1 Pharmacy Department, Heart Hospital, Hamad Medical Corporation, 3050 Doha, Qatar
2 Cardiology Department, Heart Hospital, Hamad Medical Corporation, 3050 Doha, Qatar
Academic Editor: Leonardo De Luca
Abstract
Cardiogenic shock (CS) is a hemodynamically complex and highly morbid syndrome characterized by circulatory collapse and inadequate end-organ perfusion due to impaired cardiac output. It is usually associated with multiorgan failure and death. Mortality rate is still high despite advancement in treatment. CS has been conceptualised as a vicious cycle of injury and decompensation, both cardiac and systemic. Interrupting the vicious cycle and restoring the hemodynamic stability is a fundamental treatment of CS. Acute coronary syndrome (ACS) is the most frequent cause of CS. Early coronary revascularization is a cornerstone therapy that reduces mortality in patients with ACS complicated by CS. Early diagnosis of CS accompanied with invasive hemodynamics, helps in identification of CS phenotype, classification of CS severity, stratification of risk and prognostication. This can guide a tailored and optimized therapeutic approach. Inotropes and vasopressors are considered the first-line pharmacological option for hemodynamic instability. The current availability of the mechanical circulatory support devices has broadened the therapeutic choices for hemodynamic support. To date there is no pharmacological or nonpharmacological intervention for CS that showed a mortality benefit. The clinical practices in CS management remain inconsistent. Herein, this review discusses the current evidence in the diagnosis and management of CS complicating ACS, and features the changes in CS definition and classification.
Keywords
- Acute myocardial infarction
- Cardiogenic shock
- Inotrope
- Mechanical circulatory support
- Pulmonary artery catheter
- Vasopressor
Shock in the general term is a circulatory failure due to impaired oxygen utilization by the body cells, which affects approximately one third of critically ill patients. The most common mechanisms of shock are hypovolemia, cardiac factors, obstruction, and distributive factors. The distributive shock is usually characterized by high cardiac output (CO), reduced systemic vascular resistance (SVR), and altered oxygen extraction. Whereas the three other mechanisms lead to low CO and insufficient oxygen transport (Table1) [1]. Cardiogenic shock (CS) is a clinical condition that results from ventricular failure due to acute coronary ischemia [1, 2], which eventually leads to inadequate peripheral tissue perfusion, tissue and cellular ischemia, end-organ damage and multiorgan system failure [3, 4]. Up to 40,000 and 80,000 CS patients are annually admitted to hospitals in the United States and Europe, respectively [2].
| Type examples | Percentage | CO or SvO |
CVP | ECHO |
| Distributive (Vasodilation) Severe sepsis, anaphylaxis | 62% | Normal or high | - | - Normal cardiac chambers |
| 4% |
- Preserved contractility in most of the cases | |||
| Hypovolemic Internal or external loss of volume (plasma or blood) | 16% | Low | Low | - Small cardiac chambers |
| - Normal or high contractility | ||||
| Cardiogenic (Ventricular failure) Acute MI, end-stage CM, myocarditis | 16% | Low | High | - Large ventricles |
| - Poor contractility | ||||
| Obstructive (Obstruction) Pericardial tamponade, PE, pneumothorax | 2% | Low | High | - Tamponade: pericardial effusion, small ventricles, dilated inferior vena cava |
| All | - Arterial hypotension | |||
| - Signs of tissue hypoperfusion (altered mental status, mottled and clammy skin, oliguria) | ||||
| - Tachycardia, elevated blood lactate, circulatory shock | ||||
Abbreviations: MI, myocardial infarction; CM, cardiomyopathy; CO, cardiac output; CVP, central venous pressure; ECHO, echocardiography; LV, left ventricle; PE, pulmonary embolism; RV, right ventricle; SvO | ||||
Acute coronary syndrome (ACS) is the most common cause of CS, accounting for up to 80% of the cases [5]. CS complicates 4–12% of ST-segment elevation myocardial infarction (STEMI) [2, 3, 6, 7, 8] and 2–4% of non-STEMI patients [2, 7, 8, 9, 10]. Most of acute myocardial infarction (MI) cases develop CS after hospital admission [6, 7, 9, 11, 12] (e.g., 62–89%) [5, 6, 11], and usually within 24 hours of the event [12]. CS may also occur after coronary reperfusion [13]. Non-ACS etiology accounts for one-fifth of the cases [5]. Illnesses or conditions that may cause CS include but not limited to, cardiac tamponade, primary idiopathic pericarditis, myocarditis, acute heart failure, end-stage severe congestive heart failure, severe infections with and without septicaemia, or cor pulmonale due to pulmonary emboli [14]. Figs.1,2 present the historical perspective of shock, acute MI and their basic aspects of management [14, 15, 16, 17, 18].
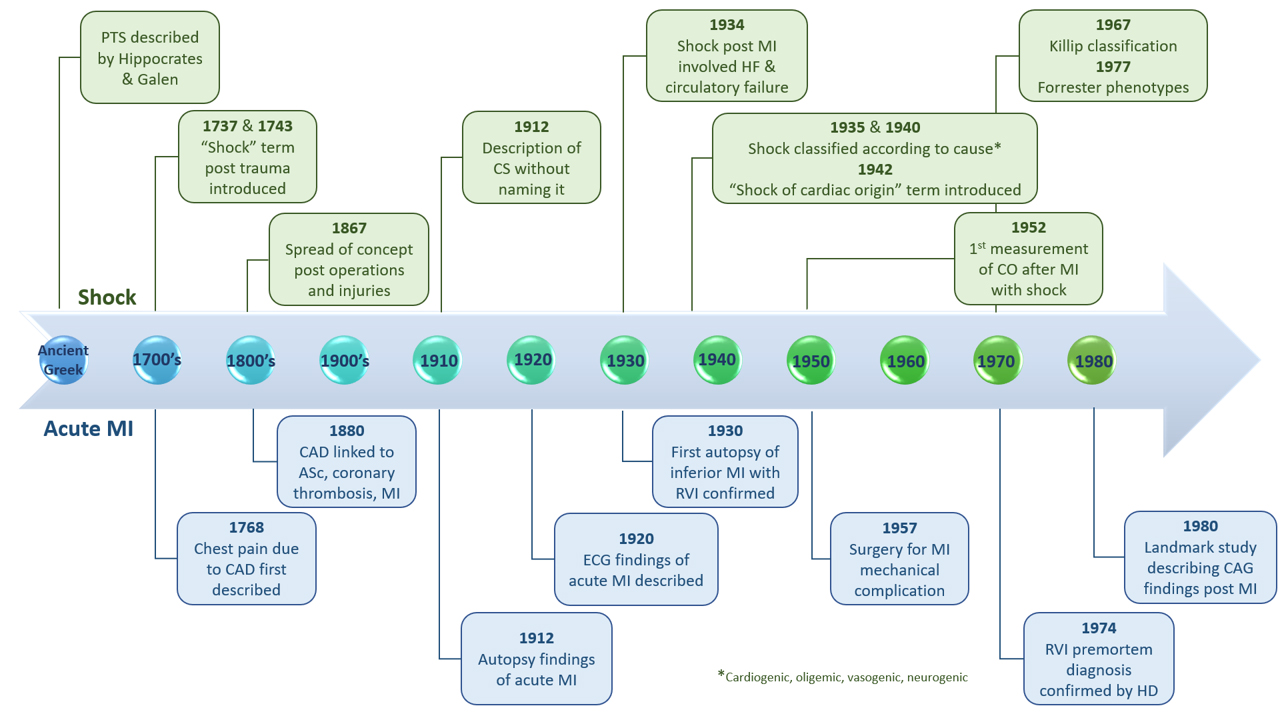 Fig. 1.
Fig. 1.Historical perspective of shock and myocardial infarction. ASc, atherosclerosis; CAD, coronary artery disease; CO, cardiac output; CS, cardiogenic shock; ECG, electrocardiogram; HD, hemodynamics; MI, myocardial infarction; PTS, posttraumatic syndrome; RVI, right ventricular infarction.
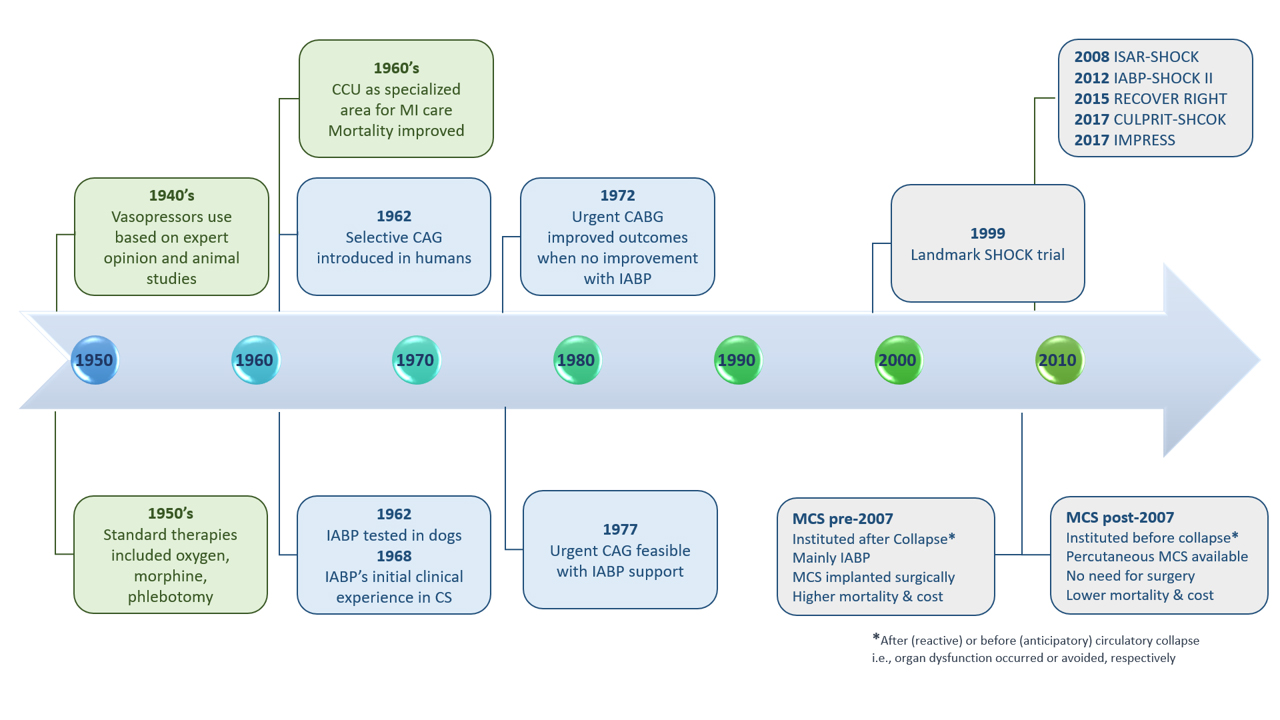 Fig. 2.
Fig. 2.Historical perspective of cardiogenic shock management. CAGB, coronary artery bypass surgery; CAG, coronary angiography; CCU, Coronary Care Unit; CS, cardiogenic shock; IABP, intra-aortic balloon pump; MCS, mechanical circulatory support.
Mortality rates had varied widely overtime (i.e., from 50% to 90%), probably due to the non-unified definition of CS between studies [15]. However, early mortality due to CS complicating acute MI remains high, in up to 50% of patients [5, 6, 13, 19, 20], even after more than 20 years of the SHOCK trial publication [21]. With recent advancement in STEMI management, the reported rates of early mortality in the recent studies are in the range of 40% [22, 23]. Whereas, the in-hospital mortality is lower (24%) in CS that is not secondary to ACS [23]. Fig. 3A projects the mortality rates over years and Fig. 3B projects rates over short (i.e., 1–4 years) and long (i.e., 10–30 years) periods of time [6, 8, 11, 21, 22, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39]. CS and its consequences have a considerable economic impact as well [4]. The objective of this review is to discuss the current evidence in the diagnosis and management of CS complicating ACS, and feature the changes in CS definition and classification.
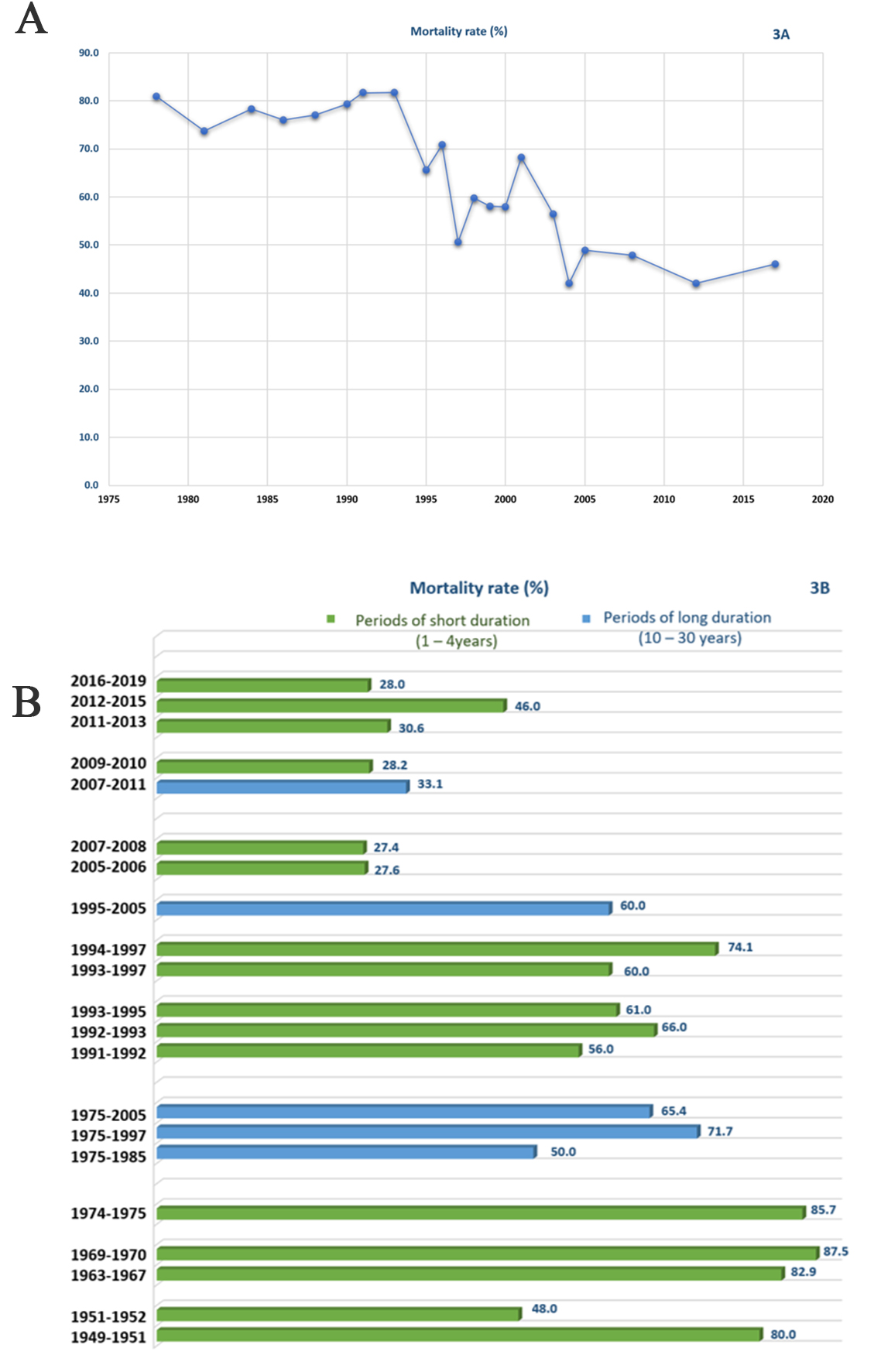 Fig. 3.
Fig. 3.(A) Mortality rate over years. (B) Mortality rate over periods of year. Short period is defined as 1–4 years and long period as 10–30 years.
The main mechanism of CS is acute MI [1, 2, 5, 40], including its mechanical complications [5, 33, 41, 42], that causes left ventricular (LV) pumping failure [1, 2, 40]. Mechanical complications of an ACS event (e.g., ventricular septal rupture (VSR), free-wall or papillary muscle rupture) account for 12% of CS cases [40] and frequently occur within 24 hours of hospital admission [3]. As an example, VSR has the highest risk of mortality (87%) [40, 42] with a median of 16 hours to occurrence from MI onset. However, other timing data reported in literature varied from three to eight days [42]. The underlying pathology of MI in CS has been studied. Stepwise or progressive myocardial damage and injury may occur. In patients who die due to CS, the extent of damage is greater than in those who die from acute infarction. LV infarction mass was 51% (range 35–68%) in CS non-survivors. Losing half of LV myocardium may explain the clinical and hemodynamic consequences of CS [43]. Moreover, the LV infarct size exceeded 40% even after coronary reperfusion [44]. Severe ischemia leads to elevated ventricular filling pressures, reduced CO, hypotension, and systemic tissue hypoperfusion, that eventually affect all body organs [2, 3]. With persistent CS, there are further exacerbation of ischemia, coronary perfusion defect, ongoing cell death, and deterioration of systolic and diastolic functions, resulting in further increments in ventricular filling pressures and worsening of CO [2]. This uninterrupted maladaptive vicious cycle is eventually deadly [2, 4]. Although peripheral vasoconstriction (i.e., early compensatory mechanism) can ameliorate the perfusion, both coronary and peripheral, this is achieved at the expense of an elevated afterload [3]. Circulatory compensatory mechanisms are usually insufficient and may worsen the situation [3, 40]. The acute cardiac event can also provoke a systemic inflammation that leads to pathological vasodilation [2, 3, 4], due to the release of inflammatory mediators and nitric oxide (NO) [4, 45]. Inflammatory mediators include tumor necrosis factor and interleukins [46]. High levels of NO produced by NO synthases and the cytotoxic NO-derived species (i.e., peroxynitrite) have many deleterious effects such as inappropriate vasodilation with reduced systemic and coronary perfusion pressures [47]. Fig. 4 summarizes the pathophysiology of infarct-related CS and treatment targets [2, 3, 40, 47].
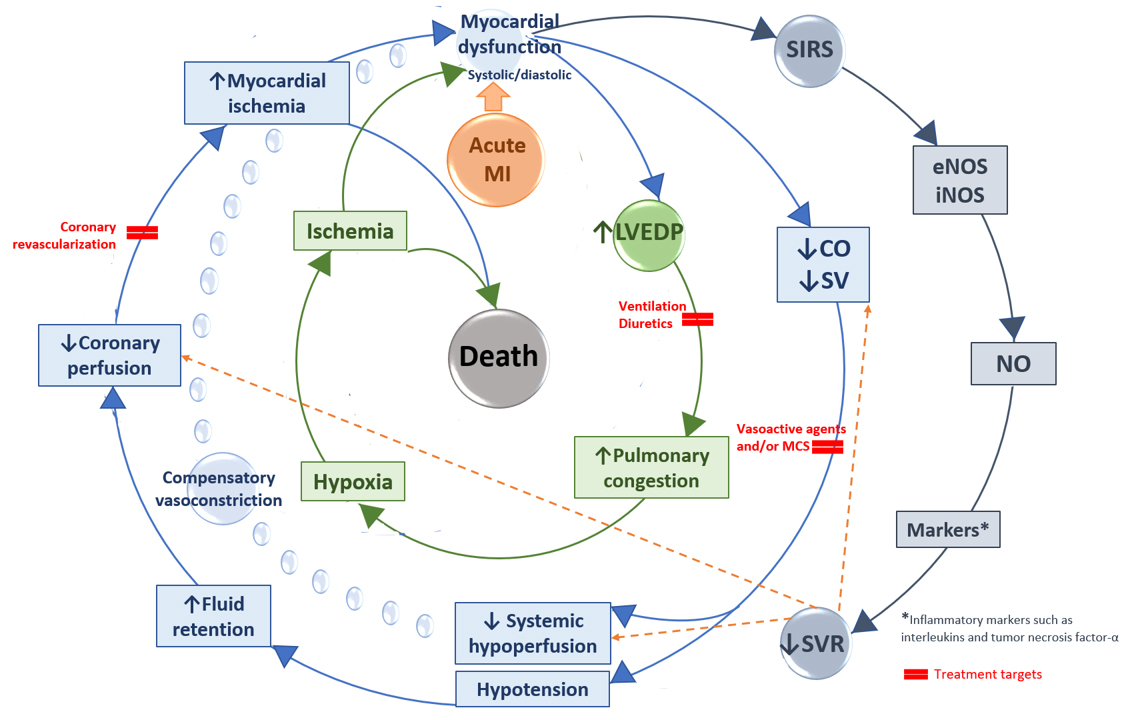 Fig. 4.
Fig. 4.Pathophysiology of cardiogenic shock and treatment targets. CO, cardiac output; eNOS, endothelial NO synthase; iNOS, inducible NO synthase; LVEDP, left ventricular end-diastolic pressure; MCS, mechanical circulatory support; MI, myocardial infarction; NO, nitric oxide; SIRS, systemic inflammatory response syndrome; SV, stroke volume; SVR, systemic vascular resistance.
With regards the consequences of CS on body organs, elevation in the LV filling pressure causes pulmonary edema and congestion, due to rise in pulmonary capillary hydrostatic pressure [2]. Pulmonary vasoconstriction, as a result of hypoxia and inflammation, increases myocardial oxygen consumption and afterload of both ventricles. Renal glomerular filtration rate is reduced, and renin-angiotensin-aldosterone system is activated secondary to renal hypoperfusion. Consequently, tubular sodium reabsorption and fluid overload increase, thus attenuating the response to diuretics. Splanchnic vasoconstriction induced by sympathetic nervous system causes further deterioration of fluid overload, through blood redistribution to the central circulation [2, 4], and precipitation of septic reaction by translocating bacteria or their toxins. Cerebral hypoperfusion is the reason for the altered mental status in CS [2].
CS is a clinical condition of impaired primary cardiac function with ineffective CO that hinders sufficient blood perfusion to the end-organs (i.e., tissues hypoxia) to meet their metabolic demands [2, 3, 23, 40, 48, 49]. In many cases, the patients are not hypovolemic (i.e., have adequate intravascular volume) [2]. The definition of CS has evolved over years [23] and CS-defining criteria have varied among clinical trials and societal guidelines [4, 49] as summarized in Table2 (Ref. [2, 3, 6, 7, 16, 21, 24, 26, 27, 32, 35, 36, 38, 39, 48, 50, 51, 52, 53, 54, 55, 56]). The widely accepted definition of CS includes clinical signs and symptoms of tissue hypoperfusion (e.g., altered mental status, oliguria, high lactate level) and elevated LV filling pressures (e.g., pulmonary congestion). In addition to hemodynamic parameters (e.g., persistent hypotension with severely reduced cardiac index (CI)) [2, 23, 40].
In 1967, Killip et al. [26] proposed a clinical classification of
severity for patients presenting with acute MI based on their hemodynamic status.
Patients were classified in one of four classes: (I) no clinical signs of cardiac
decompensation; (II) heart failure diagnosed by rales, venous hypertension, and
S3 gallop; (III) severe heart failure, characterized by frank pulmonary edema;
and (IV) CS, characterized by hypotension (i.e., systolic blood pressure (BP)
Given that CS presentation encompasses a wide range of clinical and hemodynamic parameters, the definition of CS should consider a continuum of stages rather than a binary diagnosis such as the classic construct of “cold and wet” phenotype [23]. The classification systems by Killip and Forrester also assess congestion and perfusion through physical exam findings and hemodynamic parameters. However, such classifications do not gauge CS severity, allow better hemodynamic-guided management of CS, or address the timely use of mechanical circulatory support (MCS) devices [62]. Thus, other terminologies have been proposed to address the broad range of CS presentation and describe progression that can assist in escalating management from the use of vasoactive agents to MCS devices. One proposal suggested that stages such as pre-, mild, profound, and refractory shock can be more appropriate [23]. In response to such unfulfilled demands and in search of a new lexicon and uniform system to define CS severity, the Society for Cardiovascular Angiography and Interventions (SCAI) has published a five-stage classification system based on expert opinion. This consensus document was endorsed by the American College of Cardiology (ACC), the American Heart Association (AHA), the Society of Critical Care Medicine (SCCM), and the Society of Thoracic Surgeons (STS). The purpose of the SCAI classification was to offer a simplified scheme in order to facilitate a clear and easy communication about patients’ status and a better differentiation between patients’ subsets in clinical trials. Other aims were, to assist in rapid patient assessment, reassessment and re-classification, to be applicable to multiple clinical settings and retrospectively to prior clinical trials, and to have a prognostication potential of different CS subsets [13].
| Prior to SHOCK trial | ||||
| Griffith 1954 [24] | Binder 1955 [50] | Malach 1960 [16] | Goldberg 1991 [27] | Hochman 1995 [32] |
| Killip 1967 [26] | (1975 to 1988) | Holmes 1995 [6] | ||
| Holmes 1999 [7] | ||||
| - Marked hypotension for |
- SBP ≤80 mmHg | - SBP |
- SBP |
- SBP |
| - In normotensive patients: SBP |
- Pulse rate |
- Hypotension (SBP |
- SBP increased to | |
| - In hypertensive patients: SBP |
- Clinical signs of peripheral circulatory collapse | oliguria, cyanosis and diaphoresis [26] | ||
| - No improvement for 30 min after pain relief and O |
||||
| SHOCK trial and beyond | ||||
| Hochman1999[21, 51] (SHOCK) | Burkhoff2006[52] | Thiele2012[38, 53] (IABP-SHOCKII) | Thiele2017[39, 54] (CULPRIT-SHOCK) | Ouweneel2017[36](IMPRESS) |
| - SBP |
- CI |
- SBP |
- SBP |
- SBP |
| - CI |
- End-organ hypoperfusion or need for high-dose pressor and/or inotropic support | - Clinical signs of pulmonary congestion, and impaired end-organ perfusion | - Clinical signs of pulmonary congestion, and signs of impaired organ perfusion | |
| Registries and guidelines | ||||
| Menon2000[55] | Wayangankar2016[35] (CathPCIRegistry) | 2016 ESC HF [48] | 2017AHA[3] | 2020ACCAPosition[2] |
| (SHOCKtrialregistry) | 2017 ESC STEMI [56] | ScientificStatement | Statement | |
| - SBP |
- SBP |
- SBP |
- Persistent hypotension unresponsive to volume replacement with clinical features of end-organ hypoperfusion requiring pharmacological or mechanical support | - SBP |
| - Evidence of tissue hypoperfusion with adequate or elevated LV filling pressures | levels | - Hemodynamic parameters are not mandatory but help confirm diagnosis | - Pulmonary congestion and signs of end organ failure | |
| Abbreviations: ACCA, Acute Cardiovascular Care Association; AHA, American Heart
Association; AV, atrioventricular; CI, cardiac index; CS, cardiogenic shock; ESC,
European Society of Cardiology; HF, heart failure; hr, hour; IMPRESS, IMPella
versus IABP Reduces mortality in STEMI patients treated with primary PCI in
Severe cardiogenic SHOCK; LV, left ventricle or ventricular; MAP, mean arterial
pressure; min, minute(s); O | ||||
The SCAI classification, inspired by the ACC/AHA classification of heart failure
[63] and the Interagency Registry for Mechanically Assisted Circulatory Support
(INTERMACS) classification [64], is the first standardized set of definitions for
CS. The SCAI scheme stratifies CS into five stages: at-risk (stage A), beginning
(stage B), classic (stage C), deteriorating (stage D), and extremis (stage E).
Each stage has been described in three domains: physical exam or bedside finings,
biochemical markers, and hemodynamic parameters (Fig. 5). Furthermore, the
cardiac arrest as an important prognosis modifier (i.e., the (
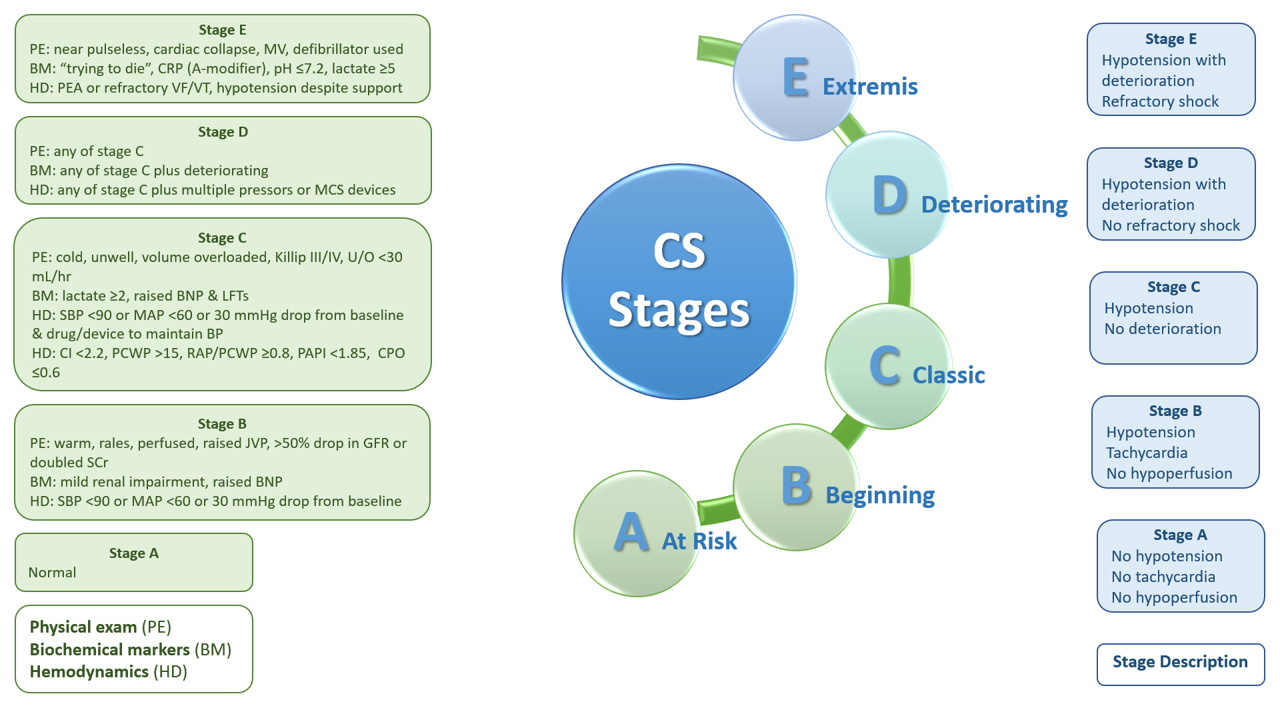 Fig. 5.
Fig. 5.SCAI classification of cardiogenic shock. BNP, brain natriuretic peptide; BP, blood pressure; CI, cardiac index; CPO, cardiac power output; CPR, cardiopulmonary resuscitation; CS, cardiogenic shock; GFR, glomerular filtration rate; JVP, jugular venous pressure; LFTs, liver function tests; MAP, mean arterial BP; MCS, mechanical circulatory support; MV, mechanical ventilation; PAPI, pulmonary artery pulsatility index; PCWP, pulmonary capillary wedge pressure; PEA, pulseless electrical activity; RAP, right atrial pressure; SBP, systolic BP; SCr, serum creatinine; U/O, urine output; VF/VT, ventricular fibrillation/tachycardia.
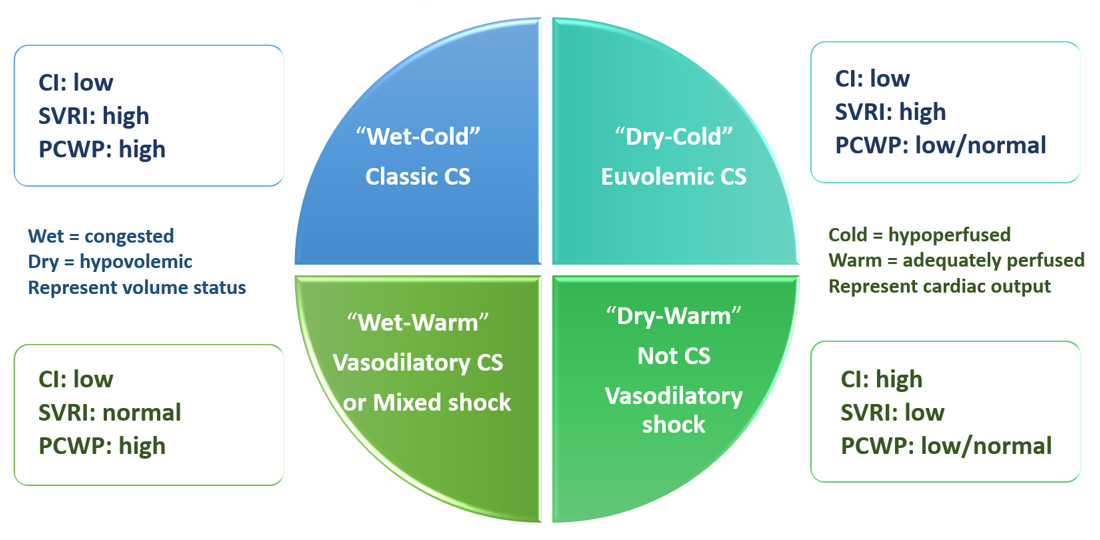 Fig. 6.
Fig. 6.Phenotypes of cardiogenic shock. CI, cardiac index; CS, cardiogenic shock; ECG, electrocardiogram; PCWP, pulmonary capillary wedge pressure; SVRI, systemic vascular resistance index.
The diagnosis of CS is usually differentiated from other types of shock (i.e.,
distributive, hypovolemic, non-obstructive) based on history, physical
examination, laboratory data, and electrocardiogram characteristics. As any
condition leading to profound LV or RV impairment can result in CS [40], various
causes should be identified such as acute MI, acute decompensated heart failure,
post-cardiotomy shock, atrial or ventricular arrhythmias, or valvular diseases
[2, 59]. CS diagnosis should also distinguish between CS and mixed shock type due
to other contributing factors [2], such as infection, bowel ischemia, or
hemorrhage in the setting of MI. Interestingly, CS may develop as an iatrogenic
illness. Medication classes that are used to treat acute MI can be associated
with the development of shock such as beta-blockers and angiotensin-converting
enzymes inhibitors [40]. Regardless of its type, shock is diagnosed based on
clinical, hemodynamic, and biochemical components [1]. The clinical signs of
cutaneous, renal and brain tissue hypoperfusion are featured as cold and clammy
skin, cyanosis due to vasoconstriction, oliguria (i.e., urine output
The differentiation between the hemodynamic phenotypes of CS needs invasive
hemodynamic monitoring using pulmonary artery (i.e., Swan-Ganz) catheter (PAC)
[3, 13, 40]. Hemodynamic monitoring can confirm CS diagnosis when uncertain or when
there is no response to therapy. In addition, it can direct therapy and evaluate
the need for MCS [2, 40, 72, 73, 74, 75]. Among all CS phenotypes, the common hemodynamic
characteristic is low CI, while other parameters (e.g., volume, SVR, pulmonary
capillary wedge pressure (PCWP)) may vary. Although the widely accepted CI
cut-off in CS is
Historically since 1970, PAC was the first device used in critically ill
patients to classify the hemodynamic parameters. In patients presented with acute
MI, the use of PAC consistently and significantly increased from 7.2% to 19.9%
in 1975 throughout 1984, respectively [76]. Despite its widespread use for
diagnosis and decision-making in management, PAC use has progressively decreased
thereafter [74]. A multicentre longitudinal study (n = 15,006) found a decrease
in PAC use from 16.4% in 2002 to 6.5% in 2006 (i.e., decrease by
| Agent | Target | Effect |
| Vasopressor/inotropes | ||
| Dopamine | D |
- Inotropy, dromotropy, chronotropy, and vasoconstriction |
| Dose-dependent agonism | - ↑ to ↑↑ CO, ↑ to ↑↑ SVR | |
| Epinephrine | - Inotropy, chronotropy, dromotropy, and vasoconstriction | |
| - ↑↑ CO, ↑↑ SVR | ||
| Norepinephrine | - Inotropy, chronotropy, dromotropy, and vasoconstriction | |
| - ↑ CO, ↑↑SVR | ||
| Inodilators | ||
| Dobutamine | - Inotropy and mild vasodilation | |
| - ↑↑ CO, ↓ SVR, ↓ PVR, ↓ MAP | ||
| Enoximone | PDEi | Inotropy and inodilator |
| Milrinone | - ↑ CO, ↓ SVR, ↓ PVR, ↓ MAP | |
| Levosimendan | Myofilament Ca |
- Inotropy and inodilator |
| - ↑ CO, ↓ SVR, ↓ PVR, ↓ MAP | ||
Early hemodynamic support is essential for patients in shock regardless of the cause. Priorities and goals of therapy usually target four phases. The salvage phase, to obtain acceptable BP through life-saving measures (e.g., coronary revascularization; discussed below); the optimization phase, to ensure adequate cellular oxygenation through hemodynamic resuscitation (e.g., optimizing cardiac output); the stabilization phase, to minimize complications through organ support; and the de-escalation phase, to achieve negative fluid balance [1]. The “VIP” approach succinctly describes the initial resuscitation steps; “ventilate” by administering oxygen, “infuse” by fluid resuscitation, and “pump” to restore cardiac competence. The postscript (i.e., “PS”), follows with “pharmacologic treatment” such as vasoactive agents to improve perfusion and “specific or surgical management” of the primary causes [88].
Circulatory support, either pharmacologic or nonpharmacologic, should be promptly employed to manage hypotension and maintain tissue perfusion [89]. Fig. 7 summarizes the overall management approach to CS [48, 90, 91].
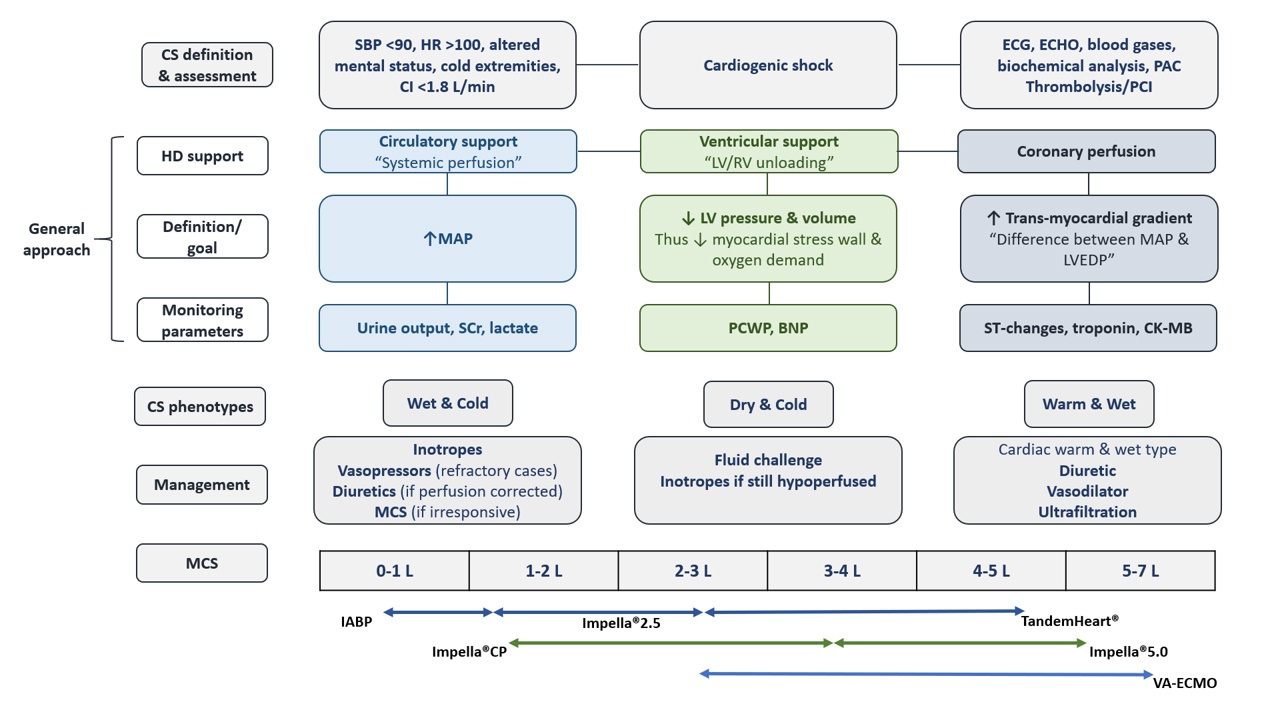 Fig. 7.
Fig. 7.Management approach to cardiogenic shock. BNP, brain natriuretic peptide; CI, cardiac index; CK-MB, creatine kinase-MB; CS, cardiogenic shock; HR, heart rate; ECG, electrocardiogram; ECHO, echocardiogram; HD, hemodynamics; HR, heart rate; IABP, intra-aortic balloon pump; LVEDP, left ventricular end-diastolic pressure; MAP, mean arterial blood pressure; MCS, mechanical circulatory support; PAC, pulmonary artery catheter; PCI, percutaneous coronary intervention; PCWP, pulmonary capillary wedge pressure; SBP, systolic blood pressure; SCr, serum creatinine; VA-ECMO, veno-arterial extracorporeal membrane oxygenation.
Vasoactive agents (i.e., intravenous inotropes and vasopressors) remain the
initial hemodynamic support in CS unresponsive to fluid resuscitation [4, 23],
with an administration rate in almost 90% of patients [5, 92]. They
preserve end-organ tissue perfusion through increasing myocardial contractility
and CO, and decreasing filling pressures [4]. Vasopressors or inoconstrictors,
via
The use of temporary MCS devices is in the rise for the purpose of maintaining hemodynamics in CS [4, 119]. They are usually used as a bridge-to-decision for myocardial recovery, heart transplantation, palliation, or a durable ventricular assist device (VAD) [4, 23, 90]. Various devices have been developed and studied after the SHOCK trial which was conducted when only intra-aortic balloon pump (IABP) device was available [13]. The devices included axial LV-to-aorta pumps (Impella®), left atrium (LA)-to-aorta assist devices (TandemHeart®), right atrium (RA)-to-aorta pump (VA-ECMO) and devices for RV support [13, 119]. The devices are usually classified according to the pump type: volume-displacement pumps (i.e., IABP), continuous-flow pumps (i.e., Impella®) or centrifugal-flow (i.e., TandemHeart®, VA-ECMO) MCS [120]. In the National Cardiovascular Data Registry, 2.4% and 0.7% of percutaneous coronary intervention (PCI) cases were provided IABP and other MCS devices, respectively. IABP was mostly (63.3%) initiated after the start of PCI, while the other MCS devices were mostly (77.6%) inserted prior to PCI [121]. MCS devices reduce ventricles pressure and volume (i.e., unloading). These devices have potential ability to perverse vital organs perfusion, support circulation, amplify coronary perfusion, contain infarct size, reduce congestion and pulmonary edema by decreasing intracardiac filling pressures, and decrease LV volumes, wall stress, and myocardial oxygen demand [120]. Centres’ initial experiences with MCS devices demonstrated their benefit [37, 52, 122, 123, 124]. However, mortality benefit was not shown in the subsequent pivotal RCTs [36, 38], or meta-analyses [125, 126]. Table5 (Ref. [4, 23, 36, 37, 38, 52, 90, 119, 120, 124, 127, 128, 129, 130, 131, 132, 133]) summarizes the features and evidence from pivotal studies of the available temporary MCS devices.
| Study | Sample size (N) | Interventions | Conclusion | Jadad scale |
| Sites number (S) | Follow-up (F/U) | Population | (0–5) | |
| Vasopressors and inotropes | ||||
| De Backer 2010 [97] | N = 1679 | - Dopamine versus norepinephrine | - No difference in death | 5 |
| SOAP II | CS subgroup (17%) | - Shock in general | - More adverse events with dopamine use | |
| S = 8 | F/U: 28 days | - CS subgroup: higher mortality rate with dopamine use | ||
| Levy 2011 [98] | N = 30 | - Epinephrine versus NE – Dobutamine | - Similar efficacy between groups in term of global hemodynamic effects | 2 |
| S = 1 | F/U: 26 months | - Dopamine-resistant CS without ACS | - Transient lactic acidosis, higher HR and arrhythmia, and inadequate gastric mucosa perfusion with epinephrine | |
| - NE-dobutamine may be more reliable and safer | ||||
| Levy 2018 [99] | N = 57 | - Epinephrine versus NE | - Epinephrine use compared with NE was associated with similar effects on | 5 |
| OptimaCC | F/U: 60 days | - CS after acute MI | arterial pressure and CI but higher incidence of refractory shock | |
| S = 9 | ||||
| Inodilators | ||||
| García-González 2006 [100] | N = 22 | - Levosimendan versus dobutamine | - Levosimendan improved CPO and CI [100] | 1 |
| S = 1 | F/U: at 24 and 30 hr | - CS in STEMI patients treated with PPCI | - Levosimendan significantly reduced IVRT, and increased E/A ratio [101] | |
| - No difference in improving long-term survival [102] | ||||
| Fuhrmann 2008 [103] | N = 32 | - Add-on levosimendan versus enoximone | - Levosimendan may improve survival compared with enoximone | 3 |
| S = 1 | F/U: at 30 days | - CS complicating acute MI | ||
| Husebye 2013 [104] | N = 61 | - Levosimendan versus matching placebo | - Levosimendan improved contractility post ischemia | 5 |
| LEAF | CS subgroup (15%) | - STEMI treated with PPCI complicated by HF | - Levosimendan did not increase arrhythmias | |
| S = 1 | F/U: 42 days | - Similar results obtained in CS subgroup | ||
| Mathew 2021 [105] | N = 192 | - Milrinone versus dobutamine | - No difference in composite of in-hospital death from any cause, resuscitated cardiac arrest, receipt of a cardiac transplant or MCS, nonfatal MI, TIA or stroke, or initiation of RRT (primary outcome) | 5 |
| DOREMI | F/U: index hospitalization | - CS (SCAI stages B, C, D, or E), including patients with ACS | - No difference in the individual components of primary composite outcome | |
| S = 1 | (secondary outcomes) | |||
| Nitric oxide synthase inhibitors | ||||
| Cotter 2003 [106] | N = 30 | - Supportive care plus L-NAME versus supportive care only | - NOSi significantly reduced mortality and improved MAP and UOP | 1 |
| LINCS | F/U: at 30 days | (no treatment) | ||
| S = 1 | - ASC complicated by refractory CS | |||
| Dzavík 2007 [107] | N = 79 | - L-NMMA (in 5 regimens) versus matching placebo (normal | - L-NMMA resulted in modest increases in MAP at 15 min but no differences | 4 |
| SHOCK-2 | F/U: 30 days | saline) | at 2 hr | |
| S = 22 | Dose-ranging study (Phase II) | - Acute MI complicated by persistent CS | - No difference in 30-day mortality | |
| Alexander 2007 [108] | N = 398 | - Tilarginine (L-NMMA) versus matching Placebo | - Tilarginine did not reduce mortality rates | 4 |
| TRIUMPHb | F/U: 6 months | - MI complicated by refractory CS despite opening the IRA | - Early mortality rates were high | |
| S = 130 | - Note: enrolment was terminated at 398 patients based on a prespecified futility analysis. | |||
| aJadad scale: 3-item scale examines randomization, blinding, and patient
disposition; 5-point score: 0 and 2 (poor quality) and 3 to 5 (good quality)
[109, 110, 111].
bThe study was terminated prematurely for futility analysis. Abbreviations: ACS, acute coronary syndrome; CI, cardiac index; CPO, Cardiac power; CS, cardiogenic shock; F/U, follow-up duration; hr, hour; HF, heart failure; HR, heart rate; IRA, infarct-related artery; IVRT, isovolumetric relaxation time; LEAF, LEvosimendan in Acute heart Failure following myocardial infarction; LINCS, L-NAME (a NO synthase inhibitor) In the treatment of refractory Cardiogenic Shock; L-NAME, N | ||||
The best evidence for IABP use informing about mortality benefit comes from the IABP-SHOCK II trial [39, 130, 131] which did not show any benefit, leading to downgrading of guidelines’ recommendations on routine IABP use [134]. However, IABP may improve outcomes in the presence of mechanical complications. Thus, the guidelines consider IABP for those patients [56]. Multiple observational studies [135, 136, 137], RCTs [138, 139, 140], and meta-analyses [141, 142, 143, 144] have concluded the lack of mortality benefit as well. As compared with IABP, Impella®LP 2.5 significantly improved hemodynamics but not 30-day mortality (46%) in the pilot ISAR-SHOCK study [37]. Whereas, 30-day mortality rate was higher (64.2%) in those who received Impella®LP 2.5 in a multicentre registry [145]. Routine use of Impella®CP, in the exploratory IMPRESS study, did not show 30-day or six-month mortality benefit [36]. In addition, findings from two meta-analyses of RCTs did not report statistical difference in 30-day [126, 146] or six-month mortality when compared with IABP [146]. Evidence from observational studies on Impella® devices use versus IABP support showed conflicting results (i.e., increased harm such as in-hospital mortality and major bleeding [147], improved survival with early initiation of MCS [148, 149] or lack of association with 30-day mortality [150]). Similarly, studies on patients undergoing high-risk PCI, but not presenting with CS, showed inconsistent results with the use of Impella® devices [151, 152, 153, 154, 155, 156]. When acute MI patients presenting with CS were randomized to either TandemHeart® or IABP support, the former device improved hemodynamic parameters but not 30-day mortality [52, 124, 126]. The reported rates of 30-day and six-month mortality were more than 40% [157]. There are few published non-comparative studies that described the experience with TandemHeart® device in patients undergoing high-risk PCI including those who developed CS [158, 159, 160]. The evidence for VA-ECMO use in acute MI complicated with CS, including those who experienced cardiac arrest, is based on observational studies. In the cardiac arrest setting, VA-ECMO may be considered in patients who are refractory to cardiopulmonary resuscitation (CPR) (i.e., extracorporeal CPR (E-CPR)). A recent systematic review of cohort studies concluded that the use of VA-ECMO in CS complicating acute MI may have survival benefit [161]. The first mortality risk score, ENCOURAGE score, has been proposed to dictate the decision on the indication of VA-ECMO in acute MI patients based on pre-ECMO factors that have been correlated with mortality [162]. The EURO SHOCK (NCT03813134) is a multi-center, open-label, RCT that is underway and compares early initiation of VA-ECMO plus standard pharmacological therapy after acute PCI with standard pharmacological therapy alone. The primary endpoint is 30-day mortality. In addition, analysis of the cost-effectiveness will be conducted [163]. Earlier cohort studies in patients with ACS complicated by refractory CS or cardiac arrest who were on ECMO, concluded that in-hospital survival rate was improved [164, 165], and early initiation of ECMO would result in better outcomes and successful ECMO weaning [165]. In the aforementioned setting, the results of two meta-analyses of observational studies showed that ECMO has improved survival [166, 167]. However, one of the meta-analyses showed favourable neurological outcomes [167], while the other one reported a significantly higher complications rate, (i.e., neurological deficit and kidney impairment) [166]. Several studies found that E-CPR in acute MI patients with CS and cardiac arrest resulted in acceptable survival rates and improved outcomes [168, 169, 170, 71]. Nonetheless, prior-ECMO support in patients with VAD was associated with postoperative complications especially RV and respiratory failure [172]. Finally, combining IABP with VA-ECMO is gaining more interest and has been associated with successful VA-ECMO weaning and improved mortality rates [173].
Acute RV dysfunction or failure may eventuate from different clinical setting such as acute MI, decompensated heart failure, fulminant myocarditis, orthotopic heart transplant, or following left VAD (LVAD) procedure [90, 174]. RV failure contributes to CS via three ways: RV infarction due to obstruction of proximal right coronary artery in the absence of LV failure; elevated pulmonary vascular resistance and/or PCWP leading to elevated pulmonary afterload; and RV failure complicating primary LV failure [59]. RV failure is associated with increased mortality and morbidity [59, 120]. The approach for managing RV failure includes treating the cause, sustaining adequate preload, decreasing RV afterload and improving RV contractility [120]. This is achieved by inotropic therapy, pulmonary vasodilation, and optimized volume status [174]. When medical therapy is insufficient, temporary MCS or destination therapy should be considered [120, 174]. Historically, MCS support has been limited to IABP [120]. At-present, examples of RV-support devices include CentriMag®, PROTEK Duo®, and Impella®RP [23, 90, 129, 133]. The CentriMag® ventricular assist system provides temporary right, left, or biventricular support. A preliminary study showed that CentriMag®, in patients with CS, provided temporary support with low rates of device-related complications without device failure events [175]. PROTEK Duo® catheter has an extracorporeal pump, and its use in the setting LVAD implantation and CS due to severe pulmonary hypertension has been described in case reports [133]. TandemHeart® adapted for RV support improved hemodynamic status without any reported intra-procedure mortality [176]. Following the promising initial experiences, Impella®RP device in the pivotal RECOVER RIGHT trial promptly improved hemodynamic parameters in patients with life-threatening RV failure and all patients were alive at 180 days follow-up [132].
| MCS device | Description | Hemodynamic effects | Characteristics | Pivotal/RCT (PICO) |
| LV support | ||||
| IABP | - Device: counterpulsation pump placed in descending aorta | - LV unloading (modest) | - Advantages: easy insertion, low cost, rare vascular complications | Thiele 2012 (IABP-SHOCK II) [38] |
| - Has 2 major components, balloon catheter and pump console to control the balloon | - Cardiac power ↑ | - Disadvantages: increase in CO is small, requires native heart beat and stable rhythm, arrhythmias mitigate its usefulness | - N = 600 | |
| - CO support/flow: 0.3–0.5 L/min | - Afterload ↓ | - Complications: spinal cord ischemia, infection, bleeding, retroperitoneal haematoma, limb ischemia, compartment syndrome, vascular trauma, stroke, thrombocytopenia | - P: CS complicating acute MI (STEMI/NSTEMI) and early revascularization. | |
| - Mechanism: balloon inflation and deflation (aorta) | - MAP ↑ | - Contraindications: AR, severe PAD or aortic disease | - I&C: IABP versus conventional treatment | |
| - Insertion: femoral artery, axillary artery | - LVEDP ↓ | - O-efficacy: no difference in 30-day mortality | ||
| - Cannula size: 7–9 Fr (arterial) | - LV preload - | (39.7% vs. 41.3%) | ||
| - Implantation: percutaneous | - Coronary perfusion ↑ | - Long F/U: no difference in mortality at 12 months | ||
| - Timing of balloon inflation and deflation is based on ECG or pressure triggers | [130] and 6.2 years [131] | |||
| Impella®LP 2.5 | - Device: micro-axial pump that decompresses LV and pumps | - LV unloading | - Advantages: axillary approach allows long-term support | Seyfarth 2008 (ISAR-SHOCK) [37] |
| blood into ascending aorta | - Cardiac power ↑↑ | - Do not require ECG or arterial waveform triggering | - 2-center, pilot, N = 26 | |
| - CO support/flow: 1.0–2.5 L/min | - Afterload ↓ | - Complications: haemolysis, valvular lesions, device migration, | - P: CS post-acute MI | |
| - Mechanism: axial flow continuous pump (LV-to-Aorta) | - MAP ↑↑ | CNS hemorrhage, CNS infarction, brain death, seizures | - I&C: Impella®LP 2.5 versus IABP | |
| - Insertion: femoral artery, axillary artery | - LVEDP ↓↓ | - Contraindications: mechanical aortic valve or LV thrombus, AR or stenosis, severe PAD | - O-efficacy: significant improvement in CI after 30 min with Impella®. Mortality at 30-day was 46% in both groups | |
| - Cannula size: 12–14 Fr | - LV preload ↓↓ | - O-safety: haemolysis and transfusion significantly | ||
| - Implantation: percutaneous | - Coronary perfusion ↑ | higher with Impella®LP 2.5 | ||
| - Max implant duration: 7–10 day | ||||
| Impella®CP | - Device: micro-axial pump that decompresses LV and pumps blood into ascending aorta | - As above | - As above | Ouweneel 2017 (IMPRESS) [36] |
| - CO support/flow: 3.7–4.0 L/min | - N = 48 | |||
| - Mechanism: axial flow continuous pump (LV-to-Aorta) | - P: CS post-acute MI | |||
| - Insertion: femoral artery, axillary artery | - I&C: Impella®CP versus IABP | |||
| - Cannula size: 12–14 Fr | - O-safety: more bleeding events and haemolysis | |||
| - Implantation: percutaneous | with Impella®CP | |||
| - Max implant duration: 7–10 day | - O-efficacy: no difference in 30-day survival and 6-month mortality (50%) in both groups | |||
| Impella®LP | - CO support/flow: 5.0 L/min | - As above | - As above | - No RCTs |
| 5.0 | - Mechanism: axial flow continuous pump (LV-to-Aorta) | |||
| - Insertion: femoral or axillary artery | ||||
| - Cannula size: 21–22 Fr | ||||
| - Implantation: surgical cutdown of artery prior to insertion of sheath | ||||
| - Max implant duration: 2–3 week | ||||
| Impella® 5.5 | - CO support/flow: 5.5–6.0 L/min | - LV unloading | - As above | - No RCTs |
| - Mechanism: axial flow continuous pump (LV-to-Aorta) | - Cardiac power ↑↑ | |||
| - Insertion: femoral or axillary artery | - Afterload ↓↓ | |||
| - Coronary perfusion ↑ | ||||
| TandemHeart® LV-FA | - Device: centrifugal pump with inflow cannula placed in LA and outflow cannula in one or both femoral arteries across inter- | - LV unloading | - Advantages: does not require ECG or arterial waveform triggering | Thiele 2005 [124] |
| atrial septum. | - Cardiac power ↑↑ | - Disadvantages: need for transseptal puncture, risk of dislodge- | - N = 41 | |
| - Pumps blood from LA to iliofemoral arterial system | - Afterload ↑ | ment of LA cannula | ||
| - MAP ↑↑ | - Complications: air embolism, cardiac perforation, tamponade, | - P: CS post-acute MI | ||
| - Has 4 components: a 21-F transseptal cannula, a centrifugal | - LVEDP ↓↓ | residual atrial septal defect, massive RV-to-aorta shunt, thrombo-or | - I&C: TandemHeart® versus IABP | |
| pump, a femoral arterial cannula, and a control console | - LV preload ↓↓ | air-embolism, haemolysis, vascular trauma, limb ischemia | - O-efficacy: hemodynamic improvement is greater | |
| - CO support/flow: 2.5–5.0 L/min | - Coronary perfusion - | - Contraindications: profound coagulopathies, bleeding diatheses | with TandemHeart®. Mortality at 30 days were similar | |
| - Mechanism: centrifugal flow continuous pump (LA-to-Aorta) | e.g., HIT or DIC, RA or LA thrombus, severe PAD | (43% vs. 45%) | ||
| - Insertion: femoral artery or femoral vein | - O-safety: significantly more limb ischemia, blood | |||
| - Cannula size: 12–19 Fr (arterial), 21 Fr (venous) | transfusions, DIC in TandemHeart® arm | |||
| - Implantation: transeptal puncture | ||||
| - Max implant duration: 2–3 week | Burkhoff 2006 [52] | |||
| - 12-center, N = 42 | ||||
| - P: refractory CS (post-acute MI; 70%) | ||||
| - I&C: TandemHeart® versus IABP | ||||
| - O-efficacy: significantly greater increases in CI and greater decreases in PCWP over first 16 hours with TandemHeart®. Survival at 30-day was not significantly different (53% vs. 64%) | ||||
| - O-safety: no difference in severe adverse events or bleeding | ||||
| RV support | ||||
| CentriMag® | - Device: centrifugal pump with magnetically levitated propeller | - RV unloading | - Advantages: easy insertion and maintenance, reliable, low thrombosis risk | - No RCTs |
| - Has centrifugal pump, electric motor, and console. | - Complications: infection, Bleeding, Systemic heparinization re | |||
| - CO support/flow: up to 9.9 L/min | quired, Limited patient mobility, Arrhythmia | |||
| - Mechanism: centrifugal-flow | ||||
| - Insertion: femoral-to-femoral bypass | ||||
| Impella®RP | - Device: axial catheter-based pump or RV assist device that pumps blood from RA to PA | - RV unloading | - Advantages: need for only single venous access site | Anderson 2015 (RECOVER RIGHT) [132]- 15-center, prospective, non-RCT, N = 30 |
| - CO support/flow: 4.0–5.0 L/min | - P: refractory RV failure following acute MI, car- | |||
| - Mechanism: axial flow continuous pump (RA-to-PA) | diotomy or LVAD implantation | |||
| - Insertion: femoral vein | - I&C: Impella®RP versus none | |||
| - Cannula size: 22 Fr (venous) | - O-efficacy: immediate hemodynamics improvement with significant increase in cardiac index and decrease in CVP. Overall survival at 30 days was 73.3%. All patients discharged were alive at 180 days | |||
| PROTEK | - Device: | - RV unloading | - | - No RCTs |
| Duo® | - CO support/flow: | |||
| - Mechanism: extracorporeal centrifugal-flow (RA-to-PA) | ||||
| - Insertion: superior vena cava | ||||
| - Cannula size: double lumen, 29 or 31 Fr | ||||
| TandemHeart® | - CO support/flow: 4.0 L/min | - RV unloading | - Complications: infection, bleeding, ischemia of lower extremities | - No RCTs |
| RA-PA | - Mechanism: extracorporeal centrifugal-flow continuous pump (RA-to-PA) | |||
| - Insertion: internal jugular vein | ||||
| - Cannula size: 29 Fr (venous) | ||||
| LV and RV support | ||||
| VA-ECMO | - Device: heart-lung bypass machine | - RV unloading | - Advantages: metabolic derangement and deleterious systemic effects | - No RCTs |
| - Has centrifugal pump and membrane oxygenation | - Cardiac power ↑↑↑ | of CS can be corrected within hours of initiation | ||
| - V-V for oxygenation only or V-A for oxygenation and circulatory support | - Afterload ↑↑↑ | - Disadvantages: afterload increase may worsen PCWP and LV func- | ||
| - CO support/flow: 7.0 L/min | - MAP ↑↑ | tion, vasodilators or MCS e.g., IABP or Impella® may be needed to reduce afterload | ||
| - Mechanism: centrifugal flow continuous pump (RA-to-Aorta) | - LVEDP |
- Complications: air embolism, LV dilation, LV blood stasis, pulmonary edema, circuit clots, haemolysis, acquired von Willebrand disease, | ||
| - Insertion: femoral vein or femoral artery | - LV preload ↓ | HIT, VTE, GI or pulmonary bleeding, sepsis, DIC | ||
| - Cannula size: 14–19 Fr (arterial), 17–21 Fr (venous) | - Coronary perfusion - | - Contraindications: significant aortic insufficiency, severe PAD | ||
| - Implantation: percutaneous or surgical cutdown | ||||
| - Max implant duration: 3–4 week | ||||
| Abbreviations: AR, aortic valve regurgitation; CI, cardiac index; CNS, central nervous system; CO, cardiac output; CS, cardiogenic shock; CVP, central venous pressure; DIC, disseminated intravascular coagulation; ECG, electrocardiogram; ECMO, extracorporeal membrane oxygenation; FA, femoral artery; Fr, French; F/U, follow-up; GI, gastrointestinal; HIT, heparin induced thrombocytopenia; IABP, intra-aortic balloon pump; IMPRESS, IMPella versus IABP Reduces mortality in STEMI patients treated with primary PCI in Severe cardiogenic SHOCK; LA, left atrium; LV, left ventricle or ventricular; LVAD, left ventricular assist device; LVEDP, left ventricle end-diastolic pressure; MAP, mean arterial blood pressure; Max, maximum; MCS, mechanical circulatory support; MI, myocardial infarction; NSTEMI, non-ST segment elevation myocardial infarction; PA, pulmonary artery; PAD, peripheral artery disease; PCWP, pulmonary capillary wedge pressure; PICO, population, intervention, comparison, and outcomes; RA, right atrium; RCTs, randomized controlled trials; RECOVER RIGHT, Impella RP Right Ventricular Heart Failure Trial; RV, right ventricle; SHOCK-IABP II, Intraaortic Balloon Pump in Cardiogenic Shock II; STEMI, ST-segment elevation myocardial infarction; V-A, veno-arterial; VTE, venous thromboembolism; V-V, Veno-veno. | ||||
Taken together, a retrospective study documented a five-year experience of MCS devices as a bridge-to-decision in patients with refractory CS. Acute MI was the etiology of CS in 49% of patients. Initially, the use of temporary VAD was in 49% and VA-ECMO in 51% of patients. Implantable VAD and heart transplantation were performed in 26% and 11% of patients, respectively. Survival to discharge from hospital was 49% [177]. There are few controversial questions related to temporary MCS in the care of CS patients, the main ones focus on device selection and time of device initiation [4, 23]. Device selection in patients with severe hemodynamic instability, should be guided by several factors such as, patient’s hemodynamic status, advantages and disadvantages of the device, its technical feasibility, and the overall goals of therapy. As a general approach, IABP is usually the initial choice given the familiarity with it. However, the pharmacologic support is usually required but not with the Impella® devices. Thus, as a next step Impella®LP 2.5 or CP may offer a more powerful support. With the continuous deterioration, TandemHeart®, VA-ECMO, or Impella®LP 5.0 can be considered. Early insertion and initiation of MCS device and before PCI can result in significant hemodynamic improvement and mitigate ischemia and the worsening cardiac function [120]. The recent European guidelines recommended the short-term use of MCS in selected patients with ACS and CS, and did not recommend routine IABP use [118, 178], which should be considered in patients with mechanical complications [56, 178]. Destination therapy with durable MCS (i.e., LVAD) or heart transplantation is warranted in refractory CS despite revascularization, inotropic therapy and temporary MCS [4].
Early, successful myocardial revascularization in ACS complicated by CS is the only therapy with proven mortality benefit [21, 32, 179]. Over 23 years from 1975 to 1997, despite the non-significant change in the rate of CS complicating acute MI, the survival rate increased in parallel with the increased use of coronary reperfusion strategies [29].
Thrombolytic or fibrinolytic therapy should be considered for STEMI patients if timely PCI is delayed or not feasible [90, 180]. Furthermore, data on the efficacy of thrombolytics in CS patients is very limited as they were frequently excluded from the respective clinical trials [181]. In Cath-PCI registry, there was a significant reduction in thrombolysis use, from 4% to 1.2%, between 2005 and 2013, respectively [35].
In patients presenting with ACS complicated by CS, emergency coronary angiography is recommended [118, 178]. The landmark SHOCK trial has shown significant six-month mortality benefit on long-term follow up [182], but not at 30 days, with early revascularization using either PCI or coronary artery bypass grafting (CABG) [21]. In Cath-PCI registry, the number of patients who underwent PCI for acute MI complicated by CS increased dramatically between 2005 and 2013 [35]. Currently, immediate CABG has been reported in less than 4% of patients [90]. Emergency CABG is usually indicated when coronary arteries are not amenable to PCI [118, 178].
The majority of patients with CS (i.e.,
Patients with acute MI complicated with CS are usually excluded from studies examining PCI arterial access site, stent type or aspiration thrombectomy. Trans-radial access has been endorsed by the AHA and advocated by CS care centres as the access of preference [189, 190]. Trans-radial access has been associated with lower rates of mortality, major adverse cardiac and cerebrovascular events [191, 192], and major bleeding [191]. In Cath-PCI registry, the use of radial access significantly increased from 0.4% to 4.2% between 2005 and 2013, respectively [35]. Interestingly, in a study from England and Wales the increase in radial access use with primary PCI was from 24.6% in 2007 to 76.5% in 2014 [193]. Routine aspiration thrombectomy is not recommended as there is no convincing evidence to support it in CS [189]. Drug-eluting stents, apparently, are favoured over bare-metal stents despite the indefinite evidence in CS [189, 194, 195, 196].
Recommendations for pharmacologic therapy in CS are similar to those for ACS without CS, since there are no dedicated RCTs in CS. Antithrombotic therapy (i.e., antiplatelets and anticoagulation) is a must prior, during, and after PCI [2, 90, 189]. Once the shock status is resolved, beta-blockers and renin-angiotensin system inhibitors should be considered as tolerated [2].
Interventional cardiologist ideally collaborates with general cardiologist, cardiothoracic surgeon, intensivist, heart failure specialist, and specialized nurses in a multidisciplinary approach (i.e., the shock team) [4, 90, 119] to decide on the most effective acute interventions and destination therapy as appropriate [90, 119].
Initial risk stratification and prognosis of patients with CS can be assessed by several risk scores. However, not all of them were validated [2, 59]. IABP-SHOCK and CardShock scores were investigated. The IABP-SHOCK score stratifies the risk of 30-day mortality (low, intermediate, high) by using six variables (age, prior stroke, Thrombolysis In Myocardial Infarction flow grade post PCI, serum levels of lactate, creatinine, and glucose) [197]. The CardShock score estimates the risk of short-term mortality (low, intermediate, high) by using seven variables (age, prior CABG, previous MI, ACS etiology, LV ejection fraction, lactate level, confusion) [5].
The evidence for management of patients presenting with MI and CS is usually extrapolated from trials that enrolled hemodynamically stable MI patients. Although not optimal, it is considered an acceptable approach given the lack of other alternatives [189]. Conducting RCTs in CS patients is difficult because CS includes broad spectrum of patients of variable etiologies and severities with consequent variability of treatment outcomes [13]. Some unanswered clinical questions include, initial selection of a vasoactive agent [96], most appropriate MCS device strategy [23], or routine use of MCS devices as adjunct to coronary revascularization. Trials are underway that address some of these issues [120].
Acute MI with myocardial dysfunction is the most frequent cause of CS. It is characterized by circulatory collapse and inadequate end-organ tissue perfusion due to impaired CO. CS triggers unfavourable compensatory mechanisms that create a vicious cycle which is difficult to reverse and eventually leads to death. Interrupting this vicious cycle and restoring hemodynamic stability is the fundamental comprehensive treatment of CS. Although it has been declined over time, the mortality rate is still unacceptably high despite the advancement in MCS modalities and coronary revascularization practices. Early identification of CS, rapid diagnosis, and prompt initiation of therapy may improve prognosis. Clinical assessment with physical examination and early invasive hemodynamics, helps in the identification of CS phenotype, risk stratification, and severity classification according to SCAI taxonomy. Thus, this can guide a tailored and optimized therapeutic approach in critically ill patients. To date there is no pharmacological or nonpharmacological intervention that showed a mortality benefit.
Vasoactive agents are considered the initial management of hemodynamic instability. There is no convincing evidence of the superiority of one agent over another. The current availability of MCS devices has broadened the therapeutic choices for hemodynamic support. Their early initiation instead of escalating the doses of vasoactive agents may mitigate further deterioration. Appropriate MCS device should be carefully selected and paired with the right patient at the right time. Device selection process should be dictated by several factors. Early coronary revascularization, by PCI or CABG, is the cornerstone therapy which improves mortality in patients with acute MI complicated by CS. Adequately powered RCTs are urgently needed to address the controversial and unanswered questions.
CardShock, Cardiogenic Shock; COMPLETE, The Complete versus Culprit-Only Revascularization Strategies to Treat Multivessel Disease after Early PCI for STEMI; DOREMI, Dobutamine Compared with Milrinone; ENCOURAGE, prEdictioN of Cardiogenic shock OUtcome foR AMI patients salvaGed by VA-ECMO; GUSTO, Global Utilization of Streptokinase and Tissue plasminogen activator for Occluded coronary arteries; IMPRESS, IMPella versus IABP Reduces mortality in STEMI patients treated with primary PCI in Severe cardiogenic SHOCK; RECOVER RIGHT, Impella RP Right Ventricular Heart Failure Trial; SHOCK, Should We Emergently Revascularize Occluded Coronaries for Cardiogenic Shock; SHOCK-IABP II, Intraaortic Balloon Pump in Cardiogenic Shock II.
RK: design, literature search, literature summaries, writing, tables summary, figures, revision, and responses to reviewers. SE: design, general writing and critical revision.
Not applicable.
Thanks to Leen Abu Afifeh (medical student at Qatar University) for her support in literature search. Thanks to all the peer reviewers for their time in the first place and for their valuable opinions and suggestions which helped refine and improve the manuscript.
Open Access funding provided by the Qatar National Library.
The authors declare no conflict of interest.







