1 Department of Neurology, Johns Hopkins University, Baltimore, MD 21794, USA
2 Montreal Heart Institute, Université de Montréal, Montreal, QC H1B, Canada
Academic Editor: Peter A. McCullough
Ticagrelor was compared to clopidogrel in the PLATO trial, which randomized
18,624 patients with ACS to either ticagrelor (180 mg loading dose plus 90 mg
twice daily maintenance dose) or clopidogrel (300–600 mg loading dose plus 75 mg
once daily maintenance dose) both on top of aspirin [1]. Ticagrelor leads to a
significant reduction in the primary endpoint (a composite of death from vascular
causes, myocardial infarction, or stroke) compared to clopidogrel (9.8% vs.
11.7%, 95% confidence interval (CI): 0.77–0.92, p
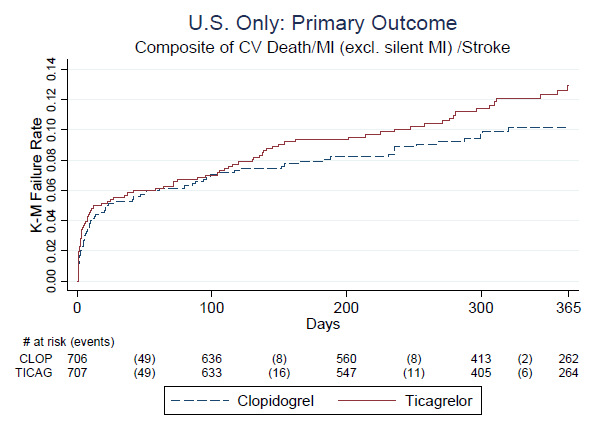 Fig. 1.
Fig. 1.PLATO outcomes in the USA. No early benefit of ticagrelor and growing over time superiority of clopidogrel. Source: R. Fiorentino, Clinical reviewer.
The controversy surrounded this observation led to multiple post-hoc
analyses, which generated a questionable hypothesis that higher daily aspirin
dose of 300–325 mg in the US (53.6%) compared to the rest of the world (1.7%)
was the exclusive key factor (out of 37 variables explored) to explain the
geographical interaction of the reason why ticagrelor was harmful in the US trial
population [2]. In the ticagrelor-aspirin
Moreover, a highly significant diabetic-aspirin interaction in PLATO has later
challenged the ticagrelor-high dose aspirin hypothesis [5]. In fact, diabetics
(regardless of treatment arm) had a highly significant reduction in major adverse
cardiovascular events, all-cause mortality, and vascular mortality if they
received higher aspirin doses
We finally gained access to the detailed dataset of 938 PLATO deaths reported to the FDA. Those records were matched with original local patient-level data from sites controlled by the sponsor revealing that actual existence, precise dates and proper causes of some deaths in PLATO were inaccurately reported in favor of ticagrelor [9]. Moreover, for the first time we gained access to deaths dependent on the monitoring source. Somehow per country deaths on ticagrelor and clopidogrel were never disclosed to public, despite the fact that PLATO Investigators acknowledged “geographical” differences in trial outcomes [10]. The CRO’s reported outcomes from USA, Russia, Georgia, and most of Ukraine, while sites in other 39 countries were monitored by the sponsor. Such method used in the PLATO trial [10] led to death reporting from combined “North America” mixing true unbiased US data monitored by CRO with heavily misreported outcomes in Canada [9] which were controlled by the sponsor. We compare if there were any differences in aspirin doses when deaths were reported by the sponsor versus independent CRO’s. The details are outlined in the Table 1.
| Source/Country | Enrolled (n) | High-dose Aspirin (%) | Deaths | Deaths | ||
| (Clopidogrel) | (Ticagrelor) | |||||
| CRO: | ||||||
| United States* | 1413 | Yes (53.6%) | 24 | 29 | 5 | |
| Russia** | 678 | None (0%) | 19 | 29 | 10 | |
| Georgia** | 519 | None (0%) | 7 | 12 | 5 | |
| Ukraine*** | 163*** | None (0%) | 6 | 5 | –1 | |
| Total | 2773 | Yes (27.6%) | 56 | 75**** | 19 | |
| Sponsor: | ||||||
| Total | 15,851 | 781 (4.9%) | 462**** | 345 | –117**** | |
| *: ReSearch Pharmaceutical Services (Fort Washington, Pennsylvania, USA). **:
Evidence CRP, now Worldwide Clinical Trials (Morrisville, North Carolina, USA).
***: CRO (163 patients, 9 deaths); sponsor (6 patients, 2 deaths). ****:
p | ||||||
These FDA-issued data indicate that CRO’s reported deaths numbers are in the opposite direction than PLATO sponsor reported death numbers, and this difference was highly significant. Such observation suggests that the “surprising” results were not PLATO-US outcomes, but those primary events misreported outside US. Lack of such obvious analyses (CRO’s versus Sponsor) not conducted by the FDA is unexplained and intriguing. Indeed, there were 39 variables tested to explain PLATO-US phenomenon [2], but surprisingly why such a simple variable was never explored and disclosed to public. Why the deaths distributions by country were never disclosed despite almost 100 of secondary PLATO publications? Releasing such death distributions in the countries monitored by independent CRO’s could justify a thorough review of all sponsor-reported trial outcomes. Importantly, ticagrelor inferiority in Russia and Georgia was even stronger than in the US, but this result had nothing to do with the higher aspirin doses. Both Russia and Georgia are governed by European Society of Cardiology ACS recommendations, and aspirin in the daily use of over 100 mg for cardiac indications is not used in both countries. In fact, local pharmacies in both countries are not even offering aspirin in the 325 mg dosage. In the formularies, there are cardio aspirins with 100 mg/pill or “аспирин кардио” for Russia, and “ ასპირინი კარდიო” in Georgia (Bayer), or less popular cardiomagnyl by Takeda containing aspirin daily 75 mg dose in both countries. Gaining access to the complete PLATO death dataset issued by the FDA was determinant to explore in depths the reason(s) for possible discrepancies in deaths reporting. With these new data we were able to disclose the striking difference in mortality by each country, and to link this difference to the monitoring source. Excess ticagrelor deaths in the countries with no high-aspirin dose controversy strongly suggest the artificial nature of the existing hypothesis to explain inversed US-PLATO outcomes. The comparison between the FDA records and local patient-level data from sites controlled by the sponsor revealed that actual existence, precise dates and proper causes of some deaths in PLATO were inaccurately reported in favor of ticagrelor [9]. Moreover, there is a massive discrepancy between primary death causes reported to the FDA, and those utilized by the PLATO Investigators for numerous secondary reports published in top journals for over decade [11]. Examining cancer deaths reveled that at least 8 clopidogrel events were misreported in PLATO favoring ticagrelor as well [12].
In summary, dose of aspirin has nothing or very little to do with ticagrelor “benefit”. Outcomes misreporting may be much more important. We now know that it is not only the US paradox, but the outcomes in the countries not monitored by the sponsor consistently exhibited ticagrelor inferiority in mortality over clopidogrel. In both Russia and Georgia aspirin is not used at the dose over 100 mg/daily for any cardiac indication, but ticagrelor deaths were higher than in the US.
VS wrote the initial draft and analyzed the FDA-issued PLATO deaths dataset. JT made a critical revision and reassessed the evidence.
Not applicable.
Our true appreciation to clinical and statistical FDA reviewers on ticagrelor New Drug Application 22-433 for their integrity a courage with the “no approval” recommendation.
This research received no external funding.
The authors declare no conflict of interest.

