1 Truth for Health Foundation, Tucson, AZ, USA
2 Union Square Medical Associates, San Francisco, CA, USA
3 Yale School of Public Health, New Haven, CT 06510, USA
Keywords
- SARS-CoV-2
- COVID-19
- Hydroxychloroquine
- Ambulatory treatment
- Mortality
Hydroxychloroquine is the most widely prescribed intracellular anti-infective for human SARS-CoV-2 infection and COVID-19 syndrome. There have been 296 studies, 220 of which are peer reviewed, 246 comparing treatment and control groups [1]. This agent is successfully used in both prophylaxis and early therapy (Fig. 1). As a general principle, the earlier hydroxychloroquine is started in the course of illness, the larger treatment effects can be observed. These effects are greatly enhanced by the use of agents in combination to address SARS-CoV-2 replication, cytokine storm, and thrombosis [2, 3]. Early treatment of SARS-CoV-2 infection has the largest opportunity to control the outbreak since efforts are placed on those acutely ill with COVID-19 where there are interventions to control spread of disease, reduce the intensity and duration of illness, generate natural immunity, and prevent hospitalization and death, resulting in greater population protection from SARS-CoV-2 [4].
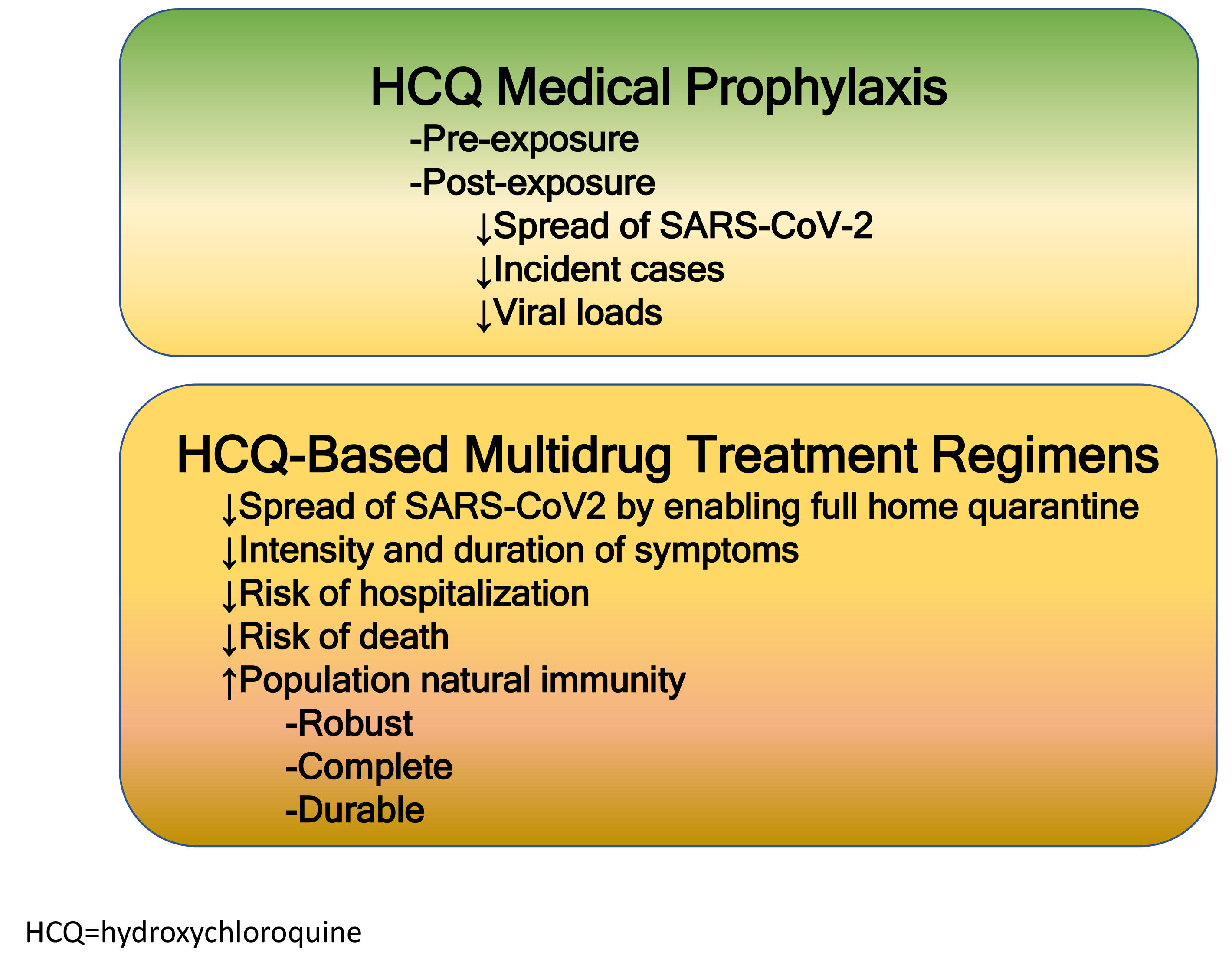 Fig. 1.
Fig. 1.Roles of hydroxychloroquine in the prevention and treatment of SARS-CoV-2 infection and the COVID-19 syndrome.
In this issue of Reviews in Cardiovascular Medicine, a paper from
Million et al. [5] describes the impact of hydroxychloroquine as a direct treatment
for early COVID-19 in ambulatory patients presenting to an expert day hospital
(Institut Hospitalo-Universitaire Med́iterraneé Infection) for early therapy
with the goal of reducing hospitalization and death. Among 10,429
patients who took on average 4 days to present for medical care and another day
to receive SARS-CoV-2 test results, 8315 were treated with hydroxychloroquine
and azithromycin and this group had an observed case fatality fraction of 0.06%.
The combination of hydroxychloroquine and azithromycin was associated with a
93% reduction in mortality (adjusted OR = 0.17, 95% CI 0.06–0.48, p = 0.0007,
n = 2015 age
The large study conducted over nine months from Million and
colleagues demonstrates that very low rates of mortality can be achieved in a
dedicated outpatient “hospital” even if therapy is started relatively late into
the course of viral replication [7]. A weakness of the study by Million et al.
is that it is from a single center and not randomized. These findings are
complemented by the contemporary early use of hydroxychloroquine in combination
with anti-inflammatory agents, and antithrombotics in some cases to extend the
range of hydroxychloroquine to higher risk patients and reduce the risks of both
hospitalization and death [7]. Thus in aggregate, the body of
literature supporting early ambulatory therapy for high-risk COVID-19 patients is
compelling and at this point, only large (n
PAM drafted, edited, finalized the manuscript, RBS provided edits and references, HAR provided edits.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.

