1 German Centre for Heart and Circulatory Research (DZHK) Partner Site Berlin, 10785 Berlin, Germany
Academic Editors: Yan Topilsky and Fabian Sanchis-Gomar
Abstract
Echocardiography (ECHO) is indispensable for evaluation of patients with terminal chronic heart failure (HF) who require transplantation or mechanical circulatory support by a left- or biventricular assist device (LVAD or BiVAD, respectively). In LVAD candidates, ECHO represents the first-line investigation necessary for a timely discovery of heart-related risk factors for potentially life-threatening post-operative adverse events, including identification of patients who necessitate a biventricular support. ECHO is also required for intra-operative guiding of VAD implantation and finding of the most appropriate setting of the device for an optimal ventricular unloading, postoperative surveillance of the VAD support, and monitoring of the RV changes in LVAD recipients. Thanks to the ECHO, which has decisively contributed to the proof that prolonged VAD support can facilitate cardiac reverse remodeling and functional improvement to levels which allow successful weaning of carefully selected patients from LVAD or BiVAD, the previous opinion that chronic non-ischemic cardiomyopathy (NICMP) is irreversible could be refuted. In patients with normalized and stable right heart catheter-derived hemodynamic parameters obtained at short-term interruptions of VAD support, ECHO has proved able to predict post-weaning long-term freedom from HF recurrence in patients with pre-implant terminal chronic NICMP. The purpose of this article is to offer an actualized theoretical and practical support for clinicians engaged in this particularly challenging and topical issue especially due to the new practical aspects which have emerged in conjunction with the growing use of long-term ventricular assist devices as bridge-to-transplantation or as destination therapy, as well as the increasing evidence that, in some patients, such VAD can become a bridge-to-recovery, allowing the removal of the device after a longer support time.
Keywords
- echocardiography
- heart failure
- heart transplantation
- ventricular assist devices
- myocardial recovery
- weaning from assist device
Heart failure (HF) is a clinical syndrome of varying etiologies resulting from the loss of compensation for acute or chronic cardiac dysfunction due to structural and/or functional cardiac abnormalities, leading to low cardiac output (CO) and/or high filling pressures [1]. The management of severely symptomatic end-stage chronic HF refractory to medical therapy and implantable electronic heart rhythm management devices (i.e., stage D) is demanding, requiring specialized treatment strategies such as heart transplantation (HTx) and ventricular assist device (VAD) implantation [2]. Despite its well known limitations, HTx is still the optimal therapy for advanced refractory chronic HF. Long-term event-free survival is still better after HTx compared with mechanical circulatory support. However, the increasing use of long-term VADs as destination therapy (DT) for patients who are not eligible for HTx has substantially improved the management strategies for end-stage HF [1, 2]. In addition, there is clear evidence that some of the VADs initially designed as DT or as a bridge-to-transplantation (BTT) can turn into a bridge-to-recovery allowing their explantation after several weeks or months [3, 4, 5, 6, 7].
Echocardiography (ECHO) is a major tool for cardiac assessment in HTx and VAD candidates and is recommended as the key investigation for monitoring of VAD recipients [2]. Before implementation of a left ventricular assist device (LVAD) therapy, ECHO is indispensable for decisions regarding the need for an additional temporary or long-term right ventricular (RV) assist device, and in HTx candidates, ECHO can help in optimizing the listing of HTx (e.g., timing and prioritization) [8, 9, 10, 11].
The purpose of this article is to provide an updated overview on the usefulness and limitations of the currently available ECHO techniques for assessment of failing hearts due to primarily altered LV structure and function in patients with chronic non-ischemic cardiomyopathy (NICMP). Particular attention is paid to the importance of ECHO for detection and evaluation of ventricular reverse remodeling associated with improvement of contractile function during VAD support, as well as for weaning decision-making in patients with reversal of ventricular dilation and evidence of relevant and stable functional improvement.
Despite the beneficial effects of neurohormonal antagonists, and cardiac resynchronization therapy (CRT) with or without combined ICD, many patients eventually progress to an advanced HF stage, characterized by severe clinical symptoms, marked hemodynamic impairment, and high mortality [12, 13]. Early identification of patients at high risk for rapid cardiac deterioration leading to dependency on continuous inotrope infusions, and finally to the need for HTx or VAD implantation is crucial for successful management of end-stage HF. Continuous comprehensive and optimally timed ECHO monitoring is particularly suitable for this purpose [8, 14, 15, 16, 17, 18, 19, 20].
Because the key features of HF resulting from cardiac structural and/or
functional damages are low CO and high cavity filling pressures, it appears
logical that the severity of both reduction of stroke volume (SV) and diastolic
dysfunction can indicate the risk for rapid deterioration of heart function
towards life-threatening stage D chronic HF. Stable patients with chronic NICMP
reveal a lower base-line left atrial volume index (LA
Development of pressure overload-induced RV dilation and failure (RVF) in HF
syndrome triggered by LV dysfunction indicate accelerated disease progression
associated with a two- to threefold increase in risk of cardiac death, regardless
of the degree of LVEF impairment [21]. In patients with either
reduced or preserved LVEF (HFrEF or HFpEF, respectively), the ratio between the
ECHO-derived tricuspid annular plane systolic excursion and the pulmonary
arterial systolic pressure (TAPSE/PASP), which allows the estimation of
RV-pulmonary artery (RV-PA) coupling by plotting fiber shortening (TAPSE) vs. the
force generated for overcoming the imposed load (PASP), was identified as an
independent predictor of HF aggravation and fatal outcome [22]. CRT
can improve RV–PA coupling in patients with mismatched TAPSE and PASP and
the TAPSE/PASP ratio appeared independently associated with outcomes in
CRT recipients [21]. A low TAPSE/PASP ratio (
In HF with preserved LVEF (HFpEF), where the right atrial pressure (RAP)
reflects specifically the cumulative burden of abnormalities in the left heart,
pulmonary vasculature, and the right heart, the RAP estimated from inferior vena
cava morphology and its respiratory change [estimated right atrial pressure
(eRAP)] was found particularly useful for prediction of HF worsening
[15]. In a recent study, eRAP
Assessments of the short-term (6 months) prognostic value of cECHO and speckle tracking echo-cardiography (STE) in HTx candidates with idiopathic DCM revealed that stable patients have at baseline higher LV end-systolic global longitudinal strain (GLS) and peak systolic strain rate values, as well as lower systolic circumferential and longitudinal intraventricular dyssynchrony indexes [8]. Stable patients reveal also longer trans-mitral E-wave deceleration times, lower E/A flow velocity ratios and LV diastolic early/late strain rate ratios, as well as higher LV late diastolic strain rate values, which all indicate a less altered myocardial compliance with less restrictive LV filling [8]. The superiority of STE over cECHO for evaluation of LV systolic dysfunction and prediction of HF aggravation was meanwhile confirmed by several other studies [19, 20, 26, 27, 28].
The reduction of GLS and increase of the ratio between the transmitral E-wave
velocity and the early diastolic LV longitudinal strain rate (E/E’sr) in patients
with reduced LVEF were found more predictive for cardiac worsening (mortality or
urgent HTx) than the alteration of LVEF and/or the E/e’ ratio [28]. An ECHO score (including cECHO, STE and 3D-ECHO parameters) for prediction of
major adverse cardiac events (including urgent need for a VAD or HTx) related to
advanced chronic HF, underlines the significance of the left-sided heart filling
pressure increase and the associated right-sided heart alterations (size and
geometry changes plus RV systolic dysfunction) as prognostic indicators
[20]. Thus, together with the LA
A large portion of patients with advanced HF has severe mitral regurgitation (MR), which acts as a driving force in inducing and also further aggravating the HF in a vicious cycle [14].
Although ECHO data reflecting LV size provide important information on chronic NICMP-related LV remodeling, the cavity size cannot predict rapid deterioration of heart function toward end-stage HF. Similar degrees of LV dilation can be associated with varying degrees of systolic and diastolic dysfunction, depending mainly on the impairment of myocardial contractility, the severity of secondary MR, and the severity of filling restriction due to the reduced LV myocardial compliance. The latter is mainly a consequence of high filling pressure-induced LV overdistension and/or extensive MF, but it can also be affected by ventricular diastolic interactions [29, 30, 31]. LV dilatation is the main cause of significant MR and requires adequate medication plus close ECHO monitoring already in the early stages of LV dilation and dysfunction.
Contrary to the LV cavity size, secondary MR appeared consistently associated
with higher mortality regardless of its severity and has proved to be a strong
predictor of deleterious events in chronic HF initiated by a severe LV systolic
dysfunction [30, 31, 32]. MR was found to be a powerful independent predictor of
12-months mortality, and the risk of death among elderly persons with
moderate/severe secondary MR can be 4-fold higher than in those with absent/mild
MR [30]. The optimal time for MV repair in patients with secondary
chronic MR is difficult to determine because even patients with mild secondary MR
have a worse prognosis and also because the smaller regurgitant volume (RegVol)
due to the impaired LV systolic function, may lead to underestimation of MR
severity [32, 33]. During the compensatory phase of MR, the increased preload and
reduced or normal afterload as a consequence of blood regurgitation into the LA
can allow a temporary preservation of the forward SV (
In secondary MR, in contrast to primary MR, the relationship between the LV volume overload and the EF is altered by the fact that LV dilation and dysfunction are rather the cause than the result of MR [36]. Thus, although restoration of MV competence will not be curative, timely interruption of the vicious circle of LV dilation and MR in carefully selected patients may delay the aggravation of HF [33]. According to the current guidelines, MV repair might be considered in severe symptomatic secondary MR (stage D). However, the optimal criteria for defining severe MR (Table 1, Ref. [33, 35, 36, 37, 38, 39]) and also those for the benefit/risk assessment of surgical therapy are still a controversial issue, and the identification of appropriate patients for MV repair or replacement remains challenging [33, 36, 37, 38, 39, 40]. The fact that secondary MR is dynamic and load dependent additionally complicates the evaluation of its severity [32, 35, 38]. Grading of functional MR is also more challenging due to the lack of structural abnormalities of the MV leaflets and chords and also because symptoms, LV and LA dilation, pulmonary congestion, as well as alterations of the pulmonary venous flow pattern, may be caused by the underlying cardiomyopathy, and are thereby less useful for establishing the severity of MR [35].
| Guidelines and studies | Threshold values of major ECHO-derived variables which indicate severe MR | ||
| EROA | RegVol | RegFrac | |
| Nishimura et al. [33] | |||
| 2017 AHA/ACC guidelines | |||
| Baumgartner et al. [37] | |||
| 2017 ESC/EACTS guidelines | |||
| Zoghbi et al. [38] | |||
| 2017 ASE guidelines | 30–39 mm |
45–50 mL if 3 of 4 other specific ECHO criteria* are present or if elliptical ROA | 40–49% if 3 of 4 other specific ECHO criteria* are present or if elliptical ROA |
| Bartko et al. [36] study | |||
| 2019 Unifying concept | |||
| Bonow et al. [35] | |||
| 2020 ACC Expert Consensus | 30–39 mm |
45–50 mL if 3 of 4 other specific ECHO criteria* are present or if elliptical ROA | 40–49% if 3 of 4 other specific ECHO criteria* are present or if elliptical ROA |
| Vahanian A. et al. [39] | |||
| 2021 ESC/EACTS guidelines | or |
or | |
| ECHO, echocardiography; MR, mitral regurgitation; EROA, effective regurgitant
orifice area; RegVol, regurgitant volume; RegFrac, regurgitant fraction; ROA,
regurgitant orifice area; AHA, American Heart Association; ACC, American College
of Cardiology; ASE, American Society of Echocardiography; ESC, European Society
of Cardiology; EACTS, European Association for Cardio-Thoracic Surgery. * the 4 criteria are: vena contracta | |||
Because no single ECHO parameter is sufficient for quantifying MR in individual patients, integration of multiple parameters is mandatory for assessment of MR severity [35, 36, 40]. However, because the choice of the most appropriate therapeutic strategy for severe HF related to chronic NICMP associated with severe secondary MR (e.g., MV repair, VAD implantation or listing for HTx) is based on multiple factors; ECHO-derived data, although indispensable, represent only a part of the information requested for a final decision-making.
The impact of MR on the validity of EF for the evaluation of LV contractile
function was already recognized decades ago [41]. In view of the
high incidence of secondary MR in NICMP, this problem needs particular
consideration in order to avoid misinterpretation of measurements, because LVEF
can be misleading in patients with severe secondary MR [39]. The
reason for the negative impact of MR on the validity of EF as a parameter of LV
contractile function is the fact that, in the presence of MR, the
Overrating of LV pump function can be prevented by quantification of the forward
EF (
MR also affects the validity of E/e’ by increasing the velocity of early diastolic displacement of the mitral annulus in accordance with the increase of diastolic flow volume across the mitral valve [50]. This can lead to underestimation of LV diastolic dysfunction and can adversely affect the value of E/e’ for timely prediction of rapid deterioration of heart function toward end-stage HF.
In patients with advanced HFpRF it is likely that the frequently detectable mild or moderate MR, which appeared associated with greater hemodynamic severity of cardiac dysfunction, is caused by progressive mitral annulus stretching as a result of LA remodeling and dysfunction (i.e., LA myopathy) [51]. Because even mild functional MR is associated with upstream impairments in pulmonary vascular function, RV dysfunction and higher incidence of TR, its presence in HFpEF (even without atrial fibrillation) can be considered as a clear evidence of a higher risk for further cardiac worsening [51].
Timely referral of suitable patients is a key to good outcomes of HTx. The
persistence of a low CO syndrome unmanageable by the available therapeutic
options (e.g., maximal medical therapy, CRT, MV repair), is a main indication for
HTx and, therefore, close monitoring of SV by ECHO can be particularly useful for
timely placement of patients on the HTx waiting list [14]. First it
has to be decided whether the patient is eligible for both HTx and VAD
implantation, or only for one of them. Those eligible for HTx should be timely
listed, but VAD support (LVAD
There is general agreement about the importance of ECHO in ascertaining the necessity and feasibility of LVAD support, taking decisions about the need for an additional assist device also for the right-sided heart in LVAD candidates, intraoperative guiding of VAD implantation and adjustment of the assist device flow, as well as post-operative monitoring of the ventricular support including accurate monitoring of the right-sided heart in LVAD recipients (Table 2, Ref. [3, 4, 5, 6, 7, 11, 22, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120]) [2, 54, 121, 122, 123]. In addition, ECHO is the first-line tool for detection and estimation of ventricular reverse remodeling and functional improvement in response to the VAD support, selection of potential weaning candidates, weaning decision-making and monitoring of cardiac function after VAD explantation [54].
| Role of ECHO | Usefulness | Challenges and limitations |
| Selection of LVAD candidates [55, 56, 57, 58, 59, 60, 61] | - Detection of cardiac abnormalities (e.g., thrombi, PFO, endocarditis, valvular abnormalities) and aortic diseases (e.g., aneurisma, atheroma, coarctation) that increase the risk for complications. | - The low AR velocities resulting from the low ∆P between the aorta and the LV during the diastole hamper the quantification of AR. Routine calculation of both |
| Pre-operative prediction of RVF after LVAD implantation [11, 22, 60, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 81, 85, 88] | - Pre-operative RVEDD |
- RVEDD, FAC |
| - The use of integrative parameter combinations, which allow the evaluation of RV alterations in connection with RV afterload, appeared more reliable for prediction of RV anatomical and functional responses to the LVAD support. TAPSE/PAPsyst, PSSrL • ΔP |
- The distinctly load dependency of RV geometry, size and function is the main reason for the low predictive value of the individual RV anatomical and/or functional ECHO-derived parameters for the persistence or new occurrence RVF during LVAD support. This also includes the impact of TR, which can induce RVEF, FAC | |
| Decision on the necessity for additional RVAD [11, 22, 54, 65, 69, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88] | - ECHO-derived data on cardiac anatomy and function are mandatory for decision making in favor or against the additional consideration of a mechanical support also for the RV. ECHO mainly enables the identification of patients without the need of RVAD support after LVAD implantation. The LAI |
- The prediction of the need for RV mechanical support by ECHO and/or by any other available tool for evaluation of the right-sided heart is particularly challenging because many patients with post-LVAD RVF can improve and finally even normalize their RV function thanks to the elimination of pulmonary congestion and the possibility to use maximum pulmonary vasodilation therapy. Another important aspect is the fact that patients who need post-operatively an emergent implantation of a mechanical support for the RV are only a minority of LVAD recipients with RVF and the pre-operative identification of these patients still remains a major challenge because both unnecessary BiVAD implantation and delayed transition from LVAD to BiVAD support may have adverse impact on patient outcome. |
| Assessment of recovery and weaning decision-making [3, 4, 5, 6, 7, 54, 57, 81, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120] | - ECHO is the main tool for selection of weaning candidates, evaluation of the functional relevancy and stability of recovery, and together with RHC, also mandatory for decision-making in favor of or against VAD explantation. | - No single anatomical or functional ECHO parameter allows alone the evaluation of recovery. |
| - Multiparametric ECHO evaluation of cardiac anatomy and function plus integrative interpretation of measurements are essential requirements for assessment of recovery. Pre-explant ECHO can predict post-explant outcome only in patients with normal off-pump RHC data. | ||
| ECHO, echocardiography; LVAD, left ventricular assist device; PFO, patent
foramen ovale; | ||
Preoperative ECHO is indispensable for LVAD candidate selection which is a
demanding task due to the fact that end-stage HF caused by
severe LV dysfunction is a multifaceted syndrome with
multiple risk factors for an unfavorable course of
LVAD therapy [55, 56, 57, 58]. Small LV chamber dimensions in patients with
restrictive cardiomyopathy pose particular challenges for a LVAD
implantation. Thus, LVEDD
ECHO is the preferred screening tool for detection of pathological alterations of the heart and thoracic aorta that enhance the risk for potentially life-threatening events, or may even contraindicate LVAD insertion [58]. However, in patients with evidence of aortic abnormalities, ECHO alone can be insufficient for accurate assessment which will necessitate the use of additional imaging techniques (e.g., CT scan).
TTE and TEE (transthoracic and transesophageal echocardiography, respectively) are particularly useful for pre-implant detection of possible thrombi, patent foramen ovale (PFO), valve abnormalities, endocarditis or aortic diseases. Special attention should also be paid on indications that suggest the presence of PFO [56, 57, 58, 59].
Exclusion of relevant (
RVF is a major cause of morbidity and mortality in LVAD recipients [54]. Because of their particularly strong load dependency, the RV size, geometry, and function are often altered already before LVAD implantation. Reverse remodeling and functional improvement of the RV during mechanical support of the LV depend on the reversibility of both the RV myocardial alterations and the pathologic circulatory and metabolic changes triggered by imbalanced neurohumoral/inflammatory reactions to the insufficient CO and the HF-related end-organ failure (especially renal and liver failure) [54, 125]. Conversely, the reversibility of pathologic neurohumoral activation and end-organ failure during mechanical LV support depend on the reversibility of RV dysfunction. Prediction of the impact of mechanical LV support on RV function and on patient outcome without additional RV support becomes therefore particularly difficult. This also explains the fact that the majority of the scoring-systems currently used for supporting the decision in favour of an LVAD or BiVAD therapy are based rather on parameters related to the pre-implant severity of HF-related multiorgan dysfunction than on parameters directly related to RV contractile performance and its potential reversibility [10, 54, 62, 63, 126, 127]. This could be the reason for the differences regarding the risk prediction for RVF during LVAD support revealed by different studies using the same scoring-system which did not include parameters of right-sided heart anatomy and function.
Several risk-scores (with and without incorporated ECHO-parameters) for preoperative prediction of RVF during LVAD support can also predict the mortality risk after LVAD insertion [63, 64, 65, 127, 128, 129, 130, 131]. The best capability to identify LVAD candidates at high risk for postoperative death was found for complex scoring-systems that include preoperative hemodynamic, laboratory and clinical parameters that reflect mainly end-organ dysfunction which on its part can remain a serious risk for death even after improvement of RV function due to the LVAD-promoted reduction of the PVR [57, 63, 129, 130, 131]. Nevertheless, addition of ECHO-derived variables for evaluation of RV size, geometry and function to different risk-scores based exclusively on symptoms parameters and reflecting the preoperative severity of HF-related multiorgan dysfunction, can improve the preoperative prediction of patient outcome after LVAD implantation [64, 66, 67].
Preoperative prediction of myocardial recovery during prolonged LVAD or BiVAD support, sufficient for later explantation of the VAD is not possible, neither by ECHO, nor by any other investigation [4, 5, 6, 17, 54].
ECHO allows pre-implant identification of LVAD candidates with and without the
necessary preconditions to become and/or remain free from RVF after LVAD
implantation, because those with inadequate responses of the RV to the support of
the LV have already pre-operatively more altered morphological and functional
right-sided heart parameter values [62, 63, 64, 67, 68, 126, 132]. Thus, patients with RVF
during mechanical LV support showed preoperatively larger RV end-diastolic
diameters (RVEDD) with greater S/L (short/long axis) ratios, greater RV/LV
diameter ratios, lower RVEF and FAC
Preoperative identification of LVAD candidates who necessitate mechanical assistance for both ventricles is essential for optimal postoperative outcomes. Compared to BiVADs, LVADs are safer for the patients and offer a higher quality of life, but even if LV myocardial alterations and dysfunction were the primary cause of advanced HF, RV dysfunction of different severity is nearly always present. Unfortunately, although several ECHO parameter alterations which are recognized as risk-factors for RHF after LVAD implantation can predict preoperatively the absence of RVF after LVAD implantation, only few of them can also predict the occurrence of RVF during the LVAD support [11]. Prediction of the need for additional mechanical support also for the RV is difficult, because many patients with post-LVAD RVF can improve and finally even normalize their RV function thanks to the elimination of pulmonary congestion and the possibility to use a maximum pulmonary vasodilation therapy [11, 69, 73]. However, such prediction would be important because LVAD recipients who receive a RVAD much later after LVAD surgery have a less favorable outcome than those with simultaneous LVAD and RVAD insertion [54, 139]. It should also be considered that in many patients at high risk for RHF after LVAD surgery, the concurrent insertion of a durable LVAD and an easily removable temporary RVAD can avoid the implantation of a durable BiVAD [54, 139].
There are major difficulties and methodological weaknesses in the evaluation of
the RV by ECHO which can greatly influence the diagnostic and prognostic
reliability of this technique in VAD candidates [74, 75, 76, 77, 78]. Because of the
distinctly high dependency of RV size, geometry and pump function on hemodynamic
loading conditions, a major limitation for the diagnostic and prognostic value of
individual RV parameters is their frequent alteration already before any
impairment of RV myocardial contractility. This contributes to the limited
predictive value of pre-operatively measured ECHO parameters which had proved to
be relevant risk-factors for RV failure after LVAD insertion. Parameters like
FAC
The ECHO-based assessment of the RV can be improved by using also different
combinations of parameters which include measures that reflect the RV afterload
[11, 22, 80, 81, 82, 83, 84, 85, 139, 140]. Because the RV stroke work index (SWI
Other composite ECHO-derived parameters and indexes, which combine longitudinal displacement and afterload (i.e TAPSE/PAPS and TAPSE/PVR), or myocardial shortening velocity and load (i.e., “afterload corrected PSSrL”) were also found appropriate for evaluation of the RV myocardial contractile ability [11, 77, 84, 140]. The ratio of TAPSE/systolic PAP appeared to be an independent predictor of mortality in patients with congestive HF due to primary impaired LV function [65]. Because PVR has a decisive impact on RV systolic function, its inclusion into ECHO-derived composite parameters can be useful for evaluation of the relationship between increased afterload and RV function [22]. A composite ECHO-derived parameter which reflects that relationship in a simplified manner is the “RV ejection efficiency” (RVEe), defined as RVEe=TAPSE/PVR [22]. Using TAPSE as a surrogate for RV ejection and ECHO-derived PVR (PVR=TR peak velocity/RV outflow tract VTI) as a surrogate for the RHC-derived PVR, the easy calculable RVEe could be useful for assessment of RV functional abilities [86, 87]. However, future studies are needed to determine whether the ECHO-derived RVEe can be able to predict RV function post-LVAD RV function.
The RV free wall “afterload-corrected PSSrL”, defined as PSSrL •
Evaluation RV ability to improve or even normalize its function after lowering
its afterload is enabled by calculation of the LAI
The replacement of
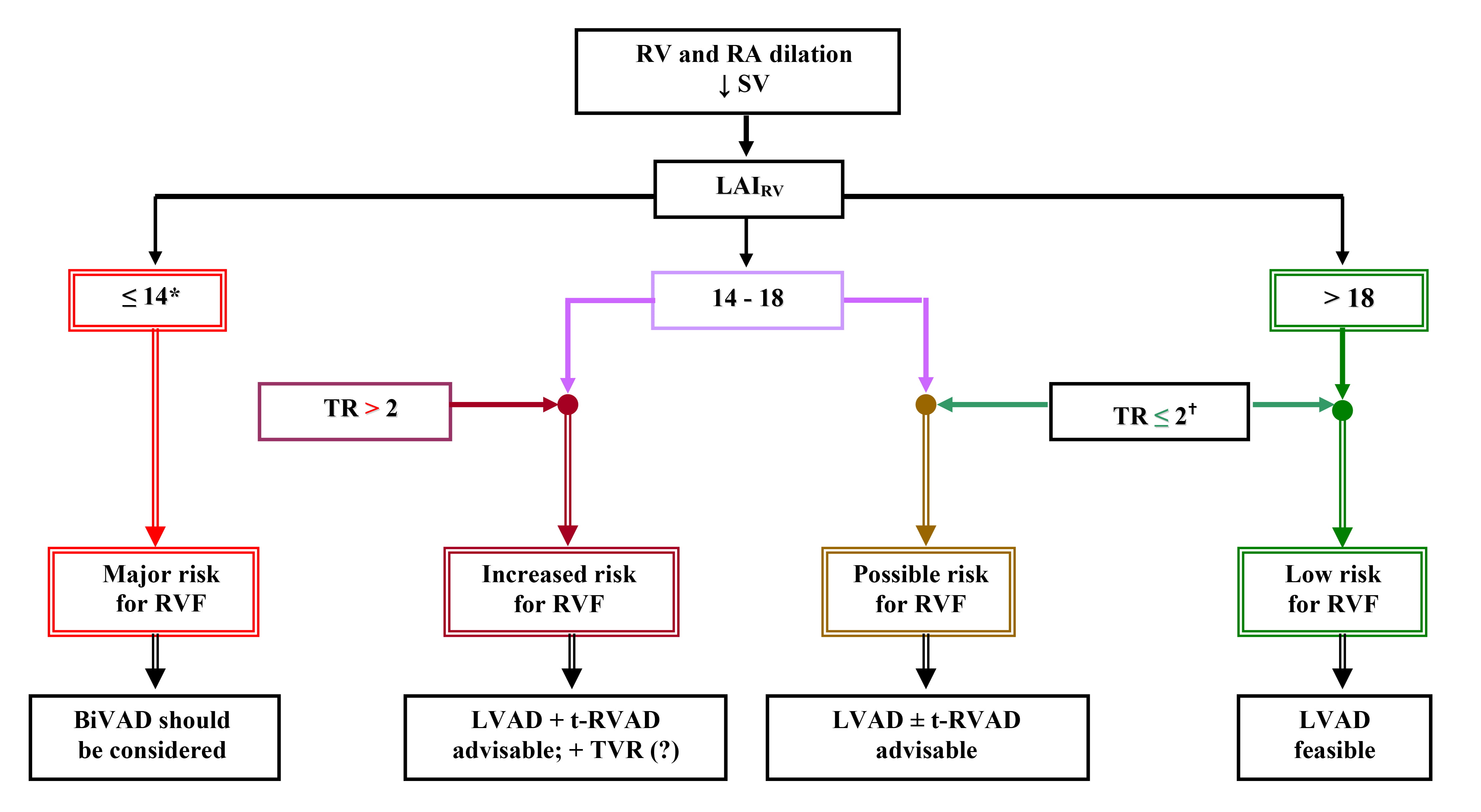 Fig. 1.
Fig. 1.ECHO-based clinical algorithm for device selection in patients
with end-stage non-ischemic chronic cardiomyopathy [9, 11, 79, 88, 91]. ECHO,
echocardiography; RV, right ventricle; RA, right atrium;
TEE is useful for intraoperative exclusion of a PFO that may have been masked by increased LA pressure, as well as final evaluation of cardiac valve and RV function. Intraoperative TEE also helps in guiding cannula placement and optimization of ventricular support by the VAD.
Examination of inflow and outflow cannula (IC and OC, respectively) position and anastomoses is particularly important [57, 58, 142, 143, 144, 145, 146, 147]. The IC, should be parallel to the septum, directed toward the MV and not too close to the ventricular walls [58, 142, 143, 144]. Excessive IC deviation towards the septum necessitates intra-operative revision. Short-term reduction of LV volume by increasing the pump rate facilitates the confirmation of the correct position of the IC [145]. Real time 3D-ECHO allows better visualization of the IC within the LV [58].
Flow-Doppler techniques allow the assessment of blood flow at the cannulas. The normal color flow-Doppler pattern and velocities, as well as the flow alterations in pathological circumstances are shown in Table 3 (Ref. [57, 58, 139, 144, 145, 146, 148, 149, 150, 151, 152, 153, 154]).
| Optimal LV support [58, 145, 149] | Underfilling/Excessive unloading [139, 144, 146, 151, 152, 153] | Overfilling/Impaired LVAD support [145, 146, 150, 154] |
| ● For CF-LVADs, the normal flow pattern at the cannulas revealed by flow-Doppler techniques is characterized by a laminar and unidirectional blood flow with systolic augmentation and peak velocity of 1–2 m/s (depending on intrinsic LV function and preload). Axial-flow shows at the IC a peak filling velocity between 0.7–2 m/s. At the OC, the normal flow peak velocity varies between 0.5–2 m/s. | ● Rapid RV geometry alteration with TR increase and worsening of RV function associated with left-ward septum-shift, no AV opening, and extreme LV diameter decrease ( |
● The combination of low pump flow with increased power values resulting in impaired LV support can arise by LVAD malfunction or by increased afterload. |
| ● The major causes of LV overfilling are cannula regurgitation, cannula obstruction and LVAD malfunction. | ||
| ● For pulsatile pumps, the peak flows in the IC and OC are usually higher than for CF-LVADs, but not beyond 2.3 m/s. | ● The combination of low pump flow with normal or low power (and low PI) values indicates reduced LVAD preload (i.e., under-fillling) which is most commonly related to RV failure, significant TR, or hypovolemia. | ● LVAD malfunction can result from primary failure of different device components or by thrombi inside the pump and/or the cannulas. |
| ● A neutral septum position between the ventricles denotes adequate LV decompression. | ||
| High right-sided heart pressure, dyssynchrony from inter-ventricular contraction delay, ischemic wall motion abnormalities, post-sternotomy state, pericardial constriction, and RV pacing can complicate the assessment of septum position. | ● Underfilling of the supported LV also shrinks the cavity size and can cause obstruction of the IC orifice by suction of the myocardial or chordal structures. | ● In CF-LVADs cannula regurgitation can occur in patients with high systemic vascular resistance. In pulsatile VADs, failure of the one-way valves, lead to regurgitation. |
| ● Reduced aortic valve opening frequency and degree. | ● ECHO signs of underfilling are: | ● Cannula obstruction can result from mal-position, kinking or thrombosis (complete, partial, or intermittent). |
| - reduction of LVEDD | ||
| - reduction of LA size | ● Aliased high velocity turbulent flows at the cannulas orifices (i.e., | |
| - leftward septum shift | ||
| - reduction of AV openings | ||
| - signs for reduced filling pressure | ||
| (decrease in E-wave velocity, decrease in E/E’ ratio prolongation of E-DT) | ||
| ● Major ECHO signs for impaired LVAD support are LV dilation, more frequent AV openings, rightward septum-shift, increased spontaneous “ECHO-contrast” in the left-sided heart, LA dilation, new appearance or increase of MR, RV and RA diameter increase, increasing TR with increase in the peak velocity of the TR-jet, as well as inferior vena cava dilation. | ||
| ● Differentiation of systemic vasoconstriction from mechanical problems is crucial because of the completely different therapeutic approaches. The AV response, which will stay closed in patients with severe vasoconstriction, is particularly useful for differentiation. | ||
| LVAD, left ventricular assist device; CF, continuous flow; LV and RV, left and right ventricle, respectively; IC and OC, inflow and outflow cannula, respectively; TR, tricuspid regurgitation; AV, aortic valve; ECHO, echocardiography; LVEDD, left ventricular end-diastolic diameter; PI, pulsatility index; LA and RA, left and right atrium, respectively; E-wave, early diastolic peak mitral flow velocity; E/E’, early diastolic peak mitral flow to mitral annular velocity ratio; E-DT, E-wave deceleration time; MR, mitral regurgitation. | ||
During LVAD implantation, TEE is mandatory for intraoperative assessment and optimization of LV unloading, monitoring of RV size, geometry and function, as well as close observation of possible changes in TR and/or RA size [139].
The goal of LVAD implantation is to increase cardiac output while decreasing
filling pressures. The main parameters for assessment and monitoring of LV
unloading are ventricular and atrial septum position, LVEDD and AV opening
[144, 146]. After LVAD initiation, sudden excessive LV unloading can induce
immediate alterations of RV geometry associated with RV dysfunction (up to acute
RVF). RV overdistension should therefore be absolutely prevented. Neutral
ventricular and atrial septum position is essential for prevention of RV and/or
TV ring geometry alterations [146, 148, 149]. Preoperative RV pathological
remodeling and dysfunction is often reversible by LV pressure relief-induced
reduction of RV afterload plus enhanced pulmonary vasodilation medical therapy
[53, 155]. If LVAD initiation remains without any impact on RV size, geometry and
function, additional insertion of a temporary RVAD might be useful because its
placement concomitantly with LVAD implantation increases the postoperative
survival chances compared to delayed RVAD implantation [156, 157, 158]. LVAD-induced
decrease of RV afterload is usually followed by regression of TR. Nevertheless,
persistence of more than moderate TR has a negative impact on RV improvement and
facilitates further aggravation of TR, which often will need a subsequent
surgical correction [148, 149, 156]. Simultaneous LVAD insertion and TVr in
patients with pre-operative
After starting the LVAD, TEE is mandatory for re-evaluation of AV coaptation,
because the reduction of LV pressure increases the transvalvular
LVAD initiation can create a pressure gradient from the RA to the LA, with
right-to-left shunting in the presence of a PFO. Intraoperative TEE is therefore
mandatory because PFO needs to be closed during VAD surgery [148, 163]. To improve
the visualization of that shunting by TEE it is useful to induce a short-term
shifting of the atrial septum to the left by increasing the LVAD flow
[148]. After starting the LVAD, TEE is also necessary for guiding pump
speed adjustment for optimizing LV unloading without any septum shift and, if
possible, with maintaining a systolic AV opening at least once every 3 heart
cycles [57, 163, 164]. Even if a maximum attainable LVAD support could be more
advantageous for promoting LV reverse remodeling and increasing patient exercise
capacity, sub-maximal support in order to keep a near physiologic AV function
should be preferred in outpatients, because a full support enhances the risk of
AV leaflet fusion, suction alarms, aortic rood thrombosis and also the emergence
and/or aggravation of AR by increasing the
ECHO is the major cardiac imaging technique for routine monitoring of VAD therapy. It is necessary for evaluation and optimization of LV support, surveillance of the right-sided heart, and early identification of certain abnormalities which could affect the results of VAD therapy. It is also a cornerstone for assessment and valuation of unloading-promoted myocardial recovery and decision-making about a possible weaning of certain patients from the VAD.
Accurate LV support is decisive for patient outcome after LVAD implantation and ECHO is the preferred method for routine surveillance of LVAD recipients. Of major importance for evaluation of LVAD function are the septum position (particularly the interatrial septum), the status of AV opening, the flow pulsatility in the OC of axial-LVADs, TR velocity, estimation of RA pressure, as well as LVAD system output (most useful by direct TTE-derived measurement) and total cardiac output estimation [144].
Impairment of LVAD support causes LV overfilling, whereas, underfilling is most commonly related to RVF, significant TR, or hypovolemia [144, 145, 146, 150, 151, 152, 153]. The major ECHO-signs for overfilling and underfilling are described in Table 3. LV unloading is best reflected by LVEDD, septal position and AV opening which are therefore also the most often used parameters for optimization of LVAD settings [165, 166]. For CF-LVADs, the adjustment of the rotor speed should strive to bring the septum in a neutral position and to ensure that the AV opens minimally for a very short time and/or to a very low frequency at the lowest possible LVEDD [145, 165].
Regular follow-up controls of AV opening are essential for surveillance and if
necessary also for readjustment of LVAD support. AV opening depends on the
Many potentially life-threatening anomalies inside and outside de supported heart, especially tamponade, intracardiac thrombi, vegetations, AR and cannula obstruction are easily identifiable with ECHO.
Timely identification of partial, intermittent or complete cannula obstructions, resulting from kinking, malposition, thrombosis, or vegetations is decisive for achieving an optimal outcome for LVAD recipients. Excessive unloading-induced reduction of LV size which increases the risk of IC obstruction by cardiac structures (“suckdown” of papillary muscles, trabeculations, septum and chordal structures) plus preload reduction by tamponade or dehydration, which also increase the susceptibility for IC orifice obstruction, are easily demonstrable by ECHO [57, 145]. In some patients with unloading-promoted amelioration of LV myocardial contractility, the increasing amount of blood ejected by the LV into the aorta can lead to excessive unloading which necessitates readjustment of the LVAD function to avoid suction events. In patients with CF-LVADs, low diastolic flow velocity and high systolic/diastolic flow velocity ratio suggest thrombosis-related LVAD malfunction [154]. If a misalignment between flow and ultrasound beam direction or a relevant SV provided by the supported LV can be excluded, intermittent low peak flow velocity may suggest LVAD malfunction. Low velocities at the IC may indicate thrombosis inside the IC. Backflow at the IC indicates malfunction of the device. Whereas in pulsatile devices, the malfunction of the one-way valve leads to regurgitation, in CF-LVADs regurgitation occurs when pumps cannot overcome the systemic vascular resistance. For the IC the most optimal Doppler-angle is provided by apical views but measurements can be impaired by reverberation-artifacts from the pump. For the OC in the ascending aorta, the best visualization by TTE is often provided by right parasternal views but adequate imaging is usually quite challenging [148, 151].
Continuous ECHO controls are important for the monitoring of AV function because relevant AR may occur also months after LVAD insertion and newly developed AS, as a result of cusp fusion, can also be a complication of LVAD support [145, 167, 168]. Because the lack of AV opening is an important risk factor for thrombosis, ECHO-surveillance of LVAD recipients with irrelevant AV opening should be particularly focused on the search for LVOT, AV and aortic root thrombi [168, 169].
In LVAD recipients, AR can be present also during LV systole if LV pressure remains below the aortic pressure. Conventional severity grading of AR using the vena contracta proximal isovelocity surface area and the jet-width/LVOT-diameter ratio could underestimate the severity of AR because it does not consider the pancyclic nature of AR-jets in LVAD recipients [167]. Newly developed ECHO parameters like systolic-to-diastolic peak velocity ratio (S/D-ratio) and OC diastolic acceleration and can be more reliable for AR grading in LVAD-supported patients [167].
The evaluation of LVEDD, AV opening-time and mitral inflow E-DT changes during ECHO ramp-tests (changes of device speed) can also be helpful in diagnosing CF-LVAD malfunction in the setting of pump thrombosis [170]. Nevertheless, small thrombi can induce false negative results, whereas high mean arterial pressure or AR can generate false positive ramp-tests [171].
Even though LVAD initiation can lower the PVR already intraoperatively, and a
durable LVAD support can further diminish the PVR and thereby also promote
reverse remodeling and functional improvement of the RV, neither early nor late RVF after LVAD insertion are completely evitable, even by an
additional use of enhanced pulmonary vasodilative therapy [139, 172]. Continuous
monitoring of the right-sided heart is therefore mandatory and TTE is
particularly useful for early detection of RV anatomical and functional
alterations [9, 53, 54]. Progressive increase of RVEDD, RA volume and TR are key
indicators for imminent or already present right-sided HF and additional signs
for non-optimal LV unloading suggest that the alteration of RV function could be
the consequence of inadequate LV mechanical support [9, 53, 54]. Persistence of
right-sided heart dilation and no reduction of TR, despite optimal LV unloading,
indicate either reduced RV myocardial contractility or insufficient reduction of
RV hemodynamic overload or both. Main causes for RV overload despite proper LVAD
function are pressure overload by high pre-capillary vascular resistance
(irreducible by LV unloading) and/or volume overload (more often caused by severe
kidney dysfunction and/or TR). In such patients, normal or reduced PVR indicate
that reduced contractile function must be the major cause for RV dysfunction.
Reduced LAI
Compared to the prediction of early RVF, the prediction of late RVF is more challenging, particularly in patients supported by a CF-LVAD in the presence of a CRT-defibrillator prior to the LVAD implantation which was found associated with significantly higher incidence late RVF [173, 174]. Given that the underlying pathomechanisms of this more frequent complication is not clarified, closer long-term ECHO monitoring of the right-sided heart is of particular importance in these patients.
By lowering the myocardial wall-tension and optimization of blood flow into vital organs, LVADs and BiVADs can remove the major pathophysiological stimuli for cardiac remodeling and interrupt the vicious circle of ventricular dilation and reduction of the efficiency of myocardial contraction [89, 90, 91, 92, 93, 94]. All this facilitates reverse remodeling even in patients with chronic NICMP and can be accompanied by clinically relevant reversal of LV structural and functional alterations allowing sometimes even removal of the VAD. Weaning from long-term VADs was initially performed only in patients with acute forms of HF [95]. This is quite understandable because the reverse of acute HF during a longer VAD support is not unusual whereas, until quite recently, cardiac remodeling processes and functional alterations in chronic HF and were thought to be progressive, unidirectional and irreversible.
The first worldwide elective explantations of LVADs in patients with idiopathic
DCM were carried out in Berlin, in 1995 (4 adult men, age 47
ECHO is the major tool for evaluation of myocardial recovery during LVAD support
with a key role in both selection of weaning candidates and weaning
decision-making [6, 54, 81, 94, 108, 109]. Regular TTE screenings with normal LVAD
support are necessary for identification of potential weaning candidates.
Potential weaning candidates are those with signs of LV reverse remodeling
(including normalization of the LVEDD), improvement of wall motion (LV fractional
shortening
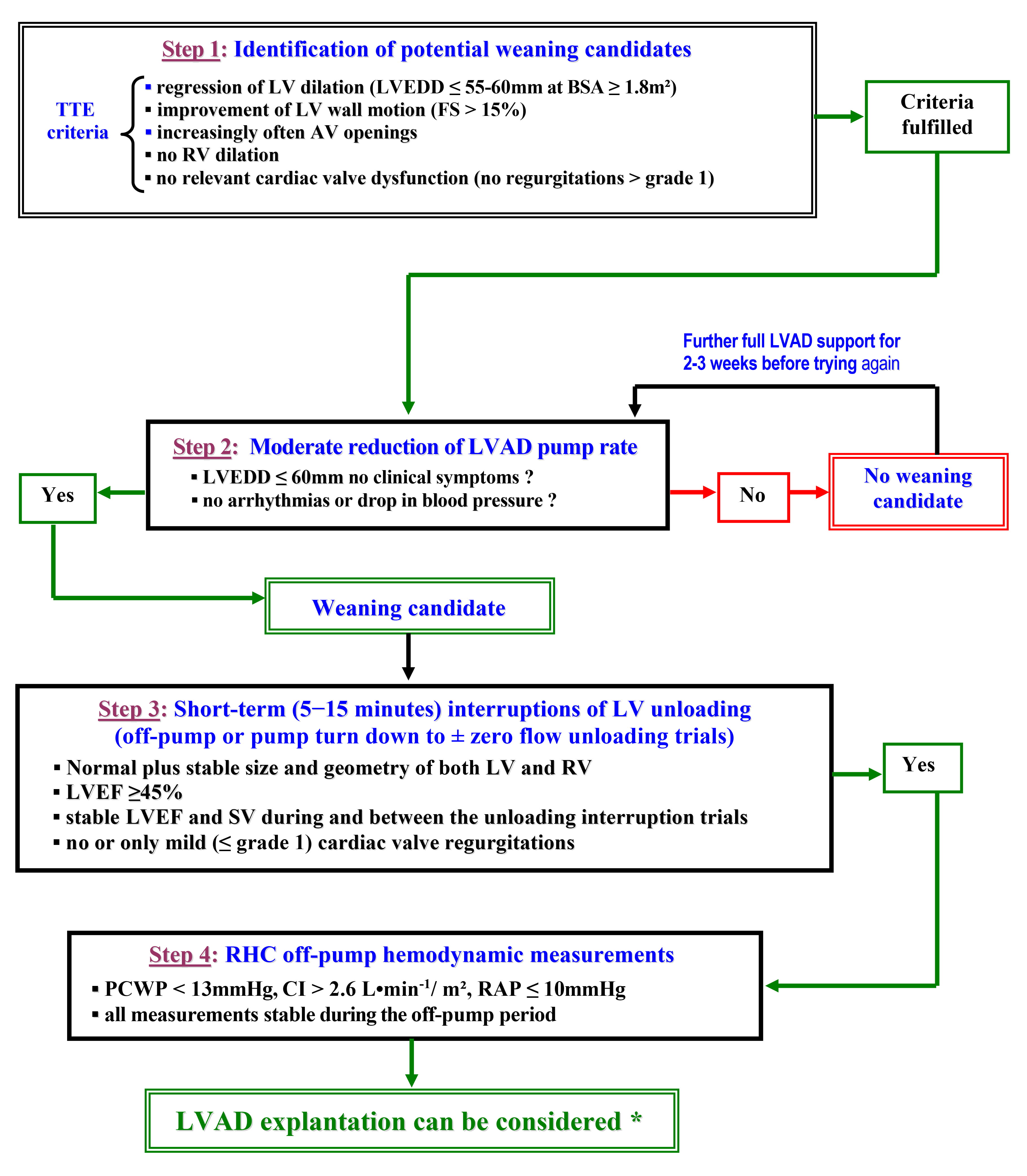 Fig. 2.
Fig. 2.Major steps for evaluation of cardiac improvement in LVAD recipients [6, 91, 94, 102, 107, 111, 119]. TTE, transthoracic echocardiography; LV and RV, left and right ventricle, respectively; LVEDD, LV end- diastolic diameter; BSA, body surface area; FS, fractional shortening; AV, aortic valve; LVAD, LV assist device; LVEF, LV ejection fraction; SV, stroke volume; RHC, right heart catheterization; PCWP, pulmonary capillary wedge pressure; CI, cardiac index; RAP, right atrial pressure. * particularly in patients with adequate renal, hepatic, and pulmonary function.
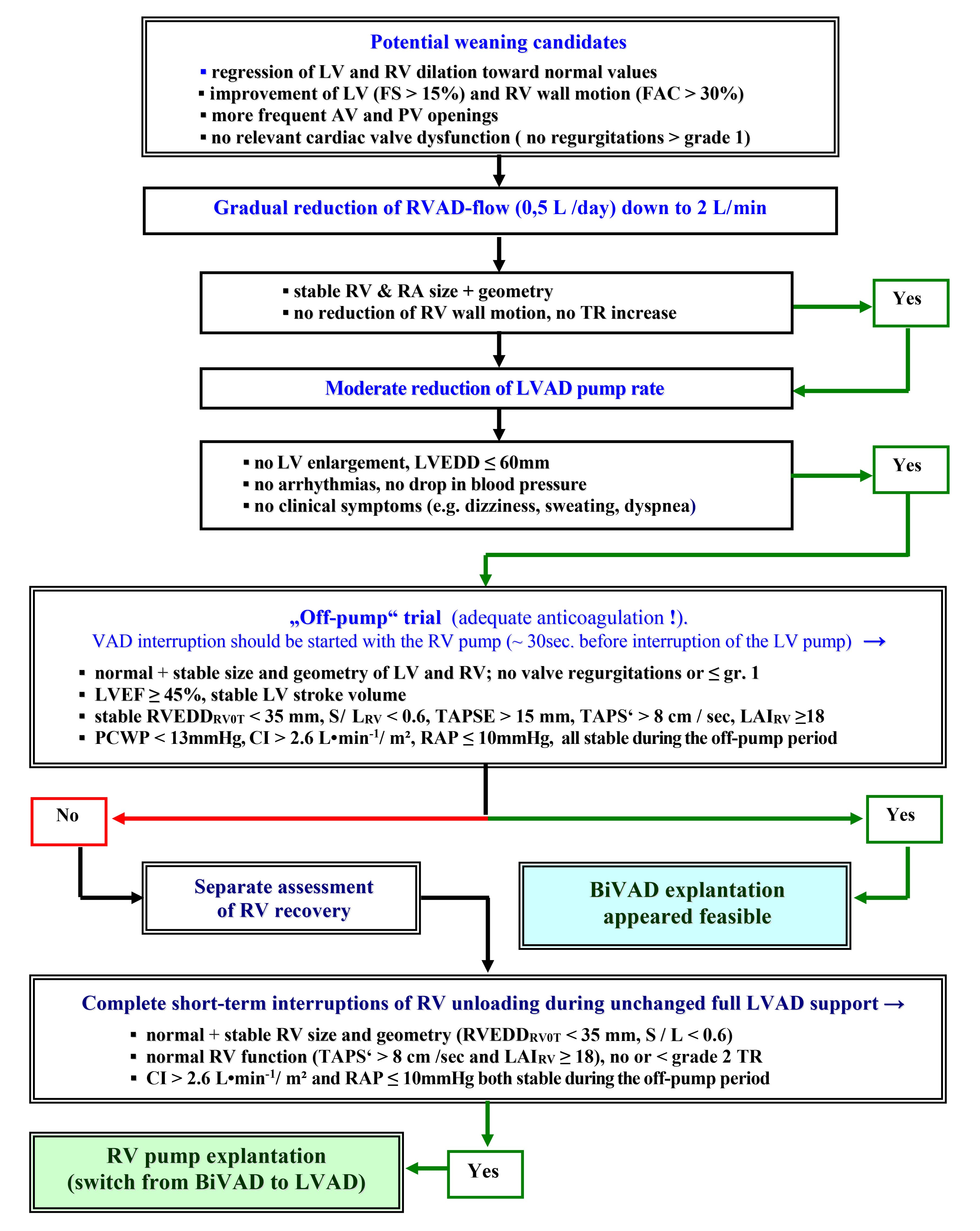 Fig. 3.
Fig. 3.Recovery assessment and weaning from biventricular assist
devices [91, 94, 102, 107, 119]. LV and RV, left and right ventricle, respecively;
FS, fractional shortening; FAC, fractional area change; AV, aortic valve; PV,
pulmonary valve; LVAD, RVAD and BiVAD, LV, RV and biventricular assist device,
respectively; RA, right atrium; TR, tricuspid regurgitation; LVEDD, LV
end-diastolic diameter; TAPSE and TAPS’, tricuspid annulus peak systolic
excursion and velocity, respectively; RVEDD
Before the first interruption of VAD support, a stepwise ECHO-guided pump-rate reduction is useful to determine whether complete interruptions are feasible. If a gradual reduction of the support already provokes dizziness, sweating or arrhythmias, a complete stop of VAD support is both risky and useless. If the patient stays asymptomatic but the LVEDD rises beyond 60 mm, and/or the right-sided heart shows morphological and/or functional instability (RA dilation, increasing TR, SV reduction), VAD support interruption trials are also senseless, because the patient is at present not a weaning candidate [94].
In weaning candidates, ECHO-assessment of recovery is usually based on the
findings gained during repeated short-term zero-unloading trials (pulsatile pump
stop or CF-pump turn-down until reaching a
Before LVAD support interruption trials, heparin administration is necessary to
prevent thrombus formation inside the pump (60–100 IU/kg according to the
prothrombin time) [6, 175]. Patients with heparin-induced thrombocytopenia should
receive a synthetic thrombin inhibitor (e.g., argatroban infusion 2
ECHO evaluation of cardiac reverse remodeling and functional recovery should be as extensive as possible with inclusion of tissue-Doppler imaging (TDI) and STE measurements [111, 115, 175]. Advantages of STE are its ability to differentiate between active and passive movement of different myocardial segments, the angle independency of measurements, the possibility to quantify intra-ventricular asynchrony and dyssynergy, as well as the possibility to evaluate visually not assessable components of myocardial function (particularly longitudinal deformation) [75, 114, 116, 117, 118]. A major limitation of STE is the dependence on image quality. Therefore, not all suitable parameters for weaning decisions can be reliably measured in all patients especially if Doppler and STE examinations had to be performed during rotor-speed reduction (as in case of CF-LVAD recipients), instead of complete pump stop [106].
ECHO and RHC are fundamental cornerstones for weaning decisions
[81, 113, 117, 175]. The predictive value of ECHO data for the stability of
post-explant heart function are shown in Table 4 (Ref. [4, 5, 6, 7, 54, 81, 91, 94, 102, 106, 111, 118, 120, 175]). At rest, and without LVAD support, LVEF 45% plus normal
LVEDD are weaning requirements [81, 94, 106, 111, 119]. Their stability after
maximum improvement over the next 2 to 4 weeks between, and also during follow-up
off-pump tests, appeared predictive for long-term post-explant outcome in
patients with a CI
| ECHO data obtained at rest during pre-explant LV support interruption trials | |||
| Indicative for successful weaning ( |
Indicative for HF recurrence during the first 3 years after explantation* | ||
| Individual or combined parameters | Predictive value | Individual or combined parameters | Predictive value |
| LVEF |
74%–79% [6, 94, 175] | LVEF 35%–45% at the last off-pump |
87%–88% [6, 94] |
| Stable LVEF |
80%–86% [6] | LVEF 35%–45% in patients with |
Up to 100% [6] |
| LVEF |
78%–88% [6, 175] | Unstable LVEF |
Up to 90% [94] |
| LVEF |
87%–88% [6] | LVEF |
89% and 82%, respectively [6, 94] |
| Stable LVEF |
87% [94, 111] | LVEF |
87% and 85%, respectively [94] |
| Stable LVEF |
94% [94, 175] | LVEF |
83% and 90%, Respectively [94] |
| Stable LV SV (i.e., stable PW Doppler-derived VTI in the LVOT) | ▪ These data are alone not predictive for long-term freedom HF recurrence [94] | LVEF 45%–50% with concurrent MR grade I–II (possible misleading overestimation of LVEF) | ▪All are validated risk factors for early recurrence of HF after LVAD removal [94] |
| Absence or |
▪ Nevertheless, all of them are required pre-conditions for successful weaning [94, 111] | Systemic AP |
▪ Currently there are no accurate figures available for their predictive value for early recurrence of HF after LVAD explantation [94] |
| Absence of or |
Relevant LV diastolic stiffness despite optimal LVEF ( | ||
| No RV dilation (RVOT-EDD |
SV reduction and/or asynchrony/dyssynergy of LV contraction | ||
| RV size and geometry alterations and/or deficient RV adaptation to increased afterload during the last off-pump* trial | |||
| Global radial, circumferential and longitudinal LV strain and strain rate. LV synchrony and synergy of contraction | Their usefulness is undisputed. Their PV has not yet been established [94] | TR (new appearance or accentuation) with or without increase in jet velocity (evidence of pulmonary arterial pressure increase as an answer to higher resistance in the pulmonary circulation) during the last off-pump trial | |
| le,respectively; HF, heart failure; LVEF, LV ejection fraction; LVEDD, LV
end-diastolic diameter; Sm, peak systolic wall motion velocity (measured by the
tissue Doppler at the basal LV posterior wall; SV, stroke volume; VTI,
velocity-time integral; LVOT, LV outflow tract; AR, aortic valve regurgitation;
MR, mitral valve regurgitation; PR, pulmonary valve regutgitation; RVOT-EDD, RV
outflow tract end-diastolic diameter; S/L * Additional details related with the below items can be found in the references: [4, 5, 7, 54, 81, 91, 102, 106, 118, 120]; | |||
ECHO is indispensable for assessment of cardiac anatomy and function in patients referred for HTx or VAD implantation (as BTT or DT) due to end-stage HF related to chronic NICMP. In VAD candidates, ECHO is the first-line screening tool for detection of cardiac risk factors, like PFO, thrombi, valvular abnormalities (e.g., AR and relevant TR) and endocarditis that affect the LVAD therapy, as well as identification of patients who necessitate mechanical support also for the RV.
ECHO is also required for intraoperative guiding of device implantation and optimization of its supporting function, postoperative surveillance of VAD support, surveillance of the right-sided heart in LVAD recipients, search for signs of myocardial recovery and assessment of its clinical significance, as well as for weaning decision-making in patients with relevant cardiac improvement.
Because the preoperative prediction of possible persistence or even aggravation of RV dysfunction during LVAD support is still one of the most difficult problems, a major future objective should be the further validation of the newly introduced ECHO-based composite parameters which focus on RV adaptability to increased loading conditions, and to include into established RHF risk scores, based exclusively on parameters reflecting the severity of HF-related multi-organ dysfunction, also ECHO measurements which reflect the right-sided heart anatomy and function. All this would improve the preoperative prediction of post-implant RHF in LVAD candidates.
Thanks to ECHO, which has decisively contributed to the key finding that prolonged LVAD support can trigger and further promote myocardial reverse remodeling and improvement of ventricular function up to levels which allow successful LVAD explantation, even in patients with pre-implant chronic NICMP, the previous opinion that end-stage chronic cardiomyopathy is irreversible could be refuted. In patients with normalized and stable RHC-derived hemodynamic parameters measured during short-time decommissioning of the LVAD, ECHO parameters of heart anatomy and function (including their stability between and during the LVAD support interruption trials) can predict post-explant freedom from HF recurrence.
MD drafted, edited, finalized the manuscript.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Michael Dandel is serving as one of the Editorial Board members/Guest editors of this journal. We declare that Michael Dandel had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Yan Topilsky and Fabian Sanchis-Gomar.



