1 Cardiology Department, Zagazig University, 44519 Zagazig, Egypt
Abstract
Background: The most common cause of coronary artery diseases (CAD) is
atherosclerosis. The synergy between percutaneous coronary intervention with
TAXUS™ and cardiac surgery (SYNTAX) score was used to assess
complex CAD lesions. The renal resistive index (RRI) is a Doppler ultrasound
parameter calculated to assess renal haemodynamics. The direct relationship
between CAD complexity and RRI was not yet investigated. The aim of our study was
to investigate this relationship between RRI and SYNTAX score in stable CAD
patients. Methods: This study included 214 patients with stable CAD and
subsequent coronary angiography done at our institution. Regarding CAD
complexity, these patients were classified into 166 patients with low SYNTAX
score (SYNTAX
Keywords
- coronary artery disease
- renal resistive index
- SYNTAX score
Cardiovascular diseases (CVD) are the commonest cause of morbidity and mortality worldwide. The commonest underlying mechanism of coronary artery diseases (CAD) is atherosclerosis. Atherosclerotic plaques develop over years leading to a long-term clinically silent coronary obstruction, up to sudden plaque rupture or erosion with subsequent development of acute coronary syndrome (ACS) [1].
Chronic coronary syndromes (CCS), unlike ST-segment elevation myocardial infarction (STEMI), have different predictors of long-term outcomes [2] including left ventricular ejection fraction (LVEF), age, complete revascularization [3], associated acute left ventricular failure, peak troponin level and degree of ST-segment deviation [4]. Also, chronic kidney diseases (CKD) are increasingly becoming a key predictor of cardiac morbidity and mortality [5].
The synergy between percutaneous coronary intervention with TAXUS™ and cardiac surgery (SYNTAX) score was calculated to evaluate both CAD extent and complexity. Moreover, this score could be considered as a strong predictor of major adverse cardiac events (MACE) [6] and cardiovascular death in patients subjected to percutaneous coronary intervention (PCI) [7]. Each coronary lesion could be scored separately using SYNTAX scoring system, and then the total SYNTAX score is achieved by summing all these calculated scores.
The cardio-renal syndrome is the coexistence of both cardiac and renal pathology, and is related to both humoral and neural signaling [8]. Analysis of micro- and macrovascular circulatory parameters could be used for early detection of CVD and related vascular damage. Among these parameters, it was found that renal resistive index (RRI) could be considered as the most reproducible and clinically relevant index of renal hemodynamics and vascular stiffness [9].
RRI is a simple non-invasive renal Doppler Ultrasonography (USG) parameter reflecting renal haemodynamic changes and allowing assessment of renal vascular damage and resistance [10]. Also, RRI was dependent on numerous factors including renal vascular stiffness, age, brady- and tachyarrhythmias, pulse pressure, significant valvular lesions and pathological lesions within renal parenchyma [11].
High RRI was found to be correlated with impaired renal haemodynamics [12]. This could be used to allow accurate prediction of the onset of acute kidney injury and its persistence [13], arterial remodeling and stiffness [9], and adverse CVD events in elderly patients especially those having a hypertensive renal disease [14]. Moreover, in a large cohort enrolling CKD patients, high RRI was significantly associated with increased mortality [15].
Of note, to our knowledge, none of previously published studies assessed the relationship between RRI and CAD. In our study, the aim was to prove the hypothesis that using intra-renal flow parameters may define complex CAD in stable CAD patients by evaluating the association between RRI and extent and complexity of CAD, defined by SYNTAX score.
Between February 2021 and August 2022, a total of 254 consecutive patients with the inclusion criteria of CCS [16] and a clinical indication for coronary angiogram based on former non-invasive stress tests, have been included in this cross-sectional study with subsequent coronary angiography done at our hospital.
Patients with pulmonary edema, cardiogenic shock, any type of respiratory
failure, previous coronary artery bypass grafting (CABG), CKD defined as
proteinuria level
Finally, 214 patients were enrolled and classified according to the CAD
severity; revealed by coronary angiogram and assessed by SYNTAX score, into two
main groups: group (1) including 166 patients with low SYNTAX score (SYNTAX
Baseline demographics and clinical characteristics of all included patients were
obtained from our hospital records. Cardiovascular risk factors of all patients
were identified. The diagnostic criteria of diabetes mellitus were confirmed in
all patients receiving active treatment or having an abnormal fasting glucose
(
Venous blood samples were withdrawn from all patients on admission prior to
coronary angiography. The following laboratory parameters were obtained from all
patients: complete blood count, lipid panel, serum albumin level, and creatinine
level. The eGFR was measured according to the abbreviated Modification of Diet in
Renal Disease (aMDRD) equation: aMDRD = 186
A resting ECG was obtained all patients on admission. Transthoracic echocardiography (TTE) was done before coronary angiography by an experienced blinded cardiologist according to the current practice guidelines [20] to assess the diastolic functions and LVEF by the modified Simpson’s rule.
Coronary angiography was done using Siemens (Axiom Sensis XP, Berlin, Germany) device at our catheterization laboratory via either radial or femoral access using 6 or 7 Fr sheaths and catheters with administrating 0.2 mg of intracoronary nitroglycerin. Multiple projections were used for adequate analysis of target lesions characteristics by two experienced blinded interventional cardiologists.
According to lesion classification system based on the American College of
Cardiology/American Heart Association (ACC/AHA) [21], patients with left main
coronary artery (LMCA) stenosis
All included patients underwent the renal ultrasound study using a 5C probe (4.4–6.7 MHz). The Doppler analysis of blood flow indices within arcuate or interlobular renal arteries was done by an experienced operator. After 6 hours of fasting, the patients were evaluated after resting for at least 20 minutesin supine position and the following parameters were assessed: (a) the morphology of kidneys to obtain their length, width, parenchymal thickness and potential structural abnormalities requiring further evaluation, (b) renal artery hemodynamics screening using peak systolic velocity (PSV) and end-diastolic flow velocity (EDV) within the main renal artery, and (c) mean velocity (MV), acceleration time (AT), augmentation index (AI) of intra-renal arteries using a 2–4 mm pulsed wave Doppler.
These mentioned Doppler parameters were used to calculate both RRI and renal pulsatility index (RPI) based on these formulas: RRI = (PSV – EDV)/PSV and RPI = (PSV – EDV)/MV. These indices were obtained 3 times involving the interlobular renal arteries located in upper, mid and lower kidney poles using a pulsed-wave Doppler with the calculation of arithmetic means of these measurements [24].
Data distribution was first assessed using the Kolgormonov-Smirnov test. Then,
categorical data were compared based on the chi-square test or Fisher exact test.
Continuous variables were also compared according to an unpaired Student’s
t-test or Mann–Whitney U-test. Data were expressed as mean
A total of 214 patients with stable CAD underwent coronary angiography at our
institution were enrolled in the present study. Subsequently, out of these 214
patients, 166 patients had low SYNTAX score (SYNTAX
Baseline demographic data and clinical characteristics are shown in Table 1. This study population included 142 males (66.4%) and 72 females (33.6%). Out of high SYNTAX score group, 62.5% were males compared to 67.5% in low SYNTAX score group (p = 0.52). Similarly, no differences were significantly noted between two groups regarding the mean age of patients (p = 0.97).
| Variable | All Patients | SYNTAX |
SYNTAX |
p value | ||
| (n = 214) | (n = 166) | (n = 48) | ||||
| Clinical characteristics | ||||||
| Age, mean |
64.02 |
64.04 |
63.96 |
0.97 | ||
| Male sex, n (%) | 142 (66.4%) | 112 (67.5%) | 30 (62.5%) | 0.52 | ||
| Cardiovascular risk factors, n | ||||||
| Hypertension | 174 (81.3%) | 138 (83.1%) | 36 (75.0%) | 0.20 | ||
| Smoking | 104 (48.6%) | 73 (44.0%) | 31 (64.6%) | 0.01 | ||
| Dyslipidemia | 104 (48.6%) | 73 (44.0%) | 31 (64.6%) | 0.01 | ||
| Diabetes Mellitus | 100 (46.7%) | 69 (41.6%) | 31 (64.6%) | 0.005 | ||
| Family history of CAD | 80 (37.4%) | 58 (34.9%) | 22 (45.8%) | 0.17 | ||
| Obesity | 36 (16.8%) | 26 (15.7%) | 10 (20.8%) | 0.39 | ||
| BMI (kg/m |
26.1 |
25 |
27 |
0.27 | ||
| SBP, mmHg | 131.59 |
126.15 |
133.16 |
0.16 | ||
| DBP, mmHg | 72.43 |
67.73 |
73.80 |
0.03 | ||
| HR, bpm | 87.64 |
84.02 |
88.69 |
0.12 | ||
| LVEF, % | 55.00 |
58.20 |
51.80 |
0.12 | ||
| LV diastolic dysfunction | ||||||
| Grade 1 | 98 (45.8%) | 82 (49.4%) | 16 (33.3%) | |||
| Grade 2 | 90 (42.1%) | 67 (40.4%) | 23 (47.9%) | 0.09 | ||
| Grade 3 | 26 (12.1%) | 17 (10.2%) | 9 (18.8%) | |||
| Medications | ||||||
| Beta-blockers | 210 (98.1%) | 164 (98.8%) | 46 (95.8%) | 0.18 | ||
| CCBs | 204 (95.3%) | 158 (95.2%) | 46 (95.8%) | 0.85 | ||
| Nitrates | 168 (78.5%) | 134 (80.7%) | 34 (70.8%) | 0.14 | ||
| Aspirin | 166 (77.6%) | 126 (75.9%) | 40 (83.3%) | 0.28 | ||
| ACEIs/ARBs | 24 (11.2%) | 22 (13.3%) | 2 (4.2%) | 0.08 | ||
| Statins | 172 (80.4%) | 130 (78.3%) | 42 (87.5%) | 0.16 | ||
| Laboratory characteristics | ||||||
| Hemoglobin, gm/dL | 12.84 |
13.08 |
12.60 |
0.5 | ||
| Leukocytes ( |
8.14 |
7.56 |
8.72 |
0.24 | ||
| Platelet count ( |
226.50 |
225.18 |
231.08 |
0.63 | ||
| TGs, mg/mL | 155.02 |
156.14 |
151.13 |
0.58 | ||
| LDL-C, mg/dL | 129.08 |
122.55 |
151.64 |
|||
| HDL-C, mg/dL | 38.86 |
38.91 |
38.65 |
0.83 | ||
| Serum albumin, g/L | 35.73 |
35.56 |
36.28 |
0.11 | ||
| Creatinine, mg/dL | 1.02 |
1.03 |
1.01 |
0.82 | ||
| eGFR, mL/min/1.73 m |
94.01 |
91.37 |
103.17 |
0.13 | ||
ACEIs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin receptor blockers; bpm, beats per minutes; CAD, coronary artery disease; CCBs, calcium channel blockers; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; HDL-C, high density lipoprotein cholesterol; HR, heart rate; LDL-C, low density lipoprotein cholesterol; LV, left ventricle; LVEF, left ventricular ejection fraction; SBP, systolic blood pressure; TGs, triglycerides; BMI, body mass index; SYNTAX, percutaneous coronary intervention with TAXUS™ and cardiac surgery.
Compared to those with low SYNTAX scores, diabetes mellitus, dyslipidemia and smoking habit were more frequent in high SYNTAX scores patients (64.6% vs. 41.6%, p = 0.005 & 64.6% vs. 44.0%, p = 0.01 & 64.6% vs. 44.0%, p = 0.01, respectively). No significant differences were noticed regarding other demographic and clinical data between these two groups.
Laboratory features are shown in Table 1. Compared to low SYNTAX score group, it
was noticed that LDL-C level was significantly higher in patients included in
high SYNTAX score group (151.64
Angiographic features are shown in Table 2. The mean SYNTAX score value was
15.16
| Variable | All Patients | SYNTAX |
SYNTAX |
p value |
| (n = 214) | (n = 166) | (n = 48) | ||
| Angiographic characteristics | ||||
| Multi-vessel CAD | 101 (47.2%) | 71 (42.8%) | 30 (62.5%) | 0.02 |
| Left main CAD | 26 (12.1%) | 14 (8.4%) | 12 (25.0%) | 0.002 |
| SYNTAX score | 15.16 |
11.39 |
28.21 |
|
| Gensini score | 41.34 |
24.44 |
99.79 |
CAD, coronary artery disease; SYNTAX, percutaneous coronary intervention with TAXUS™ and cardiac surgery.
Of note, multi-vessel CAD was noted in 47.2% of all included patients, and it was more frequent in those with a higher SYNTAX score than those with a lower SYNTAX score (62.5% vs. 42.8%, p = 0.02). Similarly, left main CAD was noticed in 12.1% and its prevalence is significantly higher in high SYNTAX score group compared to that with low SYNTAX score (25.0% vs. 8.4%, p = 0.002).
Renal USG and Doppler characteristics are shown in Table 3. Regarding USG
characteristics, the mean values were 44.08
| Variable | All Patients | SYNTAX |
SYNTAX |
p value |
| (n = 214) | (n = 166) | (n = 48) | ||
| Renal USG characteristics | ||||
| Renal width, mm | 44.08 |
43.96 |
44.20 |
0.91 |
| Renal length, mm | 104.22 |
103.18 |
105.26 |
0.58 |
| Renal parenchymal thickness, mm | 14.51 |
14.48 |
14.54 |
0.95 |
| Aorta PS, cm/s | 65.57 |
66.96 |
59.51 |
0.12 |
| PSV, m/s | 57.46 |
57.48 |
57.36 |
0.99 |
| RPI | 1.37 |
1.38 |
1.34 |
0.79 |
| RRI | 0.632 |
0.614 |
0.699 |
PS, peak systolic; PSV, peak systolic velocity; RPI, renal pulsatile index; RRI, renal resisitive index; USG, ultrasonography; SYNTAX, percutaneous coronary intervention with TAXUS™ and cardiac surgery.
Regarding renal Doppler characteristics, there were no significant differences
between two SYNTAX groups regarding mean values of PSV (57.48
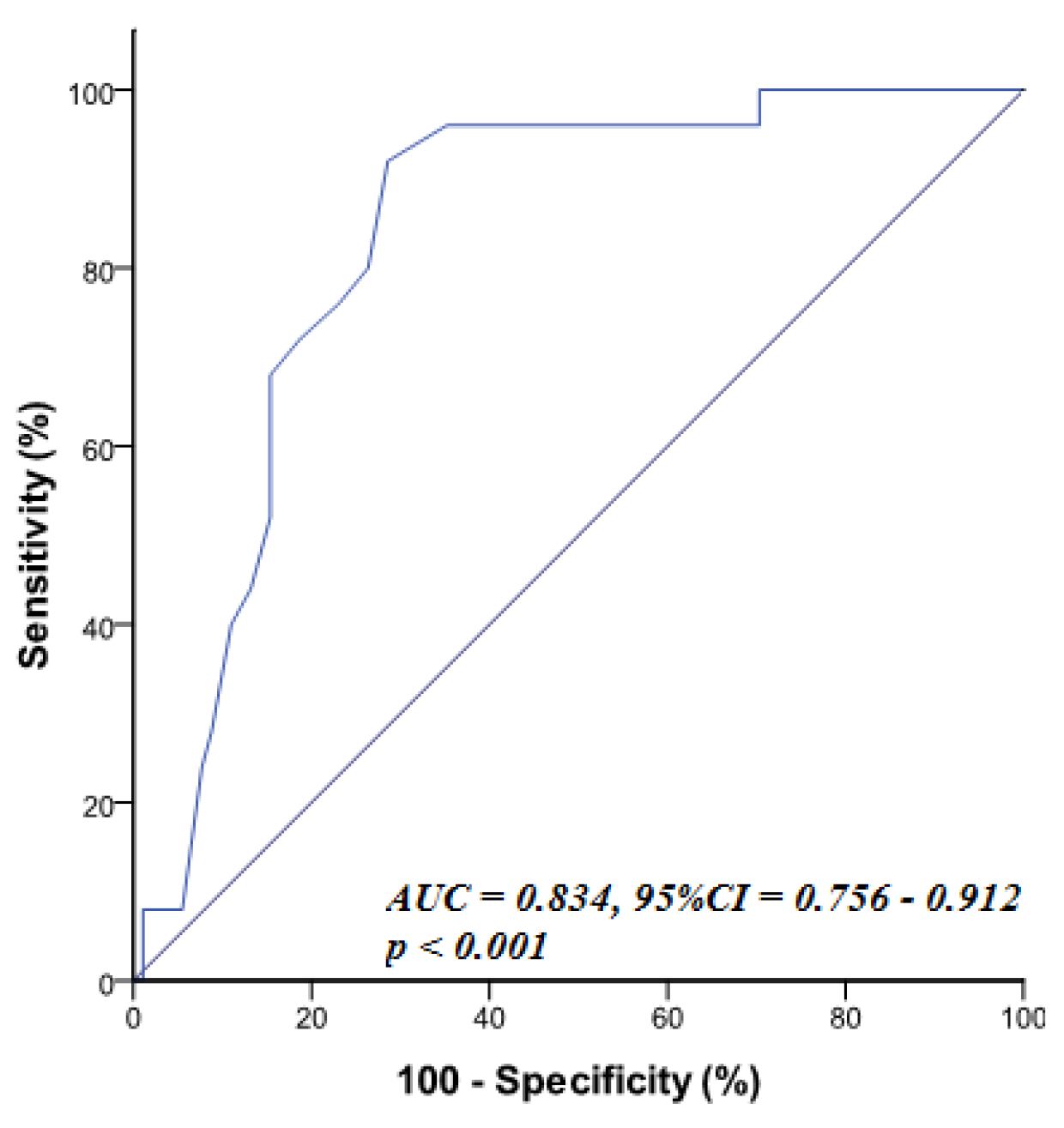
ROC statistical analyses (Table 4)
showed that RRI
| Variable | AUC | p-value | 95% CI | Cut off | Sensitivity | Specificity |
| RRI | 0.834 | 0.756–0.912 | 80.0% | 73.6% |
RRI, renal resistive index; SYNTAX, percutaneous coronary intervention with TAXUS™ and cardiac surgery; CAD, coronary artery disease; AUC, area under curve.
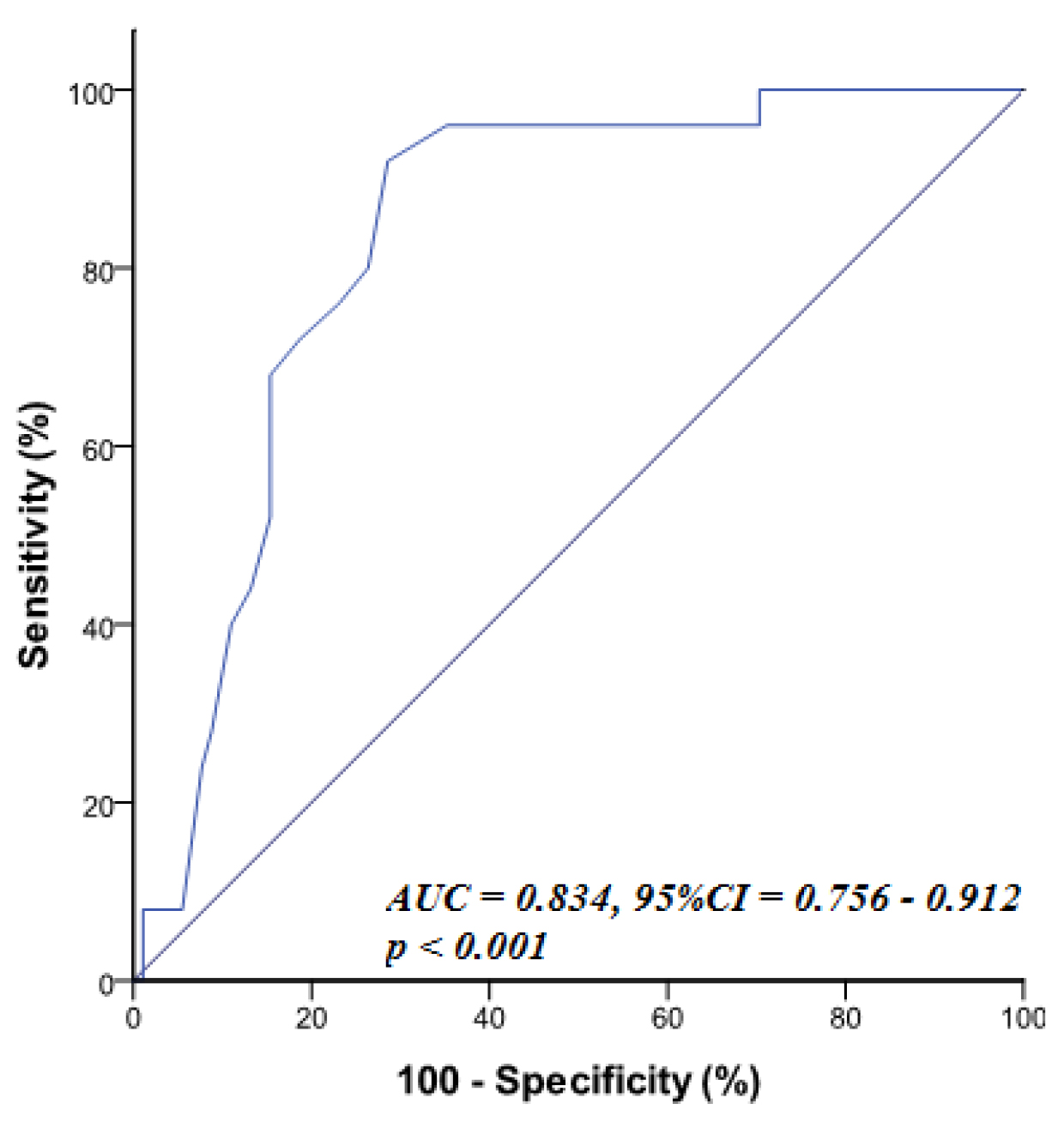 Fig. 1.
Fig. 1.ROC curve of RRI for prediction of high syntax score (SYNTAX
The independent predictors of complex CAD in stable CAD patients; defined by
SYNTAX score
| Variables | Univariate | Multivariate | ||
| p-value | OR | p-value | OR | |
| (95% CI) | (95% CI) | |||
| Diabetes mellitus | 0.005 | 2.564 | 0.040 | 4.401 |
| (1.315–4.996) | (1.081–17.923) | |||
| Smoking | 0.01 | 2.323 | 0.087 | 2.717 |
| (1.193–4.523) | (0.799–27.847) | |||
| LDL-C, mg/dL | 1.035 | 0.027 | 2.957 | |
| (1.020–1.050) | (1.920–8.995) | |||
| Multi-vessel CAD | 0.02 | 2.230 | 0.001 | 2.113 |
| (1.152–4.316) | (1.241–2.280) | |||
| Left main CAD | 0.002 | 3.619 | 0.279 | 0.874 |
| (1.543–8.487) | (0.527–3.176) | |||
| Gensini score | 3.073 | 0.002 | 6.539 | |
| (1.944–4.859) | (1.977–21.626) | |||
| RRI | 1.751 | 0.010 | 4.440 | |
| (3.628–8.450) | (1.418–13.903) | |||
CAD, coronary artery disease; LDL-C, low density lipoprotein cholesterol; RRI, renal resistive index; OR, odds ratio; SYNTAX, percutaneous coronary intervention with TAXUS™ and cardiac surgery.
In the multivariate logistic regression analysis, diabetes mellitus (odds ratio, OR = 4.401, 95% CI: 1.081–17.923, p = 0.04), LDL-C (OR = 2.957, 95% CI: 1.920–8.995, p = 0.027), multi-vessel CAD (OR = 2.113, 95% CI: 1.241–2.280, p = 0.001), Gensini score (OR = 6.539, 95% CI: 1.977–21.626, p = 0.002), and RRI (OR = 4.440, 95% CI: 1.418–13.903, p = 0.010) were found to be strong independent predictors of a higher SYNTAX score in stable CAD patients.
The main findings of our study were: (1) in stable CAD patients, diabetes
mellitus, dyslipidemia, and smoking were more frequent in those with high SYNTAX
score group (SYNTAX
Atherosclerosis is a chronic immune-inflammatory and fibro-proliferative disease with a systemic and progressive behaviour. It is heterogeneously distributed in coronary plaques and frequently affects peripheral vasculature as renal arteries in addition to aorta and coronary arteries. In coronary arteries, atherosclerosis is characterized by its fatty streaks in childhood. However, in adulthood, fibrous plaques are more noted with more complex and advanced coronary lesions [25]. Concurrently, the kidneys are characterized by their significant arterial vascular structure, and also are affected by atherosclerosis lie coronary arteries where it leads to both anatomical and functional effects in the kidneys [26].
Our study included 214 patients with stable CAD who had been admitted to our
catheterization laboratory to perform coronary angiography. According to
complexity of atherosclerotic coronary lesions defined by the SYNTAX score, these
patients were subsequently divided into those with SYNTAX score
Within the same context, the present study reported a higher frequency of
smoking (64.6% vs. 44%, p = 0.01) and diabetes mellitus
(64.6% vs. 41.6%, p = 0.005) among patients with stable CAD
and having complex coronary lesions (SYNTAX
RRI is a non-invasive renal Doppler modality used to assess renal hemodynamics. Moreover, RRI could be considered as a result of complex haemodynamic interactions involving both the kidneys and the systemic vasculature, and most of those interactions are not entirely understood yet. Kintis et al. [30] stated that RRI provides also a prognostic information regarding the systemic vasculature and could be considered as a promising marker for vascular damage, where high RRI could result in adverse clinical outcomes, especially in elderly, hypertensive, and diabetic patients.
This strong association between elevated RRI and systemic vascular damage was
reported in many other previous studies where Prejbisz et al. [31]
reported that a higher RRI was noticed in patients having a truly resistant
hypertension than those with a well controlled systemic hypertension (0.62
More recently, Geraci et al. [34] reported that the Doppler-based intra-renal flow parameters were greatly associated with severity of atherosclerotic lesions assessed in coronary angiography, but only noted among those patients having less pronounced atherosclerosis. Of note, this significant correlation was found to be valid for RPI, but not RRI, which needs further explanations. On this basis, there are no sufficient studies discussing the correlation between extent and severity of atherosclerotic CAD and RRI, and thus, we hypothesized in this study that RRI values determined by renal Doppler studies could be used to investigate renal vasculature and correlating it with the atherosclerotic CAD severity to assess the RRI value to predict its progression.
Accordingly, the present study shed light on the relationship between RRI and
the complex coronary lesions in patients with stable atherosclerotic CAD. It was
also found that RRI was higher in patients having more complex coronary lesions
(SYNTAX
However, our results came in contrast with Demirtaş and Bulut who reported
that RRI in the low-risk ACS patients (mean SYNTAX Score
The present study has a great clinical implication where it documents that RRI could be considered as a novel strong independent predictor of high SYNTAX score in stable atherosclerotic CAD, besides other traditional predictors as diabetes mellitus, LDL-C level, multi-vessel CAD, and Gensini score. In line with these findings, the cohort study stated by Pearce et al. [14] revealed a significant association between Doppler renal parameters; including RRI and RPI, and adverse cardiovascular events. Also, Akgul et al. [38] proved a relationship between RRI and both cardiac risk factors and carotid IMT. It was stated that a mild renal dysfunction has been also reported to be a strong independent risk factor for cardiovascular mortality, even after proper controlling of other atherosclerotic risk factors [39].
The main underlying mechanisms illustrating the higher SYNTAX score associated with high RRI values are still uncertain. However, this could be attributed to a histological study showing that the reno-vascular atherosclerosis is considered as independent risk factor for an increased RRI. Moreover, atherosclerosis is a systemic process, and its main pathophysiology is nearly the same in all involved patients [40]. Also, recent studies including hypertensive patients documented a significant correlation between arterial stiffness index [41], central pulse pressure, aortic stiffness [42] and RRI. Accordingly, the RRI could be a novel indicator of a systemic vascular injury rather than only a renal vascular damage. Moreover, Wybraniec et al. [24] stated that the mechanism underlying this predictive role of RRI could be attributed to its correlation with both vascular remodelling (stiffness) and vascular sympathetic tone (renal artery constriction). It was stated that a higher sympathetic tone within renal arcuate and interlobular arteries could lead to both lower EDV and higher RRI values in high risk atherosclerotic CAD patients [24]. The present study has a unique design as it compensated for intrarenal flow variations by excluding those patients having valvular heart disease that could be considered as a significant determinant of RRI and also adverse long-term outcomes.
Alan et al. [36] analysed the diagnostic clinical performance of RRI
values in ROC analyses to differentiate CAD severity according to Gensini score.
They found that the best cut-off value was 0.605 with sensitivity of 80.60% and
specificity of 66.70% [36]. Furthermore, Calabia et al. [43] found high
specificity and sensitivity values for RRI in determining arterial vascular
stiffness. In this study, the best cut-off RRI value to differentiate patients
with a high SYNTAX score was
The present study had some limitations. First, it is a single-centre study including small number of certain patients with stable CAD. Thus, multicentre studies including more patients could have more significant data and results to investigate the correlation between increased RRI and cardiovascular mortality and adverse events. Second, RRI is limited by its dependence on numerous variables altering the intra-renal hemodynamics. However, patients with severe valvular lesions of any kind, high pulse pressure, tachy- and bradyarrhythmias were excluded from this study. Third, this study didn’t evaluate the potential effects of drugs taken by some patients prior to RRI assessment. Lastly, the measurements of Doppler parameters as PSV and EDV and their derivatives are influenced by inter- and intra-observer variability. Thus, measurements were repeated in kidneys using the arithmetic mean of these measurements.
Atherosclerosis is commonly associated with the affection of both coronary and renal vasculatures. In patients with stable atherosclerotic CAD, the RRI is significantly associated with both angiographic extent and complexity of CAD; and not only a specific marker for renal vascular damage. Thus, this RRI could be considered as a diagnostic modality which is easily accessible to improve the stratification of cardiovascular risk and provide additional prognostic information in stable CAD patients referred to invasive procedures.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
HR designed the research study and analyzed data. AT has made substantial contributions to data acquisition, and data analysis and interpretation. HR and AT revised the article critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in adherence to the principles of the Declaration of Helsinki on Biomedical Research Involving Human Subjects. The Ethics Committee of Zagazig university approved the study protocol (ZU-IRB #62/5). All study population already gave written informed consents prior to study participation.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.