NOX4) in cardiomyocytes. It also improved the AMPK and SIRT1 signaling pathway and prevented changes in the structure and function of the myocardium [76].
Apigenin is a natural flavonoid found in many dietary plant foods. Apigenin have
been reported to be beneficial a variety of CVD, such as atherosclerosis,
hypertension, ischemia/reperfusion-induced myocardial injury, DCM, and
drug-induced cardiotoxicity [117]. Apigenin effectively mitigated
diabetes-induced myocardial inflammation, oxidative stress, fibrosis, and
apoptosis, both in vivo and in vitro. The internal mechanism is
that apigenin suppresses the phosphorylation of the NF-
Myricitrin is a member of the flavonol class of flavonoids, which is commonly derived from vegetables, fruits. Myricitrin exerts cardioprotective effects against DCM through the anti-inflammatory, anti-oxidative stress and inhibition of apoptosis. Its mechanism of action is through attenuating the Nrf-2 inhibition in DCM, by the regulation of AKT and extracellular signal-regulated kinase (ERK) phosphorylation [78].
Nobiletin is a polymethoxyflavone primarily present in citrus fruits. Nobiletin
mitigates cardiac dysfunction and interstitial fibrosis in DCM. These effects of
nobiletin may be attributed to the suppression of JNK, P38, and NF-
Myricetin is a
hexahydroxyflavone
and isolated from the leaves of Morella rubra Lour. Myricetin possesses
potential protective effects in DCM, which attributed to alleviate oxidative
stress, inflammation, apoptosis, and fibrosis.The underlying mechanisms of it at
least partly associated to the inhibition of the
I
Baicalein is a trihydroxyflavone derived from the roots of Scutellaria baicalensis Georgi. Baicalein was effective in preventing damage to DCM caused by oxidative stress and inflammation, and the PI3K/AKT signaling pathway may have been involved in mediating these effects [81].
Sciadopitysin is an amentoflavone-type biflavonoid contained in Taxus
chinensis, which exerts anti-inflammatory and anti-oxidative effects.
Sciadopitysin alleviated HG-caused oxidative stress and apoptosis in
cardiomyocytes by activating the PI3K/PKB/GSK-3
Spiraeoside, also known as quercetin-4-O-
Chrysin is a
dihydroxyflavone
which occurs naturally in many plants, honey, and propolis. The binding of AGE to
its receptor AGE (RAGE) enhances oxidative stress, thereby causing damage to cells
and tissues. Chrysin significantly ameliorated isoproterenol-induced myocardial
injury through anti-inflammatory and anti-oxidative stress. The PPAR-
Kolaviron, an important component of the seed of Garcinia kola Heckel, possesses a variety of biological activities, including anti-inflammatory and anti-oxidant stress properties. Kolaviron attenuated oxidative and inflammation cardiovascular injury in DCM [85].
Galangin is a naturally occurring flavonol glycoside found in Alpinia officinarum Hance. Galangin ameliorated HG, hyperlipidemia, oxidative stress, inflammation and apoptosis, and prevented myocardial damage in DCM [86].
Taxifolin is a dihydroflavonol commonly found in onion, Silybum marianum (L.) Gaertn., Tamarindus officinalis Gaertn., and Larix gmelinii (Rupr.) Kuzen. Taxifolin exerted cardioprotective effects against DCM by anti-oxidant stress and inhibition of apoptosis [87].
Wogonin is a flavonoid acting as a yellow color pigment, obtained from the roots of the plant Scutellaria Baicalensis Georgi. The anti-apoptotic, anti-inflammatory, anti-fibrosis, and anti-oxidative stress bioactivities of wogonin are expected to alleviate the progression of DCM [88].
Hydroxysafflor yellow A is the main bioactive compound of a traditional Chinese medicine (TCM) obtained from Crocus sativus L. Research has shown that the pharmacokinetics of Hydroxysafflor yellow A changed significantly in DCM, which may improve the anti-oxidative stress effect of the drug [89].
Terpenoids are the largest and most diverse group of natural products, attracting extensive attention due to their various biological activities [118, 119]. Terpenoids, which are composed of five carbon isoprene units, are classified into various subclasses based on their distinct chemical structures, including hemiterpenoids, monoterpenoids, sesquiterpenoids, diterpenoids, sesterterpenoids, triterpenoids, and tetraterpenoids. Terpenoids have been widely used in the treatment of numerous diseases because of their extensive range of biological activities, including their anti-microbial, anti-cancer, hypotensive, anti-hyperlipidemic, anti-hyperglycemic, anti-inflammatory, anti-oxidant, anti-parasitic, immunomodulatory, and anti-cholinesterase activities [120]. The significant pharmacological effects of terpenoids have been further demonstrated in DCM studies. A total of 19 terpenoids had effective therapeutic intervention effects on DCM, including 1 iridoid, 1 sesquiterpenoid, 6 diterpenoids, 9 triterpenoids, and 2 tetraterpenoids. Table 2 (Ref. [121, 122, 123, 124, 125, 126, 127, 128, 129, 130, 131, 132, 133, 134, 135, 136, 137, 138, 139, 140, 141, 142, 143, 144, 145, 146, 147, 148, 149, 150]) provides the basic information and mechanisms of these 19 terpenoids from recent studies on DCM, while Fig. 3 presents the chemical structures of the 19 terpenoids.
| Number | Terpenoid Subclass | Compounds | Molecular formula | Weight (g/mol) | Resources | Animal/Cell model | Dosage (mg/kg/d; µm) | Dosing cycle | Target/Pathways/Mechanism | Effects | Reference |
| 1 | Diterpenoid | Triptolide | C |
360.40 | Tripterygium wilfordii Hook. f. | STZ-induced diabetic rats | 100, 200, and 400 (µg/kg/d) | 6 weeks | Up-regulated MAPK signaling pathway | Improved myocardial energy metabolism | [121] |
| STZ-induced diabetic rats; HG-induced H9c2 cells | 100, 200, and 400 (µg/kg/d); 20 (ng/mL) | 6 weeks; 48 h | Suppression of NF- |
Anti-inflammatory, decreased myocardial fibrosis | [122] | ||||||
| HG and high-fat + STZ-induced diabetic rats | 50, 100, 200 µg/kg/d | 8 weeks | Inhibition of TLR4-induced NF- |
Regulated immune system, anti-inflammatory, decreased myocardial fibrosis, improved left ventricle function | [123] | ||||||
| 2 | Triterpenoid | Ginsenoside-Rb1 | C |
1109.30 | Panax ginseng C. A. Mey. | HFD + STZ-induced diabetic mice | 40 | 8 weeks | / | Improved calcium signaling | [124] |
| Diabetic db/db mice | 25, 50, and 100 | 12 weeks | Regulated adipocytokine pathway | Anti-inflammatory, decreased myocardial fibrosis, ameliorated apoptotic, anti-oxidative stress, reduced cardiac hypertrophy | [125] | ||||||
| 3 | Triterpenoid | Ginsenoside-Rg1 | C |
801.00 | Panax ginseng C. A. Mey. | HFD + STZ-induced diabetic rats | 10, 15, and 20 | 12 weeks | / | Decreased myocardial apoptosis, ameliorated oxidative stress | [126] |
| HFD + STZ-induced diabetic rats | 10, 15, and 20 | 12 weeks | / | Decreased myocardial apoptosis, reduced ER stress | [127] | ||||||
| 4 | Triterpenoid | Ginsenoside Rh2 | C |
622.90 | Panax ginseng C. A. Mey. | STZ-induced diabetic rats; HG-induced H9c2 cells | 5; 50 | 4 weeks; 24 h | Suppression of PPARδ/STAT3 Signaling pathway | Decreased myocardial fibrosis | [128] |
| 5 | Triterpenoid | Astragaloside IV | C |
785.00 | Astragalus membranaceus var. mongholicus (Bunge) P. K. Hsiao | HFD + STZ-induced diabetic rats | 80 | 8 weeks | / | Anti-inflammatory, decreased myocardial fibrosis, improved lipid metabolism | [129] |
| HG-induced H9c2 cells | 25, 50, and 100 | 24 h | Regulated miR-34a/Bcl2/(LC3II/LC3I) and pAKT/Bcl2/(LC3II/LC3I) pathway | Anti-oxidative stress, inhibition autophagy | [130] | ||||||
| STZ-induced diabetic rats; HG-induced H9c2 cells | 10, 20, and 40; 20, 40, and 80 | 16 weeks; 24 h | Regulated PGC-1 |
Regulate energy metabolism | [131] | ||||||
| 6 | Diterpenoid | Crocin | C |
977.00 | Crocus sativus L. | HFD + STZ-induced diabetic rats; HG-induced adult rat cardiac myocytes | 10 and 20; 1 and 10 (mmol) | 2 weeks; 3 h | / | Decreased myocardial apoptosis, inhibition autophagy | [132] |
| HFD + STZ-induced diabetic rats | 50 | 8 weeks | / | Anti-oxidative stress | [133] | ||||||
| STZ-induced diabetic rats | 40 | 4 weeks | Activation of PPAR |
Anti-inflammatory, anti-oxidative stress | [134] | ||||||
| 7 | Triterpenoid | Ursolic acid | C |
456.70 | Arctostaphylos uva-ursi (L.) Spreng., Prunella vulgaris L., Ilex rotunda Thunb., etc. | STZ-induced diabetic rats | 35 | 8 weeks | / | Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis | [135] |
| HFD + STZ-induced diabetic mice | 100 | 8 weeks | / | Anti-inflammatory | [136] | ||||||
| 8 | Triterpenoid | Glycyrrhizin | C |
822.90 | Glycyrrhiza uralensis Fisch. | ZDF rats; HG-induced AC16 human CMs cell | 50; 50 | 4 weeks; 24 h | Activation of Nrf-2 signaling pathway, inhibition of CXCR4/SDF1 and TGF- |
Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis | [137] |
| HS + HFD-induced diabetic mice | 150 | 8 weeks | Inhibition of HMGB1 | Anti-inflammatory | [138] | ||||||
| 9 | Tetraterpenoid | Fucoxanthin | C |
658.90 | Brown seaweed | STZ-induced diabetic rats; HG-induced H9c2 cells | 200; 1 | 12 weeks; 48 h | Regulation of BNIP3/Nix and Nrf-2 signaling pathway | Decreased myocardial fibrosis, reduced cardiac hypertrophy, reversed morphological and functional abnormalities of mitochondria, improved mitophagy | [139] |
| STZ-induced diabetic rats; HG-induced H9c2 cells | 200 and 230; 1 | 12 weeks; 48 h | Regulation of Nrf-2 signaling pathway | Decreased myocardial fibrosis and hypertrophy, anti-oxidative stress | [140] | ||||||
| 10 | Triterpenoid | Oleanolic acid | C |
456.70 | Canarium oleosum (Lam.) Engl., etc. | STZ-induced diabetic rats | 80/2 d | 2 weeks | Regulation of HO-1/Nrf-2 signaling pathway and GS/GP signaling pathways | Anti-oxidative stress | [141] |
| 11 | Triterpenoid | Chikusetsu saponin IVa | C |
795.00 | Swartzia simplex (Sw.) Spreng., Anredera baselloides (Kunth) Baill. | HG-induced H9c2 cells and rat primary cardiomyocytes | 12.5, 25 and 50 | 24 h | Activation of SIRT1/ERK1/2 and Homer1a signaling pathway | Anti-oxidative stress, decreased myocardial apoptosis, ameliorated Ca |
[142] |
| 12 | Triterpenoid | Betulin | C |
442.70 | Betula platyphylla Sukaczev | Diabetic db/db mice; HG-induced H9c2 cells | 20 and 40; 30 (mmol/L) | 12 weeks; 24 h | Reversed the SIRT1/NLRP3/NF- |
Anti-inflammatory, improved insulin resistance and hyperglycemia | [143] |
| 13 | Diterpenoid | Kirenol | C |
338.50 | Sigesbeckia orientalis L., Sigesbeckia glabrescens Makino, Sigesbeckia pubescens Makino, etc. | The GK rat; HG‐induced CMs and CFs from rats | 0.5 and 2; 20 and 40 | 8 weeks; 12, 24, or 36 h | Suppression of NF- |
Anti-inflammatory, decreased myocardial fibrosis, ameliorated apoptosis | [144] |
| 14 | Iridoid | Catalpol | C |
362.33 | Rehmannia glutinosa (Gaertn.) Libosch. ex Fisch. & C. A. Mey. | HS + HFD + STZ-induced diabetic rats; HG-induced Mouse cardiomyocytes | 10; 1, 2, and 4 (mg/mL) | 12 weeks; 24 h | Regulated Neat1/miR-140-5p/HDAC4 axis | Decreased myocardial apoptosis | [145] |
| 15 | Diterpenoid | Isosteviol | C |
318.40 | Stevia rebaudiana (Bertoni) Bertoni | STZ-induced diabetic rats | 8 | 11 weeks | Inhibition of ERK and NF- |
Anti-inflammatory, anti-oxidative stress | [146] |
| 16 | Tetraterpenoid | Bixin | C |
394.50 | Bixa orellana L. | HFD-induced diabetic rats; HG-induced H9c2 cells | 50, 100, and 200; 20, 40, and 80 | 14 weeks; 24 h | Activation of Nrf-2 signaling pathway | Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis | [147] |
| 17 | Sesquiterpenoid | C |
204.35 | Citrus |
STZ-induced diabetic rats | 100 and 200 | 4 weeks | Inhibition of NF- |
Anti-inflammatory, anti-oxidative stress, decreased myocardial fibrosis | [148] | |
| 18 | Diterpenoid | Andrographolide | C |
350.40 | Andrographis paniculata (Burm. f.) Wall. ex Nees in Wallich | STZ-induced diabetic mice; HG-induced H9c2 cells | 1, 10, and 20; 1, 5 and 10 (µM) | 12 weeks; 48 h | Suppression of NF- |
Anti-inflammatory, anti-oxidative stress, decreased myocardial apoptosis | [149] |
| 19 | Diterpenoid | Forskolin | C |
410.50 | Coleus forskohlii (Willd.) Briq. | STZ-induced diabetic mice | 2 | 4 weeks | / | Decreased myocardial fibrosis, anti-oxidative stress | [150] |
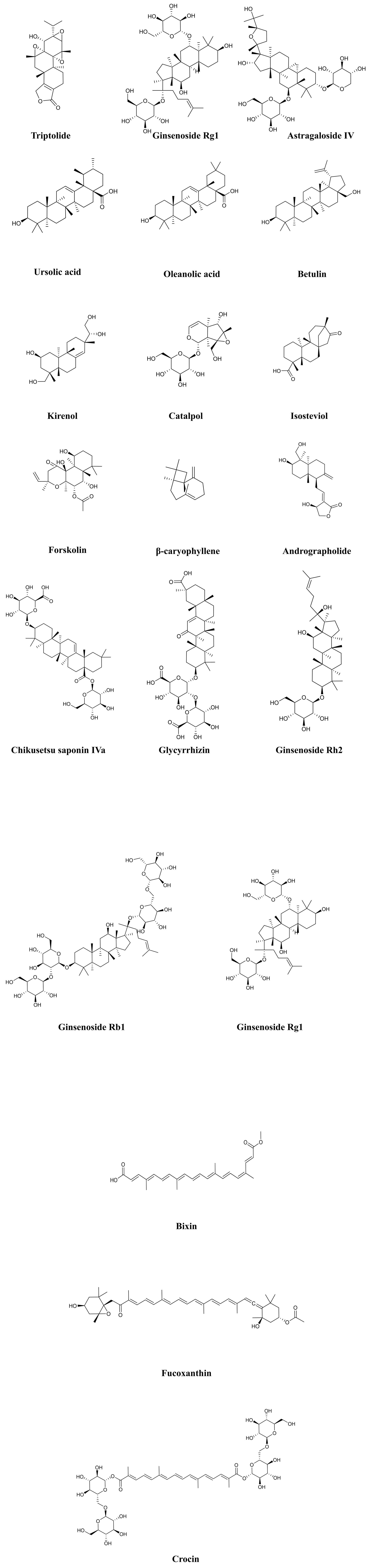 Fig. 3.
Fig. 3.Chemical structures of considered terpenoids. Note: Due to the complex chemical structure of terpenoids, we have rearranged the order.
Traditional herbal medicine (THM) provides a fertile ground for modern drug development, and triptolide is one of the “poster children” that exemplifies the potential and promise of transforming THM into modern drugs [151].
The loss of metabolic flexibility leads to a decrease in the utilization of
cardiac matrix and the efficiency of ATP production in DM patients. Triptolide
therapy improved cardiac function and increased cardiac energy metabolism,
through up-regulation of MAPK signaling transduction in vivo [121]. The
activation of NF-
Ginsenosides derived from the roots and rhizomes of Panax ginseng C. A. Meyer, have been utilized as an adjuvant treatment for DM in China. Ginsenosides can provide myocardial protection in DM through anti-oxidant stress, improved cardiac function, attenuated myocardial fibrosis, and reduced apoptosis [152].
Ginsenoside Rb1 is the most abundant triterpenoid saponin, which belongs to ginsenoside type A. One study has suggested that ginsenoside Rb1 could serve as a viable adjunctive therapeutic agent for DCM. The activity of RyR2 and SERCA 2a was regulated by Ginsenoside Rb1, which improved calcium signaling [124]. Adipocytokines are secreted from adipose tissue, which play critical roles in diabetes and obesity. Ginsenoside Rb1 reduced lipid levels through apoptocytokine signaling and reduced oxidative stress, hypertrophy, inflammation, fibrosis, and apoptosis in DCM [125].
Ginsenoside Rg1, which belongs to ginsentriol type B, has significant myocardial protective effect of DCM and its efficacy was associated with reduced oxidative stress and attenuated myocardial apoptosis [126]. Ginsenoside Rg1 treatment attenuated diabetic myocardial damage in DCM by reducing ERS-induced apoptosis [127].
Ginsenoside Rh2, which belongs to the ginsenodiol saponins, is suitable for the
development of an alternative remedy for myocardial fibrosis. Research has
indicated its effectiveness in improving cardiac function and fibrosis, through
increasing PPAR
Astragaloside IV (AS-IV), one of the main compounds from Astragalus membranaceus var. mongholicus (Bunge) P. K. Hsiao, is a cycloartane-type triterpene glycoside chemical.
The changes in metabolic pattern affect the cardiac remodeling and functional
change. Astragaloside IV can prevent myocardial injury caused by T2DM, and its
mechanism may involve improving myocardial lipid metabolism [129]. In addition,
astragaloside IV can regulate the release of peroxisome proliferator-activated
receptor-
The light chain 3 (LC-3) plays an important role in autophagy and is used as a molecular marker of autophagy. B-cell lymphoma-2 (Bcl2), a protein that also plays a significant role in autophagy, interacts with a variety of co-factors to trigger a cascade of autophagy proteins. Astragaloside IV inhibits HG-induced oxidative stress and autophagy, through the miR-34a/Bcl2/(LC3II/LC3I) and phosphorylated-Serine473-AKT (pAKT)/Bcl2/(LC3II/LC3I) pathways in vitro [131].
Crocin, an abundant anti-oxidant ingredient of Crocus sativus L. (saffron), exhibits significant protective effects against myocardial injury, especially in DCM. Crocin enhances cardiac dysfunction by restoring autophagy and preventing apoptosis in DCM [132]. Crocin resulted in a higher increase of anti-oxidant levels and a more reduced lipid peroxidation rate (malondialdehyde (MDA) content) in the heart of T2DM, and it was also revealed that a combination of crocin with voluntary exercise was more effective than crocin therapy alone [133].
Crocin, as an anti-oxidant compound, protects the myocardium against diabetes
complications through activation of PPAR
Ursolic acid is a natural pentacyclic triterpenoid, which possesses diverse
pharmacological actions. Ursolic acid is capable of improving the cardiac
structure and function in vivo by attenuating oxidative stress, inflammation, and
fibrosis [135]. In addition, ursolic acid had an obvious protective effect on
myocardial injury in DCM, and its mechanism may be associated with the inhibition
of NLRP3 inflammasome activation, reduced IL-1
Glycyrrhizin, also called glycyrrhizic acid, is a triterpenoid saponin mainly
isolated from Glycyrrhiza uralensis Fisch. Glycyrrhizin has presented
cardioprotective effects in diabetic cardiac atrophy, which may be mediated
through activation of Nrf-2 and inhibition of C-X-C chemokine receptor type 4
(CXCR4)/stromal cell-derived factor-1 (SDF1) as well as the TGF-
Fucoxanthin, as the natural product of carotenoids, can potentially be obtained
from marine algae. The NIP3-like protein X (Nix) is a key protein for mitophagy
during the maturation of reticulocytes. Fucoxanthin reduced the accumulation of
TGF-
Oleanolic acid is a naturally occurring pentacyclic triterpenoid that is widely distributed in plants. Treatment with oleanolic acid blunted HG-induced oxidative stress, apoptosis, and the ubiquitin–proteasome system in heart cells [154]. In recent years, the value of oleanolic acid in the field of DCM has been gradually explored, and its protective effect against cardiac injury caused by oxidative stress has been revealed. Glycogen synthase (GS) and glycogen phosphorylase (GP) are two key enzymes for glycogen synthesis and metabolism. Oleanolic acid protects against DCM, through the HO-1/Nrf-2 and GS/GP signaling pathways [141].
Chikusetsusaponin IVa is a natural product found in Swartzia simplex (Sw.) Spreng., Anredera baselloides (Kunth) Baill., and other plants. Chikusetsusaponin IVa protected cardiomyocytes from HG-triggered oxidative stress and calcium overload. The underlying mechanisms of Chikusetsusaponin IVa-mediated cardioprotection might be attributable to the regulation SIRT1/ERK1/2/Homer1a signaling pathways [142].
Betulin is a natural triterpenoid product contained in several medicinal plants,
including Betula platyphylla Sukaczev. Betulin significantly protected
against DCM by effectively improving insulin resistance, HG, and inflammation.
Research has shown that Betulin plays the heart-protective role described above
by regulating the SIRT1/NLRP/NF-
Kirenol is an ent-pimarane-type diterpenoid that has been reported from
Sigesbeckia orientalis L., Sigesbeckia glabrescens Makino,
Sigesbeckia pubescens Makino, and others. The Goto–Kakizaki (GK) rat is
a non-obese, non-hypertensive model of T2DM, like humans, it has a susceptibility
locus on chromosome 10. The cardioprotective effect of kirenol in GK rats is
mediated by regulation of the NF-
Catalpol is an iridoid glycoside extracted from the roots of Rehmannia glutinosa (Gaertn.) Libosch. ex Fisch. & C. A. Mey. Catalpol might stimulate the Neat1/miR-140-5p/Histone deacetylase 4 (HDAC4) signaling pathways, thereby leading to inhibition of HG-induced myocardial apoptosis [145].
Isosteviol, an ent-beyerane diterpenoid found in Stevia rebaudiana
(Bertoni) Bertoni, has been repeatedly reported as possessing potent
cardioprotective activity. Isosteviol sodium (STVNa) is an improved formulation
with higher solubility and bioavailability, which therapeutic effect is achieved
by reducing oxidative stress and inflammation in DCM. The mechanism is based on
inhibiting ERK and NF-
Bixin, a natural carotenoid extracted from Bixa orellana L., possesses anti-oxidant stress and anti-inflammatory effects. Bixin might be a novel and protective agent with therapeutic activity against DCM which acts by suppressing fibrosis, anti-inflammatory and anti-oxidative stress. Its related intervention mechanism is mediated by Nrf-2 signaling pathway activation [147].
Andrographolide is a labdane diterpenoid that is produced by the plant Andrographis paniculata. Andrographolide treatment exerts cardioprotective effects through modulation of the NADPH oxidase (NOX)/Nrf-2 signaling pathway. The therapeutic potential of andrographolide in the treatment of DCM is demonstrated by its ability to attenuation oxidative stress, inflammation, and apoptosis [149].
Forskolin is a labdane diterpenoid isolated from the Indian Coleus plant Coleus forskohlii (Willd.) Briq. Forskolin treatment in DCM significantly blocked oxidative stress and reduced myocardial fibrosis [150]. However, the specific signaling pathway mechanism underlying the role of forskolin remains to be elucidated.
Alkaloids is an extensive group of secondary metabolites, containing more than 12,000 different compounds [155]. Alkaloids are generally extracted from plants of the Ranunculaceae, Papaveraceae, Apocynaceae, Rutaceae, Fangke, Solanaceae, Leguminosae, and Polygonaceae families. Of course, they are also found in some animals [156]. Alkaloids’ chemical backbones have the potential to engage in interactions with an extensive array of proteins pertaining to glucose homeostasis. This has made them a highly visible and reliable candidate in the field of diabetes drug discovery, which is receiving increasing attention [157]. Some alkaloids can intervene in the insulin signal transduction pathway, reverse molecular defects resulting in insulin resistance and glucose intolerance [158]. Along with the in-depth research and application of alkaloids in the field of diabetes, the value of alkaloids in the treatment of DCM has become increasingly apparent. A total of 7 alkaloids were found to effective therapeutic intervention effects in the context of DCM. Table 3 (Ref. [159, 160, 161, 162, 163, 164, 165, 166, 167, 168, 169, 170, 171, 172, 173]) provides the basic information and mechanisms of the 7 alkaloids from recent studies on DCM, while Fig. 4 shows the chemical structures of 7 alkaloids.
| Numbers | Terpenoid Subclass | Compounds | Molecular formula | Weight (g/mol) | Resources | Animal/Cell model | Dosage (mg/kg/d; µm) | Dosing cycle | Target/Pathways/Mechanism | Effects | Reference |
| 1 | Isoquinoline alkaloid | Berberine | C |
336.4 | Coptis chinensis Franch. | HG-induced H9c2 cells | 40 | 4 weeks | Activation of AMPK signaling pathway | Improved insulin resistance | [159] |
| HFD + STZ-induced diabetic rats; HG-induced adult rat neonatal cardiac fibroblasts | 200; 12.5, 25, 50, 100 | 4 weeks; 24 h | Down-regulating myocardial IGF-1 receptor-regulated MMP-2/MMP-9 expression | Decreased myocardial fibrosis, alleviated cardiac diastolic and systolic dysfunction | [160] | ||||||
| STZ-induced diabetic rats | 50, 100, 150 | 12 weeks | Down-regulation of the expression of TGF- |
Decreased myocardial fibrosis | [161] | ||||||
| HFD + STZ-induced diabetic rats; palmitate-induced H9c2 cells | 100; 10 | 16 weeks; 48 h | Activation of 5 |
Decreased myocardial fibrosis and hypertrophy | [162] | ||||||
| HS, HFD + STZ-induced diabetic rats | 30, 200 | 4, 10, 16, 22 weeks | Regulated PGC-1 |
Decreased myocardial fibrosis, alleviated cardiac diastolic and systolic dysfunction, regulated lipid metabolism disorders | [163] | ||||||
| 2 | Pyridine alkaloid | Matrine | C |
248.36 | Sophora flavescens Aiton | STZ-induced diabetic rats; HG-induced mouse myocardial cells | 5 | 10 weeks | Down-regulation of the TGF‐ |
Anti-inflammatory, ameliorated apoptotic | [164] |
| STZ-induced diabetic rats; Primary cardiac fibroblasts from LV in rats | 200; 0.25, 0.5, 1.0, 1.5, 2.0, 2.5 mmol/L | 10 days (drug administered before molding); 48 h | suppression activation of ATF6/calreticulin/NFAT signaling pathway | Decreased myocardial fibrosis, inhibited ECM synthesis | [165] | ||||||
| STZ-induced diabetic rats | 200 | 10 days (drug administered before molding) | Suppression TLR-4/MyD-88/caspase-8/caspase-3 signaling pathway, suppression ROS/TLR-4 signaling pathway | Anti-oxidative stress, decreased myocardial apoptosis | [166] | ||||||
| STZ-induced diabetic rats | 300 | 10 days (drug administered before molding) | Inhibition of TGF- |
Decreased myocardial fibrosis | [167] | ||||||
| AGEs-induced diabetic rats | 50, 100, 200; 0, 0.5, 1.0, 2.0 mmol/L | 20 days; 24 h | Reduced ryanodine receptor 2 activity | Decreased cardiac apoptosis, attenuated calcium overload | [168] | ||||||
| 3 | Organic amine alkaloids | Betanin | C |
550.5 | Beta vulgaris L. | High fructose-induced diabetic rats | 25, 100 | 60 days | Inhibition of TGF- |
Decreased myocardial fibrosis | [169] |
| 4 | Quinolizidine alkaloid | Sophocarpine | C |
246.35 | Sophora flavescens Aiton, Styphnolobium japonicum (L.) Schott, etc. | STZ-induced diabetic mice; HG-induced H9c2 cells | 20; 0.01–10 mM | 16 weeks; 48, 96 h | Suppression NF- |
Anti-inflammatory, decreased cardiac apoptosis | [170] |
| 5 | Organic amine alkaloids | Capsaicin | C |
305.4 | Capsicum annuum L. | STZ-induced diabetic rats; HG-induced mouse vascular endothelial cells | 5; 1 | 8 weeks; 24 h | Up-regulated TRPV1/eNOS signaling pathway | Anti-oxidative stress, decreased myocardial fibrosis, ameliorated apoptosis | [171] |
| 6 | Pyridine alkaloid | Piperine | C |
285.34 | Piper nigrum L. | STZ-induced diabetic rats; HG-induced H9c2 cells | 10, 20, 40 | 4 weeks | Regulated Bcl2, Bax/Bcl2, and caspase-3 signaling pathway | Anti-oxidative stress, decreased myocardial apoptosis | [172] |
| 7 | Isoquinoline alkaloid | Sinomenine | C |
329.4 | Sinomenium acutum (Thunb.) Rehd et Wils. (S. acutum), Sinomenium acutum (Thunb.) Rehd. et Wils. var. cinereum Rehd. et Wils, etc. | STZ-induced diabetic rats | 30, 60, 120 | 10 weeks | Deactivation of NF- |
Anti-inflammatory | [173] |
AGE, advanced glycation end-product; ATF6, activating transcription factor 6;
AMPK, AMP activated protein kinase; Bax, BCL2-Associated X; Bcl2, B-cell
lymphoma-2; CTGF, connective tissue growth factor; eNOS, endothelial nitric oxide
synthase; HFD, high-fat diet; HG, hyperglycemia; LV, left ventricles; MMP-2,
matrix metalloproteinase-2; MMP-9 , matrix metalloproteinase-9; MyD88, myeloid
differentiation factor 88; NFAT, nuclear factor of activated T cells;
NF-
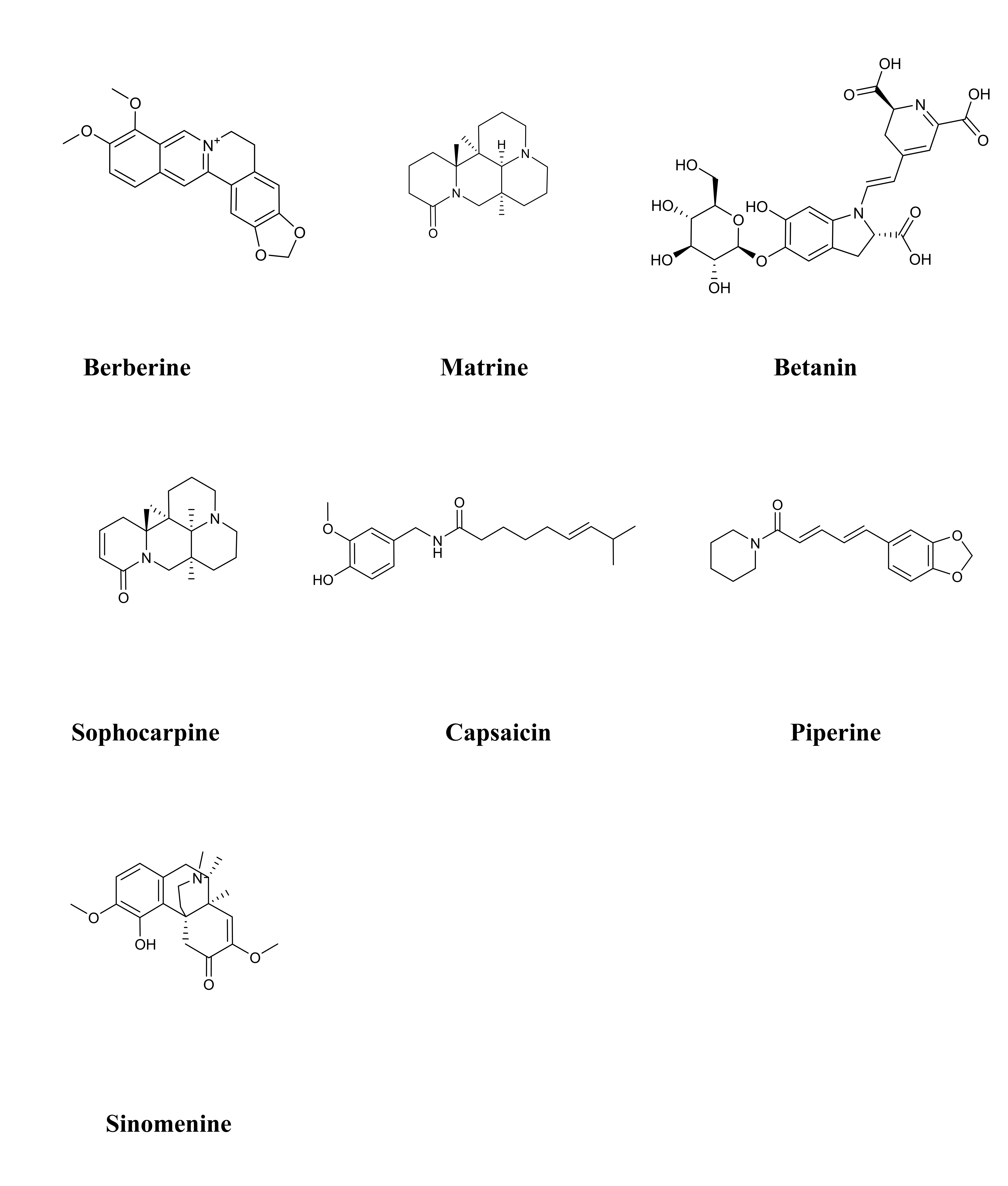 Fig. 4.
Fig. 4.Chemical structures of considered alkaloids.
Coptis chinensis Franch. is a THM that has been frequently used in many TCM formulas for the treatment of DM for thousands of years [174]. Berberine, the main active component of Coptis chinensis Franch., has been shown to have potential for the treatment of DM and its complications.
Insulin resistance is one of the most important risk factors for DCM. Berberine
can improve blood sugar status via increasing insulin sensitivity in peripheral
tissues. Berberine improves insulin resistance in cardiomyocytes, by increased
AMPK signaling pathway activity [159]. The MMPs family is a group of enzymes
involved in ECM degradation. Berberine down-regulated insulin-like growth
factor-1 (IGF-1) receptor expression and MMP-2/MMP-9 levels in cardiac
fibroblasts, suggesting a novel mechanism for anti-fibrotic cardioprotection of
berberine in DCM [160]. In additon, berberine improves myocardial fibrosis
through suppressing of TGF-
Matrine is a bioactive component of THM, such as Sophora flavescens and Radix Sophorae tonkinensis. Emerging evidence has suggested that matrine possesses anti-inflammatory, anti-oxidant stress, anti-fibrotic, anti-allergic, anti-nociceptive, hepatoprotective, cardioprotective, and neuroprotective properties [175].
The protein kinase RNA-like endoplasmic reticulum kinase (PERK) signaling
pathway plays a role in ERS-mediated apoptosis. Matrine could serve as a
potential anti-inflammatory and anti-apoptosis agent in the pathological
processes of DCM through down-regulation of the TGF-
Betanin is a water-soluble alkaloid extracted from Beta vulgaris L.
Anti-myocardial fibrosis is its key effect in DCM treatment. Betain showed
significant antifibrotic effects on myocardium, which is related to inhibition of
NF-
Sophocarpine is a natural quinolizidine alkaloid derived from Sophora
flavescens Aiton, Styphnolobium japonicum (L.) Schott, and other
plants. Sophocarpine may be effective against DCM as it can suppress inflammation
and inhibit the NF-
Capsaicin is a natural protoalkaloid, derived from Capsicum annuum L. Capsaicin might protect against HG-induced endothelial dysfunction and DCM through the transient receptor potential vanilloid 1 (TRPV1)/eNOS signaling pathway in DCM [171].
Piperine is the source of the distinctive sharp flavor of Piper nigrum L. The therapeutic effects of piperine on DCM are mediated by regulation of the caspase-3, Bcl2, and Bax/Bcl2 signaling pathways. Piperine attenuates STZ-induced DCM by reducing oxidative stress, maintaining the activity of mitochondria, and preventing apoptosis [172].
Sinomenine is one of the most widely known alkaloids, due to its prominent
anti-inflammatory activities. Sinomenine significantly improved cardiac function,
which attributed to the de-activation of NF-
Quinones are a class of compounds widely distributed in nature, being found in a wide variety of plants as well as fungi, bacteria, and animals. Quinones are molecules comprised of a basic benzoquinone chromophore, which is an unsaturated cyclic structure with two carbonyl groups [177]. Natural quinones can be mainly divided into benzoquinones, naphthoquinones, anthraquinones, and phenanthraquinones [178]. Quinones have been reported to exhibit numerous biological activities, such as cardioprotective, antidiabetic, hepatoprotective, neuroprotective, anti-cancer, anti-inflammatory, trypanocidal, anti-viral, anti-tubercular, anti-fungal, anti-bacterial anti-filarial, anti-malarial, and so on [179]. A total of 3 quinones have been reported as having effective therapeutic intervention effects in DCM. Table 4 (Ref. [180, 181, 182, 183, 184]) provides the basic information and mechanisms of the three quinones from recent studies on DCM, while Fig. 5 shows the chemical structures of the three quinones.
| Number | Terpenoid Subclass | Compounds | Molecular formula | Weight (g/mol) | Resources | Animal/Cell model | Dosage (mg/kg/d; µm) | Dosing cycle | Target/Pathways/Mechanism | Effects | Reference |
| 1 | Benzoquinone | Thymoquinone | C |
164.2 | Nigella damascena L. | STZ-induced diabetic rats; HG-induced H9c2 cells | 50 | 12 weeks | Up-regulation of Nrf-2 signaling pathways | Anti-oxidative stress, anti-inflammatory | [180] |
| STZ-induced diabetic rats | 50 | 30 days | Up-regulation of PI3K/AKT signaling pathways | Anti-oxidative stress, anti-inflammatory, decreased myocardial apoptosis | [181] | ||||||
| STZ-induced diabetic rats | 20 | 5 weeks | / | Anti-oxidative stress, anti-inflammatory | [182] | ||||||
| 2 | Anthraquinone | Emodin | C |
270.24 | Rheum palmatum L., Reynoutria japonica Houtt., and Pleuropterus multiflorus (Thunb.) Nakai. | High fructose + HFD + STZ-induced diabetic rats | 50, 100 | 16 weeks | Up-regulation of AKT/GSK-3 |
Regulated glycolipid metabolism | [183] |
| 3 | Anthraquinone | chrysophanol | C |
254.24 | Rheum palmatum L., Senna tora (L.) Roxb., Aloe vera (L.) Burm. f., etc. | HFD-induced Nrf-2-knockout (Nrf-2-/-) DCM mice, HG-induced H9c2 cells | 25, 50; ① 0, 10, 20, 40, 80, 160 , 320; ② 320 | 20 weeks; ① 24 h, ② 0, 6,12, 24, 36, 48, and 72 h | Up-regulation of Nrf-2 signaling pathways | Decreased myocardial fibrosis, anti-oxidative stress, anti-inflammatory | [184] |
①, ②: This distinction is made because the study used two dosing methods
and contents for cellular intervention. Treatment of H9c2 cells with different concentrations of
chrysophanol as indicated (0, 10, 20, 40, 80, 160 and 320 µm) for 24 h. In addition, the cells
were cultured with chrysophanol at 320 µm for different time (0, 6, 12, 24, 36, 48 and 72 h).
After various treatments, all cells were harvested for cell viability.
AKT, protein kinase B; GSK-3
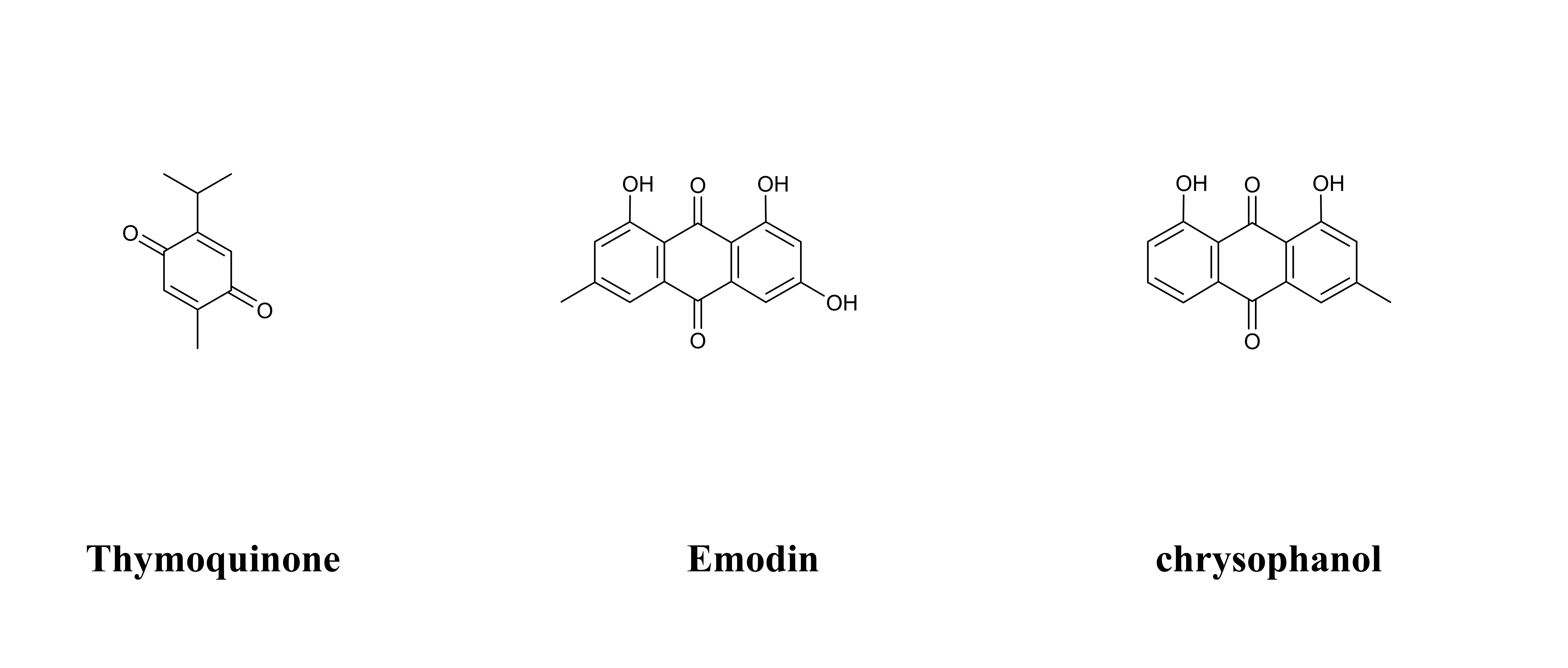 Fig. 5.
Fig. 5.Chemical structures of considered quinones.
Thymoquinone, a phytochemical compound obtained from Nigella sativa,
has received attention for its anti-inflammatory, analgesic, anti-cancer,
anti-oxidant, and anti-pyretic activities [185]. Thymoquinone diminished
oxidative damage by improving the anti-oxidant power of cardiac muscle,
consequently protecting the cardiac muscles and alleviating the inflammatory
process. The mechanism of action in this research was mainly through
up-regulation of Nrf-2 signaling pathways [180]. The protective impact of
thymoquinone enhances cardiovascular performance while mitigating oxidative
stress, inflammation, and apoptosis through mediation of the PI3K/Akt signaling
pathway [181]. Thymoquinone is a pharmacological agent that has potential for the
treatment of DCM, and its healing power further increases even more when combined
with
Emodin is a natural anthraquinone derivative that occurs in many widely used
herbs, such as Rheum palmatum L., Reynoutria japonica Houtt.,
and Pleuropterus multiflorus (Thunb.) Nakai. DM and its cardiovascular
complications are closely related to impairment of the AKT/GSK-3
Chrysophanol is a naturally occurring anthraquinone found in various herbs, including Rheum palmatum L., Senna tora (L.) Roxb., and Aloe vera (L.) Burm. f. The anti-oxidant stress, anti-inflammatory, and anti-fibrosis effects of chrysophanol can be explained by its regulation of Nrf-2 signaling pathway in DCM [184].
In addition to the flavonoids, terpenoids, alkaloids, and quinones discussed above, many other kinds of natural products can also be used to treat DCM. Table 5 (Ref. [186, 187, 188, 189, 190, 191, 192]) provide the basic information and mechanisms of seven additional natural compounds derived from recent studies on DCM, while Fig. 6 shows the chemical structures of the seven natural products.
| Number | Flavonoid Subclass | Compounds | Molecular formula | Weight (g/mol) | Resources | Animal/Cell model | Dosage (mg/kg/d; µm/24 h) | Dosing cycle | Target/Pathways/Mechanism | Effects | Reference |
| 1 | Glycoside | Apocynin | C |
166.17 | Iris tectorum Maxim., Cannabis sativa L., etc. | HFD + STZ-induced diabetic mice; HG-induced NRCMs and CFs | 10; 400 | 3 months, 48 h | Suppression of ASK1-p38/JNK signaling pathways | Anti-oxidative stress, decreased myocardial apoptosis, decreased myocardial fibrosis, attenuated cardiomyocyte hypertrophy | [186] |
| 2 | Glycoside | Gypenosides | C |
963.2 | Gynostemma pentaphyllum (Thunb.) Makino | STZ-induced diabetic rats; HG-induced H9c2 cells | 200; 100, 200, 400 mg/L | 8 weeks; 48 h | Inhibition of NLRP3 inflammasome activation | Anti-inflammatory | [187] |
| 3 | Glycoside | Sulforaphane | C |
177.3 | Brassica oleracea var. italica Plenck, Brassica oleracea L., etc. | HFD + STZ-induced diabetic mice | 0.5 | 3 months | Activation of AMPK/AKT/GSK3 |
Anti-oxidant stress, anti-inflammatory, decreased myocardial fibrosis and hypertrophy | [188] |
| 4 | Glycoside | Polydatin | C |
390.4 | Reynoutria japonica Houtt. | Sirt3 knockout (Sirt3-/-) mice; HG-induced primary neonatal mouse ventricular cardiomyocyte | 7.5; 10 | 28 d; 48 h | Up-regulated SIRT3 signaling pathway | Increased autophagy, improved mitochondrial bioenergetics | [189] |
| 5 | Glycoside | Mangiferin | C |
422.3 | Anemarrhena asphodeloides Bunge, Mangifera indica L., etc. | HFD + STZ-induced diabetic rats | 20 | 16 weeks | De-activation of NF- |
Anti-inflammatory, decreased myocardial fibrosis | [190] |
| 6 | Phenylpropanoid | Salidroside | C |
300.3 | Rhodiola rosea L. | Diabetic db/db mice; AGEs-induced H9c2 cells | 0.025, 0.05; 0.1, 1, 10 | 12 weeks; 6 h | Activation of AKT/Nrf-2/HO-1 signaling pathway | Decreased myocardial fibrosis, decreased cardiac apoptosis | [191] |
| 7 | Phenylpropanoid | Skimmin | C |
324.28 | Artemisia caruifolia Buch.-Ham. ex Roxb., Astragalus membranaceus var. mongholicus (Bunge) P. K. Hsiao, etc. | STZ-induced diabetic rats; HG-induced primary neonatal cardiomyocytes from rats | 15, 30; 2, 10 | 16 weeks; 24 h | Suppression of NLRP3 inflammasome activation | Anti-inflammatory, anti-oxidative stress, increased autophagy, inhibited pyroptosis | [192] |
AKT, protein kinase B; ASK1, apoptosis signal regulating kinase 1; CFs, cardiac
fibroblasts; GSK-3
 Fig. 6.
Fig. 6.Chemical structures of other natural compounds.
Apocynin is a naturally occurring glycoside found in Iris tectorum Maxim. and Cannabis sativa L. Apocynin may act as a potential inhibitor of apoptosis signal regulating kinase (ASK), and attenuated cardiomyocyte hypertrophy, myocardial fibrosis, and cardiac dysfunction in by inhibiting the ASK1-p38/JNK signaling pathways [186].
Gypenosides are the main active ingredients of Gynostemma pentaphyllum (Thunb.) Makino, which is a TCM commonly used in China. Gypenoside inhibited HG-induced myocardial damage through anti-inflammatory effects. The mechanism of action may be the inhibition of NLRP3 inflammasome activation and NLRP3 [187].
Sulforaphane is mainly present in the sprouts of many cruciferous vegetables,
belonging to the isothiocyanate family. AMPK is indispensable for the
sulforaphane-induced prevention of cardiomyopathy in T2DM, and the activation of
Nrf-2 by sulforaphane is mediated by the AMPK/AKT/GSK3
Polydatin is a glycoside isolated from Reynoutria japonica Houtt. Polydatin improved cardiac dysfunction, increased autophagy flux, and regulated mitochondrial bioenergetics, by up-regulating Sirt3 signaling pathways in DCM [189].
Mangiferin, a bioactive glycoside compound present in mango, has been reported
to be valuable in treating DCM. Mangiferin ameliorated DCM by preventing the
release of inflammatory cytokines, inhibiting ROS accumulation, reducing AGE/RAGE
production, and regulating NF-
Salidroside is isolated from Rhodiola rosea L., which has been used for a long time as an adaptogen in TCM. Salidroside treatment protects against cardiomyocyte apoptosis and ventricular remodeling in the hearts of diabetic patients. This cardio-protective effect of salidroside is dependent on activation of the AKT/Nrf-2/HO-1 signaling pathways [191].
Skimmin, a natural coumarin derivative, has been shown to possess protective effects against experimental DCM. Skimmin has the potential to prevent DCM by reducing NLRP3 inflammasome activation and promoting autophagy in heart tissues, as well as potentially inhibiting pyroptosis [192].
A total of 72 natural compounds were discussed in this study, divided into five categories based on their chemical structure characteristics: Flavonoids, terpenoids, alkaloids, quinones, and others. Flavonoids are the largest group of natural products reported for the treatment of DCM, with a total of 36 flavonoids retrieved. The number of terpenoids in this study ranked second, with a total of 19 species. The quantitative distribution of various types of natural products also provides scope and guidance for the future exploration and development of new drugs for use in the treatment of DCM.
Various studies have found that the efficacy of a natural product in the
treatment of DCM mainly depends on its properties in terms of anti-oxidant
stress, anti-inflammatory, regulation of programmed cell death (including
apoptosis, necroptosis, pyroptosis, and autophagy), regulation of glucose and
lipid metabolism, regulation of Ca
Natural products have the advantages of being multi-pathway, multi-link, and
multi-target agents in the treatment of DCM, and can play various roles through
different signaling pathways. However, there is some commonality in the
intervention signaling pathways when treating the same disease. NF-
Overall, this paper provided a brief overview of the main directions of natural product research related to DCM to date, as well as pointing out promising avenues for future research. It must be noted that studies existing in the literature have been restricted to animal and cell experiments, with a lack of clinical research. Further clinical research utilizing natural products could provide more insight into the effectiveness of their complicated pharmacological properties, enabling natural products to be used safely and efficiently.
ADSC, adipose-derived stem cells; AGEs, advanced glycation end-products; AKT,
protein kinase B; AMPK, AMP activated protein kinase; ARE, antioxidant response
element; ASK1, apoptosis signal regulating kinase 1; ATF6, activating
transcription factor 6; ATP, adenosine triphosphate; Bax, BCL2-Associated X;
Bcl2, B-cell lymphoma-2; BNP, brain natriuretic peptide; CFs, cardiac
fibroblasts; cGMP, cyclic guanosine monophosphate; CHD, coronary heart disease;
CK-MB, serum creatine kinase MB isozyme; CRP, reactive protein; CTGF, connective
tissue growth factor; CVD, cardiovascular disease; CXCR4, C-X-C chemokine
receptor type 4; Cyt c, cytochrome c; DCM, diabetic cardiomyopathy; DKD, diabetic
kidney disease; DM, diabetes mellitus; EC, (-)-epicatechin; ECG,
(-)-epicatechin-3-gallate; ECM, extracellular matrix; ECs, endothelial cells; ED,
endothelial dysfunction; EETs, epoxyeicosatrienoic acids; EGC,
(-)-epigallocatechin; EGCG, (-)-epigallocatechin-3-gallate; eNOS, endothelial
nitric oxide synthase; ER, endoplasmic reticulum; ERK, extracellular
signal-regulated kinase; ERK1/2 , extracellular signal-regulated kinase1/2; ERS,
endoplasmic reticulum stress; ESC, European Society of Cardiology; FA, fatty
acid; FFA, free fatty acids; GP, glycogen phosphorylase; GS, glycogen synthase;
GSK-3
PYY, XNY, and YQ concepted and performed this study. XNY, and YQ designed the study and revised the manuscript. All authors participated in writing or revising the manuscript. All authors read and approved the final manuscript. All authors have agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research was supported by the National Natural Science Foundation of China (No: 81302940), Shandong Natural Science Foundation (No: ZR2021MH386, ZR2021LZY028), and Special Fund of TCM High-level Talents Cultivation Project of Shandong Province (No. 143).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






