1 Arrhythmia Unit, Onassis Cardiac Surgery Center, 17674 Athens, Greece
2 Department of Cardiology, University of Ioannina Medical School, 45110 Ioannina, Greece
3 Kent and Medway Medical School, CT2 7FS Canterbury, UK
4 Department of Cardiology, Tianjin Key Laboratory of Ionic-Molecular Function of Cardiovascular disease, Tianjin Institute of Cardiology, Second Hospital of Tianjin Medical University, 300211 Tianjin, China
5 Department of Cardiology, Medical School, University of Nicosia, 2408 Nicosia, Cyprus
6 Department of Cardiology, Larnaca General Hospital, 7036 Larnaca, Cyprus
7 Second Department of Cardiology, Evangelismos General Hospital of Athens, 10676 Athens, Greece
8 Electrophysiology and Ablation Unit, Bordeaux University Hospital (CHU), 33600 Bordeaux, France
9 Division of Cardiology, Queen's University, Kingston General Hospital, Kingston, K7K Ontario, Canada
Academic Editor: Peter A. McCullough
Abstract
Brugada syndrome (BrS) is a complex arrhythmogenic disease displaying electrical and micro-structural abnormalities mainly located at the epicardium of the right ventricular outflow tract (RVOT). It is well-known that fibrosis, fatty infiltration, inflammation and reduced gap junction expression have been demonstrated at the epicardial anterior aspect of the RVOT providing the arrhythmogenic substrate for ventricular arrhythmic events in BrS. A number of models have been proposed for the risk stratification of patients with BrS. Endocardial unipolar electroanatomical mapping is an emerging tool that has been reintroduced to identify and quantify epicardial electrical abnormalities. Interestingly, current findings correlate the presence of large-sized endocardial unipolar electroanatomical abnormalities with either ventricular fibrillation inducibility during programmed ventricular stimulation or symptom status. This review aims to present existing data about the role of endocardial unipolar electroanatomical mapping for the identification of RVOT epicardial abnormalities as well as its potential clinical implications in risk stratification of BrS.
Keywords
- Brugada syndrome
- Electroanatomical mapping
- Risk stratification
Brugada syndrome (BrS), a complex arrhythmogenic disease displaying electrical
and micro-structural abnormalities mainly located at the epicardium of the right
ventricular outflow tract (RVOT), is characterized by J-point elevation in the
right precordial leads on the 12-lead electrocardiogram (ECG) and an increased
risk of sudden cardiac death (SCD) due to ventricular fibrillation (VF) [1, 2, 3, 4, 5].
BrS is an inherited disease that is usually transmitted in an autosomal dominant
manner [6]. The BrS related mutations are mainly associated with encoding of
sodium channel, calcium channels and potassium channels [7]. The most important
mutation is the SCN5A, encoding the sodium channel
Fibrosis, fatty infiltration, inflammation and reduced gap junction expression have been demonstrated at the anterior aspect of the RVOT epicardium providing the arrhythmogenic substrate for sustained VF in BrS [4, 5, 11, 12]. Epicardial substrate catheter ablation has been associated with electrocardiogram normalization and ventricular arrhythmia non-inducibility in patients with BrS supporting the current consideration of an epicardial disease [13, 14, 15]. Endocardial unipolar electroanatomical mapping (EAM) is an emerging tool that has been reintroduced into clinical practice for the identification and quantification of epicardial electrical abnormalities in specific cardiomyopathies with several clinical implications, including risk stratification for sudden cardiac death [16]. Risk stratification for BrS patients, and especially of asymptomatic ones, remains challenging [17]. Due to certain limitations of the current risk factors predictors, including programmed ventricular stimulation (PVS), multiparametric scores have been established for risk stratification [18, 19]. This review article questions the accuracy of endocardial unipolar EAM for the identification of RVOT epicardial electrical abnormalities as well as its potential clinical implications in risk stratification of BrS.
Accumulating data suggest that mild structural abnormalities including inflammation, fatty infiltration, and fibrosis, mainly located at the epicardial aspect of the RVOT, provide the arrhythmia substrate in BrS. Lymphocytic myocarditis has been detected in endomyocardial biopsy samples in 77% of patients with BrS without structural abnormalities assessed by imaging techniques [3]. Nademanee et al. [4] studied autoptic samples from BrS patients and showed an epicardial to endocardial gradient of collagen deposition, indicating a progression of the pathological process from the epicardium to endocardium, which correlated with low expression of Connexin 43 (Cx43). Interestingly, BrS has been associated with increased collagen content throughout the right and left ventricular myocardium [12]. Miles et al. [12] evaluated 28 whole hearts from consecutive SCD cases attributed to BrS and 29 hearts from a control group comprised of non-cardiac deaths. Cardiac tissue from BrS decedents displayed a higher amount of collagen in relation to control subjects. The highest collagen accumulation was observed at the epicardial RVOT (geometric mean area 23.7%) [12]. Pieroni et al. [11] performed EAM-guided endomyocardial biopsies demonstrating histopathological abnormalities, including fibrosis and lymphomononuclear infiltrates in the majority of cases. EAM abnormalities manifested as low voltage unipolar and bipolar areas correlated with myocardial inflammation. Zumhagen et al. [20] showed that 55% of BrS patients and normal imaging of the right ventricle (RV) display histopathologic abnormalities (fibrosis, inflammation, and fatty tissue). A similar study identified fatty tissue infiltration, interstitial fibrosis, and lymphocyte infiltration in 52% of BrS with inducible VF [21].
Imaging studies have also provided evidence that subtle cardiac structural abnormalities exist in BrS patients. Computed tomography studies demonstrated wall motion abnormalities within the RVOT in most BrS patients with cardiac arrest [22]. Mild reduction of RVOT ejection fraction, dilatation of the RV inflow tract diameter, and increased RV end-systolic volume have been reported in BrS patients by cardiac magnetic resonance imaging (MRI) [23, 24]. Electromechanical substrate abnormalities during ajmaline challenge including an increase of the electrical substrate in combination with worsening of the mechanical function of the RV, particularly in the anterior free wall of RVOT, have been recently demonstrated [25].
EAM studies have consistently demonstrated the presence of
abnormal substrate at the epicardium of the RVOT. In Nademanee’s seminal report,
abnormal electrograms characterized by low voltage (
Lambiase et al. [29], using high-resolution non-contact endocardial mapping, have demonstrated significant regional conduction delays, reduction in the activation gradient, and formation of lines of functional conduction block at the anterolateral free wall of the RVOT compared to the body and the apex of the RV. Postema et al. [30] have shown conduction slowing and abnormal conduction velocity restitution in the RV in BrS. These data are consistent with the depolarization hypothesis regarding the pathophysiology of BrS [17]. The cardiac intercalated disc is the host of a protein interacting network, called “the connexome”, where different molecules including desmosomes, fascia adherence junctions, gap junctions, and voltage-gated sodium channels interact together to control excitability, electrical coupling, and intercellular adhesion in the heart. Although BrS is considered a primary electrical disease, microstructural abnormalities may enhance the electrical heterogeneity by affecting any of the “connexome” components [31, 32]. Sodium channel activity could be affected by the disruption of any “connexome” components. Cx43 expression that is required for Nav1.5 stability in the intercalated disk membrane is impaired in BrS [4].
The wide “field of view” of endocardial unipolar EAM allows identifying tissue
abnormalities deeper in the mid-myocardium and epicardium. Previous studies,
using different methodologies (either using the 95% confidence interval in
healthy patients or direct comparison of adjacent endocardial and epicardial
points), have proposed different endocardial unipolar voltage cut-off values for
the identification of abnormal epicardial abnormalities of the RVOT. Polin
et al. [33] have initially suggested the 5.5 mV cut-off criterion by
using the 95% confidence interval in healthy control patients. Of note, mapping
was performed using a 4-mm-tip electrode ablation catheter (2-mm-ring electrode
separated by 1-mm spacing), and the sampling points ranged between 105 and 164.
In a high-density EAM study (1019.1
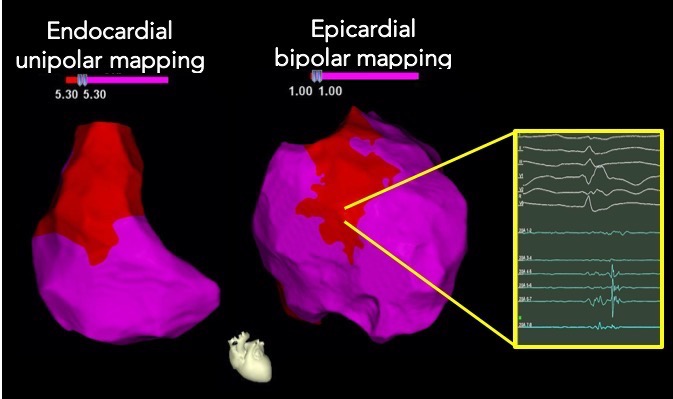 Fig. 1.
Fig. 1.Combined endocardial-epicardial RV/RVOT high density
electroanatomical mapping in BrS. Endocardial unipolar voltage abnormalities
(
The accuracy of endocardial unipolar EAM to identify distant epicardial scar is compromised by several factors, including tissue thickness, the electrode orientation with respect to the tissue, the presence of endocardial scar (low bipolar voltage areas), the electrode size of the mapping catheter, the tissue contact, and the number of sampling points (high-density) [16]. Therefore, the exact “field of view” of unipolar recordings is not completely predictable. A compact area of unipolar voltage abnormalities possibly reflects real anatomic abnormalities deeper into the endocardial layer [3]. By lowering the unipolar voltage slider bar down (from 5 to 4 or 3 mV) and still demonstrating abnormalities, an intramural and/or epicardial scar is suggested with a higher degree of certainty (Fig. 2).
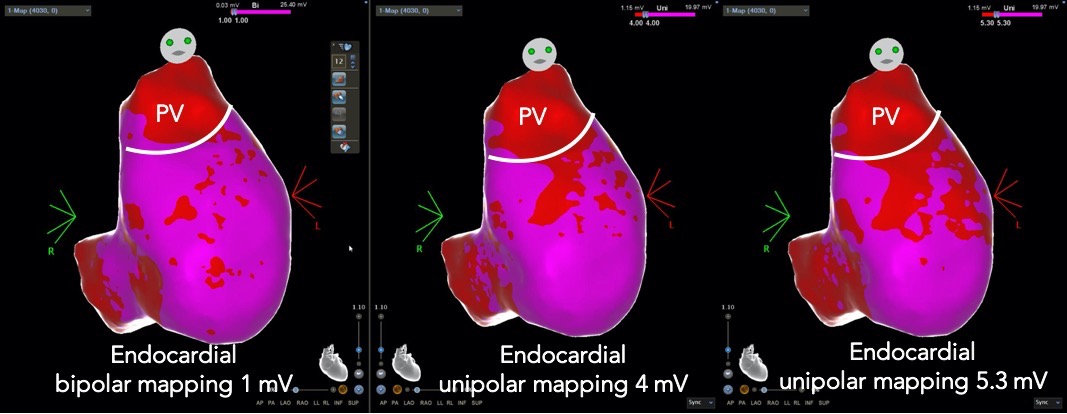 Fig. 2.
Fig. 2.High density electroanatomical mapping showing patchy areas of
low-voltage bipolar signals (
The prognostic significance of VF inducibility during PVS is under continuous debate. There are numerous reasons for the discrepancy of data regarding PVS [16]. First, methodological differences in the stimulation protocols, including the number of extra stimuli, the minimum coupling interval used, and the stimulation site, have a significant impact on PVS results [39]. Second, the inducibility rate may be correlated to the BrS ECG type on the day of the procedure [40]. Third, data regarding the reproducibility of PVS are very limited and do not correlate with clinical presentation [41]. Finally, a false positive VF induction cannot be ruled out.
Endocardial unipolar EAM may improve the prognostic accuracy with respect to
true positive PVS results in BrS. We have initially tested the hypothesis that
RVOT abnormalities detected by high-density endocardial unipolar EAM mapping
predict VF inducibility during PVS [42]. The study population consisted of 17
asymptomatic probands with spontaneous type 1 BrS ECG pattern referred for risk
stratification with PVS. A comprehensive evaluation including late gadolinium
enhancement cardiac MRI ruled out structural heart disease in all patients. An
EAM was considered abnormal in the presence of low-voltage areas
Risk stratification of BrS patients still remains puzzling and represents a great challenge. The prognostic significance of spontaneous type-1 ECG pattern, QRS fragmentation, family history of SCD, VF inducibility during PVS, sinus node dysfunction, and even syncope merit special consideration as single risk factors before implanting an ICD [16]. Multiparametric scores, including the markers mentioned above, have been introduced into clinical practice for a better selection approach [18, 19]. However, novel markers are mandatory to improve risk stratification of BrS patients, especially of asymptomatic ones.
In a recent study including 14 patients with BrS and aborted SCD in the setting
of documented VF and 40 asymptomatic individuals with spontaneous type-1 ECG
patterns, we evaluated the prognostic significance of RVOT electroanatomical
abnormalities [38]. A combined high-density endocardial-epicardial mapping
procedure was performed in all cases with aborted SCD. All symptomatic patients
with aborted SCD displayed abnormal endocardial-epicardial maps. The endocardial
unipolar low-voltage areas (
In BrS, the presence of both repolarization and depolarization abnormalities constitute the functional and anatomical substrates that are possibly implicated in VF maintenance [17]. The mechanisms that maintain VF are partially clarified. The presence of arrhythmogenic substrates related to structural abnormalities is considered a key factor for sustained VF [43, 44]. Fibrillatory mechanisms are strongly related to the underlying electro-architectural substrate including fibrosis patterns and the degree of abnormal gap junction coupling [44]. VF is sustained by a continuous spectrum of mechanisms ranging from organized fibrillation sustained by stable rotational activities to disorganized fibrillation without stable rotational activities [44]. Previous studies have demonstrated that rotational activities are mainly localized to areas of greater fibrosis in perfused cardiomyopathic hearts and low voltage areas during EAM [45, 46]. Based on these findings, the extent of the abnormal substrate may play an essential role in VF maintenance in BrS. A proposed workflow for risk stratification of asymptomatic individuals with BrS referred for an electrophysiological study is shown in the Fig. 3. Subjects displaying both abnormal RVOT substrate and induced VF may be at higher risk for future arrhythmic events.
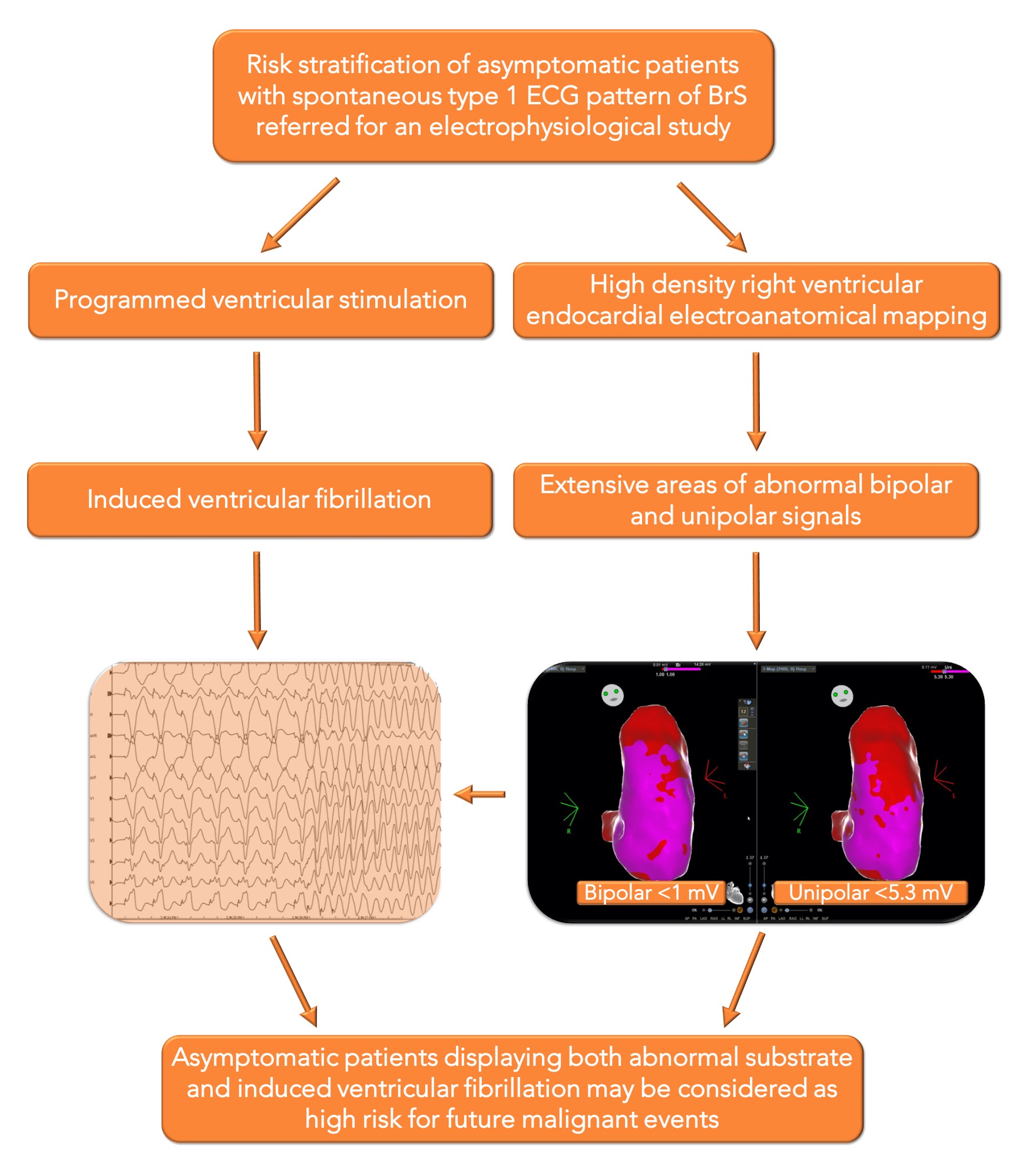 Fig. 3.
Fig. 3.Proposed workflow for risk stratification of asymptomatic individuals with BrS referred for an electrophysiological study. The presence of abnormal RVOT substrate revealed by high-density EAM predicts VF inducibility and is possibly related to symptom status. Subjects with abnormal substrate and induced VF may be at higher risk for future arrhythmic events. Abbreviations, BrS, Brugada syndrome; EAM, electroanatomical mapping; ECG, electrocardiogram; RVOT, right ventricular outflow tract; VF, ventricular fibrillation.
Preliminary findings correlate the presence of large-sized electroanatomical abnormalities with both VF inducibility during PVS and/or symptom status in patients BrS. Due to the certain limitations of PVS, the identification of microstructural alterations in BrS by different imaging modalities, including endocardial unipolar EAM, may assist in the risk stratification of patients with BrS.
KPL—conception of the idea, wrote the first draft, critical revisions, final approval; KV, ME, SD, PK, GT, TL, GB, PN, EP, FS, MH, AB—critical revision, final approval.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Konstantinos P. Letsas is serving as one of the Guest editors of this journal. We declare that Konstantinos P. Letsas had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Peter A. McCullough.



