1 Department of Surgery, Division of Cardiac Surgery, University of Maryland School of Medicine, Baltimore, MD 21201, USA
Academic Editors: Jerome L. Fleg and John Lynn Jefferies
Abstract
The first human heart transplantation was performed by Christian Barnard in 1967. While the technical aspect had been worked out, allograft rejection was a major limitation in the early days of heart transplant. The discovery of cyclosporine revolutionized the field and led to the modern era of transplant. Heart transplantation now offers the best survival benefit for patients with end-stage heart failure with a median survival over 12 years. However, there are still limitations including the impact of limited availability of graft, graft dysfunction, and rejection, and long-term non-cardiac complications. This review serves as an update on the short- and long-term outcomes following heart transplantation focusing on the new donor allocation system, efforts to expand the donor pool, primary graft dysfunction, acute cellular and antibody-mediated rejection, cardiac allograft vasculopathy, and post-transplant malignancy and renal dysfunction.
Keywords
- Heart transplantation
- Donation after cardiac death
- Hepatitis C virus donation
- Donor heart allocation system
- Immunosuppression
Heart transplantation (HTx) has evolved from a basic experimental model and miracle in human medicine to an expected choice on the menu of treatment for end-stage heart disease. This review aims to discuss HTx with a focus on outcomes after the new allocation system and expansion of the donor pool; utilizing donors with hepatitis C virus, donors following circulatory death, and high-risk donors. This review also presents a discussion on desensitization and serves as an update on outcomes following HTx with an emphasis on primary graft dysfunction (PGD), acute cellular rejection (ACR), antibody-mediated rejection (AMR), cardiac allograft vasculopathy (CAV), and post-transplant malignancy and renal dysfunction. We also provide a brief outline of our regimens and management of each.
Since the first heart transplant in 1967 by Barnard, the field of HTx developed by Shumway now results in survivals that average more than a decade. The groundbreaking discovery of cyclosporine by Calne [1], followed by its application in the 1980s by Starzl [2], ushered the modern era of reasonably well-tolerated effective immunosuppression. Median survival post-transplantation increased from 8.6 years in 1982–1991 to 12.5 years in 2002–2009 [3]. In addition, the advent of mechanical circulatory support (MCS) has not only offered an alternative therapy to patients with a failing heart, but also widened the candidacy for HTx. Advancements in MCS has led to improved 1-year survival of patients with ventricular assist devices (VAD) from 10.2% in 1996–2000 to 70% in 2011–2017 [4].
In recent years, the number of transplants has stabilized to ~5500 procedures worldwide [3]. Mortality on the waitlist has decreased from 15–20% to 7–10% since 2006 [4], likely due to widespread use of automatic implantable cardioverter defibrillators, bridging with mechanical pumps, and overall improved care of end-stage heart failure. To maintain appropriate prioritization of candidates, the allocation system was recently revised [5]. This was done in the context of advances in durable MCS, percutaneous VADs and extracorporeal membrane oxygenator (ECMO) [6]. The medical treatment of heart failure has improved the survival and quality of life [7] for patients who were historically hospital bound with limited life expectancy.
In October 2018, United Network for Organ Sharing (UNOS) revised the donor heart allocation system from a three-tier to a six-tier system (Table 1, Ref. [5, 8]). Prior to the change, it was learned that candidates without MCS and with durable or temporary MCS; including those with ECMO and intra-aortic balloon pump (IABP), had similar post HTx survival, freedom from CAV, and frequency of treated rejection episodes [9]. Such findings encouraged a change to prioritize candidates with temporary MCS given their acuity. In addition, for status 1 and 2 candidates, donor hearts are now drawn from a wider geographic area (500 miles) to more equitably utilize allografts. The change in allocation system has shortened the length of time on the waitlist between 2017 and 2019 from a median of 93 days to 41 days [10]. Distance travelled for procurement increased from a median of 83 miles to 225 miles [10], and correspondingly donor ischemic time increased from a median of 3 to 3.5 hours [10, 11].
| New allocation system [5] | Status | Previous allocation system | Status | ||
| VA ECMO | less than 7 days | 1 | ECMO | 1A | |
| more than 7 days | 3 | ||||
| Non-dischargeable, surgically-implanted, non-endovascular MCS device | biventricular device | 1 | Elective VAD within 30 days | 1A | |
| LVAD less than 14 days | 2 | MCS beyond 30 days | 1B | ||
| LVAD more than 14 days | 3 | ||||
| Dischargeable LVAD | for discretionary 30 days | 3 | |||
| without discretionary 30 days | 4 | ||||
| IABP, or percutaneous endovascular MCS device | less than 14 days | 2 | IABP | 1A | |
| more than 14 days | 3 | ||||
| MCS device with: | life-threatening VT/VF | 1 | MCS with complications | 1A | |
| malfunction or mechanical failure | 2 | Exceptions to status 1A/1B | |||
| one of the following: | 3 | ||||
| Device infection | |||||
| Hemolysis | |||||
| Pump thrombosis | |||||
| Right heart failure | |||||
| Mucosal bleeding | |||||
| Aortic insufficiency | |||||
| Other MCS devices | TAH | 2 | Inpatient TAH | 1A | |
| BiVAD | Outpatient TAH | 1B | |||
| RVAD | |||||
| VAD for single ventricle patients | |||||
| Ventricular arrhythmia (VT/VF) | no MCS required | 2 | Exceptions to status 1A/1B | ||
| Inotropic support (1 high-dose or 2+ lower-dose inotropes) | with continuous hemodynamic monitoring | 3 | With PA catheter in place | 1A | |
| without hemodynamic monitoring | 4 | Continuous IV inotropes | 1B | ||
| Exceptions to status 1A/1B | |||||
| Miscellaneous criteria | re-transplant | 4 | Exceptions to status 1A/1B | ||
| one of the following: | 4 | ||||
| Congenital heart disease | |||||
| Ischemic heart disease + intractable angina | |||||
| Hypertrophic cardiomyopathy | |||||
| Restrictive cardiomyopathy | |||||
| Amyloidosis | |||||
| dual organ transplant candidate | 5 | ||||
| All remaining candidates | 6 | 2 | |||
| Abbreviations: VA, veno-arterial; ECMO, extra-corporeal membrane oxygenator;
MCS, mechanical circulatory support; LVAD, left ventricular assist device; VAD,
ventricular assist device; IABP, intra-aortic balloon pump; VT, ventricular
tachycardia; VF, ventricular fibrillation; TAH, total artificial heart; BiVAD,
biventricular assist device; RVAD, right ventricular assist device; PA, pulmonary
artery; IV, intravenous. | |||||
Hospital length of stay seems to be longer due to the prioritization of higher acuity patients under the new system [10]. However, the incidence of treated acute rejection prior to discharge after HTx did not differ (11.9% pre vs. 11.7% post) [10]. Early studies reported lower 90-day (94.5% vs. 87.6%) and 180-day (93.4% vs. 77.9%) survival after the policy change [12, 13]. This initially raised concern that the prioritization of higher acuity patients would result in a worsening of HTx outcomes; however, more recent data suggest similar 30-day (97.2% vs. 96.9%), 90-day (94.9% vs. 95.0%), and 180-day (93.6% vs. 92.8%) survival rates prior to and after the change, respectively [10, 13, 14]. The difference in outcomes between these studies was attributed to differences in the number of patients included, time periods analyzed, follow-up time, time/survivor ascertainment bias, non-informative censoring of the data, and the factors included in multivariate models [15]. Ultimately, more time is required to accurately analyze short- and long-term outcomes of HTx after the change in listing policy.
An unintended consequence of the change in allocation system has been the shift in practice to utilize temporary, rather than durable, MCS to support candidates on the waitlist. HTx recipients with pre-HTx implanted left ventricular assist devices (LVADs) decreased from 41.8% to 21.2% [11], and candidates receiving LVADs while on the waitlist decreased from 14.5% to 10.8% [16]. Concurrently, there has been an increase in candidates on temporary MCS from 11.3% to 22.9% [11, 16]. In particular, use of IABP to bridge to HTx has increased from 7.6% to 26.2% [10, 17], and bridging with Impella CP (Abiomed, Danvers, MA, USA) has also increased from 0.8% to 9.8% [18].
Under the new allocation system, fewer candidates on ECMO support are clinically-worsening to preclude HTx or dying on the waitlist [18, 19]. Median waitlist duration decreased for these patients from 10 to 5 days [18], while 30-day (76.4% vs. 94.2%) and 6-month (74.6% vs. 90.6%) survival have increased. However, freedom from re-transplantation or death did not differ in landmark analysis after the initial 30 days [18, 19]. They also had similar 1-year survival rates post-HTx compared to recipients who were not on ECMO support or who were converted from ECMO to right VAD, LVAD or total artificial heart prior to HTx [20]. Similar improvements have been reported with the use of other temporary MCS devices, with better survival outcomes post-HTx in the current era compared to previous time periods [21]. For candidates who were supported with the Impella 5.0 (Abiomed, Danvers, MA, USA), survival at 6 months post-HTx is reportedly similar to recipients who were not on MCS devices [22].
For LVAD patients, 1-year survival on the waitlist did not significantly change under the new system (80.1% pre vs. 86.2% post) [16], with reported lower clinical deterioration while awaiting organ availability [23]. Most of these candidates were status 4 at the time of listing (55.9%). However, at the time of HTx, they were generally upstaged to status 3 (47.4%), status 2 (20.4%) or status 1 (4.5%), which could be related to potential clinical deterioration [16]. For these recipients, 1-year survival after HTx was lower under the new system (91.7% vs. 83.4%), and the rate of 1-year graft failure was also higher (8.7% vs. 15.7%). However, rejection rates were similar pre- and post-change (18.7% vs. 19.7%) at 1 year after HTx [16]. These changes could be related to deterioration that has required upstaging pre-transplant and the de-prioritization of stable LVAD patients. Further analyses are needed to confirm and determine potential causes of such findings.
Approximately 92% of recipients per transplant center have a body mass index
(BMI)
Recipients who undergo HTx for idiopathic dilated cardiomyopathy have lower 1-year mortality [25], while those with ischemic cardiomyopathy have higher long-term mortality post-HTx [29]. Other preoperative recipient risk factors associated with worse survival after HTx include pulmonary arterial hypertension [34, 35], renal dysfunction [26, 27, 36, 37, 38, 39], prior sternotomy [40, 41, 42], prolonged ventilator support or intensive care unit (ICU) stay [25, 26, 27, 36, 43, 44], and MCS use [25, 27, 36, 37, 39, 43]. In recipients with pre-HTx MCS use, allograft dysfunction has not been associated with pre-HTx device-related complications [45].
The gender distribution of heart donors (70% male and 30% female) has not
changed from 1992 to 2018 [46]. Although the isolated effect of overall donor
female gender does not seem to worsen survival post HTx [36], meta-analysis and
registry reports show lower survival in gender-mismatched transplants, with a
more pronounced effect in male recipients of female allografts [47, 48]. Median
age and BMI have been increasing over time [46]. Donor age
In the United States (US) from 2000 to 2012, there has been a steady increase in the number of heart donors who underwent prior cardiopulmonary resuscitation (CPR), with a cumulative utilization rate of 28.8% [54]. The trend in increased utilization of such donor hearts has been noted internationally, such as in France (9% in 2004 to 19% in 2012) [55]. Of note, heart donors who underwent CPR were more often classified as Center for Disease Control (CDC) high-risk donors or had a history of cocaine use [54]. With a mean CPR duration of 15–20 minutes, there was no difference in left ventricular ejection fraction (LVEF) of donor hearts despite undergoing CPR [54, 55]. A UNOS registry analysis showed similar 30-day (95.2% vs. 94.7%), 1-year (88.2% vs. 87.7%), and 5-year (72.8% vs. 74.2%) survival between recipients of donors who underwent CPR and those who did not undergo CPR [54, 55].
CDC high-risk donors are donors who confer a high risk for transmissible
infection to recipients, and include males who have sex with males, donors with
history of intravenous drug use, donors with history of bleeding diathesis
disorders who have been transfused with human-derived clotting factors, donors
who have sex with multiple persons or with persons suspected to have human
immunodeficiency virus (HIV) infection, donors who have been exposed to
HIV-infected blood products, and inmates of correctional facilities [56, 57]. In
2007, transmission of HIV and hepatitis C virus (HCV) was reported from a
seronegative donor with high risk features after liver transplantation, and a
highly-publicized article reported 4 transplant recipients contracting HIV [58, 59]. This led to a further increase in the controversy of using organs from
high-risk donors and public reluctance. A survey study shortly thereafter showed
20–65% of surgeons had used hearts/lungs from high-risk donors, and 10–50% of
surgeons stated they accept
With recent improvements in treatments for HIV and HCV, the use of high-risk donor organs has been rising. The number of donors with history of cocaine use increased from 11% in 2000 to 27% in 2018, along with the number of donors with history of non-intravenous non-prescription street drug use which has doubled from 25% to 57% [46]. Notably, there is no difference in short- and long-term survival outcomes between recipients of donors with and without history of cocaine use [27, 43]. Multiple studies, including single-center and registry reports, have showed similar survival between recipients of high-risk donors and of standard risk donors at 1 year (84.3% vs. 83%) and 5 years (71.2% vs. 65.5%) post HTx [56, 57]. Some high-volume transplant centers have even adopted a “say yes” policy, where all candidates are informed about the possibility of a high-risk donor and all suitable organs are accepted [57].
In 2006, HTx recipients of allografts from donors with HCV had more than double the mortality at 1, 5, and 10 years compared to recipients from non-HCV donors [61]. Deaths were commonly due to liver disease and CAV. At the time, interferon and ribavirin constituted the only therapeutic regimen for HCV infection [62, 63]. In 2011, protease inhibitors were introduced as a first generation direct acting antiviral (DAA) for use in conjunction with interferon and ribavirin. When second generation DAA therapies were released in 2014 [63, 64], they gained popularity due to their favorable side-effect profile, higher efficacy to cure multiple genotypes of HCV infection, and stand-alone regimented use. With this therapy, more than 90–95% of HCV patients reach aviremia [62, 63].
In light of this, transplant centers are utilizing solid organs from donors with
HCV who are either NAT+ (viremic, active infection, and infectious) or NAT–
(non-viremic, no active infection, and non-infectious). The frequency of HCV+
donors in HTx increased from 0.6% in 2016 to 11.45% in 2019 [65]. This was
accompanied by an increase in utilization rate of hearts from non-viremic HCV+
donors (27.6%) and viremic HCV+ donors (30.9%), both of which are now
equivalent to utilization of HCV– donors (31.7%) in 2019 [66]. Concurrently,
the willingness of HTx candidates to accept allografts from HCV+ donors increased
from
Transmission of HCV viremia after HTx is reportedly 95.7–100% in recipients of NAT+ donor hearts over a median of 5 days (range of 1–14 days), while none of the recipients of NAT– donor hearts acquired HCV viremia [68, 69]. Providing DAA therapy after accepting allografts from HCV donors seems to be more cost effective in the overall care of end-stage heart failure than not accepting HCV organs [70]. The International Society for Heart and Lung Transplantation (ISHLT) consensus of 2020 recommends routine testing for recipient HCV status and either a prophylactic or a preemptive approach to managing donor-derived HCV infection [62]. Length of treatment is suggested to be 4, 8, 8–12, or 12 weeks depending on the regimen and the approach of management utilized [62, 64, 68, 69, 71, 72, 73]. For those who completed DAA treatment, 100% reach aviremia [68, 69].
Single center and UNOS registry analyses show similar 30-day (93.7% vs. 96.2%), 1-year (90.7% vs. 90.5%), and actuarial 3-year (87.2% vs. 84.8%) survival between recipients of HCV+ and HCV– donor hearts and between recipients of NAT+ and NAT– donor hearts [67, 69, 74, 75]. Rates of treated rejection episodes within 1 year were similar between recipients of HCV+ and HCV– donor hearts (22.1% vs. 21.1%) [67]. When comparing recipients of NAT+ and NAT– donor hearts, the rate of acute rejection episodes within index hospitalization were also similar (22.4% vs. 21.4%) [75]. In addition, the rate of CAV at 1 year post-HTx was similar between recipients of HCV+ and HCV– donors (16.25% vs. 10.69%) [69]. Recipients did not develop worsened renal dysfunction either due to HCV infection or in response to treatment with sofosbuvir; an essential drug in DAA regimens [76].
For screening at our institution (Fig. 1), we obtain serology testing for
recipients of NAT+ allografts on postoperative day 3, and if negative will repeat
on postoperative days 7–10, and 21–28. Although some evidence suggests a
shorter course of treatment (i.e., 4 weeks) when regimen is immediately initiated
postoperatively, we are unable to do so due to insurance coverage being
conditional on positive serology with genotype analysis. Upon approval, either
glecaprevir/pibrentasvir or sofosbuvir/velpatasvir is initiated and continued for
12 weeks. We avoid using sofosbuvir/velpatasvir in patients with renal
insufficiency; however, new evidence suggests potential safety even in patients
on dialysis [77]. Cyclosporine may increase glecaprevir levels, so doses
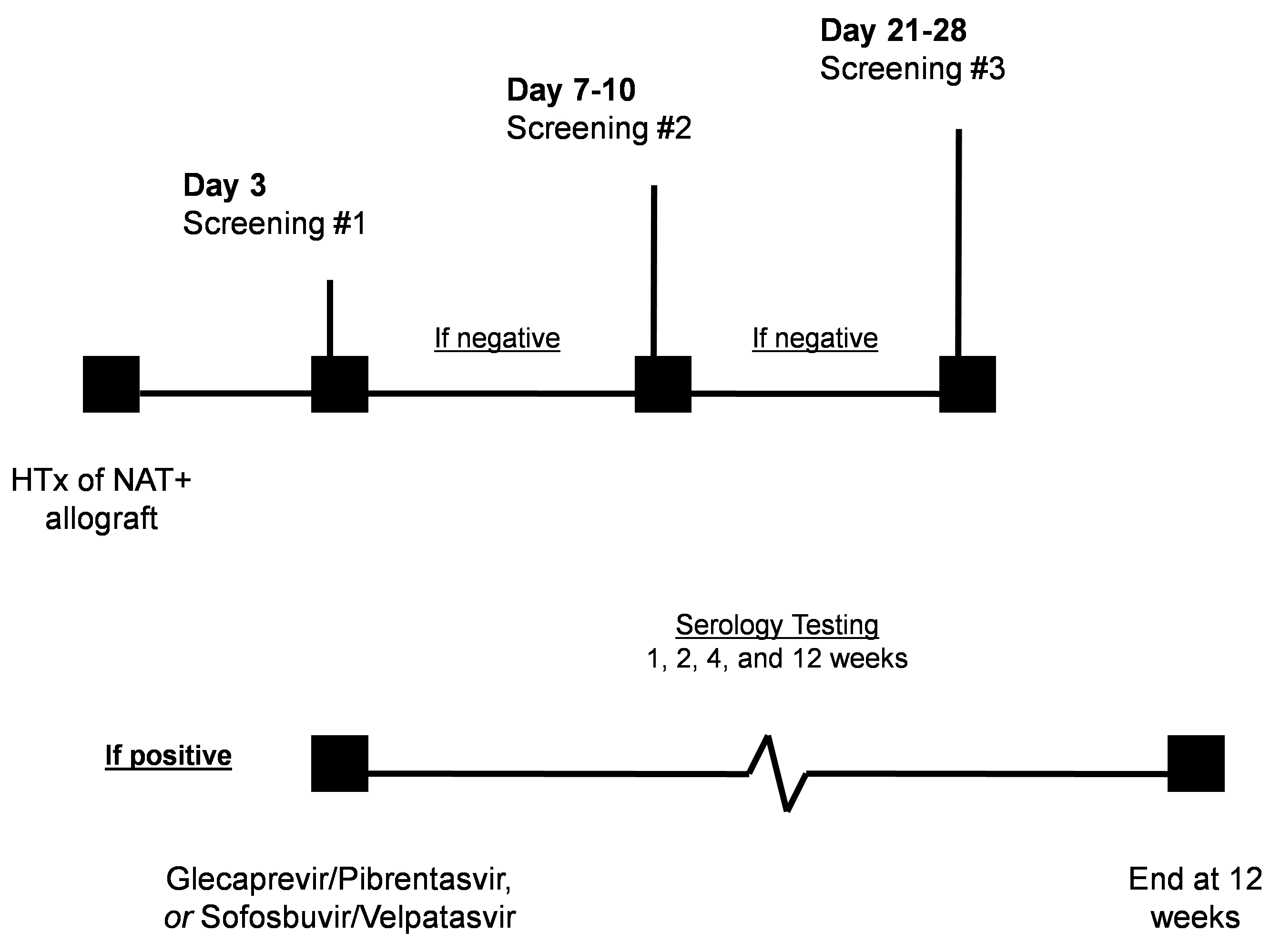 Fig. 1.
Fig. 1.Protocol at our institution for transplantation from donors with hepatitis C virus. Serology testing is obtained on postoperative day 3. If negative, it will be repeated on postoperative days 7–10, and 21–28. If serology testing is positive, treatment is initiated upon approval based on insurance coverage and continued for 12 weeks, with serology testing for response at 1, 2, 4, and 12 weeks. Abbreviations: HTx, heart transplantation; NAT, nucleic acid test (viremic, active infection, and infectious).
Of late, heart donation after circulatory death (DCD) has been cautiously gaining acceptance in the US, but this practice is not a novelty to HTx in Europe. The use of DCD allografts has increased the rate of HTx in the United Kingdom (UK) by 48% from 2015 to 2020 [78]. Use of DCD allografts is estimated to increase the number of available hearts for transplant by ~20% once it becomes well-established [79].
There are three different approaches to DCD procurement, all of which occur after the declaration of death and the appropriate “hands-off” period [78, 79, 80, 81]. Modified conventional procurement consists of rapid chest access and administration of a cardio-protective solution; where after, procurement ensues in a conventional fashion. Normothermic regional perfusion (NRP) consists of rapid chest access and clamping off blood supply to the brain before institution of cardiopulmonary bypass (CPB). This allows for cardiac reperfusion and the assessment of cardiac function, both visually and echocardiographically, prior to procurement. Direct procurement and perfusion (DPP) consists of rapid chest access and procurement of the heart followed by ex-situ reperfusion using the Organ Care System (TransMedics, Andover, MA, USA). This allows for ex-situ evaluation of the heart, analogous to ex-vivo lung perfusion, to determine organ suitability for transplant.
The UK experience adheres to the following donor heart functional criteria for
DCD: allograft LVEF
Outcomes of HTx from DCD donors have been comparable to those from donation after brain death (DBD) donors thus far. In the experience at Royal Papworth Hospital in the UK, Messer and colleagues noted that the number of recipients requiring MCS post-HTx did not significantly differ between recipients of heart allografts from DBD or DCD donors [78]. ICU and hospital length of stay, as well as median first post-HTx LVEF (at 60%), were similar between both groups [78]. Chew et al. [81] observed similar findings in the St. Vincent’s Hospital experience in Australia. Both studies found similar rates of rejection within 1 year post-HTx between the DCD group and the DBD group (25% vs. 23% in the UK experience). In addition, both recipient groups had similar 30-day (97% vs. 99%), 90-day (95% vs. 94%), and 1-year (91% vs. 89%) survival rates, respectively [78]. Comparing recipients of DCD organs via DPP to NRP, there was no difference in immediate post-HTx cardiac function, need for MCS, LVEF post-HTx, and 30-day, 60-day, and 1-year survival rates. However, recipients of DPP allografts had longer ventilatory need, ICU and hospital length of stay, and more frequent need for hemofiltration compared to NRP allografts [78].
PGD is defined as graft dysfunction occurring within 24 hours of HTx with an
LVEF
Donor risks for developing PGD and graft failure include longer donor ischemic
time, older age, left ventricular hypertrophy, cardiac dysfunction, and inotropic
support pre-procurement. Recipient risks include older age, history of diabetes
mellitus, baseline creatinine
Although developing PGD does not seem to affect rates of AMR, ACR
For management of PGD, an escalation algorithm can be used, starting with initiation of inotropic support, followed by temporary MCS with potential VAD placement, and finally re-transplantation as a last resort [98, 99]. Among 90-day survivors of PGD, median allograft recovery time is 3.5 days [86]. At our institution, we utilize veno-arterial ECMO for recipients who cannot be weaned off CPB intraoperatively or who require additional support despite maximal inotropic use.
Induction therapy is the term given for loading the recipient early after implantation of the allograft with depleting anti lymphocyte or thymocyte serum (anti-thymocyte globulin), or monoclonal antibodies that target allo-recognition by the host T lymphocytes such as basiliximab which targets interleukin-2 receptor subunit alpha (IL2RA), and alemtuzumab which targets CD52. In general, only 46% of recipients receive induction therapy preoperatively, as it does not seem to affect survival post-HTx [100]. More acute rejection episodes were observed in recipients who received induction therapy using rabbit anti-thymocyte globulin (ATG) or IL2RA therapy (20.2% vs. 19%, respectively) compared to those who did not receive any induction (16.8%) prior to HTx [101]. Similar findings were also reported in the ISHLT registry report of 2019, with no difference in treated rejection episodes observed between recipients who received ATG as opposed to IL2RA therapy [3]. The higher rates of rejection in recipients receiving induction therapy could be a selection bias due to the higher baseline sensitization profile or panel reactive antibody (PRA) levels. Notably, both registry analyses lack such data.
Our group uses induction (Fig. 2) when the recipient is presensitized to the donor and when the risk of acute kidney injury is considered high due to preexisting chronic renal disease or when the transplant has been complicated and inotropes are required. The latter indication is sparing of calcineurin inhibitors (CNI) which worsen renal function perioperatively. We currently deliver a 500-mg methylprednisolone dose prior to allograft perfusion. We use ATG instead of steroid induction if sparing of CNI is planned; typically dosed for a total of 4.5 mg/kg depending on recipient leukocyte and platelet counts. CD3 count is usually screened for adequate T-cell depletion. Currently, we seldom use basiliximab for induction.
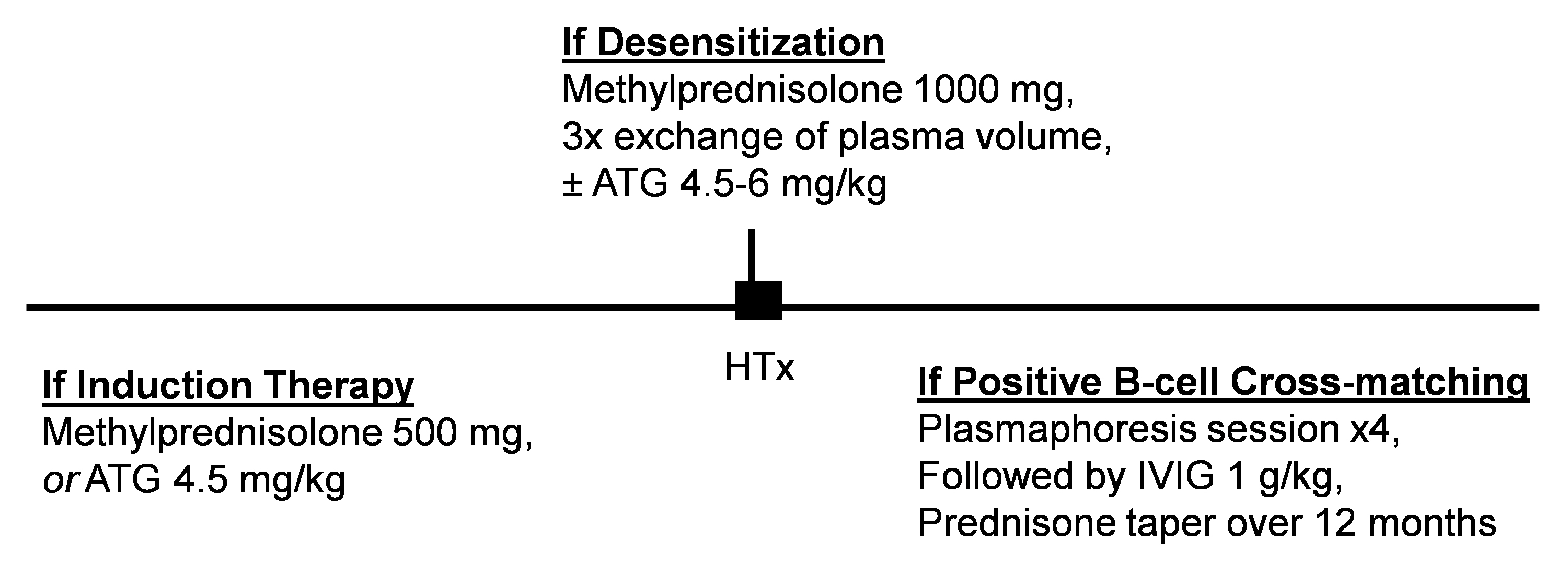 Fig. 2.
Fig. 2.Desensitization and induction protocols at our institution. For induction therapy, ATG is used instead of steroid treatment if sparing of calcineurin inhibitors is planned based on preexisting renal function. For desensitization, ATG is delivered intraoperatively if cross-matching was positive for B lymphocytes. Abbreviations: HTx, heart transplantation; ATG, anti-thymocyte globulin; IVIG, intravenous immunoglobulin.
Maintenance therapy usually consists of tacrolimus; a second generation CNI, an anti-metabolite; more commonly mycophenolic acid than azathioprine, and a short-term corticosteroid taper; from high doses followed by a slower taper of a maintenance dose [102, 103]. Notably, more treated rejection episodes within 1 year were observed when using the CNI cyclosporine as opposed to tacrolimus [3]. Other maintenance regimens include mammalian target of rapamycin inhibitors (mTORi), sirolimus and everolimus, when there is concern for renal dysfunction as they lack the renal toxicity associated with CNI. Sirolimus has been shown to have similar effects on myocardial fibrosis compared to CNI and is associated with improvements in diastolic function and pulmonary arterial pressure [104].
Our current maintenance regimen consists of tacrolimus, mycophenolic acid, and a
prednisone taper over 6 months. The initial tacrolimus therapeutic goal is 10–12
ng/mL unless induction therapy was delivered, in which case it is 8–10 ng/mL.
Therapeutic levels are usually reduced to a goal of 4–6 ng/mL by 3 years post
HTx. Mycophenolic acid is dosed at 1000 mg twice a day initially and is reduced
to 500–1000 mg twice a day after 1 year post-HTx. For recipients experiencing
recurrent infections, we usually reduce tacrolimus, mycophenolic acid, or steroid
dose by 25–50%. At 6–12 months, post-HTx, we attempt to transition patients
from tacrolimus to mTORi due to its renal-sparing effect, and prevention and
delayed progression of CAV, except in patients whose glomerular filtration rate
has dropped to
Allograft rejection consists of antibody-mediated or cellular rejection, and can also be classified as acute or chronic; the latter usually suggesting the development of CAV. Cellular rejection, being more common, is primarily T-cell mediated, while AMR is a B-cell immunologic release of antibodies. Hyperacute rejection occurs if there are preformed antibodies against donor antigens. This occurs more commonly in patients who have received multiple blood transfusions and in multiparous women. Preformed antibodies can be donor specific antibodies (DSA) directed against human leukocyte antigen (HLA) class I or II, or non-DSA directed against other donor antigens. A few of the rarely occurring non-DSA are angiotensin II type 1 receptor, endothelin 1 type A receptor [105], major histocompatibility complex class I chain related gene A, endothelial cell antigens, and vimentin [106]. Though prominent in the early days of HTx, hyperacute rejection is now a relatively rare occurrence. Advances in the field such as prospective cross-matching and PRA analyses have reduced the likelihood. The most common desensitization protocols used for pretreatment of candidates with high PRA levels include plasmapheresis, intravenous immunoglobulin (IVIG), rituximab (chimeric monoclonal anti-CD20 antibody), and bortezomib (proteasome inhibitor) [107, 108, 109]. Potential emerging therapies include eculizumab (complement C5 inhibitor), obinutuzumab (monoclonal anti-CD20 antibody) and tocilizumab (IL6 receptor binder) [108].
The ISHLT consensus of 2018 showed that 21% of centers use PRA levels
Our center’s decision implementing desensitization is based on positive virtual or actual cross-matching [110]. Desensitization consists of a 1000-mg methylprednisolone dose and three exchanges of plasma volume performed between induction of anesthesia and allograft reperfusion (Fig. 2). If cross-matching was positive for B lymphocytes, 4.5–6 mg/kg ATG is also delivered. Postoperatively, recipients undergo four plasmaphoresis sessions followed by 1 g/kg dose of IVIG. Steroids are given with each dose of ATG and tapered over 12 months. For follow up, DSA is obtained at 21 days post HTx.
Endomyocardial biopsy is the gold standard for diagnosing rejection post-HTx. Based on the severity of biopsy results, cellular rejection is classified as grade 0R to 3R [111], while AMR is classified as grade 0 to 3 pathologically [106]. In recent years, an area of research focus has been in the use of noninvasive diagnostic options such as micro-RNA (mi-RNA), cell-free DNA, and gene expression profiling [112, 113, 114]. For patients who have not experienced rejection during the first 3 months post-HTx, our center routinely uses monthly gene expression profiling, cell-free DNA, and echocardiograms, in lieu of routine biopsies.
Subherwal et al. [115] reported a reduction in ACR incidence from 54% in 1990–1991 to 5% in 2000, owing to improvements in immunosuppression regimens. The number of treated rejection episodes within 1-year after HTx has been decreasing over time from nearly 25% in 2004–2006 to 12.6% in 2010–2016 [3]. Risk factors associated with developing rejection include female donor gender, recipient elevated BMI, and recurrent ACR grade 1R [24, 36, 116, 117]. Factors not associated with increased rejection episodes include donor CPR status or history of cocaine use, and recipient history of prior sternotomy or prior MCS use (durable or temporary) [9, 40, 43, 54]. Donor ischemic time does not seem to affect rejection-related mortality after HTx [52]. While history of treated rejection episodes within 1-year post-transplant is known to be associated with sudden cardiac death [118], asymptomatic AMR does not affect actuarial 5-year survival [119]. However, overall AMR is associated with an 8% incremental increase in cardiovascular mortality per episode [120]. In addition, recurrent ACR grade 1R is associated with lower rates of freedom from rejection and survival at 5 years post-HTx [117].
The mainstay of treatment for cellular rejection is corticosteroids and T-cell depletion therapy, most commonly rabbit ATG. Treatment of AMR includes corticosteroids alongside antibody depletion and B-cell targeting; using IVIG, bortezomib, or rituximab [106]. Our management of rejection episodes is based on the designated grade. We typically increase tacrolimus therapeutic goal by 25–50% during ACR and mycophenolic acid dose by 25–50% during AMR. For patients who completed their steroid taper, we restart the regimen during ACR or AMR. If patients are still continuing the steroid taper, we increase the dosing accordingly. ATG may be added during acute rejection as well.
CAV is considered the most common cause of mortality in HTx recipients after the first year of transplantation. CAV is a form of coronary occlusive disease that occurs in the allograft heart after transplantation. Opposed to typical CAD, it is more of a diffuse and concentric process. Proximal lesions are thought to be related to donor-derived CAD, while mid and distal lesions seem to be related to an immune-inflammatory response causing CAV [91]. Local and systemic inflammation cause endothelial injury, which in turn increases smooth muscle cell proliferation and reduction in vascular lumen caliber, thus leading to the development of CAV [121]. CAV is primarily diagnosed using coronary angiography; however, upcoming modalities include intravascular ultrasound, computed tomography (CT) angiography, and endomyocardial biopsy with immune biomarkers [122].
Restrictive physiology can be observed in severe CAV [122]. Frequent rejection
episodes, chronic rejection, chronic allograft failure, and need for
re-transplantation are related to CAV. Elevated hemoglobin A1C is associated with
severity of CAV [121]. Cumulative CAV rates are reportedly 7.7% at 1 year, 29%
at 5 years, and 46.8% at 10 years [3]. Donor risk factors associated with
developing CAV include male gender, older age, and tobacco consumption. Recipient
risk factors include presence of anti-HLA DSA, dyslipidemia, ischemic
cardiomyopathy etiology, re-transplantation, pre-HTx dialysis requirement,
post-HTx HTN, ACR
Management of CAV is primarily through medication regimens for hypertension and dyslipidemia. A meta-analysis of three randomized controlled trials showed a risk reduction in mortality post-HTx in recipients on statin therapy compared to those not on statins [125]. In addition, Wenke and colleagues [126] reported improved 11-year survival in recipients on simvastatin compared to those on dietary modifications alone (77% vs. 56.8%). Incidence of CAV identified on coronary angiography was 34.6% in recipients on simvastatin compared to 73.9% in their counterparts. The culmination of this evidence has led to statins as part of the routine medication regimen of all HTx recipients. Based on platelet activation and aggregation in CAV, some suggest acetylsalicylic acid (ASA) as a potential medication to delay progression of the disease and reduce CAV-related graft failure [127]. Of note, surgical revascularization is associated with increased mortality, while percutaneous coronary intervention (PCI) is challenging due to the diffuse nature of CAV. Although drug-eluting stents have reduced the risk of in-stent restenosis compared to bare-metal stents, there is no effect on survival and no sufficient evidence to suggest that PCI carries a survival benefit over medical management in HTx recipients [128]. Ultimately, treatment for severe CAV is re-transplantation. At our institution, all patients are placed on ASA and statin therapy. In addition, as previously described, we utilize early conversion to mTORi to prevent progression of CAV.
In HTx, malignancy is a concern due to immunosuppression. Male gender, re-transplantation, prior malignancy, and immunosuppression are identified risk factors for malignancy post-HTx. Protective factors against malignancy include acyclovir prophylaxis, induction with ATG therapy, use of sirolimus or everolimus, and female gender [129]. Overall malignancy rate post-HTx seems to be 5.1% for 1-year survivors, 16% for 5-year survivors, and 27.7% for 10-year survivors [3]. The most common malignancy for HTx recipients is skin cancer, with a rate of 1.7% in 1-year survivors, 9.6% in 5-year survivors, and 18.5% in 10-year survivors [3]. Risk factors associated with developing cutaneous squamous cell carcinoma include older recipient age, male gender, HLA mismatch, pre-HTx history of malignancy, and hypertrophic or restrictive cardiomyopathy, and re-transplantation [130]. Predictors of developing basal cell carcinoma include older recipient age, male gender, and history of malignancy pre-HTx [131].
For HTx recipients with history of malignancy, recurrence post-transplant is
highest at 63% in patients who achieved remission within 1 year pre-HTx,
compared to 26% if remission was between 1 and 5 years, and 5% if remission was
Current therapies of such malignancies revolve around immunotherapy, which could be detrimental to the allograft, causing generalized cardiotoxicity, allograft failure or rejection [129]. Balance between immunotherapy for cancer and immunosuppression for rejection is required in such patients. Of note, use of mTORi for maintenance therapy seems to confer anti-oncogenic properties [132, 133]. The most common malignancy seen at our institution is skin cancer followed by PTLD. For recipients who develop any malignancy, we typically discontinue mycophenolic acid and reduce tacrolimus goal accordingly. Usually, reduction is more significant in case of PTLD given the higher likelihood of receiving chemotherapy as well. If recipients are not on it, we would initiate mTORi at the time.
Renal failure is a concern in the HTx patient population due to the nephrotoxic effects of CNI therapy. Although freedom from severe renal dysfunction has improved in recent years, 6.7%, 15.7%, and 22.3% of recipients suffer from the disease by 1, 5, and 10 years post-HTx, respectively. It has also been associated with worse conditional 1-year survival after HTx [3]. Among HTx recipients who die anytime after 1 year of transplantation, cause of death due to renal failure is estimated to be 1.3–9.9% [3]. Risk factors include donor female gender, longer donor ischemic time, history of prior sternotomy, elevated recipient pre-HTx creatinine, temporary MCS use pre-HTx, receiving preoperative induction therapy, dialysis requirement prior to discharge from index HTx, and use of long-term corticosteroid taper as maintenance therapy post-HTx [3, 21, 36, 40, 42, 52, 101, 103, 134]. Requiring renal replacement therapy pre-transplant is not associated with ESRD or survival post-HTx, while requiring it post transplant is associated with both [135]. Need for chronic dialysis is 1.5%, 2.9%, and 6%, while 0.1%, 0.6%, and 2% receive renal transplantation at 1, 5, and 10 years, respectively [3]. Efforts to either change immunosuppression regimen, especially CNI, or reduce drug target levels should be made in order to reduce likelihood of acquiring renal dysfunction post-HTx. Recent developments in renal-sparing immunosuppression, which are currently being used for kidney transplant recipients, such as belatacept [136], may further reduce the risk of renal dysfunction in the future.
Orthotopic heart transplantation remains the ultimate modality of treatment for patients suffering from heart failure. While rates may differ among etiology of heart failure, particularly among those requiring re-transplantation [3], overall survival at 10, 15, and 20 years has been reported at 50.2%, 30.1%, and 17.2%, respectively [29]. Organ shortage is the major limiting factor to the epidemiologic impact of heart transplantation. The new allocation system, implemented since October of 2018, has a positive effect on waitlist survival in multiple patient populations, including patients on temporary mechanical support. Although it may have a positive effect on early post transplant survival especially in patients with ECMO, more time is needed to accurately analyze this outcome. The utilization of high-risk, HCV, and DCD donors has expanded the pool of available organs for cardiac transplantation, without affecting survival outcomes, and hopefully will continue to do so once more experience is gained. There are associated risk factors; however, they seem to be minimal in the current era and are outweighed by the potential benefit in well-matched donor/recipient pairs.
Graft dysfunction is the most common cause of early mortality after transplantation, while allograft vasculopathy is the most common cause of late mortality. Allograft rejection is one of the most common complications after transplantation. The field of immunosuppression is continuously evolving and new drug regimens are being searched for in order to reduce the side-effect profile of such medications.
The continued advancement in LVAD technology may lead to equivalent outcomes to cardiac transplantation in the not-so-distant future. However, for the time being need for anticoagulation and risk of driveline infection make it a suboptimal solution. Currently, heart transplantation remains the best treatment for patients with end-stage heart failure. Other avenues being investigated at this time include xenotransplantation and bioengineered organs, though there still remain many hurdles, including maintaining graft function long term, genetic modifications of organ source, and higher-than-standard immunosuppression regimens. Promising endeavors are currently underway to alleviate such limitations.
MAA, AS and BPG: writing—original review and editing. All authors have read and agreed to the published version of the manuscript.
Not applicable.
We would like to express our gratitude to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.


