1 Section of Heart Failure & Transplantation, Cleveland Clinic Foundation, Cleveland, OH 44195, USA
2 Department of Clinical Therapeutics, National Kapodistrian University of Athens, 15771 Attica, Greece
3 Department of Nursing, Cleveland Clinic Foundation, Cleveland, OH 44195, USA
4 Department of Internal Medicine, University of Iowa, Iowa City, IA 52246, USA
Academic Editor: Peter A. McCullough
Abstract
In the context of the COVID-19 pandemic, many barriers to telemedicine disappeared. Virtual visits and telemonitoring strategies became routine. Evidence is accumulating regarding the safety and efficacy of virtual visits to replace in-person visits. A structured approach to virtual encounters is recommended. Telemonitoring includes patient reported remote vital sign monitoring, information from wearable devices, cardiac implantable electronic devices and invasive remote hemodynamic monitoring. The intensity of the monitoring should match the risk profile of the patient. Attention to cultural and educational barriers is important to prevent disparities in telehealth implementation.
Keywords
- Telemonitoring
- Heart failure with reduced ejection fraction
- Telemedicine
- COVID-19
Heart failure (HF) is a chronic disease characterized by high mortality, often associated with multiple comorbidities that require a multidisciplinary treatment approach [1, 2, 3, 4, 5]. Approximately 6 million adults live with HF in the United States. Assuring access of those patients to specialized care, potentially life-saving therapies, and overcoming psychosocial, economic, and geographical barriers is a significant challenge [6, 7, 8, 9, 10]. Health care systems have developed disease management programs organized as spoke and hub models to overcome these limitations [11]. Telemedicine and telemonitoring are an integral part of those programs, and their application increased exponentially in 2020–2021 during the COVID-19 pandemic [12, 13].
This review focuses on telemedicine’s current role in HF management and provides a practical framework for its application.
Telemedicine entails utilization of information technology to provide medical care by enabling communication between providers in one location and a patient or provider at another site [14]. Telemedicine encounters can be asynchronous (e.g., reviewing an echocardiogram, laboratory data, or responding to a patient call) or synchronous, real-time interaction (e.g., video consultation of a patient with a left ventricular assist device in a rural area with the advanced HF cardiologist). Telemonitoring is the use of information technology to monitor patients at a distance. Telemonitoring can be continuous or intermittent and occur in the inpatient or outpatient setting [15] (Fig. 1). A disease management program (DMP) is characterized by multidisciplinary involvement of HF nurses and physicians,s integrates all aspects of care and promotes adherence to clinical practice guidelines [16]. Current guidelines support the development and implementation of DMP in HF. Telemedicine usually occurs in the context of a DMP [17, 18, 19].
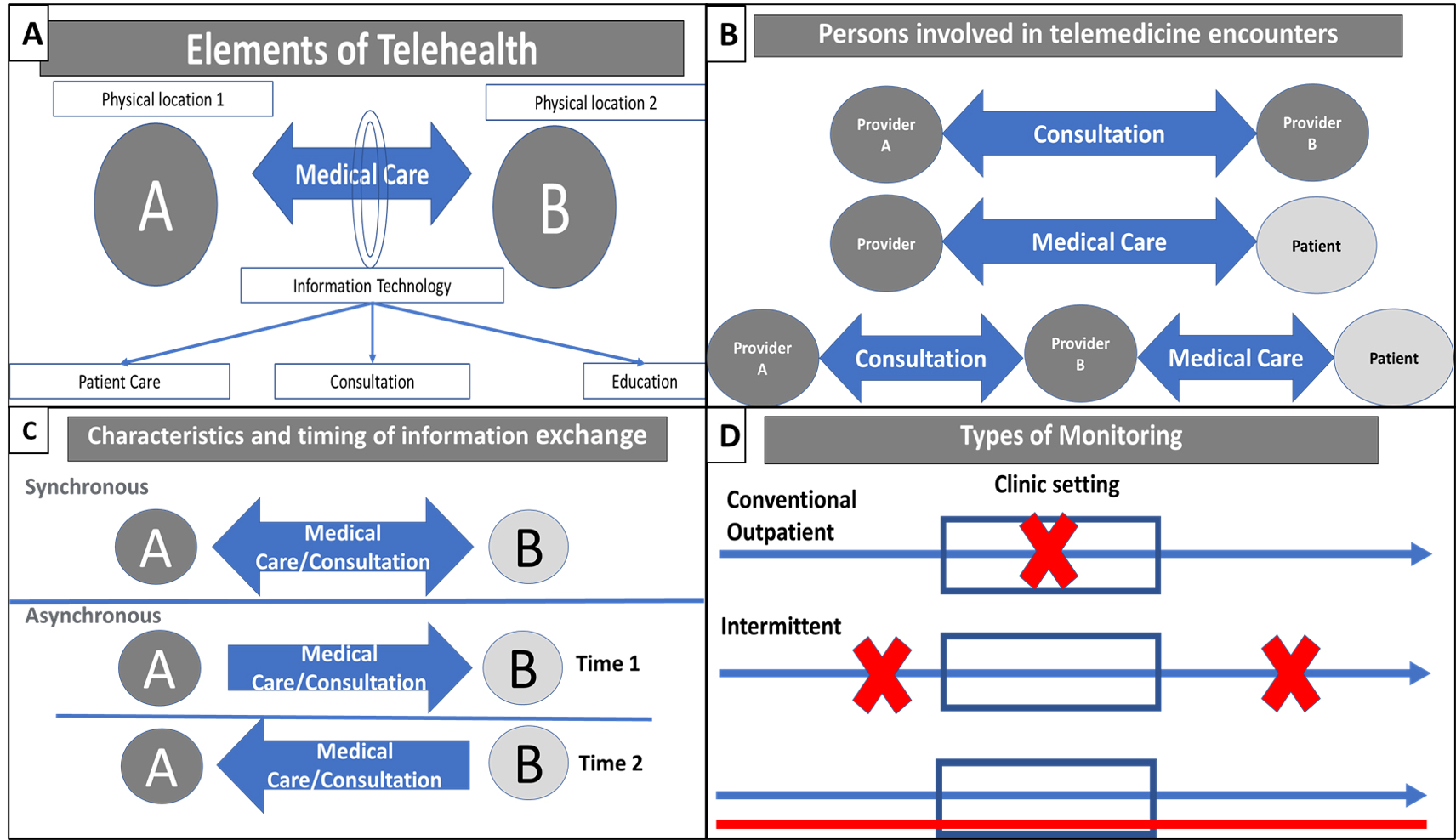 Fig. 1.
Fig. 1.Framework for telehealth implementation. (A) Elements of Telehealth. (B) Persons involved in telemedicine exchange. (C) Timing of information exchange. (D) Types of Telemonitoring.
Intermittent patient-provider encounters characterize ambulatory patient care.
Virtual visits (VV) are defined as synchronous audio/video interactions between
the provider and the patient. The uptake of virtual visits as an alternative to
in-person visits before the COVID 19 pandemic was relatively low. The lack of
familiarity with technology, regulatory, legal, and reimbursement concerns were
among the factors that hinder its widespread application. COVID -19 pandemic led
to the disappearance of many of the barriers mentioned above and the rapid
adoption of VV [20]. A recent HFSA statement summarizes current evidence and
provides recommendations for a successful VV preparation and billing codes to
assure the program’s sustainability in the pandemic context [21]. Follow-up
within 14 days of a HF hospitalization is required to reduce readmissions and
improve outcomes [22]. Recent pre-pandemic evidence shows that virtual visits may
be a safe alternative to in-person visits in post-discharge follow-up [23, 24].
The frequency of VV is variable and determined by professional judgment. In the
The Randomized Trial of Telephone Intervention in Chronic Heart Failure (DIAL),
the phone call frequency ranged from weekly in patients with New York Heart
Association (NYHA) III-IV symptoms, recent hospitalization, weight gain of more
than 2 Kg, and severe edema to monthly in patients with NYHA I, not hospitalized
within the previous year,
After the initial or between follow-up in-person or VV encounters, monitoring strategies may help detect parameters associated with increased decompensation risk and provide the opportunity to change trajectories [30, 31]. Early detections and recognition of congestion have been the target of telemonitoring in HF. The first event of congestion is increased filling pressures, followed by autonomic adaptation, decreased intrathoracic impedance, weight gain, and symptoms [32]. Below, we will review the different parameters and strategies that are available for remote monitoring of HF patients.
Current guidelines recommend daily weight in patients with HF to promote self-monitoring, adjust diuretic therapy, and volume overload detection [18]. A nested case-control study showed that increases in body weight of 2 or more pounds are associated with hospitalization for HF. The weight gain begin at least one week before admission [33]. The interval between weight gain and hospitalization may preclude the prescription of therapeutic interventions, especially if there are barriers to healthcare access. Significant congestion can develop in patients without significant weight gain due to sympathetically mediated volume redistribution in acute HF [31].
Hypertension is one of the most frequent comorbidities in patients with HF [34]
24-hour ambulatory blood pressure monitoring analysis has shown that most HF
patients have blunted blood pressure dipping during sleep [35]. The importance of
blood pressure control HF outcomes is increasingly recognized [36]. Home-blood
pressure management with goals
Wearables are external sensors that capture continuous functional or physiological data. The sensors are incorporated in different form factors such as patches, clothes, or smartwatches. The sensors are connected to other platforms that transmit, collect and interpret the data [40, 41]. Data from wearables include but are not limited to heart rate, blood pressure, activity, lung water content, and arrhythmias. Data processing and analysis may help to diagnose decompensation, monitor response to treatment, and detect potential factors associated with increased risk of decompensation [42, 43].
Atrial fibrillation affects approximately ¼ of patients with
heart failure. The presence of atrial fibrillation in heart failure is associated
with increases in stroke risk, heart failure hospitalization, and all-cause
mortality [44, 45, 46]. The Apple Heart Study that recruited 419,297 participants
over eight months showed that an irregular pulse detected by a commercial
smartwatch had a positive predictive value of 84% for detecting atrial
fibrillation on electrocardiogram (ECG) simultaneously with a subsequent
irregular pulse notification. Only 0.52% of the patients included in the study
received an irregular pulse notification. Although the proportion of patients
with HF was low (
The LINK-HF study analyzed the accuracy of a wearable multiparametric sensor to predict HF hospitalization [48]. One hundred patients in the Veteran Affairs Health System were enrolled, and 74% had HF with reduced ejection fraction. The intervention consisted of a disposable multisensor patch placed on the chest that collected continuous ECG waveform, continuous 3-axis accelerometry, skin impedance, skin temperature, and information on activity and posture. Data were uploaded continuously to a cloud via a smartphone. Primary information includes heart rate, heart rate variability, arrhythmia burden, respiratory rate, gross activity, walking, sleep, body tilt, and body posture. Machine learning was used to build a model that would predict heart failure hospitalization. The platform had sensitivity between 76% and 88% and a specificity of 87% to detect heart failure hospitalization precursors with a median time between alert and admission of 6.5 days. The impact of this technology to improve outcomes remains to be tested.
The remote dielectric sensing (ReDS
CIED such as implantable cardioverter defibrillators (ICD) and cardiac
resynchronization therapy defibrillators (CRT-D) improve survival in selected
patients with HF and reduced ejection fraction and provide HF-related diagnostic
information [18, 53] (Fig. 2A–C). Thoracic impedance is among the HF diagnostic
parameters provided by ICD and CRT-D. Intrathoracic fluid content influences the
electrical field’s impedance created by the current between the implantable pulse
generator and the pacing electrode’s tip. Congestion is associated with a
decrease in thoracic impedance [54, 55]. Intrathoracic impedance and intracardiac
filling pressures are inversely correlated. The decline of thoracic impedance
starts approximately 2 weeks before the onset of clinical congestion and has a
sensitivity of 76.9% to predict HF hospitalization. Persistent low thoracic
impedance is associated with increased mortality [56, 57]. The OptiVol fluid
index (Medtronic Inc., Minneapolis, MN, USA) calculates the difference between a
measured and a reference impedance and is inversely correlated with thoracic
impedance. A value greater than 60 is associated with increased mortality [58].
In the OptiLink HF trial, OptiVol telemonitoring failed to reduce HF
hospitalization or mortality in patients with advanced HF [59]. Of note less than
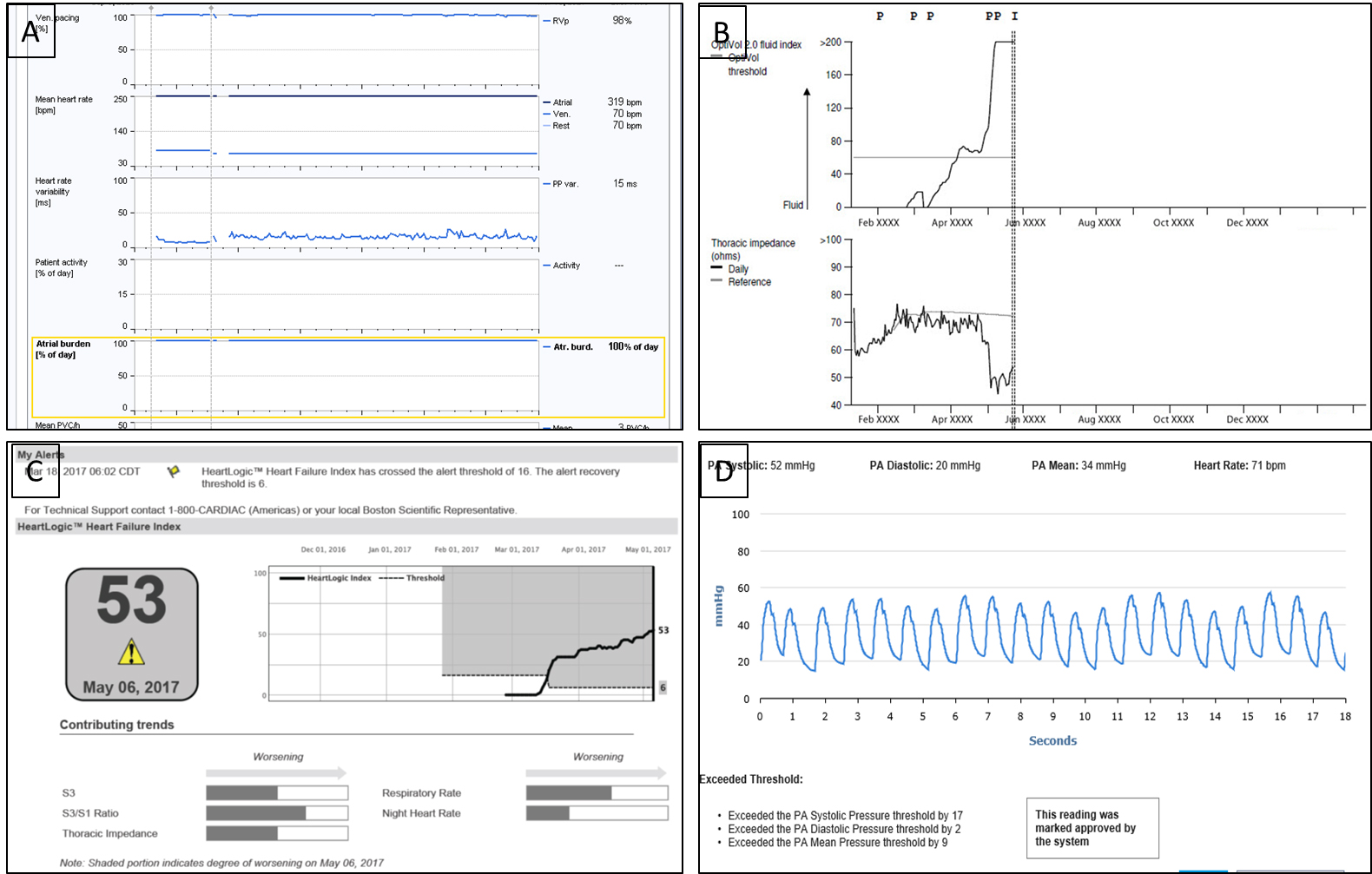 Fig. 2.
Fig. 2.Telemonitoring Interfaces. (A) CRT-D interrogation showing persistent atrial fibrillation. (B) Optivol tracing: Showing decrease in thoracic impedance and increase in Optivol Fluid index. (C) Multiparametric HeartLogic interface showing an alert parameter. (D) CardioMems interface showing a pulmonary artery pressure waveform and related parameters.
The Program to Access and Review Trending Information and Evaluate Correlation
to Symptoms in Patients With Heart Failure) (PARTNERS) HF prospective cohort
study evaluated the hypothesis that using a combined HF diagnostic algorithm
would improve the prediction of HF hospitalizations in patients with a left
ventricular ejection fraction of
The “INfluence of home moniToring on mortality and morbidity in heart failure
patients with IMpaired lEft ventricular function (IN-TIME)” trial randomized in
a 1 : 1 fashion 664 patients with NYHA functional class II or III, LVEF
HeartLogic HF algorithm (Boston Scientific, Boston, Mass, USA) incorporates information from multiple sensors embedded in the commercially available ICD or CRT-D devices [64, 65]. Information includes first and third heart sounds, respiration rate, rapid shallow breathing index (the ratio of respiration rate to tidal volume), thoracic impedance, heart rate, and patient activity. Sensor changes from the patient’s baseline were calculated and aggregated in the HeartLogic HF index. An alert is triggered if the HeartLogic HF index crosses a user-configurable threshold. Patients on alert state and high NT-pro BNP had a 50 fold increased risk of an HF event [65]. The median time from alert onset to HF event was 34 days creating the potential to intervene and reduce the risk of hospitalization [64, 66].
2.2.7.1 Right ventricular pressure. The Chronicle (Medtronic Inc., Minneapolis, MN, USA) system consisted of a programmable device that processed and stored information and a transvenous lead with a pressure sensor incorporated near its tip. The lead was positioned in the septum or right ventricular outflow tract. The initial pilot study showed that the device was safe but failed to significantly reduce hospitalizations and emergency or urgent care visits requiring intravenous therapy [67]. The sponsor terminated the pivotal trial because of the development of lead failures in patients included in previous trials [68].
2.2.7.2 Left atrial pressure. Left Atrial Pressure Monitoring to Optimize Heart Failure Therapy Study (LAPTOP-HF) evaluated the safety and efficacy of a left atrial pressure sensor monitoring. A HeartPOD implantable sensor lead (ISL) (St Jude Medical Sylmar, Sylmar, CA, USA) measured the left atrium waveform, core temperature, and the intracardiac electrogram. The ISL could be used as a stand-alone system or as a part of a cardiac resynchronization defibrillator device. The system included a patient advisor module (PAM) of the small phone size that powered the device and allowed the patient to communicate with the implant. One of the unique features of the trial was that the PAM provided interactive medical therapy adjustment recommendations according to the left atrial pressure level and the investigator’s input [69]. Although a significant reduction of HF hospitalizations was achieved in the telemonitoring arm, the Data Safety and Monitoring Board stopped the pivotal trial after enrolling 486 patients because of a perceived excess of procedure-related complications [70].
2.2.7.3 Pulmonary arterial pressure. The CardioMEMS
In the hemodynamic treatment group, optivolemic state was defined as:
-Pulmonary artery systolic pressure 15–35 mm Hg.
-Pulmonary artery diastolic pressure 8–20 mm Hg.
-Pulmonary artery mean pressure 10–25 mm Hg.
Definitions and treatment recommendations for hypervolemic or hypovolemic states
were provided to the investigators. The trial met the safety and efficacy
endpoints at six months with a 98.6% and 100% freedom from device-related
complications and sensor failures, respectively, and a 28% risk reduction of
heart failure hospitalizations [72]. In 2016, the investigators reported the
complete follow-up results from the CHAMPION randomized trial showing that when
PA pressure data was available to guide therapy, HF hospitalizations in the
initial control group were reduced by 48% [73] An analysis of the Medicare data
showed similar reductions in HF hospitalizations and the potential reduction in
HF hospitalizations costs [74]. The real world CardioMEMS
Circumstances that should be considered as potential contraindications include:
The presence of a chest circumference
A Cochrane Library systematic review of structured telephone support or non-invasive telemonitoring for patients with HF published in 2015, which included 41 randomized controlled trials, concluded that those interventions reduce the risk of all-cause mortality and HF -related hospitalizations [81]. Another systematic review using network meta-analysis technique that included 29 studies arrive at similar conclusions [82]. Hereine, we discuss the findings of five studies that were not included in those reviews (Table 1, Ref. [83, 84, 85, 86, 87]). “Efficacy of telemedical interventional management in patients with heart failure” (TIM-HF2) was a randomized, controlled, parallel-group unmasked trial that tested a multifaceted telemonitoring intervention compared with usual care [83]. This trial built upon the TIM-HF experience that showed that remote patient monitoring compared with standard care was not associated with reducing all-cause mortality [88]. A subgroup analysis of TIM-HF showed that patients without depression or recent HF hospitalization were most likely to benefit from telemedicine interventions. The primary endpoint was the percentage of days lost due to unplanned cardiovascular hospital admissions or all-cause death.The intervention consisted of a remote Bluetooth monitoring system (Physio-Gate® PG 1000, GETEMED Medizin- und Informationstechnik AG, Teltow, Germany) that transmitted vital signs and electrocardiogram information directly to the monitoring center. The telemonitoring system consisted of four Bluetooth-equipped measuring devices: a 3-channel ECG, pulse oximeter, blood pressure monitor, and digital Scale. A proprietary custom-made software triaged the information to identify patients that required immediate attention. Patients were provided with a mobile phone to call the monitoring center in case of an emergency [89]. The percentage of days lost due to unplanned cardiovascular hospital admissions and all-cause death was 4.88% (95% CI 4.55–5.23) in the remote patient management group and 6.64% (6.19–7.13) in the usual care group (ratio 0.80, 95% CI 0.65–1.00; p = 0.0460). Most of the intervention’s benefit was reducing all-cause mortality and unplanned hospital admissions due to worsening HF. One year after stopping the intervention, there were no significant differences in the trial’s primary outcome [90].
| Author (Ref.) | Year | n° | Design | Primary endpoint | Patients | Intervention | Comparator | Outcome |
| Ong [86] | 2016 | 1437 | RCT | Readmission for any cause within 180 days after discharge | Adults hospitalized for heart failure | Conducted by Registered Nurses: 1-Predischarge education; 2-Regularly schedule telephone coaching; 3-Home tele monitoring of weight, blood pressure, heart rate and symptoms | Pre-discharge education and follow up phone call | No significant difference in primary outcome |
| Koehler [83] | 2018 | 1571 | RCT | Percentage of days lost due to unplanned cardiovascular hospital admissions or death from any cause, during the individual patient follow-up time | Ambulatory patients; NYHA II or III; heart failure hospitalization within 12 months before randomization, and LVEF 45% or lower (could be higher if patients were on diuretics). Depression or recent hospitalization |
Daily transmission of bodyweight, blood pressure, heart rate, analysis of the heart rhythm, SpO2 and a self-rated health status to the tele-medical center; a definition of a patient’s risk category; patient education; and co-operation between the tele-medical center, and the patient’s general practitioner and Cardiologist | Usual Care defined as follow up in accordance with the current guidelines for the management and treatment of patients with heart failure | Percentage of days lost was statistically reduced in patients allocated to telemedicine |
| Galinier [85] | 2020 | 937 | RCT | All‐cause mortality or unplanned hospitalizations at 18 months | Hospitalized for acute HF within 12 months before inclusion, had access to a landline telephone or General Packet Radio Service network | Tele-monitoring program which consisted in: (1) electronic devices transmitted weight and heart failure symptoms to a specialized system that generated alerts and communicated with the nurses | Usual Care | No significant difference in primary outcome |
| (2) Educational HF component: Information pack and phone calls | ||||||||
| Rahimi [84] | 2020 | 101 | Two-armed partially blinded parallel RCT | Use of guideline-recommended medical therapy for chronic HF and major comorbidities, measured as a composite opportunity score | High risk heart failure patients. Defined by functional class, NT pro BNP and |
Tele-monitoring system with integrated risk prediction and disease management service which provided alert and support to healthcare practitioner | Tele-monitoring system without integrated risk prediction and disease management service | No significant difference in primary outcome |
| Lee [87] | 2020 | 2372 | RCT | Readmission for HF within 30 days after discharge | Adults hospitalized for heart failure | Structured telephone visit with a nurse or pharmacist to guide follow-up. Telephone appointments included a structured protocol enabling medication titration, laboratory ordering, and booking urgent clinic visits as needed under physician supervision | In person visit within 7 days | No significant difference in 30-day readmission or mortality. Telemedicine visits non-inferior to in person-visits |
| RCT, Randomized Controlled Trial. | ||||||||
The SUPPORT-H2 Study recruited high-risk HF patients and showed that a telemonitoring system with the capacity to contact the clinical staff failed to improve the adherence to guideline-directed medical therapy [84]. The small sample size is a limitation of this report. In “Optimization of the Ambulatory Monitoring for Patients With Heart Failure by Tele-cardiology” (OSICAT), patients with a history of HF hospitalization within the prior 12 months were randomized to telemonitoring or usual care. The telemonitoring intervention failed to reduce all-cause death or hospitalization for heart failure. The telemonitoring system provided an alert that nurses evaluated in working days, and if appropriate, they advised the patient to contact their general practitioner or cardiologist [85]. This contrasted with the intensive monitoring and readily available physician support of TIM-HF2. The Better Effectiveness After Transition – Heart Failure (BEAT-HF) was a negative clinical trial of post- HF hospitalization telemedicine monitoring. Of note, the telemonitoring strategy was not integrated with physician care to allow rapid medical treatment changes [86]. Finally, “The Heart Failure Readmission Intervention by Variable Early Follow-up” (THRIVE) Study showed that a structured phone interview by a pharmacist or a nurse within seven days of discharge had comparable clinical outcomes at 30 days (readmission and death) with an in-person visit with a physician [87].
COVID-19 pandemic revitalized the interest in telemedicine and prompted
regulatory changes needed for rapid deployment. Along with significant
biotechnological advances, digital health has an established role in clinical
practice [91]. A reduction of HF hospitalization was observed in early 2020 [92].
This was interpreted secondary to the patient’s fear of going to a healthcare
facility. During the same period, a 154% increase in virtual visits was
documented [93]. Virtual visits and telemedicine changed from being rare to
become one of the primary forms of health care delivery. This transition was not
done without difficulties, and health disparities became more evident [94]. For
example, female sex and median household income
Synchronous virtual encounters are followed by telemonitoring with or without a formal chronic disease management program [21]. For a virtual visit to be successful, there should be patient consent, and the patient should be familiar with the virtual platform. Although most insurances prefer video, the telephone is a valid and valuable option. The VV should follow a pre-defined structure. Medication review is of paramount importance. The patient can check the vital signs during the encounter and if the patient has a CIED, access the latest remote interrogation of the device. Monitoring of activity level is feasible, and if a step counter is available the average steps per day can be noted [97]. In patients with a wearable defibrillator, evaluation of daily step count and changes in body position in addition to electrophysiological parameters can be performed [98]. Use of the video capability to detect jugular venous distention or lower extremity swelling can also be helpful. Benzinger et al. [99] described the “Telehealth ten”, a patient-assisted medical examination with ten components. During the virtual encounter, the clinician guides the patient to perform a modified physical examination. An after-visit summary with the therapeutic plan and follow-up should be emailed to the patient.
The functional capacity, psychosocial factors and clinical stability dictate the intensity of monitoring. If a patient has a CIED, every effort should be made to maximize the information obtained through device interrogation. If the patient had a recent hospitalization for HF, implantation of a pulmonary arterial pressure monitoring device should be considered (Fig. 3). Of note, barriers for compliance such as financial factors, health literacy, dietary transgressions should be evaluated, and specific interventions should be implemented [100, 101].
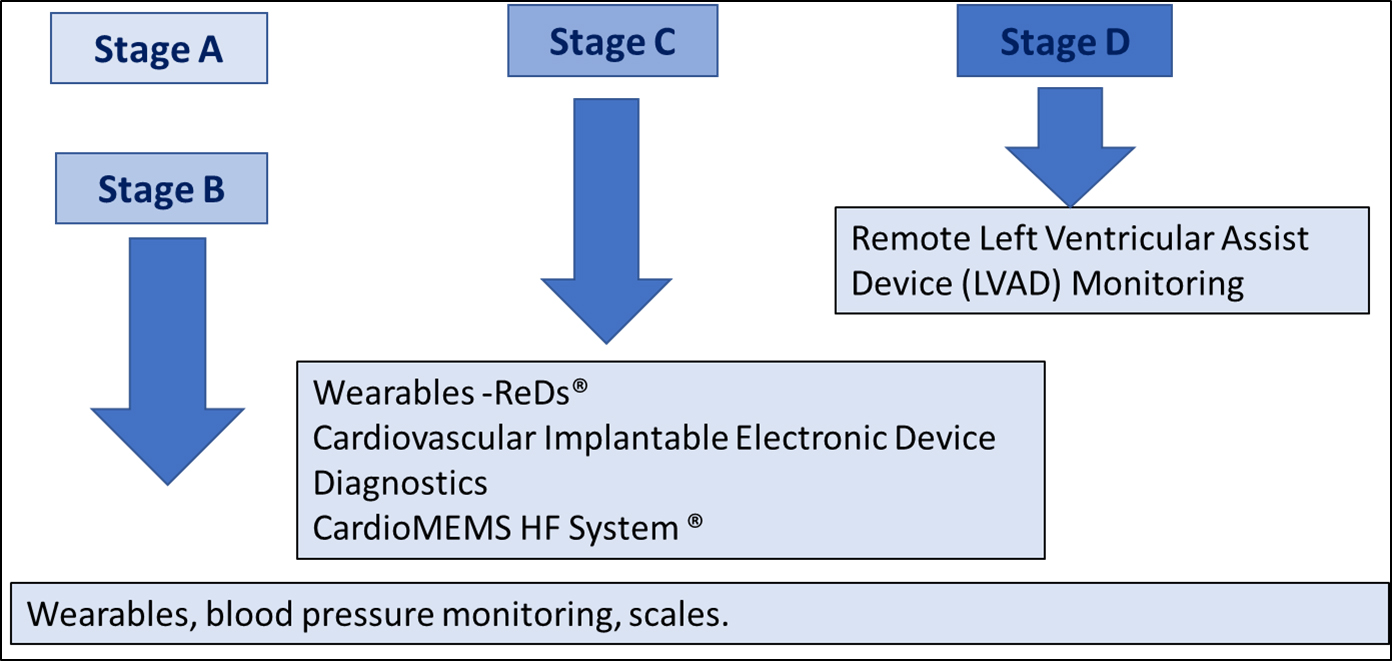 Fig. 3.
Fig. 3.Telemonitoring in Heart Failure according to AHA/ACC Heart failure stages.
Virtual visits have increased exponentially during the COVID-19 pandemic. Current evidence support the use of virtual visits to replace post discharge in-person visits. A structured approach is recommended by current guidelines. Tele-monitoring interventions can range from low to high complexity and should match the risk profile of the patient. Regardless of the tele-monitoring platform, the integration with clinical support for medical therapy optimization cannot be overemphasized. Racial and social disparities in telemedicine implementation have been reported and further research is needed.
PA, AB conceived and designed the content; JB reviewed the manuscript and contributed with graphical material; PA, AS, AA wrote the manuscript; PA, AB, supervised and prepared the final version for submission. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.



