1 School of Medicine, Health Research Institute, University of Limerick, V94 T9PX Limerick, Ireland
Academic Editor: Hirofumi Tanaka
Abstract
Hypertension and physical inactivity are leading causes of premature mortality. While both are modifiable risk factors for cardiovascular disease, their prevalence remains high. As populations grow older, they are more likely to develop hypertension and to become less physically active. Scientific advances have contributed to understanding of how physical activity improves blood pressure and the clinically relevant ambulatory blood pressure, but this is not reflected in hypertension guidelines for clinical management of hypertension. The aim of this paper is to clearly present up to date knowledge from scientific studies that underpin the role of physical activity in hypertension management. Longitudinal studies in this review demonstrate a protective effect of higher physical activity levels as well as higher levels of cardiorespiratory fitness. Interventional studies report improvements in blood pressure associated with aerobic, resistance and concurrent exercise; the improvements in some studies were greatest among participant groups with established hypertensions; the effect was observed for groups with treatment-resistant hypertension also, a clinically important subgroup. The most recent research provides evidence for the synergy between physical activity and pharmacotherapy for the treatment of hypertension, providing an opportunity for clinicians to promote physical activity as an adjunctive treatment for hypertension as well as a preventative strategy. This review critiques the evidence and summarises the most up to date literature in the field of physical activity and hypertension.
Keywords
- physical activity
- exercise
- cardiorespiratory fitness
- hypertension
- blood pressure
- preventative medicine
- lifestyle medicine
- cardiovascular disease
Hypertension is the main modifiable risk factor for preventing cardiovascular disease (CVD) and death [1, 2]. Poorly controlled blood pressure (BP) increases the incidence of CVD, end stage kidney disease, and all cause death/CVD death [3, 4, 5]. Elevated Ambulatory Blood Pressure Monitoring (ABPM) readings [6, 7, 8], Home Blood Pressure Monitoring (HBPM) readings [9], and office based readings [10], lead to higher incidence of CVD. The finding is consistent across age and ethnic groups [11, 12]. Furthermore, Framingham data demonstrates the effects of elevated BP on increasing incidences of coronary artery disease, cerebrovascular accidents, peripheral artery disease and heart failure with a reduced ejection fraction [13]. On the other hand, small improvements in BP control, even to the levels of 2 mmHg in systolic BP, lead to reductions in CVD events [14, 15]. A person is considered hypertensive, if systolic blood pressure (SBP) is persistently greater or equal to 140 mmHg and/or diastolic blood pressure (DBP) is persistently greater or equal to 90 mmHg; and, in the USA, stage 1 hypertension is defined as a SBP of 130–139 mmHg or DBP of 80–89 mmHg [16].
The prevalence of hypertension amongst the adult population is estimated at 31–38% [17, 18], with greater than 1 billion people globally diagnosed with the condition; two thirds of those with hypertension live in low- and middle-income countries [19]. Prevalence rises with age: in the elderly, it may be as high as 64%, with only 45% aware that their blood pressure readings are elevated. Only half of those diagnosed with hypertension take the medications prescribed to treat the condition, and of those who are treated, only half have their blood pressure optimally controlled [20]. The proportion of hypertensive persons that have their BP controlled may be as low as 20% in some cohorts [19, 21].
Analysis of the National Health and Nutrition Survey (NHANES) database demonstrates significantly higher all-cause, stroke and CVD disease mortality for uncontrolled hypertensives compared to controlled hypertensives or normotensives; notably, no significant differences in mortality were observed between controlled hypertensives and normotensives [22]. Treating and preventing elevated BP is a major public health priority. To this end, lifestyle measures—addressing physical activity, diet, body mass index and alcohol—have proven benefit for BP reduction [23]. In the 1950s, Morris and colleagues reported reduced death from cardiovascular disease among active London bus conductors compared to their seated driver colleagues [24], and the evidence base for addressing physical inactivity for the prevention and treatment of chronic conditions and prevention premature mortality has accelerated in recent years.
The Lancet refers to a pandemic of physical inactivity [25]; it is the fourth biggest cause of mortality worldwide, and 1.4 billion people are at risk of developing or exacerbating chronic illness due to being physically inactive [26]. There is strong evidence for the association between physical inactivity and mortality, risk of cancer, depression, dementia and more chronic illnesses [27]. The WHO guidelines for physical activity and sedentary behaviour reflect significant advances in scientific knowledge relating to physical activity and health [28]. The guidelines state that a minimum of 150 minutes of moderate intensity physical activity or 75 minutes vigorous intensity physical activity per week are required for health. They emphasise a reduction in sitting time and replacing this with any physical activity. Significantly, strength training on at least two days per week is also recommended for all adults. Internationally, there is a consensus on the weekly minimum ‘dose’ of physical activity: the 2018 joint European Society of Cardiology/European Society of Hypertension guidelines recommend 30 minutes of moderate intensity physical activity on five to seven days per week in addition to resistance training on two to three days per week [29].
Some of the terminology used in this field is important to clarify:
• physical activity refers to any bodily movement involving skeletal muscles and energy expenditure [30];
• exercise is a subset of physical activity that is “planned, structured, and repetitive and has as a final or an intermediate objective the improvement or maintenance of physical fitness” [30];
• cardiorespiratory fitness is another term for aerobic fitness and is most commonly measured by V02 max (the maximum volume of oxygen that can be used);
• aerobic training is a form of training that involves cardiovascular conditioning, e.g., walking, swimming and cycling;
• resistance training is a form of exercise to strengthen muscles by working them against a weight or force;
• isometric resistance training is a form of resistance training where the muscles work without changing length, e.g., a ‘wall sit’ – holding a seated position against a wall;
• dynamic resistance training is a form of resistance training where the muscles work by contracting, resulting in the movement of a body part, e.g., a squat;
• concurrent resistance training is a combination of aerobic and dynamic resistance training;
• light intensity physical activity refers to activities such as household tasks;
• moderate physical activity equates to a brisk walk;
• vigorous physical activity includes activities like running [28].
Understanding of how physical activity benefits health is advancing rapidly; research reports that even small amounts of physical activity can reduce mortality [31]. Analysis of data from studies using accelerometers to measure physical activity reports that physical activity, at any level, is associated with reduced mortality and that the relationship is non-linear dose response, with greatest benefits observed for getting the most physically inactive somewhat active [32]. Furthermore, recognition of the importance of cardiorespiratory fitness (CRF) in preventing adverse health outcomes is growing [33]. Medical organisations have called on clinicians to record physical activity [34] and CRF [33] as risk factors on patient health records—a view endorsed by the Lancet series on physical activity [35]. Researchers and clinicians grounded in exercise medicine advocate for exercise prescribing [36, 37].
While knowledge of the devastating effects of hypertension is known and accepted, and evidence for the role of physical activity in its management is steadily emerging, it appears that the prominence of physical activity is understated in guidelines and underutilised as a primary and secondary prevention strategy among clinicians. However, this year European cardiology societies have advocated more research to promote personalised exercise prescriptions for the prevention and management of hypertension [38]. The aim of this paper is to clearly present up to date knowledge from scientific reports that underpin the role of physical activity/exercise in hypertension management. Recognising the emergence of CRF as a related factor, we will include evidence for its role where relevant. For physicians, exercise is a management strategy to be used in conjunction with pharmacological therapy as opposed to a direct choice between the two; therefore, the synergy between pharmacological and physical activity approaches. The following sections will present epidemiological studies, interventional studies and underlying physiological mechanisms and will conclude with recommendations for future directions.
Hypertension is more commonly diagnosed in females, and a significant factor in this is a higher rate of healthcare utilisation [39]. Hypertension is also more common among lower socio-economic groups, particularly among people with lower educational levels [40]. On the other hand, physical activity levels are higher among males and higher socio-economic groups; race, genetics, interpersonal and environmental factors all contribute to physical activity behaviour across the lifespan [41]. Age is a recognised risk factor for the development of hypertension, and in the National Health and Nutrition Survey, over 60% of older adults reported a diagnosis of hypertension [42]. In terms of physical activity, 1.4 billion people are insufficiently active [26], and longitudinal studies from Europe [43, 44], the USA [45] and the UK [46] report a decline in physical activity with age.
A systematic review and meta-analysis of longitudinal cohort studies investigated the relationship between physical activity and incident hypertension [47]. It included 330,222 individuals and 67,698 cases of incident hypertension. For subjects who achieved minimum weekly physical activity guideline requirements, a 6% reduction (relative risk, 0.94; 95% confidence interval, 0.92–0.97) in incident hypertension was observed. The authors concluded that additional protective benefits against hypertension were accrued with higher levels of physical activity.
Huai and colleagues conducted a meta-analysis of longitudinal studies involving 136,846 persons who were normotensive at the outset, with 15,607 developing hypertension [48] during follow up. The study reported an inverse dose response relationship between recreational physical activity levels and the risk of developing hypertension. When comparing high versus low physical activity levels, the risk reduction was 0.81: RR, 0.81; (95% CI); and, for moderate versus low, risk reduction was 0.89 (95% CI). No significant relationship was noted between physical activity accumulated at work and hypertension risk. A longitudinal study involving over 5000 participants with a 20-year follow-up period reported that both cardiorespiratory fitness and physical activity levels are inversely associated with the development of hypertension [49].
A separate review of longitudinal studies examined the relationship between physical activity and CVD mortality among subjects with established hypertension [50]. It reported that inactive individuals had double the risk of mortality compared to active. However, the authors cautioned about lack of consistency in reporting of intensity, duration and type of exercise, as well as BP status. Recently, using data from the UK Biobank study on 39,294 individuals with hypertension, researchers reported an inverse dose-relationship between mortality and accelerometer-measured physical activity [51]. The study had a median follow-up time of 6.25 years, and utilised accelerometer-measured physical activity, factors that support the reliability of the findings. Further, the authors noted that higher levels of physical activity were not associated with harm, a finding that should encourage clinicians to promote physical activity to individuals with hypertension.
The evidence base is advancing also for CRF and BP; Holmlund and colleagues observed that a large improvement in CRF was associated with an 11% lower risk of incident hypertension compared to people who maintained the same fitness levels; the finding was reported after adjustment for smoking, BMI, diet, stress and exercise habits [52]. This study involved a retrospective analysis conducted at two time points on the Swedish Health Profile Assessment database, involving 91,728 participants with a mean age of 40.7 years. Similarly, a contemporaneous systematic review and meta-analysis reported an inverse dose-response relationship between CRF and risk of developing hypertension [53]. In that review, the high CRF group had a 37% risk reduction compared to the low CRF group; and, the moderate CRF group had a 15% risk reduction compared to the low CRF group. Interestingly, BMI was noted as a modifier in the relationship, suggesting that those with higher BMI may gain more from becoming more cardiorespiratory fit. The review involved nine large cohort studies but could not distinguish between effects for gender. Furthermore, the authors acknowledged heterogeneity of how CRF measurement between studies [53]. Similarly, a longitudinal study of adolescents reported an inverse relationship between CRF and SBP [54]. The implication for the clinician is that encouraging patients to become physically fit across the lifespan is likely to prevent, or at least delay, the onset of hypertension and its negative health sequelae.
For millennia, aerobic exercise, particularly walking, has been recommended for cardiovascular prevention and longevity. Up until the past decade most research concerning physical activity and blood pressure focused on aerobic exercise. Accordingly, aerobic exercise has been recommended by systematic reviews and guidelines as the most appropriate form of physical activity for BP reduction [55]. Further large reviews of exercise, including aerobic exercise are discussed in the following sections. Some of the larger studies published recently are presented in Table 1 (Ref. [56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70]); Fig. 1 outlines key findings from the studies.
| Study, year | Intervention | Participant profile, number | Outcome |
| Aerobic continuous v inactive control | |||
| Williamson [64], 2022 | Aerobic (moderate-high continuous) | Young normotensive adults (N = 203) | No differences in ABPM between control and aerobic exercise cohorts at 16 weeks or 52 weeks |
| 3 | |||
| Blumenthal [62], 2021 | Aerobic (moderate continuous) | Treatment resistant hypertensives (N = 140) | ABPM SBP was reduced in the aerobic cohort [–7.0 (95% CI, –8.5 to –4.0) mmHg], no change in control (p = 0.001) |
| As part of cardiac rehabilitation and in conjunction with dietary input | |||
| Control | |||
| Lopes [63], 2021 | Aerobic (moderate continuous) | Treatment resistant hypertensives (N = 53) | ABMP SBP for the aerobic group was reduced by 7.1 mmHg (95% CI, –12.8 to –1.4; p = 0.02). Additionally, 24-hour ambulatory diastolic ABPM DBP was reduced (–5.1 mmHg; 95% CI, –7.9 to –2.3; p = 0.001) |
| 3 | |||
| Dimeo [61], 2012 | Aerobic (moderate continuous) | Treatment resistant hypertensives (N = 50) | SBP reduced in the aerobic cohort [6 mmHg (+/–12 mmHg) p = 0.03] and DBP reduced in the aerobic cohort [3 mmHg (+/–7 mmHg) p |
| Control | |||
| Aerobic continuous v isometric interventions | |||
| Goessler [65], 2018 | Aerobic (moderate continuous) 150-minutes of CME/week | Young normotensive adults (N = 61) | Similar improvements in office SBP and ABPM |
| Isometric daily 2-minute contractions ( |
Office DBP improved only in aerobic cohort | ||
| Pagonas [66], 2017 | Aerobic (moderate continuous) 3–5 |
Hypertensive adults (N = 75) | No statistical improvement was noted in the isometric cohort |
| Isometric 5 |
A significant SBP reduction was noted in the aerobic cohort (p = 0.025) using ABPM | ||
| HIIT interventions | |||
| Reljic [67], 2020 | HIIT 2 sessions (cycling) | Mixed hypertensive and non-hypertensive (N = 65) | Reduced SBP [–12 mmHg (95% CI –16 to –8 mmHg), p |
| Inactive control cohort | |||
| Control cohort was unchanged | |||
| Ghardashi Afousi [68], 2018 | HIIT 3 sessions (cycling) v Aerobic (moderate continuous) 42-minutes |
Hypertensive and pre hypertensive adults (N = 75) | Both groups significantly improved SBP compared to control: HIIT [–3.66 mmHg] and CMIT [–3.7 mmHg] |
| Inactive control | Both groups significantly improved DBP compared to control: HIIT [–5.5 mmHg] and CMIT [–4.8 mmHg] (p | ||
| Shepherd [69], 2015 | HIIT 3 sessions | Mixed hypertensive and non-hypertensive (N = 90) | Reduced SBP [–4 mmHg (–7 to –2 mmHg), p = 0.01] was noted in the Moderate continuous group only |
| (cycling)/week | |||
| Continuous aerobic | No changes were noted in the HIIT cohort; no changes were noted for DBP | ||
| 5 |
|||
| Isometric exercise interventions v inactive control | |||
| Correia [56], 2020 | Isometric 3 |
Mainly hypertensive (N = 102) | Reduced DBP [–3 mmHg, p = 0.04] in the isometric cohort |
| Reduced SBP in isometric cohort but not statistically significant | |||
| Farah [57], 2018 | Isometric 3 |
Medicated hypertensive adults (N = 72) | Reduced SBP from [132 mmHg +/–4 mmHg to 120 mmHg +/3 mmHg] and reduced DBP [71 mmHg+/–2 mmHg to 66+/–2 mmHg (p |
| Supervised v home-setting | |||
| Inactive control | |||
| Medication v non – medication trial | |||
| Mora-Rodriguez [70], 2022 | 3 HIIT sessions/week | Hypertensive adults (N = 178) | The medicated and non-medicated group each demonstrated similar reductions in BP: |
| Medicated (n = 103) | Medicated group [–15 mmHg (95% CI –12 to –19); p | ||
| No medication (n = 75) | The higher the baseline SBP the greater the reduction after training | ||
| Combined exercise | |||
| Ruangthai [60], 2019 | Aerobic (moderate) |
Mixed hypertensive and normotensive (N = 54) | SBP reduced significantly in the aerobic (–7.9%, p |
| Strength |
At 12-week follow-up post intervention, only the combined cohort maintained significant SBP reduction (–7.5%, p | ||
| Combined (aspects of both within a single session) | |||
| Lima [58], 2017 | Aerobic group |
Normotensive (N = 44) | SBP (wake and sleep) decreased similarly for the aerobic cohort and combined cohort, measured by ABPM (p |
| Combined group – as above plus dynamic resistance exercise circuit | DBP (wake only) decreased by 4 mmHg in both the aerobic and combined cohorts | ||
| True control | No change was seen for night DBP [58] | ||
| Sousa [59], 2013 | Aerobic group (moderate-vigorous) |
Mixed hypertensive and normotensive (N = 59) | SBP reduced in the aerobic cohort [149.4 mmHg ( |
| Combined group – as above with one session replaced with dynamic resistance exercise | |||
| Control group | DBP reduced in the aerobic cohort [80.4 mmHg ( | ||
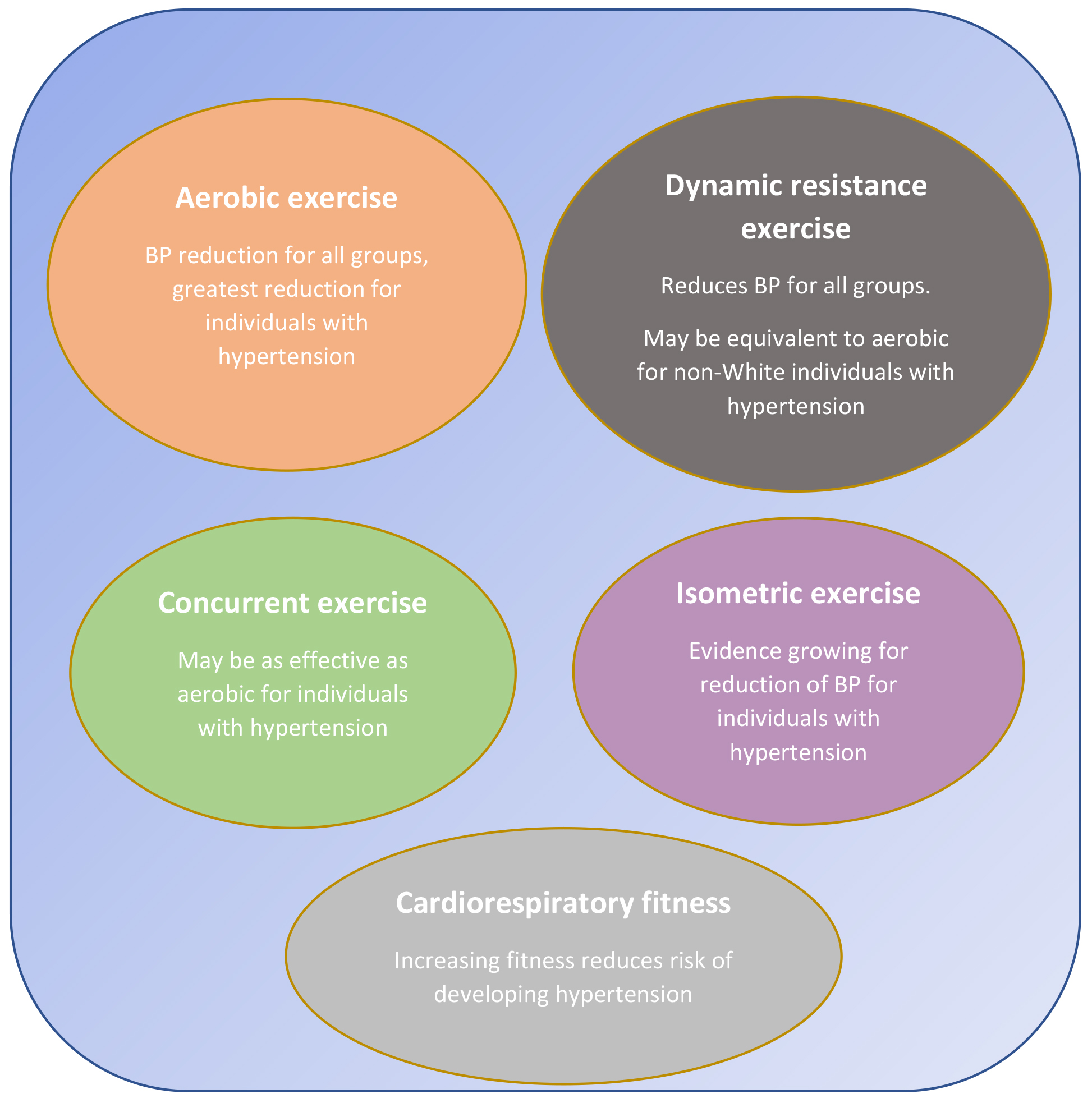 Fig. 1.
Fig. 1.Physical activity options for BP reduction.
The question of whether HIIT or MICT training is most effective for blood pressure has been the subject of four systematic reviews. Leal and colleagues investigated differences in BP reduction between HIIT and MICT, reviewing 15 trials of individuals with hypertension (mean age 55.8 years): HIIT and MICT reduced SBP with comparable effect but HIIT was more effective at reducing DBP for this population [71]. Another review involving 14 trials that were mainly composed of younger and middle aged adults reported a greater effect for HIIT in the hours following the bout of exercise, but no difference on nocturnal BP [72]. A systematic review conducted on studies involving an elderly population, concluded that both modalities were effective with comparative success [73].
Cornelissen and colleagues (2011) examined 28 randomized-controlled trials (with
33 study groups and 1012 participants) in a systematic review and meta-analysis
[74]. Only five of the 33 study groups had hypertension—all others were either
normotensive or pre-hypertensive. Dynamic resistance training (squats, push ups,
bicep curls) was the dominant intervention in trials (n = 25 trials), and static
resistance training (isometric hand grip) was examined in three trials. The
authors remarked that the quality of trial reporting was poor overall (all were
published before 2010). The combined effect for all modalities of resistance
training were SBP (–3.9, 95% CI, –6.2; –1.5)/DBP (–3.6, 95% CI, –5.0;
–2.1) mmHg, (p
Cornelissen and Smart repeated the meta-analysis in 2013 to include more recent
studies and widened the inclusion criteria to include dynamic aerobic endurance
(jogging, swimming), dynamic and isometric resistance, and combined
resistance-endurance training [75]. Overall, it involved 93 trials (105
endurance, 29 dynamic resistance, 14 combined, and 5 isometric resistance
groups), with 5223 participants (3401 exercise and 1822 control). SBP was reduced
after endurance (–3.5 mmHg; 95% CI –4.6 to –2.3), dynamic resistance (–1.8 mmHg; 95% CI –3.7 to –0.011), and isometric resistance (–10.9 mmHg; 95% CI –14.5 to
–7.4) but not after combined training. Reductions in DBP were seen in all four
modalities examined. Once again, isometric resistance training was the most
effective modality reported but, interestingly, BP reductions after endurance
training were greater (p
MacDonald’s systematic review of dynamic resistance training included 64 studies, reported a modest SBP and DBP reduction, and concluded that the greatest reduction was observed among non-White hypertensives [76]. A recent systematic review, focussing specifically on isometric exercise and blood pressure, concluded that this exercise modality has a statistically significant and clinically important SBP lowering effect [77]. Two large trials investigated hand grip training (a form of isometric exercise) for individuals with hypertension and prehypertension; compared to inactive controls, statistically significant reductions in DBP [56, 57] were reported in both, and SBP reported in one [57].
A meta-analysis of 68 trials reported that concurrent resistance training is as useful as aerobic exercise for the for reducing BP in people with hypertension [78]. Utilising ABPM for measurement, Lima and colleagues compared aerobic, combined and inactive control; they reported similar SBP reductions for both during wake, as well as similar sleep and similar waking DBP reductions [58]. A second trial, conducted among an older cohort with more hypertension at baseline, reported a greater SBP and DBP reduction among the combined exercise cohort [59]. Most interestingly, the third study reported similar SBP reductions for aerobic and combined cohorts (but no significant reduction for strength only); the SBP reduction was maintained at 12-weeks follow-up. In practical terms, this is relevant as the unsupervised period during which benefits were maintained is reflective of real-life settings, where in most instances continuous support is not available [60].
There is no agreed frequency of ‘exercise dose’ for achieving BP reduction. Lee & Chae reported that optimum frequency of physical activity for BP lowering effect is three times per week [79]. For SBP, effect sizes depending on exercise frequency were: –6.10 mmHg (95% CI = –8.83 to –3.37) for twice weekly; –9.16 mmHg (95% CI = –11.66 to –6.66) for three times weekly’ and –6.96 mmHg (95% CI = –9.27 to –4.64) for four or more times weekly. For DBP, effect sizes based on exercise frequency were: –4.38 mmHg (95% CI = –7.09 to –1.66) for twice weekly; –5.55 mmHg (95% CI = –6.87 to –4.23) for three times weekly; and, –4.50 mmHg (95% CI = –6.80 to –2.20) for four times weekly. Cornelissen & Smart suggest that even less than three exercise sessions per week provides maximum BP benefit; this meta-analysis included various frequencies of exercise intensity, ranging from 1 to 7 times per week across 93 trials (n = 5223) [75]. In the trials reviewed for this paper, most aerobic interventions were 3–5 times per week, whereas HIIT sessions were 2–3 times weekly.
Lee & Chae categorized aerobic training intensity into low-intensity, moderate-intensity, and vigorous intensity in their meta-analysis [79]. They reported that only moderate- [–8.97 mmHg (95% CI = –11.11 to –6.84)] and vigorous- [–6.85 mmHg (95% CI = –12.03 to –1.66)] intensity exercise improved SBP. A similar trend was observed regarding DBP: [–5.75 mmHg (95% CI = –7.18 to –4.32)] for moderate-intensity exercise; and, for vigorous-intensity exercise [–4.36 mmHg (95% CI = –5.94 to –2.79)]. Moderate-intensity exercise had the most impact on reduction of both SBP and DBP. This is consistent with findings from Cornelissen & Smart, who reported lower intensity training to be associated with the smallest effect size on SBP (p = 0.032) and DBP (p = 0.03) [75]. Their meta-analysis involved trials with intensity from 35% to 95% peak oxygen consumption for endurance training, between 30% and 100% of 1-repetition maximum for dynamic resistance training, and between 10% and 40% for isometric resistance training.
Lee & Chae report that intervention duration is important in mediating BP
reductions of aerobic physical activity [79]. This meta-analysis included trials
with intervention duration varying between 4 and 37 weeks. Their results show
that exercise durations of 4–7 weeks [–3.04 mmHg (95% CI = –5.14 to –0.95)],
8–11 weeks [–9.12 mmHg (95% CI = –14.09 to –4.16)], 12–23 weeks [–8.77 mmHg
(95% CI = –11.06 to –6.49)] and 24 or more weeks [–8.24 mmHg (95% CI =
–10.77 to –6.27)] all reduce SBP and DBP (–3.5 mmHg: 4–7 weeks, –5.4 mmHg;
8–11 weeks, –4.8 mmHg: 12–23 weeks, –7 mmHg: 24 or more weeks). The
intervention duration of 8–11 weeks showed the greatest effect in reducing SBP
whereas 24 weeks or longer had the most impact in lowering DBP. These findings
contrast to the results of Cornelissen & Smart (2013) who found program duration
of
In 2020, a review conducted by Saco-Ledo and colleagues reported that 24-hour blood pressure, DBP and night-time blood pressure [81]. Two interesting and, perhaps unexpected, findings were noted from subgroup analyses: firstly, significant improvements in ABP were only noted for participants treated with anti-hypertensive medication and secondly only aerobic exercise produced a significant improvement [81]. A separate review involving 17 study groups and 633 participants reported ABP improvements with exercise for daytime but not night-time BP [82].
A meta-analysis conducted by Faggard and Cornelissen, that included 72 RCTs and 105 study groups, investigated the effects of exercise training on BP. The authors concluded that the greatest reductions in BP were observed among hypertensive study groups [83]. The finding was consistent with a later systematic review and meta-analysis by Cornelissen and Smart, that grouped a total of 5223 participants into hypertensive (26 study groups), pre-hypertensive (50 study groups) and normotensive (29 study groups) [75]. The study reported statistically higher SBP and DBP reductions among the hypertensive group, followed by the pre-hypertensive group. However, current guidelines have reclassified what was pre-hypertension as hypertension, making interpretation of older reports more complex [84]. A systematic review reported greater benefits from exercise for people with pre-hypertension compared to normotensives, but that all categories demonstrated improvements [85].
A recent review focussed only on older hypertensive adults, involving 69 RCTs and 2272 participants [86]; it reported significant decreases in both SBP and DBP after exercise. Similarly, a contemporaneous review of training in normotensive adults demonstrated that exercise produced beneficial effects on BP [87].
Treatment resistant hypertension is associated with increased organ damage and was defined as having uncontrolled blood pressure despite taking three anti-hypertensive medications or having controlled blood pressure and taking at least four anti-hypertensives; it has been the subject of high-quality trials and systematic reviews. An RCT used ABPM to measure the impact of an 8–10-week programme on treatment resistant hypertension: significant reductions in SBP and DBP were observed [61]. Recently, a larger trial involving 140 individuals with treatment resistant hypertension reported significant improvements in SBP as measured by ABPM [62]. In 2021, two trials with a combined total of 193 individuals with treatment resistant hypertension, were randomised to a moderate intensity aerobic exercise intervention or a control; the trials were of three to four months in duration and both reported statistically significant ABPM measured SBP reductions close to 7 mmHg [62, 63]. A systematic review, published in 2022, involving 10 studies and a combined population of 380 participants, observed that exercise appears to be effective in reducing BP for people with treatment resistant hypertension [88].
It is likely that synergy will exist if both these treatment modalities are optimized in single patients. In 2020, Magalhães reported that aerobic exercise significantly increased urinary levels of ACE (angiotensin converting enzyme) and plasma levels of ACE2. This may indicate the synergistic mechanism of renin-angiotensin-aldosterone inhibitor based anti-hypertensive drugs and exercise [89]. Whilst many trials based on hypertensive patients have shown that aerobic exercise reduced BP [82, 90], most have not attempted to peel this back and assess effects of medication, aerobic exercise or a combination of both.
A network meta-analysis investigating the efficacy of exercise versus medications in lowering BP suggested that medications are more effective (mean difference –3.96 mmHg, 95% CI –5.02 to –2.91); the authors noted that all types of exercise lowered BP and that individuals who were taking anti-hypertensive medications had the best BP lowering responses [91]. A second network meta-analysis on this topic acknowledged that ‘the current evidence base with a bias towards medication research may partly explain the circumspection around the efficacy of exercise in guidelines and practice’ [92].
While a recent systematic review purported that the benefits of exercise and pharmacotherapy are not additive and are not synergistic [93], a series of small trials have demonstrated improvements in BP from combining exercise with pharmacological management. The most recent of these (which post-dated the review) concluded that both medication and physical activity (in this instance HIIT) have independent and additive effects in lowering BP (24 hour Mean Arterial Pressure: –5.7 mmHg) [94].
Current evidence suggests that pharmacologically treated hypertensive people should aim to exercise. This may reduce the need for escalation of anti-hypertensive drug doses or the prescription of additional drugs over time, as BP remains controlled. Issues remain on the generalisability of knowledge from these trials to real world settings-as in these trials most patients were well controlled hypertensive patients, largely taking renin-angiotensin-aldosterone inhibitor drugs, and participant numbers remain small. Further large trials are required and should include hypertensive populations that are sub-optimally controlled (between 130/80 to 145/90 mmHg etc.), and those with co-morbidities.
Postexercise hypotension (PEH), the phenomenon whereby a single session of exercise can generate a transient reduction in BP [95], which occurs in both hypertensive and normotensive individuals [96], but seems to have a greater and longer lasting effect in the latter group [97]. The primary mechanism proposed in PEH is the central baroreflex pathway, and this is mediated by a complex system involving an exercise-induced neuroplasticity in the brainstem medulla [98]. During exercise, the sympathetic system is activated, initiating catecholamine release, inducing peripheral vasoconstriction [99], thereby raising BP. Subsequent cessation of exercise initiates reactivation of parasympathetic mechanisms and sympathetic deactivation [100]. Importantly from a clinical perspective, sustained elevations in BP (as in hypertensives), have been found to alter the central baroreflex pathway by resetting to higher activation threshold [101], thereby reducing reflex sensitivity.
Sustained postexercise vasodilation refers to the persistence of increased blood flow to vascular beds following exercise cessation; local vasodilatory mechanisms - namely nitric oxide (NO) release and histamine activation pathway are implicated in sustained postexercise vasodilation [102].
NO is produced and released by endothelial cells, exhibiting vasodilatory effects through vascular smooth muscle relaxation and sympathetic neural constrictor antagonism [103]. Exercise stimulates NO release through increased shear stress leading to endothelial cell deformation [104, 105]. The hypotensive effect mediated by NO release in response to exercise appears to be related to exercise intensity [106] and may have genetic and gender influences. Positive effects on BP, mediated by increases in NO levels have been reported for normotensive [107] as well as hypertensive [108] populations.
Activation of skeletal muscles, as occurs during exercise, promotes the formation and release of histamine, a chemical that plays a key role in sustained postexercise vasodilation [109, 110]. Research also points to two other potential mediators of physical activity on BP: angiogenesis, whereby physical activity is responsible for morphological changes in the vascular tree, thereby augmenting blood flow; and the sensitising effect of physical activity on insulin [111]. A contemporaneous review reported that aerobic exercise improves vascular remodelling across the layers of the arterial wall, hence reducing peripheral vascular resistance, by reducing inflammation, proliferation and fibrosis [112].
Physical inactivity and hypertension are extremely common, and both are drivers for increased cardiovascular morbidity and mortality. Engagement in any modality of physical activity has a BP lowering effect, but a combination of moderate to vigorous intensity aerobic, in conjunction with resistance training, over at least three days of the week, appears to be the most successful. The greatest BP lowering effects are seen in those who are already deemed to have high BP. Hypertension is more common in the elderly, and in those from lower socio-economic groups. Getting these specific groups more active will ultimately benefit the individual and society. Incorporating behaviour change into healthcare consultations is key to doing this. The promotion of the prescription of exercise as a treatment modality is ever-increasing, but further work is needed-it is not suggested however the physical activity alone should be a substitute for the use of anti-hypertensive medication. Nor do we suggest that healthcare professionals should be expected to shoulder the enormous task of increasing population physical activity levels alone – physical inactivity is a complex, multi-level public health problem that requires a strategic multi-system response, including co-operation between state departments such as transport, urban planning and education, as well as regional and local organisations.
A problem identified in this review is the heterogeneity of studies. While
several high-quality systematic reviews and meta-analyses of both RCTs and
longitudinal studies exist, major disparities exist between population groups.
Physiology, genetics, gender, age and morbidity levels are likely correlates in
the relationship between physical activity/exercise and hypertension. However,
with most trials involving healthy people with normal BP, the effect reported
will be lower than it would be if a homogenous population with hypertension was
recruited. Further research should involve high quality, well powered, RCTs
examining those with grade 1 hypertension (
It is also clear further examination of the synergistic effect of medication plus increased exercise needs analysis as a clear scientific basis for this hypothesis now exists. Although at an early stage of investigation-it is likely that some synergy exists between anti-hypertensive medication and increased exercise in reducing BP. From a clinical viewpoint, physicians routinely promote exercise as well as prescribing medications for hypertension.
Technology is broadening the scope of research in this field; home BP monitoring is an effective way of reducing BP and could effectively be managed with the collaboration of healthcare professionals; exploring how this can be done safely and effectively, perhaps by involving synchronous and asynchronous mechanisms, is a priority for clinical researchers. Medical organisations are advocating for investment in technology as well as training and reimbursement for healthcare professionals to build health information capacity. In this context, wearables have become commonplace and have potential to bring about awareness of personal physical inactivity and to motivate and bring about behavioural change in relation to physical activity and exercise. Technology can facilitate data collection on BP and physical activity levels, and healthcare professionals of the future will need to be adaptable in order to harness its potential. Through judicious use of technology, future research could provide evidence to inform more precise recommendations for the physical activity needed for BP reductions. With the rapid development of devices and opportunities, an exciting future awaits clinicians and researchers in the fields of physical activity and hypertension.
Robust evidence from RCTs and systematic reviews supports exercise for the management and treatment of hypertension. All types of physical activity produce blood pressure lowering effects and improvements are observed for people in every blood pressure category. Traditionally, evidence was strongest for aerobic exercise, but as research in this field expands, resistance (both dynamic and isometric) and concurrent exercise, have demonstrated improvements in blood pressure. Achieving healthy blood pressure levels is a major health challenge; to achieve this, clinicians have sufficient evidence to encourage patients to become more physically active, as part of a wider strategy.
Conceptualization—PH and AOR; content design—PH and AOR; writing — original draft preparation—PH, KMK, AF, AK and AOR; writing — review and editing—AOR, and PH; supervision—AOR and PH. All authors have read and agreed to the published version of the manuscript.
Not applicable.
The authors wish to acknowledge the support and encouragement of Professor Liam Glynn.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

